Abstract
Chronic kidney disease (CKD) is characterized as a progressive dysfunction of the kidney. The estimated glucose disposal rate (eGDR) is widely recognized as a dependable marker of insulin resistance (IR). Nonetheless, the potential link between eGDR and CKD incidence remains insufficiently clarified. This study utilized data from the China Health and Retirement Longitudinal Study (CHARLS). The outcome of this study was CKD events. We performed adjusted Cox proportional hazards regression, restricted cubic splines (RCS), and mediation analyses. Among the 6,737 participants followed for a median of 108 months, 1,356 (20.13%) developed CKD. Relative to the lowest quartile (Q1) of eGDR, the adjusted HR for the highest quartile (Q4) was 0.85 (95% CI: 0.72–0.99). Each standard deviation increase in eGDR was linked to a 7% reduction in CKD risk (HR: 0.93, 95% CI: 0.88–0.99). The RCS curve indicated a linear relationship between eGDR and CKD risk (threshold = 8.21). The cardiovascular disease (CVD) significantly mediated 27.0% of the association between eGDR and CKD risk. This study demonstrates a significant inverse correlation between eGDR levels and CKD risk in middle-aged and elderly individuals within the Chinese population. Moreover, CVD emerges as a key intermediary linking eGDR and the heightened risk of CKD.
Similar content being viewed by others
Introduction
Chronic kidney disease (CKD) is marked by sustained and progressive impairment of renal function, resulting in structural and functional alterations. In advanced stages, patients often progress to end-stage renal disease (ESRD), requiring hemodialysis for survival1,2. In China, the CKD prevalence among adults stands at 10.8%3, while globally, it reaches 14.3%, with a 41.5% rise in CKD-related mortality4. In 2017, an estimated 697.5 million individuals were affected by CKD worldwide, with 1.2 million deaths from the disease5. CKD is projected to become the fifth leading global cause of mortality by 20406. Characterized by its asymptomatic onset, alongside high morbidity and mortality rates, CKD imposes significant health and economic burdens both in China and globally, escalating into a critical public health issue7,8. Early identification of high-risk populations is essential to mitigate the global impact of CKD; however, existing risk factor models are increasingly inadequate in addressing the growing CKD patient population. Consequently, there is an urgent need for the development of simplified and effective markers to improve risk prediction in CKD.
Insulin resistance (IR) refers to the diminished physiological response to insulin, which is strongly linked to the onset and progression of CKD9,10. Although the hyperinsulinemic-euglycemic clamp is considered the gold standard for assessing IR, its high cost limits its use in large-scale clinical settings11. Recently, eGDR has gained traction as a more practical alternative to evaluate IR. It offers lower costs, ease of measurement, and suitability for extensive clinical research, with comparable diagnostic accuracy to the hyperinsulinemic-euglycemic clamp technique12,13. Studies have identified IR as a significant risk factor for CKD and revealed a complex interplay between cardiovascular disease (CVD) and CKD14,15,16,17. Nevertheless, the association between eGDR and CKD remains unclear, particularly across varying glucose regulation states, and the potential mediating role of CVD in the relationship between eGDR and CKD warrants further investigation.
This study explored the association between eGDR and CKD utilizing data from the China Health and Retirement Longitudinal Study (CHARLS), while also evaluating the mediating role of CVD in this relationship. Additionally, it analyzed how glucose regulation status influenced the eGDR-CKD link, addressing the knowledge gap on eGDR and CKD in a nationally representative cohort and reinforcing its potential for real-world clinical application.
Methods
Study population
Data for this cohort study were derived from the CHARLS, an ongoing nationwide longitudinal survey targeting Chinese residents18. CHARLS sampled participants from 450 communities across 150 districts in 28 provinces, ultimately enrolling 17,708 individuals from 10,257 households. The cohort encompassed detailed information on sociodemographic characteristics, lifestyle variables, and health-related metrics specific to this age group. Participants were followed every two to three years after baseline. The study initially enrolled 17,708 individuals from the 2011 (Wave 1) baseline survey, with data gathered through standardized questionnaires during personal interviews. Of these, 10,971 were excluded based on the following criteria: (1) presence of CKD or missing CKD data, (2) incomplete age data, age under 45, or absence of serum creatinine and gender data, (3) missing data on glycated hemoglobin (HbA1c), hypertension, and waist circumference, (4) Estimated glomerular filtration rate (eGFR) below 60 mL/min/1.73 m2. Ultimately, 6,737 participants remained for analysis and were stratified into four subgroups based on the quartiles (Q) of eGDR.
The Biomedical Ethics Review Committee of Peking University, China, granted approval for this study, with all participants providing written informed consent prior to enrollment. The study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines19. We confirm that all methods were performed in accordance with the relevant guidelines. The subject registration process was illustrated in Fig. 1.
Flow chart of the study population. CHARLS China Health and Retirement Longitudinal Study, CKD chronic kidney disease, HbA1c Hemoglobin A1c, eGFR estimated glomerular filtration rate.
