Abstract
Amidst growing animal rights movements, the release of captive cetaceans, particularly killer whales and dolphins, into their natural environments has gained increasing support from activists due to ethical concerns. However, there is a notable lack of quantitative studies on the interactions between wild and captive dolphins during rehabilitation before release. This study assesses the rehabilitation process of captive dolphin during its stay in the sea pen using advanced surveillance techniques. Methods including CCTV, hydrophone monitoring, and drone observations were utilized to document interactions between rehabilitating and wild dolphins. A convolutional neural network-based detector for dolphin whistle sound was used to automatically detect the whistle from underwater sound recorded at the rehabilitation site, which significantly expedited the identification of wild dolphin encounters compared to traditional manual methods. This technology also facilitated synchronized analysis of underwater sounds, CCTV footage, and drone videos, providing comprehensive visual and acoustic observations of captive and wild dolphins during encounters. This research offers insights that can guide future dolphin rehabilitation monitoring and release strategies.
Similar content being viewed by others
Introduction
In recent decades, the animal liberation and animal rights movements have revolutionized public perceptions of animals and their treatment, sparking a more thorough examination of the conditions faced by captive animals. This heightened scrutiny has given rise to ethical considerations regarding the practice of confining wild animals. Notably, concerns surrounding cetacean captivity have been extensively documented1,2,3,4,5. Studies suggest cognitive capacities at least comparable to great apes6,7,8,9 featuring traits like self-awareness and abstract thinking10–12. This further amplifies ethical concerns, particularly in the context of captive cetaceans within public display industries. Non-governmental organizations, notably those opposing public display practices, have passionately campaigned for the cessation of captivity and the advocacy for ethical treatment of these beings. Consequently, there is a growing interest, both among the public and the scientific community, in releasing captive cetaceans back into the wild.
A successful release program for captive cetaceans is defined by the ability of the released animals to maintain health, manage stress levels, and exhibit feeding patterns similar to their counterparts in the wild2. It also involves showcasing normal predator avoidance behaviors and ultimately participating in reproductive activities. The rehabilitation process for reintroduction to the wild should aim to eliminate any unnatural behaviors developed in captivity, acquire new skills such as catching live fish in their natural habitat, and readapt to conspecifics in the wild. The duration of rehabilitation period should be flexible depending on the health condition of the dolphin and the progress of rehabilitation process. Captive dolphins and killer whale had occasionally been released from marine parks, but few of these releases had been properly conducted and documented.
A case in Florida in 1990, considered successful, involved the release of two bottlenose dolphins (Tursiops truncatus). However, the experiment was conducted under controlled circumstances with young animals held in captivity for a short duration13. The short-term captivity may not be long enough to eliminate their natural survival skills which most of the long-term captive dolphins are incapable of. In another case, a total of nine bottlenose dolphins were released to local habitat water after ten years of captivity14. Six of them were no longer resighted from the 45th day after release. These dolphins were likely not adopted by the local dolphins and their survival remains unknown. The other three were recaptured and moved to the local aquarium because of severe loss of body weight. Incapable of proper foraging in the habitat most likely the reason for losing weight although they all showed live fish-catching capability during rehabilitation in the sea pen. Keiko, the killer whale that played the role of Willy in the 1993 film Free Willy was released back into the ocean15. Keiko, accustomed to following a caretaker’s boat and engaging in open ocean swims, demonstrated proximity to wild whales, but failed to integrate into a wild group, repeatedly returning to the bay pen and its premature death by Pneumonia was determined at age of 27. Given the highly social nature of cetaceans, conspecific interaction and adoption by the wild group may play a pivotal role in survival after release.
In South Korea, the Indo-Pacific bottlenose dolphin Tursiops aduncus bycatch from local water were sold to the marine parks in 1990. Under the growing pressure from local press and activists, the Korean government classified the bottlenose dolphin as a protected marine species in 2012 and launched the dolphin release project. Subsequently, eight dolphins were reintroduced into their natural habitat Jeju Island from 2013 to 202216–18. Notably, a female dolphin named Sampal emerged as a prime example of successful release, thriving in her natural environment. Sampal’s return underscores the possibility of rehabilitating and releasing marine mammals that have endured prolonged periods of captivity. However, not all released dolphins have followed a similar trajectory, with some remaining unobserved or failing to survive19,20. Despite the successful cases of release, insufficient systemic observation and the absence of available material hinders the quantitative analysis throughout those release cases.
The geographical disparity between Jeju Island and the preceding case holds the potential to enhance the success of a release program in South Korea. The relatively small size of Jeju Island, coupled with the shallowness of the coastal waters, imposes constraints on the long-range travel of released dolphins into deeper sea areas. This restriction consequently amplifies the likelihood of encounters with wild dolphins, given that the indigenous dolphin population is predominantly observed in the vicinity of the island throughout the year21,22. While the heightened frequency of encounters may not necessarily culminate in direct reunions with wild dolphins, it nonetheless proves advantageous in augmenting the adoption rate. Employing quantitative measures to observe wild dolphin encounters throughout the rehabilitation phase enhances our comprehension of the dynamics between wild and captive dolphins. The systematic observation of interactions with wild dolphins during the rehabilitation process emerges as a crucial criterion in determining both the suitable release location and its timing.
The primary objective of this study was to observe the interactions between wild dolphins and a captive dolphin undergoing rehabilitation training for eventual release. Various surveillance methods, including continuous closed-circuit television (CCTV) camera recording, hydrophone monitoring, and opportunistic drone-based observations, were employed to meticulously document every event occurring in the vicinity of the sea pen. Despite the absence of subsequent sightings of the released dolphin after its release, the methodologies outlined in this study for monitoring wild dolphin encounters during rehabilitation have the potential to offer valuable insights for future dolphin release projects.
