Abstract
The US FDA recommends assessment of abuse liability (AL) for premarket tobacco product applications (PMTAs) to determine whether a new tobacco product is appropriate for the protection of public health (APPH). To assess the AL and nicotine uptake of Vuse Alto electronic nicotine tobacco products (ENDS) which offer e-liquid pods that vary in nicotine concentration, we conducted two clinical laboratory studies. The primary objective of Study 1 was to assess measures of product abuse liability (AL) in real time during and following product use. This was achieved through timed subjective effect questionnaires, physiological measures (blood pressure and heart rate), and pharmacokinetic assessments for Golden Tobacco flavor 2.4% and 5% nicotine concentration Vuse Alto products. Study 2 evaluated PK and Overall Product Liking (OPL) of four Alto flavor variants with 1.8% and 2.4% nicotine concentrations. The studies were designed as open-label, randomized, crossover studies with 9 and 7 days of confinement for Study 1 and Study 2 respectively, were employed to assess nicotine PK, subjective effects, and physiological measures (Study 1 only) for 4 h after a 10-minute ad libitum ENDS product use. Study 1 included high (usual brand [UB] cigarettes) and low abuse liability (nicotine gum) comparators. Nicotine PK of Vuse Alto products were generally similar across the three nicotine concentrations and four flavors tested in these studies. In Study 1, nicotine PK and several subjective effects (product liking, OPL, product positive/negative effects and urge to smoke) for Vuse Alto 2.4% and 5% products were determined to be in between UB cigarettes and nicotine gum. Increases in heart rate, systolic and diastolic blood pressure were observed for both products. In Study 2, nicotine PK and the OPL for Vuse Alto four flavors at 1.8% and Golden Tobacco at 2.4% were similar and comparable to the Alto products tested in Study 1. These results indicate the AL of Vuse Alto, as assessed by nicotine PK and subjective effects measures of Vuse Alto products at three nicotine concentrations and of four flavors, is significantly lower than cigarettes, and higher than nicotine gum. ENDS products deliver nicotine in a manner that satisfies smokers’ preferences while exhibiting lower AL than cigarettes. Compared to pharmaceutical smoking cessation aids, such as nicotine gum, the Vuse Alto products demonstrate nicotine PK profiles characterized by a more rapid onset and higher peak concentrations, with subjective measures such as product liking, fall between those of nicotine replacement therapy gum and combustible cigarettes. Collectively, the AL assessments indicate that Vuse Alto products may support tobacco harm reduction by providing an alternative that may help smokers migrate away from cigarette smoking.
Similar content being viewed by others
Introduction
The 2009 Family Smoking Prevention and Tobacco Control Act has created the Premarket Tobacco Product Application (PMTA) pathway for marketing of new tobacco products that are determined to be appropriate for the protection of public health (APPH) by the FDA1. Considering that combustible cigarettes are the most harmful class of tobacco products, alternative tobacco products that do not generate combustion-related toxicants have been suggested as less harmful alternatives for smokers who choose not to quit smoking as part of a comprehensive harm reduction strategy2,3. While the best option to reduce harm from cigarette smoking is complete cessation4a continuum of risk is recognized wherein the combustible tobacco products (e.g., cigarettes) are the most harmful. Within the risk continuum, non-combustible tobacco products (e.g., e-cigarettes) fall in between the least harmful FDA- approved nicotine products (e.g., gum) and combustible products (e.g., cigarettes)5,6.
Electronic Nicotine Delivery Systems (ENDS) products are an emerging class of alternative, inhaled, non-combustible tobacco products that produce far fewer and significantly lower quantities of harmful and potentially harmful constituents (HPHCs)7. For example, some ENDS products have been shown to contain fewer toxicants8,9 and reduced exposure to HPHCs10,11. Several studies suggest that ENDS can help reduce cigarette consumption and support smoking cessation12,13,14,15,16,17. A recent commentary further articulated evidence on nicotine e-cigarettes as a tool for smoking cessation18. However, some reports indicate that ENDS are only as effective as FDA-approved medicinal products19,20. Thus, ENDS have a potential to reduce the adverse health effects of cigarette smoking21. Recent favorable PMTA decisions, in general, reflect the appropriateness of this product category as APPH22.
ENDS are a heterogeneous category of products that differ in their e-liquid composition (e.g., nicotine concentration and flavors) and product design. Abuse Liability (AL) assessment is primarily focused on the pharmacological effects of the substance or product that may contribute to effects such as reinforcement, pleasure, physical dependence and withdrawal. However, factors such as initiation, patterns of use, and persistence of product use involve many non-pharmacological factors including product form and flavor, which can be important influences on tobacco product use23. It is reported that nicotine delivery and flavors are among the factors that may influence AL and product appeal24. Abuse liability refers to the potential of a substance to result in addiction and be used repeatedly or even sporadically with undesirable effects25. A certain level of AL and product appeal are necessary for adult consumers to substitute and adopt ENDS use when moving away from more harmful cigarette smoking and eventually to smoking cessation3,21,25. Although some previous reports have suggested that flavored ENDS are associated with uptake of ENDS by youth26,27a more recent systematic review summarized evidence demonstrating that e-liquids with higher nicotine concentrations and flavors were helpful in migrating smokers away from cigarette smoking24. Further, two national surveys also suggest that the use of flavored e-cigarettes, particularly menthol, is associated with the chance of quitting smoking successfully28,29.
Determination of AL is among the required assessments for PMTAs30. Abuse liability studies indicate the degree to which users of the tobacco product are likely to use or develop an addiction to the product and whether a given tobacco product is APPH30. Assessments of tobacco product AL typically include nicotine pharmacokinetics (PK), subjective measures of product liking (PL), and physiological measures31,32. The pharmacological profile of nicotine is critical to understanding the behavioral effect of a tobacco product, as it impacts use behavior and AL, with the AL impacted by both nicotine level and speed of absorption33. Subjective responses to questionnaires on PL, intention to use, relief from smoking urges and cravings and physiological responses provide insights into abuse liability31,34,35.
Previous reports indicate that, in general, ENDS exhibit significantly lower AL measures (such as subjective measures) compared to cigarettes but substantially higher than nicotine gum, an FDA-approved nicotine replacement therapy (NRT)36,37. Most recent studies on Vuse Solo ENDS products have shown that their PK and subjective measures were intermediate between usual brand (UB) cigarettes and nicotine gum, irrespective of nicotine concentrations and e-liquid flavors38,39,40,41. Specifically, subjective measures such as PL, intent to use again and positive product effects (PEs) for Vuse Solo ENDS products were lower than UB cigarettes, but similar to nicotine gum38,39.
