Abstract
This study retrospectively analyzed children admitted to the Fourth Affiliated Hospital of Guangxi Medical University for CO (carbon monoxide) poisoning from January 2018 to December 2022 and followed up on their neurological sequelae for a long time. The study was approved by the Ethics Committees of the Fourth Affiliated Hospital of Guangxi Medical University (the identification code was KY2023131) and informed consent was obtained from all participants and/or their legal guardians. The study complied with the Declaration of Helsinki. Through Global Deterioration Scale [GDS], we further compared the differences between children with and without cognitive impairment, and identified some risk factors for long-term cognitive impairment in children after CO poisoning. The GDS score of the patient was based on the follow-up score, and we only conducted one follow-up and recorded the GDS score throughout the entire study period. The follow-up time interval is defined as the time from the first discharge of the patient to our follow-up. A total of 113 children were encompassed in the study, with an average follow-up of 3.6 years (3.6 ± 1.5 years). Among them, 13 children (11.5%, 13/113) had cognitive abnormalities. The utilization of gas water heaters in enclosed bathrooms (101 cases, 89.4%) constituted the most frequent cause of CO poisoning among children in this study, followed by heating with fire (11 cases, 9.7%). Furthermore, one child was left by his father in a running car, thereby resulting in poisoning. The clinical manifestations of CO poisoning in children were mainly consciousness disorders (67 cases, 59.3%), dizziness or headache (37 cases, 32.7%), and other manifestations including irritability, crying, vomiting, limb weakness, and limb twitching, a total of 9 cases. The duration of consciousness disorders in children with cognitive abnormalities was mostly more than one day, with a median of 5 days, and the hospitalization time was longer. Children with cognitive abnormalities had higher C-reactive protein (CRP) levels, higher D-dimer levels, and higher liver enzyme levels. The most common imaging change after CO poisoning in children was cerebral edema, with two cases of subarachnoid hemorrhage observed and one case of demyelinating changes observed. For children with coma time less than one hour, there were few abnormal changes in cranial imaging. Children with cognitive abnormalities were more likely to develop epilepsy (38.5%, 5/13) and other system damage (53.8%, 7/13) during hospitalization, including pulmonary infection (3 cases), stressful gastrointestinal bleeding (2 cases), electrolyte imbalance (2 cases), dysfunction of liver, kidney or myocardial (3 cases), and some children had multiple system damage at the same time. There were statistical differences in the admission CO hemoglobin level, fibrinogen, D-dimer, high-sensitivity CRP, neuron enolase, alanine aminotransferase or aspartate aminotransferase (ALT or AST), lactate dehydrogenase, length of hospital stay, discharge and admission Glasgow Coma Scale (GCS), seizure frequency, duration of consciousness disorders more than one day, cranial imaging changes, use of ventilators, presence of other system damage, the number of hyperbaric oxygen (HBO) treatments, and whether the patients were transferred to another hospital between the two groups of children. Multivariate logistic regression analysis showed that head imaging changes and consciousness disorders lasting for more than a day were statistical differences. For children with unconsciousness lasting for more than one hour, it is advisable to contemplate conducting a head imaging examination as soon as possible within 3 days after CO exposure to guide the treatment during the acute phase.Characteristic alterations in cranial imaging and a longer duration of consciousness disorders (exceeding one day) might be correlated with subsequent neurological sequelae. For children with CO poisoning presenting these characteristics, active treatment can be implemented, encompassing but not restricted to HBO treatments, to minimize subsequent damage to the greater extent possible. So, for children who were unconscious for more than one day or presented characteristic changes in cranial imaging, long-term follow-up should be carried out to determine whether delayed encephalopathy or subsequent cognitive impairment occurs.
Similar content being viewed by others
Background
Carbon monoxide (CO) poisoning is a common gas poisoning in emergency rooms during winter. For CO poisoning in children, if long-term cognitive impairment occurs, it will have a detrimental effect on children’s neurodevelopment and long-term healthy growth.
Introduction
Through literature search, there are not many studies on long-term follow-up of children with CO poisoning, and researchers have paid insufficient attention to the cognitive impairment of children after CO poisoning. This study focuses on the cognitive impairment of children with CO poisoning, which can provide some theoretical basis for the treatment of children with cognitive impairment during the rehabilitation period after carbon CO poisoning.
CO poisoning is the most common gas poisoning in emergency rooms during winter. Although public health departments have made great efforts to promote the prevention of CO poisoning, emergency rooms still receive many patients with CO poisoning whenever the weather becomes cold. Epidemiological studies have shown that in China, infants (0–4 years old) and elderly people (70 years old and above) have a higher risk of DALYs (disability-adjusted life years), while young people (15–24 years old) have a higher risk of CO poisoning1. According to our retrospective study statistics, the most common causes of hospital admissions in our hospital were using gas water heater in a closed bathroom (89.4%) and employing charcoal fire for heating purposes (9.7%), which is also consistent with previous studies2. The most prevalent symptoms of mild poisoning are dizziness and headache, brief loss of consciousness (not exceeding 60 min), and the main symptom of severe poisoning is prolonged loss of consciousness and possible neurological sequelae. Although the global age-standardized mortality rate caused by unintentional CO poisoning decreased by nearly 50% during the 20-year period studied, the improvement in various regions was uneven3, so we still need to continue to work hard in preventing and treating carbon monoxide poisoning.
