Abstract
The Luangwa giraffe (Giraffa tippelskirchi thornicrofti), a subspecies of the Masai giraffe endemic to the Luangwa Valley of northeastern Zambia, inhabits an increasingly human-modified landscape. Accurate and current population estimates are critical to evaluating their status and identifying effective conservation strategies. However, sparse monitoring since the early 1900s has limited inferences about population size, structure, and range. To address this, we conducted the most spatially extensive and systematic survey to date of Luangwa giraffe across its distribution, extending survey effort 120 km south of their officially recognized extent. Using spatial capture recapture modeling, we estimated 651–890 giraffe and an overall density of 0.04–0.05 giraffe/km2. Density decreased to nought beyond 7.5 km from permanent rivers, consistent with preferred forage concentrated in riparian areas. Increasing giraffe density estimates up to a threshold of the Human Footprint Index suggested that limited human presence may have negligible consequences on movement and resource selection. This was likely due to suitable habitat and minimal conflict despite human presence. However, without mitigating land-use planning, rapid land conversion threatens human-giraffe coexistence. An even sex ratio and small proportion of subadults implied a stable population, but sex-biased and temporal dynamics in space use, impacts of predation, and stochastic risks necessitate continued monitoring. This study highlights the value of systematic large-scale monitoring and opportunities for data integration across long-term monitoring programs to evaluate factors driving Luangwa giraffe dynamics and to inform science-based conservation of this unique and isolated population.
Similar content being viewed by others
Introduction
The giraffe (Giraffa spp.) is an iconic African megaherbivore with widespread natural distribution across 21 countries in sub-Saharan Africa. However, giraffe have experienced population declines of approximately 25–30% since the 1990 s1, from an estimated 155,000, to 117,000 in 20202,3. Key threats to giraffe are predominantly anthropogenic, ranging from habitat loss and fragmentation due to agriculture and urban/infrastructure development, to poaching, civil unrest, anthroponotic disease, and climate change1. Historically, giraffe existed largely outside protected areas, but are now increasingly confined to smaller, isolated protected areas which in turn threatens population viability (Newmark 2008). Subsequently, the International Union for Conservation of Nature (IUCN) uplisted giraffe as a single species from Least Concern to Vulnerable in 20161. Long-term, accurate, and up-to-date estimates of population density (i.e., abundance and distribution) for all four giraffe species are critical to monitoring and better managing their conservation status.
The Luangwa (or Thornicroft’s) giraffe (G. tippelskirchi thornicrofti) is a unique subspecies of the Masai giraffe (G. tippelskirchi)4,5,6, and consists of a single isolated population in the Luangwa Valley of northeastern Zambia7. The Great Rift Valley and its escarpment biogeographically isolate the population from its closest neighboring populations that are more than 400 km to the northeast in Tanzania. Elevational changes of the continental rift physically hinder movement and act as a barrier to gene flow for many taxa, ranging from avian8to small and large mammals9, including giraffe10. As a result, the endemic Luangwa giraffe at the southern end of the Great Rift Valley is vulnerable to stochastic risks such as disease and natural catastrophes that can amplify genetic inbreeding depression, bottlenecking, and the threat of extinction11. Natural forces such as climate patterns across multiple time scales12,13 can influence giraffe population viability, while anthropogenic pressures also threaten giraffe habitat14, alter their movement15,16, and harm their health17.
Luangwa giraffe are listed as Vulnerable on the IUCN Red List18, with an apparent stable population size. In the early twentieth century, abundance estimates of the Luangwa giraffe fluctuated widely due to differences in survey regions and methodologies. Estimates ranged from fewer than 100 individuals at the turn of the century to 150 in the 1960s, but also upwards of 500 in the 1920s and more often closer to 30019,20,21. In the 1970s, the population was estimated at approximately 30022. Field efforts in 1973–2003 to survey giraffe using vehicle-based approaches were generally restricted to their core range along the eastern bank of the Luangwa River in South Luangwa National Park (SLNP)23. In 2013, following a similar field method, Berry and Bercovitch23 estimated approximately 110 giraffe along the Luangwa River, which they extrapolated to a population size of 500–600 for the entire valley. Their approach involved a density index to partially address imperfect detection but not sighting distance or the spatial variation in giraffe distribution. Subsequent unstructured road surveys in 2016–2018 estimated 555 Luangwa giraffe in a limited area of SLNP (mean of 407–645 across years24). However, variation in spatial survey effort was not modeled, potentially reducing accuracy and precision25,26. Conservation partners including research organizations, law enforcement, and private tourism and landowners have also been collecting giraffe sightings from distance-based surveys, aerial patrols, and opportunistic community science, with sightings helping to initiate a long-term database of giraffe identities and encounters. Yet, systematic survey and robust estimation of the entire Luangwa giraffe population that account for variation in effort and their distribution have been lacking.
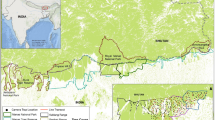
An important consideration in monitoring population size is accurate range demarcation. Historical (twentieth century) and contemporary (twenty-first century) accounts indicate that the Luangwa giraffe range extends along the western bank of the permanent Luangwa River27, having expanded north and south. However, assessing the pattern of range expansion has been challenging due to the lack of spatially comprehensive and standardized surveys and records. Berry and Bercovitch23 described a presumed range centered around the Luangwa River in SLNP between the Chibembe and Msanzara Rivers (Fig. 1). However, opportunistic and research sightings in recent decades suggest that the giraffe range extends farther, and on both sides of the Luangwa River across a patchwork of different land uses. The landscape includes a network of national parks (NPs), private game reserves (PGRs), and game management areas (GMAs) that buffer NPs while allowing specific, limited human activities. Multiple non-commercial community forests managed by locally-elected groups also occur within the range. The most current published range2,3 extends the presumed range by approximately 50 km north past the Chibembe River and SLNP to include Luambe NP (LNP) east of the Luangwa River. Sightings farther north in North Luangwa NP have been rare and are likely vagrants28, but are reflected in the IUCN Red List range1. To the south, the most current range description maintains the extent of Berry and Bercovitch3,23, while the IUCN Red List range extends farther south into Chisomo GMA1. However, giraffe have been consistently sighted even farther south for the past two decades, and on both sides of the river by community members (West Petauke GMA and PGRs on the western and eastern sides, respectively; N. Carruthers personal communications). As the Luangwa Valley has a matrix of varying habitat and anthropogenic pressures, and given the variation in density of Luangwa giraffe across its core and broader range, a comprehensive range assessment is needed to support effective monitoring and conservation.
Map of the road-based search-and-encounter survey conducted in August to November 2023 of Luangwa giraffe in the Luangwa Valley in northeastern Zambia. The region includes four national parks, eight game management areas, and seven private game reserves. Surveys extended beyond the current documented range by O’Connor et al.3. The town of Mfuwe (red pin) serves as the primary tourism hub for South Luangwa National Park. Basemap shows the terrain elevation.