With advancing product innovations, the ENDS marketplace has evolved considerably from the original cig-a-like systems (resembling cigarettes in shape and size) to more advanced pod systems which come with a more powerful battery, various nicotine concentrations and flavors. The earlier cig-a-like systems and subsequent types of pod system ENDS varied in their nicotine delivery to consumers42,43. The pod systems have the potential to deliver nicotine at levels that match or are closer to those of combustible cigarettes and it is important to assess the AL of this category of ENDS. Vuse Alto is a pod-type family of ENDS products, marketed by R.J. Reynolds Vapor Company. At the time of study conception, conduct and manuscript preparation, Vuse Alto products were commercially available in limited flavors and nicotine concentrations. In this manuscript, as a part of the PMTA process, we investigated nicotine PK and several measures of subjective effects to determine the appropriateness of Vuse Alto products in the context of APPH.
Materials and methods
Study design
Measures associated with the AL profile of Vuse Alto products were assessed at different nicotine concentrations and flavors in two independent clinical studies, termed Study 1 and Study 2 (Supplemental Table S1). Both of these studies were randomized, open-label, crossover in-clinic confinement studies, conducted in accordance with the Declaration of Helsinki and applicable sections of US Code of Federal Regulations and ICH E6 Good Clinical Practice standards. The study design features for Study 1 and Study 2 are graphically presented in Supplementary Fig. 1.
Study 1 (ClinicalTrials.gov registration number; date: NCT 04094363; 18/09/2019) was approved by IntegReview IRB (Austin, TX), and conducted with a confinement period of 9 days at 2 study sites located in the US. Recruitment and execution occurred between 03/10/2019 and 26/11/2019. In addition to the two ENDS products, Study 1 included a high AL comparator (UB cigarette) and a low AL comparator (nicotine gum). Study 1 evaluated nicotine PK over 4 h after the start of the test or comparator product use. In addition, subjective and physiological measures were assessed (Supplemental Table S1). The primary endpoints of Study 1 were PL and Overall Intent to Use Again (OIUA). The secondary endpoints were nicotine uptake parameters (AUC, Cmax and Tmax) and additional subjective measures (Product Effects [PE], Urge to Smoke [UTS], and Overall Product Liking [OPL]). Physiological measures (blood pressure and heart rate) were also included as non-subjective assessments, as indicated in the FDA’s guidance on PMTAs25,44.
Study 2 (ClinicalTrials.gov registration number; date: NCT04751461; 12/02/2021) was approved by Advarra IRB (Columbia, MD), and was conducted at a single study site located in the US. Recruitment and execution occurred between 23/02/2021 and 20/04/2021. Nicotine PK (primary endpoint) and OPL (secondary endpoint) were assessed on five Vuse Alto products. (Supplemental Table S1).
Enrolled subjects in both studies used all study products during Product Acclimation Sessions. The day or days preceding each Test Session, product-specific Acclimation Periods were conducted where subjects used the products assigned to the next Test Session. Following an overnight 12-hour nicotine/tobacco product abstinence, Test Sessions with the assigned products were conducted.
In Study 1, approximately 1.5 days before each Test Session, subjects participated in a product Acclimation Period. The CRO statistician generated the random allocation sequence using a Williams design with four Investigational Products (IPs) and four test sessions. Subjects were then randomized by a block size of 4 to one of the IP sequences (ABCD [12 subjects], BDAC [12 subjects], CADB [13 subjects], and DCBA [13 subjects]) to use one IP in each session. For both primary and secondary endpoint assessments, all 50 randomized subjects were included in the safety analysis population, while 49 subjects were included in the PD (subjective and physiological) and PK analysis populations; one subject was excluded due to non-participation in the ABCD product use sequence. The mechanism used to implement the random allocation sequence was via a central computer and personnel who enrolled and who assigned participants had access to the random allocation sequence. The study was unblinded by necessity due to the very different visual appearances of the study products. Subjects were not given information on the specific Vuse Alto product (flavor or nicotine content) to which they were randomized. Upon randomization, the subjects were instructed to use their randomized product a minimum of six times (two times on the first 0.5 day and four times on the second day) to provide familiarity with each product (except for UB cigarette) prior to the corresponding clinical Test Session. In each AL Test Session, subjects used the assigned product, administered by trained site personnel, that was either a Vuse Alto product for 10 min ad libitum, high AL comparator (UB cigarette) ad libitum until completion or until 10 min, whichever came first, or low AL comparator (nicotine gum) for approximately 30 min as per product insert. AL-related subjective measures, physiological measures, and blood samples for nicotine uptake measurements were collected during and after product use over a 4-hour sampling period.
For Study 2, starting on Day 1 and through Day 2, subjects were required to use each of the five Vuse Alto products at least once for product acclimation during the pre-study product Acclimation Period. The CRO statistician generated the random allocation sequence using a Williams design with five IPs and five test sessions. Subjects were then randomized by a block size of 3 to one of the IP sequences, (sequence[subjects]): (ABECD [3], BCADE [3], CDBEA [4], DECAB [3], EADBC [3], DCEBA [3], EDACB [4], AEBDC [3], BACED [3], and CBDAE [4]) to use one IP in each session. All 33 randomized subjects were included in the safety, PK and the OPL analysis populations for both primary and secondary endpoint measurements. The mechanism used to implement the random allocation sequence was via a central computer and personnel who enrolled and who assigned participants had access to the random allocation sequence. The study was unblinded by necessity due to the distinct visual differences between the study products. However, subjects were not informed of the specific Vuse Alto product to which they were randomized, including flavor or nicotine content. For the remaining study days, Days 3 through Day 7, study procedures included a Test Session which began each morning, followed by an afternoon product Acclimation Period (excluding Day 7 in which subjects were released from the study). Over Day 3 through Day 7, subjects participated in five separate daily Test Sessions for nicotine PK and OPL assessments, one Test Session per day. For the Test Sessions, trained site personnel administered subjects their assigned Vuse Alto product for 10 min of ad libitum use. Blood samples for nicotine PK assessments were drawn and subjective measures for OPL were collected during and after product use for 4 h. Following each Test Session, subjects were required to use the assigned Vuse Alto product to be tested the next day at least twice during daily product Acclimation Periods. During the product Acclimation Periods, subjects also had access to their UB cigarettes for ad libitum use.
Subjects
Generally healthy adult (21 to 60 years of age) males and females who primarily smoked on average at least 10 filtered menthol or non-menthol cigarettes per day for at least 6 months were recruited. At Screening, subjects who responded to Fagerström Test for Nicotine Dependence (FTND) Question 2 (“How soon after you wake up do you smoke your first cigarette?”) with either “Within 5 minutes” or “6–30 minutes” were accepted into the study. Females who had a positive pregnancy test, were pregnant, breastfeeding, or intended to become pregnant during the course of the study were excluded.