Assessment of eGDR, eGFR and CVD
Participants were instructed to fast overnight, with venous blood samples collected by trained personnel the following morning and immediately transported to a local laboratory for storage at 4 °C. After processing, samples were transferred to the Chinese Center for Disease Control and Prevention in Beijing within two weeks for standardized testing. Waist circumference was measured by a qualified healthcare provider. The eGDR was calculated using the following formula20: eGDR (mg/kg/min) = 21.158 − (0.09 ∗ WC) − (3.407 ∗ HT) − (0.551 ∗ HbA1c) [where WC represented waist circumference in cm, HT referred to hypertension status (yes = 1/no = 0), and HbA1c was expressed as a percentage]. The eGFR was derived using the Japanese-coefficient modified MDRD equation as follows21,22: eGFR (mL/min/1.73 m2) = 194 × (serum creatinine) −1.094 × (age)−0.287 (multiplied by 0.739 for females).
CVD encompassed both heart disease and stroke, which were evaluated through two key questions: (1) “Have you ever been diagnosed by a physician with heart attack, coronary artery disease, angina, heart failure, or any other cardiac conditions?“; (2) “Have you ever been diagnosed with stroke by a physician?“.
Assessment of incident CKD
The outcome of this study was the occurrence of CKD events, determined either by self-reported physician diagnosis or personal eGFR measurements from Wave 2 to Wave 5. CKD identification was based on respondents answering the question, “Have you ever been told by a doctor that you have kidney diseases?” A positive response, or an eGFR < 60 mL/min/1.73 m2, classified participants as having CKD. The date of CKD onset was defined as the period between the last interview and the interview in which the CKD event was first reported.
Covariates
Demographic data, health status, and functional capacity were gathered through standardized questionnaires administered by trained interviewers. Variables collected included gender, age, and place of residence (urban or rural), alongside marital status and education level, which was stratified into five categories: no formal education, primary school, middle school, and high school or higher. Marital status was grouped into two categories: married and non-married (never married, separated, divorced, or widowed). Health-related variables encompassed smoking and drinking status, both categorized as never, former, or current. Physician-diagnosed medical conditions such as hypertension, diabetes mellitus (DM), dyslipidemia, heart disease, stroke, and kidney disease were self-reported, along with the use of medications for hypertension, DM, and dyslipidemia. Laboratory assessments included fasting blood glucose (FBG), uric acid (UA), serum creatinine (SCR), blood urea nitrogen (BUN), total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), HbA1c, white blood cell count (WBC), platelet count (PLT), hemoglobin (HB), and C-reactive protein (CRP).
Hypertension was characterized by systolic blood pressure (SBP) ≥ 140 mmHg, diastolic blood pressure (DBP) ≥ 90 mmHg, the use of antihypertensive medication, or a physician-diagnosed history of hypertension23. Dyslipidemia was defined by TG ≥ 150 mg/dL, TC ≥ 240 mg/dL, HDL-C < 40 mg/dL, LDL-C ≥ 160 mg/dL, use of lipid-lowering medication, or a confirmed medical history of dyslipidemia24. Glucose regulation was categorized into normal glucose tolerance (NGT), prediabetes (Pre-DM), and DM. NGT was defined as FBG < 5.6 mmol/L or HbA1c < 5.7%. Pre-DM was classified as FBG 5.6–6.9 mmol/L or HbA1c 5.7–6.4%25.DM was defined by meeting at least one of the following criteria: (1) FPG ≥ 126 mg/dL, or (2) HbA1c ≥ 6.5%, (3) and/or current use of antidiabetic medications, (4) and/or self-reported diabetes diagnosed by a doctor26.
Statistical analysis
A total of 1.84% (124 out of 6737) of data points were missing, and the k-Nearest Neighbor (KNN) interpolation method was applied to address this issue and minimize potential bias. Continuous variables were presented as medians with interquartile ranges or as means ± standard deviations, while categorical variables were reported as frequencies with percentages. Group differences were assessed using one-way ANOVA, chi-square tests, and the Kruskal-Wallis test.
Kaplan–Meier curves were employed to estimate CKD survival probabilities. Collinearity among variables was evaluated using tolerance levels and variance inflation factor (VIF)27. The standardization process of eGDR involves subtracting the mean and dividing by the standard deviation. COX regression models assessed the association between eGDR and CKD risk, with three models constructed based on different covariate adjustments. Model 1 included no covariate adjustments. Model 2 adjusted for gender, age, residence, marital status, and education level. Model 3 further incorporated smoking status, drinking status, WBC, PLT, HB, HDL‑C, BUN, CRP, UA, and body mass index (BMI). After adjusting for covariates (Model 3), the dose-response relationship between eGDR and CKD was analyzed using restricted cubic splines (RCS). Mediation analysis of CVD’s role in the eGDR-CKD association was conducted via the R package “mediation.” The area under curve (AUC) of the receiver operating characteristic (ROC) curve was used to evaluate the incremental effect of incorporating the eGDR and CVD into the traditional risk model on diagnostic prediction. Predictive improvement beyond the base model was assessed using Net Reclassification Improvement (NRI) and Integrated Discrimination Improvement (IDI) indices28,29.The clinical benefits were compared using decision curve analysis (DCA). which plots net benefits against different threshold probabilities. Net benefit associated with any threshold can thus be calculated as follows: Net Benefit = Number of true positives/ Sample size – (Number of false positives/Sample size x Threshold/ 1-Threshold)30.