In 2023, we conducted the most spatially extensive and systematic survey of Luangwa giraffe across their known distribution. We implemented a road-based search-and-encounter survey during the dry season along the Luangwa River, including SLNP, LNP, adjacent GMAs, and PGRs (Fig. 1). In search-and-encounter surveys, detections are made by searching along routes rather using a set of detectors at fixed locations, such that detections can occur anywhere along the survey routes29. In particular, we extended the survey effort south of the current range description. We used spatial capture-recapture (SCR) to estimate density, thereby accounting for imperfect detection probability due to the varying distances between giraffe locations and survey effort30,31. The SCR framework also enabled assessment of human pressure and resource availability on space use at multiple ecological scales32,33. We considered proximity to permanent rivers, expecting greater giraffe density closer to water due their dependency on riparian forage34,35,36 and the importance of surficial water during the dry season when water is scarce. We also evaluated the Human Footprint Index37 to consider the impact of anthropogenic pressure, ranging from lower in NPs to intermediate and heavy in agricultural and developed environments. We expected lower giraffe density with larger ranges of individual movement in areas with increased human footprint16 given the heterogeneous land use15 and associated risks of environmental and land use change14. We discuss findings and compare the updated Luangwa giraffe abundance and distribution against previous estimates, while acknowledging differences in spatial extent and analytical methods. Finally, we offer recommendations for future research, conservation, and management of the single existing population of Luangwa giraffe across the extent of its range.
Methods
Study site
The Luangwa Valley is part of the East African Rift Valley and is characterized by the free-flowing Luangwa River with numerous oxbow lakes, lagoons, and alluvial floodplains. The region includes 58,163 km2 of protected land, comprising four NPs (29%): South Luangwa (9,050 km2), North Luangwa (4,636 km2), Luambe (331 km2), and Lukusuzi (2,614 km2); eight GMAs (69%, 39,915 km2); and seven PGRs (3%, 1,617 km2). With no major fencelines, wildlife roam freely between NPs, GMAs, and PGRs. Diverse habitat includes riparian forests (Trichilia emetica, Tamarindus indica, Diospyros mespiliformis, Faidherbia albida, and Kigelia africana), mopane forests (dominated by Colophospermum mopane), munga woodlands (Combretum, Terminalia and Vachelliaspp.), thickets, scrub, and open grasslands38.
Photographic surveys
From August-November 2023, we conducted an individual-based photographic survey of Luangwa giraffe. We divided the road network into 10 fixed survey routes covering SLNP, LNP; Lupande, Lumimba, and Sandwe GMAs; the Luembe hunting block in West Petauke GMA; and Nyakolwe, Nyamvu, Nkalamu, Munyamadzi, and Kazumba PGRs (Fig. S1). Teams endeavored to drive the entirety of any given route daily for three consecutive days, with daily driven lengths ranging 26–120 km. A total of 2,919 km was driven on 1,408 km of track, with distances up to 25 km from the river to enable sufficient effort along and away from rivers for avoiding alignment of survey effort with landscape features that could bias abundance estimation39. Surveying across the 4-month period balanced logistics for maximizing survey area and capturing density patterns over the dry season with demographic closure for SCR analysis. The survey effort extended southwest along the Luangwa River and 120 km beyond the southern extent of the range described by O’Connor et al.3. In comparison, the core range, between the Chibembe and Msanzara Rivers along the Luangwa River, is approximately 170 km long.
Each survey team included at least one dedicated observer and one driver. Driving 20–40 km/h, teams collected data on all giraffe visible within approximately 300 m of either side of the road. Teams photographed the right and left lateral sides of each encountered giraffe, documenting age class, sex, and herd composition. The right flank of giraffe was preferred to increase consistency for identification purposes, but left flanks were also photographed when the right was not possible (e.g., on the other side of the river). The team’s location upon observation and distance and bearing to the giraffe were recorded with a rangefinder and integrated GPS and compass in a Canon 7D Mark II to subsequently project the location of the giraffe. Sex and age classes (adult, subadult, calf) were determined based on physical characteristics including size, height, genitalia, and ossicone size and shape40.
Giraffe coat patterns are unique and constant, enabling individual identification41. To determine identities from photographs, we used a pattern recognition software developed for giraffe called GiraffeSpotter42 which includes a large database of existing Luangwa giraffe individuals from sightings since 2019. We based identification on uniqueness of right sides amongst the photographed giraffe or confirming at least the right side through the database, and excluded detections of individuals with only left-side photographs from the survey that could not be matched to an individual in the database with both right- and left-side images available. By discarding left-side-only detections that could not be matched to a known individual, we avoided doubly counting individuals but may also have introduced slight negative bias in the estimation through unmodeled individual heterogeneity in detection probability43.
Spatial capture-recapture (SCR)
We estimated Luangwa giraffe population size and distribution (collectively, density) with an SCR model. SCR models have separate sub-models for the distribution of individuals’ activity centers and the detection process. To define the region of inference, or state-space, we applied a 15-km buffer around the survey routes and 263 aerial sightings from independent tracking flights in 2023 (Zambian Carnivore Programme, unpublished data), with the 15-km buffer exceeding previously observed daily movements of Luangwa giraffe by five times and the conservative radius of a male home range by three times assuming a circular shape27. We removed two pixels (50 km2) in the southern part of the state-space that corresponded to the majority (86%) of a 29 km2 fenced conservation area that excluded giraffe. Three other pixels that contained the remaining 14% of the fenced area were kept in the state-space as the fenced area made up only 3–7% of each of the pixels, thus leaving the majority of those pixels accessible to giraffe. As a result, the state-space was a total of 17,000 km2. South of the state-space, a fenced PGR (Pia Manzi) and unsuitable rocky hills were not considered available habitat for Luangwa giraffe. This essentially encompassed the entire possible Luangwa giraffe range and ensured inclusion of all individuals potentially detected over the course of the survey.
We used an inhomogeneous point process to model the distribution of individuals in order to consider habitat covariates on density44. We overlaid a 5 × 5 km grid over the state-space, resulting in J = 2,242 grid cells. The intensity μ of the point process, representing the number of expected activity centers per grid cell, was modeled as a log-linear function of habitat covariates β45,46. Drivers of giraffe resource selection, and therefore where individuals place their activity centers, include the availability of water with associated woody vegetation, and anthropogenic impacts. Therefore, habitat covariates on μ included the linear effect of distance to the permanent Luangwa and Chibembe (also known as the Mupamadzi) Rivers, and the linear and quadratic effects of the Human Footprint Index (HFI37). Distance to permanent rivers in the surveyed cells ranged 0–36.1 km [mean = 7.7 km, standard deviation (SD) = 7.0 km]. HFI is a composite metric at the 1 km2 resolution that ranges 0–50 and quantifies human pressure on the landscape37. Areas are considered ‘wilderness’ if HFI < 1, intact if HFI < 4, and modified if HFI ≥ 447. HFI values were averaged within each surveyed grid cell (Fig. S2; values ranged 0.6–18.6 ;mean = 6.7, SD = 3.1) and were scaled and centered to facilitate parameter estimation. HFI and distance to river were not correlated (Pearson’s r = −0.01, t = −0.35, df = 680, p = 0.73), i.e., areas of higher HFI were not necessarily close to permanent rivers.
