Abstract
Rheumatoid arthritis (RA) is a chronic inflammatory disease associated with subsequent damage to the bone joints with a global incidence rate of 0.5–1%. To combat with RA various commercially available anti-inflammatory drugs like non-steroidal anti-inflammatory drugs (NSAIDs) are commonly practiced; however, their long-term exposure often results in eliciting gastrointestinal and renal toxicity. Given this, the scientific communities are searching for an alternative therapeutic option that seems less injurious and equally efficacious. Equisetum diffusum D. Don, from the Equisetaceae family, is a native pteridophyte species of the sub-Himalayan region of India and has been reported to have a diverse array of pharmacological properties. The decoction and macerated whole plant extract has been used by various tribal communities (like Tagin, Galo, Adi, Kukis, Meiteis, Mulam) throughout the tropical and sub-tropical Asian countries for the treatment of arthritis, bone fractures, back pain, and abrasives. The present research is focused on determining the anti-arthritic potential of E. diffusum whole plant methanol extract (EDME) in adjuvant-induced arthritic rats. Oral administration with 250 and 500 mg/kg b.w. doses of EDME significantly restored paw-edema, arthritic scoring, and normal body weight. EDME treatment also normalized the haematological, biochemical, radiological, and histological status when compared to the arthritic control rats. The methanol extract significantly (p < 0.05) attenuated arthritis progression by downregulating the gene expression of pro-inflammatory mediators, like COX-2, TNF-α, IL-6, and upregulating the expression of anti-inflammatory mediators PPAR-γ, Iκβ in the blood, in a dose-dependent manner. Significantly, EDME also reduced the serum concentration of COX-2, TNF-α, and IL-6 compared to arthritic control rats (p < 0.05). The treatment with EDME also normalized the oxidative stress level in liver by restoring the GSH level, CAT and SOD activities and reducing the elevated MDA level. HPTLC and LC–MS/MS analyses of EDME confirmed the presence of potent polyphenols (chlorogenic acid, 4-hydroxycinnamic acid) and flavonoids (kaempferol), thereby suggesting the anti-arthritic property of the plant. All our findings established the anti-arthritic potential of E. diffusum extract in chronic arthritis model on a strong ground.
Similar content being viewed by others
Introduction
Rheumatoid arthritis (RA) is a complex chronic inflammatory joint disease characterized by synovial inflammation and subsequent damage of the bone-joints associated with progressive destruction of cartilage and bones, gradually leading to chronic disability1. Presently, about 0.5–1% of the world population is affected by the disease1. Although the exact etiology of the disease is unknown, it has been associated with various genetic and external factors2. RA is linked to an abnormally elevated concentration of pro-inflammatory mediators such as cyclooxygenase (COX)-2, tumour necrosis factor (TNF)-α, interleukin (IL)-63,4. Moreover, the nuclear-factor-kappa B (NF-κB), a pro-inflammatory transcription factor stimulates the expression of pro-inflammatory mediators like cyclooxygenase (COX-2), which generates prostanoids and contribute to the pathogenesis of the inflammatory process5. By masking the nuclear localization signal (NLS), the Iκβ (inhibitor of nuclear factor kappa B) protein keeps the NF-κB in inactive state in the cytoplasm and blocks its ability to bind to DNA, resulting in downregulation of the pro-inflammatory mediators6. In RA condition, the NF-κB expression is usually elevated while the Iκβ shows decreased expression7,8. Peroxisome proliferator-activated receptor gamma (PPAR-γ) is another transcription factor that acts as a pleiotropic regulator and can reduce the NF-κB nuclear translocation, by inhibiting Iκβα phosphorylation and thereby attenuating RA progression9. The pathogenesis of RA is also linked with the involvement of reactive oxygen species (ROS) and other free radicals10. Elevated levels of ROS lead to oxidative stress which then activates NF-κB via the phosphorylation and subsequent degradation of Iκβ, ultimately resulting in cellular and tissue damage in joints11. The generation of ROS in arthritic patients seems to decrease the endogenous anti-oxidants levels of reduced glutathione (GSH), catalase (CAT), superoxide dismutase (SOD), thereby triggering the inflammatory responses, particularly in the articular cartilage12. The current drug regime for RA includes administration of commercially available anti-inflammatory medicines, such as non-steroidal anti-inflammatory drugs (NSAIDs) or steroids. However, long-term use of these medications has a series adverse reaction, including gastrointestinal and renal toxicity13,14. To surpass these issues, alternative therapeutic options are often explored, that seem to be less injurious and equally efficacious. Adjuvant-induced arthritis (AIA) is a widely accepted polyarthritis rat model that closely mimics human RA, making it useful for studying RA pathogenesis and treatments15. The introduction of adjuvant, commonly Freund’s complete adjuvant (FCA), triggers the TLR activation on the innate immune cells such as macrophages and dendritic cells, thereby inducing the synthesis of pro-inflammatory cytokines such as TNF-α and IL-6, contributing to chronic inflammation, synovial hyperplasia, and joint erosion15. Therefore, targeting NF-κB and ROS may prove advantageous to mitigate inflammation and RA progression.
Equisetum diffusum D. Don is a member of the Equisetaceae family, commonly known as ‘Himalayan horsetail’, native to the Himalayan mountains16. The plant is a shrub with hollow monomorphic stem and shiny branched rhizome, and popularly referred to as ‘Kurkure Jhar’ in Nepali17,18. The plant can be found at elevations ranging from 1500 to 8500 feet and is widely distributed throughout the tropical and subtropical region of south-east Asian countries including in India18,19. Many species of Equisetum have been utilized as traditional medicine around the world due to the presence of abundant silicic acid16. Various tribal communities around Eastern-Himalayan Biodiversity hotspot of India have been utilizing the plant for its diverse array of health benefits. The tribal communities of Arunachal Pradesh (Tagin, Nyshi, Galo, Adi) and Manipur (like Kukis, Meiteis, Nagas) use the whole plant extract in the treatment of bone fractures, back pain, and abrasives20,21,22. The plant finds its use in the treatment of arthritis, bone fracture and dislocation, backache and muscular pain, kidney ailments; and gonorrhea by various ethnic groups of Sikkim, Jammu-Kashmir, and Jharkhand states of India19,23,24,25. The boiled concoction of the roots and rhizomes of the plant are taken on empty stomach by the local communities of Darjeeling (like Lepchas, Bhutias, Sherpa, Gurung) for the treatment various inflammatory-related complications in urinary tract infections (UTI)26. The whole plant and root juice has also been used by the Nepali, Bhutia, and Lepcha communities of Sikkim for curing chest complaints, liver problems, mouth sores, indigestion, and useful in preventing kidney stones25. In Guangxi, China, the Mulam people apply the ground fresh plant parts to the affected area for its anti-inflammatory effects27. The plant also finds its place in the Vietnamese folk medication for its anti-inflammatory and hypertensive properties28.
Our previous studies on the phytochemical analysis of E. diffusum whole plant methanol extract revealed the presence of tannin, saponin, phenols, and flavonoids along with strong antioxidant activity against the DPPH, ferrous chelation, and ABTS radical29. Other studies have confirmed the anti-microbial and anti-fungal properties of the plant30. The first time gas chromatography-mass spectrometry (GC–MS) analysis of the whole plant methanol extract showed the presence of anti-inflammatory compounds like hexadecanoic acid, methyl ester; 1,2-benzenedicarboxylic acid, bis(2-ethylhexyl) esters; oleic acid, (Z)-, TMS derivative; stigmasta-3,5-dien-7-one and most of them showed good binding scores against key inflammatory mediators such as COX-2, IL-6, and TNF-α in the docking study29. The plant’s anti-inflammatory activity was also validated from our previously conducted in vitro anti-inflammatory (protein denaturation inhibition, hypotonicity-induced, and heat-induced haemolysis) and in vivo anti-inflammatory (carrageenan-induced paw edema) tests29. The detailed toxicity profiling of the methanol extract of the plant on both acute and sub-acute oral toxicity animal models indicates that the concerned methanol extract of the plant shows no significant toxic effects31.
Despite the validation of the anti-inflammatory activity of methanol extract of E. diffusum (EDME), data related to anti-arthritic properties of the plant were not been documented to date. So, the prime objective of the present study was to evaluate the anti-arthritic properties of the whole plant methanol extract of E. diffusum in RA scenario at the molecular level. Moreover, the present study also tried to screen the detailed chemical profiling of bioactives present in whole plant of E. diffusum via high-performance thin layer chromatography (HPTLC) and liquid chromatography-mass spectrometry (LC–MS/MS) techniques.