CO has an affinity for hemoglobin that is 210 times greater than that of oxygen, thereby impeding the delivery and utilization of oxygen. CO can induce cellular hypoxia, followed by oxidative stress and inflammation, and may give rise to neurological, cerebrovascular, or cardiovascular disorders, including encephalopathy, ischemia, and peripheral nerve injury. Through direct cellular damage and downstream signaling cascades, reactive oxygen species (ROS) produced during acute CO poisoning can lead to long-term neurological and cardiac sequelae. CO poisoning also causes adduct formation between myelin basic protein (MBP) and malonaldehyde, a reactive product of lipid peroxidation, resulting in an immunological cascade4, and this may lead to neurodegeneration. Currently, there are few studies focused on pediatric CO poisoning patients. We conducted a retrospective study with an average follow-up of 3.6 years to analyze pediatric patients with CO exposure in medical centers and analyzed the risk factors associated with long-term cognitive impairment after CO poisoning.
Method
Study design
We retrospectively analyzed the data of children hospitalized with CO poisoning in Liuzhou Workers’ Hospital from January 2018 to December 2022. The study was approved by the Ethics Committees of the Fourth Affiliated Hospital of Guangxi Medical University (the identification code was KY2023131) and informed consent was obtained from all participants and/or their legal guardians. The study complied with the Declaration of Helsinki. The privacy information of all patients was safeguarded, and we did not divulge any patient’s private information.
The inclusion criteria for this study were as follow: all hospitalized children (aged ≤ 18 years) diagnosed with acute CO poisoning between January 2018 and December 2022 based on medical history and COHb levels > 5%. Information regarding the first hospitalization of the children due to CO poisoning was collected. The exclusion criteria for this study were: (1) children who were still under 2 years old at the time of follow-up; (2) history of neurocognitive dysfunction before CO poisoning, including genetic diseases, neonatal hypoxic encephalopathy, etc.; (3) previous history of CO poisoning, and this poisoning was not the first occurrence of CO poisoning; (4) cardiac arrest before or during arrival at the emergency room; (5) lost to follow-up or uncooperative children or uncooperative family members during follow-up.
Based on the child’s medical history and a carboxyhemoglobin (COHb) level of > 5%, the diagnosis of acute CO poisoning was established. Some patients with CO poisoning had received 100% high-flow oxygen therapy via a mask prior to arriving at the emergency department, at which point the diagnosis of CO poisoning could be determined without relying on COHb levels. In addition, patients with any neurocognitive symptoms and signs, cardiovascular dysfunction, severe acidosis, or COHb levels of ≥ 25% can receive HBO therapy. If the child’s condition is unstable, it is regarded as perilous to employ HBO therapy, so only 100% high-flow oxygen therapy was administered.
Research methods
Through the hospitalization system, clinical information such as blood biochemical and blood gas testing information at the time of admission, length of hospitalization, source of CO poisoning, clinical manifestation, GCS scores at admission and discharge, cranial imaging data, and clinical treatment information such as whether hyperbaric oxygen therapy was utilized, the number of hyperbaric oxygen therapy sessions, and the time interval between the first hyperbaric oxygen therapy session and the time of poisoning were collected. The cognitive status of the child during follow-up was determined by councting follow-up with the child or the child’s family. Follow-up is carried out using the global deterioration scale (GDS)5, with the range of cognitive impairment ranging from none (GDS score, 1 point) to present (GDS score, 2–7 points), which is objectively confirmed through detailed interviews with experienced pediatric rehabilitation physicians. We merely conducted a one-time GDS follow-up when collecting cognitive information from the children at the end and utilized this GDS score for grouping and statistical analysis. According to the inclusion and exclusion criteria, based on the GDS scoring results, children are divided into normal group and cognitive abnormality group. The normal group has a GDS score of 1 point, indicating no cognitive impairment, with a total of 100 cases included; the GDS score ≥ 2 points for the cognitive abnormality group, with a total of 13 cases included. Compare the differences in various aspects of data between the two groups of children using statistical methods. (Table 1, 2 and 3)
Statistical analysis
The general characteristics, laboratory results(Table 1), and clinical manifestations(Tables 2 and 3) of the participants were presented as the mean ± standard deviation or median (interquartile range) for continuous variables and the frequency (percentage) for categorical variables. The two-sample independent Mann-Whitney U test was imployed for continuous variables, and the chi-square test or Fisher’s exact probability method was utilized for categorical variables to evaluate the differences between the two groups. The test for correlation between two variables employs Spearman rank correlation. After selecting a subset of variables that exhibited differences between Tables 2 and 3, binary logistic regression analysis was carried out to determine the association between cognitive impairment and clinical information related to CO poisoning in long-term follow-up (Table 4). The following is an explanation of regression analysis and its variable selection. Firstly, variables with differences were selected through univariate analysis, but due to incomplete blood test data, they were not included in the multivariate analysis. In addition to blood test data, select factors with statistical differences among the single factors and include them in the binary regression analysis. But we excluded some variables with overlapping clinical significance through clinical judgment. For example, the duration of consciousness disorders and the GCS scores of admissions and exit are variables that indicate the child’s consciousness status, so only the duration of consciousness disorders was selected for inclusion. Given the limited number of cases in this study, including an excessive number of variables might influence the regression analysis results. Consequently, some variables that presented differences in univariate analysis were ultimately excluded through the aforementioned method. The basic principle of the backward Wald test is to gradually eliminate insignificant variables until only significant variables are included in the model. This method is commonly used in regression analysis, especially when dealing with multicollinearity problems, which can help simplify the model and improve interpretability. All statistical analyses were performed using SPSS version 22, with a significant level set at 0.05.