We summarized detections as counts per giraffe per grid cell. Counts were modeled as a binomial distribution according to survey frequency of the grid cell and a detection probability. Detection probability followed a half-normal distribution, declining from a baseline probability p0 according to a movement parameter σ and the Euclidean distance between the grid center and the individual’s unobserved activity center. Given that male giraffe generally travel farther than females17,27 and age can influence movement and behavior48, we modeled p0 as a function of age class and sex covariates (α) with a complementary log–log linear regression and as covariates (δ) on σ with a log-linear regression. Females and adults were the reference sex and age class. We excluded calves because they likely violated the SCR model assumption of independence amongst individuals. While SCR can be robust to moderate levels of cohesion and herd size49,50 that emerge due to fission–fusion dynamics across all age classes in giraffe48,51, calves in this study were never detected alone and had identical detection histories with at least one adult female. We modeled the sex and age class of individuals as Bernoulli random variables with probabilitiesΨmale andΨsubadult, respectively.
We used a Bayesian approach with data augmentation to implement the SCR model52. The total population size N was modeled as a proportion of a theoretical superpopulation M. Individuals of M are members of N according to a binary indicator modeled as a Bernoulli random variable with probability Ψ. Thus, N is a derived parameter and calculated as the sum of the binary indicator over M, which must be sufficiently larger than N to avoid truncating the posterior distribution of N. Here, we used \(M=7*n\), where n is the number of detected individuals.
Data were fit to the SCR model using the ‘nimbleSCR’package v.0.2,145in R version 4.4.053. A habitat mask defined the state-space from the rectangular grid generated by the minimum and maximum x and y coordinates of the state-space. We used the ‘dbinomLocal_normal’ function to model the observed number of detections per giraffe per grid cell as a binomial draw from as many trials as the number of times the grid cell was surveyed (up to 17), thus accounting for variable effort across grid cells due to variation in repeat surveys of routes54. The ‘dbinomLocal_normal’ function also increased efficiency by considering only the local state-space for each individual and its detections when evaluating detection probability. Survey routes were discretized according to the 5 × 5 km gridded state-space, and activity centers were estimated in continuous space and then assigned to the corresponding pixels. We used vague priors (uniform and normal distributions) for parameters, and ran 3 chains each with 75,000 iterations, a burn-in of 10,000, and a thin rate of 3. This resulted in 64,998 total posterior iterations. We confirmed chain convergence with the Gelman-Rubin statistic (R-hat ≤ 1.155) and visual inspection of trace-plots, and confirmed minimum effective sample sizes of at least 400 for all parameters. We reported point estimates and 95% Bayesian Credible Intervals (BCI) for parameters, and the coefficient of variation [\(CoV= \text{log}(standard deviation/mean)\)]of estimated abundance per grid cell. We assessed goodness of fit by comparing observed numbers of individual-and-grid-specific encounters against expected values56 using 5,000 simulations. An observed to expected ratio of 1 suggests perfect fit and deviations indicate lack of fit57.
Results
The survey yielded 533 detections of individual giraffe, with a median herd (group) size of 3 (mean = 3.9; range: 1–17; interquartile range: 1–5). After removing 24 unidentifiable detections, 36 (7%) uncertain detections based on only left-side pictures, and 34 detections of 18 calves, 439 detections (82%) of n = 277 unique giraffe remained for analysis. Slightly more females than males were detected (F/M: 151/126), and more adults than subadults (adults/subadults: 242/35). Most individuals (165; 60%) were detected once, but the remaining 112 individuals were detected an average 2.4 times each (maximum 5 times), resulting in redetections making up 37% of the total detections. Distances between redetections averaged 1.1 km, but reached a maximum of 5.6 km, and 49 individuals (18%) were detected in multiple grid cells (up to 3). Most redetections (93%) of individuals occurred on the same survey route as their first detection.
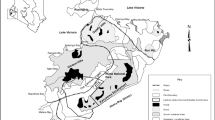
Estimated median abundance was \(\widehat{N}\) = 760 giraffe (95% BCI: 651–890) (Table S1), resulting in an overall density of 0.04 giraffe/km2 (95% BCI: 0.04–0.05, Fig. 2). Most individuals were adults (Ψsubadult = 0.10, 95% BCI: 0.07–0.15), consistent with survey observations, and evenly split across sexes (Ψmale = 0.49, 95% BCI: 0.42–0.57). Giraffe density decreased with distance from rivers (βdistance_to_river = −0.45, 95% BCI: −0.69–0.33), reaching 0 giraffe/km2 (95% BCI: 0.00–0.003) beyond 7.5 km (Fig. 3). Density increased with increasing HFI (βHFI = 1.82, 95% BCI: 1.53–2.13) up to a maximum of 0.1 giraffe/km2 when HFI = 13.6 (95% BCI: 11.1–18.1), but then decreased thereafter due to a significant parabolic effect of HFI on density (βHFI_squared = −0.86, 95% BCI: −1.14—−0.61; Fig. 3). See Fig. S3 for CoV of estimated density.
Estimated (realized) density of Luangwa giraffe in the Luangwa Valley in northeastern Zambia, based on a spatial capture-recapture analysis of a search-and-encounter survey conducted in 2023. Two crossed out cells were excluded from estimation as they correspond to the majority of a fenced area (25 km2 of 29 km2) that excluded giraffe.
Effect of distance to river (left) and human footprint index (HFI, right) on expected density of Luangwa giraffe in the Luangwa Valley in northeastern Zambia, based on a spatial-capture recapture analysis of a search-and-encounter survey conducted in 2023. Median estimates (black regression line) with 95% BCI (colored ribbons) are shown. Rugs display the distribution of habitat values within the state-space.
Estimated movement (\(\widehat{\sigma }\)) varied by age, sex, and HFI (Fig. 4, Table S1). Females and subadults had smaller ranges of movement than males and adults respectively, so adult males had the largest range of movement (\(\widehat{\sigma }\) ADM = 2.5 km, 95% BCI: 2.2–3.0) and subadult females had the smallest range of movement (\(\widehat{\sigma }\) SADF = 1.6 km, 95% BCI: 1.3–2.0). Similar to abundance, \(\widehat{\sigma }\) initially increased with greater HFI (δHFI = 0.26, 95% BCI: 0.04–0.44) before decreasing due to a significant parabolic effect (δHFI_squared = −0.18, 95% BCI: −0.34–0.01). Estimates of σADF reached a maximum of 2.2 km when HFI = 11.1 (2.5% lower limit HFI = 8.2, no upper limit) (Fig. 5).
Posterior distributions for sex and age-class specific sigma (left) and baseline detection probability (right) for Luangwa giraffe in the Luangwa Valley in northeastern Zambia, based on a spatial-capture recapture analysis of a search-and-encounter survey conducted in 2023.
Effect of human footprint index (HFI) on expected range of movement for adult female Luangwa giraffe in the Luangwa Valley in northeastern Zambia, based on a spatial-capture recapture analysis of a search-and-encounter survey conducted in 2023. Median estimates (black regression line) with 95% BCI (ribbon) are shown. Rug displays the distribution of HFI values within the state-space.
