Abstract
The rapid development in agriculture and industrial sectors has raised some serious global issues like heavy metals pollution of water resources. Cadmium (Cd) is amongst the major water pollutants worldwide. In this study, two novel sorbents were prepared by using post-consumer textile waste (PCTW). The waste denim fabric was recycled with environmentally friendly H2O2 and Ozone through oxidation under alkaline conditions to produce several functional groups at the surface (named as ODF@H202 and ODF@03 respectively) that could trap the heavy metal cations from contaminated water. The functionalized fabric sorbents were characterized by Fourier transform infrared spectroscopy (FT-IR), X-Ray diffraction (XRD), scanning electron microscopy (SEM), and energy dispersive X-Ray (EDX) that revealed the presence of carboxylic acid, hydroxyl, and amine functional groups on their surfaces. The sorption isotherm, sorption kinetics and sorption thermodynamics were carried out to unravel the sorption process mechanism. The ODF@H202 sorbent was proved more effective by giving maximum adsorption capacity of (238.09 mg g−1) compared to Ozone treatment (175.44 mg g−1) for Cd2+ and achieved within just 20 min for both sorbents. Pseudo 2nd order and Langmuir models confirmed the chemosorption as dominant mechanism for the monolayer sorption of Cd2+ ions onto ODF@H202 and ODF@03. The sorption thermodynamic revealed the sorption process as endothermic and spontaneous in nature. The results showed that both ODF@H202 and ODF@03 sorbents have an efficient potential for sorbing Cd from contaminated water. Furthermore, both ODF@H202 and ODF@03 sorbents were also tested in a regeneration study to investigate the reuse of these sorbents, and we achieved marvelous results. Both sorbents gave up to 90% of the sorption capacity even after 10 recycles. Conclusively, both sorbents can have their implications for the preparation of filters that can be used for the treatment of wastewater. This study has practical significance by tackling the two environmental problems i.e. heavy metal pollution and denim waste.
Similar content being viewed by others
Introduction
Though this is the era of growing technology, still the concentration of heavy metals in water is not within the safe range in many countries as suggested by regulatory authorities1. Mostly, the water is contaminated with various heavy metals like cadmium, arsenic, mercury, lead, and chromium that are hazardous to human health2. These heavy metals pollution of water and soil has become the hotspot of recent studies for environmental researchers due to their non-biodegradable, bioavailable, persistent, and more toxic nature3. Due to increased consumption of these heavy metals through drinking of contaminated water, diseases like diabetes, neuronal damage, cardiovascular disorders, renal injuries, and even cancer have been diagnosed3,4. Cd has been listed as the fifth harmful toxicant for human beings which causes diseases like osteoporosis, hypertension, diabetic renal dysfunction/ complications, pancreas, bladder, kidney, lung and breast cancer5,6. The main reason behind the heavy metal toxicity is the generation of reactive oxygen species that are responsible for oxidative damage and health associated disastrous effects7. The mortality rate is also increasing due to excessive utilization of various heavy metals8.
Another issue of same importance is the post-consumer textile waste (PCTW) due to fast fashion. It is reported that 27 million tons of cotton are being produced per year globally and almost the same number of textiles in the form of PCWT are disposed of every year. About 5% of all worldwide landfills are being taken up by dumping of textile waste which is creating serious environmental and health hazards due to open air dumping9,10. The degradation of textile waste particularly cotton generates greenhouse methane (CH4) gas and carbon dioxide (CO2) which are responsible for climate change while, toxic chemicals and dyes have been leached out into soil and groundwater causing soil and freshwater pollution.
Recently, every organization has been trying to achieve sustainability by recycling waste and converting it into useful products that can have minimum environmental impacts11. The denim, which is generally made of cotton, loses its aesthetic and functional value at the end of its lifecycle11,12,13. Moreover, recycling of post-consumer denim waste into new textile products is also very challenging due to loss of fiber strength and length. Denim fabric is often made from cotton having more than 90% cellulose. Cellulose is made up of several functional groups like hydroxyl, carbonyl and others. Due to the presence of hydroxyl groups on its surface the cellulose can be converted into sorbents having potential to extract positive metal ions from a contaminated medium14. These functional groups can further be oxidized to convert hydroxyl ion into carboxylic acid groups having more strong potential to sorb heavy metal ions compared to hydroxyl groups14,15,16.
