Abstract
The purpose of this study is to investigate the relationship between chronic pain and the mortality rate of cerebrovascular disease (CVD) in cancer patients. Thus, we performed a case-control investigation was conducted by utilizing data from the Surveillance, Epidemiology, and End Results (SEER) database between 1975 and 2019. Multiple demographics, pain rating and other clinical characteristics were extracted to assess predictors for the death from CVD in cancer patients. Different machine learning algorithms were applied to construct pain-related prediction model. The analysis involved 16,850 case patients and 710,729 controls. Among cancer patients, approximately 2.3% succumbed to subsequent CVD. Cancer pain (Pain rating II) was associated with a decreased risk of CVD. Univariate and multivariate COX analyses indicated that older age at cancer diagnosis, male gender, single marital status, Black or Other race, and lack of systemic therapy correlated with a higher risk of CVD-related death. Propensity score matching revealed a significantly lower proportion of Pain rating II in the case group. The logistic regression algorithm demonstrated superior predictive ability for 5-year and 10-year CVD risk in cancer patients. Notably, survival time, age, and pain rating emerged as the top three crucial variables. This study firstly investigated pain and various risk factors for CVD in cancer patients, highlighting pain as a novel and possible protective factor for CVD. The development of a risk model based on pain could aid in identifying individuals at high risk for CVD and may inspire innovative strategies for preventing CVD in cancer patients.
Similar content being viewed by others
Introduction
Cancer and cerebrovascular disease (CVD) represent significant global health challenges, ranking prominently among the primary contributors to global morbidity and mortality1,2. It is not surprising that both cancer and CVD can be diagnosed in the same patient, possibly attributed to common traditional risk factors and lifestyles like aging, smoking, and excessive alcohol consumption2. As shown by a prior epidemiological research, approximately 15% of individuals diagnosed with cancer encounter a CVD event, characterized by comparable rates of hemorrhages and infarctions3. On the contrary, an estimated rate of CVD mortality in global regions was 0.23%, suggesting a relationship between cancer and CVD4. Stroke, as the primary types of CVD events, is frequently observed in patients with various types of cancers5. Moreover, as advancements in cancer prognosis leading to longer survivorship, the issue of increased risk of CVD should be considered6,7. Therefore, enhanced precision and proactive approaches for preventing CVD are essential in the context of cancer patients; regrettably, there is presently a deficiency in specific guidelines for preventing, identifying, or managing CVD in individuals with cancer8. A better understanding of the mechanisms of CVD occurrence in cancer patients may facilitate to identify and target those patients with the highest CVD risk.
The mechanisms underlying the connection between CVD and cancer has for long captivated the interest of the medical community2,6,8,9,10,11,12,13,14,15,16,17,18. In addition to traditional risk factors, some potential mechanisms may provide insight into the connection, including hypercoagulability, non-bacterial thrombotic endocarditis, direct compression of blood vessels, and the impact of chemotherapy or radiotherapy in individuals with cancer10. However, the link between cancer and subsequent CVD is still obscure. Recent researches regarding chronic pain may shed light on this close relationship. Chronic pain is one distinctive feature of cancer patients and it can be caused by chemotherapy, radiotherapy, surgery, cancer itself and so on19. Chronic pain now is regarded as a significant contributor for the development of CVD. For instance, a surgery revealed that chronic pain can strongly increase the risk of stroke and heart attack20. This kind of phenomenon was also been reported by Von Korff et al.21 and Wang et al.22. Instead and interestingly, a fresh cohort study indicated that chronic pain could serve as a novel protective factor for certain diseases in specific cancer types23. These above effects of chronic pain are possibly related to its pleiotropic role in of chronic pain on whole-body metabolic homeostasis, central nervous system neural activity, and immune cell activity and so on24,25. Nevertheless, there is no literature examining the correlation between chronic pain and the subsequent risk of CVD in individuals with cancer. Due to the rising survival rate of cancer patients, a fresh perspective on chronic pain and other risk factors in the etiopathology of cancer and CVD could offer novel opportunities for effective therapeutic interventions.
The aim of this investigation was to explore whether chronic pain will contribute to the development of CVD in cancer patients. On the basis of pain rate and other screened risk factors, we hope to construct and validate pain-related risk prediction models for CVD death by using diverse types of machine learning algorithms. Our study may provide evidence-based recommendations for future direction, develop personalized therapeutic strategies and especially improve the prevention of CVD events in cancer patients.
Materials and methods
Data source
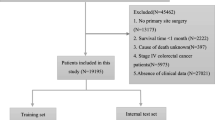
This cohort study was based on the national Surveillance, Epidemiology, and End Results (SEER) database, which covering approximately 48.0% of the United States population (https://seer.cancer.gov/about/overview.html). Authorization was obtained from SEER in June 2022. The study was a case-control cohort design and utilized the SEER database from 1975 to 2019. Inclusion criteria encompassed patients aged 18 years and older, with malignant diseases, benign, or in situ diseases. Patients who died from CVD or not were compared for analysis. The exclusion criteria included patients with malignancies who died within 12 months, or those with benign or in situ diseases who died within 1 month. Given the de-identification and public availability of SEER data for research purposes, local research ethics committee approval was not required for this study.
Study population and variables
Sociodemographic and clinical characteristics of each patient were extracted for analysis as in our previous study26. A brief description of these variables is presented as follows: age at diagnosis, sex, race (white, black, others and unknown), marriage (married, single, unknown), year of diagnosis, cancer-directed surgery (CDS) (yes, no, unknown), radiation therapy (yes, no, unknown), systemic therapy (yes, no, unknown) and survival time. For variables with missing data, such as treatment plans or patient history, we categorized them explicitly under “unknown” to maintain consistency in the dataset and ensure that all cases were included in the analysis. This approach helped mitigate data loss and ensured that the model was able to consider these cases as valid inputs. A case-control cohort study was set up to identify the risks of death from CVD. In the case group, the cause of death was specifically confined to CVD, as defined by ICD-9 and ICD-10 codes. Those who died from non-CVD over 18 were assigned to the control group.
