Abstract
The relationship between the Triglyceride-Glucose (TyG) index and Obstructive Sleep Apnea Hypopnea Syndrome (OSAHS) remains unclear. This study aimed to investigate the association between TyG index-related parameters (TyG, TyG-Body Mass Index (BMI), TyG-Waist Circumference (WC)) and OSAHS. Consecutive subjects referred to our sleep center were enrolled in this study and categorized into four groups based on the severity of OSAHS, as determined by the apnea-hypopnea index (AHI). Multivariate regression analysis was performed to identify independent risk factors associated with TyG index-related parameters in OSAHS. The study included 1250 participants, categorized into 114 without OSAHS, 212 with mild OSAHS, 257 with moderate OSAHS, and 667 with severe OSAHS. Significant differences were observed in fasting glucose levels, TyG, TyG-BMI, and TyG-WC across increasing severity of OSAHS. Multivariate regression showed that TyG-BMI and TyG-WC were independently associated with oxygen desaturation index (ODI), mean oxygen saturation (MSO2), and age (all p < 0.05). TyG was significantly associated with BMI, ODI, and sex (all p < 0.05). FPG was linked to BMI, age and MSO2, while insulin was associated with lowest oxygen saturation (LaSO2) and AHI (all p < 0.05). OSAHS-induced intermittent hypoxia is independently associated with increased TyG index-related parameters. These findings suggest that intermittent hypoxia may contribute to metabolic disturbances and insulin resistance, highlighting the need for further investigation into its clinical implications.
Similar content being viewed by others
Background
Obstructive sleep apnea hypopnea syndrome (OSAHS) is a prevalent breathing disorder characterized by repeated episodes of reduced or paused breathing during sleep. Recent analyses indicate that nearly 1 billion individuals aged 30–69 may suffer from this disorder worldwide, with approximately 425 million having moderate to severe symptoms1. Several studies have provided compelling evidence showing a strong association between OSAHS and several chronic conditions, notably cardiovascular and cerebrovascular diseases2,3,4. Delayed treatment of OSAHS significantly raises the risk of cardiovascular disease(CVD) and is associated with higher overall mortality rates5. Therefore, OSAHS is now widely recognized as a major global health concern, imposing a substantial burden on healthcare systems.
The triglyceride-glucose index (TyG), introduced and validated by Simental-Mendía et al.6 in 2008, is a combined measure utilizing fasting plasma glucose (FPG) and triglycerides. It serves as a dependable substitute for assessing insulin resistance (IR)7, 8. Studies have demonstrated that the TyG index outperforms the homeostatic model assessment of IR (HOMA-IR) in evaluating IR, which plays a crucial role in the development of CVD9, 10. Moreover, research suggests that TyG-related markers incorporating obesity metrics, such as TyG-WC, are more effective surrogate markers for reflecting IR severity compared to TyG alone11, 12. A recent meta-analysis highlighted that the TyG index is a convenient marker for assessing insulin resistance and is used in the diagnosis and prognosis of OSAHS13. Individuals with OSA generally exhibit significantly higher TyG index levels than healthy controls. Furthermore, the TyG index may independently indicate an elevated risk of OSAHS14. Additionally, it was found that elevated baseline TyG-WC levels are associated with an increased risk of first myocardial infarction, suggesting that TyG and related parameters could help identify high-risk CVD in patients with OSAHS15.
To the best of our knowledge, the relationship between TyG, its related parameters, and obstructive sleep apnea with associated hypoxia has not been comprehensively evaluated. Therefore, this study aimed to investigate the association of TyG and its related parameters, which serve as easily accessible clinical markers of insulin resistance in patients with OSAHS.
Methods
Study design and subjects
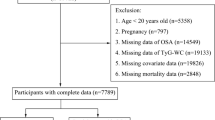
All patients who presented to our sleep laboratory with symptoms of sleep apnea between December 2013 and October 2022 were included in this study. Subjects were selected based on the concurrent completion of fasting glucose and lipid examinations. All patients underwent an Epworth Sleepiness Scale (ESS) assessment and a detailed questionnaire on sleep symptoms, history of alcohol consumption and smoking, medical history, and medication use. Exclusion criteria included a prior diagnosis of OSAHS, use of CPAP within the past three months, severe chronic cardiopulmonary disease, or acute inflammatory disease. This study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethics Committee of the First Affiliated Hospital of Fujian Medical University. Written informed consent was obtained from all participants prior to enrollment.
