Abstract
The evolving TNM classification has emphasized the tumor size’s role in NSCLC prognosis, reclassifying stage IIIA patients from the previous edition as stage IIIB (T3-4N2M0, 8th edition). However, the prognostic implications of tumor size and survival in stage III NSCLC patients undergoing neoadjuvant therapy remain unexplored. Therefore, we investigated the association between tumor size and mortality in N2 non-small cell lung cancer (NSCLC) patients undergoing neoadjuvant concurrent chemoradiotherapy followed by surgery (trimodality therapy), considering the number of metastatic N2 stations and histology. We analyzed 756 patients with stage III (T1-3N2) NSCLC who underwent trimodality therapy, excluding those with T3 tumors with invasion components or additional nodules (2003–2019). Overall survival was compared using the Cox-proportional hazards model, while the tumor size-survival relationship was estimated using restricted cubic splines. Using 8th TNM edition, 32.1%, 48.5%, and 19.3% were clinical T1, T2, and T3. During a median follow-up of 53.5 months, 398 patients died. The adjusted hazard ratios for overall survival comparing T2 and T3 to T1 were 1.46 (95% confidence interval, 1.14–1.85) and 1.48 (1.10–1.99). For the extent of clinical N2, large tumor size increased the mortality risk in patients with N2b but not in N2a. Tumor size did not increase mortality risk in squamous cell carcinoma patients; however, the mortality risk was increased with larger tumors in adenocarcinoma. These findings raise the importance of considering tumor size in treatment planning and suggesting tailored strategies.
Similar content being viewed by others
Introduction
Tumor size is an important prognostic factor for non-small cell lung cancer (NSCLC), with larger tumors being associated with poorer prognosis1. The 7th edition of the TNM classification system stratified tumor size into T2 (3–7 cm) and T3 (> 7 cm), and the 8th edition further revised this to T2 (3–5 cm), T3 (5–7 cm), and T4 (> 7 cm).2,3As a result, patients previously diagnosed with stage IIIA NSCLC were now upstaged to stage IIIB (T3-4N2M0)4. This could potentially lead to under-treatment of these patients and deprive them of a chance for cure. However, there is limited evidence on the prognostic significance of primary tumor size and the survival outcomes of patients diagnosed with stage III NSCLC who underwent neoadjuvant therapy and surgery3.
Treatment recommendations for stage III NSCLC have primarily been based on evidence from randomized trials published before 2010 when the TNM staging system (5th or 6th editions) used a 3 cm threshold to distinguish between T1 and T2 tumor classifications5,6. Moreover, tumor size was not explicitly considered as a prognostic factor, unlike the number of metastatic nodal stations and histology7,8,9. According to a large registry-based cohort study with 984 stage III NSCLC patients undergoing neoadjuvant concurrent chemoradiotherapy (CCRT) followed by surgery (trimodality therapy), survival differed by the number of metastatic nodal stations and histologic type10. While the increased risk of death was observed in patients with cN2b (N2-multistation) compared to those with cN2a (N2-single station), these findings were limited to squamous cell carcinoma and were not observed in adenocarcinoma. Thus, we evaluated the association between tumor size and mortality in N2 NSCLC patients with trimodality therapy. In addition, we investigated if the associations are different depending on the number of metastatic N2 stations or histology.
Results
Patient characteristics
The study sample had a mean age of 60.7 year [standard deviation (SD): 8.7 yr], and approximately 70.5% (n = 533) were male and 70% (n = 506) were ever-smokers. Based on the 8th edition of the TNM classification, 32.1%, 48.5%, and 19.3% were clinical T1, T2, and T3, respectively. In extent of clinical N2, 51.9%, 60.5%, and 51.4% were N2a in patients with T1, T2, and T3 tumors, respectively. Adenocarcinomas were the most common histologic type across 3 groups, it was more common in T1 (54.0%). Nodal downstaging after neoadjuvant therapy was less frequent in T1 (53.5%) than T2 (63.5%) and T3 (69.2%). Moreover, patients with T1 (81.1%) were more likely to receive adjuvant therapy than patients with T2 (69.2%) and T3 (68.5%) (Table 1).