Furthermore, different pain rating groups were compared to explore the relationship between pain and CVD mortality in cancer patients. As previous study described23, pain rating I was characterized by in situ behavior (ICD-2 codes for benign and in situ conditions), while pain rating II was identified by malignant behavior (ICD-1 codes for malignant conditions) with a survival time exceeding one year. This pain rating method was mainly based on both the degree of pain severity (as influenced by cancer aggressiveness) and pain duration (as influenced by survival time). These two factors were critical in understanding the potential influence of pain on CVD in cancer patients. Cancer behavior could be a proxy for pain severity. Pain rating I was defined for patients with in situ tumors, which typically involved less aggressive disease and thus less intense pain. In contrast, pain rating II was designated for malignant tumors, which were associated with more advanced disease stages and generally caused more severe pain due to the invasiveness and potential for nerve involvement in malignancy. Chronic and severe nature of pain in malignant cancer patients may have a more pronounced impact on the body’s physiological and neurological responses than less severe or shorter-duration pain. Additionally, survival time may serve as an indicator of pain duration. For patients with malignant cancers (pain rating II), we further specified a minimum survival time of over one year. This criterion was intended to ensure a severe and chronic pain experience in cancer patients. By focusing on patients who lived more than a year with malignant cancer, we aimed to capture pain that was not only severe but also persistent, thereby increasing the likelihood of detecting any long-term effects of chronic pain on CVD risk. These pain ratings serve as nominal definitions to distinguish between different pain characteristics without implying a severity gradient. A study flowchart of the cases included in this study was shown as Fig. 1.
Study flowchart.
Statistical analysis
The statistical analyses consisted of following steps. Firstly, descriptive statistics were employed to outline the baseline characteristics of all patients. Independent-sample tests were used for continuous variables, and a chi-square test was applied for categorical variables to evaluate potential risk factors. Secondly, both univariable and multivariable Cox regression models were utilized to examine variables associated with an elevated risk of death from CVD. Thirdly, Propensity score matching (PSM) was conducted to address differences in potential covariates and to further explore risk factors by using a 1:1 matching ratio. The selected variables for matching included age, sex, race, marriage, and year of diagnosis, which were chosen based on their relevance to the outcomes of interest. The caliper value was set at 0.001, a strict threshold to ensure precise matching and minimize bias. Although we tested broader caliper values of 0.01 and 0.005, these did not achieve adequate matching, as some key covariates still showed statistically significant differences between groups. Therefore, the 0.001 caliper was retained to ensure the most accurate matching and control for potential bias.
Finally, 5-year and 10-year risk prediction models were proposed to deeply examine whether the pain rating could be an independent predictor of CVD death. The SEER dataset was split into an 80:20 ratio to construct the predictive models. Namely, 9717 patients (80%) were designated as training data and 2430 patients (20%) were set as test data. To ensure randomness and balance in the data split, the dataset was divided randomly, and balance in key features such as cancer type, patient age, and treatment methods was maintained between the training and test sets. We adopted a multi-step approach to ensure the appropriateness of the selected statistical models and account for potential confounders. We revised the methodology section to include more detailed explanations on algorithm selection and validation processes. Several common machine-learning algorithms were applied, including backpropagation artificial neural network (BP-ANN)27, k-nearest neighbor (k‐NN)28, logistic regression, decision tree, random forest, gradient boosting decision tree model (GBDT), XGBoost, and Lightgbm, based on their effectiveness in handling classification tasks and ability to model complex relationships between features. Cross-validation procedures were implemented during model training to prevent overfitting and ensure that the models generalized well to unseen data. The predictive performance of these models was assessed using test classification scores, including accuracy, precision, recall, F1 score, and area under the ROC curve (AUC). F1 score, a measure for binary classification tasks, represents the harmonic mean between precision and recall. A 1:1 ratio PSM was applied to pain rating I and pain rating II subgroups to adjust for potential covariates, such as age, sex, race, and marital status.
Data analyses were conducted using SPSS version 27.0 (IBM, Armonk, NY, USA) and R software (R software for statistical computing, Vienna, Austria). A p-value < 0.05 was considered statistically significant.
Results
Case characteristics
A total of 16,850 cases and 710,729 controls were enrolled (Table 1). 2.3% of cancer patients were died from subsequent CVD. The case group exhibited a significantly higher average age compared to the control one (median (IQR) age, 60.00 (54.00–63.00) vs. 57.00 (50.00–62.00) years). Additionally, case patients had a significantly higher percentage of females (57.4% vs. 52.0%), individuals of race = others (7.8% vs. 7.3%), and married patients (66.3% vs. 63.2%), but a lower rate of pain rating II (91.5% vs. 95.1%). For treatments, case patients demonstrated a significantly higher rate of cancer-directed surgery but a lower rate of radiation therapy or systemic therapy when compared to controls. A notably longer survival time (193.00 (104.00-285.00) vs. 72.00 (28.00-176.00) months) was also found in cases.
Risk factors
Univariate analysis demonstrated that the mortality of CVD in cancer patients was significantly influenced by patient’s age (OR = 1.05, 95% CI 1.05–1.06), sex (OR = 0.89, 95% CI 0.84–0.94), race (OR = 1.55, 95% CI 1.43–1.69), marriage (OR = 0.84, 95% CI 0.76–0.86), year of diagnosis (OR = 0.89, 95% CI 0.84–0.94), pain rating (OR = 0.69, 95% CI 0.62–0.77) and systemic therapy (OR = 0.99, 95% CI 0.92–1.06) (Table 2). Namely, patients being older, male, race in Black and others, single, early diagnosed, with pain rating I but without systemic therapy would potentially have a higher risk of CVD death. On multivariate analysis, only age (OR = 1.04, 95% CI 1.03–1.05), race (OR = 1.56, 95% CI 1.33–1.83), marriage (OR = 0.81, 95% CI 0.72–0.92), pain rating (OR = 0.64, 95% CI 0.52–0.78), and systemic therapy (OR = 0.68, 95% CI 0.58–0.78) exhibited a statistically significant association with the increased risk of CVD mortality.
PSM analysis
After 1:1 PSM, 15,930 cases and 15,930 controls were left for comparison (Table 3). Significant disparities persisted in pain rating, CDS, and systemic therapy between the two groups (all p < 0.05). In detail, the case group had a notably larger proportion of patients with pain rating I (8.0% vs. 4.8%), a higher rate of CDS performance (84.6% vs. 74.2%), but lower rates of radiation therapy (25.8 vs. 31.9%) and systemic therapy (25.8 vs. 31.9%). Cases still had a significantly longer survival time than controls (194.00 (105.00-286.00) vs. 86.00 (31.00-206.00) months).