Anthropometric and biochemical measurements
Body weight and height were measured without shoes and in light clothing during the morning using standardized equipment. Body mass index (BMI) was calculated as weight in kilograms divided by the square of height in meters (kg/m²). Waist circumference was measured at a point midway between the lower costal margin and the iliac crest. Fasting blood samples were collected in the morning to measure serum levels of glucose, alanine aminotransferase (ALT), aspartate aminotransferase (AST), and lipid profile parameters including total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), and triglycerides (TG), and these samples were analyzed using the Modular P800 autoanalyzer (Roche, Tokyo, Japan). Insulin concentrations were determined by radioimmunoassay (Linco Research, St. Charles, MO, USA). The level of glycated hemoglobin A1c (HbA1c) was determined by high-performance liquid chromatography (VARIANT II; Bio-Rad, CA, USA). TyG, TyG-WC, and TyG-BMI were calculated according to the following formulas: TyG = ln [triglycerides (mg/dl) × glucose (mg/dl)/2]; TyG-WC = TyG × waist circumference; TyG-BMI = TyG × BMI13.
Polysomnographical evaluation
Overnight polysomnography (performed using the P Series Sleep System, Compumedics, Melbourne, Australia) involved recording several physiological parameters: electroencephalography, electrooculography, electromyography, airflow (via nasal and oral thermistors), respiratory effort (using thoracic and abdominal impedance belts), arterial oxyhemoglobin saturation (measured by pulse oximetry), snoring (via tracheal microphone), and body position (tracked using sensors). The polysomnography typically began at approximately 22:00 h (lights off) and concluded around 06:00 h (lights on). Apnea was defined as airflow reductions ≥ 90% from baseline lasting ≥ 10 s. Hypopnea was defined as a ≥ 30% reduction in airflow lasting ≥ 10 s and associated with ≥ 4% oxyhemoglobin desaturation. The apnea-hypopnea index (AHI) was calculated as the number of apneas and hypopneas per hour of sleep. The oxygen desaturation index (ODI) represented the number of ≥ 4% dips in oxygen saturation per hour of total sleep time. Polysomnographic parameters including lowest oxygen saturation (LaSO2) and mean nocturnal oxygen saturation (MSO2) were also recorded. All polysomnography studies were scored in accordance with the 2012 criteria of the American Academy of Sleep Medicine16. The severity of OSAHS was classified based on AHI as follows: no OSAHS (< 5 events/h), mild OSAHS (5 to < 15 events/h), moderate OSAHS (15 to < 30 events/h), and severe OSAHS (≥ 30 events/h).
Statistical analysis
Statistical analyses were conducted using SPSS version 26.0 (SPSS Inc., Chicago, IL, USA). Normality of all variables was assessed prior to analysis. Data were presented as mean ± standard deviation for normally distributed variables, median (interquartile range) for skewed variables, and number (percentage) for categorical variables. For normally distributed continuous variables, comparisons among multiple groups were performed using one-way analysis of variance (ANOVA). Non-normally distributed continuous variables were compared using the Kruskal-Wallis H test for multiple-group comparisons. Chi-square test or Fisher’s exact test was utilized for categorical variables. Correlations between variables were examined using the Pearson correlation analysis. All non-normally distributed descriptive data were log-transformed before multivariate analysis. Multiple linear regression analysis with stepwise selection was applied to identify independent predictors of TyG index-related parameters. Statistical significance was set at p < 0.05.
Results
Anthropometric and polysomnographic variables in all patients according to severity of OSAHS
The study enrolled 1250 participants aged 48.00 ± 12.38 years, with a mean BMI of 28.18 ± 11.05 kg/m², distributed as follows: 114 without OSAHS, 212 with mild OSAHS, 257 with moderate OSAHS, and 667 with severe OSAHS. Comprehensive demographic data for all subjects, stratified by age groups, are summarized in Table 1. Significant differences (all p < 0.05) were observed among the OSAHS severity categories in age, BMI, neck circumference, waist circumference, medical histories (including hyperlipidemia and diabetes), and smoking status. The proportion of males increased with the severity of OSAHS (63.18% in the no OSAHS group vs. 87.41% in the severe OSAHS group, p < 0.001). Furthermore, significant variations (all p < 0.05) were noted across these groups in sleep-related variables, such as AHI, ODI, LaSO2, MSO2, and indicators of excessive sleepiness (ESS).
