Abstract
The urinary bladder must remain distensible enough to expand as urine is produced yet strong enough to expel urine without rupture. Disruption in the arrangement of the bladder wall extracellular matrix (ECM) can lead to alterations in mechanical compliance, disrupting normal function and resulting in bladder dysfunction. Previously, we discovered that the mast cell activator compound 48/80 increased detrusor excitability and bladder wall mechanical compliance, resulting in increased urination frequency in mice in a mast cell-independent manner. While the effects on detrusor excitability depended on urothelial prostaglandin production, the mechanisms by which compound 48/80 altered mechanical compliance remained unclear. This study tests the hypothesis that compound 48/80 increases compliance through activation of matrix metalloproteases (MMPs) in the bladder wall. Using the Pentaplanar Reflected Image Macroscopy (PRIM) System and multiphoton microscopy, we measured ex vivo intravesical pressure, mechanical compliance, wall thickness, and permeability in bladders from C57Bl/6 mice before and after exposure to compound 48/80. Intravesical instillation of compound 48/80 increased mechanical compliance and reduced wall thickness without affecting urothelial permeability. This change in compliance was inhibited by the nonspecific MMP inhibitor doxycycline and the MMP-2 inhibitor ARP100. Bladder MMP-2 activity increased, and TIMP-2 activity decreased, after incubation with compound 48/80 as measured by zymography. Together, these findings suggest that compound 48/80 changes bladder mechanical compliance through ECM breakdown caused by rapid activation of MMP-2 and inhibition of TIMP-2. Moreover, these effects occur without altering urothelial permeability, suggesting a novel receptor-mediated mechanism and not a direct effect of compound 48/80 on MMP-2 or TIMP-2.
Similar content being viewed by others
Introduction
In addition to neuronal innervation and detrusor contractility, normal bladder function is highly influenced by the acellular components that reside in and comprise the extracellular matrix (ECM)1,2,3 The ability of the bladder’s ECM to maintain a balance between mechanical compliance and stiffness is key to normal function, as well as the progression of lower urinary tract symptoms4,5. Bladder ECM contains fibroblasts/myofibroblasts, fibrillar proteins (e.g., collagens and elastins), adhesive proteins, ECM receptors, cross-linking proteins, and matrix metalloproteases responsible for regulating ECM deposition and degradation6,7,8,9. While originally examined for its role in maintaining the integrity of the bladder wall during storage and filling, bladder ECM is now suggested to be a continuously dynamic structure that can respond to cellular signals and modulate cell-to-cell interactions10,11,12. Hence, the components of the ECM, (and the proteins that regulate it), represent a novel mechanism for understanding how bladder wall biomechanics affects bladder function and disease.
Matrix metalloproteases (MMPs) are a family of Zinc-dependent endopeptidases that are secreted in their “pro” form by many cells, including smooth muscle cells and fibroblasts13,14. These proteases play a role in ECM turnover by degrading components of the ECM, including fibrillar collagens, non-fibrillar collagens, and elastins15. MMPs are activated through removal of the pro-domain to expose the active site that is protected by a cysteine residue16. This activation takes place in the pericellular space and is triggered by multiple mediators (e.g., serine proteases, tryptases, and ATP) and inhibited by tissue inhibitors of matrix metalloproteases (TIMPs)17,18,19,20,21. Once activated, MMPs can degrade specific substrates based on their specific proteolytic sites14,22. Due to their effects on structural ECM proteins, direct MMP activation or relief of MMP inhibition by TIMPs can alter the biomechanics of soft tissue23,24. In the bladder, seven members of the MMP have been identified, but very few studies show their role in bladder dysfunction outside of bladder cancer6.
Previously, we uncovered that the mast cell activator compound 48/80 rapidly increased both bladder mechanical compliance and detrusor contractility via a mechanism that is independent of mast cells25,26. Intravesical instillation of compound 48/80 also increased urinary frequency, suggesting that the changes in mechanical compliance and detrusor excitability associated with symptoms of bladder overactivity. However, the mechanism by which compound 48/80 changes mechanical compliance remains unknown. This study seeks to determine if compound 48/80 activates MMPs and/or inhibits TIMPs to increase bladder compliance via rapid collagen remodeling. We then explore if compound 48/80-induced changes in detrusor contractility are dependent on tissue remodeling. Lastly, we investigate if compound 48/80 affects urothelial permeability to alter bladder function.
Results
Compound 48/80 increases bladder wall mechanical compliance and reduces stiffness
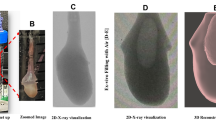
Previously, we discovered that compound 48/80 increased bladder mechanical compliance while simultaneously increasing the amplitude and leading slope of transient pressure events25. To again confirm the effects of compound 48/80 on bladder compliance were reproducible, ex vivo bladder filling in the PRIM System was performed in the absence or presence of 10 µg/mL compound 48/80 (Fig. 1A). Compound 48/80 increased mechanical compliance as noted by a rightward shift in the stress-stretch curve (Fig. 1B) and a significant increase in stretch at 10 and 25 mmHg (Fig. 1C). Additionally, stiffness is also significantly lowered at 25 mmHg (Fig. 1D).
Compound 48/80 increases bladder wall mechanical compliance. (A) Representative pressure-volume trace of ex vivo filling of C57Bl/6 mouse bladders in presence of the Mrgprb2 receptor agonist compound 48/80 (10 µg/mL). Compound 48/80 increases mechanical compliance, signified by the rightward shift in stress-stretch curve (B) and a significant increase in stretch at 10, and 25 mmHg (C, P = 0.027). Stiffness is decreased at 25 mmHg (D, P = 0.031). *P ≤ 0.05. N = 3.
