Abstract
Acute Myeloid Leukemia (AML) in infants constitutes a rare and biologically distinct subgroup. Hematopoietic stem cell transplantation (HSCT) plays a pivotal role in the treatment of infant AML. However, the source of stem cells remains insufficiently explored. This study aimed to evaluate the clinical outcomes of allogeneic HSCT from different sources. We conducted a single-center retrospective analysis of 27 infants with AML who underwent HSCT. We compared peripheral blood (PB) with/without bone marrow (BM) (Group 1, n = 16) versus umbilical cord blood (UCB) (Group 2, n = 12). There was no significant difference in 3-year overall survival (OS), disease-free survival (DFS), 3-year recurrence rates, or one-year cumulative graft-versus-host disease (GVHD) incidence between two groups (3-year OS: 80.36% for group 1 vs. 91.67% for group 2, p = 0.5474; DFS: 73.66% vs. 69.84%, p = 0.8232; GVHD: 56.25% for group 1 vs. 50.0% for group 2, p = 0.824). The group with pre-transplant minimal residual disease (MRD) status had a lower recurrence rate. This study emphasizes the efficacy of HSCT in the treatment of infant AML, with higher OS rates compared to childhood AML. It also supports UCB as a viable stem cell source. Achieving MRD-negative status before transplantation is crucial for improving post-transplant outcomes.
Similar content being viewed by others
Introduction
Acute myeloid leukemia (AML) marked by the uncontrolled proliferation of immature myeloid cells in the bone marrow and blood, represents a significant challenge in pediatric oncology. Although comprising 20–25% of childhood acute leukemias, AML’s morbidity and mortality surpass those of other leukemias in this demography1,2. Its incidence is notably lower in infants under two years—a subgroup recently delineated as infant AML3,4,5—exhibiting unique clinical and genetic features, including a predisposition to extramedullary disease and a less favorable prognosis compared to older children6,7,8. The vulnerability of infants to treatment-related morbidity, given their developing immune systems and organs, emphasizes the need for precise, age-adjusted therapies.
Treatment modalities for infant AML often parallel those for older pediatric cohorts, despite emerging evidence suggesting infants under one year should be classified as high-risk, necessitating more aggressive treatment regimens3. The absence of infant-specific protocols for AML exacerbates the risk of infection and chemotherapy-induced toxicities, culminating in suboptimal outcomes.
Hematopoietic stem cell transplantation (HSCT) represents a pivotal advancement for high-risk AML, evidenced by international data indicating survival benefits tied to treatment intensification and the efficacy of second-line treatments, notably allogeneic HSCT (alloHSCT)9,10. The integration of HSCT, particularly in first complete remission (CR1), is complex, balancing disease characteristics and treatment response against the backdrop of potential adverse effects. Nevertheless, the strategic use of HSCT in this cohort, as shown in studies like the AIEOP11, points to its efficacy in minimizing recurrence with manageable transplant-related mortality and late effects.
A significant element in HSCT for infant AML is the selection of stem cell source. Umbilical cord stem cells, with their advantages of high pluripotency12, ready availability, high HLA match tolerance13, and lower risk of GVHD14, have become a preferred stem cell source, increasingly used in the treatment of hematologic malignancies. Considering the significant impact of the number of mononuclear cells and CD34 + cells per kilogram of body weight on the success rate of transplantation, umbilical cord blood stem cell transplantation has more advantages in infant AML. This study aims to evaluate the clinical outcomes associated with different sources of alloHSCT, namely peripheral blood (PB) with/without bone marrow (BM) and umbilical cord blood (UCB). Through a single-center retrospective analysis conducted from 2013 to 2022, we compared the treatment-related complications, overall survival, and other critical metrics in a cohort of 27 infants with AML undergoing allo-HSCT using different sources of stem cell. Our findings aim to refine clinical management strategies and inform stem cell source decisions, potentially enhancing treatment efficacy for infant AML.