Survival outcomes by clinical tumor size
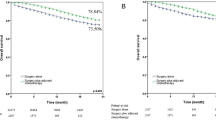
During a median follow-up of 53.5 months (interquartile range: 23.5–86.4 months), 398 patients died. The 5-yr OS was 61.8%, 52.0%, and 49.4% in the T1, T2, and T3 groups, respectively (p-value = 0.020; Fig. 1). The HRs for overall survival comparing T2 and T3 to T1 after adjusting for age, sex, smoking status, CCI, extent of clinical N2, and histology were 1.46 (95% CI: 1.14–1.85), and 1.48 (95% CI 1.10–1.99) (Table 2). Larger clinical tumor size was associated with higher risk of mortality (Fig. 1). In addition, older age, male sex, a Charlson Comorbidity Index (CCI) of 2 or high, clinical N2, adenocarcinoma, and both pneumonectomy and sub-lobar resection were significantly associated with an increased risk of mortality (Supplementary Table 1).
Kaplan–Meier plot and restricted cubic splines for overall survival by tumor size.
Survival outcomes by clinical tumor size, extent of clinical N2 and histology
Among patients with cN2a involvement, the adjusted HRs for OS comparing T2 and T3 to T1 were 1.21 (95% CI: 0.87–1.69), and 1.15 (95% CI: 0.75–1.78), respectively. The association between tumor size and the adjusted HR for mortality was linear (Tables 2 and Fig. 2; p-value for non-linearity = 0.288). Among patients with cN2b involvement, the adjusted HRs for overall survival comparing T2 and T3 to T1 were 1.69 (95% CI: 1.20–2.39), and 1.81 (95% CI: 1.21–2.71). The association between tumor size and the adjusted HR for mortality was non-linear, with a stronger association being observed with larger tumor sizes (Tables 2 and Fig. 2; p-value for non-linearity < 0.001). Specifically, the risk of mortality was rapidly increased at a tumor > 5 cm in size (Fig. 2).
Kaplan–Meier plots and restricted cubic splines for overall survival by tumor size, extent of N2 involvement, and histological type.
In the adenocarcinoma, the adjusted HRs for OS comparing T2 and T3 to T1 were 1.55 (95% CI: 1.17–2.04), and 1.50 (9 5% CI: 1.04–2.15), respectively. The association between tumor size and the adjusted HR for mortality was linear, with a stronger association being observed with larger tumor sizes (Tables 2 and Fig. 2; p-value for non-linearity = 0.111). In the squamous cell carcinoma, the adjusted HRs for OS comparing T2 and T3 to T1 1.38 (95% CI: 0.83–2.29), and 1.40 (95% CI: 0.80–2.45) with the nonlinear association (Tables 2 and Fig. 2; p-value for non-linearity < 0.001). The risk of mortality rapidly increased in patients with tumors > 5 cm in size (Fig. 2).
When stratified by extent of clinical N2 and histology, the adjusted HRs for OS comparing T2 and T3 to T1 in N2a and adenocarcinoma were 1.31 (95% CI: 0.88–1.96) and 0.95 (95% CI: 0.53–1.70). In N2a and squamous cell carcinoma, the adjusted HRs for OS comparing T2 and T3 to T1 were 1.28 (95% CI: 0.66–2.50) and 1.31 (95% CI: 0.62–1.81). In N2b and adenocarcinoma, T2 and T3 were associated with a higher risk of mortality compared to T1 (HR 1.80, 95%CI: 1.22–2.61 in T2; HR: 1.89, 95% CI: 1.16–3.06 in T3). In N2b and squamous cell carcinoma, the adjusted HRs for OS comparing T2 and T3 to T1 were 1.64 (95% CI: 0.74–3.66) and 1.71 (95% CI: 0.72–4.06) (Tables 3 and Fig. 3).