Results
Underwater sound
A total of 1,124 h of underwater audio recordings (corresponding to 1.45 TB of underwater sound files) were analyzed over the rehabilitation period. Additional 97 h of hydrophone recording during temporal relocation at the aquarium (due to typhoon) were conducted. As shown in Fig. 1, five types of dominant underwater sound including dolphin whistle, broadband signal, pen friction sound, sweep signal, and high-frequency signal were recorded. The ship engine noise and the pinger sound from Sonar, likely from nearby vessel navigation systems, are excluded. The dolphin echolocation clicks and snap shrimp sound were also recorded in audio recording but excluded from analysis. The most frequent underwater sound among the analyzed sound types is the dolphin whistle (Fig. 1a). The broadband signal (Fig. 1b) was expected to be the sound of divers’ rebreather which occurred only when the caretaker boat was docked to the pen. The pen friction sound (Fig. 1c) was thought of as one kind of dolphin vocalization because of its modulated fundamental frequency and consisted of harmonic features. However, the random shape of the fundamental and harmonic components is unlikely to be the vocalization of the dolphin, and confirmed that it was the pen friction sound. The sweep sounds shown in Fig. 1d are expected to be chirp sounds from echosounders or fish finders. Lastly, a high-frequency sound with less modulation compared to the dolphin whistle was captured by the hydrophone (Fig. 1e, f). Different from the mechanical sweep sound as shown in Fig. 1d, the high-frequency sound (Fig. 1e, f) consists of a curved frequency modulation at its beginning or ending section. Unfortunately, we could not identify the source of this sound and it needs further investigation. More examples of sweep sound and high-frequency sound can be seen in Fig.S1.
In addition, the encounter chart of each sound type is illustrated in Fig.S2. The encounter frequency of the sweeping sound (one day) and the pen friction sound (two days) is significantly low compared to the other three sound types. The dolphin whistle has the highest average encounter rate around 10.3 counts per day. The total count and the encounter percentage of daytime and nighttime of each sound type are listed in Table 1. As a result, 63% of the whistles were captured during the daytime. Except for the dolphin whistle, all other sound types were recorded in the daytime. All these sounds are within the hearing range of the dolphin.
A typical example of the five main underwater signal types recorded by hydrophone at a rehabilitation site. (a) the dolphin whistle (b) broadband signal (c) pen friction sound (d) sweep sound (e) and (f) high-frequency sound. Note that a whistle profile can be seen at the bottom of (f).
As shown in Fig. 2a, a total of 15 typical whistle types are visually classified from detected whistles. The count of each whistle type is listed in Fig.S3, and the original whistle spectrogram image of each whistle type is shown in Fig.S4. The percentage of these classified whistles from the whole detected whistles is 31%. The overlapped whistles did not show a clear profile in the spectrogram image and were neglected in the whistle-type classification. A total of 14 signature whistle were identified based on Signature identification (SIGID)23 method, and the distribution of inter-whistle intervals of all whistles and signature whistles is illustrated in Fig. 2b. The whistle type WT10 was not classified as a signature whistle since only two whistles of this pattern were detected, which dissatisfies the criteria of SIGID method. These signature whistle types serve as acoustic identifiers of individual dolphins, indicating that at least 14 different dolphins (each signature whistle is unique to an individual dolphin) were recorded near the rehabilitation pen. Although we could not identify which whistle belonged to which dolphin, this result suggests that the captive dolphin was exposed to a rich wild dolphin vocalization during rehabilitation training.
The dolphin whistle recorded during rehabilitation. (a) A collection of 15 typical whistle types by using visual classification. The start position of the individual whistle profile on the time axis (horizontal axis) was manually adjusted in each type to gain the best alignment. (b) The inter-whistle interval distribution of all whistle type and the signature whistle. A total of 14 signature whistles were identified based on SIGID method (except the WT10).
Daily caretaking duration
The duration of caretaking activity including feeding, watching, maintenance of the net, and replacement of hydrophones was measured by counting the time duration of any ship approaching the pen and leaving the pen on the CCTV footage. Since the rehabilitation pen was located at the position close to the entrance of the Sindo port, unexpected visits of tour boats or passing local fishing ships are also counted. An observer without any knowledge of the caretaking schedule was employed to watch the CCTV footage to identify the caretaker’s ship and count each operation duration. As shown in Fig. 3, the total average caretaking duration and caretaking count, which is the total count of caretaking done in one day, are 38.2 min per day and 1.5 counts per day, respectively. The rehabilitation was split into two periods because of typhon encounter. The first and last days of the two rehabilitation periods were excluded because it required more operation time to set up the pen and transport the dolphin back and forth from the pen. The average caretaking duration in the second rehabilitation period (24.7 min per day) is significantly reduced compared to the first period (44.4 min per day). The average caretaking counts on two rehabilitations are all 1.5 counts per day. The longest caretaking duration was 163 min a day and the most frequent operation count was the 3 counts a day.
The timeline shows the daily caretaking duration and caretaking count during (a) the first and (b) the second rehabilitation period. The caretaking includes feeding, watching, and scuba diving to change the hydrophone. The caretaking count indicates the total count of caretaking on each day.