Materials and methods
Ethical considerations
The study adhered to the Declaration of Helsinki’s principles and received approval from the review board of the Capital Institute of Pediatrics. All guardians provided informed consent, and patient data were anonymized for confidentiality.
Study population and data collection
The patients in this study were screened from the infants diagnosed with AML in our center at the Capital Institute of Pediatrics from November 2013 to April 2022. Criteria for enrollment of patients included infants diagnosed before two years of age who were classified as high-risk (HR) at initial diagnosis, or those who were initially diagnosed as intermediate-risk (IR) followed with persistent MRD-positive status, or those who had relapsed. HSCT was subsequently performed in these patients (n = 27). The IR and HR classification were determined according to the CCLG-AML 2019 protocol, which was based on cytogenetic/molecular characteristics and response to treatment. The MRD-negative status refers to the MRD detected by flow cytometry being lower than 10–4. Persistent MRD-positive refers to MRD remaining positive after at least three chemotherapy courses. Data were retrospectively collected from electronic medical records, covering demographics, disease characteristics, treatment history, HSCT details (type of transplant, conditioning regimen, and stem cell source), laboratory reports, and imaging studies. Cases with incomplete records were excluded.
Definitions and evaluations
Diagnosis of relapsed and refractory AML
AML relapse can be diagnosed on one of the following conditions: 1. Leukemia cells reappear in peripheral blood after complete remission (CR) of AML; 2. ≥ 5% BM blasts after CR; 3. localized leukemic infiltrates in extramedullary sites post-CR. Refractory AML can be diagnosed when: 1. initial treatment failure after two standard therapy courses; 2. relapse within 12 months post-consolidation; 3. relapse after 12 months but unresponsive to conventional chemotherapy; 4. twice or multiple times relapses; 5. persistent extramedullary leukemia15.
Graft-versus-host disease (GVHD) diagnosis
Acute and chronic GVHD were graded by modified Glucksberg16 and NIH consensus17 criteria, respectively.
Engraftment criteria
Successful engraftment included neutrophil engraftment with sustained peripheral blood neutrophil counts > 1.0 × 109/L for three days, and platelet engraftment with independence from platelet transfusion for seven days with counts > 20 × 109/L. Donor cell engraftment was confirmed via DNA fingerprinting and qPCR.
Additional definitions such as overall survival (OS), disease-free survival (DFS), CR, non-response (NR) and MRD were standardized. MRD was assessed using flow cytometry (FCM) and qPCR18, with positivity defined as FCM ≥ 1 × 10–4 and/or mutated genes presence.
Transplantation protocols
PB with/without BM transplantation and UCB transplantation were the two primary HSCT approaches used in this study. The choice of transplant regimen depends on the MRD status of the children before transplantation, the HLA matching degree, and the stem cell source, as shown in Table 1.
Outcome measures
Primary: OS. Secondary: DFS (time from HSCT to relapse, graft failure, or death), transplant-related mortality (death within 100 days post-HSCT), and complications including MRD status, GVHD, and infections.
Data analysis
Patient demographics, disease characteristics, and treatment histories were summarized using descriptive statistics. The Kaplan–Meier method generated survival curves, with group differences assessed via log-rank (Mantel-Cox) tests. Cox proportional hazards regression determined factors influencing OS and EFS. Treatment-related complication rates were calculated as percentages, and group comparisons were made using chi-square tests. Statistical analyses were performed with R software (R 4.1.3) and GraphPad Prism (GraphPad Prism 9.4.0), setting p < 0.05 as the threshold for significance.
Results
Patient characteristics
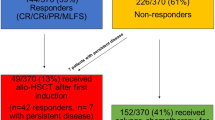
In this study, 27 infants with AML undergoing allogeneic HSCT were evaluated, including 24 high-risk cases, characterized by various genetic anomalies, and 3 intermediate-risk cases.