Overall survival by tumor size, extent of N2 involvement, and histological type.
Discussion
In this study, we classified clinical tumor size into clinical T1, T2, and T3 using the 8th edition of the TNM staging classification and found that larger tumor size was associated with higher risk of mortality in patients with N2 NSCLC undergoing trimodality therapy. However, the mortality risk across the tumor size differed according to the extent of clinical N2 involvement and histologic subtype. While tumor size was not associated with mortality risk in patients with N2a, larger tumors increased the risk of death in patients with N2b. Similarly, there was no association between tumor size and mortality in patients with squamous cell carcinoma, whereas the risk of death increased with larger tumor size in adenocarcinoma.
Based on the analysis of upfront surgery cohort for IASLC proposed 8th edition of TNM classification, which refined tumor size, notable outcome differences became evident even with small increases in tumor size3. This has led to the subclassification of stage I patients into four subgroups and the upstaging of node-negative tumors larger than 5 cm to stage II, while a reclassification of tumors larger than 5 cm with N2 disease as stage IIIB4. However, the applicability of these findings to patients undergoing neoadjuvant therapy remains uncertain due to the potential influence of neoadjuvant therapy on tumors. Nonetheless, previous studies have consistently suggested that tumor size can serve as a predictive factor for patient prognosis, even before start of neoadjuvant therapy15,16,17,18,19. In our study, an increase in tumor size was associated with a greater risk of death in patients with N2 NSCLC undergoing trimodality therapy, suggesting that it may function as an independent prognostic factor that needs to be considered for planning neoadjuvant therapy for N2 NSCLC.
From the 7th edition of the TNM classification system, an enhanced focus was placed on tumor size, and the 8th edition further refined this by classifying tumors between 3 and 5 cm as T2, 5 and 7 cm as T3, and those exceeding 7 cm as T43. This led many patients previously staged as IIIA NSCLC to be upstaged to stage IIIB. Patients with IIIB were not usual candidates for neoadjuvant therapy: while patients with T1 and T2 tumors would receive neoadjuvant therapy, patients with T3 would not be eligible for neoadjuvant treatment20,21,22,23. However, our study suggests that the prognostic value of tumor size can differ with the extent of nodal metastasis. Specifically, tumor size was not associated with an increased risk of death in N2a patients, indicating that even patients with T3 tumors might benefit from neoadjuvant therapy if N2 involvement is limited to a single station. This finding aligns with the NCCN guideline, which recommends neoadjuvant therapy for patients with T1-T3 NSCLC with single N2 disease24. In contrast, the ESMO guideline recommends definitive CCRT for patients with N2b25. In our study, while the risk of death increased with tumor size in N2b patients, N2b patients with T1 tumors showed similar survival rates to cN2a patients. This suggests that patients with N2b should not be considered as one group and may require stratification based on tumor size for precise risk assessment. For example, neoadjuvant therapy needs to be considered for N2b patients with the tumors 3 cm or smaller. Thus, clinicians need to consider not only tumor size but also nodal metastasis extent when planning treatment for patients with N2 NSCLC.
We also observed that T1 patients were more likely to receive adjuvant therapy compared to those with T2 or T3 tumors. T1 patients in our cohort had a higher incidence of adenocarcinoma and a large proportion of nodal non-responders. Although adjuvant therapy following trimodality treatment is not an established standard, these characteristics may have contributed to the increased use of adjuvant therapy in T1 patients, as clinicians may aim to mitigate recurrence risks associated with adenocarcinoma and nodal non-response. Previous studies have similarly shown that treatment response and prognosis can vary significantly by histologic type, with squamous cell carcinoma often showing a better nodal response to trimodality therapy than adenocarcinoma, which may benefit more from additional systemic treatments after resection26.
