Abstract
Despite recent accumulation on cardiovascular (CV) safety data on xanthine oxidase inhibitors (XOIs) among gout patients, assessment has been largely limited to macro- than micro-vascular disorders, prompting the investigation on the latter outcome. Therefore, we aimed to compare the risk of retinal microvascular disorders between allopurinol and febuxostat initiators among gout patients. Using the 2011–2019 Korean National Health Insurance service database, we conducted a cohort study on gout patients initiating allopurinol or febuxostat. The primary outcome was a composite of retinal microvascular disorders. After 1:1 propensity-score (PS) matching in non-diabetic (non-DM) and diabetic (DM) subgroups, pooled and subgroup-specific hazard ratios (HRs) and 95% confidence intervals (CIs) were reported, comparing allopurinol and febuxostat. We included 118,376 PS-matched pairs of febuxostat and allopurinol initiators (mean 57.3 years, 83.7% male; 89,642 pairs in non-DM and 28,734 pairs in DM subgroup). During a mean follow-up of 223 days, the incidence rate per 100 person-years of the primary outcomes was 0.88 among allopurinol users and 0.93 among febuxostat users. The corresponding HR (95% CI) was 0.98 (0.83–1.15). The HR (95% CI) was 0.94 (0.76–1.15) in non-DM subgroup and 1.05 (0.80–1.39) in DM subgroup. The result for DM retinopathy also showed a similar risk (HR 0.86, 95% CI 0.67–1.11). In this large population-based cohort study on patients with gout, we did not find any difference in the risk of retinal microvascular disorders between allopurinol and febuxostat initiators.
Similar content being viewed by others
Introduction
Gout is the most common inflammatory arthritis resulting from hyperuricemia1. Notably, patients with gout have a significantly higher risk of cardiovascular (CV) events compared to those without2. Although the mechanisms linking gout and the increased CV risk are not fully understood, the role of urate lowering therapy (ULT) in modifying the CV risk has been an issue of interest2.
The most commonly used ULTs are the xanthine oxidase inhibitors (XOIs) including allopurinol and febuxostat3,4. The CARES trial was the first randomized controlled trial (RCT) that compared the CV safety between these two XOIs with different urate lowering efficacy in high CV risk population5. Unexpectedly, the trial showed a higher risk of all-cause and CV mortality in the febuxostat than allopurinol group. However, subsequent FAST and ALL-HEART trials have shown neutral CV results between these two treatments or between those receiving versus not receiving allopurinol6,7. Given these contradictory findings, this issue remains a topic of ongoing debate. Despite the high volume of interests on this issue, the CV endpoints of the contemporary studies, however, have mainly focused on macrovascular events such as myocardial infarction, stroke, and CV deaths, while little data are available for the microvascular events3,4,5,6,7.
Although macro- and micro-vascular disorders are interconnected showing a high concordance rate, the two CV diseases are distinctive entities in terms of the anatomical and structural damages and functional consequences8,9: they harbor different positions in the continuum of vascular damage. Recently, studies have reported an association between retinal microvascular disorders and gout/hyperuricemia. For example, patients with gout or hyperuricemia exhibit choriocapillaris flow deficit or lower retinal vessel density compared with those with normal serum uric acid levels when examined with optical coherence tomography angiography10,11. Also, epidemiologic studies have shown an association between gout and age-related macular degeneration12,13,14. Therefore, it would be necessary to generate more thorough information on the comparative CV effects of different XOIs by investigating microvascular end-points. To this end, we aim to compare the risk of retinal microvascular disorders between allopurinol initiators versus febuxostat in patients with gout.
Methods
Data source
We conducted a cohort study in patients with gout using the 2011–2019 Korean National Health Insurance service (KNHIS) database. The KNHIS is mandatory for all citizens in Korea, and the database contains longitudinal patient data from birth to death, including demographics, International Classification of Diseases, 10th Revision (ICD10) diagnosis codes, procedures, pharmacy dispensing records, type of medical utilization (outpatient, inpatient, or emergency department)15. The Institutional Review Board of the Seoul National University Bundang Hospital approved this study and waived the need to obtain informed consent (No. X 2008-628-902), based on the fully deidentified database. This study was conducted according to the principles set in the Declaration of Helsinki.