Specifically, the high-risk group included 7 cases with MLL rearrangements other than t(9;11), 2 cases with t(9;11) accompanied by myeloid sarcoma, 8 cases presenting complex karyotypes, 1 case identified as transformation AML, and 6 cases with MRD > 10% following one induction cycle. Within the cohort, treatment outcomes varied, with 2 achieving second complete remission (CR2), 4 classified as non-responsive (NR; 5% < blasts in bone marrow < 20%), and 4 with MRD-positive status (MRD range: 0.01% to < 5%).
The 27 cases underwent a total of 28 HSCTs (including a case of relapse one year post-haploidentical HSCT, followed by a second UCBT), with 16 utilizing PB ± BM (group 1) and 12 employing UCB (group 2) as the stem cell source. The specific clinical data of the patients are shown in Table 2.
Engraftment outcomes
Among the 16 patients in group 1, all achieved stable engraftment (confirmed by DNA fingerprinting as complete donor chimerism). One patient in group 2 showed mixed chimerism, while the remaining 11 patients achieved stable engraftment. The median time to neutrophil and platelet engraftment in group 1 were 13 days (range: 11–23) and 12 days (range: 8–47), respectively. In group 2, the median time were 13 days (range: 9–18) and 30 days (range: 10–73), respectively (Table 2).
Survival and relapse outcomes
Clinical treatment outcomes of HSCT using PB ± BM vs. UCBT shown in Table 3. Of the 27 patients, 23 survived, with 3 deaths due to relapse and one due to transplantation-related complications. Five patients relapsed, with one patient surviving three months post-relapse under tumor burden, and another achieving 40 months of DFS following a second HSCT.
The 3-year OS and DFS rates were 84.20% and 72.06% (Fig. 1a), respectively, without significant differences between the groups (3-year OS: 80.36% for group 1 vs. 91.67% for group 2, p = 0.5474; DFS: 73.66% vs. 69.84%, p = 0.8232) (Fig. 1b, c). The relapse rates within 3 years were also comparable between the two groups (19.79% vs. 21.43%, p = 0.93) (Fig. 1d).
(a) Survival and relapse rate of the patients. Overall survival rates (b) 3-year OS rates (c) 3-year DFS rates (d) 3-year relapse rates (e) Cumulative incidence of GVHD (f) Cumulative relapse rates of MRD + vs MRD- group (g) 3-year OS rates of MRD + vs MRD- group.
Outcomes of GVHD
In group 1, eight patients (50%) developed acute GVHD (aGVHD), with chronic GVHD (cGVHD) occurring in four cases (25%; 3 mild, 1 moderate). In group 2, six (50%) experienced aGVHD and two (16.67%) were cGVHD (1 mild, 1 moderate), including one death due to aGVHD. The one-year cumulative incidence of GVHD was 56.25% in group 1 and 50.0% in group 2, without a significant difference (p = 0.824) (Fig. 1e).
Infection status
Both groups reported equal cases of sepsis (four cases) and gastrointestinal infections (two).
Group 1 had eight (50%) bacterial infections, while group 2 had seven cases (58.33%). CMV viremia was observed in 9 cases (56.25%) and 5 cases (41.67%) in each group, with group 1 also reporting one case each of CMV retinitis and EBV viremia.
Impact of pre-transplant status on prognosis
Out of 28 transplants, 21 were CR/MRD-negative, 4 NR, and 4 MRD + (0.01% < MRD < 5%). The MRD-positive group showed a significantly higher cumulative relapse rate of 55.0% compared to 6.66% in the MRD-negative group (p = 0.006) (Fig. 1f).
Of the NR patients, one achieved 24-month DFS post-transplant, two relapsed and died, and one converted to MRD-positive status six months following transplantation, surviving with active disease as of this study.
Among MRD-positive patients, two maintained DFS for 74 months, while two relapsed; one of these received a second UCBT and achieved 39-month DFS, and the other survived with disease for 3 months. Only one relapse was observed in the MRD-negative group. The 3-year OS rates for MRD-positive and MRD-negative groups were 72.91% and 88.66%, respectively, without significant difference (p = 0.316) (Fig. 1g).