To address the issues of heavy metals in water and denim waste environmental footprint, the present study is aimed at recycling denim waste and preparing a sustainable and cost-effective sorbent using environmentally safe hydrogen peroxide and ozone for generation of carboxylic acid groups on denim waste. Moreover, the present study also explores the possibility to regenerate sorbent once it is saturated with heavy metals during removal process. This study has a practical significance as it will tackle two environmental problems at the same time. This study is novel as it involves preparing two sustainable sorbents by recycling waste material into useful and reuseable sorbents having strong sorption potential for metal ions. Both sorbents are environmentally friendly as no harmful chemicals are used during their synthesis and will have minimum environmental impacts.
Results and discussion
Selection of efficient oxidation treatment
The effect of different oxidation treatments on the removal of Cd was studied by varying the oxidation treatments for H2O2 (0, 10, 25, 50, 100, 200, 400 ml/L), and for Ozone (0, 15, 30, 45, 60, 75 min). The adsorbent dose (0.01 g), shaking time (1 h) and Cd concentration (100 ppm) were kept constant and different H2O2 and ozone treatments were tested as described above. These samples were run on Atomic Absorption Spectrophotometer (AAS) and Cd removal percentage was calculated (Fig. 1a and b). We achieved maximum Cd removal percentage (96%) with H202 treatment 200 ml/L (ODF@H202) and 89% with Ozone treatment of 75 min (ODF@03) compared to fabric with untreated denim fabric (UDF) which gave only 29% Cd removal percentage. Both above samples were chosen for further use in the experiment.
Cadmium removal percentage achieved with different levels of H2O2 (a) and Ozone treatments (b).
Analysis of samples
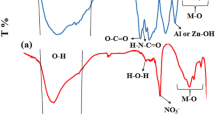
Once the samples with the highest capacity to absorb cadmium ions from water have been shortlisted, various analysis of those samples were conducted. Figure 2 shows the FTIR of untreated and treated denim fabrics. The FTIR spectrum of untreated denim fabric exhibits peaks that originated from cellulose macromolecule and indigo dye at 3345 cm−1 attributed to O–H stretching, 2941 cm−1 related to C–H aromatics, 1589 cm−1 for N–H stretching, 1433 cm−1 and 1058 cm−1was due to C–N and C-O stretching respectively17. From the FTIR spectrum of (ODF@03) and (ODF@H202), it can be observed that the peak at 1058 cm−1 i.e. the β-glucosidic bond in the cellulose macromolecules do not show any major variation between the untreated and treated fabrics. Furthermore, it was noted that the band at 3345 cm−1 due to OH stretching experienced a minor shift in the position to 3312 cm−1 and 3344 cm−1after the treatment with ozone and hydrogen peroxide respectively. This slight modification results in a reduction of the hydrogen bonds between the cellulose macromolecules18.
FTIR spectra of UDF, ODF@H202, and ODF@03.
Figure 3a shows the scanning electron microscopic image of UDF. Figure 3b shows the fabric treated with ODF@03 whereas, Fig. 3c is that of ODF@H202. The ozone treatment made the surface of fibers very rough due to oxidation of cellulosic chains. The treatment with hydrogen peroxide happens to be very drastic. It causes breakage of cellulosic chains due to excessive oxidation which leads to generation of beads on fibers. EDX analysis of UDF, ODF@H202, and ODF@03 was performed to analyze the different elements and their weight percentages. Figure 3d and e, and 3f show the results of treated and untreated denim fabrics. The weight% of oxygen elements enhanced in ODF@03 due to the oxidation of denim fabric Fig. 3f while, ODF@H202 has less weight% of oxygen elements Fig. 3e relative to the UDF Fig. 3d, and ODF@03 Fig. 3 f. which may be attributed to loss of oxygen due to formation of smaller volatile molecules like carbon dioxide. The exact values of weight% of different elements are represented in Fig. 3d and e, and f.
SEM images of UDF (a), ODF@H202 (b) ODF@03 (c) and EDX analysis of UDF (d), ODF@H202 (e) ODF@03 (f).