Compound 48/80 decreases bladder wall thickness without increasing urothelial permeability
In blood vessels, substances that activate MMPs to degrade the basal lamina propria also alter endothelial permeability27,28. To test if compound 48/80 elicited a similar response in the bladder, we tested its effects on urothelial barrier function by measuring extravasation of 4 kDa FITC-dextran using confocal/multiphoton microscopy (Fig. 2). In order to recapitulate the conditions in which in vivo intravesical compound 48/80 caused bladder overactivity in mice25, a concentration of 50 µg/mL was used in these experiments. Also, an incubation time of 10 min was used to align with prior findings and those presented in Fig. 125. In ex vivo pressurized bladders exposed to 50 µg/mL compound 48/80, FITC-dextran fluorescence remained high in the lumen and low within the bladder wall, indicative of maintained barrier function (Fig. 3A, B). As a positive control, the surfactant Triton X-100 was used to permeabilize the bladder wall. Triton X-100 (1%) caused a migration of FITC-dextran fluorescence from the lumen into the bladder wall, indicative of a loss of urothelial barrier function (Fig. 3C). The absence of effect of compound 48/80 was not due to insufficient delivery of drug to the lumen, as pressurized bladders exposed to compound 48/80 had a statistically significant decrease in bladder wall thickness that was not recapitulated in bladders permeabilized with Triton X-100 (Fig. 3D).
Permeability of the bladder wall to FITC-Dextran. Representative images of urothelial permeabilization and bladder wall thickness using confocal images of FITC Dextran (4 kDa; purple) and second harmonic generation (SHG; yellow), respectively. Each panel shows collagen alone (left), FITC-Dextran alone (middle), and overlay of both channels (right). (A–C) An intact bladder pressurized to 25 mmHg in the absence of any drug. (D–F) The Mrgprb2 receptor agonist compound 48/80 (C 48–80, 50 µg/mL) administered intravesically for 10 min did not permeabilize the urothelium, signified by similar FITC signal as compared to controls. (G-I) Intravesical infusion of the detergent Triton X-100 (Triton, 1%) for 10 min permeabilized the urothelium, signified by the colocalization of FITC and SHG signal and a loss if FITC-Dextran from the lumen. Images represent a total n = 4 images taken from N = 4 mouse bladders. L: bladder lumen. A: bladder adventitia.
Compound 48/80 reduces bladder wall thickness without permeabilizing the wall. (A–C) Plots of FITC-Dextran fluorescence intensity as a function of distance across the bladder wall for bladders exposed to vehicle, the Mrgprb2 receptor agonist compound 48/80 (0 µg/mL), and 1% Triton X-100. Neither vehicle (A) nor 50 µg/mL compound 48/80 (B) permeabilized the bladder, as shown by the stark drop in fluorescence intensity at the transition from bladder lumen to bladder wall. Triton was used as positive control and it permeabilized the urothelium to let FITC into the wall (C). The different colors of the curves represent the Ns. (D) Quantification of bladder wall thickness after being exposed to vehicle, compound 48/80 and triton. 50 µg/mL compound 48/80 significantly reduced the thickness of bladder wall collagen whereas 1% Triton had no effect on wall thickness (P = 0.022 for Control vs. C-48/80 and P = 0.333 for Control vs. Triton, N = 4). Between group comparisons were made via two-tailed, paired, Student’s t test.
Compound 48/80 alters bladder wall mechanical compliance by activating MMP-2
To determine if the increase in bladder wall mechanical compliance caused by compound 48/80 was due to MMP activation, ex vivo bladder filling was performed in the presence of the nonselective MMP antagonist doxycycline (20 µM) before exposure to compound 48/80 (10 µg/mL) (Fig. 4A). Doxycycline prevented the increase in compliance caused by compound 48/80 (Fig. 4B), as well as preventing the increase in stretch (Fig. 4C) and the decrease in stiffness (Fig. 4D). To explore which MMPs may be responsible for effects of compound 48/80, this experiment was repeated using the specific MMP-2 inhibitor ARP100 (200 nM) (Fig. 5). In the presence of ARP100, compound 48/80 did not increase wall compliance nor did it alter stretch or stiffness (Fig. 5).
The effects of Compound 48/80 on bladder wall compliance are mediated by matrix metalloproteases. (A) Representative pressure-volume trace of ex vivo filling of C57Bl/6 mouse bladders in presence of the MMP inhibitor doxycycline (20 µM) alone and after addition of the Mrgprb2 receptor agonist compound 48/80 (10 µg/mL). Doxycycline blocks the increase in compliance driven by compound 48/80 signified by the absence of rightward shift of stress-stretch curve (B) and no significant increase in stretch at 5, 10, and 25 mmHg (C, P = 0.706). Stiffness remains unchanged in both groups (D). N = 6.
The MMP-2 inhibitor ARP100 blocks the increase in mechanical compliance driven by compound 48/80. (A) Representative pressure-volume trace of ex vivo filling of C57Bl/6 mouse bladders in presence of the selective MMP-2 inhibitor ARP100 (200 nM) alone and after addition of the Mrgprb2 receptor agonist compound 48/80 (10 µg/mL). ARP100 blocks the increase in compliance caused by compound 48/80, signified by the absence of rightward shift of stress-stretch curve (B) and no significant increase in stretch at 5, 10, and 25 mmHg (C, P = 0.093). Stiffness remains unchanged in both groups (D). N = 6.