Results
In vivo evaluation of anti-arthritic properties of E. diffusum whole plant methanol extract (EDME)
Effect of EDME on paw edema and arthritic scoring
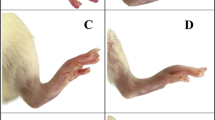
There is a significant increase (p < 0.05) of paw edema in the control arthritic rats compared with the extract-fed rats. Comparing extract-fed rats with the arthritic control rats, paw edema ameliorated significantly (p < 0.001) with EDME treatment at both 250 mg/kg and 500 mg/kg body weight doses (Fig. 1A). The paw circumference (in mm) of rats and the percentage inhibition (%) of paw-edema in various experimental groups were summarised in Supplementary Table S1. At the end of the experiment (day 27), it was found that treatment with both doses of extracts (250 and 500 mg/kg) has shown a 20.65% and 28.63% inhibition of paw-edema, respectively, when compared to the arthritic control group. However, the two extract-fed groups showed significant (p < 0.05) differences in their paw-edema amelioration when compared with each other following Tukey’s multiple comparisons test (Fig. 1A). Figure 1B shows significant higher arthritic scores in arthritic control group. However, treatment with EDME significantly (p ≤ 0.01) reduced the arthritic scores in both the extract-fed groups compared to the arthritic control group. However, no significant difference in the arthritic scores was observed between the two extract-fed rats (Fig. 1B). Arthritic scores of all experimental rats were summarised in Supplementary Table S2. The paw images of the rats of the experimental groups are provided as a Supplementary Fig. S1.
Effect of EDME on different experimental rat groups (n = 6). (A) Inhibition of paw-diameter by methanol extract. (B) Arthritic score of different experimental groups treated with methanol extract. Results are represented as Mean ± S.E.M and analysed by two-way ANOVA following Dunnett’s and Tukey’s multiple comparisons test. The symbol ‘*’ indicates the comparison between the dose groups and arthritic control group and ‘#’ indicates the inter-group comparison between all the experimental groups. α/### indicates p ≤ 0.001; β/## indicates p ≤ 0.01; and γ/# indicates p ≤ 0.05.
Effect of EDME on body and organ weight
The results of body and organ weight of various experimental groups were depicted in Fig. 2. From our results, it was clearly found that the body weight of arthritic control rats decreased steadily (6.56%) with the arthritis progression when compared to vehicle control rats (8.65% increment) (Fig. 2A). On the other hand, treatment with EDME at 500 mg/kg (high dose group) significantly (p < 0.05) restored body weight (no net change) compared to arthritic control rats at day 28 (Supplementary Table S3). When compared with the vehicle control rats, the body weight of both extract-fed rats showed a significant (p < 0.05) difference. However, no significant alterations in the body weight were observed between the extract-fed dose groups (Fig. 2A). The kidney index showed no alternation among the extract-fed groups similar to the arthritic control rats (Fig. 2B). The liver index of the control arthritic rats was found to be significantly (p < 0.05) low compared with vehicle control rats (Fig. 2C). The weight of immune organ (spleen) of arthritic control rats showed significant (p < 0.01) elevation compared to vehicle control rats (Fig. 2D). However, both the extract-fed groups showed non-significant changes in their respective organ indices when compared with each other as well as in contrast to the arthritic control group.
Effect of EDME on body and organ weight on different experimental rat groups (n = 6). (A) Changes in body weight of arthritic rats fed with EDME. Data are represented as Mean ± S.E.M and analysed by two-way ANOVA following Dunnett’s and Tukey’s multiple comparisons test. (B–D) Changes in organ weight (kidney, liver, and spleen) of different experimental groups treated with methanol extract. Data are represented as Mean ± SD and analysed by one-way ANOVA following Dunnett’s post hoc and Tukey’s multiple comparisons test. The symbol ‘*’ indicates the comparison between the dose groups and arthritic control group and ‘#’ indicates the inter-group comparison between all the experimental groups. α/### indicates p ≤ 0.001; β/## indicates p ≤ 0.01; and γ/# indicates p ≤ 0.05; ns indicates p > 0.05.
Effect of EDME on haematological and biochemical parameters
Haematological parameters like total RBC and WBC counts, haemoglobin (Hb) content, and total platelet count were measured and summarised in Fig. 3A–D and Supplementary Table S4. The RBC count of both low dose (7.61 ± 0.264) and high dose (7.65 ± 0.187) groups showed a significant (p < 0.01) increase when compared with the arthritic control group (7.1 ± 0.222) (Fig. 3A). A similar significant (p < 0.01) increment of Hb content was found in both the EDME-fed low dose (13.3 ± 0.548) and high dose (13.5 ± 0.423) groups when compared with the arthritic control group (12.5 ± 0.172) (Fig. 3B). However, the WBC count showed significant normalization in high dose (4.96 ± 0.577; p ≤ 0.01) group than the low dose (5.17 ± 0.516; p < 0.05) group when compared with arthritic control group (6.08 ± 0.640) (Fig. 3C). Similarly, the platelet count showed similar trend with the high dose (686 ± 70.8) group of EDME showed more prominent reduction (p ≤ 0.01) than the low dose (716 ± 59.4) when compared with arthritic control (803 ± 39.1) (Fig. 3D). The vehicle control group showed significant (p ≤ 0.01) changes in all the haematological parameters compared to the arthritic control group. All the biochemical parameters were also depicted in Fig. 3E–H and summarised in Supplementary Table S5. From our results, a significant (p < 0.05) decrease of serum total protein level was found in the arthritic control group (6.30 ± 0.076) which were normalized in both the low dose (6.90 ± 0.493; p < 0.05) and high dose (6.96 ± 0.282; p ≤ 0.01) groups, like the vehicle control group (7.04 ± 0.295; p ≤ 0.01) (Fig. 3E). The albumin levels also showed similar trend and were normalized back to normal on treatment of EDME when compared with arthritic control group (Fig. 3F). For the acute phase protein, it was found that the treatment with plant extracts has normalized the elevated serum ceruloplasmin level in both the low dose (24.5 ± 1.58; p ≤ 0.01) and high dose (22.2 ± 1.25; p ≤ 0.001) groups when compared with the arthritic control rats (28.0 ± 1.64) (Fig. 3G). However, the creatinine level showed no alteration among the experimental rat groups when compared with the arthritic control (Fig. 3H). Similarly, no significant alterations in both the haematological (Fig. 3A–D) and biochemical (Fig. 3E–H) parameters were observed between the extract-fed dose groups following Tukey’s multiple comparisons test.
Effect of EDME on haematological and biochemical parameters of different experimental rat groups (n = 6). (A) Total RBC count, (B) Hb content, (C) total WBC count, and (D) total platelet count, (E) total protein (g/dl), (F) albumin (g/dl), (G) ceruloplasmin (mg/dl), and (H) creatinine (mg/dl) concentration. Analysis was done by one-way ANOVA following Dunnett’s post hoc and Tukey’s multiple comparisons tests, where the symbol ‘*’ indicates the comparison between the dose groups and arthritic control group and ‘#’ indicates the inter-group comparison between all the experimental groups. ***/### indicates p ≤ 0.001, **/## indicates p ≤ 0.01, and */# indicates p ≤ 0.05, ns indicates p > 0.05.
Effect of EDME on radiological analyses
From the radiological image, it was clear that the arthritic control rats developed significant irregular joint spaces, prominent bone erosion, soft-tissue swelling, and bending of appendages (Fig. 4A,E). However, the joint space and appendages of the low dose (Fig. 4B,F) and high dose (Fig. 4C,G) group rats showed evidence of normalcy with lesser bone erosion and bending in contrast to the arthritic control rats. The vehicle control group however showed normal bone structure (Fig. 4D,H).
Radiological imaging of right hind-paw ankle joints of different experimental rat groups. Arrow-marks indicate the degradation and erosion of bone cartilage in experimental animals. Upper panel (A–D) shows the horizontal view and lower panel (E–H) shows the angular view of the same bone joints; where, (A,E) Arthritic control group, (B,F) Low dose group, (C,G) High dose group, and (D,H) Vehicle control group.
Effect of EDME on histopathological analyses
The images of the histological sections of the paw-ankle joints, kidney, and liver of different experimental groups were depicted in Fig. 5. It was clear from Fig. 5A, that the paw joints of arthritic control rats showed irregular cartilage lining along with high infiltration of the immune cells due to severe arthritis progression. The paw ankle joints sections of both the extract-fed groups (Fig. 5B,C), however showed normalization of cartilage lining with lesser infiltrating cells in contrast to control arthritic rat. However, the paw sections of the vehicle control rat (Fig. 5D) showed prominent joint cartilage lining without immune cells infiltration. The kidney sections showed well organized Bowman’s capsule cells (Fig. 5E–H) and the liver sections showed well-organized hepatic lobules with no abnormalities among the experimental groups (Fig. 5I–L).
