Abstract
In this study, cellulose was extracted from Populus nigra plant biomass and subsequently modified for use as an adsorbent for the removal of pesticide acetamiprid from wastewater. The extracted cellulose was purified using sodium oxychloride and hydrogen peroxide, acetylated with acetic acid, and further functionalized with polyvinyl alcohol. The prepared adsorbent was characterized via FTIR, UV-Vis spectroscopy, EDX, SEM, and XRD. The effects of various parameters, including the initial concentration of pesticide, adsorbent dosage, contact time, pH, and temperature on adsorption were examined using batch adsorption approaches. Maximum removal was obtained at 80 mg/L pesticide concentration, pH 8, 60 min contact time, and 0.08 g adsorbent dosage. The isothermal data for acetamiprid is matched perfectly to the Freundlich, Temkin and Harkins-Jura isotherm models with regression constant values approaching to 1, whereas good agreement was observed for kinetic data with the pseudo-second-order model. Thermodynamic analysis indicated the adsorption process as favorable and exothermic, with ∆H° = -81.65 kJ/mol, ∆S° = 305.62 J/mol·K, and ∆G° = − 116.90, -150.17 and 180.20 kJ/mol at 293, 313 and 333 K respectively. These findings suggest that the prepared cellulose-based adsorbent is an effective and promising material for pesticide removal from wastewater.
Similar content being viewed by others
Introduction
Environmental pollution problem is a burning issue these days especially the poisoning of water supplies by pesticides from agricultural practices. At the same time, due to the enormous pressure of population growth, globalization, and the development of social civilization, pesticides have become essential tools for the long-term sustainable production of large quantities of agricultural food. So, it is not possible to ban the use of pesticides, instead the reclamation of pesticide polluted water seems to be a better solution in such circumstances. These pollutants are often present in trace amounts in surface water and occasionally in groundwater, which is the major source of world drinking water. The authorities, the public, and everyone involved in the supply of drinking water are extremely concerned about the frequent discovery of various pesticide residues in natural water resources1.
It is preferable to prevent pollution of water resources rather than treat contaminated aquifers. From this point of view, it is becoming increasingly important to develop environmentally friendly remediation techniques that do not contaminate water during filling and cleaning and prevent water leakage on land. Adsorption has been proven to be more attractive to remove various organic pollutants from environmental matrices than other physical, biological, and chemical technologies, such as precipitation and coagulation2, membrane filtration3,4, and photochemical degradation5, because of its simplicity and ease of use.
Contamination of persistent pesticides in water resources may badly hurt ecosystems and public health. Some insecticides contain substances that change hormones structures. Developed countries now have very strict regulations for water treatment, about pesticide chemicals (levels of 0.1 mgL− 1)6. Adsorption, photocatalytic degradation7,8, electrochemical degradation9, oxidation10,11, membrane filtration, nanofiltration12, etc. are methods commonly used to eliminate pesticides from effluents.
Adsorption has an advantage over other techniques due to its simple design, water-free environment, and potential for low initial and land investment costs13. Although it is expensive to produce activated carbon (AC) but is a very effective adsorbent for the elimination of pollutants. A variety of cheap adsorbents have been developed, including clay, industrial waste, and by-products, as well as agricultural products and by-products.
In this study, modified cellulose obtained from Populus nigra biomass and was used as an efficient adsorbent to remove acetamiprid from wastewater. The approach used here is economic and efficient as cellulose has high surface reactivity and is a low-cost material as obtained from a non-food plant (Populus nigra) and is abundantly available biomass material for production of efficient biosorbent. Compared to conventional adsorbents, the modified cellulose is environment friendly and a cost effective solution with strong reusability and structural stability14. The advantage of the study includes eco-friendly processes, inexpensive raw material, and maximum pesticide removal efficiency. The study also has some disadvantages such as limitation in regeneration cycles and scaling challenges for industrial use. The study presents an economically viable and reliable approach for treating wastewater specially in agriculture sector effected by pesticide contamination. Also, to the best of our knowledge such modified cellulose has not been utilized by other researchers for selected pesticide.
Methods and material
Most of the chemicals utilized for this research were of analytical grade and no purification was required while performing chemical reactions. Distilled water, methanol, chloroform, and ethanol were used as a solvent. The glassware used in this study were round bottom flasks, volumetric flasks, beakers, funnels, graduate cylinders, etc.
The chemicals used were polyvinyl alcohol (Sigma Aldrich), Ammonium oxalate (Sigma Aldrich), Diphenyl ammine (Sigma Aldrich), Acetyl chloride (Sigma Aldrich), Sulphuric acid (98% purity, Sodium hydroxide, hydrogen peroxide (37%) and hydrochloric acid (37%).
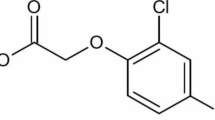
The acetamiprid insecticide is removed in this study from model wastewater. It is an organic compound having 99% purity, chemical formula (C10H11ClN4), molecular weight 222.678 gmol− 1, with maximum UV absorbance (Lambda max = 250 nm) and was obtained from the local industry of PubChem (Peshawar, Pakistan) while further purification process was not applied. The structure formula is shown in Fig. 1.
The instruments used were a double beam Spectrophotometer (Perkin Elmer Lambda 25 Double Beam), digital balance (Shimadzu AY220), hot plate (IKA C-MAG HS 7), thermometer (Mercury Thermometer/Digital Probe), furnace (LabTech LMF-3), oven (Memmert UN110), etc.
Chemical structure of acetamiprid.
Extraction of cellulose
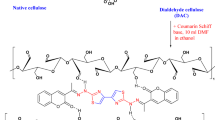
Wood biomass was collected from the local market to extract cellulose using a documented procedure14. About 30 g powdered Populus nigra wood was collected and cleaned thoroughly with hot distilled water (50 °C) and dried for 24 h in an oven. Using a solution of 300 mL of n-Hexane and 150 mL methanol solution, the dried wood was placed in a beaker (500 mL) and shaken for 6 h. Subsequently, the solid wood biomass was filtered, weighed, and washed multiple times with worm distilled water. The solid wood biomass was then transferred to another beaker along with a solution containing 5% each KOH, NaOH, Na2CO3, and ammonia (33%) in an aqueous solution, then stirred at 80 °C for 5 h. Neutralization of the medium was achieved through the addition of H2SO4 (10%) solution in a 250 mL flask, followed by the introduction of H2O2 solution (2%) for bleaching purposes, and the mixture was stirred for an hour. The mixture was finally passed from Whatman filter paper and washed multiple times with distilled water to attain neutrality. The solid biomass containing cellulose was then dried in an oven at 50 °C for 24 h. The solid biomass containing cellulose was further subjected for incorporation of organic functional groups such as polyvinyl alcohol15.
