Abstract
This study investigated the frequency of idiopathic epistaxis onset and its severity relative to the time of day. Idiopathic epistaxis was defined as epistaxis in the emergency department, with no epistaxis diagnosis in any hospital 12 months before incident epistaxis. The timing of epistaxis onset was divided into four categories: morning (6:01–12:00), afternoon (12:01–18:00), evening (18:01–24:00), and overnight (00:01–6:00). The chi-square test was used to analyse the time distribution of epistaxis (p ≤ .05).. During the study period, a total of 1684 patients with a median age of 56 years developed epistaxis. Epistaxis incidence was highest in December (n = 213), lowest in July (n = 95), and highest in winter, followed by fall, spring, and summer. Epistaxis occurred most frequently overnight (n = 823, 48.8%), followed by evening (n = 410, 24.3%), morning (n = 254, 15.1%), and afternoon (n = 197, 11.7%; p < .001). Afternoon was selected as the reference. After adjustment for covariates, odds ratios (ORs) and 95% confidence intervals (CIs) of the risk of epistaxis were 1.34 (95% CI, 1.26–1.43), 1.47 (1.38–1.56), and 3.52 (3.14–3.91) in the evening, morning, and overnight, respectively. Epistaxis rates overnight were significantly strongest and positively proportional to blood pressure levels between 0:01 am and 6:00 am (r = 18.1, p < .001), followed by overnight rates (r = 11.5, p = .017). Patients who developed epistaxis overnight were more likely to develop posterior epistaxis and to receive endoscopic electrocoagulation haemostasis (p < .001). In this cohort, these results suggest that adult idiopathic epistaxis occurred most frequently at overnight, especially in the winter, and more need management to immediately control the bleeding in ENT emergency departments. In addition, this study found that high frequent of epistaxis at overnight associated with serious blood pressure between 0:01 am and 6:00 am. This analysis supports that health care professionals and caregiver should be aware of individuals with hypertension, and the status of blood pressure at overnight should be considered in preventing nasal bleeding’s risk.
Similar content being viewed by others
Introduction
An epidemiologic study revealed that epistaxis occurs in up to 60% of the general population and that approximately 6% of individuals with epistaxis seek medical attention1. Studies have found a bimodal age distribution for epistaxis. Individuals aged younger than 10 years or 70 years or older are at most risk of epistaxis; older adults with epistaxis are three times more likely to require medical assistance than are children with epistaxis2,3. Men are slightly more likely than women to develop epistaxis4.
Epistaxis is classified as primary or secondary. Most patients (approximately 85%) develop primary epistaxis. Primary epistaxis is mostly idiopathic and arises spontaneously without any obvious precipitating insult, although environmental factors are likely to contribute to the onset of primary epistaxis. The causes of and risk factors for secondary epistaxis are classified as local (e.g., dry air, alcohol abuse, infection, chemical irritants, inflammation, allergy, trauma, cancer, and intranasal medication application) or systemic (e.g., use of anticoagulants and antiplatelet agents, blood dyscrasias, leukaemia, atherosclerosis, and congestive heart failure). Recurrent epistaxis may be the first sign of local or systemic neoplastic disease. Several reports have suggested that hypertension is a crucial systemic disease associated with the development of epistaxis. Additionally, several studies have reported a significant positive association between epistaxis and atmospheric pressure or relative humidity and seasonal changes5,6,7. Individuals in colder regions are considerably more likely to develop epistaxis in winter. This is most likely a result of the colder climate and exposure to indoor heating in winter, which results in low ambient humidity and more water evaporation from the nasal mucosa; these factors increase the risks of desiccation and epistaxis. According to anecdotal reports from caretakers, otolaryngologists, and primary care doctors treating severe epistaxis, epistaxis seems to occur more frequently in the evening or overnight. Although the frequency and management of epistaxis have been extensively studied, data on the timing of epistaxis occurrence are lacking. By being aware of the time of day when epistaxis is most likely to occur, caregivers and medical professionals can implement more careful monitoring of patients, thereby enhancing clinical outcomes. Accordingly, we conducted this retrospective study to assess the time of day of epistaxis onset in patients at a hospital within the Chinese tertiary academic health system to determine whether epistaxis occurs more commonly in the evening. We also evaluated the predictors of the timing of epistaxis.
