Abstract
Akabane virus (AKAV) is an arbovirus belonging to the genus Orthobunyavirus within the family Peribunyaviridae, order Elliovirales1, capable of infecting ruminants such as cattle, sheep, and goats. This arthropod-borne virus is transmitted by mosquitoes or midges and has been recognized as a contributor to outbreaks of reproductive disorders. This study investigates the seroprevalence of AKAV infection in cattle across various regions in Egypt, along with an analysis of potential risk factors contributing to the infection. Serum samples from 540 cattle were collected randomly from four governorates and tested using a commercial competitive ELISA kit. The seroprevalence of AKAV infection was found to be 24.4%. Univariable statistical analysis indicated that factors such as locality, age, sex, breed, season, and history of abortion (p < 0.05) were significantly associated with the infection, while herd size did not show a significant association (p > 0.05). In addition, the multivariable logistic regression analysis indicated that the likelihood of AKAV infection was significantly higher in Alexandria (OR = 2.9, P = 0.001), in cattle over 5 years of age (OR = 3.7, P = 0.002), among females (OR = 2.7, P = 0.034), in Holstein breeds (OR = 2.1, P = 0.003), during the winter season (OR = 3.7, P < 0.0001), and in cattle with a history of abortion (OR = 2.5, P < 0.0001). These findings emphasize the need for ongoing monitoring of the disease in ruminants to accurately assess its burden and raise awareness. Such efforts will promote early detection and support the development of effective control measures for the disease in this region.
Similar content being viewed by others
Introduction
Akabane virus (AKAV) is arthropod-borne, negative-sense RNA virus with segmented structure. This virus belongs to the Simbu serogroup of the Orthobunyavirus genus, family Bunyaviridae, and is similar to Schmallenberg virus (SBV) that emerged in 2011 2,3,4. Although biting midges (Culicoides spp.) seem to be the main vectors, AKAV has been isolated from mosquitoes on multiple occasions5,6. Cattle, buffalo, sheep, goats, horses, deer, and pigs are among the many wild ruminants and animals that are infected with AKAV7,8.
Akabane disease (AD) primarily affects cattle and, in rare cases, sheep and goats. Adult cattle infected with AKAV show no specific symptoms after short transit fever9. But some AKAV strains can pass through the placenta of pregnant cows and cause several problems in the developing foetus, including abortion. These birth problems include stillbirths, premature births, and various brain abnormalities such microcephaly and porencephaly, depending on the stage of pregnancy2,10.
Akabane disease has caused significant economic losses in several countries. In addition, the main financial losses associated with AKAV infection in dairy cattle are infertility, preterm abortion, dystocia during delivery, and a marked decline in the milk yield of infected cows11,12.
The virus was firstly discovered in 1959 in Gumma Prefecture, Japan, in mosquitoes (Aedes vexans and Culex tritaeniorhynchus)9. It is commonly recognized that AKAV is widely distributed throughout East Asian in tropical and subtropical regions, Australia, the Middle East, and Africa13,14. AKAV has been detected in wild animals in Africa using virus isolation, molecular techniques, and serology15, as well as domestic animals in Tanzania16, Nigeria17, Kenya18, Sudan19 and Egypt20.
Various diagnostic approaches have been developed for diagnosing AD in different animal species. Serological approaches, including as viral neutralization tests, immunohistochemistry staining, ELISAs, and dot immunobinding assays are routinely used to diagnose AD21,22,23,24. Polymerase chain reaction (PCR) methods, including real-time RT-PCR [31], multiplex RT-PCR25, nested reverse transcription PCR (RT-PCR)26, and reverse transcription loop-mediated isothermal amplification assay (RT-LAMP)27, have been successfully used to identify the AKAV genome.
Although the disease has been reported within the country, there is presently no vaccination program or established insect management control in place to prevent its spread. This is particularly concerning given the frequent reporting of AD in neighboring countries and other countries from which Egypt imports animals.
Therefore, the purpose of this study is to evaluate the seroprevalence of AD in cattle in four Egyptian governorates, as well as to assess the associated risk factors for infection.
Materials and methods
Ethical statement
This study was authorized by the Benha University Faculty of Veterinary Medicine’s ethical committee. Furthermore, all procedures followed the guidelines and regulations of Benha University’s ethics council. Consent and permission from the owner were obtained for all animals involved in the study. The research followed ARRIVE criteria.
