Abstract
Boriding is a thermochemical process by which the surface of metallic materials is strongly improved for many applications. When boriding is applied to ferrous alloys, a boride layer containing mainly a single-phase Fe\(_{2}\)B or a double-phase Fe\(_{2}\)B/FeB grows on the metallic substrate. The thickness of the boride layers depends primarily on the temperature and the treatment time. On the other hand, the thermal diffusivity is related to the materials’ density and thermal conductivity. Therefore, as the thickness of the boride layers increases, their density decreases, and so does their thermal diffusivity. In this work, the change in thermal diffusivity of the layered material, as a function of thermochemical treatment, is assessed. Samples of AISI 1018 steel were exposed to boriding treatment during 2, 4, 6, and 8 h at a constant temperature of 950 \(^\circ\)C. The crystalline structure of the layers was analyzed by X-ray diffraction and optical microscopy. The mechanical properties and the thermal diffusivity of the steel-boride layered materials were evaluated by Vickers micro indentation and Photothermal Spectroscopy, respectively.
Similar content being viewed by others
Introduction
Boriding is a thermochemical processes that modify surface characteristics such as hardness, young modulus, wear resistance, and corrosion resistance of metallic materials. This process consists of exposure of a metallic material (ferrous and non-ferrous) to high temperature conditions in a boron-rich environment (liquid, gaseous or solid) and its exposition to high temperatures (800–1100 \(^\circ\)C). During the process, the surface of the base material is saturated with boron to promote diffusion from the surface to the substrate1,2. As a result of the boriding process, boride layers are obtained on the base material. For ferrous alloys, monophasic layers of Fe\(_{2}\)B or biphasic layers of FeB/Fe\(_{2}\)B can be obtained. The Fe\(_{2}\)B layer exhibits a tetragonal structure. Meanwhile, the FeB layers have an orthorhombic structure; the highest atom density is in the [0 0 1] direction of the Fe\(_{2}\)B phase2. The layer’s structure depends on experimental conditions such as time and temperature, the proportion of available active boron, the chemical composition of the substrate and the technique employed3.
For the Fe\(_{2}\)B formation, a medium of low boron concentration is necessary in the boriding agent. Meanwhile, FeB formation requires a high boron potential and a high concentration of steel alloying elements2. The monophase Fe\(_{2}\)B layer is more convenient than the FeB phase or a double phase FeB/Fe\(_{2}\)B layer because the FeB is harder and, therefore, more brittle, and consequently, their exposure to mechanical or thermal shock could generate cracks and detachments. According to the literature, the convenient thickness of the Fe\(_{2}\)B layer will depend on industrial applications, considering that the tribological properties of layered materials improved, thus extending the useful life of the base materials. The control of the boride layer thickness is achieved by assessing the effect of experimental conditions of time and temperature4.
Thermal diffusivity is a property that determines the capability of a material to transfer thermal energy under non-stationary conditions. This property is important in designing heat exchangers or thermal insulation materials, where some of the used materials consist of a base substrate and alternating layers of other materials, such as metal oxides generated by exposure to thermal processes, such as metal boriding. The thermal diffusivity values of the boride layers produced by the process described above have not been reported. In this work, a self-normalized photoacoustic technique5,6 was applied to measure the effective thermal diffusivity of the multilayered system resulting from the boridig processes in AISI 1018 steel as a function of time. This thermal property was determined as a function of the thickness of the layers of Fe\(_{2}\)B. These measurements allowed the establishment of the kinetics of layer growth of Fe\(_{2}\)B as a function of the effective thermal diffusivity of the multilayered system and, at the same time, the thermal diffusivity of Fe\(_{2}\)B.
This study aimed to establish a correlation between thermal diffusivity and thickness of a Fe\(_{2}\)B layer type obtained as a function of treatment time through the thermochemical boriding process on AISI 1018 steel. The knowledge of the relationship between thermal diffusivity and the thickness of the Fe\(_{2}\)B layer should allow a better description of the capacity of the boride layer to conduct or dissipate heat which is a critical aspect in the engineering applications of these materials.