Study population
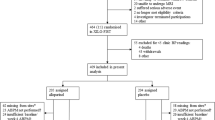
Figure 1 shows the study cohort selection process. Patients aged ≥ 40 years with ICD10 diagnosis codes of gout who initiated febuxostat or allopurinol were eligible (see Supplemental Table S1 for the ICD10 codes to define inclusion and exclusion criteria for study population). They were required free of any ULT use for at least 12 months prior to the first dispensing date of either allopurinol or febuxostat (= index date). We excluded patients who had diagnoses of any choroid and retinal disorders including diabetic retinopathy, type 1 diabetes (T1DM), cancer, or chronic kidney disease during the 12-month pre-index period (= baseline period) (Supplemental Table S1). The patients were then, stratified into non-DM and DM subgroups according to the baseline T2DM status defined based on the presence and absence of ICD10 codes for T2DM. Those without ICD10 codes for T2DM but who used anti-diabetic drugs were excluded from the analysis. The pooled cohort was then constructed by combining DM and non-DM subgroups.
Study cohort selection process. Patients aged ≥ 40 years with ICD10 diagnosis codes for gout who initiated febuxostat or allopurinol were eligible for inclusion. Patients were required to be free of any urate-lowering therapy for at least 12 months prior to the first dispensing date. Those with a diagnosis of any retinal disorders, type 1 diabetes (T1DM), cancer, or chronic kidney disease during the 12-month pre-index period were excluded.
Outcomes
Due to the unique retinal vascular structure formed by arborizing microvasculature of capillary plexus, neo-angiogenesis from pre-existing capillaries is a common complication of retinal disorders including diabetic retinopathy, retinal vascular occlusion, and age-related macular degeneration16,17,18. Thus, we defined the primary outcome as a composite of the retinal disorders that represent microvascular complications, which are defined as at least two occasions with the following ICD10 diagnosis codes: retinal vascular occlusions (H34.x), other retinal disorders encompassing retinal degeneration, hemorrhage, and other changes in retinal vascular appearances (H35.x), and other retinal disorders in diseases classified elsewhere (H36.8) including atherosclerotic retinopathy (see Supplemental Table S1 for the ICD10 codes used to define outcomes). The secondary outcomes were individual components of the primary outcome. We note that DM is more prevalent in gout patients than in the general population19, and that DM retinopathy might progress more in the presence of gout based on the common pathogenic mechanisms of oxidative stress from both conditions2,20,21. Therefore, DM retinopathy was investigated as a secondary outcome only in the DM subgroup (Supplemental Table S1).
Covariates
During the 365-day pre-index baseline period, we measured > 60 variables using the ICD10 codes, drug prescriptions, and healthcare use claims: patients’ demographics, index calendar year, common ophthalmic conditions (glaucoma, cataract), traditional CV risk factors (hypertension, diabetes, dyslipidemia) and CV diseases (angina, myocardial infarction, stroke, peripheral vascular disease), non-CV comorbidities (chronic obstructive pulmonary disease, asthma, liver disease, chronic kidney disease, osteoporosis), DM status and complications (retinopathy, neuropathy, nephropathy), medications for gout, DM, and other medical conditions, and healthcare service utilizations (Table 1). We also estimated a Charlson–Deyo score for multi-morbidities22. These covariates represent potential confounding factors that can affect retinal microvascular structure and be disproportionately distributed between the two treatment groups, thus were adjusted for by propensity-score (PS) matching.
Statistical analysis
To adjust for confounding, we used a 1:1 PS matching on > 60 covariates in each of non-DM and DM subgroups to generate a pooled main cohort. To estimate a PS, we constructed a multivariable logistic regression model within individual subgroups that included all of the covariates listed in Table 1 plus the index year. The covariate balance between the two groups was evaluated by standardized mean differences: a balanced covariate distribution was considered achieved with a standardized mean difference of < 0.123.
In our primary as-treated analysis, patients were followed from the day after the index date to the first occurrence of the following events: outcome occurrence, death, administrative end of the database (31st December 2019), discontinuation of the index treatment, adding any other ULT agent over the index treatment. Drug discontinuation was defined as no dispensing within 90 days from the expected refill date. Patients who discontinued the study medication were followed up until the last expected refill date plus 30-day grace period. The expected refill date was calculated by adding days’ supply to the last dispensing date of the study medication. For the secondary intention-to-treat (ITT) analysis, we followed patients up to 365 days after the index date without censoring on drug switching, adding, or discontinuation.
