Abstract
Identifying the drivers of population declines in migratory species requires an understanding of how individuals are distributed between periods of the annual cycle. We built post- (fall) and pre-breeding (spring) migratory networks for the blackpoll warbler (Setophaga striata), a Neotropical-Nearctic songbird, using tracking data from 47 light-level geolocators deployed at 11 sites across its breeding range. During pre-breeding migration, two stopover nodes (regions) on the U.S. eastern seaboard received high scores in our network metrics (betweenness centrality and time-adjusted node weight), likely acting as key refuelling areas for most of the global blackpoll warbler population before their multi-day flights over the Atlantic Ocean. During post-breeding migration, highly ranked stopover nodes in the southeastern U.S. acted as a geographical bottleneck before birds dispersed to their boreal breeding destinations. Nodes located in northern Colombia and Venezuela were also ranked highly during both migrations and were likely used to prepare for (pre-breeding) and recover from (post-breeding) Atlantic flights. Blackpoll warblers showed a crosswise migration pattern, whereby individuals from western breeding populations tended to spend the nonbreeding season in the eastern part of the nonbreeding range and vice-versa. Despite this, the strength of migratory connectivity between the breeding and nonbreeding grounds ranged from moderate to low, largely because many individuals used more than one node during the ‘stationary’ nonbreeding period. Our results suggest that the number of breeding populations affected by a threat in the blackpoll warbler’s range will strongly depend on where and when this threat occurs. Consequently, our migratory network should be key to inform future conservation planning and population monitoring efforts.
Similar content being viewed by others
Introduction
Determining how environmental change influences migratory animal populations, many of which have shown sharp declines in recent decades1,2,3,4, requires knowledge of how individuals are spatially distributed between periods of the annual cycle (i.e. ‘migratory connectivity’)5. A species may show ‘strong’ migratory connectivity, whereby individuals from different breeding populations use distinct and separate migratory routes and nonbreeding areas, or ‘weak’ connectivity, whereby individuals from different breeding populations mix extensively during the migratory and nonbreeding periods5. Estimating the degree of connectivity has important implications for predicting the dynamics of migratory populations6,7. For example, a species showing weak connectivity may be more buffered from environmental change (e.g., habitat loss) occurring at a single location compared to a species showing strong connectivity8,9. Furthermore, estimating the origin of individuals from a previous period in the annual cycle provides the ability to understand how events in one period of the year may carry over to influence individual success and demography in the following period10,11,12,13.
Assessing migratory connectivity in small long-distance migratory birds, such as songbirds (passerines), has been particularly challenging because most do not weigh enough to carry devices that record and transmit their position accurately and regularly (e.g., daily). In the past decades, the development of miniaturized archival light-level geolocator tags (hereafter “geolocators”) has provided the opportunity to estimate movements for birds weighing as little as 10 g based on the timing of twilights14, often revealing impressive feats of migration15,16. While progress has occurred since the introduction of geolocators17, including the development of miniaturized GPS18 and multi-sensor geolocators19, less than a third of migratory songbirds in North America have been tracked throughout their entire life cycle with animal-borne tags20. Additionally, a complete understanding of migratory connectivity requires knowledge of movements from individuals across a species’ range21 but the number of studies that have collected tracking data at this scale remains small22,23,24,25,26, likely due to the challenges associated with deploying and recovering archival tags over large ranges. This limits our ability to make strong inferences about the factors driving the dynamics of any given population or why populations in some parts of a species range are declining while others are not.
Recently, network analysis has emerged as a useful framework for describing and analyzing migratory connectivity across the annual cycle23,27,28,29,30,31. Migratory networks use concepts from graph theory to characterize movements8, whereby “nodes” represent stationary periods of the annual cycle (breeding, nonbreeding, migratory stopovers), and “edges” represent the movements of individuals between nodes. Both nodes and edges can be weighted based on their relative use (abundance) and the characteristics of the network can be quantified using network metrics32. For example, network metrics can identify nodes that serve as key connectors between other nodes (i.e., bottleneck) in a network23,28,33, or parts of a network that form distinct and highly connected groups of nodes (e.g. migratory flyways)23,34. Despite their potential, the use of network metrics to describe and analyze range-wide patterns of connectivity in migratory birds remains uncommon23,28,29,31,33,35,36.
The blackpoll warbler (Setophaga striata; hereafter ‘blackpoll’) is a small (~ 12 g) songbird that breeds in North America’s boreal and northeastern montane spruce-fir forests and spends the nonbreeding season in northern South America37. While blackpolls are widespread, estimates from the North American Breeding Bird Survey (BBS) suggest they have declined by 2.3% per year (95% CI 8.0–0.4%) on average between 1970 and 2022 in their breeding range, one of the fastest rates amongst North American birds38. Declines appear to be most pronounced in eastern Canada and the northeastern United States (U.S.)38,39,40 but large portions of the blackpoll breeding range lie north of survey routes, leaving geographic differences in population trends somewhat uncertain. Constructing and analyzing a migratory network for blackpolls is a critical step toward determining how environmental changes occurring in the species’ breeding, nonbreeding, and stopover areas are contributing to observed population declines.
Here, we combine previously published tracking data of blackpolls from 23 light-level geolocators deployed at six locations on the breeding grounds16,41 with new data from 24 additional geolocators, most of which were deployed at five new locations. Together, these data provide an unprecedented coverage of the blackpoll range. We used these data to complete the following objectives: (1) build migratory networks that delineate breeding, stopover, and nonbreeding areas and the flow of individuals moving between them during the post-breeding and pre-breeding migrations, (2) evaluate the strength of migratory connectivity across the networks, and (3) identify areas that are most important to the integrity of the migratory networks. Achieving these objectives will improve our ability to link threats in specific parts of the range to population declines and prioritize areas for broad-scale conservation planning.
Results
General description of the post- and pre-breeding migratory networks
The post-breeding (southbound) and pre-breeding (northbound) migratory networks were built by clustering estimates of stationary locations from each tracked bird (Fig. 1), resulting in nodes ranging in diameter (maximum distance between stationary locations) between 394 and 2516 km in the post-breeding migration network, and 440 and 2052 km in the pre-breeding migration network. While we describe population-scale movements between nodes as part of our analysis, individual movement patterns are provided in Supplementary Figs. S4–S7.
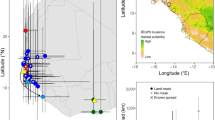
Structure of the post- and pre-breeding migratory networks. Panel (a,b) show the location and weight of network nodes. The blackpoll warbler breeding, migration, and nonbreeding distributions polygons, obtained from Birdlife International distribution maps, are also shown78. Node size and edge size are proportional to the relative abundance contributed by blackpolls that used them. Nodes are located at the median location of their stationary locations, with the exception of nodes 5, 6, and 7, which were placed at supposed coastal departure points. These three nodes and all others in North America during post-breeding migration were clustered using longitude only. Edges connect nodes but do not represent actual migratory routes. Panels (c,d) show the colour-coded and numbered clusters of stationary locations that correspond to each stopover and nonbreeding node. Each location corresponds to the median of the posterior distribution from the geolocator analysis, and the error bars show the 95% credible interval for longitude and latitude (occasionally hidden by symbols). In the post-breeding migration network, a portion of the stationary locations in the Caribbean were placed manually based on our assessment of light-level data and lacked credible intervals (See Supplementary Methods 4). Stationary locations estimated within 2 weeks of the equinox are circled in black and their latitude should be considered highly unreliable. The maps were generated in R using package sf version 1.0-16 (https://cran.r-project.org/web/packages/sf/), ggplot2 version 3.5.1 (https://ggplot2.tidyverse.org/), and lake and country polygons obtained from the Natural Earth dataset (www.naturalearthdata.com), via the naturalearth package version 1.0.1 (https://docs.ropensci.org/rnaturalearth/).
For the post-breeding migratory network, we identified 10 stopover nodes, clusters composed exclusively of sites used temporarily for a stopover, and 5 nonbreeding nodes, clusters containing at least one individual’s first site occupied for at least 2 weeks in the nonbreeding range (Fig. 1). Seven of the stopover nodes located in north America were composed of stationary locations clustered using longitude because they were occupied at a time close to the fall equinox (Fig. 1, nodes: 1, 2, 3, 4, 5, 6, 7, 8). This includes three nodes near the coast (nodes 5, 6, 7), the most likely location for stopovers before crossing the Atlantic, although individuals could have used stopovers at higher latitudes. Also due to the fall equinox, stationary locations in the Caribbean (nodes 9 and 10) were estimated by reviewing plots of latitude, longitude, and light levels (see “Methods” section). Individuals occupied 3–9 nodes (mean ± standard error (s.e.): 4.8 ± 0.2), including their breeding node, as well as at least one stopover node each. Western-breeding birds, tagged at sites west of James Bay (53.5° North, 80.5° East), used more stopover nodes (mean ± s.e.: 2.7 ± 0.5) than eastern-breeding birds (mean ± s.e.: 1.9 ± 0.4; GLM with Poisson distribution, β = 0.42 ± s.e.: 0.14, z = 3.06, p < 0.001, d.f. = 43). Although most birds completed their migration upon arrival in the first nonbreeding node they encountered (n = 27), some moved to a second nonbreeding node before stopping (n = 17) and two individuals used three nonbreeding nodes. Western-breeding birds used more nonbreeding nodes (mean ± s.e.: 1.7 ± 0.3 nodes) than eastern-breeding birds (mean ± s.e.: 1.11 ± 0.25; GLM with Poisson distribution, β = 0.46 ± 0.09, z = 5.30, p < 0.001, d.f. = 43) before finishing their post-breeding migration.