Result
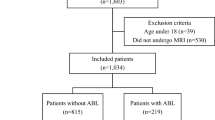
By retrieving the hospitalization system, from January 2018 to December 2022, a total of 515 patients were admitted to Liuzhou Workers’ Hospital for CO poisoning. Through age screening, a total of 179 cases were obtained in the pediatric group. In accordance with the inclusion and exclusion criteria, 41 cases were further eliminated. Among them, there were 34 cases under the age of two years old, and 7 cases of non-first-time CO poisoning. Finally, a total of 138 cases were included. Through follow-up, a total of 25 patients were lost to follow-up, and ultimately 113 patients fulfilled the study criteria. Based on the GDS classification of follow-up, 100 cases were classified into the normal cognitive group and 13 cases were classified into abnormal cognitive group.
In comparison with the normal group, the cognitive abnormality group had a longer hospitalization time, lower admission GCS scores, lower discharge GCS scores, more frequent seizures, longer periods of consciousness impairment, more common abnormal changes in cranial imaging, higher probability of using ventilators, more common occurrence of other system damage, and more children from lower-level hospitals were referred to the abnormal group. Simultaneously, children in the abnormal group also underwent more high-pressure oxygen therapy, including the number of high-pressure oxygen therapy for the first hospitalization and the total number of high-pressure oxygen therapy thereafter.
Laboratory data indicate that there are differences in fibrinogen, D-dimer, CRP, neuron enolase, ALT/AST, and lactate dehydrogenase, with higher levels in the abnormal group, while COHb levels are lower in the cognitive abnormality group than in the normal group. Multivariate logistic regression analysis indicated that hospitalization days, referral cases, head imaging alterations, and coma duration were statistically significant risk factors for children with neurological sequelae due to CO poisoning. The clinical data of the studied children are presented in Tables 1, 2, 3, 4 and 5 below.
Hospitalization days, coma duration, alterations in head imaging, other system complications were taken into consideration for inclusion in the logistic regression model in the multivariable analysis. A backward Wald selection process was employed. We found that alterations in head imaging and coma duration were statistically significant risk factors for CO poisoning children with long-term neurological sequelae.
The blood biochemical results in Table 1 were not performed in all patients, so the statistically significant blood biochemical results could not be compared between the two groups. For HBO therapy, our center tends to prefer more frequent HBO therapy when the clinical judgment of the child’s poisoning situation is more severe, so the variables related to HBO therapy were not included in the regression analysis. For alterations in cranial imaging, as some children did not undergo cranial imaging examinations, we divided this variable into two categories when conducting regression analysis: the first category showed abnormal findings in cranial imaging, and the second category showed no abnormal findings or no cranial imaging, because most children who did not undergo cranial imaging were clinically diagnosed with mild conditions and did not require cranial imaging examinations. Due to the limited number of cases in this study and the inclusion of more variables in the regression analysis compared to theory, the results of the regression analysis have certain limitations.

Comparison of the duration of consciousness disorders and changes in cranial imaging between two groups of pediatric patients.
Discussion
CO poisoning progresses more severely in children presenting neurological symptoms, increased red blood cell distribution width, and mean platelet volume. The study also indicates that satisfactory results have been achieved even in severe cases of CO poisoning through early and appropriate treatment6. Most of the children in this study did not show cognitive abnormalities during subsequent follow-up, which is consistent with the results of this study. A study encompassing 81 children with CO poisoning indicated that the most common initial symptoms of CO poisoning in children were vomiting and alterations in consciousness. Multivariate analysis demonstrated that treatment in intensive care units and the utilization of ventilators due to prolonged loss of consciousness might be associated with delayed neurological sequelae in children2. The results of this study suggest that the most prevalent initial symptom in pediatric patients is also consciousness disorders. The final regression analysis also suggests that consciousness disorders persisting more than one day may be a related factor for cognitive impairment in the later stages of the disease, which is analogous to the above research results. A case report describes a 4-month-old infant who developed delayed neurological sequelae after CO poisoning. She presented with neurological symptoms, this child presents with neurological symptoms, including opisthotonus, athetoid movements, anterior tongue thrust, and opsoclonus. These symptoms include kyphosis, hand and foot movement, anterior tongue push, and eye spasms7. Owing to the retrospective nature of this study, cognitive information of the children was gathered through a questionnaire survey, and no neurological physical examination was performed on the children. Consequently, other neurological signs were not taken into account , which is also one of the limitations of this study.
COHb level
COHb level is a reliable laboratory indicator for diagnosing acute CO poisoning. In previous studies, COHb as a biochemical indicator had a relatively weak correlation with the severity or prognosis of patient poisoning8,9,10. The clinical detection of COHb level is affected by various factors10,11. Under indoor air conditions, the half-life of COHb is 320 min12, while under 100% normal pressure oxygen conditions13, the half-life is shortened to approximately 74 min. Therefore, using COHb alone to evaluate the prognosis of CO poisoning patients has certain limitations. In this study, the median COHb level was 8.8%. The COHb level in the cognitive abnormality group was lower than that in the normal group overall. Clinically, COHb level is often used to classify CO poisoning into mild, moderate, and severe categories. However, considering that most children with cognitive impairment come from lower-level hospitals for referral, the COHb level might have significantly decreased by the time they arrive at our hospital. This also suggests that COHb level alone cannot be used to judge the severity of poisoning. A forensic study on low COHb levels of CO poisoning in Shanghai, China showed that acute CO poisoning with low COHb (less than 30%) still resulted in patient death, accounting for 18.9% (58/307) of the total cases studied14. A retrospective study conducted by the Portuguese National Institute of Legal Medicine and Forensic Science showed that there was one case of blood and internal organs with cherry red color, but the detected COHb level was only 3%15. Therefore, some studies have suggested using total blood CO (TBCO) as a surrogate biomarker for COHb11. Because for TBCO, its concentration is relatively stable during observation regardless of temperature, time, and tube headspace volume parameters. While for COHb, its concentration changes significantly during storage.