Results
X-ray diffraction (XRD)
The diffraction patterns are shown in Fig. 1, they correspond to the AISI 1018 steel samples with and without treatment.
XRD patterns of AISI 1018 steel samples exposed to the boriding processes at 950 \(^\circ\)C during (a) 0 h (un-treated), (b) 2 h, (c) 4 h, (d) 6 h and (e) 8 h.
The XRD pattern of the sample without treatment (Fig. 1a)) shows diffraction peaks at 2\(\theta\) = 44.54°, 64.69° y 82.17°. According to the literature, these peaks correspond to the crystalline planes (110), (200) and (211) of the iron structure (Fe) (4113941). However, Fig. 1b,e show the XRD patterns for the samples exposed to 2, 4, 6, and 8 h of treatment. The diffraction peaks at these time can be visualized at 2\(\theta\) = 24.65°, 35.01°, 44.91°, 49.67°, 50.44°, 56.19°, 56.88°, 73.56°, 74.17°, 79.46°, 80.60°, 84.68° and 89.39° which, according to the literature, correspond to the planes (110), (020), (121), (112), (220), (022), (130), (132), (330), (123), (141), (240), and (042), respectively. Those peaks are characteristics of the tetragonal crystalline structure of the Fe\(_{2}\)B phase (98-003-0446).
Optical microscopy (OM)
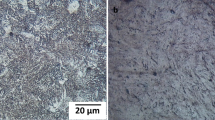
On the other hand, Fig. 2 shows OM images of the thermochemically treated AISI 1018 steel samples at different exposure times.
OM images of the AISI 1018 steel samples exposed to (a) 2 h, (b) 4 h, (c) 6 h, and (d) 8 h of treatment.
Two different regions can be observed in Fig. 2a–c. The first, on the surface (at the bottom of these figures), with a saw-tooted morphology, corresponds to the Fe\(_{2}\)B layer, typical of the boride layer formed on low carbon steels7. Likewise, in Fig. 2a–c, it is possible to observe a clear region corresponding to the substrate material (AISI 1018 steel), which is not affected by the boriding process.
Furthermore, Fig. 3a shows the behavior of the thickness of the boride layers as a function of the treatment time. Likewise, Fig. 3b shows the corresponding growth percentage. This growth percentage was estimated by considering the thickness of the samples as 100 %. The thickness of the boriding layer was measured from the digitalized images of the cross section of each sample. The thickness layers of Fe\(_{2}\)B were 63.94, 83.02, and 168.86 \(\upmu\) m for 2, 4, and 6 h, respectively. These values were used to evaluate the thermal diffusivity of the Fe\(_{2}\)B phase.
Layer thickness as a function of the treatment time (a) and behavior of the layer thickness as a function of the sample size expressed in percentage (b).
Hardness testing (HT)
The graph of the microhardness behavior of the samples through their cross-section is shown in Fig. 4a. In Fig. 4a, it can be observed that for each sample, except for the sample treated during 8 h, the microhardness is higher on the surface and decreases as the indentation moves toward the inside of the sample; this is because the Fe\(_{2}\)B layer is more compact and harder than the AISI 1018 steel. The hardness behavior of the sample exposed to 8 h (purple line) of heat treatment is due to the fact that it consists mainly of Fe\(_{2}\)B, which is why the microhardness is approximately constant. On the other hand, Fig. 4b shows the corresponding microindentations for the sample treated at 6 h.
(a) Microhardness behavior as a function of the treatment time across the cross-section of AISI 1018 steel samples with and without thermochemical treatment. (b) Corresponding microindentations for the sample exposed to 6 h of treatment.