MMP Inhibition does not affect bladder transient pressure events
Transient pressure events caused by localized contractions of the bladder wall are a major driver of sensory outflow during bladder filling29. The rate of rise of these events positively correlates to the frequency of afferent nerve firing. Using the pressure recordings from our ex vivo experiments, we analyzed the amplitude and leading slope of these events in the absence or presence of MMP inhibitors and compound 48/80. Both the increase in amplitude and leading slope of transient pressure events caused by compound 48/80 persisted in the presence of doxycycline (Fig. 6A–C) and ARP100 (Fig. 6D–F). This suggests that while compound 48/80 activates MMP-2 to degrade collagen in the bladder ECM and increase mechanical compliance, the effects of compound 48/80 on spontaneous detrusor activity occur due to other parallel mechanisms.
Compound 48/80 increases in the amplitude and leading slope of transient pressure events independent of MMP inhibition. (A) Representative traces of transient pressure events in presence of the MMP inhibitor doxycycline alone and after addition of the Mrgprb2 receptor agonist compound 48/80 (10 µg/mL). The increase in peak amplitude (B) and leading slope (C) were unaffected by doxycycline (P = 0.0002 for peak amplitude and P = 0.003 for leading slope). (D–F) The selective MMP-2 inhibitor ARP100 also had no effect (P = 0.010 for peak amplitude and P = 0.0004 for leading slope). *P ≤ 0.05. **P ≤ 0.01. N = 6.
MMP-2 is basally active in the bladder wall
MMP-1 is the primary collagenase that is expressed in the bladder to denature fibrillar collagens (type I and III). MMP-2 and MMP-9 also break down collagen type I along with their canonical substrates collagen type II and IV6. Thus, the enzymatic activity of MMP-1, −2, and − 9 was assessed using gelatin and collagen zymography (Fig. 7). Gelatin zymography showed that MMP-2 was basally active in all tissues, as shown by the light bands at a similar molecular weight as recombinant MMP-2 (Fig. 7A). Additionally, MMP-2 activity was significantly increased in samples exposed to compound 48/80 prior to protein isolation (Fig. 7B). Collagen zymography showed MMP activity at bands of a distinct molecular weight from recombinant MMP-1 but similar in weight to MMP-2 (Fig. 7C). As with gelatin zymography, compound 48/80 significantly increased MMP activity in collagen zymography (Fig. 7D). Together, these data suggest that MMP-2 is basally active in the bladder and this activity can be augmented by compound 48/80.
Compound 48/80 increases gelatinolytic and collagenolytic activity of MMP-2. (A) Gelatin zymograms showing MMP-2 activity of whole bladder protein exposed to vehicle or 10 µg/mL of compound 48/80 prior to protein isolation. Recombinant MMP-2 and MMP-9 were used as positive controls. MMP-2 active band is seen at 68 kDa. (B) Compound 48/80 significantly increases MMP-2 activity (P = 0.0208, N = 6). (C) Collagen zymograms showing MMP-2 activity of whole bladder protein exposed to vehicle or 10 µg/mL of compound 48/80 prior to protein isolation. Recombinant MMP-1 was used as a positive control. (D), Compound 48/80 increased MMP activity with a band at a molecular weight consistent with MMP-2 but different than MMP-1 (P = 0.002) *P ≤ 0.05. N = 5. Annotated images of entire gels are included in Supplemental Materials.
Compound 48/80 significantly reduces TIMP activity
MMPs are endogenously regulated by their inhibitors, tissue inhibitors of metalloproteases (TIMPs) with TIMP-2 primarily responsible for inhibiting MMP-230,31. Thus, we explored compound 48/80’s effects on TIMP-2-mediated inhibition of MMP-2 activity using reverse zymography (Fig. 8). In samples exposed to compound 48/80 prior to protein isolation, TIMP-2-mediated inhibition of MMP-2 activity was significantly reduced (Fig. 8B). These data suggest that the effects of compound 48/80 on bladder compliance are due to a cumulative effect of increased MMP-2 activation and a relief of MMP-2 inhibition by TIMP-2.
Compound 48/80 decreases TIMP-2 activity. (A) Reverse zymograms containing 0.3 mg/mL collagen type I and III and 5 ng MMP-2 as substrate to assess activity of TIMP-2 in whole bladder proteins exposed to either vehicle or 10 µg/mL compound 48/80 prior to protein isolation. TIMP-2 activity was observed at 24 kDa (arrow). (B) Compound 48/80 significantly reduced TIMP-2 activity thereby relieving the inhibition of MMP-2 (P = 0.043). *P ≤ 0.05. N = 6. Annotated images of entire gels are included in Supplemental Materials.
Discussion
In this study, we used multi-photon microscopy, biomechanical measurements, and zymography to explore the mechanisms by which compound 48/80 increases mechanical compliance of the bladder. We discovered that compound 48/80 administered intravesically reduces the thickness of bladder wall at constant pressure but without increasing urothelial permeability. While MMP inhibition (specifically MMP-2) was able to prevent the change in compliance caused by compound 48/80, the effects on bladder smooth muscle excitability were unchanged. Furthermore, compound 48/80 can both activate MMP-2 and relieve MMP-2 inhibition by TIMP-2. In all, these data suggest that compound 48/80 alters the MMP-2/TIMP-2 balance to cause rapid remodeling of bladder wall without affecting urothelial permeability.
