Abstract
This retrospective study aimed to stress the advantages of autologous hematopoietic stem cell transplantation (auto-HSCT) in treating primary MM. Ninety-four MM patients who underwent initial parallel sequential auto-HSCT were selected. Data on efficacy (efficacy evaluation, renal function and hemoglobin recovery), immune reconstitution (B-cell subsets, immunoglobulin levels, T-cell subsets, NK cells, neutrophil-to-lymphocyte ratio (NLR), lymphocyte-to-monocyte ratio (LMR)) and hematopoietic reconstitution times were collected and analyzed. Whether in all selected patients or in groups R-ISS II-III, there was a notable increase in the proportion of patients achieving in a very good partial response (VGPR) or better (P < 0.001, P = 0.02) and a complete response (CR) or better (P = 0.007, P = 0.014) after transplantation compared to the pre-transplant status. Post-Transplant Immune Reconstitution Analysis (Baseline vs. Pre-Transplant and Pre-Transplant vs. Post-Transplant): The level of CD19 + B cells, CD20 + B cells, CD22 + B cells, CD3 + T cells, IgG and LMR showed the same change trend, that is, it decreased before transplantation (P < 0.001, P < 0.001, P < 0.001, P < 0.001, P<0.007, P < 0.001) and then increased significantly after transplantation(P < 0.001, P < 0.001, P < 0.001, P < 0.001, P < 0.001, P < 0.001). CD3 + CD4 + T cells from 545.97 (342.11,708.60)/µL to 342.93 (168.38, 475.52)/µL (P < 0.001) and then to 251.48 (188.52, 406.98)/µL (P = 0.348); CD3 + CD8 + T cells from 391.36 (242.19, 563.37)/µL to 337.23 (192.54, 505.96)/µL (P = 0.065) and then to 797.96 (514.49, 1198.03)/µL (P < 0.001), so the CD3 + CD4+/CD3 + CD8 + T cell ratio still remained inverted post-transplant. NK cells changed from 309.86 (206.33, 460.96)/µL to 258.31 (160.75, 436.68)/ µL (P = 0.229) and then to 151.08 (108.17, 240.84)/µL (P = 0.007). Auto-HSCT can promote prolonged remission in patients with MM and also overcome some high-risk factors to achieve superior efficacy in group R-ISS II-III. Patients were immunodeficient before transplantation and auto-HSCT facilitated immune reconstitution.
Similar content being viewed by others
Introduction
Multiple myeloma (MM), a malignancy of plasma cells, is marked by abnormal clonal plasma cell proliferation in the bone marrow, monoclonal immunoglobulin presence in serum, suppression of normal polyclonal immunoglobulin synthesis, consequent tissue and organ damage, and immune dysfunction1. It is generally accepted that MM evolves from a premalignant condition known as monoclonal gammopathy of undetermined significance and an intermediate asymptomatic stage referred to as smoldering multiple myeloma2. Multiple myeloma accounts for about 1% of all malignancies and approximately 10% of hematological cancers, ranking as the second most common blood cancer after lymphoma3. Multiple myeloma is highly heterogeneous, prone to recurrence post-remission, and remains incurable with a poor overall prognosis. In recent decades the MM incidence in China has risen, necessitating steps to improve the patient prognosis and quality of life4. Primary treatments include Immunomodulatory Drugs (IMiDs), Proteasome Inhibitors (PIs), auto-Human Stem Cell Transplantation (HSCT), and allogeneic HSCT (allo-HSCT), with emerging therapies like monoclonal antibodies, bispecific antibodies, immune checkpoint inhibitors, antibody-drug conjugates, vaccines, and CAR T-cell therapy5.
New drugs such as IMiDs, PIs, and anti-CD38 monoclonal antibodies have improved patient outcome and prolonged survival6, raising questions about the necessity of auto-HSCT. However, studies show that auto-HSCT in the new drug era enhances response rates and survival times, remaining a standard treatment for newly diagnosed MM patients eligible for transplantation7,8,9. Current efficacy assessments follow the 2016 International Myeloma Working Group (IMWG) criteria10, evaluating therapeutic effects based on serum and urine M protein levels, free light chain levels, immunofixation electrophoresis, bone marrow plasma cell counts, and soft tissue plasmacytoma sizes, without including organ function and hematopoiesis changes.
At the microscopic level, certain research posits that the development and advancement of MM is linked to disturbances in the immune function of the organism11. Immune system reconstruction is crucial for controlling early post-auto-HSCT infection, disease recurrence, and long-term favorable prognosis. Immune function, divided into innate and adaptive immunity, can be assessed through analyzing B-cell subsets, immunoglobulins, T-cell subsets, cytokines, and NK cells. Existing studies often focus on individual immune parameters or other hematological malignancies, with fewer comprehensive observations on immune restoration post-transplant in MM patients.
Some studies have monitored immune function and the tumor microenvironment using absolute lymphocyte (ALC) and monocyte counts (AMC) in peripheral blood12,13. The ratio of ALC to AMC (LMR) can reflect immune status, while the neutrophil-to-lymphocyte ratio (NLR) is an independent factor affecting progression-free survival in MM patients due to inflammatory responses elevating ANC and cytokine secretion promoting angiogenesis in the tumor microenvironment.
This article provides a retrospective analysis to highlight auto-HSCT’s significance in MM management, addressing therapeutic effects, renal function recovery, hematopoietic recovery, and immune reconstitution post-transplant, aiming to contribute to clinical applications.
Methods
Study design and patients
Inclusion Criteria: 1. Aged > 18 years. 2. Diagnosed with MM per 2014 IMWG criteria. 3. Received chemotherapy followed by auto-HSCT at our hospital post-induction therapy between January 1, 2019 and December 31, 2021.
Exclusion Criteria: 1. Referred from another hospital after diagnosis and induction therapy with unavailable baseline data. 2. Received consolidation or maintenance therapy within 3 months after auto-HSCT. 3. Had secondary extramedullary plasmacytoma. 4. Had other tumors.