Acetylation of cellulose
Following the methods described in the reported literature, an acetyl group was added to cellulose15. About 10 g biomass-based cellulose was added in a beaker (500 mL) and to this solution, glacial acetic acid (50 mL) was introduced. The mixture was stirred up to 1 h at an ambient temperature (25 °C). Another solution was prepared from acetyl chloride (30 mL) and six drops of Sulphuric acid (98%) was added and the mixture was stirred at 0 °C using an ice bath. This solution was then mixed with cellulose mixture and shaken for three hours at 25 °C. While stirring, 50 mL of 60% glacial acetic acid was gradually added dropwise. Finally, the mixture was filtered, and the modified cellulose was weighed and several times washed with worm distilled water (50 °C) until neutrality was achieved. Finally, cellulose acetate was oven-dried for 24 h and for further application, it was collected in vials.
Incorporation of polyvinyl alcohol to cellulose acetate
To improve the adsorption efficiency of the adsorbent, polyvinyl alcohol was dispersed upon the cellulose acetate structure. Using distilled water, about 1 g cellulose acetate was dissolved and shaken for 10 min at room temperature. Separately, 0.5 g of PVA (polyvinyl alcohol) was taken in another flask and shaken for the same duration. Subsequently, the solutions were mixed in a flask at room temperature and stirred for 2 h. Finally, the mixture was filtered using Whatman filter paper and the solid biomass was thoroughly washed using hot distilled water (50 °C). The polymer-based solid adsorbent was oven dried at 50 °C and collected in vials. The dried adsorbent cellulose acetate polyvinyl alcohol was then subjected to the elimination of pesticide (acetamiprid) from wastewater.
Material characterization
For elemental composition analysis of polyvinyl alcohol incorporated cellulose acetate, EDX-sight (Energy dispersive X-ray) analyzer (High Wycombe, Oxford apparatus, UK) was used. Similarly, to observe the surface morphology of the prepared adsorbent, SEM (scanning electron microscope, JEOL Japan-JSM 5910) was used at 10 KV fixed voltage.
Furthermore, FTIR (Perkin Elmer, Cary 630, Waltham, MA, USA) analysis was performed for functional group determination of PBCA–PVA and to confirm the chemical modification of cellulose acetate.
To determine the internal structure of powdered PBCA–PVA, an X-ray diffraction machine; JOEL JDX-3532, having a Ni filter with Cu Kα radiation have a wavelength of 1.5418, a voltage of 40 kV, and a current of 30 mA was used.
Adsorption experiments
A stock solution of acetamiprid 500 mgL− 1 was prepared in a volumetric flask using double distilled water. Various standard solutions were prepared through serial dilutions. For the adsorption of acetamiprid several batch experiments were performed. Various acetamiprid concentrations were used at specific pH, temperature, and contact time. pH of the solution was adjusted using 0.1 M each HCl and NaOH solutions. In each case, a temperature-controlled shaker was used for 1 h at 170 rpm.
Various optimization studies were performed using initial pesticide concentration (10–120 mg/L), the adsorbent dosage of (0.01–0.12 g), contact time (10–120 min), and pH (2–12) to get maximum adsorption. All experiments were carried out in triplicate and the mean values are presented. The remaining concentrations of selected pesticide in solution was determined after passing over a Whatman filter paper then at 243 nm the absorbances were recorded using UV-visible spectrophotometer. Percent removal (%R) and amount of pesticide adsorbed at given time (qt in mg/g) were calculated using the following Equations16,17
The Ci and Cf are the start and end concentrations of acetamiprid, respectively; V (L) is the acetamiprid solution volume whereas m denotes the adsorbents mass in grams.
Isothermal study
To study the effect of concentration of PBCA–PVA on the adsorption of acetamiprid, different initial concentrations of pesticide were prepared (10–100 mgL− 1), while other conditions for instance 50 mL volume of solution, optimum time of contact, pH, and adsorbent dose of 0.08 g were kept constant. Using Eq. 1, their \(\:{q}_{e}\) value was calculated and plotted against equilibrium concentration. Various isotherm models for instance Freundlich, Langmuir, Jovanovic, Temkin, and Harkins-Jura were utilized to analyze the equilibrium adsorption data.
Kinetic study
To study the kinetics of acetamiprid adsorption on the prepared adsorbent, 0.08 + g of PBCA–PVA adsorbent was added to an acetamiprid solutions (50 mL) having fixed concentration. The mixture was shaken at 170 rpm for 100 min. Various kinetic models, including the pseudo-second order, pseudo-first-order, Natarajan and Khalaf, power function, and intra-particle model, were used to analyze the kinetics data of acetamiprid adsorption.
pH and adsorbent dosage effect
Different pH solutions ranging from 2 to 12 were contacted with a fixed concentration pesticide solution in 50 mL volume and shaken for an interval of time equal to equilibrium. For pH adjustment, 0.1 M each HCl and NaOH solutions were used. To study the effect of adsorbent dosage, the amount of adsorbent ranging from 0.01 to 0.08 g was contacted with fixed concentration solution and fixed time. Other experimental conditions were kept constant.
Temperature effect on adsorption of selected pesticide
In a 100 mL flask about 50 mL acetamiprid solution was stirred for 1 h with 0.08 g adsorbent at different temperatures ranging from 298 K to 333 K while other conditions were kept constant. To determine the adsorbed quantity of Acetamiprid Eq. 1 was used18.
Results and discussion
Characterization of the prepared adsorbent
The elemental analyses of plant based cellulose acetate and PBCA–PVA particles are shown in Fig. 2a and b respectively. The EDX spectra of PBCA–PVA depicts a characteristically high peak of carbon, while a second high peak of oxygen can also be observed. The EDX spectrum of plant based cellulose acetate also have minor peaks of sodium and chlorine which shows impurity during acetylation process.