Study area
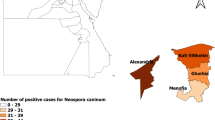
The study was conducted from March 2023 to February 2024 across four Egyptian governorates, namely Kafr Elsheikh, Menofia, Qalybia, and Alexandria (Fig. 1).
MAP showed locations of the studies governorates (MAP generated by QGIS software).
The three governorates (Kafr Elsheikh, Menofia and Qalybia) situated at Nile Delta of Egypt, which characterized by a semi-arid climate typical of the Nile Delta region. Summers are quite hot, with temperatures averaging 30 °C to 35 °C. Winters are cooler, with temperatures between 8 °C and 18 °C. Rainfall is sparse, mostly occurring during the winter months, and it is generally less than what is seen in the northern coastal regions. Alexandria has a classic Mediterranean climate with more rainfall compared to the other governorates, whereas summers are warm, with temperatures ranging from 25 °C to 30 °C, while winters are mild, with temperatures ranging from 10 °C to 18 °C. It receives significant rainfall, mostly during the winter months, with an average annual precipitation of around 200 mm to 400 mm.
Climatic changes, such as rising temperatures, increased rainfall, and humidity, significantly influence the spread of mosquitoes and the Akabane virus in the selected governorates. Without adequate vector control measures, such as mosquito management and vaccination programs, these regions are at a heightened risk of Akabane outbreaks, threatening livestock health and economic stability.
Sample size and sampling
The number of samples required to evaluate the seroprevalence of AD in cattle was calculated using the formula provided by Thrusfield28.
N = Z2Pexp(1-Pexp)/d2.
where n is the required number of samples, p is the expected prevalence, Z is the 95% confidence interval, and d is the absolute error 5%. The precision was improved by increasing the sample size from the calculated 384 to 540 samples. Blood sample (5mL) was collected from jugular vein of each examined cattle using vacutainer tube. The sera separated by centrifugation for ten min at 3000 rpm and kept at -20 C until serological analysis.
Serological analysis
The commercial ID Screen® Akabane Competition ELISA kit (ID.vet, Grabels, France) was used to identify specific Anti-G1 antibodies for AKAV in serum, according to the manufacturer’s instructions. Furthermore, this kit had no cross-reaction with other viruses in Family Bunyaviridae such as Rift Valley fever virus, Schmallenberg virus and Aino virus. In addition, it has highest correlation the virus neutralization test as verified by Li, et al.21. The results were read at an optical density (OD) of 450 nm with a microplate absorbance spectrophotometer reader (ALLSheng A101; China).
The optical densities (ODs) of each test sample were used to calculate the sample (S) to negative (N) ratio (S/N%) using the below formula.
S/N (%) = OD sample/OD negative control X 100.
Samples with a S/N% greater than 40% were judged negative, while those between 30% and 40% were regarded dubious. Samples with a S/N% less than 30% were considered positive.
Questionnaire
An epidemiological questionnaire was prepared during sampling to collect data on all animals in order to assess the risk factors associated with AKAV infection in cattle. The factors studied and their related categories are as follows: locality (Kafr ElSheikh, Menofia, Qalyubia and Alexandria), age (< 2 years, 2–5 years, >5years), sex (male/female), breed (mixed or Holstein), herd size (< 50, 50–70 and > 70), season (summer, spring, autumn, and winter) and history of abortion (yes/no).
Statistical analysis
The data was collected and statistically analyzed using Microsoft Excel. Further statistical analysis was performed with SPSS version 24 (IBM, USA). Univariable logistic regression was employed to evaluate the association between each variable and the seroprevalence of AKAV infection in cattle. P-values less than 0.05 were considered statistically significant.
A logistic regression analysis was performed to examine the relationship between AKAV antibodies and risk factors including locality, age, sex, breed, season, and history of abortion. Variables with a P-value less than 0.25 in the univariable analysis were included in a multivariate logistic regression model. The multivariate model was utilized to calculate the odds ratios (ORs) and confidence intervals (CIs) for each significant variable. The model’s fit was assessed using the Hosmer-Lemeshow goodness-of-fit test.