This retrospective study was conducted in accordance with the Declaration of Helsinki, approved by Ethical Committee of Fujian Medical University Union Hospital and informed consent was obtained from each participant.
Data collection
General Characteristics: Gender, age, classification based on M protein type, International Staging System (ISS), Revised-International Staging System (R-ISS), and genetic high-risk factors (RB1/13q14, IgH rearrangement, TP53/17p deletion, 1q21/CKS1B).
Laboratory Tests: Baseline, pre-transplant, and 3-month post-transplant peripheral blood values including creatinine levels, ANC, ALC, AMC, hemoglobin (Hb) levels, B-cell subsets, immunoglobulin levels, T-cell subsets, NK cell percentages, serum and urine M protein, FLC, protein electrophoresis, bone marrow plasma cell proportion, and soft tissue plasmacytoma size.
Statistical analysis
Data were entered and double-checked in Excel and analyzed using SPSS 25. Measurement data conforming to normal distribution were expressed as mean ± standard deviation (X ± S) and analyzed using two-sample t-tests. Non-normally distributed data were expressed as M (P25 and P75) and analyzed using rank sum tests. Enumeration data were analyzed using the Chi-squared test. P < 0.05 was considered statistically significant. All graphs were constructed using Microsoft Excel and GraphPad Prism 9.
Results
Clinical characteristics of patients
A total of 94 patients with multiple myeloma (MM) were included in the analysis, 58 males and 36 females (ratio 1.61:1). The median age at diagnosis was 55.5 years (range 38–70 years), with the largest cohort being those between 50 and 59 years, accounting for 56 cases (59.6%). Classified by M protein type, IgG was the most prevalent with 51 cases (54.3%), followed by IgA in 12 cases (12.8%), IgD in six cases (6.4%), IgM in one case (1.1%), λ-light chain in 14 cases (14.9%), κ-light chain in nine cases (9.6%), and non-secretory type in one case (1.1%). According to the ISS staging, 29 cases (30.5%) were in Stage I, 26 cases (27.7%) in Stage II, and 39 cases (41.5%) in Stage III. According to the R-ISS staging, 21 cases (22.3%) were in Stage I, 44 cases (46.8%) in Stage II, and 29 cases (30.9%) in Stage III. Genetic abnormalities detected by FISH revealed 22 cases (23.2%) without significant abnormalities, 33 cases (34.7%) with RB1 deletion, 50 cases (52.6%) with IgH rearrangement, 13 cases (13.7%) with TP53 gene deletion, and 51 cases (53.7%) with 1q21 amplification. Half of the cases received the ‘Bortezomib + Lenalidomide + Dexamethasone’ regimen (VRD) prior to transplantation. 8 (8.5%) cases were on the ‘Bortezomib + Doxorubicin + Dexamethasone’ protocol (PAD), while 5 (5.3%) received the ‘Bortezomib + Cyclophosphamide + Dexamethasone’ treatment (VCD). Additionally, 4 (4.3%) cases were treated with the ‘Bortezomib + Thalidomide + Dexamethasone’ combination (VTD). The remaining cases underwent two or more of these chemotherapy regimens during the treatment phase. More than half of the cases(59.6%) completed 4 cycles of chemotherapy, with the longest extending to 10 cycles and the shortest comprising only 2 cycles.Detailed baseline characteristics are provided in Table 1.
Transplantation efficacy analysis
According to the traditional response criteria of the 2016 IMWG, patients were divided into three groups: complete response (CR) or better, very good partial response (VGPR), and partial response (PR) or less. The number of patients achieving PR or less, VGPR, and CR or better before vs. after transplantation were 26 cases (27.7%) vs. 7 cases (7.4%), 35 cases (37.2%) vs. 39 cases (41.5%), and 33 cases (35.1%) vs. 48 cases (51.1%), respectively. There was a statistically significant difference in the overall distribution of responses before and after transplantation (P < 0.01) (Fig. 1a). The proportion of patients achieving VGPR or better increased from 68 cases (72.3%) before transplantation to 87 cases (92.6%) post-transplantation, showing a significant difference (P < 0.001). Additionally, the proportion of patients achieving CR or better rose from 35.1 to 51.1% (P = 0.007).
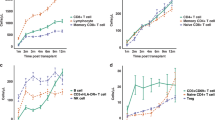
Transplantation Efficacy Analysis. Efficacy before and after auto-HSCT for the all patients collected (a). Efficacy for the R-ISS II-III patients in pre-transplant and post-transplant (b). Changes in eGFR (c) and hemoglobin (e) at baseline, before, and after auto-HSCT. Hematopoietic reconstruction time after auto-HSCT (d). PR: partial response, VGPR: very good partial response, CR: complete response, eGFR: estimated glomerular filtration rate.
Among 73 high-risk patients (R-ISS stages II-III), the efficacy rates before and after transplantation were as follows: PR or less in 17 cases (23.3%) vs. 4 cases (5.5%), VGPR in 30 cases (41.1%) vs. 31 cases (42.5%), and CR or higher in 26 cases (35.6%) vs. 38 cases (52.1%). The overall efficacy distribution among the intermediate-high risk population showed a significant difference (P = 0.001) (Fig. 1b). Notably, the proportion of patients achieving VGPR or higher increased significantly after transplantation (76.7% vs. 94.6%, P = 0.02), and those achieving CR or higher also rose significantly (35.6% vs. 52.1%, P = 0.014). Auto-HSCT can further induce disease remission and overcome intermediate-high risk factors related to the serological and genetic aspects of multiple myeloma, thus achieving superior therapeutic outcomes.