Additionally, various subgroup and interaction analyses were conducted to assess the consistency of eGDR across demographic variables, including gender, age, residence, smoking and drinking status, hypertension, DM, dyslipidemia, and BMI. The relationship between eGDR and CKD was further explored under different glucose metabolic conditions. When eGDR was analyzed as a continuous variable, the eGDR was standardized. Three sensitivity analyses were performed to evaluate the robustness of the results. First, a COX regression analysis was repeated after removing missing data to verify consistency. Second, Wave 3 data, excluding participants from Wave 1, were used in a separate COX regression to determine alignment with the original findings. Finally, the E-value for eGDR was calculated to estimate the minimum strength of association required between unmeasured confounders and both eGDR and CKD to account for the observed relationships, E-value = RR + sqrt{RR x (RR-1)}31. Statistical analyses were conducted using R software version 4.2.2, with P-values < 0.05 indicating statistical significance.
Results
Population characteristics
Baseline characteristics were compared across quartiles of eGDR: Q1 (≤ 7.37), Q2 (7.37 < eGDR ≤ 10.05), Q3 (10.05 < eGDR ≤ 11.17), and Q4 (> 11.17). This study included 6,737 participants with a mean age of 57.97 ± 8.68 years, of whom 3,258 (48.36%) were male. Participants in the higher eGDR quartiles were more likely to be male, reside in rural areas, and engage in current smoking and drinking. They also had lower prevalence rates of hypertension, DM, heart disease, stroke, and exhibited lower BMI. Detailed baseline characteristics were presented in Table 1.
Associations of eGDR with incident CKD
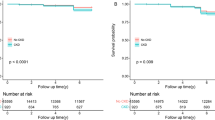
During a median follow-up of 108 months, 1,356 participants (20.13%) developed CKD. Due to VIF values exceeding 5 for TC, TG, and LDL-C, these variables were excluded from the multivariate model (Table S1). Figure S1 illustrated the cumulative CKD incidence across eGDR quartiles (Q1 to Q4), with a statistically significant difference observed (Log rank test, P = 0.006). In Model 3, adjusted for variables including gender, age, residence, marital status, education, smoking, alcohol use, WBC, Plt, HB, HDL-C, BUN, CRP, UA, and BMI, treating eGDR as a categorical variable showed that participants in Q4 had a 15% lower risk of CKD compared to Q1 (adjusted HR: 0.85, 95% CI: 0.72–0.99). As a continuous variable, each (SD) increase in eGDR corresponded to a 7% reduction in CKD risk (HR: 0.93, 95% CI: 0.88–0.99) (Table 2).
The RCS analysis demonstrated a significant linear relationship between eGDR and CKD risk after adjusting for covariates (P = 0.023; P for non-linearity = 0.171). An inflection point was identified at an eGDR of 8.21 (Fig. 2). Below this threshold, an increase in eGDR was associated with a gradual decline in CKD incidence, while above 8.21, the incidence of CKD remained relatively stable despite further increases in eGDR.
The RCS curve was used to assess the dose-response relationship between eGDR and the risk of CKD. Adjusted for gender, age, residence, marital status, educational level, smoking status, drinking status, WBC, PLT, HB, HDL‑C, BUN, CRP, UA, and BMI. The results suggest that maintaining eGDR above 8.21 may reduce CKD risk. eGDR estimated glucose disposal rate, CKD chronic kidney disease, WBC white blood cell, PLT platelet, HB hemoglobin, CRP C-reactive protein, BUN blood urea nitrogen, UA uric acid, HDL-C high-density lipoprotein cholesterol, BMI body mass index, RCS restricted cubic splines.
Mediation analyses
Mediation analysis revealed that CVD significantly mediated the relationship between eGDR and CKD. In Model 1, CVD explained 27.9% of the association between eGDR and CKD incidence. Model 2 showed a mediating proportion of 29.0%, while Model 3 indicated 27.0% mediation by CVD in this association. These results demonstrated that CVD consistently mediated the eGDR-CKD link, with the effect remaining robust even after adjusting for covariates (Table 3). The dose-response relationship between eGDR and CKD among participants with baseline CVD was assessed using RCS curves (Fig S2).
Incremental predictive performance of eGDR and CVD in the incident CKD
The predictive value of incorporating eGDR and CVD into model 3 for CKD was further assessed. The analysis demonstrated an enhancement in the model’s predictive performance for CKD with the inclusion of eGDR and CVD (AUC: 0.588 vs. 0.590 vs. 0.596) (Fig S3). Moreover, the NRI and IDI for CKD were statistically significant (all P < 0.001) (Table 4). Clinical utility was corroborated by DCA (Fig S4).
Subgroup analyses
Subgroup analyses were conducted to assess the consistency of eGDR across varying demographic profiles, along with interaction analyses to explore potential interactive effects across subgroups. Significant associations between eGDR and CKD were identified in participants aged 45–60 years, current smoker, never drinker, dyslipidemia, and BMI ≥ 28 kg/m2. Notably, interactions between eGDR, drinking status, and dyslipidemia were observed (Fig. 3).