The deposition of polyvinyl alcohol and acetate ion over the cellulose surface gives rise higher peak of carbon compared to cellulose alone. The Energy Dispersive X-ray spectra indicates that the polyvinyl alcohol and acetate group are incorporated successfully to the structure of cellulose polymer. The high peak of oxygen in the spectra indicates that the acetylation process was successful15,19.
(a) EDX spectra of plant based cellulose acetate (PBCA) sample. (b) EDX spectra of PBCA–PVA particles.
Characterization of PBCA–PVA by SEM analysis
The SEM micrograph of cellulose acetate (CA) is shown in Fig. 3a reveals the morphology of homogeneous cylindrical CA powder that contains porous holes on its surface. These porous structures resulted from the acetylation of commercial cellulose using acetic acid, which led to significant changes in the surface morphology. The morphology shown by CA in this study is somewhat different from the data obtained by Bahmind20, that is, fiber-shaped cylindrical cellulose acetate equally distributed small holes on its surface. Such variations in pore distribution may arise from differences in the acetylation process but in shape it shows similarity to the result of Dian21 and Bahmind22. However, the size and shape of the CA obtained in this study are different from the results of Dian and Bahmind because the raw material were from different sources. The presence of white regions on the CA surface may indicate residual impurities left during acetylation.
The SEM images of PBCA–PVA, presented in Fig. 3b and c which indicates random particle size, with distorted and uneven edges. The PBCA–PVA particle has high surface area and porous structure. The white dispersed spots observed within the pores suggest the successful incorporation of polyvinyl alcohol. The deposition of acetate ion and polyvinyl alcohol could be inferred from the filled pores on the PBCA–PVA surface in the SEM micrographs.
SEM images of Populus nigra powder (a), PBCA–PVA adsorbent at 430 (b) and (c) 600 X magnifications.
Characterization of Populus nigra wood powder by FTIR analysis
Figure 4 represents the FTIR spectra of Populus nigra plant. the main constituents present in Populus nigra powder include cellulose, hemicellulose, and lignin23. The distinctive IR peak representing lignin appears within the range of 1200–1300 cm− 1, while the characteristic peak for hemicelluloses at 1730–1830 cm− 1 is not observed in the spectra24, suggests that lignin and hemicellulose are entirely removed through the processes of delignification and bleaching. The primary peak at 3332 cm− 1 signifies the presence of a hydroxyl group, while other significant peaks include 2896 cm− 1 for C–H stretching in cellulose and 1423 cm− 1 attributes for C–H and C–O groups bending vibrations in the structure of cellulose polymer25. The C=O and C–H groups stretching frequency in cellulose structure was noted from peak at 1028 cm− 1. Furthermore, a peak at 894 cm− 1 is observed, suggesting the presence of β-glycosidic bond26. Since these absorption peaks correspond to cellulose’s distinct bands in the spectra, it verifies that the cellulose structure remains intact throughout the pre-treatment and acid hydrolysis processes27.
FTIR spectra of Populus nigra plant.
Characterization of plant based cellulose acetate PVA after adsorption by FTIR analysis
The prepared adsorbent was applied to eliminate acetamiprid (insecticide) from aqueous media. The spectra indicates that acetamiprid is adsorbed on the adsorbent surface via chemical interaction. Two peaks at the positions of 2892 and 3328 cm− 1 attributes to the formation of the H-bond between the acetate group of cellulose and pesticide28. The broad peak at 3328 cm− 1 position indicates the interaction of pesticide and cellulose groups. Figure 5, indicates slight blue shifts and a reduction of broadband for the OH group indicating H-bond formation29. This indicates that H-bond forms between pesticide and cellulose groups.
Furthermore, the peak of methylene moiety in 2896 cm− 1 shifts at 2894 cm− 1shows hydrophobic interaction accompanied by electrostatic interaction between the pesticide and adsorbent30. Hence the adsorption mechanism of adsorbent for acetamiprid is hydrogen bonding, electrostatic attraction, and electron transfer in this process28.
FTIR Spectra of PBCA–PVA after adsorbtion.
Characterization of PBCA–PVA by (XRD) analysis
XRD analysis was used to understand the crystalline structure and particle size of the adsorbent. Figure 6a and b shows an X-ray Diffraction plot of the Populus nigra powder and plant-based cellulose-PVA sample respectively. The XRD spectra of cellulose acetate (CA) are presented in Fig. 6a. The XRD pattern of CA indicates typical peaks of cellulose at 2θ = 15° and 20°. The XRD spectra of cotton having 0% H2SO4 concentration is like cellulose because the main constituent of cotton is cellulose. However, it indicates a broad peak and a low intensity than the spectra of cotton (hydrolyzed). Cellular crystallinity is influenced by hydrolysis of H2SO4. The highest crystallinity was achieved at 30% H2SO4 where the 2θ was 15° and 20°. The XRD pattern of CA shows 2 peaks at 2θ = 20° and 22° which are common peaks for cellulose. Furthermore, the intensity of CA XRD peaks was higher relative to that of cellulose. This influence is more likely due to the incorporation of the acetyl group into the cellulose structure31.
The XRD patterns obtained for PBCA–PVA are shown in Fig. 6b. XRD diffractogram indicates a cellulose polymer pattern showing peaks at 2θ = 22.2° and 20.2° are attributed to (200) and (110) planes of the crystal respectively32. The X-ray diffraction results for the synthesized adsorbent indicate that the primary powdered adsorbent structure is made up of OH phase, aligning with the results obtained from scanning electron microscopy (SEM) and energy-dispersive X-ray spectroscopy (EDX) analyses (Figs. 2 and 3). Due to the pasting of cellulose, distinct XRD peaks are not identifiable in the powdered adsorbent.
X-ray Diffraction pattern for Populus nigra powder (a) and plant based cellulose acetate-PVA particle (b).
Isothermal studies
Different pesticide concentrations ranging from (10 to 100 mg/L) were used in this study to analyze adsorption phenomena onto the prepared PBCA–PVA adsorbent (Fig. 7a). it was observed that adsorption increases as the concentration rises. Several isotherms were used to observe adsorption patterns such as Freundlich, Langmuir, Temkin, Jovanovich, and Harkins Jura33.