In the 94 patients, the baseline estimated glomerular filtration rate (eGFR) levels and changes before and after transplantation were 88.0 (57.0, 105.3) µmol/L vs. 104.5 (82.8, 122.0) µmol/L vs. 102.5 (81.0, 115.0) µmol/L (P < 0.001, P = 0.497), respectively. When comparing baseline with post-transplant, the values were 88.0 (57.0, 105.3) µmol/L vs. 102.5 (81.0, 115.0) µmol/L (P < 0.001) (Fig. 1c). This indicates no significant improvement in eGFR compared to pre-transplant levels in the early post-transplant period.
The median time to neutrophil engraftment post-auto-HSCT was 11.0 (10.0–12.0) days, and the median time to platelet engraftment was 14.0 (11.0–18.0) days (Fig. 1d). The two-week period following transplantation is crucial for the reconstitution of both granular and megakaryocyte lineages.
The baseline and pre- and post-transplant hemoglobin (Hb) levels (g/L) for the 94 patients were 97.53 ± 26.01 vs. 118.45 ± 19.17 vs. 113.26 ± 19.78 (P < 0.001, P = 0.444), respectively, with a significant difference between baseline and post-transplant Hb levels (P < 0.001) (Fig. 1e). This suggests no significant increase in hemoglobin levels compared to pre-transplant levels during the early post-transplantation period.
Immunological reconstitution
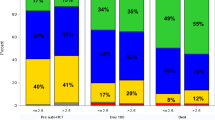
In 38 patients the comprehensive data on the changes in B cell subsets (CD19+, CD22+, CD20+, CD10+) were collected. Here are the comparisons between baseline and pre-transplantation as well as the pre-transplantation and three months post-transplantation: The changes in CD19 + B cell subsets were 89.78 (39.72, 163.04)/µL vs. 10.59 (4.75, 34.13)/µL (P < 0.001), vs. 90.93 (46.09, 193.43)/µL (P < 0.001), for CD20 + B cell subsets they were 89.66 (40.60, 154.23)/µL vs. 11.38 (7.99, 40.67)/µL (P < 0.001), vs. 102.51 (47.60, 223.85)/µL (P < 0.001), for CD22 + B cell subsets the changes were 81.25 (37.62, 150.84)/µL vs. 10.06 (3.96, 41.00)/µL (P < 0.001), vs. 104.68 (58.01, 221.47)/µL (P < 0.001), and for CD10 + B cell subsets the changes were 2.44 (0.47, 5.88)/µL, 1.35 (0.75, 2.77)/µL, and 1.46 (0, 2.83)/µL (P = 0.103), as detailed in Table 2. Before transplantation, there was a significant decrease in CD19+, CD20+, and CD22 + B cell subsets compared to baseline (P < 0.001 for all), and auto-HSCT facilitated the reconstitution of these B cell subsets. Three months post-transplantation, the numbers of CD19+, CD20+, and CD22 + B cell subsets were comparable to baseline levels (P = 0.906, P = 0.621, P = 0.088, respectively) (Fig. 2a).
Immunological Reconstitution. Changes in the number of B cell subsets (a), T Cell Subsets (c), NK Cell Subsets (d) and the level of immunoglobulin (b), NLR and LMR at baseline, before and after transplant (e). NLR: neutrophil-to-lymphocyte ratio, LMR: lymphocyte-to-monocyte ratio.
Levels of immunoglobulins IgG, IgM, and IgA were comprehensively collected at baseline, pre-transplantation, and three months post-transplantation in 77 patients. The changes in levels of the types of antibodies from baseline to pre-transplantation to three months post-transplantation were as follows: IgG 9.67 (5.67, 48.65) g/L vs. 7.62 (5.95, 10.60) g/L vs. 10.90 (8.80, 14.00) g/L (P < 0.007, P < 0.001): IgM 0.24 (0.11, 0.41) g/L vs. 0.38 (0.26, 0.56) g/L vs. 0.48 (0.30, 0.71) g/L (P < 0.001, P = 0.441): IgA 0.30 (0.14, 0.64) g/L vs. 0.48 (0.29, 0.96) g/L vs. 0.49 (0.27, 0.82) g/L (P = 0.018, P > 0.999), as detailed in Table 3. Before transplantation, IgG levels decreased compared to baseline (P < 0.007), while IgA and IgM levels increased (P < 0.001, P = 0.018). Three months post-transplantation, IgG levels rebounded to baseline levels (P = 0.512), with no significant difference observed in IgM and IgA levels compared to pre-transplantation levels (P = 0.441, P > 0.999) (Fig. 2b).
The variations in T cell subsets (CD3+, CD3 + CD4+, CD3 + CD8+) were comprehensively collected at baseline, pre-transplantation, and three months post-transplantation in 46 patients. Here are the comparisons between baseline and pre-transplantation, as well as pre-transplantation and three months post-transplantation: The levels of CD3 + T cell subset numbers were 1118.12 (695.81, 1290.2)/µL vs. 809.80 (416.25, 1139.55)/µL (P < 0.001), vs. 1194.84 (806.87, 1571.34)/µL (P < 0.001). For CD3 + CD4 + T cell subset, the levels were 545.97 (342.11, 708.60)/µL vs. 342.93 (168.38, 475.52)/µL (P < 0.001), vs. 251.48 (188.52, 406.98)/µL (P = 0.348). The CD3 + CD8 + T-cell subset the levels were 391.36 (242.19, 563.37)/µL vs. 337.23 (192.54, 505.96)/µL (P = 0.065), vs. 797.96 (514.49, 1198.03)/µL (P < 0.001). Detailed information is provided in Table 4. Before transplantation, the counts of CD3 + T and CD3 + CD4 + T cell subsets decreased significantly compared to the baseline (P < 0.001, P < 0.001), while the decrease in CD3 + CD8 + T cell subset was not significant (P = 0.065). Three months post-transplantation, the count of CD3 + T cells recovered to the baseline level (P = 0.754), the count of CD3 + CD4 + T cells had not yet recovered (P < 0.001), and the count of CD3 + CD8 + T cells even exceeded the baseline level (P = 0.001) (Fig. 2c). Baseline, pre-transplant, and three months post-transplant CD3 + CD4+/CD3 + CD8 + T cell ratios were 1.40 (0.93, 1.82) vs. 1.05 (0.61, 1.37) (P = 0.043), vs. 0.36 (0.23, 0.49) (P < 0.001), respectively, with the ratio remaining inverted.