Discussion
Historically, infant leukemia was a poor-prognosis subtype of pediatric acute leukemias. However, new treatments have improved its prognosis. This improvement has prompted a renewed focus on the efficacy and safety of new treatment strategies for infant AML in China.Data from our center over the last nine years reveal an impressive 3-year OS rate of 84.20% for infant AML treated with HSCT, notably exceeding the 65%-75% OS rates reported for childhood AML10. These outcomes challenge the conventional perception of infant AML’s poor prognosis and underscore the role of effective chemotherapy, meticulous supportive care, and HSCT in potentially enhancing long-term survival for this patient group.
The donor selections for HSCT in infant AML have broadened substantially, including haploidentical and umbilical cord blood stem cells. These developments offer new transplant possibilities, especially when a fully matched donor is unavailable. Our center has adopted these alternative sources, with a focus on haploidentical transplants using PTCy protocols and cord blood transplantation, emphasizing the critical role of GVHD prophylaxis. PTCy selectively targets alloreactive T cells to prevent GVHD while sparing ALDH-rich cells like hematopoietic stem cells and regulatory T cells.This approach reduces transplant rejection and supports immune reconstitution19.
GVHD remains a significant challenge, influencing long-term survival and quality of life post-transplant. Our data indicate that the incidence rates of both aGVHD and cGVHD within the first year for PB ± BM and UCB groups fell within a similar range of 55%-60%. However, cGVHD occurred at rates of 25% and 16.67%, respectively, with most cases classified as mild to moderate—a testament to the efficacy of our GVHD prophylaxis protocols. These rates compare favorably to historical figures reported in the literature, such as the 41.8% incidence of cGVHD (limited-type 20.0%, extensive-type 21.9%) observed with the use of cyclosporine A + MTX/MMF20.
The reduced severity and incidence of GVHD in our cohort are also attributed to the immunological characteristics of haploidentical and cord blood donors. Haploidentical donors, often immediate family members, provide a unique half-matched genetic profile that can be exploited to enhance the graft-versus-leukemia effect21,22,23 while managing the risks associated with GVHD. Cord blood transplants offer the advantage of a naturally occurring lower T-cell count24,25, which may contribute to the observed lower rates and severity of GVHD in our group. Additionally, the high degree of HLA mismatch tolerance inherent to cord blood grafts allows for broader donor selection and has been associated with a reduced incidence of severe GVHD, a factor that has been corroborated by multiple studies26,27.
The median neutrophil engraftment time—a critical marker of initial transplant success—was also identical for both groups at 13 days, indicating a robust bridging to hematopoietic and immune competence. However, a notable disparity was observed in the median platelet engraftment time, with the UCB group experiencing a more protracted recovery (30d vs 12d), highlighting the ongoing need to optimize the UCBT process further. Platelets are critical not only for preventing hemorrhage but also as an integral component of immune surveillance and wound healing. Hence, reducing the time to platelet recovery remains a priority in the CBT field.
Another factor contributing to successful engraftment rates over 90% and the comparable neutrophil engraftment time, particularly in the UCB group, may be the strategic omission of antithymocyte globulin (ATG) and methotrexate (MTX) from the conditioning regimen. ATG is known for its capacity to deplete T cells, which can impact immune reconstitution and diminish the graft-versus-leukemia (GVL) effect28, an essential mechanism by which transplanted immune cells can exert anti-cancer activity. MTX, commonly used for GVHD prophylaxis, carries a risk of inhibiting marrow recovery and contributing to engraftment failure29. By reducing the intensity of immunosuppressive conditioning, we not only preserve the immunological advantages of cord blood but also promote a more favorable environment for immune reconstitution, potentially leading to lower infection rates and improved survival outcomes. Moreover, the preferable outcomes may also be attributed to the relatively young age of the patient population (under 2) and the higher mononuclear cell/recipient weight.