Incidence rates (IRs) of primary and secondary outcomes were calculated per 100-person-years in the PS-matched study populations. We used a Cox proportional hazard model estimating the pooled and subgroup-specific hazard ratios (HRs) and 95% confidence intervals (CIs) comparing allopurinol and febuxostat24. We tested the proportional hazards assumption using the interaction term between treatment and follow-up time and the assumptions were not violated in any of the models. All analyses were performed using SAS 9.4 (SAS Institute) software.
Subgroup analysis
A subgroup of patients with a high CV risk at baseline was examined to compare allopurinol and febuxostat. The high CV risk subgroup was defined in accordance to the inclusion criteria of the representative CARES study that investigated the CV risk between allopurinol and febuxostat among gout patients at high CV risk: either men aged ≥ 50 years or women aged ≥ 55 years, who had at least one diagnosis of diabetes, angina, myocardial infarction, stroke, or peripheral vascular disease during the 1-year pre-index period5. The high CV risk subgroup was not stratified by DM status to avoid power attrition.
Results
Baseline characteristics
Figure 1 shows our study cohort selection process. A total of 405,445 new users (n = 321,365 without DM, n = 84,080 with DM) of allopurinol and 131,783 new users (n = 97,917 without DM, n = 33,866 with DM) of febuxostat were identified, who had no preexisting retinal diseases and were free of any ULT treatment at baseline. After PS-matching, 118,376 pairs of allopurinol and febuxostat initiators were generated (89,642 pairs from non-DM subgroup, 27,834 pairs from DM subgroup) with mean age 57.3 years and 83.7% male. The baseline covariates were well-balanced, showing standardized differences of < 0.1 for all covariates (Table 1). The mean age was 55.5 and 62.9 years in the non-DM and DM subgroup, respectively, with the proportion of males 86.4% and 75.2%, respectively. In the DM than non-DM subgroup, a higher prevalence of CV risk factors and CV diseases was seen. The mean comorbidity index ± standard deviation (SD) was 0.8 ± 1.0 and 2.6 ± 1.6 in non-DM and DM subgroup, respectively.
Risk of retinal microvascular disorders associated with allopurinol versus febuxostat
In pooled analyses, the IR per 100 person-years of the composite outcomes was 0.88 among allopurinol users and 0.93 among febuxostat users over 223 days of mean follow-up. The corresponding PS-matched HR (95% CI) was 0.98 (0.83–1.15) comparing allopurinol to febuxostat initiators (Table 2). In the non-DM subgroup, the IR per 100 person-years of the composite outcome was 0.77 among allopurinol users and 0.81 among febuxostat users, with a PS-matched HR of 0.94 (95% CI 0.76–1.15). In the DM subgroup, the IR of the composite outcome was 1.15 among allopurinol users and 1.26 among febuxostat users, which corresponded to a PS-matched HR of 1.05 (95% CI 0.80–1.39). The 1-year ITT results were similar showing a PS-matched HR of 0.93 (95% CI 0.83–1.03) (Supplemental Table S2).
Risk of secondary outcomes associated with allopurinol versus febuxostat
Among the individual components of the primary outcome, other retinal disorders (ICD10 H35.x, including retinal degeneration, hemorrhage, and other changes in retinal vascular appearances) were most common (Table 2). In the non-DM subgroup, its IR was 0.69 for allopurinol users and 0.72 for febuxostat users, respectively (HR 0.98, 95% CI 0.78–1.22). Among the DM subgroup, its IR was 1.04 for allopurinol users and 1.10 for febuxostat users, respectively (HR 1.05, 95% CI 0.78–1.41). Similarly, there was no significance in the risk of other secondary outcomes between the two treatment groups both in non-DM and DM subgroups. In the DM subgroup, the risk of new onset diabetic retinopathy was comparable between allopurinol and febuxostat users (HR 0.86, 95% CI 0.67–1.11).
Subgroup analysis
In the subgroup at high CV risk (see Supplemental Table S3 for their PS-matched covariate distribution), the IR of the composite outcome was 1.32 in allopurinol initiators and 1.33 in febuxostat initiators, with a HR (95% CI) of 1.08 (0.85–1.37) (Table 3). Consistent with the result from the main cohort, we also did not find any difference in the risk of secondary outcomes among the high CV risk subgroup.