Photothermal spectroscopy (PS)
From the effective thermal diffusivities measured using the photoacoustic technique described in the literature5,6, knowing the thicknesses of the materials involved and the thermal diffusivity of AISI 1018 steel, it is possible to obtain the thermal diffusivity of the Fe\(_{2}\)B layer, through Eq. (1)
Given that layers with thicknesses l\(_{1}\) and l\(_{3}\) correspond in this case to the phase Fe\(_{2}\)B, the Eq. (1) reduces to Eq. (1.2):
In this way, the thermal diffusivity (\(\alpha\) \(_{1}\)) of the layer Fe\(_{2}\)B can be obtained through the relation (Eq. (1.2))
where L\(_{effec}\) is the thickness sample, l\(_{1}\) is the thickness of the upper Fe\(_{2}\)B layer, l\(_{2}\) is the thickness of the non-treated substrate, l\(_{3}\) is the thickness of the bottom Fe\(_{2}\)B layer, \(\alpha\) \(_{effec}\) is the thermal diffusivity of the Fe\(_{2}\)B/steel sample (measured by the photoacoustic technique), \(\alpha\) \(_{1}\) = \(\alpha\) \(_{2}\) is the thermal diffusivity of the Fe\(_{2}\)B layer, \(\alpha\) \(_{3}\) is the thermal diffusivity of the substrate (AISI 1018 steel)
Figure 5 shows the representative behavior of the tangent of the self-normalized phase, in this case, for the AISI 1018 steel sample. According to the literature, the thermal diffusivity of the sample can be obtained by Eq. (2) (inset in Fig. 5):
where f is the frequency where the first discontinuity appears in the self-normalized phase graph as a function of the modulation frequency. \(\alpha\) is the thermal diffusivity of the material when it consists of a single layer, or \(\alpha\) is the effective thermal diffusivity when it consists of a multilayered material. L is the thickness of the material (single-layered or multilayered material).
Tangent of the self-normalized phase, as a function of the modulation frequency, for the untreated AISI 1018 steel sample.
These equations apply to multilayer samples using the photoacoustic technique, assuming the light surface absorption of the sample and the one-dimensional heat diffusion model (see reference6)
The value obtained for this material was \(\alpha\) \(_{1}\) = 0.234 cm\(^{2}\)/s.
Then, the effective thermal diffusivities of the boride samples obtained by the boriding process were measured for the different treatment times (see Table 1, column 1). The corresponding values were obtained in the same way as for the AISI 1018 sample. Table 1, column 5, summarizes the results. Column 6 shows the corresponding thermal diffusivity values for the Fe\(_{2}\)B calculated for each sample according to Eq. (1.2).
Discussion
The presence of XRD peaks at the described angles and their correspondence with the crystalline planes of the Fe\(_{2}\)B demonstrates that this phase was indeed formed in the thermochemically treated materials, even in the first 2 h treatment. In particular, there are no peaks of other boride chemical samples, such as FeB, as reported in other works1. The absence of FeB in the samples studied in this work may be due to the different preparation conditions. For instance, Ruiz–Trabolsi et al. used a fresh mixture of boride agents; meanwhile, in this work, a mixture of 50 % fresh boride agent and 50 % reused boride agent was employed, and the latter has a lower boron potential. Consequently, the only phase formed was Fe\(_{2}\)B because the FeB phase requires a bigger boron potential for its formation.
In fact, single Fe\(_{2}\)B layers are preferable to biphasic layer FeB/Fe\(_{2}\)B type because the FeB phase has the highest hardness and therefore the highest brittleness, as established by Quin et al.8.
However, the presence of the FeB type phase is not evident in Fig. 2a–c, which corroborates what was described above regarding the different processes carried out in this and other works. For comparison, OM images of a biphasic layer FeB/Fe\(_{2}\)B in the same AISI 1018 steel are reported by Sánchez-Islas9. They achieved the biphasic layer FeB/Fe\(_{2}\)B at a thermochemical treatment of 1,000 \(^\circ\)C, corroborating the hypothesis that the FeB phase requires a higher activation energy10.