Results
The total seroprevalence of AKAV antibodies in investigated cattle samples was 24.4% (132/450). The prevalence rates ranged from the highest 36.2% (47/130) in Alexandria to the lowest 15.6% (21/135) in Qalyubia governorate. The prevalence rate varied substantially between governorates (P = 0.001), Table 1.
Moreover, the seroprevalence of AKAV antibodies was significantly higher in cattle older than five years 35% (77/220), and in females 26.4% (126/478). Holstien cattle revealed higher significant seroprevalence rate 29.5% (96/325) in comparison to mixed breed and the winter had the greatest seroprevalence of AKAV antibodies at 36.8% (67/182) when compared to other seasons. Furthermore, the animals with history of abortion revealed higher seroprevalence rate 32.9% than other animals, Table 1.
The results of multivariable logistic regression for different significant variables associated with AKAV infection in cattle revealed that Alexandria (OR = 2.9, 95%CI: 1.53–5.46, P = 0.001), age over 5 years (OR = 3.7, 95%CI: 1.65–8.48, P = 0.002), females (OR = 2.7, 95%CI: 1.08–6.96, P = 0.034), Holstein (OR = 2.1, 95%CI: 1.29–3.36, P = 0.003), winter (OR = 3.7, 95%CI: 1.84–7.51, P < 0.0001), and history of abortion (OR = 2.5, 95%CI: 1.58–3.83, P < 0.0001) were identified as risk factors for AKAV infection in cattle, Table 2.
Discussion
Akabane diseases frequently occurred in places with tropical or subtropical climatic conditions, including Egypt. The epidemiological data on the disease in Egypt is very scarce. Thus, this study aimed to determine the seroprevalence of AD in some Egyptian governorates and assess the associated risk factors.
In the present study, the overall seroprevalence of AKAV antibodies in cattle was 24.4% and significantly varied across studied governorates with the highest seroprevalence rate of 36.2% in Alexandria and lowest seroprevalence rate of 15.6% was observed in Qalyubia governorate.
The reported seroprevalence rate was lower than previously recorded in Egypt (54.3%)20, Sudan (29.4 to 47%)19,29, and Nigeria (70.1%)17, but greater than that reported in China (21.3%)8. In addition, A serological survey of AKAV infection in two sentinel herds of calves in Saudi Arabia’s Central and Eastern regions revealed a 70% infection rate in the Al-Ahsa oasis, closer to Iran, while none of the examined cattle had antibodies to the virus30. Other studies stated that the seroprevalence rates were 20.32% in China31, 87% in Israel32, 0.14% in Turkey33.
Additionally, several studies conducted in the same study areas have reported the presence of antibodies against bluetongue virus (BTV) in cattle, and camels34,35 and against AKAV in sheep and goats36.
The variation in seropositivity for AKAV across different regions may be influenced by factors such as geographic location, sampling methods, the immunological status of the animals examined, age, climatic conditions, and the population density of vectors, particularly Culicoides34,35,36,37,38,39,40,41.
Regarding age, the seropositivity for AKAV differed significantly across the age categories of the examined cattle, with older cattle (over 5 years) showing the highest seroprevalence rate (35%) compared to the other age groups. This finding is consistent with the results of Metwally, et al.20, who reported a significantly higher seroprevalence in older cattle compared to younger and middle-aged cattle. Similarly, Karami Boldaji, et al.42 observed a significant increase in AKAV seroprevalence with age. Furthermore, Ahi, et al.43 found that infection in adult animals is widespread in endemic locations, although clinical disease reports are rare, while the neurological disease accompanied to the infection in cattle of 2 to 7 years age has been seen44. In line with our findings, Oluwayelu, et al.17 reported a higher seropositivity rate in adult cattle (86.4%) compared to younger cattle (14.3%). Conversely, Elhassan, et al.19 observed a greater seroprevalence increase in mid-aged cattle (45.3%) than in adults (34.8%). This can be explained by the fact that older animals are more likely to have been exposed multiple times to infected Culicoides vectors throughout their lives, increasing the likelihood of AKAV infection45,46,47,48.