For the pre-breeding migratory network, there were 9 stopover nodes and 5 nonbreeding nodes (Fig. 1b,d), with individuals occupying 2–6 nodes (mean ± s.e.: 4.4 ± 0.2). In this network, stopover nodes were defined as they were in the post-breeding migratory network. However, nonbreeding nodes in this network were based on the last nonbreeding site used (i.e., the point of departure for pre-breeding migration). All birds used at least one stopover node except for two individuals, both of whom bred in Québec and spent the nonbreeding season in the eastern part of the nonbreeding range (geolocator ID V8296-004 and V8296-006, Supplementary Fig. S5, S6). This does not mean that these two individuals migrated without stopping, but rather that they made no detectable stopovers of at least two days after leaving South America. Similar to the post-breeding migratory network, western-breeding birds used more stopover nodes (mean ± s.e.: 2.6 ± 0.5) than eastern-breeding birds (mean ± s.e.: 1.2 ± 0.3, GLM with Poisson distribution, β = 0.62 ± 0.16, z = 3.9, p < 0.001, d.f. = 33). Most individuals moved to a stopover node in North America without detectable stopovers in South America (n = 27), but seven birds stopped in one additional nonbreeding node, and two stopped at two nonbreeding nodes before leaving the nonbreeding range. In this case, western- and eastern-breeding birds used a similar number of nonbreeding nodes before departing South America (eastern: 1.46 ± 0.32, western: 1.22 ± 0.23; GLM with Poisson distribution, β = − 0.18 ± 0.30, z = − 0.61, p = 0.54, d.f. = 33).
Across all the nodes used during migration, blackpolls were stationary for an average of 10.3 ± 0.7 days per node during post-breeding migration (range 2–37) and 5.6 ± 0.3 days per node during pre-breeding migration (range 2–16). This longer use of stopovers during post-breeding migration is supported by overall migration durations. For individuals that we could track throughout both migrations (n = 31), post-breeding migration was estimated to last an average of 43 ± 3 days, while pre-breeding migration lasted 32 ± 2 days.
Migratory connectivity
Examining patterns of node use by individuals from different breeding regions showed that extensive inter-population mixing occurred at several nodes during both post- and pre-breeding migration. In the post-breeding migratory network, at least one individual from each breeding region co-occurred in New England (node 6) and the mid-Atlantic US (here taken to include Virginia and West Virginia, node 5, Fig. 2a). The weights of these two nodes were 0.5 and 0.43 respectively based on our propagation of abundance estimates from eBird Status and trends42 throughout the network. If we account for the fact that some individuals used both nodes and only counted them once, then the two nodes had a joint weight of 0.76. Consequently, it is likely that a large portion of the global blackpoll breeding population uses these areas, although we emphasize that this estimate is based largely on adult males. On the nonbreeding grounds, individuals from all breeding regions co-occurred extensively at multiple nodes (Fig. 2a), in particular in northern Colombia and Venezuela (node 11, weight: 0.37), and eastern Colombia (node 13, weight: 0.30), although mixing in the former diminished after the departure of western breeders to other nonbreeding nodes (Fig. 2c).
Relative node use patterns for blackpoll warblers from each of the four regions we defined in the nonbreeding grounds (Eastern, Central, Western, and Northwestern, see Fig. S1) during (a) post-breeding and (b) pre-breeding migration. Inset maps (c,d) show only edges for movements performed between nonbreeding nodes, as well as the final (after post-breeding migration) and initial (before pre-breeding migration) population-specific use and weight accounting only for individuals that stayed over 2 weeks (i.e., excluding individuals making stopovers). The communities identified by the Walktrap algorithms are shown in (e,f), with their modularity values. A modularity near 0 indicates a weak community structure, while a high value indicates more edges within rather than between communities. The maps were generated in R using package sf version 1.0-16 (https://cran.r-project.org/web/packages/sf/), ggplot2 version 3.5.1 (https://ggplot2.tidyverse.org/), and lake and country polygons obtained from the Natural Earth dataset (www.naturalearthdata.com), via the naturalearth package version 1.0.1 (https://docs.ropensci.org/rnaturalearth/).
In the pre-breeding migratory network, individuals from eastern and central breeding regions co-occurred extensively in northern Venezuela (node 10; weight: 0.53), before departing South America (Fig. 2d). At pre-breeding migratory stopover nodes, at least one individual from all breeding regions occurred in Florida (node 8) and Georgia and Alabama (node 7), nodes with weights of 0.33 and 0.37, respectively (Fig. 2b). However, 16/34 tracked birds (corresponding to a weight of 0.36) did not make detectable stopovers at these nodes, instead moving directly to stopover nodes farther north, where eastern and western breeders were more strongly segregated (nodes 2–6, Fig. 2b).
Despite instances of high-interpopulation mixing in both networks, there was still some spatial structure, with eastern and western breeding individuals being more common at certain stopover nodes. For example, eastern-breeding individuals were dominant in New England (node 5) and Hispaniola (node 9, including the nearby Turks and Caicos) during post-breeding migration, and North Carolina and Virginia (node 5) during pre-breeding migration, whereas western-breeding individuals were dominant in the mid-Atlantic (node 7) during post-breeding migration and US Midwest (nodes 2, 3, 4 and 6) during pre-breeding migration. This combination of mixing at certain nodes and an underlying spatial structure was corroborated by the Walktrap community detection algorithm43, which identified two communities of highly interconnected nodes forming distinct flyways (Fig. 2e,f). These communities separated stopovers and nonbreeding areas primarily used by eastern and western breeders, but their low modularity values suggested that movements between the communities were still common, particularly during post-breeding migration.
Population-specific patterns of node use suggested that eastern breeding individuals spent the nonbreeding season in the western part of the nonbreeding range while western breeders spent it in the east, resulting in a crosswise migration pattern. This was supported by the strong relationship between breeding and nonbreeding site coordinates: breeding site longitude was negatively correlated with the longitude of each individual’s first nonbreeding site (Spearman’s rho: rs = − 0.73, p < 0.001) and last nonbreeding site (rs = − 0.78, p < 0.001). Individuals that bred at higher latitudes also tended to have their first (rs = − 0.39, p = 0.01) and last (rs = − 0.44, p = 0.01) nonbreeding sites at a lower latitude, suggesting a pattern of leapfrog migration, whereby northern breeders spent the nonbreeding season farther south and southern breeders spent it farther north, but this relationship was weaker than the longitudinal correlations.
The strength of migratory connectivity metric (hereafter “MC metric”), used to quantify the degree of migratory connectivity between the breeding and nonbreeding grounds44, indicated that the degree of migratory connectivity was moderate between the breeding grounds and the first nonbreeding site (0.42 ± 0.08) and low between the breeding grounds and the last nonbreeding site (0.26 ± 0.12). The sample sizes from the first and last nonbreeding sites were different (n = 46 and n = 36, respectively) due to geolocator failures, so we also estimated the MC metric using only individuals that had both a first and last site (n = 35) and obtained similar results (Supplementary Method 1).
Importance of stopover and nonbreeding nodes during migration
We used a centrality metric from graph theory, betweenness centrality, to quantify the degree to which each node was located at the intersection of multiple migratory routes. In the post-breeding migratory network, the nodes with the highest betweenness centrality values were in New England (node 6), the mid-Atlantic U.S. (node 7), northern Ontario (node 4), and Nova Scotia (node 5, Fig. 3a). In the pre-breeding migratory network, Georgia and Alabama (node 7) had a higher betweenness centrality than all other stopover nodes, with Illinois and Missouri (node 3) coming second (Fig. 3b). In both networks, nodes located in northern Venezuela and Colombia (post-breeding migration: node 11, pre-breeding migration: node 11 and 10) had the highest betweenness values in South America, highlighting their importance for stopovers performed within the nonbreeding range (Fig. 3a,b).
Values of betweenness centrality in the (a) post-breeding and (b) pre-breeding migratory networks for blackpoll warblers and time-adjusted node weights in the (c) post-breeding and (d) pre-breeding migratory networks. Nodes with high betweenness centrality are located at the intersection of the shortest path between other nodes in the network. Nodes with high time-adjusted weight are expected to have a higher relative abundance of blackpoll warblers and are used for longer stopovers. The maps were generated in R using package sf version 1.0-16 (https://cran.r-project.org/web/packages/sf/), ggplot2 version 3.5.1 (https://ggplot2.tidyverse.org/), and lake and country polygons obtained from the Natural Earth dataset (www.naturalearthdata.com), via the naturalearth package version 1.0.1 (https://docs.ropensci.org/rnaturalearth/).
Stopover nodes with high betweenness values in the post- and pre-breeding migratory networks also tended to have high time-adjusted node weights (relative abundance weighed by time spent, Fig. 3c,d). However, Hispaniola (node 9) was now the third highest-ranked node in the post-breeding season, after the mid-Atlantic and New England (Fig. 3c). Additionally, North Carolina and Virginia (node 5) had a high time-adjusted weight value during the pre-breeding migration, despite having a relatively low betweenness centrality value (Fig. 3d). These differences can be explained by the fact that betweenness ranks nodes higher when they are at the intersection of multiple shortest paths, even though they may have a lower estimated relative abundance and total time used than nodes located along fewer shortest paths. The highest-scoring nonbreeding nodes were again located in northern Colombia and Venezuela in both networks (Fig. 3c,d). Complete lists of nodes and their characteristics (number of tracked individuals present, weight and time-adjusted weight, importance metrics) are available in supplementary Tables S1, S2, and S3.