Patterns of pain rating
The pain rating I and II subgroups have 1268 (8.0%) and 14,662 (92.0%) cases for comparison, respectively (Table 4). The pain rating II subgroup had a significantly higher proportion of male (43.5% vs. 24.0%), married (70.4% vs. 66.0%), early diagnosed (68.9% vs. 59.0%), receiving less CDS (83.8% vs. 92.9%) but more systemic therapy patients (1.9% vs. 1.3%) when compared with the pain rating I subgroup. A significant shorter survival period (194.00 (105.00-285.00) vs. 200 (100.00-290.00) months) was also found in pain rating II patients.
Risk prediction models performance
5-year risk prediction models
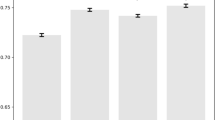
Among 8 prediction models the logistic regression one demonstrated superior predictive capability, as evidenced by the highest AUC value (0.71) (Table 5). The F1 score was suitable for assessing prediction performance in imbalanced samples. And the random forest model had the highest F1 score (0.0507), representing the best prediction performance (Fig. 2). Therefore, the random forest model was chosen as the optimal predictive model for estimating the risk of CVD-related mortality.
The prediction models for the 5-year risk. Eight machine learning algorithms were applied and compared by the accuracy, recall, precision, AUC, and F1 score. The logistic regression model demonstrated superior predictive capability, as evidenced by the highest AUC value (0.71). And the random forest model had the highest F1 score (0.0507), representing the best prediction performance.
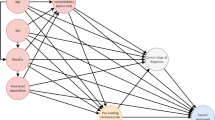
Subsequently, we conducted an in-depth analysis of feature importance using the GBDT model. The mean Shapley additive explanations (SHAP) value for each feature variable reflects its impact on the final predicted probability (Fig. 3). A higher SHAP value indicates a more pronounced influence on the model output. Notably, survival time (20.93%), age (14.66%), and pain rating (13.26%) emerged as the three most crucial feature variables in the GBDT model for predicting the cause of death. The association of categorical variables with CVD death was studied using cross-tabulation analysis (Table 6). The proportion of death from CVD rose with the increase of survival time. Conversely, with the grades of pain rating going up, the proportion of death from CVD decreased.
Features of importance derived from the 5-year GBDT model. The mean Shapley additive explanations (SHAP) value of feature variables represents the degree of influence on the final predicted probability. Survival time (20.93%), age (14.66%) and pain rating (13.26%) were the top three important feature variables in the GBDT model for predicting the death cause. Abbreviations: UN, unknown; CDS, cancer-directed surgery.
10-year risk prediction models
Furthermore, the 10-year risk prediction models were also constructed based on aforementioned algorithms (Table 7). Similar to the 5-year prediction models, the logistic regression model continued to present superior predictive performance, achieving an AUC of 0.69 (Fig. 4). Additionally, the random forest model remained the best prediction model based on the highest F1 score (0.066).
The prediction models for the 10-year risk. Eight machine learning algorithms were applied and compared by the accuracy, recall, precision, AUC, and F1 score. Similar to the 5-year prediction models, the logistic regression model presented a superior predictive performance, achieving an AUC of 0.69. The random forest model remained the best prediction model based on the highest F1 score (0.066).
GBDT model indicated that survival time (23.68%), age (16.18%) and pain rating (8.49%) were still the top three critical feature variables for forecasting the cause of death (Fig. 5). The cross-tabulation analysis still demonstrated that the proportion of death from CVD increased with the increase of survival time but decreased with the increase of grades of pain rating (Table 8).
Features of importance derived from the 10-year GBDT model. Survival time (23.68%), age (16.18%) and pain rating (8.49%) were still the top three important feature variables in the GBDT model for predicting the death cause. Abbreviations: UN, unknown; CDS, cancer-directed surgery.
Discussion
According to our best knowledge, this study stands one of the first to explore the relationship between chronic pain and CVD death in cancer patients by utilizing the SEER database. Notably, a significant inverse correlation emerged between pain rating and CVD-related mortality in cancer patients. This suggests that pain may act as an innovative protective factor against CVD in cancer patients. Additionally, by leveraging pain and other identified risk factors, we developed novel prediction models to assess the risks of CVD-related death in cancer patients. This study may shed some light on the pathogenesis of CVD and provide suggestions for the prevention of CVD events in cancer patients.
Pain’s role as a protective factor against CVD in cancer patients
As mentioned above, current evidence regarding the risk of CVD among cancer patients remains ambiguous29. Recently, a plethora of literatures have supported that cancer patients predispose to suffer from different forms of CVD events, in particular fatal stroke. For example, a fresh SEER-based study demonstrated that 57,523 (0.9%) out of 6,136,803 cancer patients succumbed to stroke, with a significantly higher mortality rate than that observed in the general population10. Similarly, Navi et al. utilized nationwide claims data and documented an elevated risk of stroke in patients diagnosed with the four most prevalent cancers when compared to those without cancer30. Some other studies also reported an increased risk of subsequent CVD among cancer patients2,31. Of note, as the survival rates for cancer patients steadily rise, it is imperative to identify cancer survivors who hold an increased risk of CVD death. Some efforts have been made to target subgroups at greatest risk of CVD. Primary factors examined for CVD risk in cancer patients encompass age, gender, race, cancer type, grade, and treatment32,33. Of note, chronic pain may serve as a risk factor of CVD and one distinctive feature of cancer patients is persisted pain. Nevertheless, there is no literature examining the correlation between chronic pain and the subsequent risk of CVD in individuals with cancer.