Histological sections of rat paw (ankle) joints (upper panel); kidney (middle panel); and liver (lower panel) obtained from different experimental groups. (A,E,I) Arthritic control group; (B,F,J) Low dose group; (C,G,K) High dose group; and (D,H,L) Vehicle control group. The 40× image are shown in the inset, while the main images show parts seen under a × 10 magnification. Scale bar of 100 µm for 10× and 25 µm for 40× are used. White arrows in the upper panel represent the immune cell infiltration and the black arrows indicate the cartilage lining in all experimental groups. Following white letters are the labelling of histological structures: vascularized Glomeruli (G); Bowman’s capsule (Bc); Urinary space (Us); Hepatocytes (H); Kupffer cell (K); Portal vein (PV); Central vein (CV).
Effect of EDME on mRNA expression of inflammatory biomarkers
The mRNA expression study of various inflammatory biomarkers is presented in Fig. 6 and summarised in Supplementary Table S6. From our results, a significant elevation in COX-2 expression (p ≤ 0.01) was observed in the arthritic control rats (3.27 ± 0.165-fold) in comparison to the vehicle control group rats. However, treatment with EDME at 250 mg/kg (1.39 ± 0.753-fold) and 500 mg/kg (1.38 ± 0.120-fold) body weights reduced the COX-2 expression as compared to arthritic control group (Fig. 6A). The expression of pro-inflammatory cytokines like TNF-α (11.0 ± 0.195-fold) and IL-6 (5.01 ± 1.24) also showed significant (p ≤ 0.01) upsurge in the arthritic control group compared to vehicle control as shown in Fig. 6B,C. Treatment with EDME significantly (p ≤ 0.01) ameliorated the TNF-α expression in high dose group (3.99 ± 1.74-fold) and reduced the IL-6 expression in both the low dose (1.80 ± 0.713-fold) and high dose group (1.02 ± 0.497-fold). A significant (p ≤ 0.001) decrease in expression of PPAR-γ was observed in the arthritic rats (14.3 ± 1.55%). Treatment with plant extract at 250 mg/kg (20.6 ± 4.00%; p < 0.05) and 500 mg/kg (68.7 ± 0.40%; p ≤ 0.001) significantly elevated the PPAR-γ expression in comparison to arthritic control group (Fig. 6D). A similar result was observed in Iκβ expression where treatment with EDME at 500 mg/kg significantly (53.3 ± 3.65%; p ≤ 0.01) up-regulated the Iκβ expression compared to the arthritic control group (31.8 ± 4.85%) (Fig. 6E). Based on the Tukey’s multiple comparisons test, it was found that both the extract-fed groups showed no significant alterations in the expression of the pro-inflammatory genes (Fig. 6A–C), however, showed significant (p ≤ 0.01) differences in the gene expression of the anti-inflammatory genes (Fig. 6D,E). This suggests a dose-dependent effect of the extract on the gene expression of the anti-inflammatory markers.
Effect of EDME on mRNA expression of various inflammatory mediators in arthritic rats (n = 6). (A) COX-2, cyclo-oxygenase-2; (B) TNF-α, tumor necrosis factor-α; (C) IL-6, interleukin-6; (D) PPAR-γ, peroxisome proliferator-activated receptor-gamma; (E) Iκβ, inhibitor of kappa β. Analysis was done by one-way ANOVA following Dunnett’s post hoc and Tukey’s multiple comparisons tests, where the symbol ‘*’ indicates the comparison between the dose groups and arthritic control group and ‘#’ indicates the inter-group comparison between all the experimental groups. ***/### indicates p ≤ 0.001, **/## indicates p ≤ 0.01, */# indicates p ≤ 0.05, and ns indicates p > 0.05.
Effect of EDME on serum concentration of COX-2, TNF-α, and IL-6
The induction of FCA in rat results in an elevated serum concentration of pro-inflammatory markers like COX-2, TNF-α, and IL-6. The effects of EDME on the serum concentration of these pro-inflammatory markers are presented in Fig. 7A–C and summarised in Supplementary Table S7. The COX-2 levels in the arthritic control group (10.3 ± 0.967 ng/ml) showed significant (p ≤ 0.001) elevation compared to the vehicle control group rats (by a 6.48-fold increase). However, treatment with methanol extract at 250 mg/kg (6.03 ± 0.287 ng/ml; 6.03-fold increase) and 500 mg/kg (5.08 ± 0.505 ng/ml; 5.06-fold increase) was found in comparison to arthritic control group as shown in Fig. 7A. At the same time, an abnormally elevated TNF-α level was observed in the arthritic control group (397 ± 36.5 pg/ml). This elevated level was significantly (p ≤ 0.001) attenuated by treatment with EDME at 250 mg/kg (272 ± 23 pg/ml) and 500 mg/kg (174 ± 32.8 pg/ml) (Fig. 7B). The IL-6 levels also significantly (p ≤ 0.001) elevated in the arthritic control group (98.7 ± 16.20 pg/ml) compared to the vehicle control group (by a 7.54-fold increase). The plant extract, however, significantly (p ≤ 0.001) restored the elevated IL-6 level in both the extract-treated rats to the levels of the vehicle control groups (Fig. 7C). The serum concentration of all the pro-inflammatory markers of both the extract-fed rats showed significant (p < 0.05) differences, when compared with the vehicle control rats (Fig. 7A–C). Similarly, significant (p < 0.05) alterations were also observed in the serum concentration all the pro-inflammatory markers between the extract-fed dose groups, suggesting the dose-dependent effect of the extract in the serum inflammatory mediators.
Effect of EDME on serum inflammatory mediators and oxidative stress in the liver of experimental rats (n = 6). (A) COX-2 level; (B) TNF-α level; (C) IL-6 level; (D) MDA level; (E) GSH level; (F) CAT activity; (G) SOD activity. Analysis was done by One-way ANOVA following Dunnett’s post hoc and Tukey’s multiple comparisons tests, where the ‘*’ indicates the comparison between the dose groups and arthritic control group and ‘#’ indicates the inter-group comparison between all the experimental groups. ***/### indicates p ≤ 0.001, **/## indicates p ≤ 0.01, and */# indicates p ≤ 0.05.
Effect of EDME on oxidative stress in the liver of rats
The ameliorating effects of EDME on the level of antioxidant enzymes in the liver of FCA-induced arthritic rats are presented in Fig. 7D–G and summarised in Supplementary Table S8. The MDA level in the arthritic control rats (45.50 ± 5.04 nmol/g tissue; p ≤ 0.001) was found to be significantly higher compared to vehicle control rats (32.00 ± 5.62 nmol/g tissue). However, upon treatment with EDME a significantly (p ≤ 0.01) reduced MDA level was observed in both the low dose (35.90 ± 4.67) and high dose (33.20 ± 3.53) groups (Fig. 7D). The GSH levels, CAT and SOD activities also normalized significantly (p ≤ 0.01) upon treatment with EDME in both the low and high dose groups compared to the arthritic control rats (Fig. 7E–G). The level of liver antioxidant enzymes (GSH, CAT, and SOD) of the low dose group rats showed significant (p ≤ 0.001) differences in comparison with the vehicle control rats (Fig. 7E–G). Similarly, significant (p ≤ 0.001) alterations were also observed in the antioxidant levels between the extract-fed dose groups (Fig. 7E–G), suggesting the dose-dependent normalization of liver oxidative stress.
HPTLC analysis of EDME
The HPTLC study of EDME detects the presence of two phenolic acids and two flavonoids. The identified phytochemicals have been further validated for quantification and their therapeutic potentials are summarised in Table 1. The HPTLC fingerprint of EDME is depicted as Supplementary Fig. S2.
LC–MS/MS analysis of EDME
The LC–MS/MS analysis of EDME enabled the identification of a total of 69 tentative compounds, out of which 29 were identified in the positive mode and 40 were recognized in the negative mode of ionization. The selected compounds in both modes [positive and negative electrospray ionisation (ESI)] are enlisted along with their chemical class and biological properties in Supplementary Tables S9 and S10. The LC–MS chromatogram for both modes is depicted in Supplementary Fig. S3. Out of 69 compounds identified in both modes (positive and negative ESI), ten (10) and thirteen (13) compounds were found to possess anti-inflammatory and/or anti-arthritic activity in respective modes. Most of the identified compounds belonged to following classes: fatty acids, glycosides, alkaloids, flavonoids, polyphenols, and terpenoids. The anti-inflammatory/anti-arthritic compounds found from the negative ESI mode are chlorogenic acid, theophylline, kaempferol 7-O-glucoside, 4-hydroxycinnamic acid, quinic acid, enprofylline, and 9S,12S,13S-trihydroxy-10E-octadecenoic acid, while paramethasone, lucidenic acid A, and isolimonic acid are identified from the positive ESI mode. Among the tentative compounds identified, chlorogenic acid (0.68 ± 0.05 µg/ml) and kaempferol (1.124 ± 0.19 µg/ml) were identified and quantified from our HPTLC analysis.