In 46 patients, Here are the comparisons between baseline and pre-transplantation, as well as pre-transplantation and three months post-transplantation: The changes in the number of CD3-CD16 + CD56 + NK cells were 309.86 (206.33, 460.96)/µL vs. 258.31 (160.75, 436.68)/µL (P = 0.229), vs. 151.08 (108.17, 240.84)/µL (P = 0.007) (Fig. 2d). No increase in the number of CD3-CD16 + CD56 + NK cells was observed at three months post-transplantation.
In 90 patients NLR and LMR were calculated at baseline, pre-transplantation, and three months post-transplantation. Here are the comparisons between baseline and pre-transplantation, as well as pre-transplantation and three months post-transplantation: The NLR values of 1.78 (1.20, 2.67) vs. 2.53 (1.46, 3.40) (P = 0.011) vs. 1.32 (0.83, 2.10) (P < 0.001), and LMR values of 4.69 (3.60, 7.20) vs. 2.70 (1.76, 3.91) (P < 0.001) vs. 5.00 (3.86, 7.44) (P < 0.001), respectively (Table 5). Pre-transplant NLR was significantly elevated compared to baseline (P < 0.001), and pre-transplant LMR was notably reduced compared to baseline (P < 0.001), indicating immune dysfunction. Auto-HSCT is beneficial for immune reconstitution, as by three months post-transplantation, both NLR and LMR returned to levels comparable to baseline (P = 0.076, P > 0.999) (Fig. 2e).
Discussion
This study found that auto-HSCT not only mitigates high-risk serological and genetic factors but also aids in immune reconstitution. However, early post-transplantation renal function and anemia status showed no significant improvement compared to pre-transplant levels, and the two-week period post-transplantation is critical for hematopoietic reconstitution in most patients. Notably, this research further explores the value of auto-HSCT in MM by examining changes in patients’ immune function status during the early post-transplantation phase. Pre-transplant the patient is immunosuppressed, a condition detrimental to long-term survival. Transplantation facilitates immune reconstruction. By the third month post-transplant the levels of B-cell subsets (CD19+, CD22+, CD20+), IgG, CD3 + T cells, and immune function indices such as LMR and NLR had returned to pre-transplant baseline levels. Conversely, CD3 + CD4 + T cells and NK cells had not fully recovered, and the ratio of CD3 + CD4 + T cells to CD3 + CD8 + T cells remained inverted.
The introduction of new drugs briefly brought into question the role of auto-HSCT in MM treatment strategies. The combination of proteasome inhibitors (PIs) and/or immunomodulatory drugs (IMiDs) is regarded as the standard induction regimen, consistently demonstrating effective responses across all phases of MM treatment (induction, consolidation, and maintenance) for nearly all patients14. Nevertheless, a meta-analysis consolidating findings from three major phase III randomized controlled trials (2000–2017) demonstrated that sequential auto-HSCT correlated with superior progression-free survival (PFS) compared to therapies solely reliant on IMiDs and PIs. Hence, even with various novel drug regimens in induction therapy, implementing sequential auto-HSCT as an additional measure can intensify the therapeutic response and improve PFS, reaffirming auto-HSCT as a crucial intervention for MM patients eligible for transplantation. Among the 94 patients who underwent single-sequential auto-HSCT in our study, there was a significant increase in the proportion of patients achieving very good partial response (VGPR) or better, as well as the proportion achieving complete response (CR) or better, compared to those after chemotherapy. This therapeutic advantage was also observed in the intermediate-high risk group (R-ISS stages II-III).
Renal impairment is the most common organ damage associated with MM. According to studies by the British Cancer Collaboration Group, 40–55% of newly diagnosed MM patients exhibit varying degrees of renal function damage, with 15–20% of patients suffering from moderate to severe renal insufficiency. Regarding the impact of transplantation on renal function, the International Bone Marrow Transplant Registry reports that of 35 MM patients requiring hemodialysis pre-transplant, 34 were able to discontinue dialysis post-transplantation15. Research has indicated that MM patients with severe renal damage undergoing auto-HSCT experience not only an increased rate of CR but also a marked reduction in urinary protein16. Among the 94 patients receiving auto-HSCT in this study, eGFR improved with disease control post-induction therapy. However, no further improvement in renal function was observed in the early post-transplantation period. The study’s follow-up period was merely three months post-transplantation. The duration for individual kidney function restoration varies depending on the time required for disease control, suggesting that renal function may still be in the process of recovery over time. Additionally, prevalent causes of renal dysfunction include diabetes, high blood pressure, and inherent kidney disorders. In cases where primary MM is well-managed post-induction therapy and transplantation where notable renal function improvement is not observed, a renal biopsy can be considered to determine whether other causes exist besides myeloma.
Hematopoietic stem cells, due to their homing property, recognize and return to the bone marrow cavity after infusion into the patient, restoring normal hematopoiesis, a process known as hematopoietic reconstitution. The recovery of hematopoietic function is the primary indicator of successful auto-HSCT17,18.Research has reported that primary engraftment failure is associated with a relatively higher mortality rate compared to secondary engraftment failure19. In this study, the median time to neutrophil engraftment post-auto-HSCT was 11.0 (10.0–12.0) days, and the median time to platelet engraftment was 14.0 (11.0–18.0) days, indicating that the two weeks following auto-HSCT is a critical period for hematopoietic reconstitution, generally consistent with findings from many centers. Neutrophil recovery typically precedes platelet recovery, and, due to hospital stay limitations, some patients were discharged before platelet engraftment occurred. Post-discharge blood count monitoring is intermittent leading to potential inaccuracies in recording the exact platelet engraftment time. Previous studies have demonstrated that factors influencing hematopoietic reconstitution post-auto-HSCT include pre-transplant disease status, the number of infused stem cells, conditioning regimen, use of hematopoietic growth factors, among others20,21,22,23,24,25,26. The most critical factor is the quantity of transplanted hematopoietic stem cells27,28. Research has shown that higher doses of CD34 + cells correlate positively with the speed of both neutrophil and platelet engraftment20,29. However, faster hematopoietic recovery does not necessarily lead to favorable transplant outcomes.