Regarding sex, females showed a higher prevalence rate than males, which is consistent with the findings of Metwally, et al.20 and Karami Boldaji, et al.42. In addition, Consistent with a recent study conducted in Sudan, the prevalence of AKAV was higher in females (33%) compared to males (14.3%)19. This suggests that dairy herds, which often breed females, provide conducive environments for the habitat of insect vectors19,49. Although Akabane virus affects both male and female cattle equally, it is often reported more frequently in males due to several management and exposure factors. Male cattle, especially breeding bulls, are typically kept for longer periods in herds compared to females, increasing their chances of exposure to the virus over time. In addition, males are often managed in environments with greater exposure to biting insects, such as Culicoides midges and mosquitoes, which are responsible for virus transmission14,19,50,51,52.
Moreover, the highest seropositivity was observed in imported Holstein cattle (29.5%) and Mixed cattle (16.7%), which come in agreement with findings of Metwally, et al.20. Dairy heifers from high-milk-producing breeds, such as Holstein, are often imported from Europe and raised for life in intensive dairy farms53. While, Mixed breeds are crossbreeds produced by the mating of native breed and foreign cattle, and they are typically raised on smallholder or semi-intensive farms, either alone or alongside other cattle breeds54. The significant variation in seropositivity among breeds may be the farming systems in which the cattle were raised. Consequently, it is anticipated that raising Mixed and Holstein breeds together under Egyptian farming conditions may heighten the likelihood of AKAV transmission between them55,56,57,58,59,60.
The results of the present study supports findings of19 that season is a risk factor for AKAV transmission. Seroprevalence rates increased significantly during the winter season compared to other seasons. Climatic condition of studied areas including warm temperatures, moderate rainfall, and higher humidity, have a significant impact on increasing the spread of mosquitoes and the Akabane virus in the selected governorates61,62.
The present finding lies in line with previous results of Elhassan, et al.19, where the seroprevalence of AKAV infection increased significantly among cattle with history of abortion or reproductive problems than apparent healthy animals. The high prevalence of infertility and abortion in Egypt underscores the importance of AKAV infection in bovine reproductive health, indicating a need for further investigation63,64. The higher prevalence of Akabane virus infection in cattle with a history of abortion is mainly attributed to the virus’s pathogenic effects on the developing fetus. Akabane virus is a well-known cause of reproductive disorders in cattle, including abortions, stillbirths, and congenital deformities65.
Conclusion
This study clearly confirmed the presence of AKAV antibodies in dairy cattle in Egypt. The lack of clinically predictable signs associated with AKAV infections, along with significant outbreaks of abortion and fetal malformations, may contribute to an underestimation of the disease’s importance in Egypt. The multivariable logistic regression model revealed that locality, age, sex, season, breed and history of abortion had significant effect on likelihood of AKAV infection in cattle. Because of livestock production is a vital sector of the Egyptian economy, further investigations into the epidemiology and socio-economic impacts of AKAV should be pursued.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Suda, Y., Murota, K., Shirafuji, H., Tanaka, S. & Yanase, T. Replication of Akabane virus and related orthobunyaviruses in a fetal-bovine-brain-derived cell line. Arch. Virol. 169, 133 (2024).
Kirkland, P. D. Akabane virus infection. Rev. Sci. Tech. 34, 403–410 (2015).
Collins, Á. B., Mee, J. F. & Kirkland, P. D. Pathogenicity and teratogenicity of Schmallenberg virus and Akabane virus in experimentally infected chicken embryos. Vet. Microbiol. 216, 31–37 (2018).
Nikitina, E. et al. Akabane and Schmallenberg diseases: similarities and differences. Сельскохозяйственная Биология, 48–52 (2013).
Sick, F., Beer, M., Kampen, H. & Wernike, K. Culicoides biting midges—underestimated vectors for arboviruses of public health and veterinary importance. Viruses 11, 376 (2019).
Yanase, T., Hayama, Y., Shirafuji, H., Tsutsui, T. & Terada, Y. Surveillance of Culicoides biting midges in Northern Honshu, Japan, during the period of Akabane virus spread. J. Vet. Med. Sci. 81, 1496–1503 (2019).
Oğuzoğlu, T. Ç. Akabane virus infection in ruminants. Anim. Health Prod. Hygiene. 7, 592–595 (2018).
Wang, J., Blasdell, K. R., Yin, H. & Walker, P. J. A large-scale serological survey of Akabane virus infection in cattle, Yak, sheep and goats in China. Vet. Microbiol. 207, 7–12 (2017).