The spatial distribution of encountered wild dolphins
The location of the rehabilitation pen was selected at one of the sites where the wild dolphin was frequently encountered on Jeju Island. As shown in Fig. 4a, the accumulated spatial distribution of wild dolphins encountered around the pen was derived from the CCTV footage. Two clear pathways of wild dolphin could be observed in Fig. 4a-b. Wild dolphins were observed in the vicinity of the sea pen on 10 days out of the 46-day monitoring period. A total of 287 encounter was assessed from a cumulative observation time of approximately 40.6 min of footage where wild dolphin were visible. The dolphin-to-pen distance was calculated based on these encounter points and illustrated as a histogram in Fig. 4c. Most of the wild dolphin-to-pen distance was concentrated between 20 m and 50 m suggesting a high exposure rate of the captive dolphin to the wild dolphins. Note that the encounter position of the wild dolphin does not necessarily represent the density of individual dolphins encountered around the pen. Multiple encounter positions of the same individual could be presented in this result.
The distribution of the wild dolphin encounter around the rehabilitation pen was captured by CCTV. (a) The distribution of wild dolphin. The MATLAB software (MATLAB version 24.2, The MathWork Inc) was used to align the wild dolphin encounter point and the map image captured from Google Earth (b) The accumulated distribution of wild dolphin (green circles) related to the pen centroid (red triangle) (c) The histogram of the distance between wild dolphin and pen centroid.
Wild dolphin encounter observation via synchronized drone and underwater sound.
Only a single synchronized drone and hydrophone recording of wild dolphin encounter was obtained on 27 Aug 2022, and following analysis refers to this single event. The distance between the wild dolphin and the captive one was derived by tracking the wild dolphin and the pen in a drone video (Fig. 5a, b). The original drone video can be seen in the Supplementary data and website of Korean Ministry of Oceans and Fisheries (https://www.meis.go.kr/mes/dolphinView.do). In the drone video, a group of approximately ten wild dolphins slowly approach the pen from 20 s until 57 s (Fig. 5c − f). During this approach, the wild dolphins were split into a leading group and a following group. The leading group consisted of 5 dolphins who formed a parallel formation (44 s) until they arrived at the closest distance to the captive dolphin (13 m). The following group followed and dived at the 45 s and disappeared from the drone video. Meanwhile, the captive dolphin (inside the pen) perceived the approaching dolphin and turned his head toward the direction of the wild dolphin at 31 and 46 s (Fig. 5d). Later, at 48 s (Fig. 5f), the captive dolphin side slapped his head twice on the water surface to make a big splash. Then the leading group of wild dolphins dived and disappeared from drone video. One of the wild dolphins, which seemed more interested in the captive dolphin was separated from the leading group and reapproached the pen from 71 s and spent some time around the pen. This separated dolphin reunion with the wild dolphins at 120 s and formed a five-dolphin group again. Interestingly, during this encounter only one type of whistle, the whistle type WT04 in Fig. 2, was repeatedly presented (Fig. 5g). More of this whistle can be seen in Fig.S5.
The interaction between wild dolphins and the captive one during rehabilitation was recorded by hydrophone, drone, and CCTV. (a) The trajectory of the pen’s left edge (blue line), the pen’s right edge (green line), and the wild dolphin (red line) over the drone footage took by one of the staff operates the drone for wild dolphin monitoring. (b) The calculated distance between wild dolphins and the centroid of the pen. The distance between the wild dolphin and the pen was determined as null while all the dolphins dived into the water. The snapshot of the drone video on (c) 25 s (d) 31 s (e) 37 s and (f) 48s. (g) The spectrogram of dolphin whistle sound recorded by a hydrophone.
Discussion
Behavioral and acoustic indicators of release readiness
The multi-modal monitoring during rehabilitation revealed quantifiable behavioral and acoustic signs that can inform a dolphin’s readiness for release. Over the 46-day sea pen rehabilitation, wild conspecifics approached the captive dolphin on 10 distinct days (≈ 22% of the monitoring period). These encounters were often brief but notable, totaling ~ 40.6 min of observation with wild dolphins visible on camera. Dolphins frequently came within 20–50 m of the pen, and on at least one occasion a group of about ten wild individuals approached as close as ~ 13 m. Such repeated visits by wild dolphins suggest a level of tolerance or curiosity toward the rehabilitant, providing the social opportunities believed to be crucial for successful reintegration. The rehabilitating dolphin’s responses during these visits – including oriented swimming, rapid turns toward the newcomers, and social signals like tail or flipper slaps – indicate an awareness of and engagement with the wild group. These visual behaviors, combined with vocal activity, are encouraging signs that the animal is re-establishing social interest and competencies. In contrast, during a temporary return to the solitary aquarium environment (due to a typhoon evacuation), the dolphin remained acoustically silent, with no whistles or clicks recorded. This stark absence of vocalization when completely isolated underscores how profoundly social context influences dolphin behavior. Bottlenose dolphins typically increase their signature whistle output when separated from companions as a contact-call to maintain cohesion24. The subject’s silence in isolation (likely a result of long-term deprivation of conspecific contact) may indicate social withdrawal or learned inactivity, which rehabilitation aimed to reverse. Our hydrophone recordings captured a high rate of whistle detections (on average ~ 10.3 whistle events per day) during the pen stay, in stark contrast to the complete quiet in captivity. This resurgence of vocal activity is a positive behavioral indicator: it suggests the dolphin was actively attempting to communicate or respond, an essential step toward re-socialization.