Detection varied by age and sex (Fig. 4, Table S1). Males had lower detection (ɑM = −0.8, 95% BCI: −1.31–0.31) than females, while subadults had higher detection than adults (ɑsubadult = 1.15, 95% BCI: 0.38–2.12).
A goodness of fit statistic = 0.97 indicated sufficient model fit (Fig. S4).
Discussion
As the most spatially comprehensive and systematic survey effort of Luangwa giraffe, this study provides a critical update on the abundance and range of the only population of this Masai giraffe subspecies. The search-and-encounter survey estimated 651–890 giraffe and confirms that the subspecies’ range extends substantially farther south than previously documented3,18. Density reached 0.5–1.0/km2 in some areas along the river in SLNP, but overall density across the landscape was 0.04–0.05 giraffe/km2: higher than the desert-dwelling giraffe of northwestern Namibia (0.01 giraffe/km258) but lower than many other giraffe populations in Africa, including elsewhere in Namibia (0.34–0.39/km2 in fenced Ongava GR59), western Uganda (1.1–1.2/km2 in unfenced Murchison Falls NP17), southern Kenya (1.2 ± 0.4/km2in partially fenced Tsavo NP60), and northern Tanzania (1.9 ± 0.1/km2 in partially fenced Tarangire NP61). This study also estimated population age- and sex-structure, reinforced aspects of basic giraffe ecology, and highlighted the need for continued monitoring and human-giraffe coexistence.
The state-space was 1.8x larger than the current documented range of 9,356 km23. The survey extended 120 km south of the range but excluded 1,095 km2 mostly at the northern end of the range towards North Luangwa NP where rugged terrain likely results in reduced habitat associations with giraffe distribution (Fig. 1). In the area overlapping the documented range, we detected 222 individuals and estimated a mean of 612, slightly higher but generally consistent with recent estimates of 500–60023,24. The population in the documented range therefore appears stable in recent years, consistent with estimates from various analytical approaches. Where we targeted survey efforts beyond the southern range border to resolve uncertainties in range limits, we detected 55 individuals and estimated a mean of 150. We expect negligible giraffe densities farther south and farther north of the study area, due to the presence of rocky hills north and south of the study area that are poor habitat for giraffe and large distances to permanent river farther north of the study area (average 16 km, with 75% of grid cells > 7.5 km). Therefore, our spatially explicit density estimates are likely to cover most, if not all, areas with Luangwa giraffe.
Patterns in Luangwa giraffe density and movement demonstrate an ability to persist in and select resources in moderately and heterogeneously human-modified landscapes. A likely factor is the lack of significant human-giraffe conflict. Giraffe density (second order resource selection32) was highest near permanent rivers and decreased to nonexistent by 7.5 km (Fig. 3), aligning with forage preference of Luangwa giraffe for riparian woodlands and alluvial areas over mopane and miombo woodlands farther afield34. Indeed, Berry27 noted that Luangwa giraffe did not travel beyond 7 km from the Luangwa River and giraffe detections in this survey did not exceed 6.2 km (Fig. S5). Large swathes of these floodplains also support subsistence agriculture62,63. Yet, Luangwa giraffe cause minimal crop damage nor other conflict with humans, in contrast to other megaherbivores such as African savannah elephant (Loxodonta africana) and hippopotamus (Hippopotamus amphibius), which can damage crops and infrastructure and directly cause loss of human life64,65,66.
Reciprocal human-giraffe tolerance is further supported by the effect of HFI on giraffe density. With widespread human footprint on the landscape and only 16% of grid cells considered wilderness or intact habitat (HFI < 4), median giraffe density increased slightly with HFI and reached a maximum of 0.1 giraffe/km2 when HFI = 13.6 (Fig. 3). In addition to minimal human-giraffe conflict, cultural aversions to consuming giraffe meat reduce direct poaching threats24. This differs from giraffe populations such as Masai giraffe in southern Kenya (Tsavo Conservation Area67) and other herbivores in the Luangwa ecosystem for which bushmeat poaching is a key threat — especially in GMAs68. Additionally, Luangwa Valley is host to a significant wildlife tourism economy69,70, especially around Mfuwe town (Fig. 1), that encourages tolerance of wildlife presence. Humans therefore pose minimal direct threat to Luangwa giraffe survival, enabling somewhat higher densities even in areas with greater human footprint.
Parabolic relationships between HFI and Luangwa giraffe density and movement (third order resource selection32) nevertheless indicate some negative effect of human footprint. Below HFI = 8.2, both density and range of movement increased with greater HFI, suggesting individuals overlapping more and travelling farther to access increasingly limited resources in a human-impacted landscape16,71. However, above this threshold, especially after HFI > 18.1, fitness risks from increased human pressure may outweigh travel benefits, thus reducing density and restricting ranges of movement. Similar movement patterns have been documented with Nubian giraffe (G. camelopardalis camelopardalis) across a gradient of HFI exposure15. Risks to increased movement as a result of a larger σ, especially in developed areas, include higher snaring by-catch17,72 and powerline electrocutions from rapid electrification and infrastructure growth24 Rachel McRobb, Conservation South Luangwa personal communication). An average of 1.6 (range: 0–4) Luangwa giraffe have been de-snared annually since 2013 (ZCP, unpublished data). Furthermore, the range in HFI values in which density and range of movement likely each reached their inflection points and began decreasing (8.2 < HFI < 18.1) suggests complex interplay between individual distribution and space use as a result of human pressure; the degree of spatial overlap between individuals and ranges of movement likely depend on the interaction of multiple fine scale environmental features, including predator–prey dynamics and spatial heterogeneity73. So, while Luangwa giraffe have and can occur in a moderately human-modified landscape, continued study and strategic and enforced land-use planning will be increasingly important for habitat protection and ensuring the ecological and economic benefits of wildlife to communities.
The systematic survey was designed to support precise and accurate inferences about population density of the Luangwa giraffe. Sufficient redetections are necessary to precisely estimate detection probability and ultimately density74,75, and repeat surveys on consecutive days increased redetections (37% of total detections) compared to previous studies that achieved fewer redetections (< 30%) over longer surveys with more effort24. However, the short interval between repeat surveys limited spatial redetections and restricted the interpretation of the range of movement to a shorter time frame compared to the more traditional home-range scale of movement when surveys are conducted over longer periods such as full seasons. Still, almost 20% of giraffe had spatial redetections at the 5 × 5 km resolution, estimates of σ were precise [(standard deviation/mean) < 0.2, Table S1], and males moved greater distances than females as expected based on previous studies (Fig. 415,16,27). Characterization of full home range size and estimation of habitat use at finer spatial resolutions would require more repeat surveys or greater time between repeat surveys, and/or GPS tagging of individuals15,76. Surveys extended into the wet season would also help to capture seasonal variation due to differences in forage availability71,77,78. Future surveys may also consider off-road transects to further increase the degree of two-dimensional sampling39 and specialized SCR models to account for strong associations with habitat patterns in highly structured environments, particularly linear features like rivers, that can result in non-symmetrical or elongated home range geometries79,80. Approaches like kernel utilization distributions81,82 and dynamic Brownian bridge movement models83,84 may be needed to accurately estimate focal area, space use, and landscape connectivity. Finally, the sex ratio was near parity during the survey, consistent with that reported by Phillips24in SLNP over a 3-year period. However, the distribution of sexes across the landscape can vary by season and habitat due to feeding ecology85, movement patterns86, and over time from sex-biased sources of predation, mortality, and injury17,87,88. While we do not expect further range expansion in Luangwa giraffe, unless habitat patterns or associations change in the future, monitoring the distribution of sexes over the landscape may detect sex-biased movements and demographic-based population processes89. Targeted study of Luangwa giraffe movement over time and space is therefore recommended to complement density estimation.