Although the current 8th edition staging guidelines do not formally distinguish between N2a and N2b, our findings support the IASLC’s initial proposal during the development of the 8th edition to further subclassify N2 into N2a1, N2a2, and N2b based on the number of metastatic N2 stations4. While this proposal was not included in the final 8th edition, it highlighted the potential prognostic importance of single versus multistation N2 disease, which our study supports. The forthcoming 9th edition of the TNM staging system is expected to incorporate this distinction, reflecting the growing recognition of its clinical significance27. Our results provide additional evidence suggesting that both tumor size and the number of N2 stations should be considered in staging and treatment planning. As treatment paradigms evolve and precision oncology advances, integrating both anatomic factors such as tumor size and nodal station number and biologic markers such as genetic and immune profiles could further enhance patient stratification and support individualized strategies.
In terms of histology, tumor size was associated with higher risk of death in patients with adenocarcinoma but not in those with squamous cell carcinoma. Interestingly, in subgroup analysis by histology and extent of clinical N2 involvement, larger tumor size increased the risk of death in cN2b patients with adenocarcinoma, while no association was found between tumor size and mortality in cN2a patients (Table 3). For squamous cell carcinoma, on the other hand, tumor size was not associated with an increased risk of death, regardless of the number of metastatic N2 stations. This might be due to the relatively small sample size of patients with squamous cell carcinoma or could reflect different biological mechanisms by histology or nodal metastasis pattern. The number of metastatic N2 stations and histology might be important staging components as well as tumor size in the precision diagnostic era. Further studies with larger sample sizes would be necessary to confirm our findings.
There are several limitations of this study. As we used the data from a lung cancer surgery registry and the standardized therapy at our institution was trimodality therapy for resectable N2 NSCLC, we included N2 patients who successfully completed trimodality therapy. There would be some N2 NSCLC patients who were excluded from this study as they were not able to receive trimodality therapy due to physical conditions or other medical reasons. Similarly, the study was conducted at a single tertiary cancer center in Korea, the results of our study might not be applicable to N2 patients with different treatments at different settings. However, characteristics of our study populations were similar to N2 NSCLC patients in Korea11,28,29and lung cancer treatment methods were standardized worldwide23,24,30. Lastly, while this is a relatively large study with N2 NSCLC with trimodality therapy, it still requires a larger sample to perform subgroup analysis. In addition, as our study period preceded the introduction of recent developments in neoadjuvant chemoimmunotherapy, the current results may not be generalized to patients who receive neoadjuvant chemoimmunotherapy. The role of clinical tumor size needs to be evaluated in these patients. Regardless of these limitations, our study has several strengths. We used data from a large registry which collected standardized and detailed information on patients, tumors, and treatments. All patients in this registry underwent PET-CT and invasive mediastinal staging to confirm N2 disease histopathologically. Furthermore, we had complete follow-up data for mortality due to national health insurance.
In conclusion, our study demonstrates that larger tumor size is associated with higher mortality in N2 NSCLC patients undergoing trimodality therapy, with risk variations depending on the extent of clinical N2 involvement and histology type. Specifically, trimodality therapy may be most beneficial for patients with N2a NSCLC regardless of tumor size and for N2b NSCLC patients with smaller tumors (T1). This finding highlights the importance of tumor size as a prognostic factor in treatment planning, particularly as treatment paradigms shift with the recent approvals of perioperative chemoimmunotherapy and targeted therapies, such as nivolumab and pembrolizumab, for resectable cases. Although our study focuses on patients treated between 2003 and 2019, when trimodality therapy was the standard, our findings remain relevant, especially as real-world data on tumor size and N2 station involvement in patients receiving chemoimmunotherapy are still limited.