The interplay between MRD status before transplantation and post-transplant outcomes is an area of intense scrutiny in the field of HSCT. Our data underscore the critical importance of MRD status as an indicator of disease burden and predictor of relapse. Notably, in our cohort, the incidence of relapse post-transplantation was significantly lower in patients who were MRD-negative prior to transplantation, with only one out of these patients experiencing a relapse. This contrasts with a 50% relapse rate (four out of eight, two with MRD < 5% and two with MRD > 5%) observed in the MRD-positive group. Besides, three out of the rest four MRD-positive patients had DFS of over 24 months after transplantation, and one had MRD conversion to positive at 6 months post-transplant, but with MRD < 1%. These results suggest that the risk of relapse after transplantation is comparable between patients with MRD < 5% and those with MRD > 5%. Achieving MRD-negative before HSCT or the use of effective tumor-clearing pretransplant conditioning regimens may be effective safeguards for achieving DFS after transplantation.
The findings from our center resonate with the broader literature that highlights the potential of preleukemic stem cells to contribute to relapse post-remission30. The burgeoning field of CAR-T cell therapy as a bridge to HSCT presents new avenues for achieving deeper remission states pre-transplant31. Patients who achieved MRD < 0.01% following CAR-T therapy demonstrated significantly higher DFS rates and cumulative relapse rates compared to those with MRD > 0.01% (70.0 ± 7.2% vs 20.0 ± 12.6%, 70.0 ± 2.6% vs 25.0 ± 0.5%), reinforcing the concept that thorough tumor clearance before transplantation is pivotal for favorable long-term outcomes.
Overall, these findings suggest that UCBT has proved to be a viable alternative to traditional stem cell sources with satisfactory engraftment rates and comparable hematopoietic and immune reconstitution outcomes, as well as a lower risk of GVHD, compared to PB ± BM transplantation. However, the balance between eradication of the disease and minimization of treatment-related toxicity is delicate for the treatment of infant AML. In young patients, the optimal conditioning regimen is one that ensures maximal tumor clearance while preserving the patient’s ability to recover and thrive post-transplant. The pursuit of such regimens, along with the integration of novel therapies, exemplifies the progressive refinement of our therapeutic strategies to improve survival outcomes and minimize relapse in infant AML.
Data availability
All data generated or analysed during this study are included in this published article [and its supplementary information files].
Abbreviations
- AML:
-
Acute myeloid leukemia
- HSCT:
-
Hematopoietic stem cell transplantation
- CR:
-
Complete remission
- PB:
-
Peripheral blood
- BM:
-
Bone marrow
- UCB:
-
Umbilical cord blood
- UCBT:
-
Umbilical cord blood transplantation
- MRD:
-
Minimal residual disease
- MRD − :
-
MRD negative
- MRD + :
-
MRD positive
- GVHD:
-
Graft-versus-Host disease
- aGVHD:
-
Acute GVHD
- cGVHD:
-
Chronic GVHD
- OS:
-
Overall survival
- DFS:
-
Disease-free survival
- NR:
-
Non-response
- PTCy:
-
Post-transplantation cyclophosphamide
- ALHD:
-
Aldehyde dehydrogenase
- MTX:
-
Methotrexate
References
Bolouri, H. et al. The molecular landscape of pediatric acute myeloid leukemia reveals recurrent structural alterations and age-specific mutational interactions. Nat Med. 24(1), 103–112. https://doi.org/10.1038/nm.4439 (2018).
Zwaan, C. M. et al. Collaborative efforts driving progress in pediatric acute myeloid leukemia. J Clin Oncol. 33(27), 2949–2962. https://doi.org/10.1200/jco.2015.62.8289 (2015).
Calvo, C., Fenneteau, O., Leverger, G., Petit, A., Baruchel, A., Méchinaud, F. Infant acute myeloid leukemia: A unique clinical and biological entity. Cancers (Basel). 2021;13(4). https://doi.org/10.3390/cancers13040777
Blais, S. et al. Is acute myeloblastic leukemia in children under 2 years of age a specific entity? A report from the FRENCH ELAM02 study group. Hemasphere. 3(6), e316. https://doi.org/10.1097/hs9.0000000000000316 (2019).