Discussion
To our best knowledge, this study is the first large population-based cohort study to compare the risk of retinal microvascular disorders associated with allopurinol versus febuxostat treatment in patients with gout. Febuxostat treatment showed a similar IR of retinal microvascular disorders compared to allopurinol in patients with gout regardless of DM status. The results were consistent for individual secondary outcomes and among those at high CV risk.
The 2020 gout guideline from the American College of Rheumatology strongly recommends allopurinol as the first-line pharmacologic agent for ULT for all patient groups, including those with chronic kidney disease25. However, febuxostat had been a preferred treatment option based on the higher potency particularly in recalcitrant gout including those complicated by chronic kidney disease26,27, and also on a better safety against allopurinol hypersensitivity syndrome28,29. The prevalence of HLA-B*5801 positivity, a strong genetic risk factor for allopurinol hypersensitivity syndrome30, has been reported to be as high as 12.2% in Korea31, adding more preference to febuxostat. However, utilization of febuxostat tended to decline following the CARES trial32. Currently, its use recovered to constitute approximately half of the new ULT prescriptions in Korea after subsequent RCTs releasing null findings for CV risk of febuxostat33. In this context, more comprehensive CV safety data of the drug are warranted.
Although microvascular and macrovascular diseases share risk factors and often concur, differences exist8,9. For example, some T2DM patients develop macrovascular diseases alone, while others develop microvascular diseases34. Moreover, the UK Prospective Diabetes Study showed that the intensive glycemic control in T2DM exerted beneficial effects only for microvascular than macrovascular diseases35. Similarly, only microvascular than macrovascular diseases at baseline predict future major peripheral vascular diseases in T2DM36, and the burden of microvascular disease increases the risk of CV death independent of the traditional risk factors for macrovascular diseases in T2DM patients37. Based on these findings, micro- and macro-vascular disease should be independently assessed despite their similarities.
Our findings of a similar risk of retinal microvascular disorders between allopurinol and febuxostat groups are consistent with the aforementioned RCTs and recent well-designed observational studies reporting a null finding for macrovascular CV diseases regardless of different ULT3,4,6,7. Due to the concordance and similarities in risk factors of microvascular and macrovascular disorders8,9, it is not surprising that the results are consistent between them. However, our findings provide more assuring CV safety data for different ULTs among gout patients. In particular, this information could be useful for the physicians’ decision making for gout patients whose ethnic backgrounds indicate a relatively higher risk for allopurinol hypersensitivity syndrome.
The strengths of this study are as follows. First, to the best of our knowledge, it is the first study to compare the risk of retinal microvascular safety of allopurinol and febuxostat among patients with gout. Second, we examined comprehensive analyses on individual components of the primary outcomes and on subgroups stratified by the presence of T2DM or underlying CV risk status, only to observe consistent results. Third, we used a nation-wide database, ensuring high generalizability of the study findings.
There are also limitations. First, retinal microvascular outcomes were identified by ICD10 diagnostic codes, raising concerns for misclassification bias. However, the accuracy of ICD10 diagnosis codes for ophthalmic diseases including DM retinopathy and macular edema was found to be high with a positive predictive value of > 90%38,39,40. Second, as an observational study that used health insurance database, direct information on disease severity (e.g., uric acid or HbA1c levels) could not be ascertained, leaving concerns for residual and unmeasured confounding. However, to avoid this, we implemented an active comparator new user design, which is known for its advantage to cope with both measured and unmeasured confounding41. Also, we used extensive PS-matching that balanced many proxy variables for gout and DM severity (use of ULTs, NSAID, and steroid, cumulative steroid dose, use of DM medications, the presence of DM complications, and the prevalence of established CV diseases and CV risk factors).
In conclusion, we did not find clinically meaningful difference in the risk of retinal microvascular disorders between allopurinol and febuxostat. We believe this study offers valuable clinical insights on drug safety when selecting ULT agents for patients with gout. Further investigation with additional endpoints may be needed to fully understand the long-term safety of both treatments.
Data availability
Patient-level data are not publicly allowed according to Data Use Agreement. Aggregate-level data can be requested to the corresponding author.
References
Dehlin, M., Jacobsson, L. & Roddy, E. Global epidemiology of gout: Prevalence, incidence, treatment patterns and risk factors. Nat. Rev. Rheumatol. 16, 380–390 (2020).