Biochemical variables in all patients according to severity of OSAHS
Table 2 presents biochemical parameters related to glucose metabolism, lipid metabolism, and liver function. Insulin and HbA1c showed no significant differences among the four groups. However, fasting glucose levels and lipid metabolism markers including TC and TG varied significantly across the four OSAHS severity groups (all p < 0.05). Furthermore, there was a statistically significant increase in the concentrations of TyG, TyG-BMI, and TyG-WC from the control group to the severe OSAHS group (all p < 0.05).
Sex-based differences in clinical and metabolic characteristics
To further explore the influence of sex on clinical and metabolic parameters, we performed a subgroup analysis based on sex. As shown in Table 3, males were significantly younger than females and had higher values of BMI, WC, NC, AHI, and ODI, along with lower LaSO2 and MSO2 (all p < 0.001). Moreover, males showed significantly higher levels of TyG, TyG-BMI, and TyG-WC (all p < 0.001). No significant sex differences were found in FPG and insulin levels. These findings suggest that sex differences may influence both the severity of OSAHS and the associated metabolic burden.
Associations between TyG index-related parameters and indices of sleep apnea
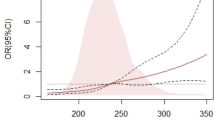
Table 4 summarizes the associations between parameters related to the TyG and indices of sleep apnea. Significant correlations were found between FPG and sleep apnea indices (all p < 0.05), albeit with relatively small correlation coefficients. Ln Insulin was only correlated with Ln LaSO2 (r=-0.227, p < 0.001). Moreover, TyG, TyG-BMI, and TyG-WC showed associations with sleep apnea indices including Ln AHI, Ln ODI, Ln LaSO2, and Ln MSO2 (all p < 0.05).
Multivariate analysis identifying predictors of glucose metabolism and TyG-related parameters
Multivariate analysis was conducted to identify independent predictors of glucose metabolism and TyG-related parameters. When ln FPG was the dependent variable, BMI and age emerged as independent risk factors (β = 0.152, p < 0.001; β = 0.178, p < 0.001), while Ln MSO2 was negatively associated with ln FPG (β=-0.075, p = 0.014). For Ln Insulin, Ln LaSO2 (β=-0.335, p < 0.001) and Ln AHI (β=-0.206, p = 0.002) were identified as significant predictors. For Ln HbA1c, BMI (β = 0.179, p < 0.001) and age (β = 0.170, p < 0.001) were significant independent predictors. Regarding TyG, BMI (β = 0.168, p < 0.001), Ln ODI (β = 0.108, p < 0.001), and gender (β=-0.085, p = 0.003) were identified as independent predictors. For TyG-BMI, Ln MSO2 (β=-0.229, p < 0.001), Ln ODI (β = 0.196, p < 0.001), and age (β=-0.133, p < 0.001) were independently associated. Similarly, for TyG-WC, Ln ODI (β = 0.225, p < 0.001), Ln MSO2 (β=-0.206, p < 0.001), gender (β=-0.077, p = 0.001), and age (β=-0.061, p = 0.027) were significant predictors (Table 5).
Discussion
This study focuses on the relationship between TyG index-related parameters (TyG, TyG-BMI, TyG-WC) and OSAHS, particularly exploring how intermittent hypoxia may influence metabolic disturbances. Our findings show significant variations in TyG-related parameters across OSAHS severities, with intermittent hypoxia indicators (ODI and MSO2) independently associated with TyG-BMI and TyG-WC, and a significant correlation between TyG index and ODI. These results suggest that intermittent hypoxia in OSAHS may contribute to insulin resistance and metabolic disturbances.