Post-migratory nonbreeding movements
Due to the change in the strength of migratory connectivity over the course of the nonbreeding season, we investigated the movements of individuals throughout this period. Based on our criteria, 18/38 individuals used more than one nonbreeding site and made a total of 24 movements with a mean distance traveled of 612 ± 61 km (Fig. 4). Most of these individuals moved only once, but four moved twice, and one moved three times. The directions of movements were not consistent across individuals: five individuals made southbound movements followed by one or more northbound movements, four individuals made southbound movements only, and 11 individuals made northbound movements only (Fig. 4). Most northward movements occurred during the mid to late winter, in March–April (14/15), while most southward movements occurred earlier, in October–February (7/9). Due to the higher prevalence of northward movements, most individuals ended the nonbreeding season at a nonbreeding site that was at a higher latitude than their first nonbreeding site (12/18).
An overview of post-migratory nonbreeding movements performed between the post- and pre-breeding migrations analyzed in the migratory networks. Locations of the initial and first stationary locations used by individuals during the nonbreeding season are shown in (a). Static points show the mean stationary locations of individuals that made no detectable post-migratory nonbreeding movements. Error bars show the 95% credible interval for the longitude and latitude of each location. The direction and timing of post-migratory nonbreeding movements are shown in (b) and movements initiated within 2 weeks of the spring equinox are indicated by dashed arrows. The maps were generated in R using package sf version 1.0-16 (https://cran.r-project.org/web/packages/sf/), ggplot2 version 3.5.1 (https://ggplot2.tidyverse.org/), and country polygons obtained from the Natural Earth dataset (www.naturalearthdata.com), via the naturalearth package version 1.0.1 (https://docs.ropensci.org/rnaturalearth/).
Transoceanic flight
Also noteworthy was that one of the two blackpolls originating from Newfoundland likely performed a transoceanic flight from northern Nova Scotia to northeastern Venezuela (geolocator V8757-096 in Supplementary Fig. S9), bypassing the Greater and Lesser Antilles. Based on the distance between the stationary locations occupied before and after the flight and the corresponding shade-free period recorded by the geolocator, we estimated that this individual flew at least 84 h non-stop, covering a distance of 4100 km over water (Supplementary Fig. S9).
Discussion
The post- and pre-breeding migrations of blackpolls represent a case of broad-scale convergence of individuals towards key stopover areas coupled with an underlying spatial structure. During post-breeding migration, blackpolls converged towards New England and the mid-Atlantic US, two adjacent nodes that likely concentrate the majority of individuals ~ 6400 km wide breeding range. These parts of North America could act as a geographic bottleneck, where disturbances such as habitat degradation could be more likely to affect a large percentage of the global population compared to other stopover sites in North America used during post-breeding migration. Interestingly, there was still some spatial structure along the Atlantic coast: eastern breeders primarily stopped in New England or further northeast and a majority of western breeders stopped in the mid-Atlantic or farther southwest. Compared to our results, stable isotope assessments of birds migrating through North America during post-breeding migration45,46,47 suggest the same general pattern, but with a higher prevalence of western breeders in New England (e.g., at sites like Metinic Island, Massachusetts)45,47. It is difficult to determine the cause of this discrepancy given the differences between tracking methods, but it could be related to our small sample size in the center of the blackpoll warbler’s range (n = 1 in Yellowknife, n = 7 in Churchill), an overestimation of the abundance of eastern breeders in our relative node use, population-specific fine-scale differences in space-use that we could not resolve here, or the lower spatial resolution of stable isotopes.
While the pre-breeding migration presented a similar pattern of convergence, it was also characterized by a degree of segregation between eastern and western breeders. During this migration, betweenness centrality was highest in Georgia and Alabama, a node used primarily by western breeders. In the rest of the network, western breeders primarily used nodes in the upper southeast and Midwest US while eastern breeders were predominant in Florida, in line with isotopic analyses from that location45,48, and in Virginia and North Carolina. Similar observations were also made by comparing the wing length of blackpolls caught at banding stations during the pre-breeding migration49, which would indicate that eastern and western populations become increasingly segregated after they arrive in North America. Conversely, a recent isotopic assay found that western breeders may migrate as far north and east as the Great Lakes before turning towards western Canada47, which would suggest more inter-population mixing in that region than was found here. Since most of the birds we tracked did not make stopovers close to the Great Lakes, it is possible that we did not track individuals from populations that frequently use this route. Regardless, it is clear that the U.S. southeast is the location of important stopovers, and it would be interesting to explore how extensive logging in this region over the last three decades50 could have influenced blackpoll abundance.
Nodes with high time-adjusted weights were largely consistent with sites that blackpolls would occupy to fuel before or recover from extended migratory flights. For instance, the highest time-adjusted weights occurred in New England and the mid-Atlantic during post-breeding migration, before birds crossed the Atlantic, and in northern Colombia and Venezuela during pre-breeding migration, before birds crossed the Caribbean Sea. High-scoring nodes in North America were consistent with records of elevated bird mass at banding stations49,51. Conversely, the high score of the node centered over Hispaniola during post-breeding migration contrasts with reports that stopovers in the Caribbean occur sporadically52,53, but this requires further verification due to the limits of our approach to identifying these stopovers. An individual’s ability to refuel at stopovers can influence the pace of migration54,55, survival56, and future reproductive success57,58. Furthermore, in some bird species, the lowest survival rates occur during migration, indicating that protecting stopovers could be essential to prevent population declines59,60. Blackpolls could be particularly susceptible to this form of regulation due to their use of a time-minimizing migration strategy involving high fat deposition rates, use of a limited number of stopovers, and long flights 46,61,62. We, therefore, suggest that further research focus on identifying fine-scale habitat use and the factors that might affect food availability at nodes with a high time-adjusted node weight, especially if they also act as points of convergence.
The nonbreeding nodes in northern Colombia and Venezuela stood out in the pre-and post-breeding migratory networks because they had high betweenness centrality and time-adjusted node weights compared to other nonbreeding nodes. The importance of northern Colombia and Venezuela during post-breeding migration was previously suggested by surveys in the Guajira and Paraguaná peninsulas showing that seasonally dry thorn scrubs and tropical forests along the coast are used as refueling sites for blackpolls arriving in South America62,63. Our results are consistent with these findings and demonstrate that this region is used by individuals from across the breeding range, highlighting the potential priority of this region for conservation. During the pre-breeding migration, the nodes in northern Colombia and Venezuela were almost exclusively used by individuals from the eastern and central breeding regions but retained relatively high scores in our metrics, even when compared with stopover nodes in North America. Blackpolls are reported to be rare in coastal areas of the Gulf of Venezuela during the pre-breeding migration, likely because this area is very dry at the end of the nonbreeding season and would not provide sufficient energy for migration63. Consequently, further studies should investigate the occurrence of pre-breeding migration stopovers further inland, along the nearby slopes of the Venezuelan Andes.
The crosswise migration pattern of blackpolls contrasts with the parallel migration that is observed in most Neotropical migratory songbirds, where western breeders winter in the west, and eastern breeders spend the nonbreeding season in the east (e.g.,22,23,25,64). The few songbird species that show crosswise migration65 either tend to show a weak pattern (e.g. purple martin, Progne subis)26 or the pattern occurs only between a subset of populations (e.g. yellow warbler, Setophaga petechia)66. While the reversal of the blackpoll’s longitudinal distribution was more pronounced than in these examples, the moderate-to-low levels of migratory connectivity that we measured suggested that there was still some mixing between individuals from different breeding regions. This contrasts with the near-complete population segregation in the nonbreeding range of some neotropical migrants with strong parallel migration patterns like the golden-winged warbler (Vermivora chrysoptera)13 or the painted bunting (Passerina ciris)67. However, if habitat loss on the nonbreeding grounds is an important driver of past declines in blackpoll populations, the crosswise pattern could be strong enough to result in differences in population trends amongst breeding populations.
Historically, habitat loss and degradation have been most pronounced in the western part of the blackpoll nonbreeding range. Colombia has lost forests at a rate of 0.62%/year between 1990 and 200568. These losses have been most pronounced at the transition between the eastern Andes and the Amazonia and Orinoquia regions69, where blackpoll abundance in the nonbreeding range is estimated to be highest according to the eBird Status and Trends dataset42. In Venezuela, forest loss has historically been most pronounced in the northwestern part of the country70. In contrast, areas to the east of the blackpoll’s nonbreeding range, in southwestern Venezuela, northern Brazil, and the Guianas are still extensively forested and remain relatively intact71. If nonbreeding habitat availability limits blackpoll abundance, we would expect blackpolls from the eastern part of the breeding range to have experienced the most pronounced population declines. Population trend estimates for the period of 1970–2022 from the Breeding Bird Survey38,39 show a more nuanced pattern, with the most severe declines in the northeastern U.S., and the Canadian Maritimes, but also with steep declines in Alaska and Yukon. Contrasting even more with our expected pattern, more recent estimates from eBird (2012–2022)42,72 report steep declines in Alaska and western Canada, and increases in eastern Canada. High uncertainty and incomplete coverage of the breeding range are an issue across these large-scale trend estimates, so the underlying pattern of declines remains ambiguous. More accurate trend estimates are needed to disentangle the effects of events occurring at stopovers, the breeding range, and the nonbreeding range.