Langmuir isotherm
The main assumption of this model explains that, at definite sites, adsorption takes place and is linearly distributed thoroughly at the surface of the adsorbent. The linear form of this model is shown by the given mathematical expression as34:
where \(\:{q}_{e}\)denotes the equilibrium adsorbed amount of acetamiprid in mg/g, while \(\:{C}_{e\:}\) indicates the equilibrium concentration in mg/L, \(\:{q}_{m}\) shows high adsorption capacity of PBCA–PVA for pesticide whereas constant \(\:KL\) shows the energy of sorption process. A diagram of \(\:Ce/qe\) versus \(\:Ce\) drawn for PBCA–PVA is shown in Fig. 7b. The \(\:q\text{m}\) and \(\:KL\) values were measured from the intercept and slope of the diagram and the corresponding values are listed in Table 1. The \(\:qm\) value indicated a moderate adsorption capacity, while the high value of the Langmuir constant L = 4.91indicates a strong affinity of the adsorbent for acetamiprid molecule. This is the best result showing effective binding interaction i.e. Hydrogen bonding and dipole-dipole interactions. The R2 value 0.8488 is not very close to 1, indicating that the Langmuir model is poorly fitted for the data not supporting the monolayer adsorption on a homogeneous surface. These information about the data strengthen the reliability of the \(\:K\text{L}\) and \(\:q\text{m}\) values.
Freundlich isotherm
This isotherm model explains adsorption onto heterogenous surface where active site have different energy offering for multilayer adsorption. The mathematical equation below represents the Freundlich isotherm35 as:
In the above equation \(\:\text{K}\text{F}\) shows Freundlich constant indicates adsorption potential, whereas n shows the empirical constant indicates the strength of adsorption. The plot is drawn between \(\:\text{l}\text{n}\text{C}\text{e}\) versus \(\:\text{l}\text{n}\text{q}\text{e}\) as presented in Fig. 7c. The value of the constant 1/ is greater than 1, indicating cooperative adsorption, whereas value less than 1 indicates favorable adsorption. The \(\:\text{K}\text{F}\) value 3.75 (mg/g) (L/mg) indicates moderate adsorption capacity. The correlation coefficient R2 = 0.993838 indicates a significantly better fit compared to the Langmuir model (R2 = 0.8488). This suggests that the Freundlich model provides the best fit for the adsorption data and confirmed the heterogeneity of the surface. The values of these constants are given in Table 1.
Selected pesticide adsorption plots: (a) Concentration Effect on adsorption ofPBCA–PVA (b) plot of Langmuir isotherm (c) Diagram of Freundlich isotherm (d)Diagram of Temkin isotherm (e) Jovanovich isotherm (f) Diagram ofHarkins–Jura isotherm.
Temkin isotherm model
This model suggests that as adsorbate molecules bind to an adsorbent surface, the surface coverage is tied with the free energy of the process. This isotherm is shown in mathematical form in the following expression as36:
In the above equation β = RT/b, where R is ideal gas constant have a value of 8.314 J/mol K, whereas T represents absolute temperature in Kelvin, while b is heating constant for adsorption. Plotting \(\:{q}_{e}\) versus\(\:\:\text{ln}Ce\), for acetamiprid adsorption on PBCA–PVA, the slope and intercept could be used to calculate values of the constants as presented in Fig. 7d and the respective values of constants are listed in Table 1. The relatively high value of binding constant (2.30 L/mg) shows strong affinity of adsorbent and acetamiprid molecule which shows the favorability of the adsorption process. The B value (37.97 J/mol) in this range typically shows physisorption with moderate interaction strength. For such interactions 20–80 J/mol is most common values. The correlation coefficient (R2 = 0.9996) is very close to 1, indicating an excellent fit for the adsorption data.
Jovanovic isotherm model
This model has the same assumption as stated by Langmuir’s model of isotherm, with further elaboration that when there is adsorption there are also mechanical interactions. The mathematical equation of this isotherm is given below16:
In the above equation \(\:{C}_{e}\) shows the equilibrium concentration of adsorbate (mg/L), while \(\:{q}_{e}\:\) shows amount adsorbed at equilibrium (mg/g), \(\:{q}_{max}\) represents the adsorption efficiency of the adsorbent. The Jovanovic constant is represented by KJ which is related to the energy of acetamiprid (pesticide) adsorption. Plotting \(\:ln{q}_{e}\) versus \(\:{C}_{e}\) shows the linearity of Jovanovic isotherm. The value of\(\:\:{q}_{max}\) and \(\:KJ\) were estimated from the slope and intercept of the diagram (Fig. 7e) and are given in Table 1. The obtained \(\:{q}_{max}\) value of 0.7835 mg/g represents the maximum monolayer adsorption capacity of PBCA–PVA for acetamiprid removal, indicating a moderate adsorption capacity. The consistency of this value across isotherm models suggests stable adsorbent behavior. The Jovanovic constant (KJ = 0.465) reflects the bonding strength between the adsorbent and acetamiprid. This value, sometimes is expressed as L/mg or unitless depending on the model derivation, indicates moderate interaction energy, stronger than typical physisorption but weaker than chemisorption, suggesting specific binding via hydrogen bonding or dipole interactions involving PVA or cellulose acetate functional groups. The correlation coefficient (R2 = 0.8594) indicates a moderate fit to the Jovanovic model, implying that assumptions such as a homogeneous surface, monolayer coverage, and minimal mechanical interaction may not be fully valid.
Harkins Jura adsorption isotherm
This model explains that adsorption occurs on heterogenous porous surface where multilayer adsorption of sorbate takes place. Equation 8 explains the isotherm model as37.
In the above equation, A and B represent Harkins–Jura constants. Table 1 shows the values of various constants. By plotting \(\:\frac{1}{{\text{q}}_{\text{e}2}}\) against \(\:\text{l}\text{o}\text{g}{\text{C}}_{\text{e}\:\:}\), the linearity of the isotherm was determined as depicted in Fig. 7f, and Table 1 contains the values of these constants. The value of AH=0.8729 is associated with surface coverage and adsorption capacity. The value less than 1 indicates moderate adsorption intensity, related to physical or multilayer adsorption process.