In our study, we illustrated that chronic pain may function as an independent protective factor against CVD-related mortality in individuals with cancer. Obviously, cancer patients frequently experience different levels of chronic pain, which influence quality of life and contribute to various psychosocial responses. As reported, the prevalence rates of cancer pain were 39.3% post-curative treatment, 55.0% during anticancer treatment, and 66.4% in advanced, metastatic, or terminal disease19. Multifactorial pathogenic mechanisms have explained the occurrence of chronic pain in cancer patients, such as toxicity as radiotherapy-related toxicity, chemotherapy-caused neuropathy, post-operative pain, the presence of cancer itself, immune-nervous system interaction, and so on34. For example, a neuropathic component is present in metastatic bone cancer35. Recently, chronic pain has garnered significant attention due to its ability to increase the risk of CVD. For instance, Chung et al. revealed that people with chronic pain were 30% more likely to have a stroke and 20% more susceptible to experiencing a heart attack compared to those without such persistent pain20. In a cross-sectional study involving 5,692 individuals in the United States, Von Korff et al. observed an elevated risk of stroke and heart disease among adults with chronic pain21. Similar results that chronic pain may increase the risk of CVD were also reported by Wang et al.’s cohort study22. Our study further explored the role of chronic cancer pain in the subsequent occurrence of CVD. We categorized pain into rating I and II based on cancer behavior and survival years23, representing both the intensity and duration of pain. Our findings indicated that cancer patients who succumbed to CVD had a lower incidence of pain rating II compared to those who died from control causes. By conducting univariate and multivariate analyses, we established a correlation between pain rating II and a reduced risk of CVD in cancer patients. The further PSM analysis also supported this finding. In addition, those cancer patients being male, married, early diagnosed, applied less CDS but more systemic therapy preferred to have pain rating II. As a result, it stands to reason that chronic pain can serve as a novel protective factor against CVD in individuals with cancer. This evidence may offer insights into the pathogenesis of CVD and the mechanisms underlying the cancer-CVD relationship.
Apparently, the preventive impact of chronic pain against CVD observed in our study contradicts previous researches that have reported a positive association between pain and CVD risk. Possible reasons for this discrepancy are as follows. The main reason may be the difference in follow-up time and chronic cancer pain have a certain degree of long-lasting effect. Our data from the SEER database appeared to have a longer follow-up time (over 1 year) than other studies. A large part of data on mortality of CVD in cancer patients was documented in a short period such as 3 or 6 months12. Particularly, the peak in subsequent CVD may occur within the first two month of cancer diagnosis and then decline over time2. In other words, the mortality rate of CVD in cancer patients seemed to be lower at 12 months or later. A marked disparity in risks, pathophysiology, and outcomes may be present between individuals with newly diagnosed cancer and those with previously identified active cancer12. This result aligned with our data, in which we enrolled pain rating II patients with survival years > 1. Another possible reason is that apart from cancer pain itself, the source of chronic pain in other studies included more types of pain such as spinal disorders, osteoarthritis, headache and so on, which may confound the effect of cancer pain on CVD risk20. In addition, the “chronic pain-enhanced CVD risk” statement is mainly based on the studies involved patients with musculoskeletal pain (e.g., spinal disorders, osteoarthritis) and neurological diseases (e.g., headache) instead of patients with cancer20. Musculoskeletal pain may be responsible for more than 2/3 of painful states in primary care36. Chronic non-cancer pain and chronic caner pain may hold different effects on the development of CVD (see below for more details). Hence, it may be reasonable to conclude chronic pain as a possible protective factor for CVD death. This is in consistent with a previous study, which revealed that pain could prevent cancer patients against the death from Alzheimer Disease23.
Currently, studies directly supporting the idea of pain as a protective factor against CVD in cancer patients are scarce. Complex interactions between chronic pain, cancer, and certain physiological changes could potentially influence CVD risk. Several hypotheses may help to explain the pathophysiological basis for the protective value of pain in CVD prevention. Firstly, in cancer patients, treatments such as chemotherapy, radiation, and certain pain management approaches (like opioid use) have been found to modulate CVD risk. However, some therapies also have cardioprotective effects particularly the widely used opioids. Opioid receptor-dependent analgesics and anesthetics may enable to target adaptive/protective responses to physiological and pathological stimuli, modulate vascular tone and induce cytoprotective states in the myocardium37. For instance, biphalin, a non-addictive synthetic opioid, could significantly decrease mean atrial pressure without reduction of renal perfusion38. A randomized double-blind study indicated that higher doses β-endorphin can induce a hypotensive effect on the blood pressure by activating opioid receptors and inhibiting vasoconstrictive substances such as norepinephrine and endothelin-139. Additionally, β-endorphin was reported to lower blood pressure and peripheral vascular resistance through the nitric oxide (NO) pathway39. Particularly, when given as a cerebroventricular injection, β-endorphin could also produce a significant fall in blood pressure40. Apart from lowering blood pressure, opioids also play an important role in protecting the endothelial function in hyperlipidemia41,42, possibly decreasing the risk of CVD. Secondly, chronic pain in cancer and non-cancer patients may be caused by different mechanisms, leading to different effects on the development of CVD. As mentioned above, chronic pain in cancer patients is commonly localized and mainly caused by cancer itself or cancer-treatments. Instead, chronic pain in non-cancer patients seems to be caused by more complex reasons and coexisted with different concomitant diseases. For instance, patients with chronic low back pain, a type of most common musculoskeletal pain, can increase pro-inflammatory cytokines and subsequent central sensitization, resulting in a further atherosclerotic change43,44. Chronic low back pain was related to an increased risk of strokes, especially ischemic stroke45. Besides, exposure of chronic non-cancer pain could induce elevated cortisol levels46. In addition, multiple studies have demonstrated that chronic non-cancer pain may play a role in depression47, which is an independent risk factor of atherosclerosis48. Furthermore, chronic pain in non-cancer patients is commonly combined with other CVD-related diseases. Patients with back pain is associated with diabetes, which is a well-known risk factor of CVD49. Other concomitant diseases including chronic obstructive pulmonary disease50, chronic kidney disease51 may also increase the risk of chronic pain-related CVD in non-cancer patients. Thirdly, inflammatory response to cancer and its related pain can involve immune system adaptations that differ from those observed in typical chronic inflammatory conditions, potentially leading to lower rates of atherosclerosis and CVD52. However, the effects vary widely depending on cancer type, progression stage, and individual patient factors. Fourthly, chronic cancer pain may hold different effect when compared with non-cancer-related chronic pain. Chronic pain is often associated with heightened sympathetic nervous system activity, leading to increased blood pressure and heart rate, which can be detrimental to cardiovascular health. However, cancer-related pain, especially severe pain, may induce different physiological responses, including immune and metabolic adaptations, that could influence CVD risk differently than chronic non-cancer pain25. In addition, properly managed pain may reduce stress levels, which is a well-known risk factor for CVD53. A better pain management in cancer patients can also lead to better sleep quality, which is important for overall cerebrovascular health54. Other mechanisms that effective pain management enables better mobility, modulates immune system, stabilizes blood pressure may explain the protective effect of chronic pain on cancer-related CVD. Finally, patients with chronic pain may be more likely to engage with healthcare services and seek care for other morbidities more frequently than those without pain55. The CVD might be prevented at the same time. These behaviors may been strengthened in cancer patients, who commonly suffer from moderate to severe pain56.