The XRD spectrum of prepared sorbents are represented in Fig. 4. XRD pattern of UDF exhibits peaks at 14.9, 16.9 22.9, and 34.8˚ which correspond to (1–10), (110), (200) and (004) crystalline planes respectively (Fig. 4a) which are associated with the crystalline structure of cellulose18. The XRD pattern of ODF@H202 is presented in Fig. 4b which shows that the peak intensity of peaks at 14.9˚ and 16.9˚ is very low whereas, there is no significant change in case of ozone treatment (ODF@03) Fig. 4c. This might be due to the oxidation of cellulose fibers in the denim fabric by hydrogen peroxide due to high concentration causing slight disintegration of crystalline structure, while the ozone treatment was just a surface treatment. However, the peak at 34.8˚ remained unaffected during hydrogen peroxide treatment which corresponds to plane (004) and is characteristic peak of cellulose (II). This plane is formed during mercerization (caustic treatment) due to staking of crystallographic layers and has very strong hydrogen bonding which resists disintegration of structure due to hydrogen peroxide treatment in ODF@H202.
XRD analysis of UDF (a), ODF@H202 (b) and ODF@03 (c).
Sorption experiment
Sorption isotherm
The effect of different initial Cd concentrations (5, 25, 50, 100, 200 and 400 ppm) on the removal of Cd from water was tested using 0.01 g of both sorbents dose, at normal pH and optimum shaking time. The plot of equilibrium metal concentration against experimental sorption capacities is presented in supplementary Fig. 1a and 1b. It was noticed that by increasing the initial concentration, the sorption capacity rapidly increased. Figure shows the high sorption efficiency at low initial concentration by exhibiting a sharp slope, while exhibiting a flat slope at higher concentration which indicates saturation of sorption at higher concentration of metal. The increment in metal sorption potential may be due to diffusion-based mechanism on both sorbents19 and the higher metal concentration also increases the contact opportunities and provides a strong driving force to react with the functional groups present on the surface of sorbents ODF@H202 and ODF@03. The maximum sorption capacity was achieved at 100 ppm metal concentration. After that a decrease in removal % was recorded. This decrease in sorption rate after 100 ppm metal concentration may be due to the insufficient binding sites pertaining to saturation at very high metal concentrations20. To further understand the sorption mechanism on ODF@H202 and ODF@03, the obtained experimental sorption results were fitted with Freudlich, Langmuir and Temkin models. The Langmuir model assumes the monolayer sorption of adsorbate on the surface of adsorbent21. The Langmuir equation can be represented as
where, qe (mg g−1) represents the equilibrium sorption capacity of metal ions on surface, qmax (mg g−1) represents the maximum sorption capacity of metal ions at complete monolayer coverage. Ce (mg L−1) is the concentration of metal ions at equilibrium. KL (L mg−1) is the Langmuir constant related to the heat of sorption21.
The Freundlich model describes the sorption behavior for sorbents having heterogenous surfaces and assumes multilayer sorption of adsorbate on the heterogenous surface of adsorbent. The Freundlich equation can be represented as:
where, qe (mg g−1) represents the equilibrium sorption capacity of metal ions on surface. Ce (mg L−1) is the concentration of metal ions at equilibrium. Kf (mg g-1) is the Freundlich sorption isotherm constant related to sorption capacity. 1/n represents the magnitude of driving force for sorption and indicates the heterogeneity of the data distribution.
The Temkin equation can be represented as
where, qe (mg g−1) represents the equilibrium sorption capacity of metal ions on surface. Kt (L g−1) is the Temkin sorption isotherm constant. T is the absolute temperature (K). R represents the ideal gas constant 8.314 (J mol−1K). bT is the constant related to the heat of sorption (Jmol−1).
The fitting results for three models have been presented in Fig. 5a and f. and the model’s calculated parameters have been presented in Table 1. It is clear from Fig. 5c and d, and Table 1 that Langmuir model best fitted with the sorption of Cd on ODF@H202 and ODF@03 as compared to Freundlich (Fig. 5a and b) and Temkin model (Fig. 5e and f) as indicated by the higher correlation coefficients values (Table 1). This indicates that the sorption of Cd on ODF@H202 and ODF@03is monolayer and homogenous22,23,24. The maximum sorption capacities for Cd calculated from Langmuir model were 238.1 and 175.4 mg g−1 for ODF@H202 and ODF@03 respectively indicating that ODF@H202 has 36% more sorption potential for Cd compared to ODF@03. The separation factor RL was calculated for Langmuir model to estimate the extent of sorption using the following Eq.