Moreover, our findings suggest that previously unresectable N2 NSCLC may become resectable with effective multimodal treatment strategies, and surgery may further improve survival by increasing locoregional control rates. As treatment strategies evolve, additional studies specifically focusing on the role of tumor size and N2 station involvement in patients receiving chemoimmunotherapy are needed to refine staging and treatment guidelines. Our results also support the shift toward biologic staging, as traditional TNM classification alone may no longer fully capture prognosis in the precision oncology era. Integrating both anatomic and biologic factors, such as molecular profiling and immune response markers, could enhance patient stratification and treatment individualiz1ation31. This study thus advocates for a refined NSCLC staging approach that combines tumor biology with traditional TNM criteria, enabling more personalized and effective treatment strategies for N2 NSCLC patients.
Methods
Study population
This cohort study used data from the Registry for Thoracic Cancer Surgery, which includes clinical information on all patients who have undergone surgery for lung cancer at the Samsung Medical Center, Seoul, Korea, since 1995. The study population included patients with histologically confirmed clinical stage III (T1-3N2) NSCLC who underwent trimodality therapy between January 2003 and February 2019. Among the 1,134 patients who initially treated neoadjuvant CCRT during the period, 1,022 completed neoadjuvant CCRT followed by surgery. Of these, patients with clinical T4 tumors (n = 93), those who had not undergone positron emission tomography (PET; n = 19) or invasive mediastinal (n = 58) examination prior to neoadjuvant therapy were excluded. Additionally, patients with T3 tumors involving invasive components (n = 20) or additional pulmonary nodules (n = 39), as well as those with histology other than adenocarcinoma or squamous cell carcinoma (n = 64) were excluded. The final study sample included 756 patients (Fig. 4). The study was approved by the Institutional Review Boards of the Samsung Medical Center (IRB file number 2023-09-086). Informed consent was waived as we only used the deidentified data.
Flow chart of the study.
Staging workup and treatment protocol
Clinical staging was based on chest computed tomography (CT), positron emission tomography (PET)-CT, and brain magnetic resonance imaging. Mediastinal nodes were confirmed histologically using mediastinoscopy, endobronchial ultrasound-guided transbronchial needle aspiration, or thoracoscopy. TNM classification was determined according to the 8th edition of the American Joint Committee on Cancer Staging Manual4.
Our institution has adopted a multidisciplinary approach to managing patients with lung cancer with trimodality therapy being the primary recommendation for medically suitable patients with resectable N2 NSCLC11,12. The neoadjuvant CCRT regimen included both chemotherapy (comprising of 5 cycles of platinum-based doublet therapy) and thoracic radiotherapy (comprising of a total dose of 4,500 cGy for 5 weeks [between 2003 and 2009] or a total dose of 4,400 cGy for 4.5 weeks [after 2009])11. Chest CT and PET-CT were repeated after completion of neoadjuvant CCRT to allow tumor restaging, and surgery was performed 4 to 6 weeks later unless the restaging workup showed evidence of disease progression. The surgical procedures included anatomic pulmonary resection depending on the extent of the tumor and systematic nodal dissection of the ipsilateral hilum and mediastinum. Adjuvant therapy was added for patients with persistent N2 disease, multiple N2 with extracapsular extension, or close or positive resection margins on pathological reports. Surveillance after surgery included physical examination, blood tests, chest CT, and/or PET-CT every 3 months for the first 2 year and every 6 months thereafter11,12.
Clinical tumor size
The clinical tumor size was determined by measuring the longest diameter on the pretreatment CT imaging according to the recommendations of the International Association of Study for Lung Cancer (IASLC)13. Then the clinical tumor size was classified using the 8th edition of the TNM staging system (i.e., T1: ≤ 3 cm; T2: > 3 cm–≤ 5 cm; and T3: > 5–≤ 7 cm)4.
Extent of clinical N2 involvement
The extent of clinical N2 involvement was categorized into cN2a (single station N2) or cN2b (multiple station N2) based on the number of metastatic N2 stations adopting the proposed pathologic N subclassification by IASLC14. Clinical diagnosis of nodal metastasis was made through PET-CT and invasive mediastinal examination, with positive nodal metastasis being defined as either a positive finding upon histological examination or suspicion of nodal metastasis following PET-CT.