The severity of CO poisoning
Previous studies have shown that Poisoning Severity Score is a good indicator for predicting the prognosis of CO poisoning16,17, but they mainly predict short-term adverse outcomes. Therefore, there are limitations in predicting the long-term outcome of patients. A retrospective study of 331 children showed that GCS, white blood cell count, and troponin T level are factors associated with the severity of CO poisoning in children, which may help predict the clinical outcome of children with CO poisoning18. This study defined severe poisoning as the utilization of positive inotropic drugs, mechanical ventilation therapy, or multiorgan failure, but did not conduct a follow up on the long-term prognosis of children. It only analyzed factors that may be related to severe poisoning. A study on CO poisoning in patients aged 16 or above demonstrated that the main endpoint outcome was neurocognitive sequelae in the subsequent 4 weeks, with GDS scores ranging from good (1–3 points) to poor (4–7 points)19. The results showed that age greater than 50 years (1 point), GCS score less than or equal to 12 points (1 point), shock (1 point), serum creatine kinase level greater than 320 U/L at emergency department visit (1 point), and no use of HBO therapy (1 point) were still significantly associated with poor outcomes, and the scoring system was named COGAS. The area under the receiver operating characteristic curve for the COGAS score was 0.862 (95% CI, 0.828–0.895) for the derivation cohort and 0.870 (95% CI, 0.779–0.961) for the validation cohort. This study had a longer follow-up period (3.6 ± 1.5 years), and divided the patients into cognitive impairment group (11.5%, 13/113) and normal group (88.5%, 100/113) based on long-term GDS scores. Based on the data from this study, consciousness changes persisting for more than one hour, combined with damage to other systems, and characteristic alterations in cranial imaging seem to imply more severe CO poisoning and a poorer prognosis. The GDS scores of the 13 cases in the cognitive impairment group are shown in the table below (Table 5).
About the sequelae of nervous system
A retrospective study from Taiwan on children showed that acute seizures, severe metabolic acidosis, significantly low blood pressure, prolonged unconsciousness, and longer hospitalization time were associated with delayed neurological sequelae (DNS)20. DNS refers to the symptoms of varying degrees of cognitive impairment, personality changes, motor disorders, and focal nervous system defects that occur in patients with acute CO poisoning after a period of false recovery after significant recovery from poisoning. It generally occurs between 2 and 60 days after acute CO poisoning. Typically, if the duration of this sequelae exceeds three months, it might develop into PNS(permanent neurological sequelae or long-term neurological sequelae). A total of 30 children were included in the study5, and only 5 (16.7%) children developed DNS, but all recovered completely within 2 months. Owing to the limited number of cases, no further analysis of risk factors was carried out. Another study regarding DNS or PNS in children after CO poisoning indicated that treatment in the intensive care unit due to prolonged loss of consciousness was the only independent risk factor for patients with DNS2. The use of ventilators was the only independent risk factor for patients with PNS. A total of 81 children were included in the study, and patients with PNS were followed up for more than one year, similar to our follow-up duration2. Although the study concluded that prolonged loss of consciousness was the only independent risk factor for the occurrence of DNS, it did not analyze the impact of specific duration of consciousness loss on the long-term neurological prognosis of children. A systematic review included 2328 patients21, and the results showed that low initial GCS scores in patients with CO poisoning were associated with the occurrence of delayed neurological sequelae, and the incidence of delayed neurological sequelae in GCS < 9 group was significantly higher than that in the control group (OR 2.80, 95% CI 1.91–4.12, I²= 34%). This study demonstrated through univariate analysis that children with consciousness disorders for more than 1 day were associated with long-term neurological sequelae. Moreover, multivariate analysis revealed that duration of consciousness disorders was an independent risk factor for long-term neurological sequelae. Most of the coma duration in cognitive impairment group was more than 1 day, with a median of 5 days (120 h). Among the 13 children with long-term neurological sequelae, only 2 were treated with ventilators during hospitalization, and one of them had a GCS score of only 3 points upon discharge. However, we failed to identify a correlation between the utilization of ventilators and PNS, which might be attributed to an insufficient number of cases.
Explanation of inspection indicators
Although some test results between the two groups presented statistical differences in univariate analysis, due to the absence of some test results, the test results were not incorporated into the regression analysis, which constitutes one of the limitations of this study. However, through univariate analysis of some test results, it was shown that children with cognitive impairment had higher levels of CRP, higher liver enzyme indicators, higher neuron-specific enolase (NSE) levels and higher D-dimer and fibrinogen indicators. A previous study reported that the levels of NSE and S100B proteins increased after traumatic brain injury in children22. A study in children showed that the level of NSE increased in children with hypoxic brain injury related to CO poisoning23, which might indicate that NSE might be a meaningful indicator for detecting cerebral ischemia and anoxia injury after CO poisoning. In a study of adults (aged 53.48 ± 19.29 years), the incidence of DNS was 29.2% (84/288)24. This study indicated that neutrophil-to-lymphocyte ratio was an significant independent predictor of DNS in CO poisoning patients, which also reflected from the side that CO poisoning patients with higher inflammatory indicators may be more prone to subsequent neurological damage.