When the value of RL is 0 the sorption is irreversible, beneficial when 0 < RL < 1 and adverse when RL > 125. The RL values we achieved for both ODF@H202 and ODF@03 were between 0 and 1, so the sorption of Cd on both sorbents is beneficial. Furthermore, both of our sorbents exhibited more sorption capacities compared to most of the previously reported sorbents. The comparison of maximum sorption capacities of ODF@H202 and ODF@03 for Cd metal with other reported sorbents has been presented in supplementary Table 1.
Sorption isotherms for Freundlich (a and b) Langmuir (c and d) and Temkin (e and f) for the sorption of Cd on ODF@H202 and ODF@03 respectively.
Sorption kinetics
The effect of contact time on the sorption of Cd on ODF@H202 and ODF@03 was investigated using different shaking times (0, 5, 10, 20, 40, 60 and 120 min). Figure 6a shows the sorption capacities (qt) achieved as a function of time at three different initial concentrations 5, 50, and 100 mg L−1). It is clear from the figure that the sorption capacities rapidly increased with increasing contact time reaching to maximum at 20 min of contact time, after that became constant for both ODF@H202 and ODF@03by reaching at equilibrium time22. The very.
short equilibrium time (20 min) as we achieved in this study for heavy metal removal by both ODF@H202 and ODF@03 is beneficial for practical application of these sorbents for wastewater treatment. To further explore the sorption mechanism the experimental results were modeled by Pseudo first and Pseudo second order kinetic models. The equations for both models are given as under
Where K1 (min−1) and K2 (g mg−1 min−1) represent the constants of reaction rate for Pseudo first and Pseudo second order models respectively. The plots between time (min) and ln(qe-qt) for pseudo first order and between time (min) and t/qt for pseudo second order have been presented in (supplementary Fig. 2a and 2b (ODF@H202) and Fig. 2b and c (ODF@03). The calculated parameters for these models are given in Table 2. It is clear from Table 2 that Pseudo second order gave best fit for the sorption of Cd on both ODF@H202 and ODF@03, exhibiting correlation coefficients (R2) values 0.9997 and 0.9993 for ODF@H202 and ODF@03respectively compared to Pseudo fort order R2(0.6278 and 0.5674) respectively26;27, Furthermore, the qe values calculated from Pseudo second order (181.8 and 161.3 mg g−1) for ODF@H202 and ODF@03 respectively at initial concentration 100 mg L−1 are very close to the experimental values i.e. (180.5 and 160.5 mg g−1) respectively indicating best fitting of Pseudo second order model for the Cd sorption. These findings can be supported by previous published work for Pb sorption22and Cd sorption23. So, the Cd sorption kinetics in our study followed pseudo second order kinetics and is governed by chemical interactions between metal ions and the abundance of COOH groups present (Fig. 2) on the surfaces of ODF@H202 and ODF@03.
Effect of pH
The solution pH is an important factor affecting the sorption of Cd by ODF@H202 and ODF@03 sorbents. The surface electrical properties of ODF@H202 and ODF@03 as well as the forms of Cd in the solution can vary with the change in solution pH. To investigate the effect of different solution pH on the sorption of Cd by ODF@H202 and ODF@03, we changed the pH from 2 to 7 while other batch conditions were kept constant as in previous sections and the results are shown in Fig. 6b. The minimum Cd sorption capacities were recorded at pH 2 i.e. 37 and 14 mg g−1 by ODF@H202 and ODF@03respectively. As at low pH the surface of both sorbents is positively charged and Cd is least sorbed on these surfaces due to electrostatic repulsion28. A sharp increase in the sorption capacities was noted with increasing solution pH and maximum sorption capacities (180.5 and 160.5 mg kg−1) for both ODF@H202 and ODF@03 were recorded at Ph 6. When we increase the pH the surface functional groups of both ODF@H202 and ODF@03are deprotonated making their surfaces gradually negatively charged and at high pH due to electrostatic attraction the positively charged Cd ions are more sorbed on the surfaces of both sorbents. These findings can be supported by23,26.