Other risk factors for CVD in patients with cancer
Following both univariate and multivariate analyses, other risk factors such as age, race, marital status, and systemic therapy were identified as correlates with CVD mortality in cancer patients. Detailly, patients being older at cancer diagnosis, male, single, race in Black and Others and without systemic therapy linked to an increased AD risk. The roles of age and race in CVD development have been well discussed by other studies32,33. While systemic therapy could potentially contribute to cancer-associated CVD, its impact on increasing the risk of CVD remains uncertain. Kitano et al. reported in their study that patients in the chemotherapy group had a higher stroke risk compared to those in the non-chemotherapy group (hazard ratio = 1.84). However, this difference became statistically insignificant after adjusting for cancer status57. An additional investigation found that chemotherapy, particularly platinum-based regimens, independently posed a risk for stroke in patients with ovarian cancer58. Other investigations have also examined the impact of both traditional and nontraditional chemotherapeutic agents on the occurrence of stroke59.
Of note, chronic pain may interact with these risk factors (including age, race, marital status, and systemic therapy, etc.), possibly contributing to CVD risk in cancer patients. For instance, epidemiological studies found that older adults with metastatic cancer reported lower pain intensity ratings than their younger counterparts60. In other words, older cancer adults might experience less chronic pain and loss its protective effect against CVD death. Racial and cultural disparities can play a significant role in pain management. As shown by a recent study, minority groups may receive different pain assessments and treatments compared to their counterparts, which can affect the management and perception of chronic cancer pain61. Systemic treatments such as chemotherapy, hormone therapy, or immunotherapy can exacerbate existing pain or introduce new pain syndromes as side effects19. However, the side effects of systemic treatments may outweight the possible protective effect of chronic pain on CVD. More studies are necessitated to explore the interaction between chronic pain and other risk factors in cancer patients.
Risk model performance
Given the increased risk of subsequent CVD in cancer patients, it is imperative to employ multidisciplinary approaches. An effective strategy involves the identification of a subset of patients at high risk for such events. Leveraging the identified risk factors, we developed and validated 5-year and 10-year prediction models for the risk of CVD death in cancer patients. Among 8 types of machine learning models, the logistic regression 5-year and 10-year ones had the best prediction ability, with an AUC at 0.71 and 0.69, respectively. The subsequent importance of features analysis revealed important feature variables and highlighted the importance of pain rating in the risk models. Based on the relatively high AUC, we believe that our pain-related risk models may be a tool to assist clinicians in the identification of high-risk CVD patients with cancer. Currently, the QSTROKE scores62 and several prediction models63,64,65,66 are available to quantify CVD risk. However, most of these models were specifically designed to determine a relatively short period of stroke risk in general population or a set of patients with certain type of cancer when compared with our risk models. Our unique pain-related CVD risk model was based on a large group of general cancer patients, with the ability of a long-term CVD risk prediction and a wide range of application. Undoubtedly, further studies are necessary to validate and adjust our risk model, particularly identify the applicability of this model in different types of cancer.
Limitations
Admittedly, several limitations in this study should be considered. The primary one is the inherent limitation of SEER database, which provides basic patients’ characteristics, without information regarding comorbidities, biomarkers (e.g., D-dimer levels), full cancer treatment plans, pain assessment and its management strategies, detailed types of CVD. More detailed analyses on risk factors of CVD could not be executed. Of note, currently no other databases contain such information67. Anyway, SEER data were beneficial to use for our type of analysis. In addition, we enrolled patients diagnosed between 1975 and 2019. Treatment paradigms for cancer patients have changed over time such as chemotherapy and radiotherapy, likely affecting the result analyses. Moreover, due to the lack of pain assessment in database, we evaluated the pain degree based on cancer behavior and survival time. Of note, this assessment of pain was indirect and semi-quantitative, partly confounding results analysis, losing detailed evidence and reducing the confidence power. Particularly, data about pain management strategies were missed, therefore we could not figure out whether pain treatments or pain itself hold the protective effect on CVD related. The relationship between certain analgesics and CVD mortality was also not explored. Besides, the prediction ability of pain-related risk model is still not fully satisfactory at present, because the AUCs were not too strong to meet the clinical applications. Enrolling more covariates, such as lab tests68 and comorbidities, may be helpful to improve the pain-related risk model. Furthermore, we did not detailly analyze whether chronic pain exhibit the same pain-protective effect in different types of cancer. This was a pilot study to explore the role of chronic pain in CVD, not limited to certain type of cancer. Future study is necessary to identify the applicability of our pain-related model across different types of cancer. In addition, our study was lack of a power analysis to justify the sample size. However, our study was based on the SEER database and after set up the inclusion and exclusion criteria, all patients meeting these criteria were drawn from the SEER from 1975 to 2019. In all, we enrolled 16,850 cases and 710,729 controls. This extensive dataset can not only meet the necessary requirements for our research but also facilitate easy data acquisition. Consequently, we believe that our conclusions are well-supported and reliable, and an additional justification of the sample size appears to be unnecessary.
Despite the above limitations, the main strength of our study is the identification of pain as a possible protective factor for CVD in patients with cancer at the first time and conducted a pain-related risk model by using different machine learning algorithms. Based on these findings, several potential clinical impacts and strategies could be developed for clinical practice. First of all, chronic pain may serve as a marker to help stratify CVD risk among cancer patients, allowing clinicians to better tailor preventive measures and allocating resources to those at higher risk for CVD. Moreover, more personalized pain management protocols may also be needed to balance pain control with maintaining potential protective elements. Particularly, it seems necessary to develop integrated care models that consider chronic pain as part of the overall health status and risk profile of cancer patients. This involves close collaboration between oncologists, cardiologists, and pain specialists to provide comprehensive and holistic care. Furthermore, it necessitates to encourage lifestyle interventions that might contribute to CVD protection, alongside managing cancer pain. More awareness should be created on the pain management and CVD prevention in cancer patients, hoping to improve both quality of life and survival outcomes.