CO has a high affinity for hemoglobin and can cause varying degrees of tissue hypoxia when poisoning occurs. Owing to the restricted availability of oxygen in tissues, the functions of the heart, liver, nervous system, and other tissues could be negatively affected, and these effects might manifest as abnormalities in hematological tests, including but not limited to liver function abnormalities, myocardial enzyme abnormalities, coagulation function abnormalities, and so forth. CO induces the production of reactive oxygen species through three different mechanisms, leading to lipid peroxidation and reduced glutathione. The utilization of antioxidants or oxidase inhibitors can protect neurons and astrocytes from CO-induced oxidative stress and cell death25. These reactive oxygen species might cause damage to various tissues in the human body, which may manifest as abnormal hematological markers in different organs.
CO poisoning can induce reversible or irreversible myocardial injury through the direct toxic effects of myocardial hypoxia and CO. CO binds to myoglobin and decreases the availability of oxygen, which can cause arrhythmia and cardiac dysfunction26. A study on the effects of acute CO poisoning on the heart in children showed that, acute CO intoxication temporarily increases troponin I and creatine kinase isoenzyme levels, and cardiac enzyme levels are correlated with echocardiographic and electrocardiographic abnormalities. Further studies are required to evaluate the importance of these new electrocardiographic and echocar diographic parameters in children with CO poisoning27. This might be one of the reasons for abnormal myocardial enzymes caused by CO poisoning. A study on the relationship between adverse neurological outcomes after CO poisoning and the neutrophil lymphocyte ratio at admission suggests that an increase in the neutrophil lymphocyte ratio at admission is an important and independent predictor of poor neurological outcomes in CO poisoning patients24. Furthermore, this study also showed that patients with delayed neurological sequelae or elevated neutrophil lymphocyte ratio after CO poisoning had a higher incidence of acute kidney injury during hospitalization. Systemic ischemia-hypoxia injury, direct cellular injury at the level of the kidney, and inflammation can explain the pathogenesis of CO poisoning-related nephrotoxicity28.
Significance of cranial imaging
Alterations in cranial imaging are another characteristic of CO poisoning induced hypoxic ischemic encephalopathy, and such alterations might have certain value in predicting CO poisoning related delayed encephalopathy. Magnetic resonance imaging (MRI) can be used to sensitively identify CO poisoning related cytotoxic edema within 72 h after CO exposure29. The globus pallidus is the most common location of abnormal MRI signals in patients with acute CO poisoning, with abnormal signals observed in 19.9% of CO poisoning patients. Most lesions were located in the cortex, hippocampus white matter, and basal ganglia (including the globus pallidus), while lesions in the brain stem and thalamus are rare30. Among the 13 children with cognitive impairment in this study, 8 (61.5%, 8/13) had alterations in cranial imaging, while 4 (4%, 4/100) had alterations in cranial imaging in the normal group. Univariate analysis showed that there was a statistically significant difference in cranial imaging alterations between the two groups (P < 0.05). The most common imaging alteration is brain edema, with SAH observed in 2 cases (this might be related to the child falling after experiencing consciousness disorders) and demyelinating changes observed in one case. Among the 13 cases, no abnormal alterations were detected in the cranial imaging of children with coma time less than one hour. In the normal group, there were 4 cases presenting abnormal cranial imaging, and the durations of coma were ranging from 120 h, 20 h, 3 h, to 0.33 h. These data might imply a positive correlation between coma time exceeding one hour and brain hypoxic changes; that is, brain injury is more likely to be observed on imaging, which is closely related to patient prognosis. Among the imaging techniques for detecting acute ischemic changes in the brain, MRI has higher sensitivity than CT in identifying lesions31. Therefore, for children with coma time exceeding one-hour, cranial imaging should be considered, preferably cranial MRI. The examination time should be within 72 hours after CO exposure32, to determine whether there are changes of brain injury, which has certain guiding significance for the subsequent treatment of children.
The tissues of children afflicted with CO poisoning will encounter varying degrees of hypoxia, which is dictated by the high affinity between CO and hemoglobin. The imaging manifestations of cerebral hypoxia include high DWI signal intensity, shallow sulci and gyrus, cerebral edema signal intensity, and low density of brain parenchyma. These hypoxic processes might exert adverse effects on children’s development, behavior, and academic performance33. The abnormal imaging findings discovered in this study mainly include low-density shadows of brain parenchyma, brain swelling, and specific imaging changes of CO poisoning (symmetrical abnormal signal shadows of bilateral globus pallidus), which also belong to the scope of cerebral hypoxia. A resting state functional MRI study on children with CO poisoning showed that, compared to healthy children, the functional network small-world structure of the children with CO poisoning was damaged, and the brain’s ability to integrate information was reduced34. Furthermore, the results of the brain network nodes and edge attributes of CO poisoning patients were mutually verified, and the relationship between the nodes with altered characteristics and the neurocognitive performance of the patients was determined.