Critically, the signature whistle analysis provides insight into social readiness. Using an identification method based on whistle repetition patterns (SIGID, following Janik et al. 2012), we classified 15 distinct whistle types and identified 14 signature whistle types among them. Signature whistles are unique to individuals and function as social identifiers. Their detection allowed us to infer that at least 14 different wild dolphins interacted acoustically around the pen during rehabilitation. The rehabilitant was therefore exposed to a rich repertoire of wild dolphin calls, likely learning to recognize and respond to them. Notably, two whistle types (tentatively labeled WT01 and WT09) occurred in our recordings without any wild dolphins spotted on CCTV within ± 30 min of those whistles. These two whistle patterns are strong candidates for the captive dolphin’s own signature whistle – in other words, vocalizations produced by the dolphin itself, potentially attempting to hail nearby conspecifics. Dolphins in the wild commonly exchange signature whistles when encountering new individuals as a greeting or identification mechanism24. The emergence of the rehabilitant’s signature whistle during periods of no visible wild presence suggests it was actively calling out, possibly to solicit contact. This behavior marks a significant milestone in rehabilitation: the dolphin shifted from the silence of captivity to reasserting its “name” call, a signal of regained social motivation. In practical terms, the frequency of wild encounters, the duration/proximity of those interactions, and the presence of signature whistle exchanges together form a suite of metrics that reflect social engagement and adaptability. A high encounter rate with wild dolphins (and especially evidence of two-way interaction, such as the rehabilitant orienting to dolphins and producing signature whistles) is interpreted as a positive prognosis for release. These metrics indicate that the dolphin is re-acculturating to life among its wild conspecifics – it is recognizing other dolphins acoustically and visually, and is attempting to communicate. Thus, quantitative thresholds for release can be considered: for example, the individual demonstrating consistent whistle activity and appropriate responses to wild dolphin visits over a sustained period may be deemed ready to rejoin wild society. In contrast, if a rehabilitant fails to whistle or engage despite opportunities, it may signal the need for prolonged rehabilitation or alternative management.
Multi-modal surveillance: benefits and challenges
Our study highlights the value of a multi-modal surveillance approach – combining underwater acoustics, CCTV video, and aerial drones – in monitoring rehabilitation progress. Each modality contributed unique and complementary data. The hydrophone with an automated whistle detector enabled continuous, around-the-clock surveillance of dolphin presence beyond visual range. This proved especially useful for nocturnal or low-visibility conditions when wild dolphins could not be seen. The CNN-based detector significantly expedited encounter detection, automatically flagging whistle events that corresponded to wild dolphin visits, which would have been laborious to find by manual listening. The CCTV cameras, meanwhile, provided spatial and behavioral context to acoustic events. From shore-based video, we mapped wild dolphins’ approach paths and distances around the pen, quantified encounter frequency, and observed surface behaviors. For instance, video evidence allowed us to determine that wild groups often traversed two main pathways near the pen and tended to linger briefly within the 20–50 m range. By synchronizing time stamps, we could correlate these visuals with specific whistle bouts on the audio, revealing cases of both concordance (wild dolphins visible and whistling) and divergence (whistles detected with no dolphins in view, or vice versa). Such cross-verification is invaluable: it guards against false positives from acoustic detection (e.g. distant dolphin whistles not related to an actual approach) and enriches our interpretation of dolphin behavior during encounters. The occasional deployment of drones added a further dimension by capturing a bird’s-eye view of wild dolphin group dynamics around the pen. In the one detailed drone-recorded encounter, we observed coordinated group movements (a parallel formation of five dolphins) as they cautiously approached the pen, split into subgroups, and dove near the enclosure. Simultaneously, we documented the captive dolphin’s real-time reactions to this approach (orienting toward the dolphins, followed by a side flipper slap). Such fine-scale behavioral observations would have been missed by a stationary camera alone. Together, these technologies provided a holistic view of interactions: the acoustic data indicated when dolphins were near (and possibly communicating), the CCTV showed where and how they moved relative to the pen, and the drone footage captured group-level behavior and the context of the broader environment.
Limitations and future directions
While our case study demonstrates the feasibility of using surveillance technology to guide a rehabilitation, we acknowledge several limitations. The sample size is inherently small – focusing on a single individual – so caution is needed in generalizing too broadly. Individual personality and history (e.g. age, prior training) can influence rehabilitation outcomes. Future efforts should compile data across multiple rehabilitation cases to refine the proposed metrics of readiness. Additionally, our monitoring period was relatively short (several weeks) and constrained by external factors (weather forcing a temporary return to the aquarium). A longer acclimation might be beneficial for long-term captives to ensure full recovery of natural behaviors. It remains an open question how much rehabilitation time is “enough” for a given year of captivity; systematic studies varying the duration and intensity of training would be valuable. Another limitation was that our study did not encompass the post-release phase. The ultimate success of rehabilitation is confirmed only if the dolphin survives and integrates after release. Incorporating post-release tracking (via satellite tags, passive acoustic monitoring in the release area, and coordination with local sighting networks) is a vital extension of the protocol. We strongly advocate that future programs plan for long-term follow-up, potentially for years, to document survival, ranging, and social assimilation. This will not only measure success but also provide feedback to improve rehabilitation techniques. Technologically, the next generation of dolphin rehabilitation could benefit from emerging tools such as drone-based photogrammetry (to non-invasively assess body condition over time), AI-driven pattern recognition in videos (for example, to automatically log active vs. resting behavior of the captive), and acoustic AI for identifying not just whistles but also tail slaps or prey capture sounds. Environmental enrichment during pen training, beyond just exposure to wild pods, could also be expanded – e.g. simulating predatory fish presence or introducing objects to stimulate play – to encourage a wider range of natural behaviors. Finally, ethical and welfare considerations must remain central. Rehabilitation protocols should be tailored to minimize stress: even as we introduce challenges (like live fish or decreasing human contact), the animal’s wellbeing should be monitored (through cortisol levels or behavioral indicators of stress) to avoid negative welfare impacts. Balancing the goal of release with the individual’s welfare is paramount; in some cases, an animal might not exhibit the expected readiness signs, and the humane decision could be lifetime sanctuary care instead of release. By transparently acknowledging limitations and iteratively improving methods, the field of marine mammal rehabilitation can move toward evidence-based best practices.