Methods
Study population
This retrospective study was performed at the affiliated hospital of Yangzhou University, Yangzhou, China, between January 2017 and January 2022. This study was conducted in adherence to the tenets of the Declaration of Helsinki. This study was approved by the ethics committee of the affiliated hospital of Yangzhou University. Written informed consent was obtained from all participants. We screened the medical records of all patients to identify individuals who met the inclusion criteria. We selected only patients whose medical records mentioned ear, nose, throat (ENT) emergency department visits for epistaxis. Specifically, the main medical records were required to include the recorded time (hour) of epistaxis onset, as provided by the patient or caregiver, and the month of the ENT emergency department visit. Epistaxis episodes were restricted to those that occurred in outpatient settings.
Main outcome and data collection
The main outcome in this study was treatment for epistaxis episodes in the ENT emergency department. In this study, epistaxis was defined as idiopathic epistaxis diagnosed in the emergency department (International Classification of Diseases, Tenth Revision code R04.0), with no epistaxis diagnosis in any Medicare claims record (outpatient, inpatient, or health-care provider) before incident epistaxis. The emergency department arrival time was recorded, when available. Timing of epistaxis onset was divided into four categories: morning (6:01–12:00), afternoon (12:01–18:00), evening (18:01–24:00), and overnight (00:01–6:00). Season of epistaxis onset was classified as spring (March, April, and May), summer (June, July, and August), fall (September, October, and November), or winter (December, January, and February). Strategies for controlling epistaxis were classified using procedure codes as cauterisation, nasal packing (anterior or posterior), or endoscopic electrocoagulation (unipolar or bipolar). Patients were excluded if they (1) had traumatic, iatrogenic, coagulopathic, or recurrent epistaxis; (2) had other risk factors for epistaxis, including acute rhinitis, allergic rhinitis, sinonasal tumour, rhinologic surgery, an epistaxis tendency, or a coagulation disorder; (3) were receiving medical treatment that could significantly increase the risk of epistaxis (i.e., heparin, enoxaparin, clopidogrel, ticlopidine, abciximab, warfarin, and low-dose aspirin); (4) were using an intranasal spray (i.e., a spray containing steroids, antihistamines, or ipratropium bromide) for treating rhinitis and allergic rhinitis; or (5) were lacking epistaxis time and treatment option data.
Covariates
Demographic characteristics in this study included age and sex. Lifestyle risk factors included smoking status and alcohol consumption status. Body mass index (BMI; calculated as weight in kilograms divided by height in metres squared) was calculated using self-reported weight and height data. A lifestyle risk factor was the presence or absence of hypertension and diabetes mellitus.
Statistical analysis
All statistical analyses were performed using SPSS version 26.0. Categorical variables are expressed as numbers and percentages. Continuous variables are expressed as mean and standard deviation (M ± SD). Comparisons of categorical variables were performed using chi-square tests or Fisher’s exact test. The Mann–Whitney U test or unpaired Student’s t test was applied for analysing continuous variables. Analysis of variance was used to examine the relationship between the four seasons and epistaxis rates. Multiple comparisons were performed using a post hoc Tukey’s honestly significant difference test. Spearman’s correlation was estimated for determining the relationships between epistaxis rates and blood pressure variation within a day. After adjustment for demographic characteristics and lifestyle factors, a multivariate logistic regression analysis was used to calculate odds ratios (ORs) and 95% confidence intervals (CIs) for evaluating the association between the occurrence of epistaxis and the time of day. Two-sided p values of < 0.05 were considered statistically significant.
Results
Demographic characteristics of the study population
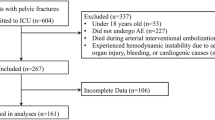
During the study period, ENT emergency department arrival times were mentioned in the medical records of a total of 1795 adult patients. A total of 38 patients were excluded due to a lack of medical records mentioning the timing of epistaxis (N = 19) or other details (N = 9). The remaining 1757 patients had records that provided the timing of epistaxis. Of these patients, 45 lacked data on the specific hour of epistaxis onset. For these 45 patients, the time of epistaxis onset was recorded as morning (n = 24, 63%), afternoon (n = 2, 5%), or evening (n = 12, 32%). The remaining 1712 patients had medical records that provided the precise timing of epistaxis onset. After 28 patients with secondary epistaxis were excluded, 1684 patients with primary epistaxis were included in the analysis.