Hyperbaric oxygen therapy
Hyperbaric oxygen therapy (HBOT) can accelerate the clearance of CO in the body by increasing oxygen partial pressure and ventilation, and is a recognized treatment method for acute CO poisoning, with many research results and theoretical support35. In terms of drugs, drugs that can alleviate downstream pathophysiological damage caused by acute CO poisoning include steroids, anti-inflammatory drugs, and mitochondrial electron transfer chain substrates36. Extremely limited research exists regarding the relationship between the utilization of HBO and prognosis in children with CO poisoning. An earlier study showed that HBOT should be carried out within 6 h after poisoning, which is related to the significant reduction of DNS incidence rate37. A study from Taiwan suggests that the acceptable HBOT time is within 22.5 h after CO poisoning, if HBOT is performed more than 48 h after CO poisoning, it is not beneficial for preventing DNS38. The overall trend of evidence is that for patients who have been assessed to require HBOT, implementing HBOT as soon as possible under possible conditions might have greater benefits. Our center launched HBOT earlier, with 86.7% (98/113) of all enrolled children receiving HBOT. The median number of HBOT hospitalizations for the first time was 8, and most of them could start HBOT within one day after poisoning. For severely poisoned children, we will urgently arrange HBOT to expel CO from the body as soon as possible. Continue HBOT after the acute phase to exert its potential neuroprotective effect, as studies have shown that high affinity hemoglobin for CO may continue to slowly release CO within hours to days after COHb returns to normal levels, continuing to cause damage to the nervous system39. However, there is no good evidence to support the number of HBOT times that should be implemented to prevent long-term neurological sequelae, and clinical judgment can only be based on actual conditions or experience.
In our study, children who did not receive HBOT had a shorter length of hospitalization and mild symptoms. Through Spearman rank correlation analysis, the duration of consciousness impairment (r = 0.012, p = 0.237), length of hospitalization(r = 0.453, p<0.001), and presence of other systemic complications(r = 0.219, p = 0.020) were positively correlated with the number of HBOT times during the first hospitalization. Univariate analysis further indicated a statistically significant difference in the number of HBOT sessions between the two groups of children. The median number of sessions was 12 for children in the cognitive abnormality group during their first hospitalization, in contrast to 5 for children in the normal group. This also implies that our center is inclined to conduce multiple HBOT session for children with more severe conditions.
The opportunities for HBOT are restricted after CO poisoning, and other forms of treatment are also necessary. For children presenting with long-term consciousness disorders (more than one day) and abnormal findings in cranial imaging subsequent to CO poisoning, greater attention and a longer treatment course might be requisite. These treatments might impose an additional burden on the families of affected children. Hence, healthcare policies ought to pay appropriate due attention to this category of child and, if necessary, augment medical insurance expenses to alleviate the burden on the families of affected children.
Conclusion
For children with unconsciousness lasting for more than one hour, it is advisable to contemplate conducting a head imaging examination as soon as possible within 3 days after CO exposure to guide the treatment during the acute phase. A longer hospitalization duration, characteristic alterations in cranial imaging, referral cases, and a longer duration of consciousness disorders (exceeding one day) might be correlated with subsequent neurological sequelae. For children with CO poisoning presenting these characteristics, active treatment can be implemented, encompassing but not restricted to HBOT, to minimize long-term damage to the greater extent possible.
Therefore, for children who were unconscious for more than one day or presented characteristic changes in cranial imaging, long-term follow-up should be carried out to determine whether delayed encephalopathy or subsequent cognitive impairment occurs.
Limitations
This study has certain limitations. We focused primarily on assessing the cognitive and learning abilities of the pediatric patients and did not follow up on the impact of CO poisoning on other systems. Firstly, the age range of the included patients was quite broad, and children’s learning abilities and cognitive levels are constantly developing. Therefore, there are significant differences in these abilities among children of different age groups. To facilitate data analysis and statistical analysis, we uniformly adopted the GDS grading system, which may not provide an accurate assessment of the patients’ cognitive and learning abilities. During the follow-up assessment of GDS scores, we primarily compared the patients with their peers of the same age to ensure the detection of any differences in cognitive and learning abilities compared to most children of the same age. Secondly, the impairment of cognitive and learning abilities in these patients was mostly mild, making it difficult to strictly distinguish between different severity levels. Therefore, this study did not further conduct subgroup analysis and instead included all patients with cognitive abnormalities into a unified group. There are certain limitations to the single follow-up GDS assessment of cognitive function in children in this study, and more comprehensive cognitive assessment methods may be needed for further research in the future. Additionally, since this study was retrospective, some patients’ blood biochemical tests were not comprehensive. We only conducted univariate analysis to identify some significant test items and did not include them in the multivariate binary regression analysis. This is another aspect where the study could be improved. Future considerations could include conducting a multicenter prospective study to more comprehensively analyze relevant risk factors. The sample size of the cognitive impairment group in this study is small, therefore larger multicenter studies are needed to further confirm the research results. More prospective studies or clinical trials are needed in the future to validate the potential of these findings.
Data availability
The datasets generated and/or analyzed during the current study are not publicly available due to protect patient privacy information, but are available from the corresponding author on reasonable request.
Change history
08 May 2025
A Correction to this paper has been published: https://doi.org/10.1038/s41598-025-99830-y
References
Cui, P. et al. Burden of carbon monoxide poisoning in China, 1990–2019: A systematic analysis of data from the global burden of disease study 2019. Front. Public. Health. 10, 930784. https://doi.org/10.3389/fpubh.2022.930784 (2022).
Chang, Y. C. et al. Risk factors and outcome analysis in children with carbon monoxide poisoning. Pediatr. Neonatol. 58, 171–177. https://doi.org/10.1016/j.pedneo.2016.03.007 (2017).