Anemia is a common clinical manifestation of MM, with studies indicating that anemia in MM patients may be related to myeloma cells infiltrating the bone marrow cavity, leading to erythropoiesis suppression, impaired renal function resulting in inadequate erythropoietin production, chemotherapy-induced myelosuppression, and immune dysfunction causing overproduction of cytokines like interleukin-63. Theoretically, auto-HSCT should aid in controlling primary MM disease and facilitating hematopoietic reconstitution. However, in this study, no further elevation in hemoglobin levels was observed in MM patients shortly post-transplantation. This can be attributed to the relatively short follow-up period and the incomplete recovery of the bone marrow microenvironment as well as renal function and immune function within three months post-transplantation.
The primary disease of multiple myeloma (MM) leads to immune dysfunction, and induction therapy results in secondary immunodeficiency (SID). The impairment of immune system function hinders the long-term remission of the primary disease and increases the incidence of infections and secondary malignancies. Reconstructing the body’s immune function is a crucial foundation for achieving further disease remission and survival benefits through auto-HSCT.
B cells constitute approximately 20% of the total peripheral lymphocyte count and are identified by surface marker molecules CD3-CD19+. Research by Heck et al.30 has shown that in patients with MM, following induction therapy, preconditioning, and transplantation, there is a significant reduction in B cell numbers. The median number of CD19 + B cells decreases by about 95% compared to baseline. Naive B cells can be activated through two mechanisms: T cell-dependent and T cell-independent activation. In T cell-dependent activation, co-stimulatory molecule CD40L, expressed by CD4 + T cells, and interleukin-21 (IL-21) play crucial roles in the interaction between T and B cells. Following auto-HSCT, reductions in IL-21 and CD40L may be significant causes of quantitative and functional B cell defects in patients who received high-dose chemotherapy. Studies have demonstrated that in patients with malignant hematological diseases undergoing auto-HSCT, CD19 + B cells exhibit a marked decline one month post-transplantation, begin to recover from the third month, and are largely restored by six to twelve months post-transplantation30. Additionally, CD 20 is a specific marker of B cells expressed at all stages of B cell development except in plasma cells and it regulates B cell proliferation and differentiation. The membrane protein CD22, specifically expressed on B cells, acts as an inhibitory receptor, negatively modulating the CD19/CD21/CD81 co-receptor complex31,32. However, limited research exists on the reconstruction of various B cell subpopulations following MM transplantation. This study noted a significant reduction in the quantity of CD19+, CD20+, and CD22 + B cells before transplantation, with these subsets’ numbers restored to pre-transplant baseline levels by the third month post-transplantation.
Following maturation, B cells are activated to transform into plasma cells that secrete immunoglobulins; hence, serum immunoglobulin levels can act as vital parameters to assess B cell functionality or the state of humoral immunity. This serves as an essential indicator in monitoring health conditions following transplantation surgeries. Retrospective investigations revealed that in patients with hematological malignancies undergoing hematopoietic stem cell transplantation, IgG levels can normalize 3–6 months post-transplantation. An increase in IgM levels above normal values can be observed as early as three months post-transplantation, while IgA levels typically take up to one year to normalize33,34,35,36. Similarly, our research uncovered a decline in IgG levels before transplantation, which subsequently rebounded to baseline levels within three months following the procedure. Pre-transplant concentrations of IgM and IgA were higher than baseline values, showing no remarkable divergence during the early stages post-transplantation. The exploration of immunoglobulin alterations in this study was somewhat rudimentary. Given that MM patients characteristically secrete abnormal monoclonal immunoglobulins, future studies necessitate more accurate quantification of the varied polyclonal immunoglobulin contents. A study by Jimenez-Zepeda VH et al.37 further emphasized that the restoration of serum immunoglobulins post-transplant serves as an independent predictor of progression-free and overall survival (PFS and OS, respectively), revealing a positive association between inferior immunoglobulin restoration and less favorable prognoses.
T lymphocytes express T cell antigen receptors along with the CD3 + complex. CD4 + T cells function as helper T cells, facilitating antibody secretion and modulating immune responses, whereas CD8 + T cells serve as cytotoxic T cells, identifying and destroying target cells. This study reveals that pre-transplant levels of CD3+, as well as CD3 + CD4 + T cells, are reduced. By three months post-transplantation, CD3 + T cell counts rebound to baseline levels, while CD3 + CD4 + T cell counts have not yet recovered. Pre-transplant counts of CD3 + CD8 + T cells showed no significant deviation from baseline but increased markedly three months after transplantation. T cell immune reconstitution after HSCT occurs through two pathways: thymus-independent and thymus-dependent38. The thymus-independent pathway involves direct and rapid proliferation of T cells within the transplanted hematopoietic stem cells via peripheral immune organs, bypassing the thymus. The thymus-dependent pathway involves precursor T cells from the graft returning to the thymus, where they undergo development and differentiation into naive T lymphocytes, leading to a stable and long-lasting immune reconstitution. The delayed recovery of CD4 + T cells compared to CD8 + T cells may be due to the generation and maturation of CD8 + T cells not being entirely dependent on the thymus, whereas CD4 + T cell production is primarily thymus-dependent39. Impaired thymic function post-transplantation is a significant cause of T cell immunodeficiency during the early period40. Compared to previous studies, this research lacks longer-term follow-up post-transplantation to further elucidate the timeline for the restoration of various T cell subsets. Moreover, further investigation into cytokine dynamics could enhance understanding of T cell function recovery.