Test products
Vuse Alto is an ENDS consisting of closed e-liquid cartridges (also referred to as pods) that work in combination with a non-adjustable power unit with a rechargeable battery (typical capacity 370 mAh). The Vuse Alto pods are non-refillable and contain approximately 1.8 mL of e-liquids of varying flavors and salt-based nicotine contents of 1.8%, 2.4% and 5.0% by weight.
The ENDS products in the Study 1 were two Vuse Alto Golden Tobacco flavor products at 2.4% and 5% nicotine concentrations. Study 1 also included a high AL (subject’s UB cigarette) comparator product and a low AL (nicotine gum [Nicorette® White Ice Mint, 4 mg nicotine]) comparator product (Supplemental Table S1).
The five test products included in Study 2 were Vuse Alto Rich Tobacco, Golden Tobacco, Menthol and Mixed Berry flavor variants at 1.8% nicotine concentration as well as Golden Tobacco at 2.4% nicotine concentration (Supplemental Table S1).
PK measurements
The following nicotine PK parameters were evaluated in both Study 1 and Study 2: overall nicotine uptake over 4 h (AUC0 − 240; area under the nicotine concentration-time curve from zero to 240 min after the start of product use), the maximum plasma nicotine concentration (Cmax), area under the nicotine concentration-time curve from time zero to 15 min after the start of product use (AUC0 − 15), and time to reach Cmax (Tmax). Blood samples for assessment of nicotine PK parameters were collected during Test Sessions at various timepoints from 5 min prior (-5) to 4 h after the start of product use. Blood sampling times coincided with select subjective and physiological assessments as described below (Subjective measures).
Subjective measures
Study 1 evaluated 5 questionnaires, each with a single question, on a numerical rating scale, ranging from 0–10 39–41. The PL (“At this moment, how much do you like the product?”), UTS (“How strong is your current urge to smoke your usual brand cigarette?”) and PE were assessed several times, coinciding with the blood draws, over 4 hours after the start of test product use; the UTS was also assessed 5 min before product use. Product effects assessments included Positive PE (“Rate the degree to which you feel positive effects of the product at this moment”) and Negative PE (“Rate the degree to which you feel negative effects of the product at this moment”). The OIUA (Rate the degree to which you would like to use the product”) and the OPL (“Overall, how much did you like the product?”) were assessed once, at the end of the Test Session, i.e., 4 h post product use. On these questionnaires, lower numerical ratings indicated minimal or no effect, whereas higher numerical ratings represented strong effects.
The AUECPL 3−240, and Emax PL were computed from the responses to PL questionnaire over the assessment time course, whereas the Eoverall IUA was derived from the responses to single OIUA questionnaire. The measures of positive PE (AUECPEpos 3−240, Emax PEpos) and negative PE (AUECPEneg 3−240 and Emax PEneg) were calculated from the responses to positive PE and negative PE questionnaires, respectively. The Emin UTS, Tmin UTS, AUECUTS 0−15, AUECUTS 0−240 were calculated from the responses to the UTS questionnaire.
The Eoverall PL was calculated from the response to the single OPL questionnaire.
Study 2 evaluated OPL as a single subjective measure.
Physiological measures
In Study 1, physiological measures (blood pressure [systolic and diastolic] and heart rate) were measured starting at 5 min prior to product use (baseline) and at various timepoints following use of the product through to the end of each Test Session (at 240 min).
Safety assessment
Safety was assessed and monitored throughout study duration, starting with subject screening assessments including vital signs, hematology, complete metabolic panel, ECG, serology, urine drug screen, serum and urine pregnancy testing, physical and oral examinations, collection and assessment of concomitant medications and medical history. During clinical conduct, subjects were assessed daily for adverse events (AEs) and with symptom-driven physical or oral examinations.
Statistical analysis
For Study 1, sample size was estimated based on data from two previous Vuse ENDS AL studies38,39. Based on those data and requirements of the randomization method (i.e., Williams Design), a minimum of 36 subjects was needed to have at least 80% power for detecting the hypothetical differences in each primary endpoint between each Vuse Alto product and UB cigarette at a two-sided 0.0042 significance level (Bonferroni-adjusted for multiple comparisons). The target number of subjects randomized for this study was 50 subjects to allow an approximate 15% dropout rate with a goal to ensure 36 subjects completed all Test Sessions. Statistical significance of p ≤ 0.05 was applied for all the other endpoints.
Subjective effects and PK parameters were calculated using a SAS® program which follows the same algorithm of noncompartmental analysis (NCA) as that implemented in Phoenix WinNonlin (Version 8.0.0.3176 or higher; Certara LP, Princeton, NJ USA).
Statistical analyses were carried out to test the null hypotheses that there were no significant differences between each Vuse Alto Golden Tobacco product (2.4% and 5.0%) and each of the high- and low-AL comparator for the primary and secondary endpoints. It is important to note that the studies were not designed to make comparisons between the Vuse Alto products.
For Study 2, data from a prior study was used as an estimate to obtain 30 subjects as the minimum number of subjects needed to have at least 80% chance of obtaining 95% confidence interval with half width up to 20% of the means for the primary endpoints46. The target number of subjects to be randomized for this study was 36 subjects to allow an approximately 15% dropout rate with a goal of ensuring 30 subjects complete all Test Sessions with evaluable data.
Subjective effects and PK parameters were calculated using a SAS® program which follow the same algorithm of NCA as that implemented in WinNonlin® Professional (Version 8.0, Pharsight Corporation, A Certara Company, St. Louis, MO). SAS results were validated using the parameters generated from WinNonlin 8.0. The PK and OPL for all five test products were summarized using descriptives statistics. No statistical comparisons were performed among these groups.
For both studies, AUC0 − 240 was not calculated for a subject if the first or last nicotine concentration of the interval was missing, or those with baseline plasma nicotine concentrations > 4 ng/mL as this suggests they may have used a nicotine-containing product during the abstinence period, thus biasing their assessment of subjective measures. In addition, subjects with a Tmax ≥ 120 min were also considered to have used other nicotine-containing products during the Test Session and were excluded from PK analysis. Missing data were considered “missing at random” and no data imputation was performed. Data were screened for potential extreme values.
Results
Demographics
In Study 1, 50 subjects were enrolled and randomized; 48 completed the study, while two subjects withdrew consent and were subsequently discontinued (see Supplementary Fig. 2). In Study 2, 33 subjects were enrolled and randomized; 32 completed the study, and one subject was early-terminated by the Sponsor (see Supplementary Fig. 2).