Interestingly, nearly half of the blackpolls we tracked appeared to perform long-distance movements during the ‘stationary’ nonbreeding season. Similar long-distance nonbreeding season movements have been reported in several long-distance migratory bird species14,73. However, to our knowledge, movements of a similar scale during the nonbreeding season have only been reported in one other New World warbler, the prothonotary warbler (Protonotaria citrea)74. Most of the movements we detected had a strong northward and southward component and should be interpreted with caution due to the spring equinox and generally higher uncertainty in latitude estimates close to the equator75. If we consider only movements that occurred more than 2 weeks before or after the equinox, most were still directed toward the northern part of the range and occurred late in the season. Consequently, these relocations were likely made toward staging sites used before departing South America, putting them in a migratory rather than “nonbreeding” context and further highlighting the potential importance of the northern part of the nonbreeding range for stopovers. However, not all movements that occurred during the nonbreeding season fell under this scenario, and their drivers warrant further study.
While our study focused on population-scale movement patterns, we also documented a transoceanic crossing over a route that had not previously been described using geolocators. A blackpoll tagged in Newfoundland performed a transoceanic flight from Nova Scotia to northwestern Venezuela during its post-breeding migration, possibly flying continuously for more than 84 h over an estimated distance of 4100 km, with an estimated flight speed of 13.5 m/s−1). This is 700 km longer than the longest previously reported non-stop flight for the species, which was for a western-breeding individual that flew nearly 3400 km over the Atlantic from the eastern U.S41. Even if the bird from Newfoundland had a short period of rest in the Lesser Antilles, this still represents an extreme feat of migration. By comparison, the other passerine thought to make a nonstop flight of a similar length between North America and Africa (up to 4000 km) is the northern wheatear (Oenanthe Oenanthe)76 but this species is more than twice as heavy as a blackpoll warbler on average (25 g vs. 12 g). We recommend further research into the lesser-known migration of blackpolls from Newfoundland, as well as the physiological characteristics needed to make such flights possible.
While our work provides new insights into the spatial structure of blackpoll migration, it is also important to consider its limitations. The most obvious one is the limited spatial and temporal accuracy and precision of our location estimates, particularly during the fall equinox. For instance, we placed stopover nodes 5, 6, and 7 where their longitude met the coastline of North America. While radio-tracking of individuals46 suggest prolonged stopover in proximity to the coast, some blackpolls may make long stopovers inland before initiating their transoceanic flight. Also due to the fall equinox, we likely underestimated the number of stopovers in northern Venezuela during post-breeding migration because movements from the coast to nearby inland nonbreeding sites occurred at a fine scale and close to the equinox for some individuals. The combination of Motus and the miniaturization of newly developed, multi-sensor geolocators, may help clarify uncertainties that remain around pre-breeding migration routes and transoceanic flights19,77. Multi-sensor geolocators collect light-level data alongside temperature and barometric pressure, allowing more accurate location estimates, and Motus provides high-accuracy point-to-point information for migrating birds.
Another limitation is the uneven nature of our sampling, which was characterized by a small number of individuals at some sites (Table 1) and tracks that were obtained during different years. Our sample was also male-biased and an inherent bias of geolocator tracking studies is the lack of data from individuals who did not return to the capture site, died, or could not be recaptured. This is particularly notable given our low rate of geolocator recoveries relative to similar studies on new world warblers24,25. While it is likely that we did not capture the full range of migratory behaviors at each breeding site, we believe that the networks effectively captured migratory behavior at a broad regional scale. The movements we detected spanned the majority of the known breeding, nonbreeding, and migratory ranges42,78, and the higher weights in the western nonbreeding nodes were also consistent with current estimates of abundance from eBird’s Status and Trends estimates42,79. At the same time, we note that eBird’s abundance estimates, which we used to estimate the weight of nodes in our network, may also include some inaccuracies that could influence our results. eBird checklists, the model’s main source of information, are scarce in northern Canada, and the model estimates a noticeably larger relative abundance in eastern Canada than the Boreal Avian Modelling Project80. Given the logistical challenge of deploying a large number of geolocators across the vast blackpoll breeding range, future large-scale tracking efforts could be made with stable-isotopes81 high-resolution genetic markers36, or Motus Automated Telemetry Network especially since these methods are less costly and only require capturing individuals once.
In conclusion, we provide new insights into the relative importance of stopovers across the blackpoll range and, for the first time, assess migratory connectivity between species’ breeding and nonbreeding ranges. Given the low precision and accuracy of geolocators, further studies should investigate fine-scale habitat use in the nodes that acted as points of convergence or important refueling areas and identify the factors that might pose a risk to individuals. In a broader context, the range-wide patterns of connectivity that we have unveiled here could be key to linking events occurring in different parts of the blackpoll range to changes in the population trends in other areas, and ultimately the development of full-annual cycle models82. However, it is also important to consider that migratory connectivity is not fixed in time but can instead change under the influence of underlying population dynamics8,83. Recent evidence suggests that such changes in connectivity have already occurred in blackpolls47,84, further stressing the importance of expanding population monitoring efforts for this species in conjunction with establishing its migratory connectivity patterns.
Methods
Geolocator deployment and recovery
Geolocators were deployed in 2013, 2016, 2018, 2019 and 2020 (Table 1). Light-level data from geolocators deployed at seven breeding sites in 2013 and 2016 were previously analyzed and reported by DeLuca et al. (2015) and (2019), respectively, and were re-analyzed here for consistency in methods. More recent deployments were performed at one site used in 2016, four new sites on the breeding range, and two new sites in Colombia. All blackpolls were captured using mist nets and audio playback of songs and calls. Geolocators were fitted to individuals using 32–36 mm leg-loop harnesses made with 0.5 mm black stretch magic (Pepperell Braiding Company, Pepperell, Massachusetts, USA). Each bird was also marked with a U.S. Geological Survey or Canadian Banding Office numbered aluminum band and different color band combinations so they could be identified for recapture. The geolocator models deployed included 8 Intigeo-W50 geolocators developed by Migrate Technology Ltd (~ 0.45 g with harness), and 286 geolocators of different models (MK6, ML6040, ML6440, and ML6740) developed by Biotrack/Lotek Wireless (~ 0.5 g with harness; Table 1).
In the years following each deployment, sites were revisited to recapture individuals carrying geolocators using the same capture techniques. A total of 56 geolocators were recovered (19% of deployed geolocators) across all 11 deployment locations, including 32 geolocators from 2013 and 2016 deployments, and 24 from later deployments. Not all geolocators collected data throughout the period during which they were deployed: 35 recorded data throughout the deployment period, two stopped recording light levels during pre-breeding migration, 9 recorded data during the post-breeding migration only, and the sole geolocator recovered on the nonbreeding grounds recorded data only during the pre-breeding migration. Additionally, 9 geolocators deployed in the breeding grounds failed before their carriers completed their post-breeding migration and were excluded from our analysis. Our final dataset included data from 47 birds, with post-breeding migration movements from 46 individuals and pre-breeding migration movements from 36. Only three (of 47) geolocators included in our analysis were carried by females. Additionally, only one geolocator was carried by a bird born in the deployment year, while one bird had an unknown age, and all others were born in years preceding the capture year.
Geolocator analysis
All analyses in this study were performed using R version 4.4.085. We used the threshold approach to estimate the location of birds based on the light data recorded by the geolocators. This approach is well suited for MK6 and ML6 geolocator models, which quantify light only within a limited range. Additionally, it is robust to shading and relies on day length and the timing of solar noon to determine latitude and longitude respectively86,87. We annotated twilights (i.e., successive sunrises and sunsets) in the light data of each geolocator using the preprocessLight function of the TwGeos package version 0.1.288 with an arbitrary light level threshold of 1.5 for MK6 and ML6 geolocators, and 3.0 for Intigeo models. In a small number of cases, large outliers in twilight estimates lying several hours from their neighbors were seen and manually adjusted during the annotation process. All other outliers were filtered automatically using the twilightEdit function with settings: window = 4 days, stationary.mins = 35 min, and outliers mins = 25 min. An average of 1.1% of twilight estimates were deleted (s.d. = 1.5%) per individual, and 0.3% were adjusted (s.d. = 0.5%) using this function.
To determine the error distribution of twilight times and the angle of the sun relative to its zenith during twilight (the “zenith angle”), we performed calibrations during two different periods. The first calibration period was performed by generating twilight error distributions while birds were known to be at their tagging sites based on plots of recorded light levels (“in-habitat calibration”)87. For birds tagged in North America, this was during the period before the post-breeding migration (range June 20th–Sept. 1st), and when possible, after the return to the breeding grounds following the pre-breeding migration (range May 25th–June 7th), yielding zenith angles of 92.65° on average (s.d. = 1.5). For most birds (n = 29/46), only the period preceding the post-breeding migration could be used due to their geolocators stopping before their return, or due to incomplete darkness at night at high latitudes. In-habitat calibration for the bird tagged in Colombia was performed in the nonbreeding ground, between April 4th–June 6th. Birds may experience different shading levels outside of their breeding grounds due to factors like vegetation and topography89. To attempt to limit the effect of these variations on our estimates, we performed a second calibration during the longest time span that each bird was estimated to be stationary in its nonbreeding range using the “Hill-Ekstrom” approach, where the angle that minimizes variation in latitude estimates is selected87,89. This yielded angles that differed from the in-habitat zenith angle by 1.32° on average (s.d. = 1.38). We used the angle derived from in-habitat calibration for the start of the post-breeding migration until the arrival in South America, and the angle derived from Hill-Ekstrom calibration from the arrival in South America to the departure during pre-breeding migration. For periods ranging from the departure from South America during pre-breeding migration to the recovery of the geolocator, we compared locations obtained with both angles and used the angle that showed the bird returning closest to the recapture location.