Among the 1684 patients with epistaxis, 1032 (61.3%) were men. The median age was 56 years (range, 18–88 years, Table 1). The age distribution of the patients was as follows: 96 (5.7%) patients were aged 18–35 years, 221 (13.1%) patients were aged 36–50 years, 483 (28.6%) patients were aged 51–65 years, 686 (40.8%) patients were aged 66–80 years, and 198 (11.7%) patients were aged ≥ 81 years (χ2 = 46.596, p < .001, Table 1). In the detailed analysis by the time of epistaxis onset, 21 (8.3%) patients aged 18–35 years developed epistaxis in the morning. Moreover, 22 (11.2%), 25 (6.1%), and 28 (3.4%, p < .001) patients aged 18–35 years developed epistaxis in the afternoon, evening, and overnight, respectively. Among patients aged 36–50 years, 48 developed epistaxis occurred in the morning (18.9%), 24 developed epistaxis in the afternoon (12.2%), 64 developed epistaxis in the evening (15.6%), and 85 developed epistaxis overnight (10.3%; p < .001). In the analysis, 66 (13.7%) patients aged 51–65 years developed epistaxis in the morning. In addition, 62 (31.5%), 108 (26.3%), and 247 (30.0%; p < .001) patients aged 51–65 years developed epistaxis in the afternoon, evening, and overnight, respectively. Among patients aged 66–80 years, epistaxis occurred in the morning, afternoon, evening, and overnight for 96 (37.8%), 71 (36.0%), 155 (37.8%), and 364 (44.2%) patients (p < .001), respectively. Among patients aged ≥ 81 years, 23 (9.1%), 18 (9.1%), 58 (14.1%), and 99 (5.9%; p < .001) developed epistaxis in the morning, afternoon, evening, and overnight, respectively.
As presented in Tables 1 and 67.8% (1142) of patients with epistaxis had a history of hypertension. The majority of these patients [82.3% (665/832)] developed epistaxis overnight, followed by epistaxis development in the evening, afternoon, and morning. In the detailed analysis by the mean of blood pressure levels, the highest mean of systolic blood pressure and diastolic blood pressure was found in the overnight group, followed by the evening, morning, and afternoon groups. Table 1 also reveals that among the 823 cases of overnight epistaxis, 296(37%) were attributed to anterior epistaxis and 527(64%) to posterior epistaxis. Of these cases, 649(78.9%) were treated with endoscopic electrocoagulation under local anesthesia, whereas 109(13.2%) necessitated surgical intervention under general anesthesia.
Of the 1684 patients, 1451 (86.2%) were immediately discharged from the emergency department after the management of epistaxis. The management strategies were endoscopic electrocoagulation haemostasis under local anaesthesia in the emergency department in 1164 patients, nasal packing in 185 patients, and observation in 102 patients. In total, 223 (13.8%) patients were admitted to the hospital for inpatient endoscopic surgical intervention under general anaesthetic, because of the inability to perform endoscopic electrocoagulation haemostasis due to severely deviated nasal septum or pain. In the follow-up in the first month after epistaxis management, the recurrent epistaxis rates were 3.1% (36/1164), 25.9% (48/185), 49% (50/102), and 3% (6/203) for patients who underwent electrocoagulation, nasal packing, observation, and surgical intervention (p < .001), respectively. As listed in Table 1, posterior epistaxis was more common than anterior epistaxis in the overnight (χ2 = 103.755, p < .001). By contrast, anterior epistaxis was more common than posterior epistaxis in the morning, afternoon, and evening (all p < .001). In the analysis by epistaxis time, no statistical difference was found in BMI, location, smoking status, alcohol consumption status, or history of diabetes between the morning, afternoon, evening, and overnight groups (all p > .05).
Timing of epistaxis
Data on the timing of epistaxis onset are provided in Fig. 1. The frequency of epistaxis episodes was 15.1% (n = 254) in the morning, 11.7% (n = 197) in the afternoon, 24.3% (n = 410) in the evening, and 48.8% (n = 823) overnight (p < .001). Afternoon was selected as the reference because the proportion of patients with epistaxis was the lowest in the day. After adjustment for all covariates, multivariate ORs of the risk of epistaxis were 1.34 (95% CI, 1.26–1.43), 1.47 (1.38–1.56), and 3.52 (3.14–3.91) in the morning, evening, and overnight groups, respectively.
The frequency of epistaxis presentations by relative time of day from Jan 2017 to Jan 2022. Morning (6:01–12:00), afternoon (12:01–18:00), evening (18:01–24:00), and overnight (00:01–6:00).
Figure 2 displays the overall number of epistaxis cases by month throughout the research period. On a monthly basis, the numbers of epistaxis cases were highest in December (n = 213) and lowest in July (n = 95). The incidence of epistaxis was highest in winter, followed by fall, spring, and summer (Fig. 2). The incidence of epistaxis in the four seasons was compared using analysis of variance. The results revealed a significant difference the incidence of epistaxis between the seasons (p < .001). Post hoc Tukey’s honestly significant difference analysis revealed significant differences in the incidence of epistaxis between summer and winter (p < .001) and between summer and fall (p < .001).