Study 1 evaluated two nicotine concentrations (2.4% and 5%) of Vuse Alto Golden Tobacco flavor products and compared the PK and subjective measures to a high- (UB cigarette) and a low- (nicotine gum) AL product (Supplemental Table S1). Study 1 population consisted of approximately 54% male subjects, and the majority were White (80%). None of the subjects were of Hispanic or Latino ethnicity. The mean age was 40.4 years, with a range of 22 to 60 years. The mean BMI was 30.4 kg/m2 (range of 19.8 to 48.5 kg/m2). Subjects reported consuming a mean of 17 cigarettes per day. The mean duration of smoking history was approximately 24 years (Supplemental Table S2). Two subjects withdrew from the study early on; one subject withdrew consent from study participation, and the other withdrew from the study due to a family emergency.
Study 2 evaluated five Vuse Alto products: Rich Tobacco, Golden Tobacco, Menthol and Mixed Berry, each at 1.8% nicotine concentration as well as Golden Tobacco at 2.4% nicotine concentration (Supplemental Table S1). Study 2 population consisted of approximately 63.6% male subjects, and the majority were White (82%). None were of Hispanic or Latino ethnicity. The mean age was 42.4 years, with a range of 26 to 58 years. The mean BMI was 31.34 kg/m2 (range of 17.8 to 46.7 kg/m2). Subjects reported consuming a mean of 17.8 cigarettes per day. The mean duration of smoking was approximately 24.9 years. Approximately 63.6% of subjects reported smoking non-menthol cigarettes, and approximately 36.4% reported smoking menthol cigarettes (Supplemental Table S2).
Study 1, nicotine pharmacokinetics
For Study 1, parameters of nicotine uptake were highest and most rapid for the UB cigarette, while these parameters were lowest for nicotine gum (Fig. 1; Table 1). The Cmax was 15.3 and 3.46 ng/ml for UB cigarette and nicotine gum, respectively. Nicotine uptake (AUC) at 15 and 240 min for UB cigarette was 147 and 1058 ng x min/mL, respectively, whereas for nicotine gum, it was 4.79 and 464 ng x min/mL, respectively. The Tmax for UB cigarette was 8 min whereas for nicotine gum it was 45 min, as consistent with the use period for these products.
Baseline adjusted mean plasma nicotine concentration profiles of Vuse Alto Golden Tobacco 2.4% and 5%, UB filtered cigarette, and nicotine gum for 4 h following start of product use. UB = usual brand. n = 44 for the two Alto Golden Tobacco groups, and n = 46 for the UB cigarette and nicotine groups.
Nicotine uptake over 15 and 240 min for the ENDS products were significantly lower compared to UB cigarette, and generally higher compared to nicotine gum. All PK parameters (Cmax [9.08 and 9.04 ng/ml], AUC at 15 [80.6 and 85.4 min x ng/ml] and 240 min [631 and 678 min x ng/ml], and Tmax [10.5 and 10.7 min]) were similar for the Vuse Alto 2.4% and 5% nicotine concentration products, respectively.
The least squares (LS) mean difference comparisons between the test and reference products were statistically significant (Fig. 1; Table 1).
Study 1, subjective effects
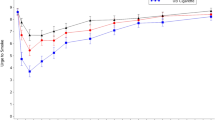
The primary endpoints of Study 1 were product liking (PL) and overall intent to use again (OIUA) (Fig. 2; Supplemental Table S3). Additional endpoints including positive and negative PE, UTS and OPL were tested as secondary endpoints (Fig. 2; Supplemental Tables S4 and S5). In these pharmacodynamic assessments, UB cigarettes were consistently rated higher than Vuse Alto 2.4% and 5% products. Compared to nicotine gum, the Vuse Alto products were generally rated higher, although some comparisons were not statistically significant (Fig. 2; Supplemental Tables S3, S4, and S5).
Product Liking and overall intent to use again (Panel A), Product Effects (Panel B) and Urge to smoke (Panel C) of Vuse Alto Golden Tobacco 2.4% and 5% nicotine concentration Products tested in the Study 1. The subjective effects for the Vuse Alto products and nicotine gum are presented as % values of UB CC (100%). Various measures used to assess subjective effects are indicated in the respective panels. * and # indicate statistically significant difference compared to UB CC and nicotine gum, respectively. For the primarily endpoints, PL and OIUA (Panel A), p ≤ 0.0042 following Bonferroni adjustment was considered significant, whereas for the other subjective effects (Panels B and C) p ≤ 0.05 was considered statistically significant. Supplemental Tables S3-S5 contain absolute values for the normalized data presented in this figure.
The maximum PL scores (Emax PL) were 9.22 for UB cigarettes and 6.62 for nicotine gum (Supplemental Table S3). The Emax PL for Vuse Alto 2.4% and 5.0% products were 7.84 and 7.78, respectively, which placed them between UB cigarettes and nicotine gum. Product liking (AUECPL 3−240) scores for UB cigarettes were highest at 1942 and lowest at 1161 for nicotine gum, whereas the Vuse Alto 2.4% and 5.0% products ratings were scored between the comparator products at 1419 and 1473, respectively.
Similarly, Overall Intent to Use Again (EOIUA) result for UB cigarettes was 8.67 compared to 4.09 for nicotine gum, and 5.95 and 5.73 for Vuse Alto 2.4% and 5.0% products, respectively (Supplemental Table S3). Compared with UB cigarettes, the PL and OIUA parameters for 2.4% and 5% Vuse Alto products were significantly lower than for UB cigarettes. However, relative to nicotine gum, the Emax PL was significantly higher for both Vuse Alto products. Additionally, the AUECPL3−240 was significantly higher for the Vuse Alto 5% product, whereas the EOIUA was significantly higher for the Vuse Alto 2.4% product (Fig. 2A).
Positive and negative PE of the Vuse Alto products were evaluated from two separate questions. The AUECPEpos 3−240 were 1060 and 1138 for Vuse Alto 2.4 and 5% products, respectively, compared to 947 for nicotine gum and 1364 for UB cigarettes; Emax PEpos values were 7.45 and 7.72 for Vuse 2.4% and 5% products, respectively, compared to 5.8 for nicotine gum, and 8.99 for UB cigarette (Supplemental Table S4). The Vuse Alto 2.4% product elicited significantly lower AUECPEpos 3−240 and Emax PEpos compared to UB cigarettes, while only the Emax PEpos was significantly higher compared with nicotine gum (Fig. 2B). The AUECPEpos 3−240 and Emax PEpos scores for Vuse Alto 5% were significantly lower compared to UB cigarettes, and were not different from nicotine gum. There were no significant differences in negative PE (Emax PEneg) for Vuse Alto 2.4% and 5% products relative to either UB cigarettes or nicotine gum (Fig. 2B).
Overall PL was assessed after product use (indicated as Eoverall PL). Vuse Alto 2.4% (6.42) and 5% (6.64) products were significantly different relative to both of the comparator products, with UB cigarettes (8.78) ranked higher in terms of OPL, and nicotine gum (5.12) ranked lower (Supplemental Table S4).