The estimated proportion of subadults and observed calves suggest population turnover that is necessary for population persistence. However, subadults (7–15% of the estimated population) appeared fewer than in other giraffe populations where they are almost twice as prevalent, such as Nubian giraffe in Murchison Falls NP (Uganda17) and Soysambu Conservancy (Kenya90). Instead, the age structure resembled that of Nubian giraffe in Lake Nakuru NP (Kenya) where high lion density influenced giraffe age structure90. With SLNP being a lion stronghold91, predation may be contributing to the stable Luangwa giraffe population in their core range by suppressing recruitment, survival, and population growth. Direct predation events are challenging to document88, and we found no evidence at the 5-km scale to suggest calves and subadults selected areas of greater human presence for safety (e.g., human-shield hypothesis92). Based on post-hoc exploration, HFI values were similar across areas where giraffe of different age classes were detected (Tukey-adjusted pairwise means comparisons: tadult/calf = 0.31, tadult/subadult = −1.19, tcalf/subadult= −1.00; all p > 0.46). Climate variability and anomalous high rainfall can also reduce giraffe survival by increasing disease prevalence and cover for predators12, altering forage nutrition and availability, and potentially even movement patterns93.
This study provides an important baseline for long-term monitoring to identify density trends, estimate survival and recruitment rates, and understand predator–prey dynamics of the Luangwa giraffe. Ongoing Luangwa ecosystem-wide research has generated local datasets (e.g., distance sampling, camera trapping, aerial surveys, opportunistic sightings) that could be combined in an integrated framework to estimate Luangwa giraffe population trends and dynamics at scale94,95. Appealing properties of integrated approaches may include increased precision and accuracy of estimates from having more data and uncertainty being propagated through the model96. New parameters may also become identifiable despite no explicit data on them being collected97,98, such that quantifying and understanding processes like source-sink dynamics become more tractable99,100. Analytical developments can be leveraged to extract signals from datasets with less individual resolution, such as detections with entirely or partially missing identities43 and count and presence-absence data from citizen science efforts101,102,103. Partner collaborations are particularly valuable for data collection in areas like range peripheries with sparse data. Integrated analysis frameworks therefore provide an efficient approach for long-term datasets to estimate population dynamics and trends critical for informing conservation.
Giraffe are ecologically important in the Luangwa Valley ecosystem, engineering habitat and vegetation structure and promoting biodiversity, in part due to their unique morphology that enables exclusive foraging access to vegetation104. As the only population of this subspecies, the endemic Luangwa giraffe has special conservation status. This range-wide survey of Luangwa giraffe provides critical updates on their population density, and underscores the value of systematic efforts to understand patterns in population abundance, structure, and impact of anthropogenic pressures. Results indicate that human-giraffe coexistence is possible to a degree, but human presence in the Luangwa Valley — as in many other regions with giraffe populations — will likely increase and continue to threaten to reduce the availability of natural habitat. Robust monitoring is critical for conserving vulnerable wildlife species and populations in the face of compounding effects from habitat degradation, resource overexploitation, climate change, inadequate environmental education, and uncoordinated or weak land use governance105,106. We recommend long-term monitoring of the Luangwa giraffe and leveraging the range of available datasets and quantitative approaches to better understand its ecology, track population status, and assess viability through robust analytical approaches.
Data availability
Data and script to run Luangwa giraffe SCR model and calculate GOF can be found on the Figshare repository at https://figshare.com/articles/dataset/27,752,955.
References
Muller Z, Bercovitch FB, Brand R, et al Giraffa camelopardalis (amended version of 2016 assessment). IUCN Red List of Threatened Species (2018).
Brown M B, Kulkarni T, Ferguson S, et al Conservation status of giraffe: evaluating contemporary distribution and abundance with evolving taxonomic perspectives. https://doi.org/10.1016/B978-0-12-821139-7.00139-2(2022).
O’Connor, D. et al. Updated geographic range maps for giraffe, Giraffa spp., throughout sub-Saharan Africa, and implications of changing distributions for conservation. Mammal. Rev. 49, 285–299. https://doi.org/10.1111/mam.12165 (2019).
Coimbra, R. T. F. et al. Whole-genome analysis of giraffe supports four distinct species. Curr. Biol. 31, 2929-2938.e5. https://doi.org/10.1016/j.cub.2021.04.033 (2021).
Fennessy, J. et al. Multi-locus analyses reveal four giraffe species instead of one. Curr. Biol. 26, 2543–2549. https://doi.org/10.1016/j.cub.2016.07.036 (2016).
Winter, S., Fennessy, J. & Janke, A. Limited introgression supports division of giraffe into four species. Ecol. Evol. 8, 10156–10165. https://doi.org/10.1002/ece3.4490 (2018).
Fennessy, J. et al. Mitochondrial DNA analyses show that Zambia’s South Luangwa Valley giraffe (Giraffa camelopardalis thornicrofti) are genetically isolated. Afr. J. Ecol. 51, 635–640. https://doi.org/10.1111/aje.12085 (2013).
Behrends, G. J., Meheretu, Y. & Manthey, J. D. The Great Rift Valley is a more pronounced biogeographic barrier than the Blue Nile Valley for six Ethiopian Highland passerines in the eastern Afromontane biodiversity hotspot. Ornithology 141, 14. https://doi.org/10.1093/ornithology/ukae030 (2024).
Cuypers, L. N. et al. Biogeographical importance of the Livingstone Mountains in Southern Tanzania: comparative genetic structure of small non-volant mammals. Front Ecol Evol https://doi.org/10.3389/fevo.2021.742851 (2022).
Lohay, G. G. et al. Genetic evidence of population subdivision among Masai giraffes separated by the Gregory Rift Valley in Tanzania. Ecol. Evol. 13, e10160. https://doi.org/10.1002/ece3.10160 (2023).
Frankham, R. Inbreeding and extinction: island populations. Conserv. Biol. 12, 665–675. https://doi.org/10.1111/j.1523-1739.1998.96456.x (1998).
Bond, M. L., Ozgul, A. & DerekE, L. Effect of local climate anomalies on giraffe survival. Biodivers. Conserv. 32, 3179–3197. https://doi.org/10.1007/s10531-023-02645-4 (2023).
Pausata, F. S. R. et al. Changes in climate extremes in Zambia during green and dry Sahara periods and their potential impacts on hominid dispersal. Quatern. Sci. Rev. 321, 108367. https://doi.org/10.1016/j.quascirev.2023.108367 (2023).