Discussion
Rheumatoid arthritis is a chronic systemic inflammatory disease that equally affects adults and the elderly population with a prevalence rate ranging between 0.5 and 1%1. The high prevalence rate of RA in low and middle-income countries are due to their inability to meet daily personal and social needs. The fundamental method for the prevention and management of RA includes the use of NSAIDs; DMARDs; Glucocorticoids. However, adverse drug effects on its long-term exposure have shifted the focus on the use of herbal medication for disease management of RA. For this, the natural products need to be screened in appropriate in vivo arthritic model to validate its efficacy. The FCA-induced Wistar Albino arthritic rat model is widely used in this respect to study the anti-arthritic properties of various compounds as it shows similar resemblance to human RA36,37.
The development of FCA-induced arthritis is biphasic: starting with pronounced swelling in the acute phase, that last for 0 to 10 days due to the release of prostaglandin followed by chronic phase which lasts for 11 to 28th day and is characterised by immune cell infiltration, synovitis, hypertrophy, bone erosion due to the disruption of inflammatory mediators38,39. In our study, treatment with EDME results in significant inhibition of arthritis progression via ameliorating the paw swelling compared to the control arthritic rats. Arthritic scoring is a major index to measure the severity of arthritic progression of FCA-induced arthritis39. Arthritic score of the extract-treated groups also showed significant reduction than FCA-treated rats, thereby supporting the anti-arthritic activities of the extract. RA is directly linked with weight loss (RA-associated cachexia) and studies showed that reduction in body weight is due to progressive swelling upon adjuvant injection which hinders absorption via the gut. However, treatment with anti-inflammatory medicines improves absorption40,41. In our study, treatment with EDME showed normal weight gain in the arthritic rats. Splenomegaly, splenitis, and lymphadenopathy are indicators of a hyper-functional immune conditions, like RA42. In our study the arthritic rats showed significant hyper-functioning of spleen compared to vehicle control rats, however, the hyper-functioning of immune organ was suppressed upon treatment with extracts.
In the control arthritic rats, both the RBC count as well as the Hb content decreased in our experimental results in contrast to vehicle control rats. This may be attributed to the disruption of the bone marrow along with the abnormal iron storage combined with the inability of bone marrow to react to anaemia37,43. However, both the RBC count and Hb content normalized upon treatment with EDME in the experimental dose groups. The significant elevation in WBC count observed in the control arthritic rats are mainly due to the increased immunogenic response against the invading antigens38. However, treatment with EDME significantly normalized the elevated leukocyte count in the extract-treated rats. The elevated platelet count in the arthritic control rats has been significantly counteracted by the treatment with EDME, thereby justifying its efficacy in arthritic conditions. Our haematological results are found to be similar to our previously published research work, where the normalization of haematological parameters was found in the dose groups, justifying the immunomodulatory effects of D. quercifolia rhizome methanol extract37. From our biochemical results, it was observed that the total serum protein and albumin level significantly decreased in the arthritic control rats indicating the onset of arthritis. This is mainly due to the involvement of several inflammatory mediators such as prostaglandins, bradykinin, histamine that elevates the vascular permeability, thereby lowering the total protein and albumin levels38. However, treatment with EDME significantly restored the total protein and albumin level in the extract-fed groups when compared to the arthritic control rats, thereby indicating anti-inflammatory effect of EDME. Ceruloplasmin is a major acute-phase protein that elevates during arthritic conditions37. This serum protein is mostly generated in the liver and released into the bloodstream following any tissue injury38. In our study the ceruloplasmin level in the control arthritic rats was significantly elevated indicating tissue injury during the chronic arthritis progression. However, EDME treatment resulted in a significant normalization in both the low and high dose group rats, demonstrating its effect on tissue repair. The serum creatinine level in our study, however, showed no significant alteration among the experimental groups, indicating normal kidney function.
The conventional radiography plays a vital role in assessing the status of RA and is still a reliable practice in indicating the severity of RA diagnosis44. The hallmark of RA includes major irregular joint space, prominent bone erosion, and soft-tissue swelling45. In our study, the radiographic patterns of the hind limb of arthritic control rats developed narrowing of joint space along with swelling of soft tissue, indicating the disease severity. Treatment with EDME showed restoration of bone erosion in both extract-fed groups with improved radiographic patterns. These radiographic alterations were also supported by histopathological sections of right paw ankle joints of the arthritic rats (Fig. 5A). The paw ankle-joint of the arthritic control rats showed irregular cartilage lining along with excessive infiltration of immune cells within the articular cartilage linings. However, treatment with EDME minimized the infiltrations of cells in both extract-fed groups, in contrast to the control arthritic rats. However, the histological sections of kidney and liver showed no significant structural changes among the experimental groups, thereby negating the toxic side-effect of EDME treatment.
In adjuvant-induced arthritic condition, COX-2 serves as pro-inflammatory immune modulator responsible for the increased production of PGE2 that results in vasodilation and recruitment of neutrophils in inflammatory arthritic joints46,47. Cytokines are key mediators of inflammatory arthritis, among which TNF-α and IL-6 are pro-inflammatory cytokines released from monocytes and macrophages that are responsible for the infiltration of immune cells in the inflamed regions and thereby initiate cartilage degradation by promoting matrix metalloproteases (MMP)-2 production8,47. PPAR-γ is a transcription factor that serves as a pleiotropic regulator controlling anti-inflammatory activity, antioxidant levels, and phagocyte-mediated cleaning functions by down-regulating the levels of pro-inflammatory mediators like COX-2, TNF-α, ΙL-6, and MMP-2 in PPAR-γ-overexpressing C2C12 skeletal muscle cells9,48. Previous studies on adjuvant-induced arthritis model have also shown that, activation of PPAR-γ expression is linked with the anti-inflammatory activity and reduction of disease severity9,49. Studies by Zhang et al. has reported that up-regulation of PPAR-γ expression can reduce NF-κB nuclear translocation, by inhibiting Iκβα phosphorylation and thereby attenuating RA progression9. NF-κB is another major transcription factor that regulates the expression of pro-inflammatory mediators such as cytokines, chemokines, inducible enzymes, and growth factors50. It is found in inactivate state due to its association with inhibitor protein Iκβ. From our current study we found that oral administration of E. diffusum extract in arthritic rats considerably down-regulated the mRNA expression of pro-inflammatory enzyme (COX-2), pro-inflammatory cytokines (TNF-α and IL-6), however, notably up-regulated the mRNA expression of anti-inflammatory mediators (PPAR-γ and Iκβ) in a dose-dependent manner, suggesting the immunomodulatory activity of the plant. Our gene expression studies also found to be similar to studies involving crude Aloe vera leaf gel extracts where the expression of COX-2 and TNF-α were found to be downregulated in adjuvant-induced Wistar rats47. In a recent study, we have demonstrated the anti-inflammatory property of E. diffusum, where the whole plant methanol extract was found to be significantly inhibiting paw-edema in carrageenan-induced acute inflammatory model and the GC–MS identified phyto-compounds also showed good inhibition against key inflammatory mediators (COX-2, TNF-α, and IL-6) in molecular docking study29. In the current study we also measured the serum pro-inflammatory mediators (COX-2, TNF-α, and IL-6) and it was found that treatment with extract has considerably down-regulated the levels of these inflammatory mediators compared to the arthritic control rats in a dose-dependent manner, thereby corroborating our previous study29.
Reactive oxygen species (ROS) are apparently linked with a wide range of chronic inflammatory diseases including RA10. In the RA state, various types of ROS, including hydrogen peroxide, superoxide anions, and hydroxyl radicals, are produced due to the hyperactivation of immune cells such as macrophages and neutrophils51. The activation of NF-κB also enhances the synthesis of enzymes such as inducible nitric oxide synthase (iNOS) and NADPH oxidase, leading to increased generation of ROS. The ROS-NF-κB cycle induces a pro-inflammatory state, exacerbating synovial inflammation and joint degradation associated with rheumatoid arthritis51. In our study, elevated liver lipid peroxidation level was observed in the arthritic rats compared to the vehicle control rats. However, treatment with EDME significantly suppressed lipid peroxidation by restoring the MDA concentration in the arthritic rats. GSH is a low molecular weight thiol that serves as the first line of defence against oxidative stress related damage generated from free radicals and organic peroxides52. CAT and SOD are common anti-oxidants that plays a vital role in protecting the cells from oxidative stress by detoxification of hydrogen peroxide and superoxide anion, respectively52. In adjuvant-induced arthritis, generation of oxidative stress results in reduction of these endogenous antioxidants. However, post-immunisation with FCA, the levels of these antioxidants were markedly restored upon treatment with both doses of E. diffusum extracts, thereby suggesting that modulation of gene expression and arthritic parameters are probably due to the scavenging of free radicals by the plant extract. But, the 500 mg/kg b.w. dose showed more prominent restoration of endogenous antioxidants when compared to the 250 mg/kg b.w. dose, thereby suggesting the dose-dependent role of the plant extract.