Once calibrations were complete, we inspected plots of the light data recorded by each geolocator alongside preliminary location estimates obtained using the thresholdPath function from the Solar/Satellite Geolocation for Animal Tracking (SGAT) R package90,91. We observed clock drift in six geolocators and differences between recorded times and expected Greenwich Mean Time in 20 geolocators but these issues were resolved before completing the rest of the analysis (see Supplementary Methods 2 and 3). We linearly interpolated latitude in our preliminary estimates for periods ranging between 1 and 41 days before and after the equinoxes (mean = 17 days, s.d. = 7). This was to limit erroneous location estimates caused by the spring and fall equinoxes (Mar. 19–21 and Sept. 21–24). Near the equinoxes, day length is similar at all latitudes, resulting in higher errors in latitude estimates derived from geolocators87.
To refine our preliminary location estimates, we used Markov Chain Monte Carlo (MCMC) simulations implemented with the “grouped threshold” model of the SGAT package version 0.1.387. This approach requires grouping locations where individual birds were stationary. We used the Geolight package version 2.1.091 and its changelight function to create a schedule of periods during which birds were stationary based on changes in the timing of twilights then used the mergesite functions to group stationary sites located close together. We could not use the same parameters for all tracked birds because certain values led to the mergesites function failing for some individuals but not others, an issue also reported by Hahn et al.92. We set the probability threshold defining movements to values between 0.55 and 0.97, choosing a value for each bird that best captured changes in longitude indicating movements. We set a minimum threshold of 1–2 days for stationary periods in Changelight and grouped stationary locations located within 150–250 km of each other with mergesites.
The MCMC simulations estimated the probability distribution of location estimates given our preliminary location estimates and a Gamma distribution of twilight errors. As additional prior information, we included a spatial mask restraining stationary locations to breeding, migration, and nonbreeding range polygons obtained from BirdLife International’s distribution maps for blackpolls78 (Fig. 1). Finally, the speed of each bird (the distance travelled between successive twilights) was modeled as a gamma distribution with a shape of 2.2 and scale of 0.08 (units: km h−1). As a result, the most probable speed is approx. ~ 16 km/h and faster speeds (up to ~ 50 km/h) are common, fitting for migratory flights, since we did not consider movements during stationary periods. We ran 1000 iterations on three chains as burn-in and a final run of 3000 iterations. For 18 geolocators, we noted large differences between the output from SGAT’s threshold group model and our preliminary location estimates. In these cases, we adjusted the zenith angle used for the grouped threshold model by increments of 0.5° and ran the MCMC sampler until the differences were minimized. We used the median latitude and longitude of stationary locations from the posterior distribution to build our networks.
Our geolocator analysis with SGAT could not accurately estimate locations over the Caribbean during Atlantic crossings unless birds departed long after the fall equinox. Consequently, we followed the procedure described by Deluca et al.16 to identify stopovers in the Caribbean. We reviewed the time series of light and longitude measurements collected by each geolocator to distinguish multi-day transoceanic flights and stopovers (Supplementary Method 4, Table S4, Fig. S8, S9, S10, S11). If a stopover was detected in this manner but not during the standard geolocator analysis, it was inserted manually in the Caribbean at the bird’s longitude during the stopover, taking advantage of the archipelago’s longitudinal extent. We recognize that this approach has limitations. Notably, since MK6 and ML6 series geolocators record light levels in a restricted range, periods of high illumination of the geolocator characteristic of migratory flights can be difficult to distinguish.
Defining nodes
We created two separate networks using the igraph package version 2.0.393,94 to distinguish between movement patterns during post- and pre-breeding migration. In both networks, the 11 sites on the breeding grounds where most geolocators were deployed and recovered were defined as breeding nodes. Additionally, we defined the estimated breeding site of the individual tagged in Meta, Colombia as a breeding node. We discarded all stationary locations estimated within 250 km of geolocator deployment sites as they could not reliably be considered stopovers due to geolocator error. To identify stopover and nonbreeding nodes, we clustered locations where blackpolls were estimated to be stationary for over two days after leaving their breeding node (i.e. when they might be roosting and foraging, but not making migratory flights), as well as the location where the individuals from, Meta, Colombia was tagged. We used the ‘partitioning around medoids’ function (pam) from the cluster package version 2.1.695. This function uses parameter k to specify the number of clusters to group locations. We iteratively clustered our network and increased the value of k until the median cluster diameter (maximum distance between stationary location estimates within a cluster) was closest to a specified minimum. This distance was set to 700 km to account for the spatial error of geolocators30. We used a slightly different clustering procedure in the post-breeding migratory network because of the fall equinox.
In the post-breeding migratory network, our movement data were most heavily influenced by the equinox while blackpolls were moving across North America, as well as during, and sometimes shortly after, their transoceanic flight to South America. As a result, we clustered stationary locations in North America using only longitude. Among the stopover nodes that we clustered using longitude, we placed those that were north of the Atlantic coast where their latitude intersected the coastline or the latitude of Cape Hatteras (35.3° N). However, stopovers could have occurred further inland and individuals could have used multiple locations varying in distance to the coastline. We set a southern limit at Cape Hatteras because very few blackpoll warblers are caught at banding stations below this latitude during the post-breeding migration, suggesting a low abundance96. One possible exception is southern Florida, a region where relative abundance reaches a low peak during the fall according to eBird Status and Trends data42. Since the five birds with stationary locations in southern Florida were present long enough after the equinox that we had confidence in their latitude estimates, we did not cluster their stationary estimates using only their longitude. We defined edges between all nodes linked by the movement of at least one blackpoll. These edges had directions matching those of the birds moving along them and were assigned a weight quantifying the flow of individuals moving between the nodes.
We classified clusters as stopover or nonbreeding nodes based on their use by tracked birds. First, for individual birds, we designated stationary locations derived from our geolocator analysis as stopover sites if they were outside of the South American nonbreeding grounds or in the nonbreeding grounds but occupied for less than 2 weeks. Stationary locations in the nonbreeding grounds and occupied for ≥ 2 weeks were defined as nonbreeding sites. We stopped tracking individuals in our post-breeding migration network when they reached their first nonbreeding site, meanwhile, we started tracking individuals in our pre-breeding migratory network when they left their last nonbreeding site. Subsequently, in both networks, we classified a cluster as a stopover node if it was composed exclusively of stopover sites, and as a nonbreeding node if it was composed of at least one nonbreeding site. Due to this classification system, a nonbreeding node could still include individual stopover sites, meaning that individuals could stop in multiple nonbreeding nodes before completing their migration in the post-breeding migratory network, or before leaving South America in the pre-breeding migratory network. Additionally, since the diameter of nodes was 700 km, a given node could include multiple stopovers performed by the same individual if those were made in close proximity.
To determine whether birds tracked from the eastern and western parts of the breeding range used a different number of stopover and nonbreeding nodes in each network, we used generalized linear models (GLMs). We defined western-breeders as including all birds from breeding sites west of James Bay and eastern-breeders as including all birds from breeding sites east of James Bay including the individual that was tracked from Colombia). We used a Poisson distribution with a log link and verified model fit using the DHARMA version 0.4.697. Models that displayed overdispersion were refit using a generalized Poisson distribution with a log link in the glmmTMB package version 1.1.998.
Network weighting
One approach to weighting nodes and edges is to use the number of individuals moving through them28,30. However, with uneven sampling and a low sample size, these weights are not an accurate estimate of real flows. To address this, we used an estimate of relative abundance in the breeding nodes to estimate our node weights22,23. An individual’s contribution to the weight of an edge, stopover node, or nonbreeding node was calculated as the relative abundance of blackpolls in the individual’s breeding node, divided by the number of birds tracked from the same breeding node. With this approach, birds from breeding nodes where blackpoll abundance was high had a higher individual contribution to weights, but this contribution was also controlled by the number of individuals from the same site moving through the network.
To estimate relative abundance at each breeding node, we first divided the blackpoll breeding range into four regions (Supplementary Fig. S1). These breeding regions were largely based on a prior study of genetic structure in blackpolls99. We first defined a northwestern and western region, with the former comprised of Alaska and the latter comprised of British Columbia and southern Yukon. Second, we defined a central region to include deployment sites in Manitoba and the Northwest Territories because the memberships of these locations in the eastern or western groups were unresolved genetically. Finally, we defined an eastern region to include Québec, Ontario, the Maritimes, Newfoundland, and the northeastern US. When outlining these regions, we relied as much as possible on physical barriers or major geographic landmarks, such as the Mackenzie Mountains in Alaska and James Bay in Ontario. We then estimated the relative abundance in each region using a raster layer of blackpoll breeding abundance obtained from eBird’s Status and Trends Dataset42. The relative abundance of each breeding node was calculated as the relative abundance of the node’s region, divided by the total number of other nodes in the same region (supplementary Table S1).