Bond, M. L., Lee, D. E. & Paniw, M. Extinction risks and mitigation for a megaherbivore, the giraffe, in a human-influenced landscape under climate change. Glob. Change Biol. 29, 6693–6712. https://doi.org/10.1111/gcb.16970 (2023).
Brown M, Fennessy J, Crego R, et al Ranging behaviours across ecological and anthropogenic disturbance gradients: a pan-African perspective of giraffe (Giraffa spp.) space use. Proceedings of the Royal Society B. https://doi.org/10.1098/rspb.2023.0912(2023).
Knüsel, M., Lee, D. & Bond, M. Correlates of home range sizes of giraffes. Giraffa camelopardalis. Animal Behav. 149, 143. https://doi.org/10.1016/j.anbehav.2019.01.017 (2019).
Kasozi, H., Linden, D. W., Roloff, G. J. & Montgomery, R. A. Evaluating the prevalence and spatial distribution of giraffes injured by non-target poaching. J. Zool. 319, 152–162. https://doi.org/10.1111/jzo.13033 (2022).
Bercovitch F, Carter K, Fennessy J, Tutchings A (2018) Giraffa camelopardalis ssp. thornicrofti. The IUCN Red List of Threatened Species (2018).
Berry PSM The Luangwa Valley giraffe. Puku 71–92 (1973).
Pitman, C. R. S. A report on a faunal survey of Northern Rhodesia (Livingstone, 1934).
Zoological Society of London. Part 4: Order: Artiodactyla, Suborder: Ruminantia, Infraorder: Pecora, Family: Giraffidae, Genus: Giraffa. Trans. Zoological Soc. London 30, 139168 (1965).
Giraffe Conservation Foundation Country profile: Republic of Zambia giraffe conservation status report. Giraffe Conservation Fund (2022).
Berry, P. S. M. & Bercovitch, F. B. Population census of Thornicroft’s giraffe Giraffa camelopardalis thornicrofti in Zambia, 1973–2003: conservation reassessment required. Oryx 50, 721–723. https://doi.org/10.1017/S003060531500126X (2016).
Phillips S Estimating giraffe (Giraffa camelopardalis) population size and survival rates in South Luangwa Valley, Zambia. All NMU Master’s Theses (2022).
Link, W. A. Nonidentifiability of population size from capture-recapture data with heterogeneous detection probabilities. Biometrics 59, 1123–1130. https://doi.org/10.1111/j.0006-341X.2003.00129.x (2003).
Moqanaki, E. M. et al. Consequences of ignoring variable and spatially autocorrelated detection probability in spatial capture-recapture. Landscape Ecol 36, 2879–2895. https://doi.org/10.1007/s10980-021-01283-x (2021).
Berry, P. S. M. Range movements of giraffe in the Luangwa Valley, Zambia. Afr. J. Ecol. 16, 77–83. https://doi.org/10.1111/j.1365-2028.1978.tb00429.x (1978).
East, R. & Estes, R. D. African antelope database 1998 (IUCN/SSC Antelope Specialist Group, Gland, Switzerland and Cambridge, 1999).
Royle, J. A., Kéry, M. & Guélat, J. Spatial capture-recapture models for search-encounter data. Methods Ecol. Evol. 2, 602–611. https://doi.org/10.1111/j.2041-210X.2011.00116.x (2011).
Efford, M. Density estimation in live-trapping studies. Oikos. 106, 598–610. https://doi.org/10.2307/3548382 (2004).
Royle, J. A. & Young, K. V. A hierarchical model for spatial capture-recapture data. Ecology 89, 2281–2289. https://doi.org/10.2307/27650753 (2008).
Johnson, D. H. The comparison of usage and availability measurements for evaluating resource preference. Ecology 61, 65–71. https://doi.org/10.2307/1937156 (1980).
Royle, J. A., Fuller, A. K. & Sutherland, C. Unifying population and landscape ecology with spatial capture–recapture. Ecography 41, 444–456. https://doi.org/10.1111/ecog.03170 (2018).
Berry, P. S. M. & Bercovitch, F. B. Seasonal and geographical influences on the feeding ecology of giraffes in the Luangwa Valley, Zambia: 1973–2014. Afr J Ecol 55, 80–90. https://doi.org/10.1111/aje.12324 (2017).
Hart, E. E., Fennessy, J., Hauenstein, S. & Ciuti, S. Intensity of giraffe locomotor activity is shaped by solar and lunar zeitgebers. Behav. Proc. 178, 104178. https://doi.org/10.1016/j.beproc.2020.104178 (2020).
Leuthold, B. M. & Leuthold, W. Ecology of the giraffe in Tsavo East National Park, Kenya. Afr. J. Ecol. 16, 1–20. https://doi.org/10.1111/j.1365-2028.1978.tb00419.x (1978).
Mu, H. et al. A global record of annual terrestrial human footprint dataset from 2000 to 2018. Sci. Data 9, 176. https://doi.org/10.1038/s41597-022-01284-8 (2022).
Astle, W. L., Webster, R. & Lawrance, C. J. Land classification for management planning in the Luangwa Valley of Zambia. J. Appl. Ecol. 6, 143–169. https://doi.org/10.2307/2401534 (1969).
Efford, M. Non-circular home ranges and the estimation of population density. Ecology https://doi.org/10.1002/ecy.2580 (2019).
Strauss, M. K. L., Kilewo, M., Rentsch, D. & Packer, C. Food supply and poaching limit giraffe abundance in the Serengeti. Popul. Ecol. https://doi.org/10.1007/s10144-015-0499-9 (2015).
Foster, J. B. The giraffe of Nairobi National Park: home range, sex ratios, the herd, and food. Afr. J. Ecol. 4, 139–148. https://doi.org/10.1111/j.1365-2028.1966.tb00889.x (1966).
Smith S, Searche RE, Iology B GiraffeSpotter: a cloud-based photo-identification analysis tools for giraffe research. Accessible at: https://giraffespotter.org (2019).
Augustine, B. C. et al. Spatial capture–recapture with partial identity: an application to camera traps. Ann Appl Stat 12, 67–95. https://doi.org/10.1214/17-AOAS1091 (2018).
Royle, J. A., Chandler, R. B., Sollmann, R. & Gardner, B. Spatial capture-recapture 1st edn. (Academic Press, 2013).
Bischof R, Turek D, Milleret C, et al nimbleSCR: Spatial Capture-Recapture (SCR) Methods Using “nimble” (2022).
Zhang, W. et al. A flexible and efficient Bayesian implementation of point process models for spatial capture–recapture data. Ecology 104, e3887. https://doi.org/10.1002/ecy.3887 (2023).
Williams, B. A. et al. Change in terrestrial human footprint drives continued loss of intact ecosystems. One Earth 3, 371–382. https://doi.org/10.1016/j.oneear.2020.08.009 (2020).
Lavista Ferres, J. M. et al. Social connectedness and movements among communities of giraffes vary by sex and age class. Anim. Behav. 180, 315–328. https://doi.org/10.1016/j.anbehav.2021.08.008 (2021).