The presence of various bioactive plant metabolites plays effective roles against various oxidative stress-related disorders such as arthritis, diabetes, and Parkinson’s disease8. So, for the analysis of these metabolites high-performance liquid chromatography (HPTLC) and LC–MS/MS separation techniques are widely practiced. HPTLC is a versatile and cost-effective separation techniques used for phytochemical and biomedical analysis53. The HPTLC analysis of E. diffusum extracts showed the presence of two important phenolics (chlorogenic acid, caffeic acid) and two flavonoids (kaempferol, apigenin). The therapeutic potential of chlorogenic acid has been reported to have antioxidant, anti-inflammatory, and anti-arthritic properties32. Recent studies have also shown the anti-inflammatory role of chlorogenic acid in rheumatoid arthritis model, in which it attenuates NF-κB expression, thereby inhibiting downstream expression of TNF-α54. Caffeic acid is another important polyphenol reported to have anti-inflammatory, anti-viral, anti-cancer, anti-oxidant, anti-microbial, anti-tumour, and immune regulatory properties33. Numerous biological characteristics of apigenin include anti-inflammatory, anti-cancer, and anti-oxidant effects34. Kaempferol is a naturally occurring flavonoid that possesses a variety of pharmacological properties, like anti-inflammatory, anti-oxidant, and anti-estrogenic properties35. Our HPTLC study corroborated the LC–MS analysis. The LC–MS technique is an efficient and useful tool for precise identification of secondary metabolites found in plant material55. The LC–MS chromatogram of the whole plant methanol extract of E. diffusum revealed the presence of pharmacologically important alkaloids (including theophylline, enprofylline); polyphenols (such as chlorogenic acid, 4-hydroxycinnamic acid); flavonoids (like kaempferol 7-O-glucoside); terpenoids (like lucidenic acid A, acetylvalerenolic acid). Studies in adjuvant-induced arthritic rats showed a 45 mg/kg/day dose of theophylline can significantly reduce arthritis index, decrease ankle joint diameter, decrease hind-paw volume, reduce inflammatory cellular infiltration in the synovium of the ankle joints and pannus formation56. Furthermore, studies in rheumatoid arthritis model suggests that p-coumaric acid (one of three isomers of hydroxycinnamic acid) can significantly ameliorates inflammation and suppress cartilage and bone erosion by downregulating both protein and gene expression of RANKL, TNF-α, IL-6, NF-κB levels in serum and ankle joint homogenate of arthritic rat57. Two compounds (chlorogenic acid and kaempferol derivatives) have been identified and quantified from our HPTLC fingerprints were also detected in our LC–MS chromatogram. Presence of these important metabolites may justify the pharmacological attributes of the plant. To the best of our knowledge, it is the first report on the detailed phytochemical analysis of E. diffusum whole plant methanol extract and the anti-arthritic effect via modulating expression of key inflammatory biomarkers in adjuvant-induced arthritic animal model.
Methods
Drug and chemicals
Freund’s complete adjuvant (FCA) containing 10 mg/mL heat-killed Mycobacterium tuberculosis was obtained from Sigma-Aldrich, USA; Carboxymethylcellulose sodium salt (CMC) was procured from HiMedia Laboratories, India. Total RNA was extracted using TRIzol LS reagent from Invitrogen, USA. cDNA was synthesized using a reverse transcriptase kit procured from Thermo Fischer (USA), and HiMedia Laboratories (India). FastStart Universal SYBR Green master dye was obtained from Roche Diagnostics, USA. For measurement of RNA yield, a Nanodrop spectrophotometer (SPECTROstar Nano, BMG LABTECH, Germany) was used. For the RT-Q-PCR study, Roche Lightcycler 96 (Roche, Switzerland) and Bio-Rad CFX96 Touch Real-Time Detection system (Bio-Rad, USA) were used. Rat COX-2 ELISA kit was purchased from MyBiosource Inc., San Diego, USA. Rat TNF-α and IL-6 ELISA kit was procured from RayBiotech, USA. All chemicals utilized in the experiments were of molecular biology grade, obtained from Sigma-Aldrich (USA), and HiMedia (India).
Plant material and preparation of whole plant methanol extract
Whole plant of Equisetum diffusum were collected from the sub-Himalayan Teari regions adjacent to Darjeeling foothills of West Bengal in November–December, 2022–2023. Dr. R. K. Gupta (Scientist-E) of Central National Herbarium, Botanical Survey of India (BSI), India identified the collected plant samples through a deposition of a voucher specimen (NBU/SS-002) (see Supplementary Fig. S4). The methanol extract of whole plant was prepared following previously published protocols and the final percentage-yield was found to be around 4.8% (w/w)29. The finally obtained concentrated E. diffusum whole plant methanol extract (EDME) was kept in an airtight container at 4 °C until future uses.
Experimental animals
Male Wistar albino rats (8–12 weeks old; 150 ± 10 g) were used for evaluating the anti-arthritic activity of EDME. The rats were obtained from authorized animal dealers (M/s Chakraborty Enterprise, Kolkata, India; Regd. No. 1443/PO/Bt/s/11/CPCSEA) and were kept in polypropylene cages (with 4 rats per cage) at standard laboratory conditions: temperature (25 ± 3 °C); relative humidity (60–70%); 12-h:12-h dark–light cycle; supplemented with standard feed and filtered water ad libitum. Before the commencement of experiments, all the animals were acclimatized for 7 days in the departmental animal house.
Dosing, experimental design, and arthritis induction
Previously acclimatized rats were randomly divided into 4 groups of 6 rats each (n = 6). Oral administration of dose was done using oral gavage. Group I (Arthritic control): Arthritic rats orally administered only with 0.9% saline water; Group II (Low dose): Arthritic rats orally administered with EDME (250 mg/kg b.w.); Group III (High dose): Arthritic rats orally administered with EDME (500 mg/kg b.w.); Group IV (Vehicle control): Rats orally administered with 0.5% carboxymethylcellulose (CMC). All the animal groups, except vehicle control group, were injected with Freund’s Complete Adjuvant (FCA, 0.1 ml) into the right hind paws (sub-plantar region) at day 0 followed by a booster dose at day 14 for arthritis induction37,47. The oral feeding-dose was selected based on previously published acute oral toxicity tests29. The EDME doses were administered once daily from day 0 till the end of experimental tenure (day 28).
Evaluation of arthritic progression
The degree of arthritis progression was evaluated by measuring paw edema using vernier calliper following standard published protocols37. To calculate the diameter of paw-edema (mm), the measurement was recorded at two different planes using the following formula: 2π [√ (A2 + B2)/2], where ‘A’ and ‘B’ were measurements of paw-edema at two different planes (ipsilateral and contralateral planes). To minimize the error rate, the mean values of three measurements were taken for each rat. The paw edema was first measured at day 0 followed by intervals of 2 days till the end of experimental tenure (day 28). The percentage-inhibition of paw edema was recorded using the following formula: Percentage inhibition of paw edema = [1 − {(Cday 27 − Cday 0) T / (Cday 27 − Cday 0) AC}] × 100, where ‘(Cday 27 − Cday 0) T’ is the difference in the size of paw edema from day 0 and day 27 in extract-treated groups and ‘(Cday 27 − Cday 0) AC’ is the difference in the paw edema in arthritic control rats58. Arthritic scoring was performed for evaluating the clinical severity of arthritic progression in the form of paw inflammation, swelling, redness and were scored from 0 to 4. A score of ‘0’ means no swelling; ‘1’ means swelling of one toe joints; ‘2’ means swelling of toes and toe joints; ‘3’ means swelling of ankle joints; and ‘4’ means swelling of entire paw, and were given on day 1, 7, 14, 21, and 28 respectively39,59.
Body weight of individual rats was measured on weekly basis at day 0, 7, 14, 21, and 28. To determine the percentage weight change, following formula was used: Percentage (%) change = [(Weight on Day 28 − Weight on Day 0)/ (Weight on Day 0)] × 100. At the end of the experimental tenure, all animals of each group were anesthetized using sodium pentobarbital (60 mg/kg; i.p.) and euthanized by cervical dislocation. Immediately after sacrifice, the spleen, kidney, and liver were dissected out, weighed and their respective organ index were calculated following previously published protocols60,61.
Haematological and biochemical analysis
Blood samples were drawn through the cardiac puncture into EDTA-coated vials and different haematological parameters (total RBC and WBC counts, Hb content, and total platelet count) were measured using an automated haematology analyser (Sysmex XN-1000, Mumbai, India) following standard protocol37. For biochemical analysis, serum was collected by centrifuging blood samples at 5000 rpm for 10 min. Total protein, albumin, and creatinine were measured using Coral Kits (Coral clinical systems, India) following standard protocols. Serum ceruloplasmin was determined following p-phenylenediamine oxidase activity62.