The frequency of epistaxis presentations by relative time of month From Jan 2017 to Jan 2022. The seasons that we defined in this study, the spring (March, April, May), the summer (June, July, August), fall (September, October, November) and winter (December, January, February).
Relationship between timing of epistaxis and hypertension
In total, 1142 of the patients had hypertension and visited the ENT emergency department for ongoing epistaxis. We investigated whether the timing of epistaxis was affected by blood pressure level variations within the day. We examined epistaxis rates and hypertension status in morning, afternoon, evening, and overnight groups. The results revealed that the overnight group was significantly more likely to be admitted to the emergency department for epistaxis than the other groups. In total, 59.3% (677/1142) of patients with hypertension were admitted overnight, followed by evening (22.6%, 258/1142), morning (9.9%, 113/1142), and afternoon (8.2%, 94/1142; p < .001). Among patients without hypertension, 28% (152/542) were admitted overnight. Patients without hypertension were more likely to be admitted to the emergency department overnight than in the evening (26.9%, 146/542), morning (26%, 141/542), or afternoon (19%, 103/542; p < .001). We further analysed the association between the site of epistaxis and hypertension. The analysis indicated that 60.9% (696/1142) of patients with hypertension had posterior epistaxis and that 53% (288/542) of patients without hypertension had anterior epistaxis.
To further examine the effects of blood pressure variations on epistaxis rates, we calculated Spearman’s correlation values to estimate the correlation between epistaxis rate and blood pressure level. Epistaxis rates in the overnight group were significantly strongest and positively proportional to blood pressure level (r = 18.1, p < .001), followed by those in the evening group (r = 11.5, p = .017). However, no correlation was found between blood pressure level and epistaxis rate in the morning and afternoon groups.
Discussion
This study used medical data from the Chinese tertiary academic health system to evaluate whether frequency and severity of idiopathic epistaxis in adults are associated with the time of day. Our study found that idiopathic epistaxis most frequently occurred overnight (0:01 am–6 am). The risk of epistaxis overnight was 3.52 times higher than the risk in the afternoon (adjusted OR, 3.52; 95% CI, 3.14–3.91), followed by the risks in the evening (OR, 1.47; 95% CI, 1.38–1.56) and morning (OR, 1.34; 95% CI, 1.26–1.43). Additionally, most epistaxis cases occurred overnight, and the majority of epistaxis cases required endoscopic electrocoagulation haemostasis. Consistent with findings from other studies, our study found that epistaxis frequency was highest in winter, followed by fall, spring, and summer. During the study period, the incidence of idiopathic epistaxis in adults was highest in December, January, and February. In addition, patients in the overnight group were more likely to develop posterior epistaxis and to receive endoscopic electrocoagulation haemostasis than the other groups (p < .001). To the best of our knowledge, the present study is the first to assess the association between epistaxis and onset time. We also observed a strong association between epistaxis and onset time. Epistaxis was nearly 3 times more likely to develop overnight than in the afternoon. Elevated blood pressure between 0:01 am and 6:00 am is a possible risk factor for overnight epistaxis (posterior epistaxis mostly) occurrence.
Our findings align with those of studies reporting on the variation in epistaxis incidence between seasons. Several studies revealed no significant association between epistaxis incidence and season8,9. Stopa and Scho¨nweiler conducted a retrospective study of 230 patients visiting a university ENT department in Essen, Germany, in 1986 and found that most patients developed epistaxis between September and March10. In another retrospective study, Walker et al. demonstrated that admission rates were lowest in summer, especially in August, and highest between November and May, with a peak in January11. In contrast to other studies, the present study is the first to identify a seasonal variation of epistaxis presentation to the ENT emergency department in a city in south China. This study demonstrated that the highest epistaxis rates were found in December to February, with a peak in December. These results are similar to those of Pallin et al.12. The higher incidence of epistaxis in the colder climate is likely related to the effects of both low temperatures and the frequent use of indoor heating, which contribute to low relative humidity. In low-humidity conditions, water tends to evaporate from the airway and nasal surfaces, resulting in mucosal desiccation and a predisposition to epistaxis. We believe that the variability of the results in the literature is related to population characteristics, variation in comorbidities in different regions, and geographical climate and environmental factors.