Bischof, R. et al. Consequences of ignoring group association in spatial capture–recapture analysis. Wildl. Biol. https://doi.org/10.2981/wlb.00649 (2020).
Sun, C. & Burton, A. C. The importance of independence in unmarked spatial capture–recapture analysis. Wildl. Biol. 2024, e01182. https://doi.org/10.1002/wlb3.01182 (2024).
Carter, K. D. et al. Social networks, long-term associations and age-related sociability of wild giraffes. Anim. Behav. 86, 901–910. https://doi.org/10.1016/j.anbehav.2013.08.002 (2013).
Royle, J. A. & Dorazio, R. M. Parameter-expanded data augmentation for Bayesian analysis of capture–recapture models. J. Ornithol 152, 521–537. https://doi.org/10.1007/s10336-010-0619-4 (2012).
R Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/ (2024).
Milleret, C. et al. A local evaluation of the individual state-space to scale up Bayesian spatial capture–recapture. Ecol. Evol. 9, 352–363. https://doi.org/10.1002/ece3.4751 (2018).
Gelman, A. & Rubin, D. B. Inference from iterative simulation using multiple sequences. Statist Sci 7, 457–472. https://doi.org/10.1214/ss/1177011136 (1992).
Choo, Y. R., Sutherland, C. & Johnston, A. A Monte Carlo resampling framework for implementing goodness-of-fit tests in spatial capture-recapture models. Methods Ecol. Evol. 15, 1653–1666. https://doi.org/10.1111/2041-210X.14386 (2024).
Molinari-Jobin, A. et al. Mapping range dynamics from opportunistic data: spatiotemporal modelling of the lynx distribution in the Alps over 21 years. Anim. Conserv. 21, 168–180. https://doi.org/10.1111/acv.12369 (2018).
Fennessy, J. T., Leggett, K. E. A. & Schneider, S. Distribution and status of the desert-dwelling giraffe (Giraffa camelopardalis angolensis) in northwestern Namibia. African Zoology 38, 184–188. https://doi.org/10.1080/15627020.2003.11657208 (2003).
Bonenfant, C., Stratford, K. & Périquet, S. Counting giraffes: A comparison of abundance estimators on the Ongava Game Reserve, Namibia. Hystrix 34, 1–7. https://doi.org/10.1101/2021.09.28.459593 (2023).
Njueini, J. K., Mwangi, E. M. & Montgomery, R. A. Seasonal variation in giraffe population density and abundance in the Tsavo landscape. Kenya. African J. Ecol. 62, e13215. https://doi.org/10.1111/aje.13215 (2024).
Lee, D. E. & Bond, M. L. Precision, accuracy, and costs of survey methods for giraffe Giraffa camelopardalis. J. Mammal. 97, 940–948. https://doi.org/10.1093/jmammal/gyw025 (2016).
de Filippo, C. et al. Genetic perspectives on forager-farmer interaction in the Luangwa Valley of Zambia. Am. J. Phys. Anthropol. 141, 382–394. https://doi.org/10.1002/ajpa.21155 (2009).
Styger, E. Rice production diagnostic for Chinsali (Chinsali District, Northern Province) and Mfuwe (Mwambe District, Eastern Province) (Atkinson Center for Sustainable Development, Cornell University, Ithaca New York, 2014).
Di Minin, E. et al. A pan-African spatial assessment of human conflicts with lions and elephants. Nat Commun 12, 2978. https://doi.org/10.1038/s41467-021-23283-w (2021).
Nyirenda, V. R., Myburgh, W. J., Reilly, B. K. & Phiri, A. I. Wildlife crop damage valuation and conservation: conflicting perception by local farmers in the Luangwa Valley, eastern Zambia. Intl. J. Biodiversity Conservation 5, 741–750. https://doi.org/10.5897/IJBC12.026 (2013).
van Houdt, S. & Traill, L. W. A synthesis of human conflict with an African megaherbivore; the common hippopotamus. Front. Conserv. Sci https://doi.org/10.3389/fcosc.2022.954722 (2022).
Muneza, A. B. et al. Socio-economic factors correlating with illegal use of giraffe body parts. Oryx 57, 457–466. https://doi.org/10.1017/S003060532200062X (2023).
Becker, M. S. et al. Wire-snare bushmeat poaching and the large African carnivore guild: Impacts, knowledge gaps, and field-based mitigation. Biol. Cons. 289, 110376. https://doi.org/10.1016/j.biocon.2023.110376 (2024).
Banda-Thole C, Taylor TK, Larsen T The late president Dr. Kenneth Kaunda’s influence on wildlife conservation and tourism in South Luangwa National Park of Zambia. Zambia Social Science Journal 9: (2024).
Chidakel A, Child B, Muyengwa S Evaluating the economics of park-tourism from the ground-up: Leakage, multiplier effects, and the enabling environment at South Luangwa National Park, Zambia. Ecological Economics. https://doi.org/10.1016/j.ecolecon.2021.106960(2021).
Mcqualter, K. et al. Home ranges, seasonal ranges and daily movements of giraffe (Giraffa camelopardalis giraffa) in northern Botswana. Afr. J. Ecol. https://doi.org/10.1111/aje.12232 (2015).
Watson, F., Becker, M. S., McRobb, R. & Kanyembo, B. Spatial patterns of wire-snare poaching: implications for community conservation in buffer zones around national parks. Biol. Cons. 168, 1–9. https://doi.org/10.1016/j.biocon.2013.09.003 (2013).
Schuette, P., Creel, S. & Christianson, D. Ungulate distributions in a rangeland with competitors, predators and pastoralists. J. Appl. Ecol. 53, 1066–1077. https://doi.org/10.1111/1365-2664.12610 (2016).
Schmidt, G. M. et al. Precision and bias of spatial capture-recapture estimates: a multi-side, multi-year Utah black bear case study. Ecol. Appl. 32, e2618. https://doi.org/10.1002/eap.2618 (2022).
Dupont, G. J. et al. Optimal sampling design for spatial capture-recapture. Ecology 102, e03262. https://doi.org/10.1002/ecy.3262 (2021).
Fennessy, J. Home range and seasonal movements of Giraffa camelopardalis angolensis in the northern Namib Desert. Afr. J. Ecol. 47, 318–327. https://doi.org/10.1111/j.1365-2028.2008.00963.x (2009).
Brown, M. B. & Bolger, D. T. Male-biased partial migration in a giraffe population. Front Ecol Evol https://doi.org/10.3389/fevo.2019.00524 (2020).
Clark, R. K. et al. Seasonal dynamics impact habitat preferences and protected area use of the critically endangered Kordofan Giraffe (Giraffa camelopardalis antiquorum). African J. Wildlife Res. https://doi.org/10.3957/056.053.0119 (2023).
Murphy, S. M. et al. Characterizing recolonization by a reintroduced bear population using genetic spatial capture-recapture. J. Wildl. Manag. 80, 1390–1407 (2016).
Sutherland, C., Fuller, A. K. & Royle, J. A. Modelling non-Euclidean movement and landscape connectivity in highly structured ecological networks. Methods Ecol. Evol. 6, 169–177. https://doi.org/10.1111/2041-210X.12316 (2015).