Migratory connectivity
We described migratory connectivity in the post and pre-breeding migratory networks by assessing population-specific patterns of node use. To do so, we estimated the relative use of each node in the post- and pre-breeding migratory network by blackpolls from the four breeding regions used to weigh the network. This required dividing the estimated abundance at each node (i.e., node weights) into the proportion contributed by individuals from each breeding region. In parallel to this, we performed a community detection analysis to determine whether the migratory networks presented any distinct group of nodes acting as “flyways” for different blackpoll populations23,28,29. We discuss the methods for this analysis in more detail in Supplementary Method 5.
We took additional steps to assess migratory connectivity between the breeding and nonbreeding grounds, this time using the nonbreeding sites of individual birds rather than nonbreeding nodes. We first focused on the distribution within the nonbreeding range. Strong migratory connectivity occurs when individuals from different breeding populations move to distinct parts of the nonbreeding range, leading to patterns such as parallel, crosswise, chain, or leap-frog migration100. To investigate the occurrence of these migration patterns, we calculated Spearman’s rank correlation coefficient between each individual’s breeding and nonbreeding longitude, as well as between their breeding and nonbreeding latitude, using both the first and last nonbreeding sites that they occupied. Due to the spatial arrangement of geolocator deployment sites along a northwest-to-southeast axis in North America, breeding latitude and longitude were highly correlated (r = − 0.89). As a result, we could not use multivariate regression to assess the effect of longitude while controlling for latitude and vice-versa.
To quantify the degree of migratory connectivity between the breeding and nonbreeding grounds, we estimated the MC metric using the MigConnectivity package version 0.4.7101. The MC metric estimates the correlation of pairwise distances between individuals during the breeding and nonbreeding seasons while accounting for uneven sampling and errors in location estimates44. Due to these properties, it is a useful tool to compare patterns of migratory connectivity between species, and it has already been used multiple times (e.g.,23,25,67). Since it approximates a correlation coefficient, the value of the MC metric ranges between − 1 and 1. Values close to 1 indicate that birds that breed close together tend to also occur in proximity during the nonbreeding season (strong migratory connectivity) and values close to 0 indicate there is no relationship between pairwise distances in the breeding and nonbreeding grounds, i.e., that individuals from different regions intermix (weak migratory connectivity). Negative values indicate that birds that are located close to one another in the breeding range tend to be further apart in their nonbreeding range. We used the same breeding regions used to weigh network edges to estimate the MC metric. In the nonbreeding range, we grouped birds whose nonbreeding sites were in the same node and defined nonbreeding regions as convex hulls around each group (supplementary Fig. S2). If any region in the nonbreeding range was composed of fewer than four individuals, it was merged with the nearest region. We again used eBird Status and Trends relative abundance data from the blackpoll breeding range42 to estimate the transition probabilities between the breeding and nonbreeding regions. A resampling procedure accounted for the bias in geolocator location estimates (longitude: − 27 ± (s.d.) 123 km, latitude: − 87 ± 121 km) and their covariance. These values were estimated by comparing estimated and actual locations while birds were still at their breeding site.
Assessing the importance of network nodes during migration
To assess the relative importance of network nodes during post- and pre-breeding migration, we used a network centrality metric and a refined version of our node weight. The centrality metric we chose was betweenness as it is useful to assess the degree to which migratory birds from different parts of a network converge toward a given node during their migration32. The betweenness of a node is defined as the number of shortest paths that pass through it to connect other nodes of the network102 and the removal of a node with a high betweenness is expected to lower network functional connectivity and impede migratory movements103. We used the inverse of edge weight estimates as intra-node distances, hence making the most used paths between nodes the shortest. Additionally, we used a version of the betweenness centrality that minimizes both the distance and number of nodes along a path, available in the tnet package version 3.0.16 (α = 0.5; Opsahl et al.104).
To obtain a refined index of relative node use, we expanded the weighting scheme to incorporate information on the time spent by individuals in each of the nodes following a similar approach to that used by Hagelin et al.105 for raster data. Using location estimates from SGAT, we quantified the period during which each bird used nodes as stationary stopovers during its migration (Supplementary Fig. S3). Since our focus was on migration, any time spent moving within a node, or using a node as a breeding or nonbreeding site was excluded. Then, for each bird, we multiplied its contribution to the node’s weight (based on relative abundance at its node of origin), by the time spent at the node. We refer to the resulting value as the “time-adjusted node weight” and it accounts for both relative abundance in the breeding range and time spent at each node. We normalized this metric by dividing it by its maximum value in both networks.
Post-migratory nonbreeding movements
Due to the difference in the location of the initial and final nonbreeding sites of many individuals in our dataset, we documented occurrences of post-migratory nonbreeding movements. These are defined as long-distance movements between distinct nonbreeding areas during the nonbreeding season that are not undertaken as part of either post- or pre-breeding migration migration73. We recorded any movements between locations occupied for at least 2 weeks during the nonbreeding period for birds with geolocators that recorded data throughout the nonbreeding season (n = 37) and the sole bird tagged in Colombia. Because 52% of these individuals did not make post-migratory nonbreeding movements and because several individuals that moved did so close to the spring equinox, we did not build a network for the nonbreeding period.
Data availability
Part of our dataset of light-level geolocator data was made accessible on MoveBank in connection with work performed by Deluca et al. in 2015 (ID: 126313959) and 2019 (ID: 959756713). The additional data we used in our analysis, from geolocators deployed after 2018, is also available on MoveBank (ID: 1988749082).
Code availability
The code used to perform our analysis is available on GitHub (Pjefferyi/Blackpoll_Warbler_migration_network_Git) and the light-level geolocator data is available on MoveBank (see “Data availability”).
References
Harris, G., Thirgood, S., Hopcraft, J., Cromsight, J. & Berger, J. Global decline in aggregated migrations of large terrestrial mammals. Endanger. Species Res. 7, 55–76 (2009).
Lascelles, B. et al. Migratory marine species: their status, threats and conservation management needs. Aquat. Conserv. Mar. Freshw. Ecosyst. 24, 111–127 (2014).
Rosenberg, K. V. et al. Decline of the North American avifauna. Science 366, 120–124 (2019).
Burns, F. et al. Abundance decline in the avifauna of the European Union reveals cross-continental similarities in biodiversity change. Ecol. Evol. 11, 16647–16660 (2021).
Webster, M. S., Marra, P. P., Haig, S. M., Bensch, S. & Holmes, R. T. Links between worlds: Unraveling migratory connectivity. Trends Ecol. Evol. 17, 76–83 (2002).
Martin, T. G. et al. Optimal conservation of migratory species. PLoS ONE 2, e751 (2007).
Runge, C. A., Martin, T. G., Possingham, H. P., Willis, S. G. & Fuller, R. A. Conserving mobile species. Front. Ecol. Environ. 12, 395–402 (2014).
Taylor, C. M. & Norris, D. R. Population dynamics in migratory networks. Theor. Ecol. 3, 65–73 (2010).
Betini, G. S., Fitzpatrick, M. J. & Norris, D. R. Experimental evidence for the effect of habitat loss on the dynamics of migratory networks. Ecol. Lett. 18, 526–534 (2015).
Marra, P. P., Hobson, K. A. & Holmes, R. T. Linking winter and summer events in a migratory bird by using stable-carbon isotopes. Science 282, 1884–1886 (1998).
Norris, D. R. & Taylor, C. M. Predicting the consequences of carry-over effects for migratory populations. Biol. Lett. 2, 148–151 (2006).
Norris, D. R. & Marra, P. P. Seasonal interactions, habitat quality, and population dynamics in migratory birds. The Condor 109, 535–547 (2007).
Kramer, G. R. et al. Population trends in Vermivora warblers are linked to strong migratory connectivity. Proc. Natl. Acad. Sci. 115, E3192–E3200 (2018).
McKinnon, E. A. & Ten Love, O. P. years tracking the migrations of small landbirds: Lessons learned in the golden age of bio-logging. Auk 135, 834–856 (2018).
Bairlein, F. et al. Cross-hemisphere migration of a 25 g songbird. Biol. Lett. 8, 505–507 (2012).
DeLuca, W. V. et al. Transoceanic migration by a 12 g songbird. Biol. Lett. 11, 20141045 (2015).
Stutchbury, B. J. M. et al. Tracking long-distance songbird migration by using geolocators. Science 323, 896–896 (2009).
Hallworth, M. T. & Marra, P. P. Miniaturized GPS tags identify non-breeding territories of a small breeding migratory songbird. Sci. Rep. 5, 11069 (2015).
Liechti, F. et al. Miniaturized multi-sensor loggers provide new insight into year-round flight behaviour of small trans-Sahara avian migrants. Mov. Ecol. 6, 19 (2018).
Scarpignato, A. L. et al. Shortfalls in tracking data available to inform North American migratory bird conservation. Biol. Conserv. 286, 110224 (2023).
Vickers, S. H., Franco, A. M. A. & Gilroy, J. J. Sensitivity of migratory connectivity estimates to spatial sampling design. Mov. Ecol. 9, 16 (2021).
Stanley, C. Q. et al. Connectivity of wood thrush breeding, wintering, and migration sites based on range-wide tracking. Conserv. Biol. 29, 164–174 (2015).
Knight, S. M. et al. Constructing and evaluating a continent-wide migratory songbird network across the annual cycle. Ecol. Monogr. 88, 445–460 (2018).
Tonra, C. M. et al. Concentration of a widespread breeding population in a few critically important nonbreeding areas: Migratory connectivity in the Prothonotary Warbler. The Condor 121, duz019 (2019).