The first meaningful finding of this study is the distribution of epistaxis onset time. We observed a strong association between epistaxis onset and time of day. The frequency of epistaxis overnight was nearly 3 times that in the afternoon. The variation of the time of day for epistaxis rates has not been reported. Although the literature suggests that infection, allergy, trauma, the use of anticoagulants and antiplatelet agents, or dehydration may disturb clot formation and lead to epistaxis13. Nevertheless, the exact mechanism underlying the association between 24-hour variations in the time of day with epistaxis rates remains unclear. According to ENT doctors, epistaxis may occur more frequently in the evening and overnight. However, this trend has not been investigated in the literature. The underlying risk factor is the physiological nasal cycle, which involves the involuntary alternating contraction and dilation of blood vessels in the mucosa of the inferior turbinate. This natural process can lead to congestion of the mucous membrane blood vessels in the respiratory area of the nasal mucosa overnight14. Thus, changes in the temperature, air humidity, and atmospheric pressure may lead to the disorder of the nasal cavity’s self-regulation mechanism, resulting in epistaxis overnight15,16. Thus, a combination of factors, including environmental factors (humidity and temperature), local factors (inflammation, deviated septum, and perforation), and systemic factors (clotting time variation), may create an environment that increases the risk of epistaxis overnight. Our study demonstrated a significant association between epistaxis onset and time of day. The study findings can enable a more nuanced investigation of the aetiology of idiopathic epistaxis. Ultimately, such an investigation can lead to changes in the management of epistaxis and can inform the prevention strategies of epistaxis, which will lead to more favourable patient outcomes.
Many mechanisms may contribute to the time-of-day variation in epistaxis rates. Several studies, with different strengths of evidence, have demonstrated that the role of the circadian rhythm in the pathophysiology of the time-of-day variation for different clinical conditions. For example, in asthma, a circadian cycle has been found for the bronchial reactivity of individuals with asthma, with a notable nighttime increase in bronchial reactivity17,18. Bronchial responsiveness in individuals with asthma may be increased by circadian fluctuations in cortisol, with the lowest bronchial responsiveness reported around midnight, and by the changes in the circulation levels of histamine and epinephrine19,20. The circadian cycle of the vagal tone also increases at night. Another example is myocardial infarction. Changes in the circadian rhythm affect the development of stroke and venous thromboembolism21,22,23. Numerous studies have demonstrated that strokes tend to occur in the morning (between 6:00 am and noon), when the circulating cortisol levels are the highest24. We demonstrated that epistaxis risk varies throughout the day. Thus, the findings can raise the awareness of family caregivers and health-care professionals regarding the management and prevention of severe epistaxis episodes.
In the present study, strong positive correlations were found between elevated blood pressure overnight and the frequency of epistaxis. Our results suggest that the circadian rhythm of blood pressure may lead to changes in epistaxis rates by the time of day. The normal circadian rhythm of blood pressure can exert protective effects for the structure and function of heart, brain, kidney, and other organs. When the circadian rhythm diminishes or disappears, the risk of target organ damage significantly increases, especially in patients with hypertension. Generally, the sympathetic nervous system regulates blood pressure in the state of awakening, leading to the release of higher plasma norepinephrine, epinephrine, and haematuria catecholamine levels. Regulation by the sympathetic nervous system tension is reduced during sleep, and regulation by the vagal nervous system is dominant during sleep. Our study found that the high blood pressure at night and overnight was significantly related to epistaxis rates, whereas high blood pressure in the morning and afternoon was not related to epistaxis rates. However, blood pressure levels were not significantly different between patients with hypertension and those without hypertension. This finding indicates that, as suggested by Tan and Calhoun25, hypertension at the time of treatment of epistaxis may be anxiety-related, and blood pressure returns to normal after the control and management of epistaxis. By contrast, systolic blood pressure may be an independent risk factor for persistent epistaxis in adult patients with idiopathic epistaxis26, which is consistent with our research results. Although the association between blood pressure and epistaxis has been a longstanding debate, chronic vascular damage has been reported to be a possible pathophysiological mechanism through which blood pressure is associated with epistaxis onset. To the best of our knowledge, studies investigating the role of hypertension in the pathophysiological mechanism of epistaxis are scant. A study indicated that chronic vascular damage, such as the end-organ damage caused by high blood pressure, may be involved in the pathophysiology of epistaxis. Some studies have revealed that chronic hypertension may increase the incidence of epistaxis. The increased incidence of epistaxis is probably because chronic hypertension has well-known vasculopathic consequences, such atherosclerosis, endothelial dysfunction, and rupture27,28,29,30. Patients who have both epistaxis and a history of hypertension may incur more severe damage to the tunica media of the arteries in the nasal cavity.