Effect of different shaking times (a) and effect of different pH (b) on the equilibrium sorption capacity of Cd by ODF@H202 and ODF@03, The plot of (1/T) versus LnKd for ODF@H202 (c) and ODF@03 (d).
Sorption thermodynamics
To investigate the effect of different temperatures on the sorption of Cd ions from contaminated water by ODF@H202 and ODF@03we conducted a thermodynamic study at three different temperatures i.e. 293.15, 313.15 and 333.15 K under optimized pH conditions. For both sorbents a significant increase in equilibrium sorption capacities was recorded with increasing temperature. The thermodynamic parameters like Entropy change, Enthalpy change, and Gibbs free energy can provide the better information regarding spontaneity and thermodynamic changes during sorption processes29. The equations being used are as under
Where, ΔSo is the entropy change, ΔHo is the enthalpy change, ΔGo represents the Gibbs free energy related to sorption process, Kd is the distribution coefficient calculated by dividing equilibrium sorption capacity (Qe) over initial Cd concentration (Co). The calculated thermodynamic parameters have been presented in Table 3.
The plot of (1/T) versus LnKd for both sorbents ODF@H202 and ODF@03 have been shown in Fig. 6c and d. The ΔHo and ΔSo were calculated from the slope and intercept respectively Table 3. As we increased the temperature the distribution ratio Kd was also increased. A direct proportion between Kdvalues and temperature indicates that the sorption process is endothermic30. We obtained negative values of Gibbs free energy (ΔGo) at all temperatures indicating the spontaneous sorption of Cd2+ ions onto both ODF@H202 and ODF@03. Furthermore, the ΔGo negative values decreased with increasing temperature which suggests that at high temperatures a stronger adsorptive force exists between sorbents and Cd2+metal cations. These findings can be supported by22. The positive values of entropy change (ΔSo) confirmed the structural changes in sorbents ODF@H202 and ODF@03 and affinity of both sorbents for Cd2+metal cations22,30. The positive values of entropy change ΔSo also confirmed the occurrence of ion replacement reaction and enhanced randomness at solid-solution interface during sorption of Cd2+ onto ODF@H202 and ODF@03 Gubbuk, (2011) results for the sorption of Cu2+ ions onto sporopollenin surface supports our findings for Cd2+ ions sorption on ODF@H202 and ODF@0331 .
Regeneration study
A regeneration study was conducted to check the recycling ability of both ODF@H202 and ODF@03 sorbents. The results of the regeneration study have been presented in Fig. 7. which shows that both sorbents exhibit minimal changes in the sorption efficiency during all 10 cycles. The sorbents ODF@H202 and ODF@03 gave adsorption efficiency of 99 and 96% after 1 cycle of desorption. Whereas, after 10 recycles the ODF@H202 and ODF@03 sorbents gave sorption efficiencies of 90 and 84% respectively. The efficient regeneration abilities of both sorbents indicate their suitability for their practical applications in wastewater treatment. Moreover, the cost of sorbent preparation from waste denim is nominal. The waste denim was collected from the local waste Collection Centre for free of cost. The treatment costs include hydrogen peroxide, sodium hydroxide and ozone treatment which is just a few cents per kilogram of sorbent.
Desorption cycles of both sorbents.
Conclusion
The present research work is designed to convert the challenge of landfill with waste denim into an opportunity to address the problem of heavy metal contamination of water. The waste denim fabric was oxidized with H202 and ozone under alkaline conditions using two simple techniques yielding ODF@H202 and ODF@03. The spectroscopic and microscopic analysis confirmed the generation of new carboxylic groups on the surfaces and complete change in the surface morphology of both functionalized sorbents. The sorption efficiency of both sorbents was evaluated in a batch laboratory experiment. Both sorbents showed excellent sorption capacities for Cd from contaminated water. The ODF@H202 sorbent was found to be more effective by giving 36% more sorption capacity compared to ODF@03. The Langmuir and pseudo second order model best fitted with the experimental sorption results. The sorption isotherm, sorption kinetics and sorption thermodynamics revealed the sorption process as spontaneous and endothermic with chemisorption as a dominant process governing the sorption process. The present work addresses a practical approach as both sorbents can effectively be implied for wastewater treatment due to having efficient sorption capacities and recycling ability of 90% even after 10 recycles. Furthermore, our findings present a cost-effective and very simple technique where a waste is converted into a sustainable material having strong potential to address heavy metal contamination of water with minimum environmental impacts.