The value of BH = 0.04758 indicates surface heterogeneity. Their lower value (near to zero) indicates little variation in adsorption energy of the surface i.e., the surface is uniform fairly and not homogenous completely. It indicates that the adsorbent has high distributed binding sites, although not same in terms of energy. The high value of R2 = 0.994 indicates that this model is best fit for experimental data which is much better than Temkin and Jovanovic models.
Considering R2 (regression-coefficient) values for the used models, it is reported that the best model that fits the experimental data are the Freundlich, Temkin and Harkins Jura isotherm models having a greater R2 value near to one38.
Kinetic study
To understand the adsorption mechanism of acetamiprid on the surface of prepared adsorbent, the kinetic study was performed by taking 60 mg/L pesticide concentration. Equilibrium adsorption time determined was 60 min. Above 60 min time the process proceed with steady rate as shown in Fig. 8a. The observation can be explained as follows; At the start of the reaction the adsorption sites on the adsorbent surface were free for pesticide adsorption and acetamiprid pesticide interacted freely with groups attached to the adsorbent surface, that why a sharp increase in transfer of pesticide molecules to adsorbent was noted39 continued up to 60 min. In subsequent time greater number of pesticides were gathered on the surface of PBCA–PVA adsorbent therefore slowing down the adsorption process40. Finally, beyond 120 min, the rate of adsorption becomes constant41.
Several kinetic models, such as Lagergren pseudo-first-order, Intra-particle diffusion, pseudo-second-order, Natarajan and Khalaf models, and power functions were applied to determine various kinetic parameters of adsorption.
Kinetic model of pseudo-first order
The expression of pseudo-first-order in the linear form is given below42,43:
In the above equation \(\:\text{q}\text{e}\) is the quantity (mgg− 1), of pesticide adsorbed at equilibrium, \(\:\text{q}\text{t}\) showing acetamiprid amount adsorbed at instant t where \(\:\text{K}1\) is the pseudo-first-order rate constant. Plotting time (t) against \(\:\text{l}\text{n}(\text{q}\text{e}-\:\text{q}\text{t})\), the value of constant \(\:\text{K}1\) and qe may be calculated from the slope and intercept of the diagram, as depicted in Fig. 8b and the values of constants are given below in the Table 2. The high value of qe =124.7 mg/g indicates theoretical equilibrium adsorption capacity for the model. If the theoretical values are different from the experimental value, then this indicates poor model fit. The moderate rate constant value k1=0.1019 min− 1 indicates reasonable adsorption rate. However, their interpretation depends on comparison with other models (such as Pseudo-second order model). The R2 = 0.7618 which is poor fit, this suggests strongly that the pseudo-first order model is poorly fit for the experimental data of adsorption of acetamiprid on PBCA–PVA.
Adsorption kinetics of selected pesticide adsorption on PBCA–PVA (a) Kinetics plot (b) Pseudo-first order plot (c) Pseudo-second order plot (d) Power function plot (e) Khalaf and Natarajan plot (f) Intra-particle model.
Pseudo-second order model
The mathematical form of this model in its linear form can be given as42,43:
In the given equation, K2 represents pseudo-second order rate constant. The plot of \(\:\frac{t}{qt\:}\) against t is given in Fig. 8c, the constant K2 value was obtained from slope and is listed in Table 2. The qe = 9.69 mg/g represents the calculated equilibrium adsorption capacity estimated from the model. Although lower than the predicted value of the pseudo-first-order model (124.7 mg/g), it is much closer to the experimental value, indicating that this model provides a more realistic representation of the adsorption process. The high-rate constant K2 = 19.16 g/mg suggests that the PBCA–PVA composite rapidly adsorbs acetamiprid, reflecting fast interaction kinetics and the presence of efficient binding sites. The high correlation coefficient (R2 = 0.994) demonstrates an excellent fit, confirming that the adsorption kinetics are well described by the pseudo-second-order model.
Power function model
Equation of power function in the linear form is given below44:
In the above equation, a and b indicates reaction constant and its values were measured from the intercept and slope of diagram plotted between \(\:log{q}_{t}\) and \(\:log\text{t}\) as shown in Fig. 8d. This model explains early-stage adsorption behavior. It does not explain a particular mechanism, such as physisorption or chemisorption, but it generally provides an explanation of how adsorption rate changes over time. The high value of initial adsorption rate, a = 4.48 shows that the acetamiprid is quickly adsorbed at the start of the process. It matches to the high value of rate constant of pseudo-second order model. The constant b = 0.335 value shows a decreasing adsorption process, which indicates fast initial binding followed by slow attachment as the active sites are occupied with the passage of time.
Natarajan and Khalaf kinetic model
Natarajan and Khalaf model in the mathematical form can be presented as45:
In this equation, \(\:Co\) (mgL− 1) represents initial concentration at t = 0, \(\:Ct\) (mg/L) indicates concentration at any time t. The Fig. 8e presents a plot of \(\:logCo/\:Ct\) and t, provides the constant \(\:KN\) value for comparison. The value of K = 0.070 min− 1indicates moderate removal of acetamiprid from aqueous media. This indicates that the adsorption is active but not too rapid, owing to surface heterogeneity, competition between surface and internal bending sites and diffusion limitation. R2 = 0.9721 value indicates that this model is best fit for experimental data.
Intraparticle diffusion model
Intra-particle diffusion model in the mathematical form can be expressed as46,47:
Here, \(\:{q}_{t}\) represents the amount of pesticide adsorbed at any time on the adsorbent surface. The value of \(\:{K}_{diff}\) (slope) and \(\:C\) (intercept) were measured from \(\:{q}_{t}\) versus t1/2 plot as shown in Fig. 8f. High R2 = 0.9921 value indicates strong fit of the experimental data, means that intra particle diffusion play a pivotal rule in the adsorption process, however, alone the high R2 value does not confirm that the intra particle diffusion is the only rate determining step. The value of C can also be interpreted. The non-zero value of C indicates that intra particle diffusion is not only the rate determining step. The high value of C = 5.09 indicates greater contribution of surface adsorption. The high value of C indicates both intraparticle diffusion and surface adsorption takes place simultaneously.
Considering various R2 values of the applied models, it is suggested that the pseudo-second order adsorption model is the most relevant model fitted for experimental data with a regression constant value close to 1.