Proposed monitoring protocol for dolphin rehabilitation and release
Drawing on the insights from this study and previous release projects, we propose a structured monitoring protocol to guide future dolphin rehabilitation and release programs. This protocol is designed to ensure comprehensive assessment of a dolphin’s readiness across health, foraging, and social dimensions, using an array of surveillance technologies and predefined criteria:
-
1.
Baseline Assessment and Preparation: Before transfer to a sea pen, thoroughly evaluate the dolphin’s health, diet, and behavior in captivity. Identify or elicit the individual’s signature whistle (e.g. by recording its vocalizations when briefly isolated from any tank mates, or during interactions with trainers) to establish a baseline vocal profile. Simultaneously, conduct field surveys (boat-based or using remote acoustic recorders) in the intended release area to catalog the local wild dolphins’ signature whistles and typical group patterns. This baseline library of calls will serve as a reference to identify known individuals or pods during rehabilitation.
-
2.
Sea Pen Acclimatization with Multi-Modal Monitoring: Place the dolphin in a secure sea pen located within its native range or an area of high wild dolphin presence. Equip the site with synchronized underwater hydrophones (ideally an array of 3 + hydrophones with GPS-synchronized clocks) to continuously record acoustic activity and allow real-time detection and localization of dolphin whistles. Install CCTV cameras covering the pen perimeter (and if feasible, an underwater camera inside the pen) to capture approaching wildlife and the rehabilitant’s behavior. If resources allow, schedule regular drone surveys (e.g. at set times of day when wild dolphins are frequently active) to document broader-scale interactions around the pen. Use automated detectors (for whistles, and optionally for video movement) to log each encounter event for subsequent analysis. Maintain detailed daily logs of the dolphin’s behavior, feeding success with live fish, and any notable events. Ensure the dolphin has started transitioning to live prey and reducing dependency on humans for food prior to sea pen introduction.
-
3.
Quantitative Criteria for Social Engagement: Throughout the rehabilitation, track key metrics of social interaction: frequency of wild dolphin visits (per week), duration of each encounter, proximity of wild dolphins to the pen, and any synchronous behaviors (e.g. the captive and wild dolphins swimming in parallel, or echoing each other’s whistles). Also monitor the rehabilitant’s vocal activity closely. A robust signature whistle exchange − where the captive dolphin produces its signature call and receives responses from wild dolphins − is a strong positive indicator of social reintegration potential24. Conversely, lack of interest from wild groups or persistent silence/withdrawn behavior from the rehabilitant should prompt concern. Set threshold goals for release (for example: wild dolphins’ approach on ≥ X days per month, the dolphin itself initiates whistles or social behaviors in ≥ Y% of encounters, and is successfully hunting live fish).
-
4.
Adaptive Management of Training: Use the monitoring data to adapt the rehabilitation regime in real-time. If the dolphin is not meeting social or foraging milestones, implement corrective actions: e.g. extend the pen training duration, modify feeding strategies, or introduce a companion animal (another compatible dolphin) to stimulate social behavior. The multi-modal surveillance allows caretakers to pinpoint specific deficiencies – for instance, if analysis shows the dolphin hears wild whistles but does not answer, trainers might play back recorded wild whistles to encourage vocal response. If wild visits are infrequent, consider relocating the pen to a busier area or arranging controlled introductions (such as temporarily opening the pen in a supervised trial when wild dolphins are present, if feasible and safe). Throughout, veterinarians should also assess physical condition; improvements in body condition and stamina (which can be observed via drone photogrammetry or routine veterinary exams) should coincide with the behavioral readiness metrics.
-
5.
Pre-Release Evaluation and Stakeholder Review: Convene a review when the dolphin consistently meets the release-readiness criteria. This team (including marine biologists, veterinarians, and if available, experts from previous release programs) should evaluate all data – health records, feeding logs, and the surveillance metrics. If the consensus is that the dolphin can likely thrive in the wild, proceed to plan the release. If not, the review might recommend extended rehabilitation or an alternative long-term care solution. Transparency in this decision process is important for ethical reasons, and the data-driven approach provides justification for the outcome.
-
6.
Release and Post-Release Monitoring: When releasing, do so during a period of suitable weather and when wild conspecifics are nearby, to maximize the chance of the dolphin joining a group. Fit a lightweight satellite25 or radio tracking tag to the dolphin prior to release (ensuring it is well-secured and tested to last several months). Continue acoustic monitoring at the release site if possible (e.g. deploy hydrophones that can detect if the dolphin’s signature whistle is heard back near the pen or along the coast). Coordinate with local networks (whale watch operators, fishers, researchers) to report sightings. In the immediate days post-release, attempt daily visual or aerial tracking to confirm the dolphin’s trajectory and associations26. This is analogous to the contingency planning done for Keiko and others, which proved essential for intervening if things go awry15. Ideally, the rehabilitated dolphin will swiftly integrate with a wild group, as was observed in past successful releases13but if it lingers alone or returns frequently to humans (e.g. approaches boats), managers should be prepared to step in. Post-release monitoring should continue for at least several months, and preferably years, with periodic reports on the dolphin’s status27. Lessons from the post-release phase should be fed back into refining rehabilitation protocols for future cases.