Materials and methods
Chemicals and materials
Hydrogen peroxide, sodium hydroxide (NaOH), sodium meta bisulfite and cadmium nitrate were procured from Merk Germany and were of analytical grade. The Cd stock and sub stock solutions were prepared in deionized water. The waste denim fabric was collected from disposed garments. All the glassware, centrifuge tubes, and laboratory consumables used in the experiment were dipped in nitric acid solution (20%) and then thoroughly washed three times with deionized water before use for quality assurance.
Preparation of sorbent
The oxidation of denim fabric with hydrogen peroxide (H2O2) (named as ODF@H202) was done in the presence of a base in a beaker with stirrer and thermometer. A 10 cm2 piece of denim fabric was treated with different levels of H2O2 (5, 25, 50, 100, 200 and 400 ml/L) with 6 g of NaOH (sodium hydroxide) dissolved in distilled water. Continuous stirring was done on medium flame using Bunsen burner. When the temperature of the solution reached 85 ℃ the flame was slowed down, and further stirring was done for 30 min. After that, the fabric was removed and rinsed with distilled water. After washing it was placed on aluminum foil and dried in oven at 80 ℃ for 15 min.
A second piece of fabric was treated with ozone to generate carboxylic acid groups (named as ODF@03). For this, an A4 size piece of moistened fabric was placed in the ozone treatment machine from Tonello (denim fading machine) that contains (03 48 g/h with 100% moisture) in air. Five samples of fabrics were run separately on ozonation machine with different spinning times (15, 30, 45, 60 and 75 min). To neutralize the fabric, sodium meta bisulfite (2 g/L) was used for 8 min. Adjusted the rotation at 27 rpm for 4 min. Then the first Drain was done by filling water 5 L/kg for 2 min. After the second drain, spinning was done at 200 rpm for 4 min, and then fabric was taken out from machine. The fabric was Squeezed and placed on aluminum foil and dried in the oven at 80 ℃ for 15 min. the schematics for the preparation of both sorbents has been presented in the Fig. 8a and b.
Schematic diagram for the preparation of sorbents.
Batch sorption experiment
A batch sorption experiment was conducted in the laboratory to optimize the different factors affecting Cd sorption on ODF@H202 and ODF@03. A stock solution (1000 ppm) of Cd was prepared in deionized water using Cd (NO3)2·4H2O from which further sub stocks of 5, 25, 50, 100, 200, and 400 ppm concentrations were prepared. A sorbent sample of 0.01 g of both ODF@H202 and ODF@03 was inserted in the 20 ml Cd solutions of different concentrations as stated above in 50 ml centrifuge tubes. The centrifuge tubes were shaken on an orbital shaker for 1 h at 180 rpm at 25 ℃. Then the sorbent material was removed from the solution and the solution mixture was filtered by filter paper. The atomic absorption spectrophotometer (AAS) was used to examine the remaining metal ion concentration after sorption. The sorption experiment was repeated 3 times and results were taken as an average of three replicates. The sorption trial was further performed using different shaking times of (0, 5, 10, 20, 40, 60, 120) min, different pH (2, 3, 4, 5, 6, and 7) different temperatures (293.15, 313.15 333.15) K while keeping the other batch conditions constant. The experimental sorption capacities (mg g−1) and metal removal % were calculated using the following Eq.
Where Co is the initial concentration and Ce is the equilibrium concentration of the cadmium, V is the volume in (L) and m is the quantity of sorbent in (g). The experimental results obtained allowed the determination of the sorption isotherm in the system Q = f(Ce) and Cd removal curves in the system of R = f(Co).