Several studies consistently show a significant association between OSAHS, insulin resistance, and type 2 diabetes17,18,19. Chronic intermittent hypoxia in OSAHS is linked to insulin resistance and pancreatic beta-cell dysfunction20, with even mild decreases in oxyhemoglobin saturation associated with metabolic dysfunction and cardiovascular issues21. The severity of OSAHS (measured by AHI) is also strongly linked to vascular changes, coronary artery disease, arrhythmias, and heart failure22. The TyG index has been demonstrated to be a valuable tool for evaluating glycemic control in patients with T2DM, with a positive correlation to HbA1c and HOMA-IR. Therefore, the TyG index offers a straightforward and cost-effective alternative for assessing glycemic control in diabetic patients23, 24. In large populations, the TyG index serves as a practical alternative for measuring insulin resistance, and its correlation with sleep disturbances has already been documented25. Furthermore, TyG-BMI and TyG-WC have been confirmed as clinically significant indicators of insulin resistance, with associations to hypertension and myocardial infarction. Based on these findings, this study aimed to evaluate the association between TyG index-related parameters and OSAHS.
We found that as the severity of OSAHS increased, there were corresponding elevations in fasting glucose, TyG, TyG-BMI, and TyG-WC, suggesting that OSAHS severity may directly influence metabolic dysfunction. A recent meta-analysis indicates that the diagnostic accuracy of the TyG index is comparable to that of other anthropometric indices13. This is consistent with a study conducted in Korea, which found that elevated TyG index levels are associated with an increased risk of developing OSAHS26. Further multivariate regression analysis revealed several significant findings. ODI, MSO2, and age were identified as significant contributors to TyG-BMI and TyG-WC. This suggests that factors related to nocturnal hypoxia and aging may influence the metabolic implications of OSAHS, as indicated by TyG in combination with BMI and waist circumference. Moreover, BMI and ODI were found to be major independent predictors of TyG, emphasizing their role in influencing insulin resistance and cardiovascular risk associated with OSAHS. These results align with prior studies, which indicate that OSAHS is linked to metabolic disturbances and cardiovascular complications, with TyG index-related parameters acting as potential mediators. Interestingly, age was found to be a protective factor for TyG-BMI and TyG-WC. This may be attributed to the fact that with advancing age, both BMI and waist circumference tend to decrease, potentially mitigating the metabolic impact of OSAHS.
Insulin plays a critical for suppressing triglyceride production in the liver. The severity of intermittent hypoxia in OSAHS is linked to increased insulin resistance, which subsequently elevates TyG index levels. Studies suggest that intermittent hypoxia induces insulin resistance and glucose intolerance, contributing to liver fat accumulation27. A small clinical trial involving OSAHS patients showed disrupted triglyceride metabolism, which improved with continuous positive airway pressure therapy28. Furthermore, research indicates that intermittent hypoxia may influence the TyG index and lipid metabolism by promoting the formation of oxidatively stressed dysfunctional lipids, which in turn affects circulating cholesterol levels29. Interestingly, our study revealed a paradoxical finding: intermittent hypoxia was positively correlated with insulin levels, while AHI showed a negative correlation. This could be due to compensatory mechanisms in severe OSAHS, where increased sympathetic activity or other regulatory changes might alter insulin secretion. Additionally, factors like body weight, lipid metabolism, and the duration and frequency of hypoxic episodes might also influence this relationship. To further validate these findings, future studies should explore the underlying mechanisms through longitudinal research, including more comprehensive metabolic assessments and monitoring of insulin and hypoxia levels over time in larger, more diverse OSAHS populations.
Building upon these findings, we also examined sex-specific differences in clinical and metabolic characteristics. Male patients exhibited significantly higher AHI, ODI, and neck circumference, and lower MSO2 and LaSO2, indicating more severe nocturnal hypoxia. Although females had slightly higher HbA1c levels, TyG, TyG-BMI, and TyG-WC were significantly higher in males, likely reflecting greater visceral adiposity and hypoxic burden. The proportion of males increased with OSAHS severity. Prior studies have reported a male predominance in OSAHS, possibly due to differences in upper airway anatomy and fat distribution30. Similarly, sex differences in the TyG index have been observed, with higher levels in males. These findings support sex-specific evaluation and management strategies in OSAHS.