Several metrics, calculated from the UTS questionnaire responses provided insight into the subjects’ UTS. At 15 and 240 min after the start of product use, the AUECUTS 0−15 and AUECUTS 0−240 values were 90 and 1666 for UB cigarettes, and 110 and 1811 for nicotine gum, respectively. The AUECUTS 0−15 and AUECUTS 0−240 values were 91 and 1630 for Vuse Alto 2.4%, and 83.6 and 1683 for Vuse Alto 5%, respectively. Emin UTS values were 4.6 and 4.13 for the Vuse 2.4% and 5% products, respectively, compared to 3.67 for UB cigarettes and 6.19 for nicotine gum (Supplemental Table S5). The UTS measures for Vuse Alto 2.4% were significantly lower than nicotine gum, but not different from UB cigarettes. For Vuse Alto 5% product, the UTS measures were not significantly different from UB cigarettes. While the AUECUTS 0−240 was not different between Vuse Alto 5% and nicotine gum, the AUECUTS 0−15 and Emin UTS were significantly different.
Physiological measures
Physiological measures, including serial blood pressure assessments (systolic and diastolic), and heart rate were performed during each Test Session (Supplemental Table S6), and the changes from the baseline measure recorded 5 min prior to the start of product use sessions are reported herein.
The mean maximal increase in systolic blood pressure were 7.61 mmHg (UB cigarette), 12.3 mmHg (nicotine gum), 10.8 mmHg (Vuse Alto 2.4%), and 11.7 mmHg (Vuse Alto 5%) across the 4-hour Test Session. A statistically significant increase in the mean maximal increase in systolic blood pressure was only detected between Vuse Alto Golden Tobacco 5.0% compared to UB cigarette but not nicotine gum. Mean maximal increase in diastolic blood pressure was least 5.1 mmHg for UB cigarettes, and most for nicotine gum (8.55 mmHg), with increases of 8.45 and 7.7 mmHg recorded for Vuse Alto products 2.4% and 5% products, respectively. Of these changes in diastolic blood pressure, only the increase in Vuse Alto 2.4% product achieved statistical significance compared to baseline.
The mean maximal increase in heart rate was 14.1 bpm for UB cigarette, 8.5 bpm for nicotine gum, 13.2 bpm for Vuse Alto 2.4%, and 14.2 bpm for Vuse Alto 5%. The mean maximal increases in heart rate for Vuse Alto 2.4% and 5% products were higher compared to nicotine gum but not UB cigarette.
Study 2, nicotine pharmacokinetics
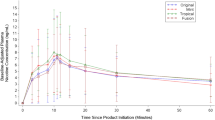
The four 1.8% nicotine concentration flavor variants (Alto Rich Tobacco, Golden Tobacco, Menthol and Mixed Berry) and the 2.4% nicotine concentration Golden Tobacco flavored Vuse Alto products displayed generally similar PK profiles and nicotine uptake parameters with overlaps of 95% CI (Fig. 3; Table 2). Geometric means of nicotine uptake at the first 15 min (AUC0 − 15) were 78.08, 81.54, 76.62 and 99.39 ng x min/mL for 1.8% Golden Tobacco, Rich Tobacco, Menthol and Mixed Berry flavored Vuse Alto products, respectively, while AUC0 − 15 was 96.26 ng x min/mL for the 2.4% Golden Tobacco product. The AUC0 − 240 values for Golden Tobacco, Menthol and Mixed Berry flavored Vuse Alto products were 679.38, 666.28, 747.33 and 850.28 ng x min/mL, respectively, while AUC0 − 240 for the 2.4% Golden Tobacco product was 798.51 ng x min/mL. The geometric mean Cmax values were 8.43, 8.89, 8.29 and 10.60 ng/mL for Golden Tobacco, Rich Tobacco, Menthol and Mixed Berry (all 1.5% nicotine), respectively, and 10.78 ng/mL for the 2.4% Golden Tobacco. The geometric mean Tmax values were between 10 and 12 min among all the Vuse Alto flavor variants at 1.8% and 2.4% nicotine concentrations (Fig. 3; Table 2). As a reference, PK parameter data of Vuse Alto Golden Tobacco products (2.4% and 5%) from the Study 1 are also included in Table 2. The AUC0 − 15, AUC0 − 240, Cmax and Tmax for these two products were approximately the same as those for the flavored Vuse Alto products, as evident from the overlap of 95% CIs.
Baseline adjusted mean plasma nicotine concentration profiles of four flavors of Vuse Alto ENDS products for 4 h following start of product use: The Study 2 evaluated Vuse Alto Golden Tobacco, Rich Tobacco, Menthol and Mixed Berry flavors at 1.8% nicotine concentration and a 2.4% nicotine concentration Golden Tobacco flavor.
Study 2, overall product liking
Study 2 assessed a single subjective measure, Eoverall PL, for OPL (Table 3). The Eoverall PL values for 1.8% Vuse Alto flavor variants were 5.5, 5.8, 5.8 and 6.9 for Golden Tobacco, Rich Tobacco, Menthol and Mixed Berry, respectively. The Eoverall PL for the 2.4% Vuse Alto Golden Tobacco product was 5.7. In comparison, OPL for the two Golden Tobacco flavored Alto (2.4% and 5% nicotine concentrations from the Study 1) were 6.4 and 6.7, respectively. Thus, the Eoverall PL scores were similar across the Vuse Alto products with different flavors and nicotine concentrations from the Studies 1 and 2, as evidenced by the overlap of their standard deviations.
Safety
In total, 13 subjects (26.5%) reported 22 AEs over the course of Study 1 (Supplemental Table S7). Of the 22 AEs, 16 were considered mild and 6 were considered moderate. The most frequently reported AE was headache (9 events in 7 subjects [14.3% of subjects]); followed by presyncope (3 events in 3 subjects [6.1% of subjects]). All other AEs occurred in 1 subject (2.0%) overall, including gastroesophageal reflux disease, nausea, vomiting, infusion site hematoma, tooth fracture, cervicogenic headache, throat irritation, lichenification, hypertension, and constipation. The overall number and nature of AEs were generally similar between products. Causally related AEs were reported by 6 subjects (12.2%): 5 subjects (10.2%) reported possibly related AEs (5 events of headaches), and 1 subject (2.0%) reported a related AE (1 event of throat irritation).
A total of 4 subjects (12.1%) reported 5 AEs during the conduct of Study 2 (Supplemental Table S7). One subject (3%) reported migraine exacerbation during the Pre-Study ENDS product Acclimation Period. All other AEs occurred in 1 subject (3.0%) overall, including nausea, dizziness, headache, and rash. Causally- related AEs were reported by 2 subjects (6%). Three AEs of migraine, nausea and dizziness were considered possibly related to the Vuse Alto product. Each of the AEs was mild and all AE symptoms resolved spontaneously without medical intervention or sequelae.