Radiological parameters
The radiological imaging of right paw ankle joint was performed using a high-resolution digital X-ray machine (Allengers 325/625, Mumbai, India). The X-ray imaging was done blind-folded at 50 mA, 50 kV peak, and with 3 s exposure time. The original X-ray images obtained were manually cropped and were analysed45.
Histopathological study
The histopathological study was performed following previously published protocols37. Briefly, the paw ankle joints were surgically removed and fixed in 10% formalin, followed by decalcification in a solution of 3% HCl. The kidney and liver samples were also collected after sacrifice and fixed in 4% formalin. After fixation, the tissues were subjected to dehydration using serial gradations of ethanol, followed by embedding in paraffin wax. Tissues were then sectioned longitudinally (for ankle joint) and transversely (for kidney and liver) at 5 μm thickness, followed by staining with haematoxylin and eosin. After that, sections were analysed by microscopic examinations using a light microscope (Nikon Eclipse E200, Nikon, Tokyo, Japan) with 10× and 40× objectives, to which a scale bar (100 µm and 25 µm) was attached respectively37.
Reverse transcriptase real-time polymerase chain reaction (RT-Q-PCR)
The animals were sacrificed on day 28 and whole blood samples was placed in an EDTA-coated tubes. The total RNA was then extracted using TRIzol LS reagent according to the manufacturer’s protocol and the yield was assessed using a Nanodrop spectrophotometer (SPECTROstar Nano, BMG LABTECH, Germany). The total RNA from animal of each group were pooled together and cDNA was synthesised using reverse transcriptase kit (Thermo Fischer, USA; Hi-cDNA Synthesis kit, India) following manufacturer’s protocol. The prepared cDNAs were used for amplification and quantification of COX-2, TNF-α, PPAR-γ, Iκβ genes using FastStart Universal SYBR Green master dye (Roche Diagnostics, USA) in Roche Lightcycler 96 (Roche, Switzerland). For IL-6 gene, the amplification and quantification were done in Bio-Rad CFX96 Touch Real Time Detection system (Bio-Rad, USA). Briefly, cDNA (5 μl); forward and reverse primers (0.6 μl each); nuclease-free water (3.8 μl); and SYBR Green master dye (10 μl) were added in MicroAmpTM optimal 8-tube microplate strips (Applied Biosystems, ThermoFisher, USA). The microplates were then subjected to 45 cycles of denaturation at 95 °C, annealing at 55 °C (for COX-2, TNF-α, IL-6, Iκβ)/57 °C (for PPAR-γ), and extension at 72 °C in a thermal cycler. The GAPDH was included as a reference gene for estimating the relative expression of the genes, which was determined by using 2−ΔΔCT method47. In case of the pro-inflammatory marker genes (COX-2, TNF-α and IL-6), their expression levels in the vehicle control group were considered as one-fold, while in case of anti-inflammatory marker genes (PPAR-γ and Iκβ), their expression levels in the vehicle control group were depicted as 100%8. The primers used in our study were designed using previously published articles and are enlisted in Supplementary Table S11.
Estimation of serum COX-2, TNF-α, and IL-6 levels
The serum COX-2 levels were measured using an ELISA kit as per manufacturer’s protocols (MyBiosource, Inc., San Diego, USA). Furthermore, for the estimation of pro-inflammatory cytokines (TNF-α and IL-6), ELISA assay was performed using commercial kit protocols (Ray Biotech, USA). The optical density was measured at 450 nm wavelength using a plate reader (Bio-Rad, USA).
Estimation of oxidative stress biomarkers in liver tissues
For the measurement of oxidative stress in the liver, 10% liver homogenate was prepared following previously published protocol60. The resultant liver homogenate obtained was used for the detection of the following oxidative stress markers. To determine liver peroxidation, the level of liver Malondialdehyde (MDA) content was assayed following the standard protocol63. The absorbance of the clear resultant supernatant was measured at 535 nm against a blank reference. The level of reduced glutathione (GSH) content was determined following the standard protocol64. The absorbance of the resultant yellow colour mixture was immediately recorded at 412 nm against a blank reference. Catalase (CAT) activities were determined following standard established protocol65. Changes in absorbance of the resultant solution were determined after 1 min at 240 nm. One unit of CAT activity was equivalent to the change in the absorbance of 0.01 unit/min. Superoxide dismutase (SOD) activities were determined following standard protocol with minor modifications66. The activity was measured spectrophotometrically by phenazine methosulfonate (PMS)-nitroblue tetrazolium (NBT) assay and expressed as unit/gm tissue.
HPTLC analysis of EDME
The HPTLC analysis of EDME was performed following previously published method67. The plate was grown employing a pre-saturated mobile phase of chloroform: ethyl acetate: formic acid (5:4:1 v/v/v) for antioxidants up to a length of 90 mm at 25 ± 5 °C in a Twin Trough Chamber (CAMAG). After drying of the plates, chromatograms were obtained using a TLC UV Cabinet 4 (CAMAG) equipped with a TLC scanner 4 operated by VisionCATS software (CAMAG; slit width 6 × 0.45 nm) at various wavelengths, including 254 nm, 366 nm, and 416 nm. The quantification of phytochemicals presented in the sample EDME was done using the linear equation of regression data of the respective standards (Supplementary Table S12)68.
LC–MS/MS analysis of EDME
The EDME was subjected to qualitative analysis of its metabolites using HR-LCMS (HRLCMS-QTOF-Agilent Technologies, USA) carried out at Sophisticated Analytical Instrument Facility (SAIF) of Indian Institute of Technology, Bombay (IIT Bombay), India. Column type used was ZORBAX Eclipse Plus-C18, 5 microns with 150 mm length and 2.1 mm diameter. For the mobile phase a combination of ‘Solvent A’ (0.1% formic acid in Milli-Q water) and ‘Solvent B’ (100% acetonitrile) were used at a flow rate of 0.3 ml/min and injection volume 5 µl. The gradient of the mobile phase started with 95% of ‘A’: 5% of ‘B’, changing to 0% of ‘A’: 100% of ‘B’ at 25 min, and finally returned to initial composition 95% of ‘A’: 5% of ‘B’ in 1 min and which was maintained at same composition for 5 min. The MS analysis was carried out in both positive (+ ve) and negative (−ve) ESI modes. The settings for the MS segment were set at: a gas flow rate of 13 l/min at 250 °C temperature; a sheath gas flow rate of 11 l/min at 300 °C temperature; and nebulizer gas pressure at 35 psig. For the data acquisition the Agilent Mass Hunter Qualitative Analysis B.06 software was used and set at MS minimum range of 126 (m/z) and a maximum of 1200 (m/z) with a MS scan rate of 1 spectra/s for identifying, profiling, and characterizing the metabolites present in the extract55,69.
Statistical analyses
Quantitative data concerning the body weight, organ weight, and paw diameter measurement was expressed as mean ± standard error mean (SEM). For the remaining assays, all data were expressed as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) or two-way ANOVA was performed following the post hoc analysis with Dunnett’s multiple comparisons test. Inter-group variations between more than two groups were measured using ANOVA followed by Tukey’s multiple comparisons test. For statistical difference values of p ≤ 0.05 were taken as standard. All the statistical analyses, graphs generation were performed using GraphPad Prism Version 7.00 (San Diego, United States of America).
Conclusion
The present study was designed to assess the effectiveness of the whole plant methanol extract of Equisetum diffusum in the amelioration of rheumatoid arthritis. From the study it can be inferred that the methanol extract of E. diffusum significantly restored the symptoms of RA such as paw edema, arthritic score, and body weight loss. Treatment with extract also improved the haematological, biochemical, radiological, and histopathological parameters without exhibiting any toxicological signs. The extract treatment also mitigated the progression of arthritis by reversing articular damage and synovitis possibly through down-regulating the gene and protein expression of pro-inflammatory mediators, like COX-2, TNF-α, and IL-6, and upregulating the gene expression of PPAR-γ and Iκβ in a dose-dependent manner. Our data also indicated that EDME upregulated PPAR-γ and Iκβ expression and therefore may modulate NF-κB functions in regulating downstream immune-regulatory markers. The modulation of the gene and protein expression can also be a result of scavenging of free radicals by the treatment of plant extract. The HPTLC and LC–MS/MS analysis of plant extract confirmed the presence of polyphenols and flavonoids such as chlorogenic acid, 4-hydroxycinnamic acid, kaempferol, suggesting the anti-arthritic activity of the plant. Hence, the current study validated the scientific basis of the use of the plant extract in folklore medications and will also serve as a reliable documentation regarding the use of extract in the treatment of RA. Our current research further points to the necessity of isolating and fractionating possible bioactives responsible for the anti-arthritic properties of the plant. In order to attenuate RA, these bioactives need to be assessed at the cellular and molecular level related to RA.