Global National mortality due to unintentional carbon monoxide poisoning, 2000–2021: Results from the global burden of Disease Study 2021. Lancet Public. Health. 8, e839–e849. https://doi.org/10.1016/s2468-2667(23)00185-8 (2023).
Thom, S. R., Bhopale, V. M., Fisher, D., Zhang, J. & Gimotty, P. Delayed neuropathology after carbon monoxide poisoning is immune-mediated. Proc. Natl. Acad. Sci. U.S.A. 101, 13660–13665. https://doi.org/10.1073/pnas.0405642101 (2004).
Reisberg, B., Ferris, S. H., de Leon, M. J. & Crook, T. Global deterioration scale (GDS). Psychopharmacol. Bull. 24, 661–663 (1988).
Güven, D. & Sarıcı, D. Clinical and laboratory characteristics Predicting the Severity of Carbon Monoxide Poisoning in children: A single-Center Retrospective Study. Pediatr. Emerg. Care. 39, 207–215. https://doi.org/10.1097/pec.0000000000002927 (2023).
Lai, Y. H., Chen, L. W. & Hsueh, Y. Y. Delayed neurological sequelae in an infant after Carbon Monoxide Intoxication. J. burn care Research: Official Publication Am. Burn Association. 42, 1035–1037. https://doi.org/10.1093/jbcr/irab074 (2021).
Ku, H. L., Yang, K. C., Lee, Y. C., Lee, M. B. & Chou, Y. H. Predictors of carbon monoxide poisoning-induced delayed neuropsychological sequelae. Gen. Hosp. Psychiatry. 32, 310–314. https://doi.org/10.1016/j.genhosppsych.2009.11.005 (2010).
Weaver, L. K., Valentine, K. J. & Hopkins, R. O. Carbon monoxide poisoning: Risk factors for cognitive sequelae and the role of hyperbaric oxygen. Am. J. Respir. Crit Care Med. 176, 491–497. https://doi.org/10.1164/rccm.200701-026OC (2007).
Hampson, N. B. & Hauff, N. M. Carboxyhemoglobin levels in carbon monoxide poisoning: Do they correlate with the clinical picture? Am. J. Emerg. Med. 26, 665–669. https://doi.org/10.1016/j.ajem.2007.10.005 (2008).
Oliverio, S. & Varlet, V. New strategy for carbon monoxide poisoning diagnosis: Carboxyhemoglobin (COHb) vs total blood Carbon Monoxide (TBCO). Forensic Sci. Int. 306, 110063. https://doi.org/10.1016/j.forsciint.2019.110063 (2020).
Peterson, J. E. & Stewart, R. D. Absorption and elimination of carbon monoxide by inactive young men. Arch. Environ. Health. 21, 165–171. https://doi.org/10.1080/00039896.1970.10667215 (1970).
Weaver, L. K., Howe, S., Hopkins, R. & Chan, K. J. Carboxyhemoglobin half-life in carbon monoxide-poisoned patients treated with 100% oxygen at atmospheric pressure. Chest 117, 801–808. https://doi.org/10.1378/chest.117.3.801 (2000).
Liu, Z. et al. Acute carbon monoxide poisoning with low saturation of carboxyhaemoglobin: A forensic retrospective study in Shanghai, China. Sci. Rep. 11, 18554. https://doi.org/10.1038/s41598-021-97436-8 (2021).
Ruas, F., Mendonça, M. C., Real, F. C., Vieira, D. N. & Teixeira, H. M. Carbon monoxide poisoning as a cause of death and differential diagnosis in the forensic practice: A retrospective study, 2000–2010. J. Forensic Leg. Med. 24, 1–6. https://doi.org/10.1016/j.jflm.2014.02.002 (2014).
Wang, I. J. et al. Poison severity score and sequential organ failure assessment score: Carbon monoxide poisoning prognosis. PloS One. 14, e0212025. https://doi.org/10.1371/journal.pone.0212025 (2019).
Cevik, A. A., Unluoglu, I., Yanturali, S., Kalkan, S. & Sahin, A. Interrelation between the poisoning severity score, carboxyhaemoglobin levels and in-hospital clinical course of carbon monoxide poisoning. Int. J. Clin. Pract. 60, 1558–1564. https://doi.org/10.1111/j.1742-1241.2006.00962.x (2006).
Akcan Yildiz, L., Gultekingil, A., Kesici, S., Bayrakci, B. & Teksam, O. Predictors of severe clinical course in children with carbon monoxide poisoning. Pediatr. Emerg. Care. 37, 308–311. https://doi.org/10.1097/pec.0000000000001580 (2021).
Kim, S. H. et al. Derivation and validation of a score for predicting poor neurocognitive outcomes in acute carbon monoxide poisoning. JAMA Netw. open. 5, e2210552. https://doi.org/10.1001/jamanetworkopen.2022.10552 (2022).
Cho, C. H., Chiu, N. C., Ho, C. S. & Peng, C. C. Carbon monoxide poisoning in children. Pediatr. Neonatol. 49, 121–125. https://doi.org/10.1016/s1875-9572(08)60026-1 (2008).
Namgung, M. et al. Association between Glasgow Coma scale in early carbon monoxide poisoning and development of delayed neurological sequelae: A Meta-analysis. J. Personalized Med. 12 https://doi.org/10.3390/jpm12040635 (2022).
Berger, R. P. et al. Neuron-specific enolase and S100B in cerebrospinal fluid after severe traumatic brain injury in infants and children. Pediatrics 109, E31. https://doi.org/10.1542/peds.109.2.e31 (2002).