Raybuck, D. W. et al. Cerulean Warblers exhibit parallel migration patterns and multiple migratory stopovers within the Central American Isthmus. Ornithol. Appl. 124, duac031 (2022).
Fraser, K. C. et al. Continent-wide tracking to determine migratory connectivity and tropical habitat associations of a declining aerial insectivore. Proc. R. Soc. B Biol. Sci. 279, 4901–4906 (2012).
Shimazaki, H. et al. Network analysis of potential migration routes for Oriental White Storks (Ciconia boyciana). Ecol. Res. 19, 683–698 (2004).
Lamb, J. S. et al. Spatially explicit network analysis reveals multi-species annual cycle movement patterns of sea ducks. Ecol. Appl. 29, e01919 (2019).
Merkel, B. et al. Strong migratory connectivity across meta-populations of sympatric North Atlantic seabirds. Mar. Ecol. Prog. Ser. 676, 173–188 (2021).
Lagassé, B. J. et al. Migratory network reveals unique spatial-temporal migration dynamics of Dunlin subspecies along the East Asian-Australasian Flyway. PLoS ONE 17, e0270957 (2022).
Gray, C. E. & Olsen, B. J. Migration routes, high-use areas, and network connectivity across the annual cycle of an arctic seabird. J. Ornithol. 164, 73–84 (2023).
Xu, Y. et al. Beyond site-specific criteria: Conservation of migratory birds and their habitats from a network perspective. Diversity 14, 353 (2022).
Xu, Y. et al. A network approach to prioritize conservation efforts for migratory birds. Conserv. Biol. 34, 416–426 (2020).
Buhnerkempe, M. G. et al. Identification of migratory bird flyways in North America using community detection on biological networks. Ecol. Appl. 26, 740–751 (2016).
Lamb, J. S. et al. Evaluating conservation units using network analysis: A sea duck case study. Front. Ecol. Environ. 22, e2648 (2024).
Ruegg, K. C., Harrigan, R. J., Saracco, J. F., Smith, T. B. & Taylor, C. M. A genoscape-network model for conservation prioritization in a migratory bird. Conserv. Biol. 34, 1482–1491 (2020).
DeLuca, W., Holberton, R., Hunt, P. D. & Eliason, B. C. Blackpoll Warbler (Setophaga striata), version 1.0. in Birds of the World (ed. Poole, A. F.) (Cornell Lab of Ornithology, Ithaca, NY, USA, 2020).
Hostetler, J. A., Sauer, J. R., Hines, J. E., Ziolkowski Jr., D. & Lutmerding, M. The North American Breeding Bird Survey, Analysis Results 1966 - 2022. U.S. Geological Survey https://doi.org/10.5066/P9IU1UZ6 (2023).
Smith, A. C. et al. North American Breeding Bird Survey. Canadian Trends Website, Data version 2021 https://wildlife-species.canada.ca/breeding-bird-survey-results/P001/A001/?lang=e (2023).
Hill, J. M. The State of the Mountain Birds Report: 2023. Vermont Center for Ecostudies, White River Junction, VT. https://mountainbirds.vtecostudies.org/ (2023).
DeLuca, W. V. et al. A boreal songbird’s 20,000 km migration across North America and the Atlantic Ocean. Ecology 100, e02651 (2019).
Fink, D. et al. eBird Status and trends, data version: 2022. Cornell Lab Ornithol. https://doi.org/10.2173/ebirdst.2022 (2023).
Pons, P. & Latapy, M. Computing Communities in Large Networks Using Random Walks. in Computer and Information Sciences - ISCIS 2005 (eds. Yolum, pInar, Güngör, T., Gürgen, F. & Özturan, C.) 284–293 (Springer, Berlin, Heidelberg, 2005). https://doi.org/10.1007/11569596_31.
Cohen, E. B. et al. Quantifying the strength of migratory connectivity. Methods Ecol. Evol. 9, 513–524 (2017).
Holberton, R. L., Van Wilgenburg, S. L., Leppold, A. J. & Hobson, K. A. Isotopic (δ2Hf) evidence of “loop migration” and use of the Gulf of Maine Flyway by both western and eastern breeding populations of Blackpoll Warblers. J. Field Ornithol. 86, 213–228 (2015).
Smetzer, J. R. & King, D. I. Prolonged stopover and consequences of migratory strategy on local-scale movements within a regional songbird staging area. The Auk 135, 547–560 (2018).
Dunn, E. H. et al. Feather isotopes (δ2Hf) and morphometrics reveal population-specific migration patterns of the Blackpoll Warbler (Setophaga striata). Avian Conserv. Ecol. 18 (2023).
Holberton, R. L., Van Wilgenburg, S. L., Leppold, A. J. & Hobson, K. A. Erratum: Isotopic (δ2Hf) evidence of “loop migration” and use of the Gulf of Maine Flyway by both western and eastern breeding populations of Blackpoll Warblers. J. Field Ornithol. 90, 286–288 (2019).
Covino, K., Morris, S., Shieldcastle, M. & Taylor, P. Spring migration of Blackpoll Warblers across North America. Avian Conserv. Ecol. 15 (2020).
Drummond, M. A. & Loveland, T. R. Land-use pressure and a transition to forest-cover loss in the Eastern United States. BioScience 60, 286–298 (2010).
Morris, S. R., Covino, K. M., Jacobs, J. D. & Taylor, P. D. Fall migratory patterns of the Blackpoll Warbler at a continental scale. The Auk 133, 41–51 (2016).
Nisbet, I. C. T. Autumn migration of the blackpoll warbler: Evidence for long flight provided by regional survey. Bird-Band. 41, 207–240 (1970).
Latta, S. C. & Brown, C. Autumn stopover ecology of the Blackpoll Warbler (Dendroica striata) in thorn scrub forest of the Dominican Republic. Can. J. Zool. 77, 1147–1156 (1999).
Schaub, M., Jenni, L. & Bairlein, F. Fuel stores, fuel accumulation, and the decision to depart from a migration stopover site. Behav. Ecol. 19, 657–666 (2008).
Gómez, C. et al. Fuel loads acquired at a stopover site influence the pace of intercontinental migration in a boreal songbird. Sci. Rep. 7, 3405 (2017).
Halupka, L., Wierucka, K., Sztwiertnia, H. & Klimczuk, E. Conditions at autumn stopover sites affect survival of a migratory passerine. J. Ornithol. 158, 979–988 (2017).
Smith, R. J. & Moore, F. R. Arrival fat and reproductive performance in a long-distance passerine migrant. Oecologia 134, 325–331 (2003).
Schaub, M., Jakober, H. & Stauber, W. Demographic response to environmental variation in breeding, stopover and non-breeding areas in a migratory passerine. Oecologia 167, 445–459 (2011).
Rushing, C. S., Ryder, T. B. & Marra, P. P. Quantifying drivers of population dynamics for a migratory bird throughout the annual cycle. Proc. R. Soc. B Biol. Sci. 283, 20152846 (2016).
Paxton, E. H., Durst, S. L., Sogge, M. K., Koronkiewicz, T. J. & Paxton, K. L. Survivorship across the annual cycle of a migratory passerine, the willow flycatcher. J. Avian Biol. 48, 1126–1131 (2017).
Bayly, N. J., Norris, D. R., Taylor, P. D., Hobson, K. A. & Morales-Rozo, A. There’s no place like home: Tropical overwintering sites may have a fundamental role in shaping migratory strategies. Anim. Behav. 162, 95–104 (2020).
Bayly, N. J., Rosenberg, K. V., Norris, D. R., Taylor, P. D. & Hobson, K. A. Rapid recovery by fat- and muscle-depleted Blackpoll Warblers following trans-oceanic migration is driven by time-minimization. Ornithology 138, ukab055 (2021).
Bosque, C. & Lentino, M. The passage of North American migratory land birds through xerophytic habitats on the western coast of Venezuela. Biotropica 19, 267–273 (1987).
Norris, D. R. et al. Migratory connectivity of a widely distributed songbird, the American redstart (Setophaga ruticilla). Ornithol. Monogr. 61, 14–28 (2006).
Vincent, J. G.-A. Clustering community science data to infer songbird migratory connectivity in the Western Hemisphere. (Carleton University, 2020). https://doi.org/10.22215/etd/2020-14223
Witynski, M. L. & Bonter, D. N. Crosswise migration by Yellow Warblers, Nearctic-Neotropical passerine migrants. J. Field Ornithol. 89, 37–46 (2018).
Sharp, A. J. et al. The strength of migratory connectivity in painted buntings is spatial scale dependent and shaped by molting behavior. J. Field Ornithol. 94, 7 (2023).
Armenteras, D., Cabrera, E., Rodríguez, N. & Retana, J. National and regional determinants of tropical deforestation in Colombia. Reg. Environ. Change 13, 1181–1193 (2013).
Rodríguez Eraso, N., Armenteras-Pascual, D. & Alumbreros, J. R. Land use and land cover change in the Colombian Andes: Dynamics and future scenarios. J. Land Use Sci. 8, 154–174 (2013).
Pacheco, C., Aguado, I. & Mollicone, D. Dinámica de la deforestación en Venezuela: Análisis de los cambios a partir de mapas históricos. Interciencia 36, 578–586 (2011).
Potapov, P. et al. The last frontiers of wilderness: Tracking loss of intact forest landscapes from 2000 to 2013. Sci. Adv. 3, e1600821 (2017).
Fink, D. et al. A double machine learning trend model for citizen science data. Methods Ecol. Evol. 14, 2435–2448 (2023).