Vinks, M. A. et al. Response of lion demography and dynamics to the loss of preferred larger prey. Ecol. Appl. 31, e02298. https://doi.org/10.1002/eap.2298 (2021).
Worton, B. J. Kernel methods for estimating the utilization distribution in home-range studies. Ecology 70, 164–168. https://doi.org/10.2307/1938423 (1989).
Goodheart, B. et al. African wild dog movements show contrasting responses to long and short term risk of encountering lions: analysis using dynamic Brownian bridge movement models. Mov. Ecol. 10, 16. https://doi.org/10.1186/s40462-022-00316-7 (2022).
Kranstauber, B. et al. A dynamic Brownian bridge movement model to estimate utilization distributions for heterogeneous animal movement. J. Anim. Ecol. 81, 738–746. https://doi.org/10.1111/j.1365-2656.2012.01955.x (2012).
Young, T. P. & Isbell, L. A. Sex differences in giraffe feeding ecology: energetic and social constraints. Ethology 87, 79–89 (1991).
Hart, E. E., Fennessy, J., Wells, E. & Ciuti, S. Seasonal shifts in sociosexual behaviour and reproductive phenology in giraffe. Behav. Ecol. Sociobiol. 75, 15. https://doi.org/10.1007/s00265-020-02954-6 (2021).
Marealle, W. N. et al. Does illegal hunting skew Serengeti wildlife sex ratios?. Wildl. Biol. 16, 419–429. https://doi.org/10.2981/10-035 (2010).
Strauss, M. K. L. & Packer, C. Using claw marks to study lion predation on giraffes of the Serengeti. J. Zool. 289, 134–142. https://doi.org/10.1111/j.1469-7998.2012.00972.x (2012).
Perez AS, Sazatornil V, Palazon S, et al. Lonely males at the front: A new open SCR model reveals the spatial sex-age structure of an expanding brown bear population. Preprint at Authorea https://doi.org/10.22541/au.171783269.95375238/v1 (2024).
Muller, Z. Population structure of giraffes is affected by management in the Great Rift Valley. Kenya. PLOS ONE 13, e0189678. https://doi.org/10.1371/journal.pone.0189678 (2018).
Rosenblatt, E. et al. Detecting declines of apex carnivores and evaluating their causes: an example with Zambian lions. Biol. Cons. 180, 176–186. https://doi.org/10.1016/j.biocon.2014.10.006 (2014).
Berger, J. Fear, human shields and the redistribution of prey and predators in protected areas. Biol. Let. 3, 620–623. https://doi.org/10.1098/rsbl.2007.0415 (2007).
Deacon, F., Smit, G. N. & Grobbelaar, A. Climatic factors affecting seasonal movements of giraffes (Giraffa camelopardalis) in a semi-arid region of South Africa. Afr. J. Ecol. 62, e13204. https://doi.org/10.1111/aje.13204 (2024).
Chandler, R. & Clark, J. Spatially explicit integrated population models. Methods Ecol. Evol. 5, 1351–1360. https://doi.org/10.1111/2041-210X.12153 (2014).
Rafiq, K. et al. Tourist photographs as a scalable framework for wildlife monitoring in protected areas. Curr. Biol. 29, R681–R682. https://doi.org/10.1016/j.cub.2019.05.056 (2019).
Zipkin, E. F. & Saunders, S. P. Synthesizing multiple data types for biological conservation using integrated population models. Biol. Cons. 217, 240–250. https://doi.org/10.1016/j.biocon.2017.10.017 (2018).
Abadi, F., Gimenez, O., Arlettaz, R. & Schaub, M. An assessment of integrated population models: bias, accuracy, and violation of the assumption of independence. Ecology 91, 7–14. https://doi.org/10.1890/08-2235.1 (2010).
Ahrestani, F. S. et al. An integrated population model for bird monitoring in North America. Ecol Appl 27, 916–924. https://doi.org/10.1002/eap.1493 (2017).
Creel, S. et al. An integrated population model reveals source-sink dynamics for competitively subordinate African wild dogs linked to anthropogenic prey depletion. J. Anim. Ecol. 93, 417–427. https://doi.org/10.1111/1365-2656.14052 (2024).
Weegman, M. D. et al. Integrated population modelling reveals a perceived source to be a cryptic sink. J. Anim. Ecol. 85, 467–475. https://doi.org/10.1111/1365-2656.12481 (2016).
Robinson, O. J. et al. Using citizen science data in integrated population models to inform conservation. Biol. Cons. 227, 361–368. https://doi.org/10.1016/j.biocon.2018.10.002 (2018).
Sun, C. C., Royle, J. A. & Fuller, A. K. Incorporating citizen science data in spatially explicit integrated population models. Ecology 100, e02777. https://doi.org/10.1002/ecy.2777 (2019).
Zipkin, E. F. et al. Integrating count and detection–nondetection data to model population dynamics. Ecology 98, 1640–1650. https://doi.org/10.1002/ecy.1831 (2017).
Mitchell, G. How Giraffes Work (Oxford University Press, 2021).
Lindsey, P. A. et al. Underperformance of African protected area networks and the case for new conservation models: insights from Zambia. PLoS ONE 9, e94109. https://doi.org/10.1371/journal.pone.0094109 (2014).
Simasiku P, Simwanza HI, Tembo G, et al The impact of wildlife management policies on communities and conservation in game management areas in Zambia: message to policy makers. Natural Resources Consultative Forum (2008).
Acknowledgements
This research was supported by the Giraffe Conservation Foundation, Zambian Carnivore Programme, and their donors. We thank the Zambia Department of National Parks and Wildlife for their continuous support and collaboration in helping monitor, manage, and conserve the Luangwa Valley Ecosystem. We appreciate Biocarbon Partners, Nsanga Conservation, and Nyamvu Safaris for survey support, and are grateful for the field assistance, knowledge, and accommodation from Munyamadzi Game Reserve (Thor Kirchner, Nicola Carruthers), Kantanta Hunting Safaris (Valerio Ventriglia), Mopane Safaris (Sarge Abdul Karim), Remote Africa Safaris (Nick Riddin and Jennifer Coppinger-Riddin), Sable Transport (Alister Norton), Shenton safaris (Derek Shenton), Sungani (Paul Davy), and The Bushcamp Company (Andy Hogg). Special thanks to Dan Linden for advice on goodness-of-fit code and Wei Zhang for insights on SCR model fitting.
Author information
Authors and Affiliations
Contributions
S. F., J. F., and M. B. B. contributed to the study conception and design. Data collection was performed by F.O., C.P., D.J., and T.R. Data curation was performed by F.O., C.M., and C.S. Analysis was performed by C.S., and figures were prepared by C.S. and C. M. The first draft of the manuscript was written by C.S., F.O., C.M., M.S.B, and M.B.B. All authors commented on previous versions of the manuscript and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sun, C., Otten, F., Hoffman, R. et al. First rangewide density estimate of the endemic and isolated Luangwa giraffe in Zambia. Sci Rep 15, 16435 (2025). https://doi.org/10.1038/s41598-025-00306-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-00306-w