By following this structured protocol, dolphin release programs can maximize the likelihood of a successful transition to the wild. The multi-faceted monitoring not only safeguards the welfare of the individual dolphin by providing continuous oversight, but also builds a rigorous evidence base on which to judge readiness. In summary, our case study demonstrates that integrating acoustic surveillance, video observation, and drone monitoring into a rehabilitation program is a powerful strategy for quantifying a captive dolphin’s progress. Coupling these technologies with established best practices from prior releases (social acclimation, local adaptation, and follow-up) leads to a comprehensive protocol. Such an approach can improve decision-making in future dolphin release efforts, ensuring that when the pen gates open, the dolphin is truly prepared – health-wise, skill-wise, and socially – to embark on a new life in the wild.
Methods
The captive dolphin and release site
A male Indo-Pacific Bottlenose Dolphin (IPBD) called Bibong was illegally captured on the coast of Biyang Island in Jeju (Fig. 6a, b) in 2005. Since then, a 17-year-long captive journey has continued at an aquarium in Jeju until being released into the sea. Before being released to the wild, rehabilitation training was performed on a circular pen (Fig. 6c, d) with an 18-meter diameter located at 33°16’43.04"N,126°10’2.42"E. The release site is in one of the areas of the highest wild dolphin encounter rate around Jeju Island. The rehabilitation training consists of gradual reduction of human interaction and transition of the daily diet from hand-fed dead fish to live fish was applied to help the dolphin gain live fishing catch capability. The rehabilitation process was expected to introduce the experience of real ocean currents and the ambient underwater sound as well as the sound of wild dolphins passing by the pen. Excluding the temporal retreat to the aquarium pool, because of Typhoon Hinnamnor a total of 46 days of rehabilitation training (from 4th Aug. 2022 to 16th Oct. 2022) was performed on the captive dolphin before its release. Since the temporal retreat was took 25 days before moving the dolphin back to sea pen, the entire rehabilitation was divided into two period when analyzing the daily caretaking duration.
The rehabilitation site for the release of the captive dolphin. (a) The map of South Korea (b) The map of Jeju Island (map captured from Google Earth). The place where the dolphin capture (Biyang Island) was marked using yellow arrow. (c) A photograph of a rehabilitation pen captured by a drone (33°16’43.04"N,126°10’2.42"E) (d) An underwater photograph of captive Indo-Pacific Bottlenose dolphin during rehabilitation training. (e) and (f) Two hydrophones attached to the frame of the pen to record underwater sound during rehabilitation (g) The location of the CCTV and the pen marked on the map (h) The photography of the employed CCTV device.
Underwater sound recording and other observation setup
To record the continuous underwater sound during the rehabilitation training, two hydrophones (SoundTrap ST300 and ST400, Ocean Instrument, New Zealand) were alternately mounted on the underwater frame of the pen (Fig. 6e, f). A sampling rate of 192 kHz was used on both hydrophones. Two CCTV devices (XRN-420 S-2T, Hanwha Vision, South Korea) were mounted on the utility poles at the Sindo port which is 300 m apart from the pen (Fig. 6g, h). A zoomed-in view and a wide view were set on two CCTV and operated 24 h to record every event around the pen. A typical snapshot of the CCTV footage of the wild dolphin encountered around the pen can be seen in Fig.S6. A brief search for wild dolphin around the sea pen was scheduled every 2 h by an operator. A search range of 1 km was used and a drone (Phantom 4, DJI, China) was used to capture the video of a wild dolphin whenever the operator encountered with wild dolphin. The underwater sound recording and CCTV recording were conducted throughout the whole rehabilitation process.
Automatic detection of the dolphin whistle on the recorded underwater sound
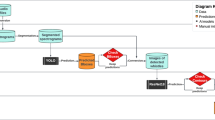
An in-house developed automatic whistle sound detector28 was used to extract the whistle profile and the time information of each whistle sound on a hydrophone-recorded underwater signal. As illustrated in Fig. 7, the original underwater sound recording file (WAV format) was cropped to a 1-second-long processing signal (one-dimensional data) and converted to a spectrogram image (two-dimensional data). Then a convolutional neural network (CNN) based whistle detector28 was used to extract the whistle profile from the spectrogram image. The WAV file of the hydrophone recorded underwater sound has a file name consisting of the information including the serial number of the operated hydrophone and the time when it started the recording. The detected whistle profile was saved as an image file with a file name including the WAV file name and time index to locate the signal within the WAV file. The cropped whistle WAV file was also saved separately for validation use.
The diagram of the detection of the dolphin whistle on hydrophone recorded underwater sound, aka passive acoustic monitoring (PAM). (a) The continuous underwater signal was recorded throughout the whole rehabilitation process. The original underwater signal (b) was trimmed to a 1-second-long signal (c) and converted to a 2D spectrogram image (d) which consists of the time axis in the horizontal axis and frequency axis in the vertical axis. (e) A convolutional neural network-based automatic whistle detector was used to detect whistle profiles in spectrogram images.
The detected whistles were further visually inspected to classify the same whistle profile into same class. Then the inter-whistle interval was calculated to identify the signature whistle based on SIGID method. In this study, the whistle type has a maximum interval between less than 10 s and repeated more than 3 times in a bout was defined as signature whistle.