Discussion
Abuse liability provides information on the potential harm caused by tobacco products at the population level, and its assessment is a necessary requirement for marketing authorization of premarket and modified risk tobacco products by the US FDA30. A nicotine product with no adverse health impact but with AL would be expected to cause minimal ‘harm’ at a population level. Key findings from these studies are: (1) Vuse Alto products, independent of their nicotine content or flavor profiles, demonstrate a lower nicotine PK profile than cigarettes, but higher than nicotine gum. (2) Consistent with the nicotine PK, subjective measures data indicate that the nicotine content of e-liquid may have limited impact on the AL of Vuse Alto products. (3) The AL of the Vuse Alto products lies between that of combustible cigarette and nicotine gum.
Smokers have varying nicotine requirements and are known to titrate their nicotine intake to match their preferences45. Since the pharmacological profile of nicotine influences product use behavior and, therefore, exposure to toxicants found in cigarette smoke, which has been linked to smoking-related diseases, it plays a critical part in the determination of abuse liability30. As expected, the use of UB cigarettes resulted in the most rapid nicotine uptake (Tmax), highest peak concentrations (Cmax), and greatest overall nicotine exposure (AUC0-240), among all tested products.; The use of nicotine gum, however, yielded the lowest PK measures. While both UB cigarette and Vuse Alto product sessions were designed to last approximately 10 min for consistency, subjects may have completed cigarette use in a shorter time frame, reflecting typical cigarette smoking behavior. However, detailed data on actual smoking times were not analyzed, limiting the ability to fully assess the impact of this difference on AL. Future studies should incorporate precise measurements of usage duration for both product types to provide a more comprehensive understanding of their impact on AL outcomes.
In general, the PK metrics values for the two Vuse Alto Golden Tobacco products at 2.4% and 5% nicotine concentrations were similar to one another and significantly lower compared to the UB cigarettes, but higher than nicotine gum. Nicotine concentration may impact nicotine exposure, but our data also indicates a potential tendency to titrate at higher nicotine levels to achieve a similar level of nicotine exposure, which demonstrates an underlying complex product use behavior. Thus, our results differ from other research showing that higher nicotine concentrations in e-liquid would result in higher nicotine delivery47,48. The similarity in PK profiles of Vuse Alto 2.4% and 5% products supports previous knowledge that consumers self-titrate their nicotine intake, based on their individual preferences45.
Study 2 evaluated nicotine PK of four Vuse Alto products, Golden Tobacco, Rich Tobacco, Menthol and Mixed Berry at 1.8% nicotine concentration, and a Golden Tobacco product at 2.4% nicotine concentration. The PK parameters of these products (AUC at 15 and 240 min, Cmax and Tmax) were very similar as illustrated by the overlap of the 95% CI of Geometric means. Moreover, the PK metrics of the Vuse Alto Golden Tobacco 2.4% and 5% products measured separately in Study 1 were also comparable to PK metrics results in the PK study (Table 2). The nicotine PK metrics for the Golden Tobacco measured at 1.8% and 2.4% nicotine concentrations (Study 2) and 2.4% and 5% nicotine concentrations in Study 1 were similar.
Thus, combined data from the two clinical studies, show that independent of different nicotine concentrations and flavors, use of Vuse Alto products produced significantly more rapid and higher attainment of plasma nicotine concentrations than nicotine gum, but lower than the UB cigarettes, suggesting that the AL of the Vuse Alto products is lower than UB cigarettes and higher than nicotine gum.
In accordance with the FDA AL guidelines, we tested several subjective effect measures in these studies. While Study 1 utilized a battery of subjective measures, whereas Study 2 assessed only OPL, the sole measure common to both studies. Overall, consistent with the nicotine PK profile, the subjective effects from the use of 2.4% and 5% nicotine concentrations of Vuse Alto products were lower than the high AL comparator, UB cigarettes, and generally higher than the low AL comparator (nicotine gum). The PL measures were significantly lower for the Vuse Alto 2.4% and 5% products than UB cigarettes, and yet Vuse Alto products were almost as effective as UB cigarettes in reducing UTS. Thus, notwithstanding the differences in nicotine PK and other subjective measures, the two Vuse Alto products tested in Study 1 were effective in reducing craving for smoking, which may help smokers transition away from cigarette smoking to use of Vuse Alto products.
The OPL (Eoverall PL) for Vuse Alto 2.4% and 5% (Study 1) was significantly lower than UB cigarettes and higher than nicotine gum. The Eoverall PL was similar across the four 1.8% nicotine concentrations products and the 2.4% Golden Tobacco product (Study 2). The Eoverall PL scores for the 2.4% and 5% products (Study 1) and the 1.8% and 2.4% products were comparable, indicating the OPL for the Vuse Alto products was independent of the flavor and nicotine concentrations, and these products may be considered effective alternatives to cigarette smoking. Evaluation of additional subjective measures (OIUA, UTS, PE and OPL) also placed the AL of the Vuse Alto products in between UB cigarettes and nicotine gum.
Independent studies conducted by Goldenson et al. reported that the PK and abuse liability parameters of ENDS systems do not vary across flavors and at different nicotine concentrations41,47. Those authors used a version of the Vuse Alto products as a comparator product in their studies and reported that Vuse Alto products had lower abuse liability than cigarettes47. In that study, a Vuse Alto product at 5% nicotine concentration with original (Golden Tobacco), menthol and mixed berry flavors were tested.
A recent study by Talih et al.49 comparatively investigated design characteristics and toxicant emissions from Vuse Solo and Vuse Alto products. They reported that the Vuse Alto Rich Tobacco 5% nicotine emitted approximately three-fold higher levels of nicotine compared to a Vuse Solo Original 4.8% nicotine product. Based on nicotine levels found in the aerosol, the authors speculate that the Vuse Alto “may present a greater abuse liability profile”. While these data do not provide a comparison between the Vuse Solo and Vuse Alto products, the results from PK and subjective measures reported herein suggest that the AL of Vuse Alto Rich Tobacco 5% nicotine and other products is lower than cigarettes and higher than nicotine gum. The AL for Vuse Solo products has also been reported as between cigarettes and nicotine gum38,39,40.
Abuse liability of tobacco products influences frequency and prevalence of use and hence, the exposure to toxicants34. While the public health impact of ENDS continues to be an active area of research and discussion, several researchers have recognized that ENDS products, in general, have the potential to reduce harm from cigarette smoking24,45. For example, ENDS have been reported to aid in smoking reduction and cessation and exposing consumers to less toxicants than CC1317 1216, . Among the variables that are reported to aid in increased usage/ adoption of ENDS are nicotine concentrations and flavors of e-liquids24. Several studies suggested that flavored ENDS facilitate switching from smoking to ENDS use29. A population-based study has shown that switchers were more likely to report exclusively using menthol/mint-flavored ENDS but less likely to report exclusively using tobacco-flavored ENDS50. Another study reported that E-cigarette use is positively associated with both making a smoking quit attempt and quit success, and those using flavored e-cigarettes, particularly menthol/mint, are more likely to quit successfully28. Thus, Vuse Alto products, due to their intermediate level AL compared to cigarettes and current pharmaceutical alternatives, can promote migration away from cigarettes.