NK cells are a vital component of the body’s innate immune system. Currently, lymphoid cells identified as human NK cells exhibit the phenotypes TCR-, mIg-, CD56+, and CD16+. Unlike T and B cells, NK cells do not specifically recognize antigens; instead, they express various regulatory receptors that activate or inhibit their functions, enabling them to directly eliminate target cells and secrete cytokines, among other activities. Based on the differential expression intensity of CD56 on their cell surface, NK cells are classified into two subsets: CD56bright and CD56dim. CD56dim represents a more mature NK cell type, primarily associated with cytotoxic activity, comprising 90% of NK cells in peripheral blood. CD56bright is an immature cellular state characterized by the secretion of various cytokines and weaker killing ability41. Orrantia et al.41 found that the total number of NK cells could recover one month after MM transplantation. Although CD56dim NK cells remain the predominant subset at all time points, there is a noticeable elevation in the number of immature CD56bright NK cells at this stage. This transition in NK cell maturity endures for over 30 days post-transplant, with the underlying mechanism of this maturation defect yet to be fully elucidated but suspected to relate to injuries inflicted upon stromal cells, T-cells, dendritic cells, among others, during the preparatory regimen before transplantation. Furthermore, the relationship between plasma IL-15 levels and NK cells is under investigation, with Orrantia et al.41 suggesting that IL-15 stimulation can enhance CD56 expression on CD56dim NK cells. This study did not continuously follow the reconstruction of NK cells beyond one, two, and three months post-transplantation; it only observed the lag in the recovery of CD3-CD16 + CD56 + NK cell counts in MM patients three months post-transplantation.
The LMR exhibits potential as an alternative marker for TAMs, where a decline in LMR may indicate tumor-induced immune impairment, whereas an increase in LMR suggests extended survival periods for MM patients42. Additionally, the amplification of pro-tumor inflammatory responses and the weakening of anti-tumor immune responses could lead to variations in NLR, reflecting not merely changes in individual cell counts but rather a systemic immune imbalance43. This study observed elevated NLR and decreased LMR prior to transplantation, indicative of immune disorder. Transplantation facilitated immune reconstitution, and by three months post-transplantation, both NLR and LMR had returned to levels comparable to baseline. High ALC levels are a protective factor for the prognosis of multiple myeloma patients, whereas elevated NLR is a detrimental factor44,45.
The advent of numerous novel medications has significantly improved the prognosis for individuals diagnosed with MM. Nonetheless, a comprehensive assessment of the benefits of auto-HSCT, encompassing aspects such as therapeutic efficacy from the patient’s perspective, implications for organ functionality, restoration of hematopoiesis, and the rebuilding of immune competence, underscores the indispensable role that auto-HSCT plays within the therapeutic regimen for MM. Central to this discussion, immune reconstruction is a multifaceted and intricately influenced procedure, intimately tied to patient prognoses. Consequently, unraveling the patterns governing post-transplant immune reconstruction bears immense significance for informing and directing clinical interventions.
Conclusion
Auto-HSCT can further facilitate remission in MM and overcome related high-risk factors, achieving improved disease remission in the intermediate-high risk group (R-ISS stages II-III). No further improvement in renal function or hemoglobin was observed compared to pre-transplant levels shortly after transplantation. Regarding immune function, the body is in an immunodeficient state before transplantation, and auto-HSCT facilitates immune system rebuilding. By three months post-transplantation, levels of IgG, certain B-cell subsets (CD19+, CD22+, CD20+), quantities of CD3 + T-cell subsets, LMR, NLR, among other indicators, have returned to baseline levels. However, the counts of CD3 + CD4 + T cells and NK cells remain unrecovered, and the CD3 + CD4+/CD3 + CD8 + T-cell ratio remains inverted.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Cowan, A. et al. Diagnosis and management of multiple myeloma: a review. JAMA 327(5), 464–477. https://doi.org/10.1001/jama.2022.0003 (2022).
Lekha, M. & James, N. K. CAR T cell therapies for patients with multiple myeloma. Nat. Rev. Clin. Oncol. 18(2), 71–84. https://doi.org/10.1038/s41571-020-0427-6 (2020).
Niels, W. C. J., vdD, Charlotte, P. & Kwee, L. Y. Multiple myeloma. Lancet 397(10272), 410–427. https://doi.org/10.1016/s0140-6736(21)00135-5 (2021).
Jiangmei, L. et al. Incidence and mortality of multiple myeloma in China, 2006–2016: an analysis of the global burden of Disease Study 2016. J. Hematol. Oncol. 12(1), 1–7. https://doi.org/10.1186/s13045-019-0807-5 (2019).
Maria-Victoria, M. et al. Overall survival with daratumumab, bortezomib, melphalan, and prednisone in newly diagnosed multiple myeloma (ALCYONE): a randomised, open-label, phase 3 trial. Lancet 395(10218), 132–141. https://doi.org/10.1016/s0140-6736(19)32956-3 (2019).
Ying, Q. et al. Successful treatment of Relapsed/Refractory Extramedullary multiple myeloma with Anti-BCMA CAR-T cell therapy followed by Haploidentical hematopoietic stem cell transplantation: a Case Report and a review of the contemporary literature. Front. Med. (Lausanne). 8, 649824. https://doi.org/10.3389/fmed.2021.649824 (2021).
Binod, D. et al. Autologous transplantation for newly diagnosed multiple myeloma in the era of Novel Agent induction: a systematic review and Meta-analysis. JAMA Oncol. 4(3), 343–350. https://doi.org/10.1001/jamaoncol.2017.4600 (2018).
Felicity, E. et al. A population-based study of the impact of dialysis on mortality in multiple myeloma. Br. J. Haematol. 180(4), 588–591. https://doi.org/10.1111/bjh.14394 (2016).