Collagen degradation and bladder wall biomechanics
The deposition, degradation, and crosslinking of ECM components contribute greatly to the stiffness, dimensionality, and geometry of the extracellular space32. As such, ECM remodeling and composition have been examined for associations with lower urinary tract symptoms (LUTS). Previous studies utilized fixed tissue samples of urinary bladder to evaluate changes in collagen expression and arrangement over periods of days to months33. However, little is known about moment-to-moment collagen turnover in healthy bladder tissue. Our previous and current work discovered that bladder remodeling can happen in minutes to hours, likely due to the large amount of MMPs present in the bladder wall25.
Though assumed to be a process that takes much longer, the bladder is not alone in its ability rearrange collagen fibers quickly. In the vasculature, collagen fibers are degraded and synthesized continuously24. The aggregation of existing soluble collagens into collagen fibers takes as little as 20 min and new procollagen can be synthesized in as little as 1–2 h34. Since blood vessels show a similar ability to distend and respond to mechanical stimuli, similar rates of turnover and synthesis are likely occurring in the bladder. Our study supports this assumption: we noted increased MMP-2 activity, decreased TIMP-2 activity, increased mechanical compliance, and rapid thinning of the bladder wall after only a ~ 10 min incubation with compound 48/80.
The reduction of bladder wall thickness could arise for one of two reasons: (1) degradation leading to a loss of total collagen; or (2) loss of collagen fiber interconnectivity. Both scenarios would increase compliance, thereby aligning with our findings. While our current results cannot differentiate between these two possible mechanisms, our findings do show that changes in compliance are largely due to changes in stretch and not stiffness. This implies that the interconnections between ECM proteins are altered but the mechanical properties of the constituent elements of bladder wall remain the same. Thus, short-term ECM dynamics in the bladder have more to do with changing how components interact with one another than the total loss or gain of collagen.
MMPs and timps in the bladder wall
MMPs have been extensively studied as regulators of ECM turnover in physiological states of other soft tissues, but studies exploring their pathological role in the bladder are largely limited to urothelial cancer35. Our study is one of the first that outlines the pathophysiological role of rapid activation of bladder MMPs and their ability to alter the biomechanical properties of the bladder wall to affect function. Additionally, our study also shows both constitutive activity of MMPs (specifically MMP-2) as well as regulation of MMP-2 by TIMPs.
During homeostasis, TIMP activity is inversely related to MMP activity to maintain the balance of ECM components31. Furthermore, spatiotemporal aspects of MMP-TIMP interactions play an important role in their activity since most MMPs are membrane bound15,36,37 with TIMPs in a 1:1 stoichiometric ratio38. Unfortunately, zymography assays cannot assess the spatiotemporal characteristics of MMP-TIMP interactions because TIMPs dissociate from MMPs during electrophoresis. As such, the MMP activity noted in our experiments may be an over-estimation of in vivo activity and instead represent “potential” activity. Even so, MMP activity was significantly increased by compound 48/80 with a parallel decrease in TIMP-mediated inhibition along with the inhibition of mechanical compliance increase in our biomechanical assays. It is also likely that these differences would be even more pronounced if better spatiotemporal analyses of MMP-2/TIMP-2 interactions were possible.
Bladder remodeling and its effects on permeability
What is most surprising about our findings is that compound 48/80 drives bladder wall remodeling without affecting urothelial permeability. In animal models, bladder pathologies are often initiated by intravesical infusion of chemicals that specifically degrade the urothelial barrier (e.g., protamine sulfate and acetic acid). The end result is inflammation, changes in compliance, and symptoms of bladder overactivity39. Our previous work showed that intravesical administration of 50 µg/mL of compound 48/80 was able to cause bladder overactivity in addition to changes in compliance and hyperexcitability25. Intravesical infusion of the same concentration of compound 48/80 had no effect on permeability. Given the relative impermeability of the urothelium to pharmacological agents40it is possible that compound 48/80 is activating a receptor-mediated signaling cascade through receptors expressed on umbrella cells.
Our previous studies support this hypothesis in several ways. First, removal of the urothelium mitigated the effects of compound 48/8026. Second, inhibition of the receptor for compound 48/80 (mas-related G protein receptor b2, (Mrgprb2)) blocked changes in ex vivo compliance25. Lastly, inhibition of purinergic receptors or prostaglandin synthesis, two key pathways activated in urothelial cell signaling, also prevented all the effects of compound 48/80 in the ex vivo bladder25,41. If such a mechanism exists, it would have broad implications for experimental models of interstitial cystitis/bladder pain syndrome, most of which rely heavily on the assumption that dysfunction must be preceded by barrier disruption42. Further experiments will explore the expression of Mrgprb2 receptors throughout the bladder wall and will determine if this orphan receptor may be a key player in transducing signals from within the urine into the bladder wall itself.
Limitations
Our study is not without limitations, many of which are already outlined above. Additionally, this study does not determine if the increase in compliance is due to rearrangement of collagen fibers or loss of collagen fibers, which will be assessed in future imaging experiments. These experiments must be done in live tissue since fixation and embedding techniques change the native arrangement of collagen thereby making it impossible to understand the dynamic nature of collagen cross-linking that affects compliance43. Also, we used transient pressure event leading slope as a surrogate measure of sensory outflow as it directly correlates29. However, we may also have disrupted mechanosensitive elements that are responsible for transducing afferent outflow. Actual measurements of afferent nerve activity are required to determine if bladder overactivity caused by compound 48/80 coincides with increased sensory outflow and not due to rearrangement of the mechanosensory mechanisms present within the bladder wall.