Effect of pH
The adsorption pattern under the influence of varying pH is represented in Fig. 9a. At neutral pH high adsorption of pesticide was observed. Highest adsorption of acetamiprid by PBCA–PVA adsorbent was achieved at 8 pH. The surface is neutral under acidic pH, while at neutral pH or somewhat basic pH, it is in ionized form which is more soluble in water, and pesticide shows greater interaction with surface groups of PBCA–PVA that leads to maximum adsorption rates48.
Point zero charge
The surface charge behavior of PBCA–PVA, determined using the mass titration method, is presented in Fig. 9b. Various amounts of PBCA–PVA were mixed with distilled water and left to equilibrate for 24 h. To study the effect of pH, 10 mL of the PBCA–PVA suspension was transferred into ten separate reagent bottles, with the pH adjusted to values ranging from 2 to 12 using 0.1 M HCl or 0.1 M NaOH. Each bottle was then supplemented with 0.1 g of acetamiprid and shaken in a thermostatic water bath at 25 °C for 24 h. After equilibrium, the pH of each solution was measured, and a graph of ∆pH versus adsorbent mass (%) was plotted. The point of zero charge (PZC) of PBCA–PVA was identified at pH 7.6, where the initial and final pH values were equal, indicating a net neutral surface charge.
Dosage effect of adsorbent
The dosage effect of PBCA–PVA on adsorption was studied by taking the adsorbent in the range of 0.01 to 0.12 g, as given in Fig. 9c. it was noted that acetamiprid adsorption increases on increasing PBCA–PVA dosage up to 0.08 g. No further increase was observed for pesticide adsorption as reported before as well49. At lower concentration the available sorption sites are few in number, and the sorption becomes independent of initial concentration, which enabled the complete acetamiprid uptake58. At higher concentration adsorbing sites were occupied which limits further adsorption. Hence 0.08 g of PBCA–PVA was chosen as an optimum dose for further experiments.
(a) pH Effect for acetamiprid (b) Determination of pH by point zero charge for PBCA–PVA particles (c) Effect of adsorbent dose (d) Van’t Hoff plots.
Thermodynamic analysis
To calculate standard enthalpy (∆H°) and entropy (∆S°) changes in the under-study adsorption process of pesticide by PBCA–PVA, Van’t Hoff’s equation was used. The linear form of Van’t Hoff’s equation is represented as50:
In the above equation, R shows the ideal gas constant with a value of 8.314 Jmol− 1k− 1, k=\(\:{q}_{e/{C}_{e}}\) indicates adsorption affinity, T denotes temperature in Kelvin. Plotting lnK against 1/T the value of ∆H° and ∆S° were determined as given in Fig. 9d while their values are listed in Table 3. ∆H° value was negative whereas that of ∆S° was positive, pointing towards the exothermic nature of the process and increase in randomness at solution and solid interface. The values of ∆G° are negative at all tested temperatures which shows the spontaneity of the pesticide adsorption by PBCA–PVA. The value of ∆G° was calculated at various temperatures as51:
The ∆G° values that is < 0 (Table 3) shows favorable adsorption of acetamiprid pesticides’ by PBCA–PVA52.
Comparison of present adsorbent with those reported in literature
Table 4 provides comparison of present adsorbent performance with the already reported ones in literature.
Regeneration study of the PBCA–PVA adsorbent
Four continuous recycling adsorption-desorption tests were conducted to study the recyclability of the PBCA–PVA, and the results are plotted in Fig. 10. The pesticide loaded adsorbent was collected by filtration after each adsorption test, washed with KOH, HCl, and ethanol solutions followed by drying. The data revealed that in each recycling step the adsorption capacity has been reduced and at 4th cycle still it was retained at 61% which clearly shows the capability of adsorbent reusability for long commercial uses.
Recycling plot of PBCA–PVA adsorbent.
Conclusions
Plant-based cellulose acetate polyvinyl alcohol nanocomposite was prepared from Populus nigra, characterized using different instrumental techniques, such as EDX, XRD, SEM, and FTIR. The adsorbent was then used as an effective adsorbent for the elimination of acetamiprid pesticides from wastewater. EDX, SEM, XRD, and FTIR data confirmed the successful acetylation of plant-based cellulose and polyvinyl alcohol incorporation. The isotherm data was fitted well by Freundlich, Temkin and Harkins Jura isotherm models (R2 = 0.9938, 0.9996 and 0.9945 respectively) whereas the kinetics data was well fitted by the pseudo-second-order adsorption model with R2 value equal to 0.994. The thermodynamic study revealed that the reaction is spontaneous and exothermic due to negative value of ∆G°= -116.90, -150.17 and − 180.20 kJ/mol and ∆H°= -81.65 kJ/mol and positive value of ∆S°= 305.65 Jmol− 1K− 1. The successful removal of acetamiprid pesticide is attributed to the porous, and large surface area and polar groups on the surface of the prepared adsorbent. More than 60% removal was observed after three times recycling. The PBCA–PVA showed better result and can be used in future as an effective adsorbent on large scale. It can be used for removal of heavy metals, drugs, organic contaminants and harmful pesticides from wastewater if different active sites are incorporated into cellulose structure. However, being environmentally friendly and with selective nature, PBCA–PVA have some challenges in adsorption capacity, stability, and regeneration.
Data availability
No/Not applicable (this manuscript does not report data generation or analysis).
References
Plakas, K. V. & Karabelas, A. J. Removal of pesticides from water by NF and RO membranes—A review. Desalination 287, 255–265 (2012).
Zolgharnein, J., Shahmoradi, A. & Ghasemi, J. Pesticides removal using conventional and low-cost adsorbents: A review. Clean–Soil Air Water 39 (12), 1105–1119 (2011).
Farrukh, S., Hussain, A. & Iqbal, N. Fabrication and characterization of microfiltration blended membranes. Desalin. Water Treat. 52 (10–12), 1833–1840 (2014).
Qiu, Y. R. & Mao, L. J. Removal of heavy metal ions from aqueous solution by ultrafiltration assisted with copolymer of maleic acid and acrylic acid. Desalination 329, 78–85 (2013).
Filipović, K. et al. Highly efficient nano sorbent as a superior material for the purification of wastewater contaminated with anthraquinone dye RB19. J. Water Process. Eng. 67, 106118 (2024).