Conclusions
This study initially investigated pain and other risk factors for CVD in individuals with cancer and determined pain as a possible protective factor against CVD. We hope to increase the awareness of high-risk CVD in cancer patients and more attempts should be made to prevent the occurrence of CVD events. Several hypotheses were proposed to underly the protective effect of chronic pain on CVD, offering a new perspective on the pathogenesis of CVD and the mechanism behind the interaction of cancer and subsequent CVD. To help identify high-risk CVD patients with cancer, we constructed pain-related risk model and believe them may inspire new approaches for CVD prevention in cancer patients. Additional research is required to validate whether chronic pain can function as a protective factor of CVD and to elucidate the clear-cut mechanisms underlying the connection between pain and CVD in individuals with cancer, and to provide evidence-based guidelines to address the distinct needs of this particular population.
Data availability
The datasets generated for this study are available to the corresponding author upon request.
References
Siegel, R. L., Miller, K. D., Wagle, N. S. & Jemal, A. Cancer statistics, 2023. CA A Cancer J. Clin. 73(1), 17–48. https://doi.org/10.3322/caac.21763 (2023).
Liu, Q. et al. Risk of cerebrovascular disease after cancer diagnosis in the United States. iScience 26(7), 107165. https://doi.org/10.1016/j.isci.2023.107165 (2023).
Grazioli, S. et al. Cancer-associated ischemic stroke: A retrospective multicentre cohort study. Thromb. Res. 165, 33–37. https://doi.org/10.1016/j.thromres.2018.03.011 (2018).
Jagannathan, R., Patel, S. A., Ali, M. K. & Narayan, K. M. V. Global updates on cardiovascular disease mortality trends and attribution of traditional risk factors. Curr. Diab. Rep. 19(7), 44. https://doi.org/10.1007/s11892-019-1161-2 (2019).
Wang, J., Kim, Y. D. & Kim, C. H. Incidence and risk of various types of arterial thromboembolism in patients with cancer. Mayo Clin. Proc. 96(3), 592–600. https://doi.org/10.1016/j.mayocp.2020.05.045 (2021).
Gon, Y. et al. Stroke mortality in cancer survivors: A population-based study in Japan. Thromb. Res. 222, 140–148 (2023).
Bhatia, S., Tonorezos, E. S. & Landier, W. Clinical care for people who survive childhood cancer: A review. JAMA 330(12), 1175–1186 (2023).
Woock, M. et al. Cancer and stroke: Commonly encountered by clinicians, but little evidence to guide clinical approach. Therap. Adv. Neurol. Disord. https://doi.org/10.1177/17562864221106362 (2022).
Sun, M. Y. & Bhaskar, S. M. M. Bridging the gap in cancer-related stroke management: Update on therapeutic and preventive approaches. Int. J. Mol. Sci. https://doi.org/10.3390/ijms24097981 (2023).
Sonbol, Y. T. et al. Stroke as a cause of death in patients with cancer: A SEER-based study. J. Stroke Cerebrovasc. Dis. 32(8), 107154. https://doi.org/10.1016/j.jstrokecerebrovasdis.2023.107154 (2023).
Shivarov, V., Shivarov, H. & Yordanov, A. Seasonal pattern of cerebrovascular fatalities in cancer patients. Healthcare https://doi.org/10.3390/healthcare11040456 (2023).
Costamagna, G. et al. Clinical and demographic characteristics, mechanisms, and outcomes in patients with acute ischemic stroke and newly diagnosed or known active cancer. Neurology https://doi.org/10.1212/wnl.0000000000207341 (2023).
Lun, R., Siegal, D., Ramsay, T. & Dowlatshahi, D. Cancer and stroke: What do we know and where do we go?. Thrombos. Res. 219, 133–140. https://doi.org/10.1016/j.thromres.2022.09.014 (2022).
Chen, Y. J. et al. Cancer-related stroke: Exploring personalized therapy strategies. Brain Behav. https://doi.org/10.1002/brb3.2738 (2022).
Martin, C. J. & Barnard, M. Potential risks of cardiovascular and cerebrovascular disease and cancer due to cumulative doses received from diagnostic CT scans?. J. Radiol. Prot. https://doi.org/10.1088/1361-6498/ac270f (2021).
Chen, N. C. et al. Risk of stroke in patients with breast cancer and sleep disorders. J. Cancer 12(22), 6749–6755. https://doi.org/10.7150/jca.63184 (2021).
Dardiotis, E. et al. Cancer-associated stroke: Pathophysiology, detection and management (Review). Int. J. Oncol. 54(3), 779–796. https://doi.org/10.3892/ijo.2019.4669 (2019).
Adams, H. P. Cancer and cerebrovascular disease. Curr. Neurol. Neurosci. Rep. https://doi.org/10.1007/s11910-019-0985-0 (2019).
van den Beuken-van Everdingen, M. H., Hochstenbach, L. M., Joosten, E. A., Tjan-Heijnen, V. C. & Janssen, D. J. Update on prevalence of pain in patients with cancer: Systematic review and meta-analysis. J. Pain Symptom Manage. 51(6), 1070-1090.e9. https://doi.org/10.1016/j.jpainsymman.2015.12.340 (2016).
Chung, K.-M. et al. Chronic pain increases the risk for major adverse cardiac and cerebrovascular events: A nationwide population-based study in Asia. Pain Med. 21(9), 1985–1990. https://doi.org/10.1093/pm/pnaa107 (2020).
Von Korff, M. et al. Chronic spinal pain and physical-mental comorbidity in the United States: results from the national comorbidity survey replication. Pain 113(3), 331–339. https://doi.org/10.1016/j.pain.2004.11.010 (2005).
Wang, H. C. et al. Increased risk of strokes in patients with chronic low back pain (CLBP): A nationwide population-based cohort study. Clin. Neurol. Neurosurg. 192, 105725. https://doi.org/10.1016/j.clineuro.2020.105725 (2020).
Xia, S., Yu, X. & Chen, G. Pain as a protective factor for Alzheimer disease in patients with cancer. Cancers (Basel) https://doi.org/10.3390/cancers15010248 (2022).