Lastly, we do not know if Mrgprb2 is the receptor by which compound 48/80 initiated its effects in the bladder wall. The pharmacology of this receptor is poor, given its status as an orphan receptor. New knockout mouse models and reporter mice will be used to determine the location of this receptor in the bladder wall and to test if it is the target of intravesical compound 48/80.
Conclusions
In conclusion, compound 48/80 increased bladder wall mechanical compliance and increased the amplitude and leading slope of transient pressure events that drive sensory outflow during filling. Alterations in compliance occur due to collagen remodeling and degradation that are driven by MMP-2 activation and TIMP-2 inhibition. These processes also lead to a reduction of bladder wall thickness. Interestingly, despite this profound remodeling, the urothelial barrier remains intact. Together, these data suggest that the mechanical properties of the bladder wall can be influenced rapidly and profoundly by compound 48/80 in a way that does not cause barrier breakdown or permeabilization of the bladder wall.
Methods
Animal care and use
All animal procedures were approved and carried out in accordance with the relevant guidelines and regulations set forth by the Institutional Animal Care and Use Committee of Michigan State University (NIH Assurance D16-0054) and the Guide for Care and Use of Laboratory Animals. All experiments were conducted in accordance with “Animal Research: Reporting of In Vivo Experiments” (ARRIVE) guidelines. Male C57Bl/6 mice (8–12 weeks old; Jackson Laboratory, Bar Harbor, ME USA; RRID: IMSR_JAX:000664) were housed in a temperature- and humidity-controlled environment with a 12-hour light/dark cycle and ad libitum access to standard chow and water. Mice were euthanized by intraperitoneal injection of pentobarbital (> 150 mg/kg) followed by decapitation prior to all experimental procedures.
Calculating mechanical bladder compliance using pentaplanar reflected image macroscopy (PRIM) system
As previously described25,44bladder wall compliance was quantified as wall stress versus stretch using the measurements obtained via ex vivo filling in the PRIM system. Whole mouse bladders were dissected, placed in ice-cold Ca2+-free HEPES buffer containing [in mM]: NaCl [134], KCl [6], MgCl2 [1.2], HEPES [10], glucose [7] (pH 7.4). Bladders were cleaned of connective tissue and cannulated through the urethra in the PRIM chamber for ex vivo filling. Throughout the experiment, the PRIM chamber was recirculated with bicarbonate buffered physiological salt solution consisting of the following [in mM]: NaCl [119], KCl [4.7], NaHCO3 [24], KH2PO4 [1.2], EDTA [0.023], and glucose [7] (pH 7.4). Buffer was warmed to 37 °C and aerated with biological atmosphere gas (20% O2, 5% CO2, and 75% N2) to maintain pH and tissue oxygenation. Bladders were then filled through the cannula at a rate of 30 µL/min until a maximum pressure of 25 mmHg was reached at which point they were emptied and allowed to rest for 10 min. Each bladder underwent 3 equilibration control fill-empty cycles prior to exposure to vehicle/antagonist. Tissues were then exposed compound 48/80 (10 µg/mL) for 10 min prior to a final fill-empty cycle recording.
Bladder wall thickness and area in empty and full configurations were derived from the PRIM system images using ImageJ (NIH). Bladder was then modelled as an ellipsoid that expands non-linearly with a constant wall volume as described previously44,45. Bladder wall stress and stretch were calculated for each time point during the ex vivo fill. To summarize data from multiple experiments, stress and stretch from all curves of the same group were averaged with reference to pressure using MATLAB R2022a (MathWorks). The pressure-volume curves obtained were used to analyze peak amplitude and leading slope of transient pressure events for all fills.
Bladder protein isolation
Whole mouse bladders were dissected, cut in half transversely from the urethra to the dome, and placed in separate tubes with Ca2+-replete HEPES buffer containing [in mM]: NaCl [134], KCl [6], MgCl2 [1.2], CaCl2 [2], HEPES [10], glucose [7] (pH 7.4). One half of each bladder was exposed to 10 µg/mL compound 48/80 at 37 °C for 30 min; the other was exposed to vehicle (dH2O) under the same conditions. Tissue was then flash frozen in liquid nitrogen and homogenized in NP-40 lysis buffer (125 mM NaCl, 30 mM TrisHCl and 10% NP-40) using a Bead Ruptor (Omni Inc.). Homogenized tissues were then centrifuged for 20 min at 12,000 g and supernatant was collected and stored at −80 °C. Total protein content for each sample was estimated using bicinchoninic acid assay.
Gelatin zymography
Bladder protein isolates (30 µg) were loaded into 1% gelatin zymogram gels (Thermo Fisher) and placed in an electrophoresis mini-gel tank (Invitrogen) filled with SDS-PAGE running buffer. In addition to bladder protein isolates, gels included recombinant MMP-1 (#901-MP), MMP-2 (#924-MP), and MMP-9 (#909-MM, R&D Systems) as positive controls. Gels were run for 2.5 h at 115 V at which point they were moved to renaturing buffer (2.5% Triton X-100) on a rocker for 30 min. Gels were then rinsed with developing buffer [in mM]: ZnCl2 [5], CaCl2 [10], Tris HCl [50], NaCl [50], and 0.2% Brij-35. Gels were next incubated in developing buffer for 30 h at 37 °C. After rinsing with distilled water, gels were stained using Coomassie blue for 1 h and destained for 2 h. Finally, destained gels were imaged using the Licor Odyssey Clx imaging system. MMP activity was visualized as light bands against the dark background.