Deriving the spatial distribution of encountered wild dolphins on CCTV footage
The spatial distribution of encountered wild dolphins near the pen was derived from the CCTV footage, particularly by discerning the dorsal fin. Due to the substantial distance (300 m) between the CCTV camera and the pen, the dorsal fin’s dimensions diminish significantly in the footage, leading to potential confusion with the sea surface. A fuzzy CCTV footage caused by the shaking of utility poles when there is strong wind increases the difficulty of identifying the wild dolphin by only watching the footage. It is also a time-consuming and tedious task to watch every second of CCTV footage to find the wild dolphin. The wild dolphin encounter time from automatic whistle sound detection was used as the reference to find the wild dolphin in CCTV footage. The CCTV footage from 2 min before to 2 min after the whistle sound encounter time was observed to identify the wild dolphin. A successive snapshot of the CCTV screen was taken while the wild dolphin was identified until the wild dolphin disappeared from the CCTV screen. Every snapshot of the CCTV screen was saved as an individual image file with a file name consisting of the time information when it took. Then the location of the wild dolphin and the two edges of the pen (left and right) were manually marked with different colors (Fig. 8a1). The centroid of the pen was derived by calculating the center point of the line connecting the two edges points of the pen, which defined as the X-axis (red line in Fig. 8a1). An additional marking point on the tetrapod was defined and a line connecting this point to the centroid of the pen was defined as the Y-Axis (yellow line in Fig. 8a1). Finally, the position of the wild dolphin related to the centroid of the pen was derived by calculating its distance to the X-axis and Y-axis.
Localization of the wild dolphin in CCTV footage and drone video. (a1) The schematic illustration of landmarks for localizing the location of wild dolphins related to the pen centroid in the CCTV footage. The X-axis was aligned with the line connecting the left and right edge of the pen and the Y-axis was defined by the line connecting the center of the tetrapod and the pen centroid. All the landmarks are manually marked. (a2) The distance from pen to CCTV and the distance from pen to tetrapod are 300 m and 193 m, respectively. The height of the camera was 5.2 m. (b1) The location of the wild dolphin in the drone video, which recorded by one of the staff who operate the drone for wild dolphin monitoring, was manually marked using the red circle on video editing software (Premiere Pro 2020, Adobe). (b2 − b3) The extraction of the center coordinate of the marked red circle in MATLAB. (b4 − b5) The automatic tracking of the left and right edges of the pen is video editing software (After Effect 2020, Adobe). (b6) The identified trajectories of two edges of the pen and wild dolphin from the drone video.
Deriving the distance between wild dolphins and captive dolphins on drone video
A drone video capturing the wild dolphin approaching the rehabilitation pen was analyzed to derive the distance between the wild dolphin and the captive one. To derive the distance, it is required to localize the position of captive dolphins and wild dolphins. First, the wild dolphins were manually marked with a red circle (Fig. 8b1) every 2 s of the video in a video editing software (Premiere Pro 2020, Adobe). Then, the manually marked video was exported as a new video file and the center coordinate of the red circle, which assumed as the position of wild dolphins, was extracted in MATLAB (Fig. 8b2, b3). Two edges of the rehabilitation pen (blue and green rectangular in Fig. 8b4) were tracked using another video editing software (After Effect 2020, Adobe), and the tracked information was exported into a text file (Fig. 8b5). Note that, since the drone video does not provide exact time information, the time information of the drone video was defined by finding the same video frame that captures the same movement of the wild dolphin on CCTV footage. As a result, the combined trajectories of wild dolphins and two edges of the pen was derived as shown in Fig. 8b6. The center point of a line connecting two pen edges was assumed as the position of the captive dolphin. Later, the distance between captive dolphins and wild dolphins, with the pixel as its unit length, was derived by calculating the pixel distance between the center point of the pen and the marked wild dolphin position (Fig.S7a). Since the relative distance between the drone and pen changes through all the video frames, the size of the pen in the drone video also changes and the variation of its horizontal length is shown in Fig.S7b. Finally, the distance between the wild dolphin and captive dolphin, meter as unit length, was then derived by multiplying the pixel distance by unit pixel length which was calculated by dividing the pen diameter (18 m) by the pixel length of the pen (pixel distance between two pen edge as shown in Fig.S7b).
Data availability
The authors declare that the data supporting the findings of this study are available within the article and its Supplementary Information files or are available from the corresponding authors upon request.
References
Peter, S. Animal liberation: A new ethics for our treatment of animals. New York Review, (1975).
Kelly, W. C. S. Ethical Issues in the Release of Animals from Captivity. BioScience 47, 7, (1997). https://doi.org/10.2307/1313022
Hoyt, E. The performing Orca - why the show must stop: an In-depth review of the captive Orca industry. Whale Dolphin Conserv. Society, (1992).
Naomi, A. R. & Parsons The Case Against Marine Mammals in Captivity, 5th edition. Animal Welfare Institute and World Animal Protection, 160 (2019).
Samantha, M. & Chris, Z. Cetacean conservation and the ethics of captivity. Biol. Conserv. 262 https://doi.org/10.1016/j.biocon.2021.109303 (2021).
Butti, C., Sherwood, C. C., Hakeem, A. Y., Allman, J. M. & Hof, P. R. Total number and volume of von economo neurons in the cerebral cortex of cetaceans. J. Comp. Neurol. 515, 243–259. https://doi.org/10.1002/cne.22055 (2009).
Krutzen, M. et al. Cultural transmission of tool use in bottlenose dolphins. Proc. Natl. Acad. Sci. U S A. 102, 8939–8943. https://doi.org/10.1073/pnas.0500232102 (2005).
Krutzen, M. et al. Cultural transmission of tool use by Indo-Pacific bottlenose dolphins (Tursiops sp.) provides access to a novel foraging niche. Proc. Biol. Sci. 281, 20140374. https://doi.org/10.1098/rspb.2014.0374 (2014).