Study outcome
The outcome of this study is overall survival (OS). Patients were followed from the date of surgery to the date of death from any cause or last surveillance by March 31, 2023.
Statistical analysis
The cumulative incidence functions (CIF) for death were estimated using the Kaplan–Meier method and compared using the log-rank test. OS was calculated as the complement of CIF for death (1–CIF). Hazard ratios (HRs) and 95% confidence intervals (CI) for OS were estimated using the Cox proportional hazards model. HRs and corresponding 95% CIs were adjusted for age, sex, smoking history (never, ever, or unknown), Charlson comorbidity index (CCI; 0, 1, or ≥ 2), extent of clinical N2 involvement (cN2a and cN2b), and histological type (adenocarcinoma or squamous cell carcinoma). We performed subgroup analysis by the extent of clinical N2 involvement and by histology. In addition, we modeled tumor size as a continuous variable using restricted cubic splines with knots at the 10th, 50th, and 90th quantiles to provide a flexible estimate of the dose-response relationship between clinical tumor size and OS by extent of clinical N2 involvement and by histology. All analyses were performed using R statistical language version 4.2.3. P-value < 0.05 was considered statistically significant.
Data availability
The datasets used and/or analysed in the current study are available from the corresponding author on reasonable request.
References
Zhang, J. et al. Relationship between tumor size and survival in non–small-cell Lung Cancer (NSCLC): an analysis of the Surveillance, Epidemiology, and end results (SEER) Registry. J. Thorac. Oncol. 10 (4), 682–690. https://doi.org/10.1097/jto.0000000000000456 (2015).
Rami-Porta, R. et al. The IASLC Lung Cancer Staging Project: proposals for the revision of the T descriptors in the Forthcoming (Seventh) Edition of the TNM classification for Lung Cancer. J. Thorac. Oncol. 2 (7), 593–602. https://doi.org/10.1097/jto.0b013e31807a2f81 (2007).
Rami-Porta, R. et al. The IASLC Lung Cancer Staging Project: proposals for the revisions of the T descriptors in the Forthcoming Eighth Edition of the TNM classification for Lung Cancer. J. Thorac. Oncol. 10 (7), 990–1003. https://doi.org/10.1097/jto.0000000000000559 (2015).
Goldstraw, P. et al. The IASLC Lung Cancer Staging Project: proposals for revision of the TNM Stage groupings in the Forthcoming (Eighth) Edition of the TNM classification for Lung Cancer. J. Thorac. Oncol. 11 (1), 39–51. https://doi.org/10.1016/j.jtho.2015.09.009 (2016).
Albain, K. S. et al. Radiotherapy plus chemotherapy with or without surgical resection for stage III non-small-cell lung cancer: a phase III randomised controlled trial. Lancet 374 (9687), 379–386. https://doi.org/10.1016/s0140-6736(09)60737-6 (2009).
van Meerbeeck, J. P. et al. Randomized Controlled Trial of Resection Versus Radiotherapy after induction chemotherapy in stage IIIA-N2 non–small-cell Lung Cancer. JNCI: J. Natl. Cancer Inst. 99 (6), 442–450. https://doi.org/10.1093/jnci/djk093 (2007).
Pless, M. et al. Induction chemoradiation in stage IIIA/N2 non-small-cell lung cancer: a phase 3 randomised trial. Lancet 386 (9998), 1049–1056. https://doi.org/10.1016/s0140-6736(15)60294-x (2015).
Xu, Y. P., Li, B., Xu, X. L. & Mao, W. M. Is there a Survival Benefit in patients with Stage IIIA (N2) non-small cell Lung Cancer receiving Neoadjuvant Chemotherapy and/or Radiotherapy Prior to Surgical Resection. Medicine 94 (23), e879. https://doi.org/10.1097/md.0000000000000879 (2015).