Robert, A. K., Vincent, S. & R Multiple myeloma. Blood 111(6), 2962–2972. https://doi.org/10.1182/blood-2007-10-078022 (2008).
Shaji, K. et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol. 17(8), e328–e346. https://doi.org/10.1016/s1470-2045(16)30206-6 (2016).
Brown, R. et al. Either interleukin-12 or interferon-gamma can correct the dendritic cell defect induced by transforming growth factor beta in patients with myeloma. Br. J. Haematol. 125(6), 743–748 (2004).
Luis, F. P. et al. Peripheral blood lymphocyte/monocyte ratio at diagnosis and survival in classical Hodgkin’s lymphoma. Haematologica 97 (2), 262. https://doi.org/10.3324/haematol.2011.050138 (2011).
M M P, L. S., K H B, A A-K, M R L, C A H, R P K, L F P, A. P. & N G, A T Prognostic relevance of Lymphocytopenia, Monocytopenia and lymphocyte-to-monocyte ratio in primary myelodysplastic syndromes: a single center experience in 889 patients. Blood Cancer J. 7(3), e550. https://doi.org/10.1038/bcj.2017.30 (2017).
Wilson, I. G. et al. Utilization of hematopoietic stem cell transplantation for the treatment of multiple myeloma: a Mayo Stratification of Myeloma and Risk-adapted therapy (mSMART) consensus statement. Bone Marrow Transpl. 54(3), 353–367. https://doi.org/10.1038/s41409-018-0264-8 (2018).
M A D, C, A. M. P. H. R. F. M. F. J. H. J. B. N. C. L. C. et al. Autologous hematopoietic cell transplantation for multiple myeloma patients with renal insufficiency: a center for international blood and marrow transplant research analysis. Bone Marrow Transpl. 52(12), 1616–1622. https://doi.org/10.1038/bmt.2017.198 (2017).
Karine, A-M. et al. Régis Peffault dL, Jérôme C Extending autologous transplantation as first line therapy in multiple myeloma patients with severe renal impairment: a retrospective study by the SFGM-TC. Bone Marrow Transplant 53 (6):749–755. https://doi.org/10.1038/s41409-018-0122-8 (2018).
Franco, L., Barbarella, L. & Pietro, M. Current and future approaches to treat graft failure after allogeneic hematopoietic stem cell transplantation. Expert Opin. Pharmacother. 15 (1), 23–36. https://doi.org/10.1517/14656566.2014.852537 (2013).
Christelle, F. et al. Outcome of graft failure after allogeneic stem cell transplant: study of 89 patients. Leuk. Lymphoma 56(3), 656–662. https://doi.org/10.3109/10428194.2014.930849 (2014).
M, R. O., D M B, R. M. S., B-M S, J. M. & O R Graft failure in the modern era of allogeneic hematopoietic SCT. Bone Marrow Transpl. 48 (4), 537–543. https://doi.org/10.1038/bmt.2012.239 (2012).
Ho Sup, L. et al. Predictive factors for rapid neutrophil and platelet engraftment after allogenic peripheral blood stem cell transplantation in patients with acute leukemia. Ann. Hematol. 92(12), 1685–1693. https://doi.org/10.1007/s00277-013-1847-5 (2013).
A M K, N E-S, H K M, M-R K, A, E. H., Abd, O. F. R. & EF, D S Impact of CD34 subsets on engraftment kinetics in allogeneic peripheral blood stem cell transplantation. Bone Marrow Transpl. 35 (2), 129–136. https://doi.org/10.1038/sj.bmt.1704755 (2004).
S Q F, C N A, J K B, J J I, D N, J. L. L. Impact of mobilized blood progenitor cell quality determined by the CFU-GM/CD34 + ratio on rapid engraftment after blood stem cell transplantation. Blood Cells Mol. Dis. 28(3), 315–321. https://doi.org/10.1006/bcmd.2002.0519 (2002).
Ulkü, E., Seçkin, C., Mustafa, P., Mümtaz, Y. & Murat, T. Factors influencing engraftment in autologous peripheral hematopoetic stem cell transplantation (PBSCT). Transfus. Apher Sci. 36(1), 23–29. https://doi.org/10.1016/j.transci.2006.08.009 (2007).
Thissiane, L. G., Dalila, M. B. & Gabriela, B. Specific factors influence the success of autologous and allogeneic hematopoietic stem cell transplantation. Oxid. Med. Cell. Longev. 2(2), 82–87. https://doi.org/10.4161/oxim.2.2.8355 (2010).
Ramaprasad, S. et al. Overcoming graft rejection in heavily transfused and allo-immunised patients with bone marrow failure syndromes using fludarabine-based haematopoietic cell transplantation. Br. J. Haematol. 133(3), 305–314. https://doi.org/10.1111/j.1365-2141.2006.06019.x (2006).
T U, K. T., H, H. Y. Y. E. H. Y., K, H. T. M. A. M. R. T. K. K. M. I. & Y, T N Expansion of human NOD/SCID-repopulating cells by stem cell factor, Flk2/Flt3 ligand, thrombopoietin, IL-6, and soluble IL-6 receptor. J. Clin. Invest. 105(7), 1013–1021. https://doi.org/10.1172/jci8583 (2000).
G, T., S, S. J. D. V. J. N. & T, L M, B C, J C, B B Peripheral blood stem cell transplants for multiple myeloma: identification of favorable variables for rapid engraftment in 225 patients. Blood 85(2), 588–596 (1995).
W B, F A, S R, R S, J S, K L, T G, T D, K S, C W Factors that influence collection and engraftment of autologous peripheral-blood stem cells. J. Clin. Oncol. 13(10): 2547–2455. (1995). https://doi.org/10.1200/jco.1995.13.10.2547
Michael, A. P. et al. Donor, recipient, and transplant characteristics as risk factors after unrelated donor PBSC transplantation: beneficial effects of higher CD34 + cell dose. Blood 114(13), 2606–2616. https://doi.org/10.1182/blood-2009-03-208355 (2009).