Clinically, epistaxis is categorised as anterior or posterior depending on its anatomic origin. In 80–90% of patients with epistaxis, the anterior nasal septum is the site of epistaxis; epistaxis in the majority of patients is caused by epistaxis involving the Kiesselbach plexus31. The majority of anterior epistaxis cases are caused by epistaxis involving smaller vessels, which are easier to compress, suitable for electrocautery, and less difficult for the emergency medical personnel to treat32. Posterior epistaxis refers to bleeding that originates from posterior sites on the lateral nasal wall or nasal septum that are not apparent on anterior rhinoscopy. Posterior epistaxis is more common in older individuals and is more challenging to treat33. A case series demonstrated the presence of posterior epistaxis in 5% of patients with epistaxis treated in the emergency department or admitted to the hospital34. Our research also found a higher frequency of posterior epistaxis among individuals aged 65 years or older and among men. We found for the first time that posterior epistaxis risk was related to the time of day, and the incidence of posterior epistaxis was the highest overnight.
A standard definition of severe epistaxis is not available. Thus, in the present study, the severity of epistaxis was indirectly assessed based on the emergency department visits and management strategies used. Regarding the severity of epistaxis, this study demonstrated that patients in the overnight group were more likely to visit the emergency department for epistaxis and be managed by endoscopic electrocoagulation haemostasis immediately compared with the other groups (Table 1). The findings are consistent with the generally held belief that nasal epistaxis is more difficult to control in patients who arrive at the emergency department overnight. Doctors should be aware of this while managing nasal haemorrhage. In the present study, significant differences were observed between the time of epistaxis onset and management. The current study compared the results between 197 patients who underwent nasal packing and observation and 1014 patients who underwent electrocoagulation using bipolar forceps under general or local anaesthesia. Notably, monopolar forceps were used on epistaxis sites where haemostasis using bipolar forceps was difficult to perform. The incidence of rebleeding was significantly lower in the electrocoagulation group (8.5%, 18/1014) than in the packing and observation group (39.6%, 73/197; p = .001). All patients with rebleeding underwent endoscopic surgery, with the successful achievement of haemostasis. In addition, 203 (13.8%) patients were admitted to the hospital for inpatient endoscopic surgical intervention under general anaesthesia because of the inability to perform endoscopic electrocoagulation haemostasis due to severely deviated nasal septum or pain. Compared with those who underwent nasal packing, the management of epistaxis in the emergency department with endoscopic electrocoagulation haemostasis has many advantages, such as lower frequency of nasal packing, lower admission rates, less pain, shorter overall average hospital length of stay, and lower total cost34,35.
Strengths and limitations
This study is the first to investigate whether idiopathic epistaxis onset is associated with time of day. The study found that epistaxis occurred most frequently between 0:01 am and 6:00 am and in winter. The findings raise awareness among family caregivers and health-care professionals of the need to manage severe epistaxis. Additionally, we found that the frequency of posterior epistaxis was higher overnight, among individuals aged 66 years or older, and among men. Regarding why the incidence of epistaxis was highest overnight, we suggest that elevated blood pressure overnight may be a risk factor for epistaxis. These results indicate that adjusting the administration time of antihypertensive drugs may reduce the incidence of idiopathic epistaxis. In this study, we also indirectly assessed the severity of epistaxis based on the management strategies. Our findings confirm that endoscopic electrocoagulation haemostasis under local anaesthesia in the ENT emergency department can effectively control epistaxis and effectively reduce the nasal and re-bleed rates. This study also has some imitations. The first limitation is the retrospective nature of the study. We relied on potentially incomplete billing and diagnostic codes in our database. The data from the assessed hospital may not be representative of the entire Chinese population. In addition, our research focused on to adult patients, and young patients were not included in our study, which may limit our analysis by age groups. In addition, this study was a single-centre study. In the next step, the author will continue to collect more epistaxis cases and will strive to work with several hospitals to study the impact of different time points on the incidence rate of epistaxis. Moreover, the author will find more appropriate management strategies.
Conclusion
In this cohort, these results suggest that adult epistaxis occurred most frequently at overnight, especially in the winter, and more need management to immediately control the bleeding in ENT emergency departments. In addition, this study found that high frequent of epistaxis at overnight associated with serious blood pressure between midnight and 6 am. The results of this study are expected to provide guidance and help for the prevention and treatment of idiopathic epistaxis. Further prospective studies are required to elucidate the pathophysiology and the exact role played by time of day.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Villwock, J. A. & Jones, K. Recent trends in epistaxis management in the United States: 2008–2010. JAMA Otolaryngol. Head Neck Surg. 139 (12), 1279–1284. https://doi.org/10.1001/jamaoto.2013.5220 (2013).