OYA, A., OKUNO, T., OGATA, T., KOBAYASHI, I. & MATSUYAMA, T. Akabane, a new arbor virus isolated in Japan. Jpn. J. Med. Sci. Biol. 14, 101–108 (1961).
Bryant, J. E. et al. Isolation of arboviruses from mosquitoes collected in Northern Vietnam. Am. J. Trop. Med. Hyg. 73, 470–473 (2005).
Horikita, T. et al. Loss of milk yield due to Akabane disease in dairy cows. J. Vet. Med. Sci. 67, 287–290 (2005).
Gorjidooz, M., Raoofi, A., Rahimabadi, P. D., Masoudifard, M. & Mardjanmehr, S. H. Study of Akabane disease in an Iranian dairy herd: a re-emerging disease. Veterinary Res. Communications, 1–12 (2024).
Yanase, T., Murota, K. & Hayama, Y. Endemic and emerging arboviruses in domestic ruminants in East Asia. Front. Veterinary Sci. 7, 168 (2020).
Kono, R. et al. Bovine epizootic encephalomyelitis caused by Akabane virus in Southern Japan. BMC Vet. Res. 4, 1–10 (2008).
Al-Busaidy, S., Hamblin, C. & Taylor, W. Neutralising antibodies to Akabane virus in free-living wild animals in Africa. Trop. Anim. Health Prod. 19, 197–202 (1987).
Mathew, C. et al. Detection of serum neutralizing antibodies to Simbu sero-group viruses in cattle in Tanzania. BMC Vet. Res. 11, 1–9 (2015).
Oluwayelu, D. O., Aiki-Raji, C. O., Umeh, E. C., Mustapha, S. O. & Adebiyi A. I. Serological investigation of Akabane virus infection in cattle and sheep in Nigeria. Advances in virology 2936082 (2016). (2016).
Davies, F. G. & Jessett, D. A study of the host range and distribution of antibody to Akabane virus (genus bunyavirus, family Bunyaviridae) in Kenya. Epidemiol. Infect. 95, 191–196 (1985).
Elhassan, A. M., Mansour, M. E. & Shamon, A. A. & El Hussein, A. A serological survey of Akabane virus infection in cattle in Sudan. International Scholarly Research Notices 123904 (2014). (2014).
Metwally, S. et al. The first serological detection and risk factors analysis of Akabane virus in Egyptian cattle. Animals 13, 1849 (2023).
Li, X. et al. Comparative evaluation of two commercial ELISA kits for detection of antibodies against Akabane virus in cattle serum. BMC Vet. Res. 15, 1–6 (2019).
Oem, J. K., Kim, Y. H., Kim, S. H., Lee, M. H. & Lee, K. K. Serological characteristics of affected cattle during an outbreak of bovine enzootic encephalomyelitis caused by Akabane virus. Trop. Anim. Health Prod. 46, 261–263 (2014).
HALIGÜR, M., Hasircioglu, S., Ozmen, O., Kale, M. & Aydoğan, A. Immunohistochemical evaluation of Akabane virus infection in aborted and new-born calves. Veterinární Medicína 59 (2014).
Kittelberger, R. et al. Evaluation of two commercial enzyme-linked immunosorbent assay kits for the detection of serum antibodies against Akabane virus in cattle. J. Vet. Diagn. Invest. 25, 645–648 (2013).
Stram, Y. et al. Detection and quantitation of Akabane and Aino viruses by multiplex real-time reverse-transcriptase PCR. J. Virol. Methods. 116, 147–154 (2004).
Akashi, H., Onuma, S., Nagano, H., Ohta, M. & Fukutomi, T. Detection and differentiation of Aino and Akabane Simbu serogroup bunyaviruses by nested polymerase chain reaction. Arch. Virol. 144, 2101–2109 (1999).
Qiao, J. et al. Rapid detection of Akabane virus by a novel reverse transcription loop-mediated isothermal amplification assay (RT-LAMP). Virol. J. 10, 1–5 (2013).
Thrusfield, M. Veterinary Epidemiology (Wiley, 2018).
Mohamed, M., Mellor, P. & P Taylor, W. Akabane virus: serological survey of antibodies in livestock in the Sudan. REVUE D ELEVAGE ET DE Med. VETERINAIRE DES. PAYS TROPICAUX. 49, 285–288 (1996).