Collagen zymography
Samples were prepared as described above. To prepare collagen zymogram gels, 0.3 mg/mL Collagen type I and III (#DS-01–0058, Ray Biotech, Georgia, USA) were added to Surecast resolving and stacking buffer packs (Thermo Fisher) according to the recipe in Table 1. Once the gels polymerized, samples were run and gels were developed, incubated, stained, destained, and imaged using the same protocol as for gelatin zymography (above). MMP activity was visualized as light bands against the dark background.
Reverse zymography
Samples were prepared as described above. Polyacrylamide gels were prepared using resolving buffer solution containing collagen types I and III (0.3 mg/mL) and recombinant MMP-2 (5 ng) according to the recipe in Table 1. Gels were run, developed, incubated, stained, and destained as described above. TIMP-dependent inhibition of MMP-2 was visualized as dark bands against a light background.
Ex vivo imaging of bladder wall permeability and thickness
Whole bladders were dissected, cannulated, and mounted in a custom-designed imaging chamber. Ca2+-replete HEPES buffer containing 10 µM Wortmannin was recirculated through the chamber and warmed to 37 °C. Bladders were filled to an intravesical pressure of 25 mmHg with Ca2+ containing HEPES buffer mixed with 4 kDa FITC-dextran (FITC-HEPES) to identify the lumen and additionally assess urothelial permeability. Full thickness Z-stacks (1 μm per slice) were captured at two randomized locations on the surface across the bladder wall using a Zeiss LSM880 multiphoton laser microscope and 40x objective (N.A. 0.8). In addition to FITC signal, second harmonic generation (SHG) of collagen was also recorded for each Z stack and served to demarcate the bladder wall46. Images were taken 10 min after intravesical installation with FITC-HEPES with compound 48/80 (50 µg/mL). As a positive control, 1% Triton X-100 was infused in the bladder at the end of the experiment and the bladder wall was imaged again. For analysis of images obtained via two-photon microscopy, each Z-stack was rotated perpendicularly to avoid the interference of bladder’s curvature for accurate calculation of SHG signal width. To calculate wall thickness, width of the SHG signal was measured based on the number of slices containing SHG signal. To calculate permeabilization, average FITC intensity was recorded inside the lumen and within the wall.
Drugs and chemicals
ARP100 (2-[((1,1’-Biphenyl)−4-ylsulfonyl)-(1-methylethoxy)amino]-N-hydroxyacetamide) was obtained from Tocris (Bristol, UK). Doxycycline, compound 48/80, and all other salts and reagents were obtained from Sigma Aldrich (Cleveland, OH). ARP100 was dissolved in DMSO and compound 48/80 and doxycycline were dissolved in water.
Statistical analysis
For comparison of two samples of equal variance, statistical significance between groups was assessed using two-tailed, paired Student’s t tests (α = 0.05). For analysis of images gathered from two-photon microscopy, 10 random images from each stack belonging to the same group were chosen. All 20 images were averaged and compared with other groups using a Wilcoxon paired sign-rank test (α = 0.05). Calculations were performed using Excel (Microsoft Corporation) or GraphPad Prism (GraphPad Software). Comparisons with P values < 0.05 were considered statistically significant. Exact P values are stated in the figure legends. For clarity, “N” represents the number of animals in each group. Distribution of data is represented as mean ± SEM.
Data availability
The datasets generated and/or analyzed during the current study are not publicly available but are available from the corresponding author on reasonable request.
References
Ewalt, D. H. et al. Is lamina propria matrix responsible for normal bladder compliance? J. Urol. 148, 544–549. https://doi.org/10.1016/s0022-5347(17)36650-8 (1992).
Murakumo, M. et al. Three-Dimensional arrangement of collagen and Elastin fibers in the human urinary bladder: A scanning Electron microscopic study. J. Urol. 154, 251–256. https://doi.org/10.1016/S0022-5347(01)67289-6 (1995).
Parekh, A., Brading, A. & Tomita, T. Studies of longitudinal tissue impedance in various smooth muscles. Prog. Clin. Biol. Res. 327, 375–378 (1990).
Parsons, B., Drake, M., Gammie, A., Fry, C. & Vahabi, B. The validation of a functional, isolated bladder model from a large animal. Front. Pharmacol. 3 https://doi.org/10.3389/fphar.2012.00052 (2012).
Drake, M. J. et al. The potential role of unregulated autonomous bladder micromotions in urinary storage and voiding dysfunction; overactive bladder and detrusor underactivity. BJU Int. 119, 22–29. https://doi.org/10.1111/bju.13598 (2017).
Aitken, K. J. & Bagli, D. J. The bladder extracellular matrix. Part I: architecture, development and disease. Nat. Rev. Urol. 6, 596–611. https://doi.org/10.1038/nrurol.2009.201 (2009).
Kadler, K. E., Hill, A. & Canty-Laird, E. G. Collagen fibrillogenesis: fibronectin, integrins, and minor collagens as organizers and nucleators. Curr. Opin. Cell. Biol. 20, 495–501. https://doi.org/10.1016/j.ceb.2008.06.008 (2008).