Sacks, D. et al. Multisociety consensus quality improvement revised consensus statement for endovascular therapy of acute ischemic stroke. Int. J. Stroke 13(6), 612–632. https://doi.org/10.1177/1747493018778713 (2018).
Martinez-Sanchez, N. et al. The sympathetic nervous system in the 21st century: Neuroimmune interactions in metabolic homeostasis and obesity. Neuron 110(21), 3597–3626. https://doi.org/10.1016/j.neuron.2022.10.017 (2022).
Wei, Y. et al. Comparison of survival outcomes and risk factors between ductal carcinoma of the prostate and acinar adenocarcinoma of the prostate: A population-based propensity score–matching study. Eur. Urol. Open Sci. 46, 88–95. https://doi.org/10.1016/j.euros.2022.10.013 (2022).
Zhang, S. Z., Chen, S. & Jiang, H. A back propagation neural network model for accurately predicting the removal efficiency of ammonia nitrogen in wastewater treatment plants using different biological processes. Water Res. 222, 118908. https://doi.org/10.1016/j.watres.2022.118908 (2022).
Ramos-Miguel, A., Perez-Zaballos, T., Perez, D., Falconb, J. C. & Ramosb, A. Use of data mining to predict significant factors and benefits of bilateral cochlear implantation. Eur. Arch. Otorhinolaryngol. 272(11), 3157–3162. https://doi.org/10.1007/s00405-014-3337-3 (2015).
Navi, B. B. et al. New diagnosis of cancer and the risk of subsequent cerebrovascular events. Neurology 90(23), e2025–e2033. https://doi.org/10.1212/wnl.0000000000005636 (2018).
Navi, B. B. et al. Association between incident cancer and subsequent stroke. Ann. Neurol. 77(2), 291–300. https://doi.org/10.1002/ana.24325 (2015).
Zöller, B., Ji, J., Sundquist, J. & Sundquist, K. Risk of haemorrhagic and ischaemic stroke in patients with cancer: A nationwide follow-up study from Sweden. Eur. J. Cancer. 48(12), 1875–1883. https://doi.org/10.1016/j.ejca.2012.01.005 (2012).
Howard, V. J. et al. Sex and race differences in the association of incident ischemic stroke with risk factors. JAMA Neurol. 76(2), 179–186. https://doi.org/10.1001/jamaneurol.2018.3862 (2019).
Gardener, H. et al. Race and ethnic disparities in stroke incidence in the Northern Manhattan study. Stroke 51(4), 1064–1069. https://doi.org/10.1161/STROKEAHA.119.028806 (2020).
Brown, M. R. & Ramirez, J. D. Neuroimmune mechanisms in cancer pain. Curr. Opin. Support Palliat. Care. 9(2), 103–111. https://doi.org/10.1097/spc.0000000000000140 (2015).
Yoneda, T., Hiasa, M., Nagata, Y., Okui, T. & White, F. Contribution of acidic extracellular microenvironment of cancer-colonized bone to bone pain. Biochim. Biophys. Acta. 1848(10 Pt B), 2677–84. https://doi.org/10.1016/j.bbamem.2015.02.004 (2015).
Hasselström, J., Liu-Palmgren, J. & Rasjö-Wrååk, G. Prevalence of pain in general practice. Eur. J. Pain. 6(5), 375–385. https://doi.org/10.1016/S1090-3801(02)00025-3 (2002).
Rawal, H. & Patel, B. M. Opioids in cardiovascular disease: Therapeutic options. J. Cardiovasc. Pharmacol. Ther. 23(4), 279–291. https://doi.org/10.1177/1074248418757009 (2018).
Bądzyńska, B., Lipkowski, A. W. & Sadowski, J. An antihypertensive opioid: Biphalin, a synthetic non-addictive enkephalin analog decreases blood pressure in spontaneously hypertensive rats. Pharmacol. Rep. 68(1), 51–55. https://doi.org/10.1016/j.pharep.2015.06.006 (2016).
Cozzolino, D. et al. Acute pressor and hormonal effects of beta-endorphin at high doses in healthy and hypertensive subjects: Role of opioid receptor agonism. J. Clin. Endocrinol. Metab. 90(9), 5167–5174. https://doi.org/10.1210/jc.2004-2554 (2005).
Laubie, M., Schmitt, H., Vincent, M. & Remond, G. Central cardiovascular effects of morphinomimetic peptides in dogs. Eur. J. Pharmacol. 46(1), 67–71. https://doi.org/10.1016/0014-2999(77)90146-7 (1977).
Bryant, H. U., Story, J. A. & Yim, G. K. Assessment of endogenous opioid mediation in stress-induced hypercholesterolemia in the rat. Psychosom. Med. Nov-Dec 50(6), 576–585. https://doi.org/10.1097/00006842-198811000-00003 (1988).
Li, J. et al. Vasculoprotective effect of U50,488H in rats exposed to chronic hypoxia: Role of Akt-stimulated NO production. J. Appl. Physiol. 114(2), 238–44. https://doi.org/10.1152/japplphysiol.00994.2012 (2013).
Wang, H., Schiltenwolf, M. & Buchner, M. The role of TNF-alpha in patients with chronic low back pain-a prospective comparative longitudinal study. Clin. J. Pain. 24(3), 273–278. https://doi.org/10.1097/AJP.0b013e31816111d3 (2008).
Giesecke, T. et al. Evidence of augmented central pain processing in idiopathic chronic low back pain. Arthritis Rheum. 50(2), 613–623. https://doi.org/10.1002/art.20063 (2004).
Wang, H.-C. et al. Increased risk of strokes in patients with chronic low back pain (CLBP): A nationwide population-based cohort study. Clin. Neurol. Neurosurg. 192, 105725. https://doi.org/10.1016/j.clineuro.2020.105725 (2020).
Van Uum, S. H. et al. Elevated content of cortisol in hair of patients with severe chronic pain: A novel biomarker for stress. Stress 11(6), 483–488. https://doi.org/10.1080/10253890801887388 (2008).
Sheng, J., Liu, S., Wang, Y., Cui, R. & Zhang, X. The link between depression and chronic pain: Neural mechanisms in the brain. Neural Plast. 2017, 9724371. https://doi.org/10.1155/2017/9724371 (2017).