Exploratory stratified analysis of the associations between eGDR and the risk of CKD. Adjusted for gender, age, residence, marital status, educational level, smoking status, drinking status, WBC, PLT, HB, HDL‑C, BUN, CRP, UA, and BMI. Significant associations between eGDR and CKD were identified in participants aged 45–60 years, current smoker, never drinker, dyslipidemia, and BMI ≥ 28 kg/m2. Notably, interactions between eGDR, drinking status, and dyslipidemia were observed. eGDR estimated glucose disposal rate, CKD chronic kidney disease, WBC white blood cell, PLT platelet, HB hemoglobin, CRP C-reactive protein, BUN blood urea nitrogen, UA uric acid, HDL-C high-density lipoprotein cholesterol, BMI body mass index.
The relationship between eGDR and CKD in different glucose regulation states
Three models were developed for NGT participants by adjusting for different covariates. In Model 1, compared to Q1 of eGDR, the adjusted HR for Q4 was 0.77 (95% CI: 0.61, 0.97). Each SD increase in eGDR correlated with an 11% reduction in CKD risk (HR: 0.89, 95% CI: 0.83, 0.96). In Model 2, the adjusted HR for Q4 relative to Q1 was 0.81 (95% CI: 0.64, 1.01), with a 9% decrease in CKD risk per SD increase in eGDR (HR: 0.91, 95% CI: 0.84, 0.99). In Model 3, the adjusted HR for Q4 compared to Q1 was 0.82 (95% CI: 0.64, 1.04), though the result was not statistically significant. Nonetheless, each SD increase in eGDR still corresponded to a 9% reduction in CKD risk (HR: 0.91, 95% CI: 0.84, 0.99). When eGDR (standardized) is analyzed as a continuous variable, no significant association was observed between eGDR and CKD risk in individuals with Pre-DM or DM (Table 5).
Sensitivity analysis
The robustness of the results was confirmed through three sensitivity analyses. First, after removing missing values, the COX regression analysis demonstrated consistency with the primary findings (Table S2). Second, an independent analysis using Wave 3 data, excluding participants from Wave 1, yielded results in agreement with the original study (Table S3). Finally, the E-value for the eGDR-CKD association, based on Model 3, was calculated to quantify the minimum strength of association required between unmeasured confounders and CKD to account for the observed relationship. An E-value of 1.36 suggested that a relatively strong unmeasured confounding effect would be necessary to explain the observed risk ratio.
Discussion
This large-scale study is the first to establish a significant link between lower eGDR levels and an increased risk of CKD in a middle-aged and elderly Chinese population, with the association remaining robust after adjusting for covariates. Cox regression analysis demonstrated that each SD increase in eGDR was associated with a 7% reduction in CKD risk. The RCS curve indicated a linear relationship between eGDR and CKD, identifying eGDR < 8.21 as an independent risk factor for CKD. Additionally, CVD mediated the relationship between eGDR and CKD risk, with mediation proportions of 27.9%, 29.0%, and 27.0% across different adjusted models. Incorporating eGDR and CVD into the baseline model significantly enhanced its predictive accuracy for CKD. Additionally, all NRI and IDI values for CKD showed statistical significance. Subgroup analysis revealed strong associations between eGDR and CKD in participants aged 45–60, current smoker, never drinker, those with dyslipidemia, and participants with a BMI ≥ 28 kg/m2. Further examination of the eGDR-CVD relationship across different glucose regulation statuses indicated a significant association between eGDR and CKD in NGT participants, while no significant association was found in Pre-DM or DM individuals. Lastly, three sensitivity analyses confirmed the robustness of the results.
The association between IR and CKD is well-established, yet the link between eGDR and CKD has been scarcely explored. Studies indicate that CKD patients with T1DM exhibit lower eGDR values compared to those without CKD, reflecting higher insulin resistance in this population32. However, the study’s cross-sectional design and the specific characteristics of the cohort limit the ability to assess causality, and the findings may not be generalizable to broader populations. A total of 6,737 participants were enrolled in a nationwide prospective cohort study in China. The analysis revealed a significant association between lower baseline eGDR levels and increased CKD incidence. The RCS curve demonstrated a linear relationship between eGDR and CKD risk. When baseline eGDR was below 8.21, CKD incidence decreased as eGDR increased, while above 8.21, the incidence remained relatively stable. These results suggest that maintaining eGDR above 8.21 may reduce CKD risk, highlighting the need for clinicians to closely monitor individuals with baseline eGDR levels below this threshold. This study is the first to establish the eGDR threshold for identifying high-risk CKD populations, offering a valuable dynamic monitoring marker for clinicians in guiding preventive strategies.