These observations underscore the clinical relevance of TyG index-related parameters in assessing the metabolic burden associated with OSAHS. Beyond their role as markers of metabolic dysfunction, these parameters may serve as indicators of cardiovascular risk in OSAHS patients. This dual perspective highlights the importance of comprehensive metabolic assessment in managing individuals with sleep-disordered breathing, aiming not only to alleviate symptoms but also to mitigate long-term cardiovascular complications. Despite these insights, several limitations should be considered. First, the cross-sectional design of our study precludes the establishment of causal relationships between TyG index-related parameters and OSAHS. Longitudinal studies are needed to clarify temporal associations and to validate the TyG index as a predictive tool for metabolic outcomes in OSAHS patients. Second, the relatively small number of female participants, particularly in the moderate and severe OSAHS groups, may have introduced selection bias and limited the statistical power of sex-specific analyses. This imbalance could potentially affect the interpretation of sex-related differences and reduce the generalizability of our findings. Future studies with larger and more balanced sex distributions are warranted to better characterize gender-specific metabolic responses in OSAHS and to minimize potential bias.
In conclusion, our study reinforces the growing evidence linking OSAHS with metabolic disturbances, with a particular focus on the role of intermittent hypoxia. The TyG index and its related parameters, such as TyG-BMI and TyG-WC, serve as promising markers for understanding the metabolic dysfunctions associated with OSAHS. Our findings suggest that intermittent hypoxia plays a significant role in the development of insulin resistance and lipid metabolism abnormalities in these patients. Future research should aim to further elucidate the clinical implications of these findings, particularly in relation to the impact of intermittent hypoxia, and evaluate the effectiveness of interventions targeting metabolic abnormalities in OSAHS patients.
Data availability
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- TyG:
-
Triglyceride-glucose
- OSAHS:
-
Obstructive sleep apnea hypopnea syndrome
- BMI:
-
Body mass index
- WC:
-
Waist circumference
- NC:
-
Neck circumference
- AHI:
-
Apnea–hypopnea index
- ODI:
-
Oxygen desaturation index
- LaSO2 :
-
Lowest oxygen saturation
- MSO2 :
-
Mean oxygen saturation
- IR:
-
Insulin resistance
- HOMA-IR:
-
Homeostatic model assessment of IR
- CVD:
-
Cardiovascular disease
- ESS:
-
Epworth sleepiness scale
- FPG:
-
Fasting plasma glucose
- TC:
-
Total cholesterol
- TG:
-
Triglycerides
- HDL-CL:
-
High-density lipoprotein-cholesterol
- LDL-C:
-
Low-density lipoprotein-cholesterol
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- HbA1c:
-
Glycated hemoglobin
References
Benjafield, A. V. et al. Estimation of the global prevalence and burden of obstructive sleep apnoea: a literature-based analysis. Lancet Respir Med. 7 (8), 687–698. https://doi.org/10.1016/S2213-2600(19)30198-5 (2019). [published Online First: 2019/07/14].
Redline, S. Screening for obstructive sleep apnea: implications for the sleep health of the population. JAMA 317 (4), 368–370. https://doi.org/10.1001/jama.2016.18630 (2017). [published Online First: 2017/01/25].
Baillieul, S. et al. Sleep Apnoea and ischaemic stroke: current knowledge and future directions. Lancet Neurol. 21 (1), 78–88. https://doi.org/10.1016/S1474-4422(21)00321-5 (2022). [published Online First: 2021/12/24].
Mesarwi, O. A., Loomba, R. & Malhotra, A. Obstructive sleep apnea, hypoxia, and nonalcoholic fatty liver disease. Am. J. Respir Crit. Care Med. 199 (7), 830–841. https://doi.org/10.1164/rccm.201806-1109TR (2019). [published Online First: 2018/11/14].
Ge, X. et al. Is obstructive sleep apnea associated with cardiovascular and all-cause mortality? PLoS One. 8 (7), e69432. https://doi.org/10.1371/journal.pone.0069432 (2013). [published Online First: 2013/08/13].
Simental-Mendia, L. E., Rodriguez-Moran, M. & Guerrero-Romero, F. The product of fasting glucose and triglycerides as surrogate for identifying insulin resistance in apparently healthy subjects. Metab. Syndr. Relat. Disord. 6 (4), 299–304. https://doi.org/10.1089/met.2008.0034 (2008). [published Online First: 2008/12/11].