Clarissa, H. et al. CD4 + T cell dependent B cell recovery and function after autologous hematopoietic stem cell transplantation. Front. Immunol. 12, 736137. https://doi.org/10.3389/fimmu.2021.736137 (2021).
Yang, Y. et al. Whole-genome sequencing of leopard coral grouper (Plectropomus leopardus) and exploration of regulation mechanism of skin color and adaptive evolution. Zool. Res. 41(3), 328–340. https://doi.org/10.24272/j.issn.2095-8137.2020.038 (2020).
Zixun, Y., Ya, Z. & Xin, W. Advances in chimeric antigen receptor T-cell therapy for B-cell non-hodgkin lymphoma. Biomark. Res. 9(1), 58. https://doi.org/10.1186/s40364-021-00309-5 (2021).
Melinda, H. et al. Homogeneous immunoglobulins following allogeneic bone marrow transplantation. Acta Haematol. 109(3), 124–128. https://doi.org/10.1159/000069285 (2003).
A-C N, D. S., O, J. M. P. L. & R, M R Allogeneic stem cell transplantation: low immunoglobulin levels associated with decreased survival. Bone Marrow Transpl. 41(3), 267–273. https://doi.org/10.1038/sj.bmt.1705892 (2007).
Mikael, S. et al. Hypogammaglobulinemia in children after allogeneic hematopoietic stem cell transplantation: a cytokine mediated immunoglobulin isotype class switch arrest? Pediatr. Blood Cancer. 62(5), 890–896. https://doi.org/10.1002/pbc.25409 (2015).
Elena, E. P. et al. Update on the use of immunoglobulin in human disease: a review of evidence. J. Allergy Clin. Immunol. 139, S1–S46. https://doi.org/10.1016/j.jaci.2016.09.023 (2017).
Jimenez-Zepeda, V. et al. Immunoparesis and polyclonal immunoglobulin recovery after auto-SCT for patients with multiple myeloma treated at a single institution. Leuk. Lymphoma 59(8), 1920–1926. https://doi.org/10.1080/10428194.2017.1403026 (2018).
Alessandro, M., Franco, L. & Lorenzo, M. Human NK cells: from HLA class I-specific killer Ig-like receptors to the therapy of acute leukemias. Immunol. Rev. 224(0), 58–69. https://doi.org/10.1111/j.1600-065X.2008.00651.x (2008).
Legrand, N., Dontje, W., van Lent, A., Spits, H. & Blom, B. Human thymus regeneration and T cell reconstitution. Semin. Immunol. 19(5), 280–288. https://doi.org/10.1016/j.smim.2007.10.001 (2007).
Mielke, S. et al. Reconstitution of FOXP3 + regulatory T cells (Tregs) after CD25-depleted allotransplantation in elderly patients and association with acute graft-versus-host disease. Blood 110(5), 1689–1697. https://doi.org/10.1182/blood-2007-03-079160 (2007).
Michael, B. et al. Up-regulation of NK cell activating receptors following allogeneic hematopoietic stem cell transplantation under a lymphodepleting reduced intensity regimen is associated with elevated IL-15 levels. Biol. Blood Marrow Transpl. 14(3), 290–300. https://doi.org/10.1016/j.bbmt.2007.12.490 (2008).
Valdés-Ferrada, J. et al. Peripheral blood classical monocytes and plasma interleukin 10 are Associated to Neoadjuvant Chemotherapy response in breast Cancer patients. Front. Immunol. 11, 1413. https://doi.org/10.3389/fimmu.2020.01413 (2020).
Wang, J. et al. Prognostic nutritional index combined with NLR to construct a survival prediction model and decision analysis of patients with muscle-invasive bladder cancer after surgery. Cancer Med. 12(13), 14207–14224. https://doi.org/10.1002/cam4.6088 (2023).
Xu, Z., Yao, X., Shi, X. & Du, X. Significance of preoperative peripheral blood neutrophil-lymphocyte ratio in predicting postoperative survival in patients with multiple myeloma bone disease. World J. Clin. Cases. 10(14), 4380–4394. https://doi.org/10.12998/wjcc.v10.i14.4380 (2022).
Yang, Y., Liu, Z., Wang, H. & Zhang, G. HLA-E binding peptide as a potential therapeutic candidate for high-risk multiple myeloma. Front. Oncol. 11, 670673. https://doi.org/10.3389/fonc.2021.670673 (2021).
Acknowledgements
This work was financially supported by National Natural Science Foundation of China (82270175, 82470148), Natural Science Foundation of Fujian Province of China (2021J02040), Joint Funds for the Innovation of Science and Technology of Fujian Province (2023Y9173), National Key Clinical Specialty Discipline Construction Program (2021-76) and Fujian Provincial Clinical Research Center for Hematological Malignancies (2020Y2006).
Author information
Authors and Affiliations
Contributions
Z. Xu designed this program. K. Chen, H. Liang, and W. Zhang conducted the data collection and analysis. Z. Yu, G. Guo, H. Zheng, Y. Huang, L. Liu, J. Lin, J. Long, R. Pan, X. Chen, C. Wang produced the manuscript which was checked by W. Zhang and Z. Xu. All the authors have confirmed the submission of this manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was approved by Ethical Committee of Fujian Medical University Union Hospital and informed consent was obtained from each participant.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, K., Liang, H., Yu, Z. et al. Analysis of early efficacy and immune reconstitution after autologous hematopoietic stem cell transplantation in multiple myeloma. Sci Rep 15, 1222 (2025). https://doi.org/10.1038/s41598-024-84047-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-84047-2
Keywords
This article is cited by
-
Advances in immune microenvironment profiling during multiple myeloma progression and therapy
International Journal of Hematology (2025)