The association between IR and CKD is well-established, and literature has confirmed the predictive value of IR for CVD, with a complex and interrelated connection between CKD and CVD33,34,35 To investigate whether CVD mediates the relationship between IR, as estimated by eGDR, and the risk of incident CKD in a middle-aged and elderly Chinese population, a mediation analysis was conducted. The results revealed that CVD mediated 27.9%, 29.0%, and 27.0% of the association between eGDR and CKD risk across various adjusted models, respectively. In 2023, the American Heart Association (AHA) introduced the concept of Cardiovascular-Kidney-Metabolic (CKM) syndrome. This syndrome is a health condition that arises from the connections between CKD, obesity, diabetes, CVD36.This fully demonstrates the complex relationship between insulin resistance and CKD, CVD. Insulin resistance can directly cause damage to renal function through metabolic effects, endothelial cell dysfunction, inflammation, and oxidative stress37,38,39. Moreover, insulin resistance has been proven to be an independent influencing factor of CVD40.The interaction between CVD and CKD is bidirectional. Insulin resistance not only increases the risk of CVD, but also leads to cardiovascular complications that further damage kidney function, such as left ventricular hypertrophy and heart failure associated with cardiovascular disease, which can cause renal hypoperfusion and worsen kidney function38,41. These may partially explain the mediating role of CVD in the correlation between eGDR and CKD. This result indicates that although our model has weak predictive ability for CKD, considering the complexity of this population and multiple potential confounding factors, AUC values between 0.5 and 0.6 can still be considered to have certain predictive value. We believe that possible reasons include sample heterogeneity, variables that were not fully considered in the model (such as genes, environmental factors, etc.)42,43, and the fact that early warning indicators for CKD may be difficult to accurately predict through a single indicator43,44. One study showed that the AUC value of major adverse cardiovascular events (MACEs) in non-diabetes patients with chronic total inclusion disease predicted by eGDR was 0.68445. Another study showed that the predicted AUC for MACEs in patients with non-ST-segment elevation acute coronary syndrome and non-diabetic patients after percutaneous coronary intervention was 0.69928. The predicted value is also less than 0.7. This indicates the importance of combining eGDR with other clinical indicators to improve prediction accuracy. Significant NRI and IDI values for CKD were observed, with DCA supporting its clinical applicability. The analysis demonstrated a substantial mediating effect of CVD, which persisted even after adjusting for covariates. CVD accounted for 27.0% of the total association between eGDR and CKD among middle-aged and older Chinese adults. Targeting CVD in public health strategies may reduce CKD risk related to IR, alleviating the overall burden of CKD.
Subgroup analyses were conducted to assess the consistency of eGDR across various demographic characteristics. Significant associations between eGDR and CKD were identified in participants aged 45–60 years, current smoker, never drinker, those with dyslipidemia, and individuals with BMI ≥ 28 kg/m2. These data indicate the need for increased clinical attention to patients within these specific groups. Evaluating the impact of eGDR levels on CKD in middle-aged and elderly populations across varying glucose regulation states holds considerable clinical importance. This study aimed to address existing gaps in the field through further investigation. The results indicate a significant inverse association between eGDR and CKD in participants with NGT, with each SD increase in eGDR associated with a 9% reduction in CKD risk. In contrast, no significant relationship was observed between eGDR and CKD in individuals with Pre-DM or DM. These results suggest that early intervention to optimize eGDR levels is essential for minimizing CKD risk, as adjusting eGDR during the Pre-DM or DM stages may have limited impact on CKD development.
The exact mechanism linking eGDR to CKD remains unclear, but it is likely influenced by IR. First, eGDR serves as a reliable marker for IR, which promotes atherosclerosis, potentially resulting in renal artery stenosis or occlusion, ultimately contributing to CKD events46,47,48. Additionally, IR disrupts glucose metabolism, leading to hyperglycemia, which triggers inflammation, oxidative stress, metabolic acidosis, and endothelial damage, all of which contribute to CKD progression49,50,51,52. Moreover, individuals with IR often present with chronic conditions such as hypertension, DM, and obesity—well-established risk factors for CKD53,54,55. Consequently, IR may induce structural and functional renal abnormalities, increasing the likelihood of CKD development.
This study presents several strengths. First, a prospective longitudinal cohort design was employed using the CHALRS database, confirming a linear association between eGDR and CKD in the middle-aged and elderly Chinese population. Moreover, the study identified an inflection point at 8.21, refining the threshold for risk assessment. Additionally, the mediating role of CVD in the eGDR-CKD relationship was clarified, offering important insights into the interplay between eGDR and CKD.
Despite the significant breakthroughs achieved in this study, several limitations remain. First, the study focused solely on the association between baseline eGDR and CKD without investigating the impact of dynamic changes in eGDR on CKD, which warrants exploration in future research. Second, although adjustments were made for numerous covariates, potential bias from unmeasured confounders, such as dietary habits, cannot be entirely ruled out. Nevertheless, the calculated E-value supports the robustness of the results. Lastly, the study population was limited to middle-aged and elderly Chinese individuals, which may restrict the generalizability of the results to younger populations or other nationalities and ethnic groups.
Conclusion
In middle-aged and elderly Chinese populations, reduced eGDR levels are strongly associated with an elevated risk of CKD, particularly in those with normal glucose regulation. CVD serves as a critical mediator in the relationship between eGDR and CKD risk. A linear association has been identified, with a threshold of 8.21 for eGDR. This metric holds potential as a reliable tool for dynamic CKD screening.
Data availability
The data supporting this study are accessible via the CHARLS website (http://charls.pku.edu.cn/).
References
Kalantar-Zadeh, K., Jafar, T. H., Nitsch, D., Neuen, B. L. & Perkovic, V. Chronic kidney disease. Lancet (London England). 398, 786–802. https://doi.org/10.1016/s0140-6736(21)00519-5 (2021).