Kostić, M. et al. Ultrasound-assisted synthesis of a new material based on MgCoAl-LDH: Characterization and optimization of sorption for progressive treatment of water. Environ. Technol. Innov. 26, 102358 (2022).
Martín, M. B. et al. Degradation of a four-pesticide mixture by combined photo-Fenton and biological oxidation. Water Res. 43 (3), 653–660 (2009).
Velinov, N. et al. Ultrasonic-assisted synthesis of wood-aluminium-based sorbents: Comparison with conventional Preparation and evaluation of chromium removal. Wood Mater. Sci. Eng. 18 (3), 1065–1075 (2023).
Velinov, N. et al. The application of wood Biowaste chemically modified by Bi2O3 as a sorbent material for wastewater treatment. Processes 12 (9), 2025 (2024).
Segura, C. et al. Imidacloprid oxidation by photo-Fenton reaction. J. Hazard. Mater. 150 (3), 679–686 (2008).
Fakhfakh, F., Raissi, S., Ben Jeddou, F., Zghal, Z., Ghorbel, A. & R., & Isotherm and kinetic modeling of cr (VI) removal with quaternary ammonium functionalized silica. J. Sol-Gel Sci. Technol. 111 (3), 921–940 (2024).
Košutić, K. et al. Removal of arsenic and pesticides from drinking water by nanofiltration membranes. Sep. Purif. Technol. 42 (2), 137–144 (2005).
Kostić, M. M. et al. Effects of ultrasound on removal of ranitidine hydrochloride from water by activated carbon based on Lagenaria siceraria. Environ. Eng. Sci. 36 (2), 237–248 (2019).
Yaşar, S. et al. Esterification of cellulose isolated from black Poplar (Populus Nigra L.) sawdust with octanoyl chloride. Turkish J. For. 22 (3), 306–310 (2021).
Hariani, P. L., Riyanti, F. & Kurniaty, A. Modification of cellulose with acetic acid to removal of methylene blue dye. J. Phys. Conf. Ser. (IOP Publishing, 2019).
Radovic, M. D. et al. Effects of system parameters and inorganic salts on the photodecolourisation of textile dye reactive blue 19 by UV/H2O2 process. Water SA. 40 (3), 571–578 (2014).
Velinov, N. et al. The influence of various solvents’ Polarity in the synthesis of wood biowaste sorbent: Evaluation of dye sorption. Biomass Convers. Biorefinery 1–12 (2023).
Wahab, M., Muhammad, Z. & Salman, S. A novel approach to remove Ofloxacin antibiotic from industrial effluent using magnetic carbon nanocomposite prepared from sawdust of Dalbergia sissoo by batch and membrane hybrid technology. Membrane Technol. 27, 28 (2019).
Kumar, A. et al. Characterization of cellulose nanocrystals produced by acid-hydrolysis from sugarcane Bagasse as agro-waste. J. Mater. Phys. Chem. 2 (1), 1–8 (2014).
Tristantini, D. & Yunan, A. Characterization of cellulose acetate based on empty fruit bunches and dried jackfruit leaves as replacement candidates for microbeads. In E3S Web. Conferences. Vol. 67, p. 04024 (2018).
Tristantini, D., Dewanti, D. P. & Sandra, C. Isolation and characterization of α-cellulose from blank bunches of palm oil and dry jackfruit leaves with alkaline process NaOH continued with bleaching process H2O2. AIP. Conference Proceedings. vol. 1904 (2017).
Bahmid, N. A., Syamsu, K. & TIP, A. M. Pengaruh Ukuran Serat Selulosa Asetat Dan Penambahan dietilen Glikol (DEG) Terhadap Sifat Fisik Dan Mekanik bioplastik. J. Teknologi Industri Pertanian 24 (3) (2014).
He, T. et al. Effect of nano precipitated calcium carbonate on the properties of hydrogels prepared with acrylamide, starch, and TEMPO-oxidized nanocellulose. Bioresources 17 (3), 5079 (2022).
Shahinur, S. et al. Effect of rot-, fire-, and water-retardant treatments on jute fiber and their associated thermoplastic composites: A study by FTIR. Polymers 13 (15), 2571 (2021).
Jafta, N. et al. Extraction Of cellulose from agricultural waste forwater purification (2021).
Castro-Guerrero, C. F. et al. Purification of cellulose from rice husk for the synthesis of nanocellulose. in IEEE 16th International Conference on Nanotechnology (IEEE-NANO) (IEEE, 2016).
Pota, G. et al. Co-immobilization of cellulase and β-glucosidase into mesoporous silica nanoparticles for the hydrolysis of cellulose extracted from Eriobotrya japonica leaves. Langmuir 38 (18), 5481–5493 (2022).
Li, P. et al. Preparation of bio-based aerogel and its adsorption properties for organic dyes. Gels 8 (11), 755 (2022).
Hokkanen, S., Bhatnagar, A. & Sillanpää, M. A review on modification methods to cellulose-based adsorbents to improve adsorption capacity. Water Res. 91, 156–173 (2016).
Cichosz, S. & Masek, A. IR study on cellulose with the varied moisture contents: Insight into the supramolecular structure. Materials 13 (20), 4573 (2020).
Zuo, W., Zuo, L., Geng, X., Li, Z. & Wang, L. Radical-polar crossover enabled triple cleavage of CF2Br2: A multicomponent tandem cyclization to 3- fluoropyrazoles. Org. Lett. 25 (32), 6062–6066 (2023).
Chong, K. Y. et al. CaCO 3-decorated cellulose aerogel for removal of congo red from aqueous solution. Cellulose 22, 2683–2691 (2015).
Wahab, M. et al. Effective removal of Tetracycline from water by batch method using activated carbon, magnetic carbon nanocomposite, and membrane hybrid technology. Z. Phys. Chemie 235 (10), 1323–1354 (2021).
Kaiblinger, N., Hahn, R., Beck, J., Wang, Y. & Carta, G. Direct calculation of the equilibrium composition for multi-component Langmuir isotherms in batch adsorption. Adsorption 30 (1), 51–56 (2024).
Nandiyanto, A. B. D. et al. Carbon particle size from galangal rhizomes as the sustainable adsorbent: Synthesis and mathematical calculation analysis in the adsorption isotherm characteristics. J. Stud. Sci. Eng. 4 (2), 21–47 (2024).