Yang, C. F. J. et al. Adding radiation to induction chemotherapy does not improve survival of patients with operable clinical N2 non–small cell lung cancer. J. Thorac. Cardiovasc. Surg. 150 (6), 1484–1493. https://doi.org/10.1016/j.jtcvs.2015.06.062 (2015).
Lee, J. et al. Clinical N-Staging subclassification among stage III-N2 Nsclc patients undergoing trimodality therapy: a good beginning is half the battle. Published online 2023. https://doi.org/10.2139/ssrn.4577979
Kim, H. K. et al. Outcomes of neoadjuvant concurrent chemoradiotherapy followed by surgery for non-small-cell lung cancer with N2 disease. Lung Cancer. 96, 56–62. https://doi.org/10.1016/j.lungcan.2016.03.016 (2016).
Lee, J. et al. Recurrence dynamics after trimodality therapy (neoadjuvant concurrent chemoradiotherapy and surgery) in patients with stage IIIA (N2) lung cancer. Lung Cancer. 115, 89–96. https://doi.org/10.1016/j.lungcan.2017.11.020 (2018).
Travis, W. D. et al. The IASLC Lung Cancer Staging Project: proposals for Coding T Categories for Subsolid Nodules and Assessment of Tumor Size in Part-Solid tumors in the Forthcoming Eighth Edition of the TNM classification of Lung Cancer. J. Thorac. Oncol. 11 (8), 1204–1223. https://doi.org/10.1016/j.jtho.2016.03.025 (2016).
Asamura, H. et al. The International Association for the study of Lung Cancer Lung Cancer Staging Project proposals for the revision of the N descriptors in the Forthcoming 8th Edition of the TNM classification for Lung Cancer. J. Thorac. Oncol. 10 (12), 1675–1684. https://doi.org/10.1097/jto.0000000000000678 (2015).
TAKEDA, S. et al. Results of pulmonary resection following neoadjuvant therapy for locally advanced (IIIA–IIIB) lung cancer. Eur. J. Cardio-Thorac Surg. 30 (1), 184–189. https://doi.org/10.1016/j.ejcts.2006.03.054 (2006).
Hsieh, C. P. et al. Prognostic factors in non-small cell lung cancer patients who received neoadjuvant therapy and curative resection. J. Thorac. Dis. 8 (7), 1477–1486. https://doi.org/10.21037/jtd.2016.05.57 (2016).
van Laar, M., van Amsterdam, W. A. C., van Lindert, A. S. R., de Jong, P. A. & Verhoeff, J. J. C. Prognostic factors for overall survival of stage III non-small cell lung cancer patients on computed tomography: a systematic review and meta-analysis. Radiother Oncol. 151, 152–175. https://doi.org/10.1016/j.radonc.2020.07.030 (2020).
Zhang, K. et al. Tumor size as a predictor for prognosis of patients with surgical IIIA-N2 non-small cell lung cancer after surgery. J. Thorac. Dis. 13, 4114–4124. https://doi.org/10.21037/jtd-21-428 (2021).
Castro, P. P. et al. Importance of tumor size in resectable stage III-N2 non–small cell lung cancer. J. Thorac. Cardiovasc. Surg. 164 (3), 629–636. https://doi.org/10.1016/j.jtcvs.2022.02.015 (2022).
Jett, J. R., Scott, W. J., Rivera, M. P., Sause, W. T. & Physicians, A. C. of C. Guidelines on Treatment of Stage IIIB Non-small Cell Lung Cancer*. Chest. 123 (1), 221S-225S (2003). https://doi.org/10.1378/chest.123.1_suppl.221s
Jett, J. R., Schild, S. E., Keith, R. L., Kesler, K. A. & Physicians, A. C. of C. Treatment of non-small cell lung cancer, stage IIIB: ACCP evidence-based clinical practice guidelines (2nd edition). Chest. 132(3 Suppl), 266S-276S (2007). https://doi.org/10.1378/chest.07-1380
Ramnath, N. et al. Treatment of stage III non-small cell Lung Cancer diagnosis and management of Lung Cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 143 (5), e314S–e340S. https://doi.org/10.1378/chest.12-2360 (2013).