Teitelbaum, C. S., Bachner, N. C. & Hall, R. J. Post-migratory nonbreeding movements of birds: A review and case study. Ecol. Evol. 13, e9893 (2023).
Wolfe, J. D. & Johnson, E. I. Geolocator reveals migratory and winter movements of a Prothonotary Warbler. J. Field Ornithol. 86, 238–243 (2015).
McKinnon, E. A. et al. Estimating geolocator accuracy for a migratory songbird using live ground-truthing in tropical forest. Anim. Migr. 2, 1–8 (2013).
Bulte, M. et al. Can wheatears weather the Atlantic? Modeling nonstop trans-Atlantic flights of a small migratory songbird. Auk 131, 363–370 (2014).
Rime, Y. et al. Multi-sensor geolocators unveil global and local movements in an Alpine-breeding long-distance migrant. Mov. Ecol. 11, 19 (2023).
BirdLife International and Handbook of the Birds of the World. Version 2022.2. Bird species distribution maps of the world. http://datazone.birdlife.org/species/requestdis (2022).
Fink, D. et al. Modeling avian full annual cycle distribution and population trends with citizen science data. Ecol. Appl. 30, e02056 (2020).
Boreal Avian Modelling Project. BAM Generalized National Models Documentation, Version 4.0. https://doi.org/10.5281/ZENODO.4018335 (2024).
Studds, C. E., Wunderle, J. M. Jr. & Marra, P. P. Strong differences in migratory connectivity patterns among species of Neotropical-Nearctic migratory birds revealed by combining stable isotopes and abundance in a Bayesian assignment analysis. J. Biogeogr. 48, 1746–1757 (2021).
Saracco, J. F. & Rubenstein, M. Integrating broad-scale data to assess demographic and climatic contributions to population change in a declining songbird. Ecol. Evol. 10, 1804–1816 (2020).
Taylor, C. M. Effects of natal dispersal and density-dependence on connectivity patterns and population dynamics in a migratory network. Front. Ecol. Evol. 7 (2019).
Gómez, C. et al. Migratory connectivity then and now: A northward shift in breeding origins of a long-distance migratory bird wintering in the tropics. Proc. R. Soc. B Biol. Sci. 288, 20210188 (2021).
R Core Team. (2024). R: A Language and Environment for Statistical Computing. Version 3.4.1. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Ekstrom, P. An advance in geolocation by light. Mem. Natl. Inst. Polar Res. Spec. Issue (2004).
Lisovski, S. et al. Light-level geolocator analyses: A user’s guide. J. Anim. Ecol. 89, 221–236 (2019).
Lisovski, S., Wotherspoon, S. & Sumner, M. TwGeos: Basic data processing for light-level geolocation archival tags https://github.com/SLisovski/TwGeos (2016).
Lisovski, S. et al. Geolocation by light: Accuracy and precision affected by environmental factors. Methods Ecol. Evol. 3, 603–612 (2012).
Sumner, M. D., Wotherspoon, S. J. & Hindell, M. A. Bayesian estimation of animal movement from archival and satellite Tags. PLoS ONE 4, e7324 (2009).
Lisovski, S. & Hahn, S. GeoLight-processing and analysing light-based geolocator data in R. Methods Ecol. Evol. 3, 1055–1059 (2012).
Hahn, S. et al. Range-wide migration corridors and non-breeding areas of a northward expanding Afro-Palaearctic migrant, the European Bee-eater Merops apiaster. Ibis 162, 345–355 (2020).
Csárdi, G. & Nepusz, T. The igraph software package for complex network research. InterJournal Complex Syst. 1695, 1–9 (2006).
Csárdi, G. et al. igraph: network analysis and visualization in R. R package version 2.0.3. https://CRAN.R-project.org/package=igraph (2024).
Maechler, M., Rousseeuw, P., Struyf, A., Hubert, M. & Hornik, K. Cluster: Cluster analysis basics and extensions. R package version 2.1.6 https://cran.r-project.org/web/packages/cluster/index.html (2023).
McNair, D. B. & Nisbet, I. C. T. Status and abundance of blackpoll warblers in autumn on the coast of the southeastern United States: an update. Southeast. Nat. 19, 241–255 (2020).
Hartig, F. DHARMa: Residual diagnostics for hierarchical (multi-level / mixed) regression models. R package version 0.4.6. https://cran.r-project.org/package=DHARMa (2022).
Brooks, M. E. et al. glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J. 9, 378–400 (2017).
Ralston, J. & Kirchman, J. J. Continent-scale genetic structure in a boreal forest migrant, the blackpoll warbler (Setophaga striata). Auk 129, 467–478 (2012).
Boulet, M. & Norris, D. R. Introduction: The past and present of migratory connectivity. Ornithol. Monogr. 61, 1–13 (2006).
Hostetler, J. A. & Hallworth, M. T. MigConnectivity: Estimate migratory connectivity for migratory animals. R package version 0.4.7 https://CRAN.R-project.org/package=MigConnectivity (2024).
Freeman, L. C. A set of measures of centrality based on betweenness. Sociometry 40, 35–41 (1977).
Xu, Y. et al. Loss of functional connectivity in migration networks induces population decline in migratory birds. Ecol. Appl. 29, e01960 (2019).
Opsahl, T., Agneessens, F. & Skvoretz, J. Node centrality in weighted networks: Generalizing degree and shortest paths. Soc. Netw. 32, 245–251 (2010).
Hagelin, J. C. et al. Revealing migratory path, important stopovers and non-breeding areas of a boreal songbird in steep decline. Anim. Migr. 8, 168–191 (2021).
Acknowledgements
We thank Nikole Freeman, Alex Sutton, Amy Newman, Lila Tauzer, Carol McIntyre, Scott Weidensaul, Iain Stenhouse, Laura Phillips, Emily Williams, Charles Grigsby, Jill Boelsma, George Gress, Darroch Whittaker, Ian Warkentin, Jean-Daniel Fiset, Lesley Howes, Camille Bégin-Marchand, Vincent Lamarre, Yves Aubry, and Nick Bayly for their work in the field and/or for logistical support, and to Brad Woodworth for an earlier analysis of geolocator data. We also thank all students, field technicians, and researchers who took part in geolocator deployments and recoveries, Elizabeth Gow, Michael Hallworth, and Daizaburo Shizuka for their advice related to the analysis, and BirdLife International for providing digital maps of the blackpoll warbler’s range. This study was supported by the U.S National Park Service Critical Connections Program, Migratory Bird Center, University of Guelph, Gabonese Scholarship Agency, SELVA: Investigación para la Conservación en el Neotrópico, Kenneth Molson Foundation, Canadian Foundation for Innovation, Birds Canada, Natural Sciences and Engineering Research Council of Canada, the Department of the Interior Northeast Climate Adaptation Science Center, Vermont Center for Ecostudies, National Audubon Society, Earl L. and Barbara Doolin, Wildlife Conservation Society Canada, Norcross Wildlife Foundation, Forest Ecosystem Monitoring Cooperative, Department of the Interior Northeast Climate Adaptation Science Center, Wildlife Conservation Society Canada, Garfield Family Foundation, Teslin Tlingit Council, the estate of Fred Bodsworth, Bering Straits Council, Solomon Native Corporations, Churchill Northern Science Centre, CNSC Northern Research Fund, Denali Education Center, Alaska Geographic, and Camp Denali.
Author information
Authors and Affiliations
Contributions
D.R.N, J.D., and W.V.D. conceived the study. W.V.D., C.A.H., K.P.M., and C.C.R. conducted fieldwork related to geolocator deployments made before 2018 and S.H., A.R.-C., J.T., B.D., H.A.C., R.B., M.H.-R., and W.V.D conducted fieldwork in 2018 and after. D.V.W., S.A.M., and W.V.D. collated the data from the various field sites. J.D. conducted the analysis and wrote the manuscript with support from D.R.N. Subsequently, W.V.D., S.H., P.P.M., A.R.-C., S.A.M., K.P.M., B.D., and P.D.T. provided feedback on the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The author(s) declare no competing interests.
Ethical statements
All geolocator deployments and recoveries made as part of this study were made under the appropriate government and academic permits, and birds were manipulated following national ethical guidelines. Deployments in British Colombia and Yukon in 2018, were carried out under an Environmental and Climate Change Canada (ECCC) permit to capture and band migratory birds (10169 BZ). Deployments in Denali National Park, in 2018, were performed under a United States Geological Survey Bird Banding Permit (24141) and National Park Service Institutional Animal Care and Use Committee authorization (“AK DENA WRST McIntyre Birds 2018.A3”). Deployments in Québec, in 2019, occurred under two ECCC Canada Animal Care Committee permits (19JT05 and 20JT05). Deployments in Newfoundland, in 20219, were performed with an ECCC permit (10616). Deployments in Yellowknife, in 2019, occurred with authorization from the Government of the Northwest Territory Wildlife Care Committee (NWTWCC 2019–011) and a Government of the Northwest Territory Wildlife Research Permit (WL500745). Deployments in Colombia, in 2020, were carried out by SELVA under a permit from the Autoridad Nacional de Licencias Ambientales (ANLA) under Resolución 00874 issued in 2018.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Duali, J., DeLuca, W.V., Mackenzie, S.A. et al. Range-wide post- and pre-breeding migratory networks of a declining neotropical–nearctic migratory bird, the blackpoll warbler. Sci Rep 14, 30229 (2024). https://doi.org/10.1038/s41598-024-80838-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-80838-9