Regeneration study
To check the sustainability of the two prepared sorbents a series of desorption steps were conducted using the same treated fabrics for all cycles. The sorbents were treated with nitric acid to remove Cd ions from fabric. For this purpose, both sorbents removed from batch study from 100 ppm Cd solution were thoroughly washed with deionized water and placed in 15 ml HNO3 (1 mol/L) for 1 h. After that the sorbents were dried in oven and reused to check the sorption capacity in new 100 ppm Cd solution. The batch conditions were kept the same as mentioned above. This desorption process was repeated for 10 cycles using the same sorbents and sorption capacity was calculated for each cycle.
Data availability
The datasets used or analyzed during this research work are available from the corresponding author upon request.
References
Liang, W. et al. Recent advances of carbon-based nano zero valent iron for heavy metals remediation in soil and water: a critical review. J. Hazard. Mater. 426, 127993 (2022).
Ghosh, S. et al. Advancing wastewater treatment with Azolla filiculoides waste: a comprehensive review of adsorption applications. Environ. Technol. Reviews. 13 (1), 359–378 (2024).
Liu, J., Cao, L. & Dou, S. Trophic transfer, biomagnification and risk assessments of four common heavy metals in the food web of Laizhou Bay, the Bohai Sea. Sci. Total Environ. 670, 508–522 (2019).
Şimşek, A., Özkoç, H. B. & Bakan, G. Environmental, ecological and human health risk assessment of heavy metals in sediments at Samsun-Tekkeköy, North of Turkey. Environ. Sci. Pollut. Res. 29, 2009–2023 (2022).
Nasab, H. et al. Association of as, Pb, Cr, and zn urinary heavy metals levels with predictive indicators of cardiovascular disease and obesity in children and adolescents. Chemosphere 294, 133664 (2022).
Nogueira, T. A. R. et al. Comparing soil-to-plant cadmium (cd) transfer and potential human intake among rice cultivars with different cd tolerance levels grown in a tropical contaminated soil. Environ. Monit. Assess. 194, 1–13 (2022).
Benettayeb, A. et al. Insights into logical method selection for modification of chitosan and alginate towards the adsorption of heavy metal ions: a review. Environ. Technol. Reviews. 13 (1), 398–420 (2024).
Nucera, S. et al. Non-essential heavy metal effects in cardiovascular diseases: an overview of systematic reviews. Front. Cardiovasc. Med. 11, 1332339 (2024).
Drzeżdżon, J., Jacewicz, D. & Chmurzyński, L. The impact of environmental contamination on the generation of reactive oxygen and nitrogen species–consequences for plants and humans. Environ. Int. 119, 133–151 (2018).
Pensupa, N. et al. Recent trends in sustainable textile waste recycling methods: current situation and future prospects. In: Lin, C. (eds) Chemistry and Chemical Technologies in Waste Valorization. Topics in Current Chemistry Collections. Springer, Cham. https://doi.org/10.1007/978-3-319-90653-9_7.
Huang, X. et al. Industrialization of open-and closed-loop waste textiles recycling towards sustainability: a review. J. Clean. Prod. 436, 140676 (2024).
Singh, V., Wyatt, J., Zoungrana, A. & Yuan, Q. Evaluation of vermicompost produced by using post-consumer cotton textile as carbon source. Recycling 7, 10 (2022).
Niinimäki, K. Textiles and garment lifetimes. In Niinimäki, K. (eds) Recycling and Lifetime Management in the Textile and Fashion Sector. CRC. 1–14. (2024).
Oussadi, K. et al. Wool keratin as a novel, alternative, low-cost adsorbent rich in various–N and–S proteins for eliminating methylene blue from water. Biomass Convers. Biorefinery 1–15. (2024).
Raj, C. S., Arul, S., Sendilvelan, S. & Saravanan, C. Bio gas from textile cotton waste-an alternate fuel for diesel engines. Open. Waste Manage. J. 2, 1–5 (2009).
Kabogo, I. T. et al. Extraction of heavy metals from water using chelating agents: a comprehensive review. Int. J. Environ. Sci. Technol. 21, 8749–8792 (2024).
Hu, Y. et al. Valorisation of textile waste by fungal solid state fermentation: an example of circular waste-based biorefinery. Resour. Conserv. Recycl. 129, 27–35 (2018).