Pallin, D. J. et al. Epidemiology of epistaxis in US emergency departments, 1992 to 2001. Ann. Emerg. Med. 46 (1), 77–81. https://doi.org/10.1016/j.annemergmed.2004.12.014 (2005).
Kasperek, Z. A. & Pollock, G. F. Epistaxis: An overview. Emerg. Med. Clin. North. Am. 31 (2), 443–454. https://doi.org/10.1016/j.emc.2013.01.008 (2013).
Sarhan, N. A. & Algamal, A. M. Relationship between epistaxis and hypertension: A cause and effect or coincidence? J. Saudi Heart Assoc. 27 (2), 79–84. https://doi.org/10.1016/j.jsha.2014.09.002 (2015).
Purkey, M. R., Seeskin, Z. & Chandra, R. Seasonal variation and predictors of epistaxis. Laryngoscope 124, 2028–2033. https://doi.org/10.1002/lary.24679 (2014).
Bray, D. et al. Epistaxis: Are temperature and seasonal variations true factors in incidence? J. Laryngol Otol. 119, 724–726. https://doi.org/10.1258/0022215054798032 (2005).
Comelli, I. et al. Influence of air temperature variations on frequency of epistaxis. Am. J. Rhinol Allergy. 29, 175–181. https://doi.org/10.2500/ajra.2015.29.4176 (2015).
Danielides, V., Kontogiannis, N. & Bartzokas, A. The influence of meteorological factors on the frequency of epistaxis. Clin. Otolaryngol. 27 (2), 84–88. https://doi.org/10.1046/j.1365-2273.2002.00535.x (2002).
Reddy, V. M., Judd, O. & Khalil, H. Investigation of the influence of ambient temperature, atmospheric pressure and water vapour pressure on epistaxis admission rate. Rhinology 48, 348–351. https://doi.org/10.4193/Rhino10.020 (2010).
Stopa, R. & Schonweiler, R. Causes of epistaxis in relation to season and weather status. HNO 37, 198–202 (1989). PMID: 2732102.
Walker, T. W., Macfarlane, T. V. & McGarry, G. W. The epidemiology and chronobiology of epistaxis: An investigation of Scottish hospital admissions 1995–2004. Clin. Otolaryngol. 32, 361–365. https://doi.org/10.1111/j.1749-4486.2007.01530.x (2007).
Pallin, D. J. et al. Epidemiology of epistaxis in US emergency departments, 1992 to 2001. Ann. Emerg. Med. 46, 77–81. https://doi.org/10.1016/j.annemergmed.2004.12.014 (2005).
Purkey, M. R., Seeskin, Z. & Chandra, R. Seasonal variation and predictors of epistaxis. Laryngoscope 124, 2028–2033. https://doi.org/10.1002/lary.24679 (2014).
Eccles, R. Nasal airflow in Health and Disease. Acta Otolaryngol. 120 (5), 580–595. https://doi.org/10.1080/000164800750000388 (2000).
McMullin, B. Atkinson P,Larivée N,et al. Examining seasonal variation in epistaxis in a maritime climate. J Otolaryngol Head Neck Surg 2019,48(1):74. https://doi.org/10.1186/s40463-019-0395-y
Unsal, A. A. et al. Doctor Google: Correlating internet search trends for epistaxis with metropolitan climates. Am. J. Otolaryngol. 40 (3), 358–363. https://doi.org/10.1016/j.amjoto.2019.02.001 (2019).
De Vries, K., Goei, J. T., Booy-Noord, H. & Orie, N. G. M. Changes during 24 hours in the lung function and histamine hyperreactivity of the bronchial tree in asthmatic and bronchitic patients. Int. Arch. Allergy. 20, 93–101. https://doi.org/10.1159/000229248 (1962).
Martin, R. J., Cicutto, L. C. & Ballard, R. D. Factors related to the nocturnal worsening of asthma. Am. Rev. Respir Dis. 141, 33–38. https://doi.org/10.1164/ajrccm/141.1.33 (1990).
Barnes, P., Fitzgerald, G., Brown, M. & Dolley, C. Nocturnal asthma and changes in circulating epinephrine, histamine, and cortisol. N Engl. J. Med. 303, 263–267. https://doi.org/10.1056/NEJM198007313030506 (1980).