Daly, M. E. et al. Management of Stage III non–small-cell Lung Cancer: ASCO Guideline. J. Clin. Oncol. 40 (12), 1356–1384. https://doi.org/10.1200/jco.21.02528 (2022).
National Comprehensive Cancer Network. Non-Small Cell Lung Cancer (Version 3.2023). Published 2023. Accessed September 1 (2023). https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf
Postmus, P. E. et al. Early and locally advanced non-small-cell lung cancer (NSCLC): ESMO Clinical Practice guidelines for diagnosis, treatment and follow-up †. Ann. Oncol. 28 (suppl_4), iv1–iv21. https://doi.org/10.1093/annonc/mdx222 (2017).
Lee, J. et al. Validation of the IASLC residual tumor classification in patients with stage III-N2 Non-small Cell Lung Cancer undergoing Neoadjuvant Chemoradiotherapy followed by surgery. Ann. Surg. 277, e1355–e1363 (2023).
Rami-Porta, R. et al. The International Association for the study of Lung Cancer Lung Cancer Staging Project: proposals for revision of the TNM Stage groups in the Forthcoming (Ninth) Edition of the TNM classification for Lung Cancer. J. Thorac. Oncol. 19, 1007–1027 (2024).
Kim, H. C. et al. Prognostic factor and clinical outcome in Stage III Non-small Cell Lung Cancer: a study based on real-world Clinical Data in the Korean Population. Cancer Res. Treat. 53 (4), 1033–1041. https://doi.org/10.4143/crt.2020.1350 (2021).
Park, C. K. et al. Korean Real-World Data on patients with unresectable stage III NSCLC treated with Durvalumab after Chemoradiotherapy: PACIFIC-KR. J. Thorac. Oncol. 18 (8), 1042–1054. https://doi.org/10.1016/j.jtho.2023.04.008 (2023).
Remon, J., Soria, J. C., Peters, S. & Committee, E. G. Early and locally advanced non-small-cell lung cancer: an update of the ESMO Clinical Practice Guidelines focusing on diagnosis, staging, systemic and local therapy. Ann. Oncol. 32 (12), 1637–1642. https://doi.org/10.1016/j.annonc.2021.08.1994 (2021).
Woodard, G. A. & Dacic, S. Should the TNM staging of NSCLC evolve beyond anatomical descriptors? J. Thorac. Oncol. 19, 663–665 (2024).
Funding
This work was supported by Future Medicine 20*30 Project of the Samsung Medical Center [#SMO1230021].
Author information
Authors and Affiliations
Contributions
Conceptualization - Jh.L. (Junghee Lee), H.K.K.; Methodology - Jh.L., J.L. (Jin Lee), Y.S.H., E.G., J.C.; Software - Jh.L., J.L, Formal analysis - Jh.L., J.L., Y.S.H.; Investigation - Jh.L., H.K.K.; Data Curation - Jh.L., G.L,; Writing-Original Draft - Jh.L., J.L.,J.C., H.K.K.; Writing-Review & Editing - all authors; Visualization - Jh.L., J.L., Y.S.H., J.C, H.K.K.; Resource - J.P, Y.J.J., S-Y.P, J.H.C., Y.S.C., J.K., Y.M.S., H.K.K., J.Y.P., Supervision - J.C., H.K.K.; Validation - all authors; Project administration - H.K.K.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The study was approved by the Institutional Review Boards of the Samsung Medical Center (IRB file number 2023-09-086). Informed consent was waived as we only used the de-identified data. All methods of this study were performed in accordance with relevant guidelines and regulations.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lee, J., Lee, J., Hong, Y.S. et al. Impact of tumor size by clinical N subclassification and histology in trimodality-treated N2 non-small cell lung cancer. Sci Rep 15, 17195 (2025). https://doi.org/10.1038/s41598-024-82946-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-82946-y