Elzein, E. A., Al-Afaleq, A., Mellor, P., El-Bashir, A. & Hassanein, M. Study of Akabane infection in Saudi Arabia by the use of Sentinel ruminants. J. Comp. Pathol. 119, 473–478 (1998).
Jun, Q. et al. A serological survey of Akabane virus infection in cattle and sheep in Northwest China. Trop. Anim. Health Prod. 44, 1817–1820 (2012).
Brenner, J., Tsuda, T., Yadin, H. & Kato, T. Serological evidence of Akabane virus infection in Northern Israel in 2001. J. Vet. Med. Sci. 66, 441–443 (2004).
Karaoğlu, T., Özgünlük, İ., Demir, B., Oezkul, A. & Burgu, I. Seroprevalence of culicoides-borne disease in cattle in European Turkey. Ankara Üniversitesi Veteriner Fakültesi Dergisi. 54, 121–125 (2007).
Selim, A. et al. Bluetongue virus infection in cattle: serosurvey and its associated risk factors. Trop. Anim. Health Prod. 55, 285 (2023).
Selim, A., Alsubki, R. A., Albohairy, F. M., Attia, K. A. & Kimiko, I. A survey of bluetongue infection in one-humped camels (Camelus Dromedarius); seroprevalence and risk factors analysis. BMC Vet. Res. 18, 322 (2022).
Selim, A., Marzok, M., Gattan, H. S. & Hereba, A. M. Factors influencing Akabane virus Seroprevalence in sheep and goats in Egypt’s nile Delta. Trop. Anim. Health Prod. 57, 1–7 (2025).
Socha, W., Kwasnik, M., Larska, M., Rola, J. & Rozek, W. Vector-borne viral diseases as a current threat for human and animal health—One health perspective. J. Clin. Med. 11, 3026 (2022).
Selim, A. & Abdelhady, A. Neosporosis among Egyptian camels and its associated risk factors. Trop. Anim. Health Prod. 52, 3381–3385 (2020).
Selim, A., Abdelhady, A. & Alahadeb, J. Prevalence and first molecular characterization of Ehrlichia canis in Egyptian dogs. Pak Vet. J. 41, 117–121 (2020).
Selim, A. M., Elhaig, M. M. & Gaede, W. Development of multiplex real-time PCR assay for the detection of Brucella spp., Leptospira spp. and Campylobacter foetus. (2014).
Selim, A., Yang, E., Rousset, E., Thiéry, R. & Sidi-Boumedine, K. Characterization of Coxiella burnetii strains from ruminants in a galleria Mellonella host-based model. New. Microbes New. Infections. 24, 8–13 (2018).
Karami Boldaji, S., Pourmahdi Borujeni, M. & Haji Hajikolaei, M. R. Seifi Abad Shapouri, M. R. Seroprevalence and risk factors of Akabane virus infection in cattle from Khouzestan Province of Iran. Iran. J. Virol. 10, 14–20 (2016).
Ahi, M. R. & Hajikolaei, H. Seifi Abad Shapouri, M. R. A serological survey on antibodies against Akabane virus in sheep in Southwest of Iran. Iran. J. Virol. 9, 20–25 (2015).
Radostits, O. M., Gay, C. C., Hinchcliff, K. W. & Constable, P. D. Veterinary Medicine E-Book: Veterinary Medicine E-Book (Elsevier Health Sciences, 2006).
Wittmann, E. & Baylis, M. Climate change: effects on Culicoides-transmitted viruses and implications for the UK. Vet. J. 160, 107–117 (2000).
Sprygin, A., Fedorova, O., Babin, Y. Y., Kononov, A. & Karaulov, A. Blood-sucking midges from the genus Culicoides (Diptera: Ceratopogonidae) act as filed vectors of human and animal diseases. Сельскохозяйственная Биология, 183–197 (2015).
Selim, A., Megahed, A., Kandeel, S., Alouffi, A. & Almutairi, M. M. West nile virus Seroprevalence and associated risk factors among horses in Egypt. Sci. Rep. 11, 20932 (2021).
Baz, M. M. et al. Novel pesticidal efficacy of Araucaria heterophylla and Commiphora molmol extracts against camel and cattle blood-sucking ectoparasites. Plants 11, 1682 (2022).