Li, L. et al. Association between Triglyceride-Glucose index and risk of periodontitis: A Cross-Sectional study. Int. J. Gen. Med. 14, 9807–9816. https://doi.org/10.2147/IJGM.S339863 (2021). [published Online First: 2021/12/24].
Yao, L. et al. The Triglyceride-Glucose index is associated with diabetic retinopathy in Chinese patients with type 2 diabetes: A Hospital-Based, nested, Case-Control study. Diabetes Metab. Syndr. Obes. 14, 1547–1555. https://doi.org/10.2147/DMSO.S294408 (2021). [published Online First: 2021/04/17].
Ormazabal, V. et al. Association between insulin resistance and the development of cardiovascular disease. Cardiovasc. Diabetol. 17 (1), 122. https://doi.org/10.1186/s12933-018-0762-4 (2018). [published Online First: 2018/09/02].
Laakso, M. & Kuusisto, J. Insulin resistance and hyperglycaemia in cardiovascular disease development. Nat. Rev. Endocrinol. 10 (5), 293–302. https://doi.org/10.1038/nrendo.2014.29 (2014). [published Online First: 2014/03/26].
Kim, H. S. et al. Triglyceride Glucose-Waist circumference is superior to the homeostasis model assessment of insulin resistance in identifying nonalcoholic fatty liver disease in healthy subjects. J. Clin. Med. 11 (1). https://doi.org/10.3390/jcm11010041 (2021). [published Online First: 2022/01/12].
Khamseh, M. E. et al. Triglyceride glucose index and related parameters (Triglyceride glucose-Body mass index and triglyceride glucose-Waist Circumference) identify nonalcoholic fatty liver and liver fibrosis in individuals with overweight/obesity. Metab. Syndr. Relat. Disord. 19 (3), 167–173. https://doi.org/10.1089/met.2020.0109 (2021). [published Online First: 2020/12/02].
Behnoush, A. H. et al. Triglyceride-glucose index and obstructive sleep apnea: a systematic review and meta-analysis. Lipids Health Dis. 23 (1), 4. https://doi.org/10.1186/s12944-024-02005-3 (2024). [published Online First: 2024/01/08].
Pei, H. et al. Association between triglyceride glucose index and sleep disorders: results from the NHANES 2005–2008. BMC Psychiatry. 23 (1), 156. https://doi.org/10.1186/s12888-022-04434-9 (2023). [published Online First: 2023/03/11].
Hu, J. et al. Association between triglyceride glucose Index-Waist circumference and risk of first myocardial infarction in Chinese hypertensive patients with obstructive sleep apnoea: an observational cohort study. Nat. Sci. Sleep. 14, 969–980. https://doi.org/10.2147/NSS.S362101 (2022). [published Online First: 2022/05/27].
Berry, R. B. et al. Rules for scoring respiratory events in sleep: update of the 2007 AASM manual for the scoring of sleep and associated events. Deliberations of the sleep apnea definitions task force of the American academy of sleep medicine. J. Clin. Sleep. Med. 8 (5), 597–619. https://doi.org/10.5664/jcsm.2172 (2012). [published Online First: 2012/10/16].
Punjabi, N. M. et al. Sleep-disordered breathing, glucose intolerance, and insulin resistance: the sleep heart health study. Am. J. Epidemiol. 160 (6), 521–530. https://doi.org/10.1093/aje/kwh261 (2004). [published Online First: 2004/09/09].
Botros, N. et al. Obstructive sleep apnea as a risk factor for type 2 diabetes. Am. J. Med. 122 (12), 1122–1127. https://doi.org/10.1016/j.amjmed.2009.04.026 (2009). [published Online First: 2009/12/05].
Togeiro, S. M. et al. Consequences of obstructive sleep apnea on metabolic profile: a Population-Based survey. Obes. (Silver Spring). 21 (4), 847–851. https://doi.org/10.1002/oby.20288 (2013). [published Online First: 2013/05/29].