Kang, E. H. & Kim, S. C. Cardiovascular safety of urate Lowering therapies. Curr. Rheumatol. Rep. 21, 48 (2019).
Zhang, M. et al. Assessment of cardiovascular risk in older patients with gout initiating febuxostat versus allopurinol: Population-based cohort study. Circulation 138, 1116–1126 (2018).
Kang, E. H. et al. Comparative cardiovascular risk of allopurinol versus febuxostat in patients with gout: A nation-wide cohort study. Rheumatol. (Oxford). 58, 2122–2129 (2019).
White, W. B. et al. Cardiovascular safety of febuxostat or allopurinol in patients with gout. N. Engl. J. Med. 378, 1200–1210 (2018).
Mackenzie, I. S. et al. Long-term cardiovascular safety of febuxostat compared with allopurinol in patients with gout (FAST): A multicentre, prospective, randomised, open-label, non-inferiority trial. Lancet 396, 1745–1757 (2020).
Mackenzie, I. S. et al. Allopurinol versus usual care in UK patients with ischaemic heart disease (ALL-HEART): A multicentre, prospective, randomised, open-label, blinded-endpoint trial. Lancet 400, 1195–1205 (2022).
Orasanu, G. & Plutzky, J. The pathologic continuum of diabetic vascular disease. J. Am. Coll. Cardiol. 53, S35–42 (2009).
Climie, R. E. et al. Macrovasculature and microvasculature at the crossroads between type 2 diabetes mellitus and hypertension. Hypertension 73, 1138–1149 (2019).
Lu, Y. et al. Retinal microvasculature and choriocapillaris flow deficit in relation to serum uric acid using swept-source optical coherence tomography angiography. Transl Vis. Sci. Technol. 11, 9 (2022).
Yang, K. et al. Association of serum uric acid with retinal capillary plexus. Front. Endocrinol. (Lausanne). 13, 855430 (2022).
Hsu, M. H., Hsu, C. A., Lai, S. C. & Yen, J. C. Gout as a risk factor for age-related macular degeneration in Taiwanese adults-a population-based study in Taiwan. Int. J. Environ. Res. Public Health. 19, 10142 (2022).
Singh, J. A. & Cleveland, J. D. Gout and the risk of age-related macular degeneration in the elderly. PLoS One 13, e0199562 (2018).
Pai, H. L., Chang, H. H. & Lin, D. P. The need to investigate hyperuricemia as a factor in the onset of age-related macular degeneration. Eye (Lond). 35, 1804–1807 (2021).
Cheol Seong, S. et al. Data resource profile: The National health information database of the National health insurance service in South Korea. Int. J. Epidemiol. 46, 799–800 (2017).
Sun, Y. & Smith, L. E. H. Retinal vasculature in development and diseases. Annu. Rev. Vis. Sci. 4, 101–122 (2018).
Bharadwaj, A. S. et al. Role of the retinal vascular endothelial cell in ocular disease. Prog. Retin. Eye Res. 32, 102–180 (2013).
Brylla, E. et al. Differences between retinal and choroidal microvascular endothelial cells (MVECs) under normal and hypoxic conditions. Exp. Eye Res. 77, 527–535 (2003).
Kim, S. C., Liu, J. & Solomon, D. H. Risk of incident diabetes in patients with gout: A cohort study. Arthritis Rheumatol. 67, 273–280 (2015).
Tang, J. & Kern, T. S. Inflammation in diabetic retinopathy. Prog. Retin. Eye Res. 30, 343–358 (2011).
Rodríguez, M. L., Pérez, S., Mena-Mollá, S., Desco, M. C. & Ortega, Á. L. Oxidative stress and microvascular alterations in diabetic retinopathy: Future therapies. Oxid. Med. Cell Longev. 2019, 4940825 (2019).
Sundararajan, V. et al. New ICD-10 version of the Charlson comorbidity index predicted in-hospital mortality. J. Clin. Epidemiol. 57, 1288–1294 (2004).
Austin, P. C. Using the standardized difference to compare the prevalence of a binary variable between two groups in observational research. Commun. Stat. Simul. Comput. 38, 1228–1234 (2009).
Austin, P. C., Lee, D. S. & Fine, J. P. Introduction to the analysis of survival data in the presence of competing risks. Circulation 133, 601–609 (2016).