Levey, A. S. & Defining, A. K. D. The spectrum of AKI, AKD, and CKD. Nephron 146, 302–305. https://doi.org/10.1159/000516647 (2022).
Zhang, L. et al. Prevalence of chronic kidney disease in China: a cross-sectional survey. Lancet (London England). 379, 815–822. https://doi.org/10.1016/s0140-6736(12)60033-6 (2012).
Ene-Iordache, B. et al. Chronic kidney disease and cardiovascular risk in six regions of the world (ISN-KDDC): a cross-sectional study. Lancet Global Health. 4, e307–319. https://doi.org/10.1016/s2214-109x(16)00071-1 (2016).
Global & national burden of chronic kidney disease. 1990–2017: a systematic analysis for the global burden of disease study 2017. Lancet (London England). 395, 709–733. https://doi.org/10.1016/s0140-6736(20)30045-3 (2020).
Kovesdy, C. P. Epidemiology of chronic kidney disease: an update 2022. Kidney Int. Supplements. 12, 7–11. https://doi.org/10.1016/j.kisu.2021.11.003 (2022).
van der Velde, M. et al. Lower estimated glomerular filtration rate and higher albuminuria are associated with all-cause and cardiovascular mortality. A collaborative meta-analysis of high-risk population cohorts. Kidney Int. 79, 1341–1352. https://doi.org/10.1038/ki.2010.536 (2011).
Gansevoort, R. T. et al. Lower estimated GFR and higher albuminuria are associated with adverse kidney outcomes. A collaborative meta-analysis of general and high-risk population cohorts. Kidney Int. 80, 93–104. https://doi.org/10.1038/ki.2010.531 (2011).
de Boer, I. H. & Mehrotra, R. Insulin resistance in chronic kidney disease: a step closer to effective evaluation and treatment. Kidney Int. 86, 243–245. https://doi.org/10.1038/ki.2014.123 (2014).
Lee, S. H., Park, S. Y. & Choi, C. S. Insulin resistance: from mechanisms to therapeutic strategies. Diabetes Metabolism J. 46, 15–37. https://doi.org/10.4093/dmj.2021.0280 (2022).
DeFronzo, R. A., Tobin, J. D. & Andres, R. Glucose clamp technique: a method for quantifying insulin secretion and resistance. Am. J. Physiol. 237, E214–223. https://doi.org/10.1152/ajpendo.1979.237.3.E214 (1979).
Komosinska-Vassev, K., Gala, O., Olczyk, K., Jura-Półtorak, A. & Olczyk, P. The usefulness of diagnostic panels based on Circulating adipocytokines/regulatory peptides, renal function tests, insulin resistance indicators and Lipid-Carbohydrate metabolism parameters in diagnosis and prognosis of type 2 diabetes mellitus with obesity. Biomolecules 10. https://doi.org/10.3390/biom10091304 (2020).
Nyström, T., Holzmann, M. J., Eliasson, B., Svensson, A. M. & Sartipy, U. Estimated glucose disposal rate predicts mortality in adults with type 1 diabetes. Diabetes Obes. Metab. 20, 556–563. https://doi.org/10.1111/dom.13110 (2018).
Matsushita, K. et al. Epidemiology and risk of cardiovascular disease in populations with chronic kidney disease. Nat. Rev. Nephrol. 18, 696–707. https://doi.org/10.1038/s41581-022-00616-6 (2022).
Vallianou, N. G., Mitesh, S., Gkogkou, A. & Geladari, E. Chronic kidney disease and cardiovascular disease: is there any relationship?? Curr. Cardiol. Rev. 15, 55–63. https://doi.org/10.2174/1573403x14666180711124825 (2019).
Liu, M. et al. Cardiovascular disease and its relationship with chronic kidney disease. Eur. Rev. Med. Pharmacol. Sci. 18, 2918–2926 (2014).
Spoto, B., Pisano, A. & Zoccali, C. Insulin resistance in chronic kidney disease: a systematic review. Am. J. Physiol. Renal. Physiol. 311, F1087–f1108. https://doi.org/10.1152/ajprenal.00340.2016 (2016).
Zhao, Y., Hu, Y., Smith, J. P., Strauss, J. & Yang, G. Cohort profile: the China health and retirement longitudinal study (CHARLS). Int. J. Epidemiol. 43, 61–68. https://doi.org/10.1093/ije/dys203 (2014).
von Elm, E. et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet (London England). 370, 1453–1457. https://doi.org/10.1016/s0140-6736(07)61602-x (2007).
Zhang, Z., Zhao, L., Lu, Y., Xiao, Y. & Zhou, X. Insulin resistance assessed by estimated glucose disposal rate and risk of incident cardiovascular diseases among individuals without diabetes: findings from a nationwide, population based, prospective cohort study. Cardiovasc. Diabetol. 23, 194. https://doi.org/10.1186/s12933-024-02256-5 (2024).
Matsuo, S. et al. Revised equations for estimated GFR from serum creatinine in Japan. Am. J. Kidney Diseases: Official J. Natl. Kidney Foundation. 53, 982–992. https://doi.org/10.1053/j.ajkd.2008.12.034 (2009).