Punjabi, N. M. & Beamer, B. A. Alterations in glucose disposal in Sleep-disordered breathing. Am. J. Respir Crit. Care Med. 179 (3), 235–240. https://doi.org/10.1164/rccm.200809-1392OC (2009). [published Online First: 2008/11/18].
Stamatakis, K. et al. Fasting glycemia in sleep disordered breathing: Lowering the threshold on oxyhemoglobin desaturation. Sleep 31 (7), 1018–1024 (2008). [published Online First: 2008/07/26].
Javaheri, S. et al. Sleep apnea: types, mechanisms, and clinical cardiovascular consequences. J. Am. Coll. Cardiol. 69 (7), 841–858. https://doi.org/10.1016/j.jacc.2016.11.069 (2017). [published Online First: 2017/02/18].
Hong, S., Han, K. & Park, C. Y. The triglyceride glucose index is a simple and low-cost marker associated with atherosclerotic cardiovascular disease: a population-based study. BMC Med. 18 (1), 361. https://doi.org/10.1186/s12916-020-01824-2 (2020). [published Online First: 2020/11/26].
Irace, C. et al. Markers of insulin resistance and carotid atherosclerosis. A comparison of the homeostasis model assessment and triglyceride glucose index. Int. J. Clin. Pract. 67 (7), 665–672. https://doi.org/10.1111/ijcp.12124 (2013). [published Online First: 2013/06/14].
Koren, D. & Taveras, E. M. Association of sleep disturbances with obesity, insulin resistance and the metabolic syndrome. Metabolism 84, 67–75. https://doi.org/10.1016/j.metabol.2018.04.001 (2018). [published Online First: 2018/04/10].
Kang, H. H., Kim, S. W. & Lee, S. H. Association between triglyceride glucose index and obstructive sleep apnea risk in Korean adults: a cross-sectional cohort study. Lipids Health Dis. 19 (1), 182. https://doi.org/10.1186/s12944-020-01358-9 (2020). [published Online First: 2020/08/11].
Fernandes, J. L. et al. Chronic intermittent Hypoxia-Induced dysmetabolism is associated with hepatic oxidative stress, mitochondrial dysfunction and inflammation. Antioxid. (Basel). 12 (11). https://doi.org/10.3390/antiox12111910 (2023). [published Online First: 2023/11/25].
Drager, L. F. et al. Obstructive sleep apnea and effects of continuous positive airway pressure on triglyceride-rich lipoprotein metabolism. J. Lipid Res. 59 (6), 1027–1033. https://doi.org/10.1194/jlr.M083436 (2018). [published Online First: 2018/04/10].
Lavie, L. Oxidative stress–a unifying paradigm in obstructive sleep apnea and comorbidities. Prog Cardiovasc. Dis. 51 (4), 303–312. https://doi.org/10.1016/j.pcad.2008.08.003 (2009). [published Online First: 2008/12/27].
Zhang, W., Phillips, A. & Wang, B. Y. Correlation analysis between airway volume and risk of sleep apnea/periodontitis. J. Oral Maxillofac. Res. 13 (2), e5. https://doi.org/10.5037/jomr.2022.13205 (2022). [published Online First: 2022/08/12].
Funding
The work was supported by the Natural Science Fundation of Fujian Province (No.2024J01573), Joint Funds for the Innovation of Science and Technology, Fujian Province (No.2023Y9070), Fujian Provincial Health Technology Project (No.2024CXA020) and National Natural Science Foundation of China (No.81870074).
Author information
Authors and Affiliations
Contributions
Hansheng Xie and Jiefeng Huang sequence/data analysis and preparation of the manuscript; Menglan Chen, study design and preparation of the manuscript; Yue Zhong, collected data; Jianming Zhao, analyzed data; Qichang Lin and Ningfang Lian, contributed in experimental design and manuscript revision; Hansheng Xie and Jiefeng Huang contributed equally to this work.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the First Affiliated Hospital of Fujian Medical University. Informed consent was obtained from all individual participants included in the study.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Xie, H., Huang, J., Chen, M. et al. Association between triglyceride glucose index related parameters and obstructive sleep apnea hypopnea syndrome in a cross sectional study. Sci Rep 15, 16345 (2025). https://doi.org/10.1038/s41598-025-01306-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-01306-6