Masetti, R. et al. Acute myeloid leukemia in infants: Biology and treatment. Front Pediatr. 3, 37. https://doi.org/10.3389/fped.2015.00037 (2015).
Rubnitz, J. E. & Inaba, H. Childhood acute myeloid leukaemia. Br J Haematol. 159(3), 259–276. https://doi.org/10.1111/bjh.12040 (2012).
Koren, G. & Schechter, T. Cancer chemotherapy in young children: Challenges and solutions. Pediatr Blood Cancer. 49(7 Suppl), 1091–1092. https://doi.org/10.1002/pbc.21349 (2007).
Creutzig, U. et al. Favorable outcome in infants with AML after intensive first- and second-line treatment: An AML-BFM study group report. Leukemia 26(4), 654–661. https://doi.org/10.1038/leu.2011.267 (2012).
Egan, G., Chopra, Y., Mourad, S., Chiang, K. Y. & Hitzler, J. Treatment of acute myeloid leukemia in children: A practical perspective. Pediatr Blood Cancer. 68(7), e28979. https://doi.org/10.1002/pbc.28979 (2021).
Rasche, M. et al. Successes and challenges in the treatment of pediatric acute myeloid leukemia: a retrospective analysis of the AML-BFM trials from 1987 to 2012. Leukemia 32(10), 2167–2177. https://doi.org/10.1038/s41375-018-0071-7 (2018).
Pession, A. et al. Treatment and long-term results in children with acute myeloid leukaemia treated according to the AIEOP AML protocols. Leukemia 19(12), 2043–2053. https://doi.org/10.1038/sj.leu.2403869 (2005).
Theilgaard-Mönch, K. et al. A comparative study of CD34+ cells, CD34+ subsets, colony forming cells and cobblestone area forming cells in cord blood and bone marrow allografts. Eur J Haematol. 62(3), 174–183. https://doi.org/10.1111/j.1600-0609.1999.tb01741.x (1999).
Barker, J. N., Scaradavou, A. & Stevens, C. E. Combined effect of total nucleated cell dose and HLA match on transplantation outcome in 1061 cord blood recipients with hematologic malignancies. Blood 115(9), 1843–1849. https://doi.org/10.1182/blood-2009-07-231068 (2010).
Locatelli, F. et al. Outcome of patients with hemoglobinopathies given either cord blood or bone marrow transplantation from an HLA-identical sibling. Blood 122(6), 1072–1078. https://doi.org/10.1182/blood-2013-03-489112 (2013).
[Chinese guidelines for the diagnosis and treatment of relapsed/refractory acute myelogenous leukemia (2021)]. Zhonghua Xue Ye Xue Za Zhi. 2021;42(8):624–627. https://doi.org/10.3760/cma.j.issn.0253-2727.2021.08.002
Przepiorka D, Weisdorf D, Martin P, et al. 1994 Consensus conference on acute GVHD grading. Bone Marrow Transplant. 1995;15(6):825–8.
Jagasia MH, Greinix HT, Arora M, et al. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: I. The 2014 Diagnosis and Staging Working Group report. Biol Blood Marrow Transplant. Mar 2015;21(3):389–401. https://doi.org/10.1016/j.bbmt.2014.12.001
Cheng, S. H. et al. Minimal residual disease-based risk stratification in Chinese childhood acute lymphoblastic leukemia by flow cytometry and plasma DNA quantitative polymerase chain reaction. PLoS ONE 8(7), e69467. https://doi.org/10.1371/journal.pone.0069467 (2013).
Shabbir-Moosajee, M., Lombardi, L. & Ciurea, S. O. An overview of conditioning regimens for haploidentical stem cell transplantation with post-transplantation cyclophosphamide. Am J Hematol. 90(6), 541–548. https://doi.org/10.1002/ajh.23995 (2015).