Upadhyay, J., Aitken, K. J., Damdar, C., Bolduc, S. & Bagli, D. J. Integrins expressed with bladder extracellular matrix after stretch injury in vivo mediate bladder smooth muscle cell growth in vitro. J. Urol. 169, 750–755. https://doi.org/10.1097/01.ju.0000051682.61041.a5 (2003).
Lu, P., Takai, K., Weaver, V. M. & Werb, Z. Extracellular matrix degradation and remodeling in development and disease. Cold Spring Harb. Perspect. Biol. 3, a005058–a005058. https://doi.org/10.1101/cshperspect.a005058 (2011).
Pompili, S., Latella, G., Gaudio, E., Sferra, R. & Vetuschi, A. The charming world of the extracellular matrix: A dynamic and protective network of the intestinal wall. Front. Med. (Lausanne). 8, 610189. https://doi.org/10.3389/fmed.2021.610189 (2021).
Hynes, R. O. The extracellular matrix: not just pretty fibrils. Science 326, 1216–1219. https://doi.org/10.1126/science.1176009 (2009).
Brunner, A., Tzankov, A. & BMI.S294. The Role of Structural Extracellular Matrix Proteins in Urothelial Bladder Cancer (Review). Biomarker Insights 2, (2007). https://doi.org/10.4137/bmi.s294
Rodriguez Faba, O. et al. Matrix Metalloproteinases and Bladder Cancer: What is New? ISRN Urol 581539 (2012). (2012). https://doi.org/10.5402/2012/581539
McCawley, L. J. & Matrisian, L. M. Matrix metalloproteinases: they’re not just for matrix anymore! Curr. Opin. Cell Biol. 13, 534–540. https://doi.org/10.1016/S0955-0674(00)00248-9 (2001).
Nagase, H., Visse, R. & Murphy, G. Structure and function of matrix metalloproteinases and timps. Cardiovascular. Res. 69, 562–573. https://doi.org/10.1016/j.cardiores.2005.12.002 (2006).
Van Wart, H. E. & Birkedal-Hansen, H. The cysteine switch: a principle of regulation of metalloproteinase activity with potential applicability to the entire matrix metalloproteinase gene family. Proc. Natl. Acad. Sci. U S A. 87, 5578–5582. https://doi.org/10.1073/pnas.87.14.5578 (1990).
Ra, H. J. & Parks, W. C. Control of matrix metalloproteinase catalytic activity. Matrix Biol. 26, 587–596. https://doi.org/10.1016/j.matbio.2007.07.001 (2007).
Hamze, A. B. et al. Constraining specificity in the N-domain of tissue inhibitor of metalloproteinases-1; gelatinase-selective inhibitors. Protein Sci. 16, 1905–1913. https://doi.org/10.1110/ps.072978507 (2007).
Johnson, J. L., Jackson, C. L., Angelini, G. D. & George, S. J. Activation of Matrix-Degrading metalloproteinases by mast cell proteases in atherosclerotic plaques. Arterioscler. Thromb. Vasc. Biol. 18, 1707–1715. https://doi.org/10.1161/01.ATV.18.11.1707 (1998).
Robinson, W. P. et al. 3rd ATP stimulates MMP-2 release from human aortic smooth muscle cells via JNK signaling pathway. Am J Physiol Heart Circ Physiol 290, H1988-1996 (2006). https://doi.org/10.1152/ajpheart.00344.2005
Mott, J. D. & Werb, Z. Regulation of matrix biology by matrix metalloproteinases. Curr. Opin. Cell. Biol. 16, 558–564. https://doi.org/10.1016/j.ceb.2004.07.010 (2004).
Patterson, M. L., Atkinson, S. J., Knäuper, V. & Murphy, G. Specific collagenolysis by gelatinase A, MMP-2, is determined by the hemopexin domain and not the fibronectin-like domain. FEBS Lett. 503, 158–162 (2001).
Hastings, G. W. Cardiovascular Biomaterials (Springer Science & Business Media, 2012).
Wagenseil, J. E. & Mecham, R. P. Vascular extracellular matrix and arterial mechanics. Physiol. Rev. 89, 957–989. https://doi.org/10.1152/physrev.00041.2008 (2009).
Saxena, P. et al. Compound 48/80 increases murine bladder wall compliance independent of mast cells. Sci. Rep. 13, 625. https://doi.org/10.1038/s41598-023-27897-6 (2023).
Jones, B. M., Mingin, G. C. & Tykocki, N. R. The mast cell stimulator compound 48/80 causes urothelium-dependent increases in murine urinary bladder contractility. Am. J. Physiol. Ren. Physiol. 325, F50–F60. https://doi.org/10.1152/ajprenal.00116.2023 (2023).
Sounni, N. E., Paye, A., Host, L. & Noël, A. MT-MMPS as regulators of vessel stability associated with angiogenesis. Front. Pharmacol. 1 https://doi.org/10.3389/fphar.2011.00111 (2011).
Soccal, P. M. et al. Matrix metalloproteinases correlate with alveolar-capillary permeability alteration in lung ischemia-reperfusion injury. Transplantation 70, 998–1005. https://doi.org/10.1097/00007890-200010150-00002 (2000).
Heppner, T. J., Tykocki, N. R., Hill-Eubanks, D. & Nelson, M. T. Transient contractions of urinary bladder smooth muscle are drivers of afferent nerve activity during filling. J. Gen. Physiol. 147, 323–335. https://doi.org/10.1085/jgp.201511550 (2016).