The lack of the FeB phase may also be related to the lower carbon content in the samples for this study; therefore, the boron does not find diffusion barriers through its path, forming a Fe\(_{2}\)B single phase layer, which is consistent with the results obtained by XRD7. Furthermore, the increase in layer thickness as a function of the treatment time is notable since the temperature remained constant throughout the process; hence, the treatment time was the only variable.
According to the literature, the orientation of the grain of the boride layers is controlled by kinetic growth3, which depends mainly on the chemical composition of the substrate and the thermochemical conditions (time and temperature)11,12.
Furthermore, for samples exposed to 8 h of treatment, the Fe\(_{2}\)B layer completely crossed the cross-section of the sample, which was expected due to the small thickness and low content of the alloying elements of the substrate. The low carbon content and low alloying concentration accelerated the kinetic growth of the layers, as reported in the literature, where similar layer thicknesses have been achieved with similar treatment conditions13.
In addition, Fig. 3a shows that the Fe\(_{2}\)B layer thickness increases with the treatment time. This behavior can be explained by the fact that the growth of boride layers is highly dependent on experimental conditions such as treatment time and temperature13,14. However, since the temperature remained constant in this work (950 \(^\circ\)C), the thickness of the layer depends only on the treatment time. As expected, the growth percentage follows the same behavior as the thickness, since the thickness growth obeys a parabolic growth law15,16; hence, the growth percentage is only influenced by the treatment time.
Otherwise, the microhardness in each region of the samples changes slightly since the average microhardness is measured between the AISI 1018 steel and the boundary of the Fe\(_{2}\)B saw-tooted layer. This behavior results from the boron migration from the surface to the volume of the substrate, as reported by Hernández-Sánchez17. The results in Fig. 4b agree well with the results reported by Ortiz-Domínguez et al.4; they exposed an AISI 9840 steel to boriding in a temperature range of 850 to 1000 \(^\circ\)C and treatment time between 2 and 8 h. However, they obtained a biphasic layer of FeB/Fe\(_{2}\)B on the surface of the treated steel. The formation of this biphasic layer is probably because AISI 9840 steel possesses higher alloying elements than the AISI 1018 steel used in the present research. In addition, treatment at higher temperatures (1000 \(^\circ\)C) probably favored the formation of the FeB phase.
Finally, it can be seen that the thermal diffusivity values obtained for Fe\(_{2}\)B from the effective diffusivities for the different samples (see Table 1, column 6) are very similar and close to the value obtained for the sample exposed to 8 h of treatment, which corresponds to the sample with the highest value of Fe\(_{2}\)B layer thickness.
The prior results exhibit the potential of the self-normalized photoacoustic technique for determining the thermal diffusivity of layered materials grown on substrates through thermochemical treatment, once the thickness of the layers involved and the thermal diffusivity of the support material have been determined. This is a significant result because obtaining this thermal property for layered materials attached to a substrate is not an easy task.
The behavior of the effective thermal diffusivity of multilayered materials of AISI 1018 steel and Fe\(_{2}\)B, as a function of the treatment time, is shown in Fig. 6. It is interesting to note that this thermal parameter decreases monotonically with the treatment time; this is a result of the increased proportion of Fe\(_{2}\)B in the AISI 1018 steel, since the thermal diffusivity of Fe\(_{2}\)B is almost one order of magnitude lower than the corresponding one for the steel substrate (0.044 cm\(^{2}\)/s and 0.234 cm\(^{2}\)/s, respectively). Then, this thermal parameter (the effective thermal diffusivity) can be used to follow the growth kinetics of the Fe\(_{2}\)B in steel.
The behavior of the effective thermal diffusivity of AISI 1018 steel and Fe\(_{2}\)B composite materials as a function of treatment time.