Zhao, X. et al. Effect of chronic graft versus host disease on relapse and survival in patients with acute myeloid leukemia. Zhonghua Xue Ye Xue Za Zhi 36(2), 116–120. https://doi.org/10.3760/cma.j.issn.0253-2727.2015.02.007 (2015).
Debeljak, M. et al. Haplotype counting for sensitive chimerism testing: Potential for early leukemia relapse detection. J Mol Diagn. 19(3), 427–436. https://doi.org/10.1016/j.jmoldx.2017.01.005 (2017).
Villalobos, I. B. et al. Relapse of leukemia with loss of mismatched HLA resulting from uniparental disomy after haploidentical hematopoietic stem cell transplantation. Blood 115(15), 3158–3161. https://doi.org/10.1182/blood-2009-11-254284 (2010).
Vago, L. et al. Loss of mismatched HLA in leukemia after stem-cell transplantation. N Engl J Med. 361(5), 478–488. https://doi.org/10.1056/NEJMoa0811036 (2009).
Salomao, M., Dorritie, K., Mapara, M. Y. & Sepulveda, A. Histopathology of graft-vs-host disease of gastrointestinal tract and liver: An update. Am J Clin Pathol. 145(5), 591–603. https://doi.org/10.1093/ajcp/aqw050 (2016).
Garderet, L. et al. The umbilical cord blood αβ T-cell repertoire: characteristics of a polyclonal and naive but completely formed repertoire. Blood J. Am. Soc. Hematol. 91(1), 340–346 (1998).
Wagner, J. E. et al. Transplantation of unrelated donor umbilical cord blood in 102 patients with malignant and nonmalignant diseases: influence of CD34 cell dose and HLA disparity on treatment-related mortality and survival. Blood 100(5), 1611–1618. https://doi.org/10.1182/blood-2002-01-0294 (2002).
Doan, P. L. & Chao, N. J. Umbilical cord blood: biology and transplantation. Expert Rev Hematol. 2(2), 197–208. https://doi.org/10.1586/ehm.09.9 (2009).
Mohty, M. & Gaugler, B. Advances in umbilical cord transplantation: the role of thymoglobulin/ATG in cord blood transplantation. Best Pract Res Clin Haematol. 23(2), 275–282. https://doi.org/10.1016/j.beha.2010.05.004 (2010).
Terakura, S. et al. GvHD prophylaxis after single-unit reduced intensity conditioning cord blood transplantation in adults with acute leukemia. Bone Marrow Transplant. 52(9), 1261–1267. https://doi.org/10.1038/bmt.2017.116 (2017).
Shlush, L. I. et al. Identification of pre-leukaemic haematopoietic stem cells in acute leukaemia. Nature 506(7488), 328–333. https://doi.org/10.1038/nature13038 (2014).
Yan, M. et al. CAR T-cell bridging to allo-HSCT for relapsed/refractory B-cell acute lymphoblastic leukemia: the follow-up outcomes. Zhonghua Xue Ye Xue Za Zhi 41(9), 710–715. https://doi.org/10.3760/cma.j.issn.0253-2727.2020.09.002 (2020).
Funding
This research is funded by Capital Health Research and Development of Special (2022–1-2091).
Author information
Authors and Affiliations
Contributions
Y.W., Y.G., and R.L.: Conceptualization, Writing—Original Draft. X.L., J.L., T.H., and Z.Z.: Methodology, Software, Data Curation. X.L. and R.L.: Writing—Review & Editing. M.H. and Z.S.: Investigation and Formal analysis. M.Y., R.T., and D.Z.: Visualization. R.L.: Supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, Y., Guo, Y., Ling, X. et al. Clinical outcomes of allogeneic hematopoietic stem cell transplant with different stem cell sources in infant acute myeloid leukemia. Sci Rep 15, 24452 (2025). https://doi.org/10.1038/s41598-025-05602-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-05602-z