Yang, L., Liu, R., Wang, X. & He, D. Imbalance between matrix metalloproteinase-1 (MMP-1) and tissue inhibitor of metalloproteinase-1 (TIMP-1) contributes to bladder compliance changes in rabbits with partial bladder outlet obstruction (PBOO). BJU Int. 112, E391–397. https://doi.org/10.1111/j.1464-410X.2012.11740.x (2013).
Bernardo, M. M. & Fridman, R. TIMP-2 (tissue inhibitor of metalloproteinase-2) regulates MMP-2 (matrix metalloproteinase-2) activity in the extracellular environment after pro-MMP-2 activation by MT1 (membrane type 1)-MMP. Biochem. J. 374, 739–745. https://doi.org/10.1042/BJ20030557 (2003).
Iskratsch, T., Wolfenson, H. & Sheetz, M. P. Appreciating force and shape-the rise of mechanotransduction in cell biology. Nat. Rev. Mol. Cell. Biol. 15, 825–833. https://doi.org/10.1038/nrm3903 (2014).
Bellucci, C. H. S. et al. Increased detrusor collagen is associated with detrusor overactivity and decreased bladder compliance in men with benign prostatic obstruction. Prostate Int. 5, 70–74. https://doi.org/10.1016/j.prnil.2017.01.008 (2017).
Gelman, R. A., Poppke, D. C. & Piez, K. A. Collagen fibril formation in vitro. The role of the nonhelical terminal regions. J. Biol. Chem. 254, 11741–11745 (1979).
Cabral-Pacheco, G. A. et al. The roles of matrix metalloproteinases and their inhibitors in human diseases. Int. J. Mol. Sci. 21 https://doi.org/10.3390/ijms21249739 (2020).
Kapoor, C. et al. Seesaw of matrix metalloproteinases (MMPs). J. Cancer Res. Ther. 12, 28–35. https://doi.org/10.4103/0973-1482.157337 (2016).
Cui, N., Hu, M. & Khalil, R. A. Biochemical and biological attributes of matrix metalloproteinases. Prog Mol. Biol. Transl Sci. 147, 1–73. https://doi.org/10.1016/bs.pmbts.2017.02.005 (2017).
Jackson, H. W., Defamie, V., Waterhouse, P. & Khokha, R. TIMPs: versatile extracellular regulators in cancer. Nat. Rev. Cancer. 17, 38–53. https://doi.org/10.1038/nrc.2016.115 (2017).
Grundy, L. et al. Experimentally induced bladder permeability evokes bladder afferent hypersensitivity in the absence of inflammation. Front. Neurosci. 14, 590871. https://doi.org/10.3389/fnins.2020.590871 (2020).
GuhaSarkar, S. & Banerjee, R. Intravesical drug delivery: challenges, current status, opportunities and novel strategies. J. Control Release. 148, 147–159. https://doi.org/10.1016/j.jconrel.2010.08.031 (2010).
Khandelwal, P., Abraham, S. N. & Apodaca, G. Cell biology and physiology of the Uroepithelium. Am. J. Physiol. Ren. Physiol. 297, F1477–1501. https://doi.org/10.1152/ajprenal.00327.2009 (2009).
Jafari, N. V. & Rohn, J. L. The urothelium: a multi-faceted barrier against a harsh environment. Mucosal Immunol. 15, 1127–1142. https://doi.org/10.1038/s41385-022-00565-0 (2022).
Turunen, M. J., Khayyeri, H., Guizar-Sicairos, M. & Isaksson, H. Effects of tissue fixation and dehydration on tendon collagen nanostructure. J. Struct. Biol. 199, 209–215. https://doi.org/10.1016/j.jsb.2017.07.009 (2017).
Hennig, G. et al. Quantifying whole bladder biomechanics using the novel pentaplanar reflected image macroscopy system. Biomech. Model. Mechanobiol. 22, 1685–1695. https://doi.org/10.1007/s10237-023-01727-0 (2023).
Damaser, M. S. & Lehman, S. L. The effect of urinary bladder shape on its mechanics during filling. J. Biomech. 28, 725–732. https://doi.org/10.1016/0021-9290(94)00169-5 (1995).
Fuentes-Corona, C. G. et al. Second harmonic generation signal from type I collagen fibers grown in vitro. Biomed. Opt. Express. 10, 6449–6461. https://doi.org/10.1364/BOE.10.006449 (2019).
Acknowledgements
We would like to extend our gratitude to Dr. William Jackson for his support and guidance throughout the multiphoton microscopy experiments.
Funding
This study is funded by the National Institute of Diabetes and Digestive and Kidney Diseases R01-DK119615, R01-DK135690, and P20-DK127554.
Author information
Authors and Affiliations
Contributions
P.S., G.M., E.F., O.J.V.R., S.R., and N.R.T. participated in research design. P.S., E.F., O.J.V.R., and A.G. conducted experiments. P.S., E.F., E.B., O.J.V.R., S.R., and N.R.T. contributed new reagents or analytic tools. P.S., E.B. and N.R.T. performed data analysis.P.S. and N.R.T. wrote or contributed to the writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
Dr. Tykocki is a paid consultant for Escient Pharmaceuticals, Inc. All other authors have no competing interests to declare.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Saxena, P., Goel, A., Broemer, E. et al. Compound 48/80 increases bladder compliance by activating MMP-2 and inhibiting TIMP-2. Sci Rep 15, 21652 (2025). https://doi.org/10.1038/s41598-025-05521-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-05521-z