The above-mentioned contrasts with that reported by Mandal et al.18, who synthesized CaCO\(_{3]}\)/rGO (calcium carbonate/reduced graphene oxide) to observe the influence of rGO on the thermal properties of CaCO\(_{3}\), finding that the thermal diffusivity increases as the percentage of rGO increases. Unlike in this work, layers of Fe\(_{2}\)B decrease the thermal diffusivity of the steel as the thermochemical treatment time increases. This contrasting result can be explained because in the case of Mandal’s work the materials’ density increases with increasing rGO percentage, while in our work the density of the material decreases with increasing the thickness of the Fe\(_{2}\)B layer increases.
Conclusion
Under experimental conditions, the boriding treatment applied to an AISI 1018 steel produces layers of a single-phase Fe2B type. These layered materials have better mechanical characteristics than a single-phase FeB type layer or a two-phase FeB/Fe2B type layer. In addition, the layer thickness increased with the treatment time, indicating that the growth kinetics of the layers depend directly on the treatment conditions.
On the other hand, in the sample exposed to 8 h of treatment, the Fe\(_{2}\)B layer completely covered the sample, forming a sample that is practically composed of the Fe\(_{2}\)B phase. The microhardness values corroborate this assumption as the Fe\(_{2}\)B phase possesses more hardness than the AISI 1018 steel.
The sample exposed to 8 h of treatment presented a higher hardness compared to the samples exposed to 2, 4, and 6 h of treatment, in addition to being almost constant along the cross-section of the sample, indicating that practically the entire sample corresponds to the Fe\(_{2}\)B phase. Concerning the thermal diffusivity values, this thermal property decreases with heat treatment time as the boriding layer increases, and the thermal diffusivity of this last material is smaller than the corresponding one for the AISI 1018 steel. So, this thermal property can probably be used to characterize the growth kinetics of the boriding layers.
It was shown that the proposed photoacoustic technique is adequate to measure the thermal diffusivity of multilayered samples, and that this thermal property can be used to follow structural changes in samples subjected to different heat treatments.
Methods
For thermochemical treatment, cylindrical samples of AISI 1018 steel were prepared using a metallographic technique consisting of sanding the surface of the samples with 120, 220, 320, 400, 500, 600, and 1000 SiC abrasive paper and polishing with Al\(_{2}\)O\(_{3}\) (0.05 \(\upmu\)m) for a mirror finish. The chemical composition of an AISI 1018 steel is approximately 0.15–0.20 wt % C, 0.60–0.90 wt % Mn, 0.15–0.30 wt % Si, 0.04 wt % P, 0.05 wt % S and Fe balance. The length of the samples used for the different exposure times (2, 4, 6 and 8 h) were 0.0980 cm, 0.0993 cm, 0.0994 cm, and 0.0403 cm, while for the untreated sample (0 h), it was 0.0904 cm , and a diameter of 7.9 mm for all the samples. The main reason for the different length of the sample exposed during 8 h of treatment was to ensure that the boride layers completely cover the sample.
The heat treatment to develop the boron layer was carried out by introducing AISI 1018 steel samples into the boron precursor (Duborid G) with a mixture of 50 % fresh boron agent and 50 % reused boride agent (first use). The treatment was carried out at a constant temperature of 950 \(^\circ\)C for periods of 2, 4, 6 and 8 h. The steel sample treated for 8 h was transformed entirely into Fe\(_{2}\)B. After thermochemical treatment, each sample was sectioned and vertically mounted in bakelite to measure the thickness of the different layers, as shown in Fig. 7.
Cross-section scheme for the mounted samples for the matter of measuring the layer thickness.
XRD, OM, HT and PS were used to characterize the structure of the samples and their physical and mechanical properties.
The crystalline structure of the phases of the materials present in the layers was analyzed by a diffractometer (Bruker, Billerica, MA, USA) with a Cu source, using a K\(\alpha\) line, to 35 kV, a 25 mA and a sweep speed of 2°/min in a range of 2\(\theta\) = 20° to 100° with increases of 0.2°.