FitzGerald, J. D. et al. 2020 American college of rheumatology guideline for the management of gout. Arthritis Care Res. (Hoboken). 72, 744–760 (2020).
Tanaka, K. et al. Renoprotective effects of febuxostat in hyperuricemic patients with chronic kidney disease: A parallel-group, randomized, controlled trial. Clin. Exp. Nephrol. 19, 1044–1053 (2015).
Kim, A. et al. Comparison of persistence rates between allopurinol and febuxostat as first-line urate-lowering therapy in patients with gout: An 8-year retrospective cohort study. Clin. Rheumatol. 39, 3769–3776 (2020).
Chohan, S. Safety and efficacy of febuxostat treatment in subjects with gout and severe allopurinol adverse reactions. J. Rheumatol. 38, 1957–1959 (2011).
Chen, C. H. et al. Hypersensitivity and cardiovascular risks related to allopurinol and febuxostat therapy in Asians: A population-based cohort study and meta-analysis. Clin. Pharmacol. Ther. 106, 391–401 (2019).
Hung, S. I. et al. HLA-B*5801 allele as a genetic marker for severe cutaneous adverse reactions caused by allopurinol. Proc. Natl. Acad. Sci. USA 102, 4134–4139 (2005).
Lee, K. W., Oh, D. H., Lee, C. & Yang, S. Y. Allelic and haplotypic diversity of HLA-A, -B, -C, -DRB1, and -DQB1 genes in the Korean population. Tissue Antigens. 65, 437–447 (2005).
Kim, S. C., Neogi, T., Kim, E., Lii, J. & Desai, R. J. Trends in utilization of urate-lowering therapies following the US food and drug administration’s boxed warning on febuxostat. Arthritis Rheumatol. 73, 542–543 (2021).
Shin, A. et al. Cardiovascular safety associated with febuxostat versus allopurinol among patients with gout: Update with accumulated use of febuxostat. Semin Arthritis Rheum. 56, 152080 (2022).
Mohammedi, K. et al. Comparative effects of microvascular and macrovascular disease on the risk of major outcomes in patients with type 2 diabetes. Cardiovasc. Diabetol. 16, 95 (2017).
Intensive blood-glucose. Control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK prospective diabetes study (UKPDS) group. Lancet 352, 837–853 (1998).
Mohammedi, K. et al. Microvascular and macrovascular disease and risk for major peripheral arterial disease in patients with type 2 diabetes. Diabetes Care. 39, 1796–1803 (2016).
Brownrigg, J. R. et al. Microvascular disease and risk of cardiovascular events among individuals with type 2 diabetes: A population-level cohort study. Lancet Diabetes Endocrinol. 4, 588–597 (2016).
Muir, K. W., Gupta, C., Gill, P. & Stein, J. D. Accuracy of international classification of diseases, ninth revision, clinical modification billing codes for common ophthalmic conditions. JAMA Ophthalmol. 131, 119–120 (2013).
Cai, C. X. et al. Effect of ICD-9 to ICD-10 transition on accuracy of codes for stage of diabetic retinopathy and related complications: Results from the CODER study. Ophthalmol. Retina. 5, 374–380 (2021).
Bearelly, S. et al. Identification of patients with diabetic macular edema from claims data: A validation study. Arch. Ophthalmol. 126, 986–989 (2008).
Yoshida, K., Solomon, D. H. & Kim, S. C. Active-comparator design and new-user design in observational studies. Nat. Rev. Rheumatol. 11, 437–441 (2015).
Funding
This study was supported by the research grant from the SK Chemicals (#06-2020-0285), but designed and drafted independently of the funding source and the authors retained the right for the final wording.
Author information
Authors and Affiliations
Contributions
Conceptualization and methodology: Eun Ha Kang Data curation, formal analysis, and software: Anna ShinPreparation of original draft: Eun Ha Kang, Min Jung Kim, Jung Yoon Pyo, Se Rim ChoiReview and Editing: All authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kim, M.J., Pyo, J.Y., Choi, S.R. et al. Comparative risk of retinal microvascular disorders in patients with gout initiating febuxostat versus allopurinol: a population-based cohort study. Sci Rep 15, 28939 (2025). https://doi.org/10.1038/s41598-025-00551-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-00551-z