Data availability
All the materials will be available for research purposes if requested to the corresponding author.
References
Smolen, J. S., Aletaha, D. & McInnes, I. B. Rheumatoid arthritis. The Lancet 388, 2023–2038 (2016).
Shah, A. & St. Clair, E. W. Rheumatoid arthritis. In Harrison’s Principles of Internal Medicine (McGraw Hill, 2018).
Fattahi, M. J. & Mirshafiey, A. Prostaglandins and rheumatoid arthritis. Arthritis 2012, 1–7 (2012).
Lawrence, T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb. Perspect. Biol. 1, a001651 (2009).
Pahl, H. L. Activators and target genes of Rel/NF-κB transcription factors. Oncogene 18, 6853–6866 (1999).
Jacobs, M. D. & Harrison, S. C. Structure of an IκBα/NF-κB complex. Cell 95, 749–758 (1998).
Miller, S. C. et al. Identification of known drugs that act as inhibitors of NF-κB signaling and their mechanism of action. Biochem. Pharmacol. 79, 1272–1280 (2010).
Saleem, A., Saleem, M., Akhtar, M. F., Shahzad, M. & Jahan, S. Moringa rivae leaf extracts attenuate complete Freund’s adjuvant-induced arthritis in Wistar rats via modulation of inflammatory and oxidative stress biomarkers. Inflammopharmacology 28, 139–151 (2020).
Zhang, L. et al. Ginsenoside Rg1 attenuates adjuvant-induced arthritis in rats via modulation of PPAR-γ/NF-κB signal pathway. Oncotarget 8, 55384–55393 (2017).
Afonso, V., Champy, R., Mitrovic, D., Collin, P. & Lomri, A. Reactive oxygen species and superoxide dismutases: Role in joint diseases. Jt. Bone Spine 74, 324–329 (2007).
Sies, H., Berndt, C. & Jones, D. P. Oxidative stress. Annu. Rev. Biochem. 86, 715–748 (2024).
Alavala, S. et al. Anti-inflammatory effect of stevioside abates Freund’s complete adjuvant (FCA)-induced adjuvant arthritis in rats. Inflammopharmacology 28, 1579–1597 (2020).
Ofman, J. J., Badamgarav, E., Henning, J. M., Knight, K. & Laine, L. Utilization of nonsteroidal anti-inflammatory drugs and antisecretory agents: A managed care claims analysis. Am. J. Med. 116, 835–842 (2004).
Zarghi, A. & Arfaei, S. Selective COX-2 inhibitors: A review of their structure–activity relationships. Iran. J. Pharm. Res. 10, 1 (2011).
Donaldson, L. F. & Chillingworth, N. L. Arthritis model, adjuvant-induced arthritis. In Encyclopedia of Pain (eds Schmidt, R. F. & Willis, W. D.) 111–115 (Springer, 2007).
Takuli, P., Khulbe, K., Kumar, P. & Pant, C. Chemical composition of essential oil of Equisetum diffusum: A noble source of phytol. Int. J. Pharm. Sci. Res. 11, 5572–5578 (2020).
Kunwar, R. M., Shrestha, K. P. & Bussmann, R. W. Traditional herbal medicine in Far-west Nepal: A pharmacological appraisal. J. Ethnobiol. Ethnomed. 6, 1–18 (2010).
Singh, B., Singh, V. N., Phukan, S. J., Sinha, B. K. & Borthakur, S. K. Contribution to the pteridophytic flora of India: Nokrek biosphere reserve, Meghalaya. J. Threat Taxa 4, 2277–2294 (2012).
Singh, B. P. & Upadhyay, R. Medicinal pteridophytes of Madhya Pradesh. J. Med. Plants Stud. 2, 65–68 (2014).
Murtem, G. & Chaudhry, P. An ethnobotanical study of medicinal plants used by the tribes in Upper Subansiri district of Arunachal Pradesh, India. Am. J. Ethnomed. 3, 35–49 (2016).
Jeyaprakash, K., Lego, Y. J., Payum, T., Rathinavel, S. & Jayakumar, K. Diversity of medicinal plants used by Adi community in and around area of D’Ering wildlife. World Sci. News 65, 135–159 (2017).
Yumkham, S. D. & Singh, P. K. Less known ferns and fern-allies of Manipur with ethnobotanic uses. Indian J. Trad. Knowl. 10, 287–291 (2011).
Sureshkumar, J. et al. A review on ethnomedicinally important pteridophytes of India. J. Ethnopharmacol. 219, 269–287 (2018).
Wani, M. H., Shah, M. Y. & Naqshi, A. R. Medicinal ferns of Kashmir, India. Int. J. Bioassays 5, 4677–4685 (2016).
Panda, A. K. Medicinal plants use and primary health care in Sikkim. Int. J. Ayurv. Herb. Med. 2, 253–259 (2012).
Srivastava, S. K. & Rao, J. A. V. P. A study of the ferns used as traditional medicine by indigenous people in Darjeeling, West Bengal, India. J. Appl. Sci. Comput. 6, 1409–1413 (2019).
Hu, R., Lin, C., Xu, W., Liu, Y. & Long, C. Ethnobotanical study on medicinal plants used by Mulam people in Guangxi, China. J. Ethnobiol. Ethnomed. 16, 1 (2020).
Nguyen, B.-L.T. et al. HPTLC fingerprinting and cytotoxicity of secondary metabolites of Equisetum diffusum D. Don extracts. Int. J. Plant Anim. Environ. Sci. 11, 596–613 (2021).
Sarkar, S., Modak, D., Haydar, M. S., Georrge, J. J. & Bhattacharjee, S. Exploring the ameliorative role of Equisetum diffusum D. Don whole-plant methanolic-extract in acute inflammation and molecular docking analysis of GC-MS-identified phytocompounds with few prominent inflammatory markers/cytokines for inspecting the potent. Pharmacogn. Res. 16, 82–97 (2023).
Mir, A. M., Ashraf, M. W. & Mir, B. A. Antimicrobial and antifungal and phytochemical analysis of various extracts of Equisetum diffusum. Trends Biomater. Artif. Organs 35, 186–189 (2021).
Sarkar, S. et al. In silico, in vitro, and in vivo acute and sub-acute toxicity profiling of whole plant methanol extract of Equisetum diffusum D. Don from the sub-Himalayan West Bengal, India, having ethnobotanical uses. BMC Complement Med. Ther. 24, 324 (2024).
Miao, M. & Xiang, L. Pharmacological action and potential targets of chlorogenic acid. Adv. Pharmacol. 87, 1 (2020).
Al-Ostoot, F. H. et al. Molecular docking and synthesis of caffeic acid analogous and its anti-inflammatory, analgesic and ulcerogenic studies. Bioorg. Med. Chem. Lett. 33, 127743 (2021).
Ginwala, R., Bhavsar, R., Chigbu, D. G. I., Jain, P. & Khan, Z. K. Potential role of flavonoids in treating chronic inflammatory diseases with a special focus on the anti-inflammatory activity of apigenin. Antioxidants 8, 1–28 (2019).
Hofer, S. et al. Pharmacological targets of kaempferol within inflammatory pathways—A hint towards the central role of tryptophan metabolism. Antioxidants 9, 1–19 (2020).
Bendele, A. et al. Animal models of arthritis: Relevance to human disease. Toxicol. Pathol. 27, 1 (1999).
Modak, D., Paul, S., Sarkar, S., Thakur, S. & Bhattacharjee, S. Validating potent anti-inflammatory and anti-rheumatoid properties of Drynaria quercifolia rhizome methanolic extract through in vitro, in vivo, in silico and GC-MS-based profiling. BMC Complement Med. Ther. 21, 1–20 (2021).
Ekambaram, S., Perumal, S. S. & Subramanian, V. Evaluation of antiarthritic activity of Strychnos potatorum Linn seeds in Freund’s adjuvant induced arthritic rat model. BMC Complement Altern. Med. 10, 1–9 (2010).
Choudhary, M., Kumar, V., Gupta, P. & Singh, S. Investigation of antiarthritic potential of Plumeria alba L. leaves in acute and chronic models of arthritis. Biomed. Res. Int. 2014, 1–12 (2014).
Yoshikawa, T., Tanaka, H. & Kondo, M. The increase of lipid peroxidation in rat adjuvant arthritis and its inhibition by superoxide dismutase. Biochem. Med. 33, 320–326 (1985).
Roubenoff, R. et al. Rheumatoid cachexia: Cytokine-driven hypermetabolism accompanying reduced body cell mass in chronic inflammation. J. Clin. Investig. 93, 2379–2386 (1994).