To facilitate handling, the samples were mounted on bakelite. The mounted samples were sanded until to exhibit their cross-section, exposing the substrate material (AISI 1018 steel) and the Fe2B layer, as is schematized in Fig. 7. Then each layer was observed using an inverted metallographic microscope (VELAB, model VE-407), to measure its thickness. This was carried out by taking cross-section images of the samples by a digital camera (Canon model EOS3 Raptor T3), each thickness layer was obtained as an average of at least 100 measurements by means of Image-Pro Plus\(^T\) \(^M\) software. Table 1, columns 2, 3 and 4 summarizes the thicknesses for the total cross-section for each sample (column 2), the Fe2B layer thickness for the upper side (in Fig. 7) of each sample (column 3) and the Fe2B layer thickness for the bottom side (in Fig. 7) of each sample (column 4).
To ensure a precise measurement, at least 100 measurements were carried out in different regions on the cross-section of the samples.
Hardness testing was performed on the cross-section of the samples from their surface to the center. According to the guidelines of the standards ASTM-E384 and ASTM-C132719,20, at least 5 indentations were carried out for each distance. A microhardness tester (CMS Metrology) was used, with a Vickers-type diamond indenter and a constant load of 25 g.
The thermal diffusivity measurement for all samples was performed using a self-normalized Photoacoustic Technique5. A semiconductor laser centered at 600 nm (Thorlabs, part numbers L904P010) was used as a radiation source. The diameter of the light spot on the sample was approximately 2 mm. The laser was powered by a current source (Standford Research Systems, LDC 202 C), controlled through the TTL port of the lock-in amplifier (Standford Research Systems, model SR-830). For data analysis, the corresponding photothermal signals were obtained in the front and transmission photoacoustic configurations in a range of 1 to 35 Hz, with increments of 1 Hz5.
Data availability
The materials and data that supports the findings of this study are available from the corresponding author, upon reasonable request.
References
Ruiz-Trabolsi, P. A. et al. Kinetics of the boride layers obtained on AISI 1018 steel by considering the amount of matter involved. Coatings 11, 1–17. https://doi.org/10.3390/coatings11020259 (2021).
Campos, I. et al. Effect of boron paste thickness on the growth kinetics of Fe\(_{2}\)B boride layers during the boriding process. Appl. Surf. Sci. 243, 1–4. https://doi.org/10.1016/j.apsusc.2004.09.099 (2024).
Dybkov, V. I., Goncharuk, L. V., Khoruzha, V. G., Samelyuk, A. V. & Sidorko, V. R. Growth kinetics and abrasive wear resistance of boride layers on Fe-15Cr alloy. Mater. Sci. Technol. 27, 1502–1512. https://doi.org/10.1179/026708310X12683157551577 (2011).
Ortiz-Domínguez, A. et al. Kinetics of boron diffusion and characterization of Fe\(_{2}\)B layers on AISI 9840 steel. Prot. Met. Phys. Chem. Surf. 53, 534–547. https://doi.org/10.1134/S2070205117030169 (2017).
Balderas-López, J. A. Self-normalized photoacoustic techniques for thermal diffusivity measurements in metals. Rev. Mex. Fis. 50, 120–126 (2004).
Balderas-López, J. A. Generalized expression for the self-normalized signal in photothermal experiments for multilayered materials in the frequency domain. J. Appl. Phys. 132, 1–11. https://doi.org/10.1063/5.0091833 (2022).
Von Matuschka, A. G. Boronizing, 1st ed (Hanser, 1980).
Quinn, J. B. & Quinn, G. D. Indentation brittleness of ceramics: A fresh approach. J. Mater. Sci. 32, 4331–4346. https://doi.org/10.1023/A:1018671823059 (1997).