Park, J. C., Joo, J. B. & Yi, J. Adsorption of acid dyes using polyelectrolyte impregnated mesoporous silica. K J. Chem. Eng. 22, 276–280 (2005).
Wahab, M. et al. Adsorption-membrane hybrid approach for the removal of Azithromycin from water: An attempt to minimize drug resistance problem. Water 13 (14), 1969 (2021).
Panagiotis Zalmas, L. et al. DNA-damage response control of E2F7 and E2F8. EMBO Rep. 9 (3), 252–259 (2008).
Velinov, N. et al. New biosorbent based on Al2O3 modified lignocellulosic biomass (Lagenaria vulgaris): Characterization and application. Env Eng. Sci. 35 (8), 791–803 (2018).
Kostić, M. et al. Ultrasound-assisted synthesis of a new material based on MgCoAl-LDH: Characterization and optimization of sorption for progressive treatment of water. Env Tech. Innov. 26, 102358 (2022).
Podder, M. & Majumder, C. Studies on the removal of as (III) and as (V) through their adsorption onto granular activated carbon/MnFe2O4 composite: Isotherm studies and error analysis. Compos. Interfaces 23 (4), 327–372 (2016).
Lagergren, S. K. et al. About the theory of so-called adsorption of soluble substances. Sven Vetenskapsakad Handingarl 24, 1–39 (1898).
Kostić, M. et al. A new catalyst with the superior performance for treatment of water polluted by anthraquinone compounds. Bull. Mater. Sci. 44, 1–10 (2021).
Ho, Y. S., Ng, J. & McKay, G. Kinetics of pollutant sorption by biosorbents. Sep. Purif. Methods 29 (2), 189–232 (2000).
Riahi, K., Chaabane, S. & Thayer, B. B. A kinetic modeling study of phosphate adsorption onto Phoenix dactylifera L. date palm fibers in batch mode. J. Saudi Chem. Soc. 21, S143–S152 (2017).
Cheung, W., Ng, J. & McKay, G. Kinetic analysis of the sorption of copper (II) ions on Chitosan. J. Chem. Technol. Biotechnol. Int Res. Process. Env Clean. Tech. 78 (5), 562–571 (2003).
Velinov, N. et al. Characterization and application of wood-ZrO2 sorbent for simultaneous removal of chromium (III) and chromium (VI) from binary mixture. Nordic Pulp. Paper Res. J. 36 (2), 373–385 (2021).
Srivastava, S., Tyagi, R. & Pant, N. Adsorption of heavy metal ions on carbonaceous material developed from the waste slurry generated in local fertilizer plants. Water Res. 23 (9), 1161–1165 (1989).
Tassone, L. et al. Clinical and genetic diagnosis of warts, hypogammaglobulinemia, infections, and myelokathexis syndrome in 10 patients. J. Allergy Clin. Immunol. 123 (5), 1170–1173 (2009).
Kara, S. et al. Modeling the effects of adsorbent dose and particle size on the adsorption of reactive textile dyes by fly Ash. Desalination 212 (1–3), 282–293 (2007).
Falcoz, Q. et al. Kinetic rate laws of cd, Pb, and Zn vaporization during municipal solid waste incineration. Env Sci. Technol. 43 (6), 2184–2189 (2009).
Lima, E. C. et al. A critical review of the Estimation of the thermodynamic parameters on adsorption equilibria. Wrong use of equilibrium constant in the Van’t hoof equation for calculation of thermodynamic parameters of adsorption. J. Mol. Liquids. 273, 425–434 (2019).
Silva, M. et al. Preliminary feasibility study for the use of an adsorption/bio-regeneration system for Molinate removal from effluents. Water Res. 38 (11), 2677–2684 (2004).
Akhtar, M. et al. Utilization of organic by-products for the removal of organophosphorous pesticide from aqueous media. J. Haz Mater. 162 (2–3), 703–707 (2009).
Ratola, N., Botelho, C. & Alves, A. The use of pine bark as a natural adsorbent for persistent organic pollutants–study of Lindane and heptachlor adsorption. J. Chem. Tech. Biotech: Int. Res. Process Environ. Clean. Tech. 78 (2-3), 347–351 (2003).
El Bakouri, H. et al. Drin pesticides removal from aqueous solutions using acid-treated date stones. Bioresource Technol. 100 (10), 2676–2684 (2009).
Islam, M. A., Sakkas, V. & Albanis, T. A. Application of statistical design of experiment with desirability function for the removal of organophosphorus pesticide from aqueous solution by low-cost material. J. Haz Mater. 170 (1), 230–238 (2009).
Sharma, R. K., Kumar, A. & Joseph, P. Removal of atrazine from water by low cost adsorbents derived from agricultural and industrial wastes. Bull. Environ. Contam. Toxicol. 80, 461–464 (2008).
El Bakouri, H. et al. Potential use of organic waste substances as an ecological technique to reduce pesticide ground water contamination. J. Hydrol. 353 (3–4), 335–342 (2008).
Deng, S. et al. Enhanced removal of Pentachlorophenol and 2, 4-D from aqueous solution by an aminated biosorbent. J. Hazard. Mater. 165 (1–3), 408–414 (2009).
Ghosh, S. et al. Adsorption behavior of Lindane on rhizopus oryzae biomass: Physico-chemical studies. J. Hazard. Mater. 172 (1), 485–490 (2009).
Acknowledgements
The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through Large Research Project under grant number RGP2/46/46.
Author information
Authors and Affiliations
Contributions
GS, MZ, SBM, AYAA, all are equal contributors and have equally contributed in experiment, conceptualization, literature survey and MS writing. All authors have approved the final draft paper for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Said, G., Zahoor, M., Moussa, S.B. et al. Plant-based modified cellulose obtained from Populus nigra as a potential adsorbent for pesticides removal from aqueous media. Sci Rep 15, 18880 (2025). https://doi.org/10.1038/s41598-025-04084-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-04084-3
Keywords
This article is cited by
-
Zinc Peroxide-based Carbon Composite for the Removal of Highly Toxic Insecticide Acetamiprid from Water
Water, Air, & Soil Pollution (2026)
-
Development of novel nanocellulose based biofunctional materials from Raphia farinifera inflorescence with enhanced structural properties for high efficiency removal of hazardous organic contaminants from water
Discover Environment (2025)