Peng, B. et al. Cellulose-based materials in wastewater treatment of petroleum industry. Green. Energy Environ. 5, 37–49 (2020).
Sayyed, A. J. et al. Cellulose-based nanomaterials for water and wastewater treatments: a review. J. Environ. Chem. Eng. 9, 106626 (2021).
Atav, R., Gündüz, Ö., Yaz, S., Çakan, G. & Sevgili, B. Eco-friendly ozone process for Denim garments as an alternative to conventional bleaching. Fibers Polym. 25, 2661–2667 (2024).
Talebi, S. & Montazer, M. Denim Fabric with Flame retardant, hydrophilic and self-cleaning properties conferring by in-situ synthesis of silica nanoparticles. Cellulose 27, 6643–6661 (2020).
El Nemr, A. et al. New magnetic cellulose nanobiocomposites for Cu (II), cd (II) and pb (II) ions removal: kinetics, thermodynamics and analytical evaluation. Nanatechnol. Environ. Eng. 6, 42 (2021).
Bojic, D. V. et al. Removal of the herbicide 2, 4-dichlorophenoxyacetic acid from water by using an ultrahighly efficient thermochemically activated carbon. Hemijska Industrija. 73 (4), 223–237 (2019).
Benettayeb, A. et al. Facile fabrication of new bioadsorbents from Moringa oleifera and alginate for efficient removal of uranium (VI). J. Radioanal. Nucl. Chem. 333, 2369–2387 (2024).
Massoud, A., Waly, S. & Abou El-Nour, F. Removal of U (VI) from simulated liquid waste using synthetic organic resin. Radiochemistry 59, 272–279 (2017).
Aziz, H., Murtaza, G., Usman, M., Basra, S. & Niaz, A. Enhancing chlorpyrifos sorption potential of agricultural soil by biochar and compost addition. Pakistan J. Agricultural Sci. 55, 833–841 (2018).
Lian, Q. et al. The effects of carbon disulfide driven functionalization on graphene oxide for enhanced pb (II) adsorption: investigation of adsorption mechanism. Chemosphere 248, 126078 (2020).
Guo, Z. et al. Fabrication of polyethylenimine functionalized magnetic cellulose nanofibers for the sorption of Ni (II), Cu (II) and cd (II) in single-component and multi-component systems. Int. J. Biol. Macromol. 184, 68–78 (2021).
Jamshidifard, S. et al. Incorporation of UiO-66-NH2 MOF into the PAN/chitosan nanofibers for adsorption and membrane filtration of pb (II), cd (II) and cr (VI) ions from aqueous solutions. J. Hazard. Mater. 368, 10–20 (2019).
Özdemir, V. T., Tuğaç, H. M. & Arar, Ö. Two-Pot oxidative Preparation of Dicarboxylic Acid containing cellulose for the removal of Beryllium (Be2+) from aqueous solution. Curr. Anal. Chem. 18, 360–369 (2022).
Li, B. et al. Remediation of cd (II) ions in aqueous and soil phases using novel porous cellulose/chitosan composite spheres loaded with zero-valent iron nanoparticles. Reactive Funct. Polym. 173, 105210 (2022).
Acknowledgements
The authors are thankful to the department of Environmental Sciences Government College University Faisalabad Pakistan for providing the research facilities.
Funding
Open access funding provided by Swedish University of Agricultural Sciences.
The open access funding was provided by the Swedish University of Agricultural Sciences, 23456 Alnarp, Sweden.
Author information
Authors and Affiliations
Contributions
H.A. and M.A. : Writing- Original draft preparation, conceptualization, conducted research, M.R. and U.R. : Methodology, Software. Data curation, validation, S.A., A.R. and J.W. H.Y. Visualization, Investigation, validation.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aziz, H., Ashraf, M., Rizwan, M. et al. Valorization of textile waste for removal of Cadmium from contaminated water. Sci Rep 14, 30611 (2024). https://doi.org/10.1038/s41598-024-82456-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-82456-x
Keywords
This article is cited by
-
Sustainable remediation of cationic dyes using Pistacia vera testa-derived nanoparticles: experimental validation and theoretical modeling
Environmental Science and Pollution Research (2025)