Pan, A., Sun, Q., Okereke, O. I., Rexrode, K. M. & Hu, F. B. Depression and risk of stroke morbidity and mortality: A meta-analysis and systematic review. Jama 306(11), 1241–1249. https://doi.org/10.1001/jama.2011.1282 (2011).
Rinaldo, L. et al. Diabetes and back pain: Markers of diabetes disease progression are associated with chronic back pain. Clin. Diabetes 35(3), 126–131. https://doi.org/10.2337/cd16-0011 (2017).
Lee, A. L., Goldstein, R. S. & Brooks, D. Chronic pain in people with chronic obstructive pulmonary disease: Prevalence, clinical and psychological implications. Chronic Obstr. Pulm. Dis. 4(3), 194–203. https://doi.org/10.15326/jcopdf.4.3.2016.0172 (2017).
Pham, P. C. et al. 2017 update on pain management in patients with chronic kidney disease. Clin. Kidney J. 10(5), 688–697. https://doi.org/10.1093/ckj/sfx080 (2017).
Cao, S., Fisher, D. W., Yu, T. & Dong, H. The link between chronic pain and Alzheimer’s disease. J. Neuroinflamm. 16(1), 204. https://doi.org/10.1186/s12974-019-1608-z (2019).
Reddin, C. et al. Association of psychosocial stress with risk of acute stroke. JAMA Network Open 5(12), e2244836–e2244836. https://doi.org/10.1001/jamanetworkopen.2022.44836 (2022).
Brunetti, V. et al. Sleep and stroke: Opening our eyes to current knowledge of a key relationship. Curr. Neurol. Neurosci. Rep. 22(11), 767–779. https://doi.org/10.1007/s11910-022-01234-2 (2022).
Kadam, U. T., Thomas, E. & Croft, P. R. Is chronic widespread pain a predictor of all-cause morbidity? A 3 year prospective population based study in family practice. J. Rheumatol. 32(7), 1341–1348 (2005).
Mercadante, S. Cancer pain treatment strategies in patients with cancer. Drugs 82(13), 1357–1366. https://doi.org/10.1007/s40265-022-01780-6 (2022).
Kitano, T. et al. The effect of chemotherapy on stroke risk in cancer patients. Thromb. Haemost. 120(4), 714–723. https://doi.org/10.1055/s-0040-1708484 (2020).
Kuan, A. S. et al. Risk of ischemic stroke in patients with ovarian cancer: A nationwide population-based study. BMC Med. 12, 53. https://doi.org/10.1186/1741-7015-12-53 (2014).
Zuo, P. Y., Chen, X. L., Liu, Y. W., Xiao, C. L. & Liu, C. Y. Increased risk of cerebrovascular events in patients with cancer treated with bevacizumab: A meta-analysis. PLoS One. 9(7), e102484. https://doi.org/10.1371/journal.pone.0102484 (2014).
Fillingim, R. B., Turk, D. C. & Yezierski, R. P. Pain in the elderly. In Advances in Geroscience (eds Sierra, F. & Kohanski, R.) 551–592 (Springer International Publishing, 2016).
Calvo, E. et al. Global pain and aging: A cross-sectional study on age differences in the intensity of chronic pain among middle-aged and older adults in 20 countries. J. Gerontol. B Psychol. Sci. Soc. Sci. 78(6), 1098–1108. https://doi.org/10.1093/geronb/gbac199 (2023).
Hippisley-Cox, J., Coupland, C. & Brindle, P. Derivation and validation of QStroke score for predicting risk of ischaemic stroke in primary care and comparison with other risk scores: A prospective open cohort study. BMJ 346, f2573. https://doi.org/10.1136/bmj.f2573 (2013).
Hunter, E. & Kelleher, J. D. Age specific models to capture the change in risk factor contribution by age to short term primary ischemic stroke risk. Front. Neurol. 13, 803749. https://doi.org/10.3389/fneur.2022.803749 (2022).
Hunter, E. & Kelleher, J. D. A review of risk concepts and models for predicting the risk of primary stroke. Front. Neuroinform. 16, 883762. https://doi.org/10.3389/fninf.2022.883762 (2022).
Zhu, Q. et al. A model for risk prediction of cerebrovascular disease prevalence-based on community residents aged 40 and above in a city in China. Int. J. Environ. Res. Public Health https://doi.org/10.3390/ijerph18126584 (2021).
Abdel-Qadir, H. et al. Development and validation of a multivariable prediction model for major adverse cardiovascular events after early stage breast cancer: A population-based cohort study. Eur. Heart J. 40(48), 3913–3920. https://doi.org/10.1093/eurheartj/ehz460 (2019).
Zaorsky, N. G. et al. Stroke among cancer patients. Nat. Commun. 10(1), 5172. https://doi.org/10.1038/s41467-019-13120-6 (2019).
Alanazi, E. M., Abdou, A. & Luo, J. Predicting risk of stroke from lab tests using machine learning algorithms: Development and evaluation of prediction models. JMIR Form. Res. 5(12), e23440. https://doi.org/10.2196/23440 (2021).
Acknowledgements
None.
Funding
This study was supported by Natural Science Foundation of Hunan Province (Grant No.2022JJ40708), Xiaoxiang Cancer Clinical Research Public Welfare Project (Grant No. P049-001, YT), Natural Science Foundation of Fujian Province (Grant No.2021J01359, YW) , Natural Science Foundation of Ningde (Grant No.2023J63, QW) and Scientific Research Project of Youth from Fujian Provincial Health Commission (Grant No. 2021QNB003, DL).
Author information
Authors and Affiliations
Contributions
Conceptualization: YW, DL, KY and YG. Formal analysis and investigation: YT, JZ, YL, QW, LZ and KK. Writing - original draft preparation: YW, DL and YT. Writing - review and editing: YW, DL, YT and YG. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Local research ethics committee approval was not required for this study, as the SEER data were de-identified and publicly available for research use. However, we have taken steps to ensure the ethical integrity of our research. The SEER data used is de-identified to protect individual privacy, and we obtained the appropriate authorization for its use. The data was exclusively used for this study and securely managed by designated personnel to maintain its confidentiality and integrity.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wei, Y., Lin, D., Lian, Y. et al. Population data study reveals pain as a possible protective factor against cerebrovascular disease in cancer patients. Sci Rep 14, 29471 (2024). https://doi.org/10.1038/s41598-024-80668-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-80668-9