Milavetz, G., Vaughan, L. M., Weinberger, M. M. & Hendeles, L. Evaluation of a scheme for establishing and maintaining dosage of theophylline in ambulatory patients with chronic asthma. J. Pediatr. 109, 351–354. https://doi.org/10.1016/s0022-3476(86)80403-6 (1986).
Raj, K. et al. Seasonal differences and circadian variation in stroke occurrence and stroke subtypes. J. Stroke Cerebrovasc. Dis. 24, 10–16. https://doi.org/10.1016/j.jstrokecerebrovasdis.2014.07.051 (2015).
Marler, J. R. et al. Morning increase in onset of ischemic stroke. Stroke 20, 473–476. https://doi.org/10.1161/01.str.20.4.473 (1989).
Fantoni, C., Dentali, F. & Ageno, W. Chronobiologic aspects of venous thromboembolism. Heart Fail. Clin. 13, 691–696. https://doi.org/10.1016/j.hfc.2017.05.005 (2017).
Mistry, P., Duong, A., Kirshenbaum, L. & Martino, T. A. Cardiac clocks and preclinical translation. Heart Fail. Clin. 13, 657–672. https://doi.org/10.1016/j.hfc.2017.05.002 (2017).
Tan, L. K., Calhoun, K. H. & Epistaxis Med. Clin. North. Am. 83, 43–56. https://doi.org/10.1016/s0025-7125(05)70086-9. (1999).
Terakura, M., Fujisaki, R., Suda, T., Sagawa, T. & Sakamoto, T. Relationship between blood pressure and persistent epistaxis at the emergency department: A retrospective study. J. Am. Soc. Hypertens. 6, 291–295. https://doi.org/10.1016/j.jash.2012.05.001 (2012).
Kikidis, D., Tsioufis, K., Papanikolaou, V., Zerva, K. & Hantzakos, A. Is epistaxis associated with arterial hypertension? A systematic review of the literature. Eur. Arch. Otorhinolaryngol. 271, 237–243. https://doi.org/10.1007/s00405-013-2450-z (2014).
Petruson, B., Rudin, R. & Svärdsudd, K. Is high blood pressure an aetiological factor in epistaxis? ORL J. Otorhinolaryngol. Relat. Spec. 39 (3), 155–160. https://doi.org/10.1159/000275350 (1977).
Knopfholz, J., Lima-Junior, E., Pre´coma-Neto, D. & Faria-Neto, J. R. Association between Epistaxis and hypertension: A one year follow-up after an index episode of nose bleeding in hypertensive patients. Int. J. Cardiol. 134, e107–e109. https://doi.org/10.1016/j.ijcard.2008.01.018 (2009).
Middleton, P. M. & Epistaxis Emerg. Med. Australas ;16, 428–440. https://doi.org/10.1111/j.1742-6723.2004.00646.x. (2004).
Schlosser, R. J. Clinical practice: Epistaxis. N Engl. J. Med. 360, 784–789. https://doi.org/10.1056/NEJMcp0807078 (2009).
Logan, J. K. & Pantle, H. Role of topical tranexamic acid in the management of idiopathic anterior epistaxis in adult patients in the emergency department. Am. J. Health Syst. Pharm. 73, 1755–1759. https://doi.org/10.2146/ajhp150829 (2016).
Cooper, S. E. & Ramakrishnan, V. R. Direct cauterization of the nasal septal artery for epistaxis. Laryngoscope 122, 738–740 (2012).
Viducich, R. A., Blanda, M. P. & Gerson, L. W. Posterior epistaxis: Clinical features and acute complications. Ann. Emerg. Med. 25, 592–596. https://doi.org/10.1002/lary.23225 (1995).
Henderson, A. H., Larkins, A. & Repanos, C. The use of bipolar electrocautery in adult epistaxis management: Using audit of one hundred and twenty-four cases to define a standardized protocol. Clin. Otolaryngol. 38, 554–558. https://doi.org/10.1111/coa.12191 (2013).
Funding
/Support: No Funding.
Additional Contributions: We are grateful to the participants and to the people involved in our study.
Author information
Authors and Affiliations
Contributions
Drs Yu had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.Concept and design: Drs Yu. Acquisition, analysis, or interpretation of data: All authors. Drafting of the manuscript: Drs Yu. Critical revision of the manuscript for important intellectual content: SY, Wang; ZH, Zhong. Statistical analysis: Drs Yu.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yu, J., Wang, S. & Zhong, Z. Frequency and severity of idiopathic epistaxis relative to time of day. Sci Rep 14, 29852 (2024). https://doi.org/10.1038/s41598-024-81570-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-81570-0