Strengths of this work include the evaluation of multiple Vuse Alto product variants, which differed in nicotine concentrations (1.8%, 2.4%, and 5% for Golden Tobacco) and flavor (Golden Tobacco, Rich Tobacco, Menthol and Mixed Berry at 1.8% nicotine concentration). The PK metrics consistently demonstrated that Vuse Alto products have lower AL than cigarettes, yet are able to provide sufficient nicotine exposure to satisfy smokers’ needs to decrease their smoking urges.
A potential limitation of the Study 2 is that it did not employ high- and low- AL comparators. However, it is well established that the AL of ENDS products is lower than the cigarettes38,39,41and the inclusion of 2.4% Golden Tobacco in both studies provides a reference for relevant PK measures relative to combustible cigarettes and nicotine gum. Additionally, the PK metrics of all tested Vuse Alto products exhibit overlapping 95% confidence intervals, attesting to similarity in their nicotine exposure profiles (Table 2). The observed similarity in PK profiles between the 2.4% and 5% nicotine concentrations suggests a potential titration effect. However, these studies did not capture data on key factors that could influence nicotine uptake, such as puff count, puff duration, or inhaling behavior. Future research incorporating these variables would provide a more comprehensive understanding of titration and potential differences in inhalation patterns among subjects. Another potential limitation is that Study 2 evaluated only OPL as a subjective measure, whereas Study 1 included a broader set of subjective measures. Nevertheless, Table 3 indicates similar OPL measures from both studies. Taken together, the similarity in nicotine PK and the OPL findings indicate that the AL of all Vuse Alto products evaluated across the two studies is comparable.
In conclusion, Vuse Alto products demonstrate lower nicotine uptake and subjective measures than cigarettes, indicating reduced AL. These findings support their potential role in aiding smokers’ transition to less harmful nicotine delivery alternatives. Furthermore, Vuse Alto products effectively deliver nicotine to satisfy smoking urges and other positive subjective effects, independent of nicotine concentration and flavor. Given that smokers are heterogeneous in their nicotine preferences, availability of different ENDS products with a range of nicotine concentrations and flavors may facilitate smoking reduction or cessation. However, the AL findings should be interpreted within the context of the study population, which consisted of highly dependent cigarette smokers with no history of using ENDS, and may not fully generalize to less dependent smokers or with individuals with greater experience using ENDS. Future studies should include a broader range of tobacco- and nicotine-use experiences to provide a more comprehensive understanding of AL across a broader spectrum of users.
Data availability
All applicable datasets generated and analyzed in Study 1 and Study 2 are presented in this manuscript and in the supplemental sections. Access to additional datasets for these studies is administered through an internal Data Sharing Committee on a reasonable request following completion of a data sharing request form and if applicable, a Data Access Agreement. Requests for data sharing and materials in the first instance should be directly emailed to the corresponding author.
References
FDA. Premarket tobacco product applications and recordkeeping requirements. In: Food and Drug Administration H, (ed). Federal register. pp 55300–55439. (2021).
Institute of Medicine. Clearing the Smoke: Assessing the Science Base for Tobacco Harm Reduction. The National Academies Press.: Washington, DC:, 2001. (2001).
Abrams, D. B. et al. Harm minimization and tobacco control: reframing societal views of nicotine use to rapidly save lives. Annu. Rev. Public Health. 39, 193–213 (2018).
United States Public Health Service Office of the Surgeon G, National Center for Chronic, Disease, P. & Health Promotion Office on S, Health. Publications and Reports of the Surgeon General. In: Smoking Cessation: A Report of the Surgeon General. US Department of Health and Human Services: Washington (DC) submitted a conflict of interest disclosure form. Unless otherwise noted below, no potential conflicts of interest were reported. Dr. Glenda Lassi reports being employed by AstraZeneca, a research-based biopharmaceutical company. Dr. Marcus R. Munafò reports receiving grant funding from Pfizer and Rusan Pharma and obtaining products for use in research from GlaxoSmithKline, a company that manufactures smoking cessation products. Dr. Judith J. Prochaska reports having served as an ad hoc advisor to Pfizer Inc, a company that manufactures smoking cessation medications., (2020).
Food and Drug Administration. Nicotine Is Why Tobacco Products Are Addictive. In: Health and Human Services, (ed), (2022).
Hatsukami, D. K. et al. Developing the science base for reducing tobacco harm. Nicotine Tob. Res. 9 (Suppl 4), S537–553 (2007).
National Academies of Sciences E, Medicine. Public Health Consequences of E-Cigarettes (National Academies, 2018).
Margham, J. et al. The chemical complexity of e-Cigarette aerosols compared with the smoke from a tobacco burning cigarette. Front. Chem. 9, 743060 (2021).
Tayyarah, R. & Long, G. A. Comparison of select analytes in aerosol from e-cigarettes with smoke from conventional cigarettes and with ambient air. Regul. Toxicol. Pharmacol. 70, 704–710 (2014).
Kanobe, M. N. et al. Part three: a randomized study to assess biomarker changes in cigarette smokers switched to vuse solo or abstinence. Sci. Rep. 12, 20658 (2022).
Round, E. K., Chen, P., Taylor, A. K. & Schmidt, E. Biomarkers of tobacco exposure decrease after smokers switch to an E-Cigarette or nicotine gum. Nicotine Tob. Res. 21, 1239–1247 (2019).
Butler, A. R. et al. Longer-term use of electronic cigarettes when provided as a stop smoking aid: systematic review with meta-analyses. Prev. Med. 165, 107182 (2022).
Foulds, J. et al. Effect of electronic nicotine delivery systems on cigarette abstinence in smokers with no plans to quit: exploratory analysis of a randomized Placebo-Controlled trial. Nicotine Tob. Res. 24, 955–961 (2022).
Hajek, P. et al. A randomized trial of E-Cigarettes versus Nicotine-Replacement therapy. N Engl. J. Med. 380, 629–637 (2019).
Han, D. H., Lee, S. H. & Seo, D. C. Within-Person longitudinal associations between electronic nicotine delivery systems use and smoking cessation efforts among US continuing adult cigarette smokers. Nicotine Tob. Res. 24, 590–597 (2022).
Myers Smith, K. et al. E-cigarettes versus nicotine replacement treatment as harm reduction interventions for smokers who find quitting difficult: randomized controlled trial. Addiction 117, 224–233 (2022).