Lee, S. et al. The association between kidney function and cognitive decline in community-dwelling, elderly Japanese people. J. Am. Med. Dir. Assoc. 16, 349e341–349e345. https://doi.org/10.1016/j.jamda.2014.12.009 (2015).
Williams, B. et al. 2018 ESC/ESH guidelines for the management of arterial hypertension. Eur. Heart J. 39, 3021–3104. https://doi.org/10.1093/eurheartj/ehy339 (2018).
Zheng, X., Han, L. & Shen, S. Hypertension, remnant cholesterol and cardiovascular disease: evidence from the China health and retirement longitudinal study. J. Hypertens. 40, 2292–2298. https://doi.org/10.1097/hjh.0000000000003259 (2022).
Zhou, Z. et al. Impact of blood lipids on 10-Year cardiovascular risk in individuals without dyslipidemia and with low risk factor burden. Mayo Clin. Proc. 97, 1883–1893. https://doi.org/10.1016/j.mayocp.2022.03.025 (2022).
ElSayed, N. A. et al. 2. Classification and Diagnosis of diabetes: standards of care in diabetes-2023. Diabetes care 46, 19–40 (2023). https://doi.org/10.2337/dc23-S002
Wax, Y. Collinearity diagnosis for a relative risk regression analysis: an application to assessment of diet-cancer relationship in epidemiological studies. Stat. Med. 11, 1273–1287. https://doi.org/10.1002/sim.4780111003 (1992).
Liu, C. et al. Predictive worth of estimated glucose disposal rate: evaluation in patients with non-ST-segment elevation acute coronary syndrome and non-diabetic patients after percutaneous coronary intervention. Diabetol. Metab. Syndr. 14, 145. https://doi.org/10.1186/s13098-022-00915-9 (2022).
Pencina, M. J., D’Agostino, R. B., Demler, O. V. & Sr. & Novel metrics for evaluating improvement in discrimination: net reclassification and integrated discrimination improvement for normal variables and nested models. Stat. Med. 31, 101–113. https://doi.org/10.1002/sim.4348 (2012).
Sadatsafavi, M. et al. Moving beyond AUC: decision curve analysis for quantifying net benefit of risk prediction models. Eur. Respir. J. 58. https://doi.org/10.1183/13993003.01186-2021 (2021).
VanderWeele, T. J. & Ding, P. Sensitivity analysis in observational research: introducing the E-Value. Ann. Intern. Med. 167, 268–274. https://doi.org/10.7326/m16-2607 (2017).
Vladu, M. et al. Insulin resistance and chronic kidney disease in patients with type 1 diabetes mellitus. J. Nutr. Metab.. 2017 https://doi.org/10.1155/2017/6425359 (2017).
Hill, M. A. et al. Insulin resistance, cardiovascular stiffening and cardiovascular disease. Metab. Clin. Exp. 119. https://doi.org/10.1016/j.metabol.2021.154766 (2021).
Ormazabal, V. et al. Association between insulin resistance and the development of cardiovascular disease. Cardiovasc. Diabetol. 17. https://doi.org/10.1186/s12933-018-0762-4 (2018).
Reiss, A. B. et al. CKD, arterial calcification, atherosclerosis and bone health: Inter-relationships and controversies. Atherosclerosis 278, 49–59. https://doi.org/10.1016/j.atherosclerosis.2018.08.046 (2018).
Ndumele, C. E. et al. Cardiovascular-Kidney-Metabolic health: A presidential advisory from the American heart association. Circulation 148, 1606–1635. https://doi.org/10.1161/cir.0000000000001184 (2023).
De Cosmo, S., Menzaghi, C., Prudente, S. & Trischitta, V. Role of insulin resistance in kidney dysfunction: insights into the mechanism and epidemiological evidence. Nephrol. dialysis Transplantation: Official Publication Eur. Dialysis Transpl. Association - Eur. Ren. Association. 28, 29–36. https://doi.org/10.1093/ndt/gfs290 (2013).
Zhang, Y. et al. Insulin resistance, incident cardiovascular diseases, and decreased kidney function among nondiabetic American Indians: the strong heart study. Diabetes Care. 36, 3195–3200. https://doi.org/10.2337/dc12-2368 (2013).
Nakashima, A., Kato, K., Ohkido, I. & Yokoo, T. Role and treatment of insulin resistance in patients with chronic kidney disease: A review. Nutrients 13. https://doi.org/10.3390/nu13124349 (2021).
Kosmas, C. E. et al. Insulin resistance and cardiovascular disease. J. Int. Med. Res. 51. https://doi.org/10.1177/03000605231164548 (2023).
Chan, D. T., Watts, G. F., Irish, A. B. & Dogra, G. K. Insulin resistance and vascular dysfunction in chronic kidney disease: mechanisms and therapeutic interventions. Nephrol. dialysis Transplantation: Official Publication Eur. Dialysis Transpl. Association - Eur. Ren. Association. 32, 1274–1281. https://doi.org/10.1093/ndt/gfv326 (2017).