Eyi, Y. E. et al. Is S100B protein level really not an indicator of brain damage due to carbon monoxide poisoning in children? Am. J. Emerg. Med. 31, 1531. https://doi.org/10.1016/j.ajem.2013.07.015 (2013).
Xu, D., Mei, T. & He, F. The neutrophil-to-lymphocyte ratio is associated with the frequency of delayed neurologic sequelae in patients with carbon monoxide poisoning. Sci. Rep. 13, 19706. https://doi.org/10.1038/s41598-023-47214-5 (2023).
Angelova, P. R., Myers, I. & Abramov, A. Y. Carbon monoxide neurotoxicity is triggered by oxidative stress induced by ROS production from three distinct cellular sources. Redox Biol. 60, 102598. https://doi.org/10.1016/j.redox.2022.102598 (2023).
Aslan, S. et al. The evaluation of myocardial damage in 83 young adults with carbon monoxide poisoning in the East Anatolia region in Turkey. Hum. Exp. Toxicol. 25, 439–446. https://doi.org/10.1191/0960327106het645oa (2006).
Ozyurt, A. et al. Effects of acute carbon monoxide poisoning on ECG and echocardiographic parameters in children. Cardiovasc. Toxicol. 17, 326–334. https://doi.org/10.1007/s12012-016-9389-4 (2017).
Chen, Y. C. et al. Acute kidney injury predicts mortality after charcoal burning suicide. Sci. Rep. 6, 29656. https://doi.org/10.1038/srep29656 (2016).
Kim, Y. S. et al. The usefulness of diffusion-weighted magnetic resonance imaging performed in the acute phase as an early predictor of delayed neuropsychiatric sequelae in acute carbon monoxide poisoning. Hum. Exp. Toxicol. 37, 587–595. https://doi.org/10.1177/0960327117722821 (2018).
Jeon, S. B. et al. Acute brain lesions on magnetic resonance imaging and delayed neurological sequelae in carbon monoxide poisoning. JAMA Neurol. 75, 436–443. https://doi.org/10.1001/jamaneurol.2017.4618 (2018).
Lansberg, M. G., Albers, G. W., Beaulieu, C. & Marks, M. P. Comparison of diffusion-weighted MRI and CT in acute stroke. Neurology 54, 1557–1561. https://doi.org/10.1212/wnl.54.8.1557 (2000).
Ahn, C. et al. Early neuroimaging and delayed neurological sequelae in carbon monoxide poisoning: A systematic review and meta-analysis. Sci. Rep. 12, 3529. https://doi.org/10.1038/s41598-022-07191-7 (2022).
Bass, J. L. et al. The effect of chronic or intermittent hypoxia on cognition in childhood: A review of the evidence. Pediatrics 114, 805–816. https://doi.org/10.1542/peds.2004-0227 (2004).
Liu, H. et al. A graph theory study of resting-state functional MRI connectivity in children with carbon monoxide poisoning. J. Magn. Reson. Imaging: JMRI. 58, 1452–1459. https://doi.org/10.1002/jmri.28706 (2023).
Sethuraman, K. & Thom, S. R. Hyperbaric oxygen should be used for carbon monoxide poisoning. Br. J. Clin. Pharmacol. 89, 939–941. https://doi.org/10.1111/bcp.15605 (2023).
Dent, M. R., Rose, J. J., Tejero, J. & Gladwin, M. T. Carbon Monoxide Poisoning: from microbes to therapeutics. Annu. Rev. Med. 75, 337–351. https://doi.org/10.1146/annurev-med-052422-020045 (2024).
Thom, S. R. et al. Delayed neuropsychologic sequelae after carbon monoxide poisoning: prevention by treatment with hyperbaric oxygen. Ann. Emerg. Med. 25, 474–480. https://doi.org/10.1016/s0196-0644(95)70261-x (1995).
Liao, S. C. et al. Targeting optimal time for hyperbaric oxygen therapy following carbon monoxide poisoning for prevention of delayed neuropsychiatric sequelae: a retrospective study. J. Neurol. Sci. 396, 187–192. https://doi.org/10.1016/j.jns.2018.11.025 (2019).
Cronje, F. J., Carraway, M. S., Freiberger, J. J., Suliman, H. B. & Piantadosi, C. A. Carbon monoxide actuates O(2)-limited heme degradation in the rat brain. Free Radic. Biol. Med. 37, 1802–1812. https://doi.org/10.1016/j.freeradbiomed.2004.08.022 (2004).
Funding
Self-funded scientific research project of the health and Health Commission of the Guangxi Autonomous Region, Grant ID: Z-B20231454.
Author information
Authors and Affiliations
Contributions
TY.W. and JH.L. designed the study and performed the data analysis. YL.W, WJ.L and TY.W. performed the medical record system data collection and follow-up. TY.W wrote the main part of the paper. TY.W. and JH.L performed the database search and drafted the manuscript. LY.P. is responsible for the planning, implementation, and proposal of revision suggestions for the entire research, and has the responsibility of supervising and coordinating the overall research. All authors have read, revised, and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: In the original version of this Article Tuoying Wen and Jinghong Liang were omitted as equally contributing authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wen, T., Liang, J., Wei, Y. et al. Analysis of prognosis of neurological sequelae in children with carbon monoxide poisoning. Sci Rep 14, 29972 (2024). https://doi.org/10.1038/s41598-024-81634-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-81634-1