Sánchez-Islas, A. et al. Dry sliding wear test on borided AISI 1018 steel under pin-on-disc configuration. Metall. Mater. Trans. A 53, 179–199. https://doi.org/10.1007/s11661-021-06511-2 (2022).
Makuch, N. et al. Growth kinetics and some mechanical properties of two-phase boride layers produced on commercially pure titanium during plasma paste boriding. Thin Solid Films 626, 25–37. https://doi.org/10.1016/j.tsf.2017.02.033 (2017).
Keddam, M. & Chegroune, R. A model for studying the kinetics of the formation of Fe\(_{2}\)B boride layers at the surface of a gray cast iron. Appl. Surf. Sci. 256, 5025–5030. https://doi.org/10.1016/j.apsusc.2010.03.048 (2010).
Milinovic, A., Marusic, V., Konjatic, P. & Beric, N. Effect of carbon content and boronizing parameters on growth kinetics of boride layers obtained on carbon steels. Materials 15, 1–17. https://doi.org/10.3390/ma15051858 (2022).
Toktas, G. & Selman, K. Indentation fracture toughness of boronized unalloyed and alloyed ductile iron. Mater. Chem. Phys. 296, 1–7. https://doi.org/10.1016/j.matchemphys.2022.127232 (2023).
Jain, V. & Sundararajan, G. Influence of the pack thickness of the boronizing mixture on the boriding of steel. Surf. Coat. Technol. 149, 21–26. https://doi.org/10.1016/s0257-8972(01)01385-8 (2002).
Sen, S., Sen, U. & Bindal, C. The growth kinetics of borides formed on boronized AISI 4140 steel. Vacuum 77, 195–202. https://doi.org/10.1019/j.vacuum.2004.09.005 (2005).
Genel, K., Ozbek, I. & Bindal, C. Kinetics of boriding of AISI W1 steel. Mater. Sci. Eng. A 347, 311–314. https://doi.org/10.1016/s0921-5093(02)00607-X (2003).
Hernández-Sánchez, E. et al. Tribological and mechanical behavior of automotive crankshaft steel superficially modified using the boriding hardening process. Coatings 14, 1–14. https://doi.org/10.3390/coatings14060716 (2024).
Mandal, S. K., Kumar, D., Bishwakarma, H., Kumar, R. & Medasetty, T. G. Influence of reduced graphene oxide on the morphology, structural, and thermal properties of calcium carbonate nanocomposites. Ceram. Int. 50, 9913–9918. https://doi.org/10.1016/j.ceramint.2023.12.292 (2024).
ASTM, E-384-05a. Standard Test Method for Microindentation Hardness of Materials (2005).
ASTM, C-1327-03a. Standard Test Method for Vickers Indentation Hardness of Advanced Ceramics (2003).
Acknowledgements
Y. Sánchez-Fuentes (CVU: 771277), L. A. Linares-Duarte (CVU:1244024) and C. Proa-Coronado (CVU: 328806) thank the Mexican Research Council (CONAHCYT) for PhD and Postdoc scholarships. Figures 3 and 5b) were obtained by optical microscopy (VELAB, model VE-407). Y. Sánchez-Fuentes created Figs. 1, 2, 4, 5a, 6 and 7. The graphics were made by OriginLab\(^T\) \(^M\) 2018, https://www.originlab.com/ .
Author information
Authors and Affiliations
Contributions
Y. S.-F. and L. A. L.-D. writing the original draft and conducting experiments, E. H.-S. and J. A. B.-A. resources and validation, C. P.-C. writing review, J. G. M.-H. and R. T.-R. investigation and analysis. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sánchez-Fuentes, Y., Linares-Duarte, L.A., Balderas-López, J.A. et al. Effect of boriding time on the effective thermal diffusivity of the borided AISI 1018 steel. Sci Rep 15, 19125 (2025). https://doi.org/10.1038/s41598-025-04105-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-04105-1