Knoers, N. et al. Genetic testing in the diagnosis of chronic kidney disease: recommendations for clinical practice. Nephrol. dialysis Transplantation: Official Publication Eur. Dialysis Transpl. Association - Eur. Ren. Association. 37, 239–254. https://doi.org/10.1093/ndt/gfab218 (2022).
Echouffo-Tcheugui, J. B. & Kengne, A. P. Risk models to predict chronic kidney disease and its progression: a systematic review. PLoS Med. 9, e1001344. https://doi.org/10.1371/journal.pmed.1001344 (2012).
Altilio, D., Tierney, A. R. & Kotler, D. P. Application of home parenteral therapies to the treatment of patients with AIDS. Nutr. Clin. Pract. 3, 171–172 (1988).
Chen, W., Liu, Y., Shi, Y. & Liu, J. Prognostic value of estimated glucose disposal rate and systemic Immune-Inflammation index in Non-Diabetic patients undergoing PCI for chronic total occlusion. J. Cardiovasc. Dev. Dis. 11. https://doi.org/10.3390/jcdd11090261 (2024).
Wu, D. et al. Characteristics and predictors of low-grade renal artery stenosis in female patients with CKD. Clin. Exp. Hypertens.. 45, 2175849. https://doi.org/10.1080/10641963.2023.2175849 (2023).
Mohan, I. V. & Bourke, V. The management of renal artery stenosis: an alternative interpretation of ASTRAL and CORAL. Eur. J. Vascular Endovascular Surgery: Official J. Eur. Soc. Vascular Surg. 49, 465–473. https://doi.org/10.1016/j.ejvs.2014.12.026 (2015).
Di Pino, A. & DeFronzo, R. A. Insulin resistance and atherosclerosis: implications for Insulin-Sensitizing agents. Endocr. Rev. 40, 1447–1467. https://doi.org/10.1210/er.2018-00141 (2019).
Tucker, B. J., Anderson, C. M., Thies, R. S., Collins, R. C. & Blantz, R. C. Glomerular hemodynamic alterations during acute hyperinsulinemia in normal and diabetic rats. Kidney Int. 42, 1160–1168. https://doi.org/10.1038/ki.1992.400 (1992).
Esteghamati, A. et al. Insulin resistance is an independent correlate of increased urine albumin excretion: a cross-sectional study in Iranian type 2 diabetic patients. Diabet. Medicine: J. Br. Diabet. Association. 26, 177–181. https://doi.org/10.1111/j.1464-5491.2008.02653.x (2009).
Kato, Y. et al. Mild renal dysfunction is associated with insulin resistance in chronic glomerulonephritis. Clin. Nephrol. 54, 366–373 (2000).
Chen, J. et al. Insulin resistance and risk of chronic kidney disease in nondiabetic US adults. J. Am. Soc. Nephrology: JASN. 14, 469–477. https://doi.org/10.1097/01.asn.0000046029.53933.09 (2003).
Barazzoni, R., Gortan Cappellari, G., Ragni, M. & Nisoli, E. Insulin resistance in obesity: an overview of fundamental alterations. Eat. Weight Disorders: EWD. 23, 149–157. https://doi.org/10.1007/s40519-018-0481-6 (2018).
da Silva, A. A. et al. Role of hyperinsulinemia and insulin resistance in hypertension: metabolic syndrome revisited. Can. J. Cardiol. 36, 671–682. https://doi.org/10.1016/j.cjca.2020.02.066 (2020).
Vestergaard, A. H. S. et al. Risk factor analysis for a rapid progression of chronic kidney disease. Nephrol. dialysis Transplantation: Official Publication Eur. Dialysis Transpl. Association - Eur. Ren. Association. 39, 1150–1158. https://doi.org/10.1093/ndt/gfad271 (2024).
Acknowledgements
This study utilized data from CHARLS. The authors extend their gratitude to the CHARLS research team and all participants involved in the study.
Funding
This work was supported by the Boxi Youth Natural Science Foundation (BXQN2023016), Scientific Research Project of Jiangsu Commission of Health (M2022010), Medical Innovation Application Research Project of Suzhou Science and Technology Bureau (SKY2022131).
Author information
Authors and Affiliations
Contributions
HS conceptualized and designed the study, and drafted the manuscript. Data analysis was conducted by HS and LZ. YZ, YH and YZ contributed to the literature search. Manuscript review was performed by HS and YC. All authors participated in the review of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The CHARLS study received approval from the Institutional Review Board of Peking University (approval numbers: IRB00001052-11015 for household surveys and IRB00001052-11014 for blood samples), with written informed consent obtained from all participants.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, Y., Zheng, L., Zhou, Y. et al. The role of cardiovascular disease in the association between estimated glucose disposal rate and chronic kidney disease. Sci Rep 15, 16034 (2025). https://doi.org/10.1038/s41598-025-00359-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-00359-x
Keywords
This article is cited by
-
Association between estimated glucose disposal rate and diabetes mellitus incidence in middle-aged and elderly adults and development of predictive model: evidence from two prospective longitudinal studies
BMC Endocrine Disorders (2025)
-
Association of estimated glucose disposition rate with aging acceleration and mortality risk in individuals with cardiovascular-kidney-metabolic syndrome: evidence from two large national population-based studies
Cardiovascular Diabetology (2025)