Issekutz, A. C. & Sapru, K. Modulation of adjuvant arthritis in the rat by 2-methoxyestradiol: An effect independent of an anti-angiogenic action. Int. Immunopharmacol. 8, 708–716 (2008).
Mowat, A. G. Hematologic abnormalities in rheumatoid arthritis. Semin. Arthritis Rheum. 1, 195–219 (1972).
Llopis, E., Kroon, H. M., Acosta, J. & Bloem, J. L. Conventional radiology in rheumatoid arthritis. Radiol. Clin. N. Am. 55, 917–941 (2017).
Khader, S. Z. A., Ahmed, S. S. Z., Arunachalam, T. & Radhakrishnan, K. Therapeutic effect of Parmotrema tinctorum against complete Freund’s adjuvant-induced arthritis in rats and identification of novel isophthalic ester derivative. Biomed. Pharmacother. 112, 108646 (2019).
Honda, T., Segi-Nishida, E., Miyachi, Y. & Narumiya, S. Prostacyclin-IP signaling and prostaglandin E2-EP2/EP4 signaling both mediate joint inflammation in mouse collagen-induced arthritis. J. Exp. Med. 203, 325–335 (2006).
Paul, S. et al. Aloe vera gel homogenate shows anti-inflammatory activity through lysosomal membrane stabilization and downregulation of TNF-α and Cox-2 gene expressions in inflammatory arthritic animals. Future J. Pharm. Sci. 7, 1 (2021).
Kim, J. S., Lee, Y. H., Chang, Y. U. & Yi, H. K. PPARγ regulates inflammatory reaction by inhibiting the MAPK/NF-κB pathway in C2C12 skeletal muscle cells. J. Physiol. Biochem. 73, 49–57 (2017).
Koufany, M., Jouzeau, J. Y. & Moulin, D. Fenofibrate vs pioglitazone: Comparative study of the anti-arthritic potencies of PPAR-alpha and PPAR-gamma agonists in rat adjuvant-induced arthritis. Biomed. Mater. Eng. 24, 81–88 (2014).
Hussein, S. Z., Mohd Yusoff, K., Makpol, S. & Mohd Yusof, Y. A. Gelam honey attenuates carrageenan-induced rat paw inflammation via NF-κB pathway. PLoS ONE 8, e72365 (2013).
Schreck, R., Rieber, P. & Baeuerle, P. A. Reactive oxygen intermediates as apparently widely used messengers in the activation of the NF-xB transcription factor and HIV-1. EMBO J. 10, 2247–2258 (1991).
Barua, C. C. et al. Anti-arthritic and anti-inflammatory activity of a polyherbal formulation against Freund’s complete adjuvant induced arthritis in Wistar rats. Indian J. Trad. Knowl. 16, 482–489 (2017).
Bhargava, A., Shrivastava, P. & Tilwari, A. HPTLC analysis of Fumaria parviflora (Lam.) methanolic extract of whole plant. Future J. Pharm. Sci. 7, 1–9 (2021).
Fu, X. et al. Chlorogenic acid inhibits BAFF expression in collagen-induced arthritis and human synoviocyte MH7A cells by modulating the activation of the NF-κB signaling pathway. J. Immunol. Res. 2019, 1–10 (2019).
Dinore, J. M. & Farooqui, M. GC-MS and LC-MS: An integrated approach towards the phytochemical evaluation of methanolic extract of Pigeon Pea [Cajanus cajan (L.) Millsp] leaves. Nat. Prod. Res. 36, 2177–2181 (2022).
Gomaa, A., Elshenawy, M., Afifi, N., Mohammed, E. & Thabit, R. Enhancement of the anti-inflammatory and anti-arthritic effects of theophylline by a low dose of a nitric oxide donor or non-specific nitric oxide synthase inhibitor. Br. J. Pharmacol. 158, 1835–1847 (2009).
Neog, M. K., Joshua Pragasam, S., Krishnan, M. & Rasool, M. p-Coumaric acid, a dietary polyphenol ameliorates inflammation and curtails cartilage and bone erosion in the rheumatoid arthritis rat model. BioFactors 43, 698–717 (2017).
Rajendran, R. & Krishnakumar, E. Anti-arthritic activity of Premna serratifolia Linn., wood against adjuvant induced arthritis. Avicenna J. Med. Biotechnol. 2, 101–106 (2010).
Zhang, Q. et al. Anti-arthritic activities of ethanol extracts of Circaea mollis Sieb. & Zucc. (whole plant) in rodents. J. Ethnopharmacol. 225, 359–366 (2018).
Ghatani, K. et al. Revealing probiotic potential of enterococcus strains isolated from traditionally fermented chhurpi and healthy human gut. Front. Microbiol. 13, 1–18 (2022).
Zhang, X. et al. Effects of dietary selenium on immune function of spleen in mice. J. Funct. Foods 89, 104914 (2022).
Sunderman, F. W. & Nomoto, S. Measurement of human serum ceruloplasmin by its p-phenylenediamine oxidase activity. Clin. Chem. 16, 1 (1970).
Draper, H. H. & Hadley, M. Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol. 186, 421–431 (1990).
Ellman, G. L. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 82, 70–77 (1959).
Aebi, H. Catalase. In Methods of Enzymatic Analysis (ed. Bergmeyer, H. U.) 673–674 (Academic Press, 1974).
Nishikimi, M., Appaji Rao, N. & Yagi, K. The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem. Biophys. Res. Commun. 46, 849–854 (1972).
Das, P. et al. High-performance thin-layer chromatography coupled attenuated total reflectance-Fourier-transform infrared and NMR spectroscopy-based identification of α-amylase inhibitor from the aerial part of Asparagus racemosus Willd.. Phytochem. Anal. 33, 1018–1027 (2022).
Ashraf, G. J. et al. High-performance thin-layer chromatography based approach for bioassay and ATR–FTIR spectroscopy for the evaluation of antioxidant compounds from Asparagus racemosus Willd. aerial parts. Biomed. Chromatogr. 35, 1 (2021).
Marulasiddaswamy, K. M. et al. HR-LC-MS based profiling of phytochemicals from methanol extracts of leaves and bark of Myristica dactyloides Gaertn. from Western Ghats of Karnataka, India. J. Appl. Biol. Biotechnol. 9, 124–135 (2021).
Acknowledgements
Authors acknowledge Department of Zoology for providing the animal house and instrument facilities for the research work. Moreover, authors also acknowledge Dr. Hari Sankar Das, Assistant Professor, Department of Chemistry, for using the Buchi-type rotary evaporator (KNF, RC600, Germany) required for plant extraction. Authors acknowledge Dr. Dhiraj Saha, Professor, Department of Zoology, for using Nanodrop spectrophotometer (SPECTROstar Nano, BMG LABTECH, Germany) required for RNA estimation. Authors also acknowledge Dr. Sourav Mukherjee, Assistant Professor, Department of Zoology, for using the plate reader (Bio-Rad, USA) required for ELISA assay. Authors also acknowledge the Council of Scientific and Industrial Research (CSIR), Human Resource Development group and University of North Bengal for granting the Junior Research Fellowships to SS [CSIR-JRF sanction no. 09/285(0094)/2019-EMR-I, dated-3rd March, 2020]; SKR [CSIR-JRF sanction no. 09/285(0088)/2019-EMR-I, dated-7th October, 2019]; and institutional fellowship to DM (University fellowship No- 259/R-2018, dated- 5th July, 2018). Authors also acknowledge University of North Bengal for granting fund (Ref. No: 2273/R-2021 dated-24/06/2021) for purchasing the ELISA kits.
Author information
Authors and Affiliations
Contributions
SB contributed to the conception and designing of experiments; RS designed the phytochemical analysis of the plant; SS and DM performed the in vivo experiments; SS, SKR and DM performed ELISA, RT-PCR study and analyzed the data; AC also performed the quantitative expression study; GJA performed the HPTLC analysis of the plant; SS prepared and revised the manuscript; interpretation of the experimental outcome, critical revision of the manuscript, and final approval of the manuscript for publication were done by SB. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
The experimental procedures were carried out from 2022 to 2023 in strict compliance with the ethical guidelines approved by the Institutional Animal Ethical Committee [Approval number: IAEC/NBU/2022/24] of CPCSEA (Committee for the Purpose of Control and Supervision of Experiments on Animals) of the University of North Bengal, West Bengal, India. All experimental methods were performed in accordance with the relevant guidelines and regulations. The ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines were followed in reporting the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sarkar, S., Modak, D., Roy, S.K. et al. Equisetum diffusum whole plant methanol extract attenuates rheumatoid arthritis by modulating the expression of key inflammatory biomarkers in rat models. Sci Rep 15, 9950 (2025). https://doi.org/10.1038/s41598-024-82951-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-82951-1