Marino, L. Convergence of complex cognitive abilities in cetaceans and primates. Brain Behav. Evol. 59, 21–32. https://doi.org/10.1159/000063731 (2002).
Reiss, D. & Marino, L. Mirror self-recognition in the bottlenose dolphin: a case of cognitive convergence. Proc. Natl. Acad. Sci. U S A. 98, 5937–5942. https://doi.org/10.1073/pnas.101086398 (2001).
Morrison, R. & Reiss, D. Precocious development of self-awareness in dolphins. PLoS One. 13, e0189813. https://doi.org/10.1371/journal.pone.0189813 (2018).
Kenneth, M. & P., S. Evidence of self-awareness in the bottlenose dolphin (Tursiops truncatus). Cambridge University Press, 18, (1994). https://doi.org/10.1017/CBO9780511565526.026
Wells, R. S., Bassos-Hull, K. & Norris, K. S. Experimental return to the wild of two bottlenose dolphins. Mar. Mamm. Sci. 14, 51–71. https://doi.org/10.1111/j.1748-7692.1998.tb00690.x (1998).
Nick, G., Kelly, W. & Clifford, S. The rehabilitation and release of bottlenose dolphins from Atlantis mark park, Western Australia. Aquat. Mamm. 19 (2), 10 (1993).
Malene, S., Hanson, M. B., Murrey, L. & Jakob, T. From captivity to the wild and back: an attempt to release Keiko from captivity to the wild and back. Mar. Mamm. Sci. 25 (3), 12. https://doi.org/10.1111/j.1748-7692.2009.00287.x (2009).
Ri, P. H. Last captive Dolphin Bibongi to be released to the wild. Korean Ministry of Culture, Sports and Tourism, (2022).
O’Barry, R. RELEASE CASE: SOUTH KOREA. Ric O’Barry’s Dolphin Project (2017).
Kang, G. & Young shin, K. Dolphins returned to the wild. Korean Ministry of Culture, Sports and Tourism, (2017).
Changbin, H. Taesan, a southern bottlenose dolphin released in Jeju, found dead after 7 years (translated title). Headline in Jeju (2022).
Eun-kyung, G. Taesan, a southern bottlenose dolphin, appears to have died seven years after being released into the water (trasnlated title). The Hankook Ilbo (2022).
Seok Gwan, C., Hyun Woo, K., Yong Rock, A. & Kyum Joon, P. Zang geun, K. Coastal resident stock of bottlenose dolphins in the Jeju Islands. Korean J. Fisheries Aquat. Sci. 42 (6), 6 (2009).
Jang, S. Behavioral ecology of Indo-Pacific bottlenose dolphin (Tursiops aduncus) in Jeju Island, South Korea. Thesis paper, Ewha Womans University (2021).
Vincent, M., Janik, S. L., King, Laela, S., Sayigh & Wells, R. S. Identifying signature whistles from recordings of groups of unrestrained bottlenose dolphins (Tursiops truncatus). Mar. Mamm. Sci. 29, 109–122. https://doi.org/10.1111/j.1748-7692.2011.00549.x (2012).
Quick, N. J. & Janik, V. M. Bottlenose dolphins exchange signature whistles when meeting at sea. P Roy Soc. B-Biol Sci. 279, 2539–2545. https://doi.org/10.1098/rspb.2011.2537 (2012).
Dunn, C. et al. Satellite-Linked telemetry study of a rehabilitated and released Atlantic spotted Dolphin in the Bahamas provides insights into broader ranging patterns and conservation needs. Aquat. Mamm. 46, 633–639. https://doi.org/10.1578/Am.46.6.2020.633 (2020).
Moore, R. B. T., Douglas, D. C., Nollens, H. H., Croft, L. & Wells, R. S. Post-Release monitoring of a stranded and rehabilitated Short-Finned pilot Whale (Globicephala macrorhynchus) reveals Current-Assisted travel. Aquat. Mamm. 46, 200–214. https://doi.org/10.1578/Am.46.2.2020.200 (2020).
McHugh, K. A. et al. Staying alive: Long-Term success of bottlenose Dolphin interventions in Southwest Florida. Front Mar. Sci. 7:624729, https://doi.org/10.3389/fmars.2020.624729, (2021).
Changzhu, J., Miyeon, K., Soojin, J. & Dong-Guk, P. Semantic segmentation-based whistle extraction of Indo-Pacific bottlenose Dolphin residing at the Coast of Jeju Island. Ecol. Ind. 137 https://doi.org/10.1016/j.ecolind.2022.108792 (2022).
Acknowledgements
This research was supported by the Basic Science Research Program of the Research Institute for Basic Sciences (RIBS) of Jeju National University through the National Research Foundation of Korea (NRF) funded by the Ministry of Education. (2019R1A6A1A10072987). This research was also supported by Korea Institute of Marine Science & Technology Promotion (KIMST) funded by the Ministry of Oceans and Fisheries (RS-2023-00256122).
Author information
Authors and Affiliations
Contributions
C.K. is involved in methodology, data preprocessing, writing original draft, writing review & editing, and visualization. B-Y.K. is involved in conceptualization, signal acquisition, review & editing. D-G.P. is involved in supervision, review & editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Material 1
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kim, C., Kim, BY. & Paeng, DG. Monitoring of wild and rehabilitating dolphin interactions during rehabilitation period using surveillance technologies. Sci Rep 15, 26161 (2025). https://doi.org/10.1038/s41598-025-06173-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-06173-9