Christensen, C. External parasites of dairy cattle. J. Dairy Sci. 65, 2189–2193 (1982).
Selim, A. & Khater, H. Seroprevalence and risk factors associated with equine piroplasmosis in North Egypt. Comp. Immunol. Microbiol. Infect. Dis. 73, 101549 (2020).
Selim, A., Weir, W. & Khater, H. Prevalence and risk factors associated with tropical theileriosis in Egyptian dairy cattle. Veterinary World. 15, 919 (2022).
Selim, A. et al. Seroprevalence and risk factors associated with canine leishmaniasis in Egypt. Veterinary Sci. 8, 236 (2021).
Abdi, A., Roushdy, S., Specialist, S., Beillard, M. & Agricultural, S. Egypt livestock and products annual 2019 Egyptian beef prices stable, US beef imports challenged. USDA Foreign Agricultural Service (2020).
Mekonnen, T., Tadesse, Y. & Meseret, S. Genetic improvement strategy of Indigenous cattle breeds: effect of cattle crossbreeding program in production performances. J. Appl. Life Sci. Int. 23, 23–40 (2020).
Owada, K., Mahony, T. J., Ambrose, R. K., Hayes, B. J. & Soares Magalhães, R. J. Epidemiological Risk Factors and Modelling Approaches for Risk Assessment of Lumpy Skin Disease Virus Introduction and Spread: Methodological Review and Implications for Risk-Based Surveillance in Australia. Transboundary and Emerging Diseases 3090226 (2024). (2024).
Selim, A., Manaa, E. & Khater, H. Seroprevalence and risk factors for lumpy skin disease in cattle in Northern Egypt. Trop. Anim. Health Prod. 53, 350 (2021).
Baz, M. M., Selim, A. M., Radwan, I. T. & Khater, H. F. Plant oils in the fight against the West nile vector, Culex pipiens. Int. J. Trop. Insect Sci. 42, 2373–2380 (2022).
Selim, A., Manaa, E. A., Waheed, R. M. & Alanazi, A. D. Seroprevalence, associated risk factors analysis and first molecular characterization of chlamydia abortus among Egyptian sheep. Comp. Immunol. Microbiol. Infect. Dis. 74, 101600 (2021).
Abd Elmohsen, M., Selim, A. & Abd Elmoneim, A. E. Prevalence and molecular characterization of lumpy skin disease in cattle during period 2016–2017. Benha Veterinary Med. J. 37, 172–175 (2019).
Hamdy, A. S., Selim, A., Shoulah, S. A. & Ibrahim, A. M. M. Sero-surveillance infectious bovine rhinotracheitis in ruminants and assessment the associated risk factors. Benha Veterinary Med. J. 42, 160–163 (2022).
Selim, A. & Radwan, A. Seroprevalence and molecular characterization of West nile virus in Egypt. Comp. Immunol. Microbiol. Infect. Dis. 71, 101473 (2020).
Selim, A. & Abdelhady, A. The first detection of anti-West nile virus antibody in domestic ruminants in Egypt. Trop. Anim. Health Prod. 52, 3147–3151 (2020).
Mathew, C. M. Infections associated with reproductive disorders in cattle in Tanzania: Occurrence, characterization and impact. (2017).
Soldan, S. S. & González-Scarano, F. Emerging infectious diseases: the Bunyaviridae. J. Neurovirol. 11, 412–423 (2005).
Kaplan, M., Çağırgan, A. A., Pekmez, K., Arslan, F. & Kalaycı, G. Survey of four important viruses causing abortion in cattle in Aegean Region. (2022).
Acknowledgements
The authors would like to acknowledge the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia (Grant No. KFU251908).
Funding
This work was supported by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia (Grant No. KFU251908).
Author information
Authors and Affiliations
Contributions
Conceptualization, methodology, formal analysis, investigation, resources, data curation, writing-original draft preparation, A.S., H.S.G., A.M.H. and M.M.; writing-review and editing, A.S., H.S.G., A.M.H. and M.M.; project administration, M.M.; funding acquisition, A.S., H.S.G., A.M.H. and M.M. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Selim, A., Marzok, M., Gattan, H.S. et al. A serological study and risk factors associated with Akabane virus infection in cattle in Egypt. Sci Rep 15, 20245 (2025). https://doi.org/10.1038/s41598-025-02732-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-02732-2