Vanderkam, P. et al. Duration of the effectiveness of nicotine electronic cigarettes on smoking cessation and reduction: systematic review and meta-analysis. Front. Psychiatry. 13, 915946 (2022).
Warner, K. E., Benowitz, N. L. & McNeill, A. Rigotti NA Nicotine e-cigarettes as a tool for smoking cessation. Nat. Med. ; e-pub ahead of print 20230213; (2023). https://doi.org/10.1038/s41591-022-02201-7
Kaplan, B., Galiatsatos, P., Breland, A., Eissenberg, T. & Cohen, J. E. Effectiveness of ENDS, NRT and medication for smoking cessation among cigarette-only users: a longitudinal analysis of PATH Study wave 3 (2015–2016) and 4 (2016–2017), adult data. Tob Control ; e-pub ahead of print 20210915; (2021). https://doi.org/10.1136/tobaccocontrol-2020-056448
Pound, C. M., Zhang, J. Z., Kodua, A. T. & Sampson, M. Smoking cessation in individuals who use vaping as compared with traditional nicotine replacement therapies: a systematic review and meta-analysis. BMJ Open. 11, e044222 (2021).
Gottlieb, S. & Zeller, M. A. Nicotine-Focused framework for public health. N Engl. J. Med. 377, 1111–1114 (2017).
Food and Drug Administration. Tobacco Products Marketing Orders. In, (2022).
HenningfieldJE, HatsukamiDK, Zeller, M. & Peters, E. Conference on abuse liability and appeal of tobacco products: conclusions and recommendations. Drug Alcohol Depend. 116, 1–7 (2011).
Gades, M. S., Alcheva, A., Riegelman, A. L. & Hatsukami, D. K. The role of nicotine and flavor in the abuse potential and appeal of electronic cigarettes for adult current and former cigarette and electronic cigarette users: A systematic review. Nicotine Tob. Res. 24, 1332–1343 (2022).
Food and Drug Administration. Premarket Tobacco Product Applications and Recordkeeping Requirements. Available from: URL (Accessed n Date Accessed Year)|.
Cooper, M. et al. Notes from the field: E-cigarette use among middle and high school Students - United states, 2022. MMWR Morb Mortal. Wkly. Rep. 71, 1283–1285 (2022).
Park-Lee, E. et al. Tobacco product use among middle and high school Students - United states, 2022. MMWR Morb Mortal. Wkly. Rep. 71, 1429–1435 (2022).
Mok, Y., Jeon, J., Levy, D. T. & Meza, R. Associations between e-cigarette use and e-cigarette flavors with cigarette smoking quit attempts and quit success: Evidence from a US large, nationally representative 2018–2019 survey. Nicotine Tob Res ; e-pub ahead of print 20221017; (2022). https://doi.org/10.1093/ntr/ntac241
Li, L. et al. How does the use of flavored nicotine vaping products relate to progression toward quitting smoking?? Findings from the 2016 and 2018 ITC 4CV surveys. Nicotine Tob. Res. 23, 1490–1497 (2021).
Food and Drug Administration. Premarket tobacco product applications and recordkeeping requirements. Fed. Regist. (2021).
Vansickel, A. et al. Approaches for meeting current regulatory recommendations. Nicotine Tob. Research: Official J. Soc. Res. Nicotine Tob. 24, 295–305 (2022).
Fant, R. V., Henningfield, J. E. & Nelson, R. A. Pickworth WB pharmacokinetics and pharmacodynamics of moist snuff in humans. Tob. Control. 8, 387–392 (1999).
BenowitzNL Pharmacokinetic considerations in Understanding nicotine dependence. Ciba. Found. Symp. 152, 186–200 (1990). discussion 200 – 189.
Carter, L. P., Stitzer, M. L., Henningfield, J. E., O’Connor, R. J. & Cummings, K. M. Hatsukami DK abuse liability assessment of tobacco products including potential reduced exposure products. Cancer Epidemiol. Biomarkers Prev. 18, 3241–3262 (2009).
Hanson, K. & O’Connor, R. Hatsukami D measures for assessing subjective effects of potential reduced-exposure products. Cancer Epidemiol. Biomarkers Prev. 18, 3209–3224 (2009).
Breland, A. et al. Abuse liability of electronic cigarettes in men who are experienced electronic cigarette users. Exp. Clin. Psychopharmacol. 28, 235–244 (2020).
Maloney, S. F. et al. Abuse liability assessment of an electronic cigarette in combustible cigarette smokers. Exp. Clin. Psychopharmacol. 27, 443–454 (2019).
Stiles, M. F. et al. Pharmacodynamic and Pharmacokinetic assessment of electronic cigarettes, combustible cigarettes, and nicotine gum: implications for abuse liability. Psychopharmacol. (Berl). 234, 2643–2655 (2017).
Stiles, M. F., Campbell, L. R., Jin, T., Graff, D. W. & Fant, R. V. Henningfield JE assessment of the abuse liability of three menthol vuse solo electronic cigarettes relative to combustible cigarettes and nicotine gum. Psychopharmacol. (Berl). 235, 2077–2086 (2018).
Campbell, C. et al. Part one: abuse liability of vuse solo (G2) electronic nicotine delivery system relative to combustible cigarettes and nicotine gum. Sci. Rep. 12, 22080 (2022).
Goldenson, N. I., Buchhalter, A. R., Augustson, E. M., Rubinstein, M. L. & Henningfield, J. E. Abuse liability assessment of the JUUL system in four flavors relative to combustible cigarette, nicotine gum and a comparator electronic nicotine delivery system among adult smokers. Drug Alcohol Depend. 217, 108395 (2020).
Farsalinos, K. E., Spyrou, A., Tsimopoulou, K., Stefopoulos, C. & Romagna, G. Voudris V nicotine absorption from electronic cigarette use: comparison between first and new-generation devices. Sci. Rep. 4, 4133 (2014).
Wagener, T. L. et al. Have combustible cigarettes Met their match? The nicotine delivery profiles and harmful constituent exposures of second-generation and third-generation electronic cigarette users. Tob. Control. 26, e23–e28 (2017).
FDA. Assessment of Abuse Potential of Drugs: Guidance for Industry. In: Research CfDEa, (ed). Silver Spring, MD, USA. pp 1–37. (2017).
Benowitz, N. L., St Helen, G., Liakoni, E. & Clinical Pharmacology of Electronic Nicotine Delivery Systems (ENDS). Implications for benefits and risks in the promotion of the combusted tobacco endgame. J. Clin. Pharmacol. 61 (Suppl 2), S18–s36 (2021).
Keyser, B. M. et al. Part two: an unblinded, parallel, randomized study to assess nicotine pharmacokinetics of four vuse solo ENDS flavors in smokers. Sci. Rep. 13, 8894 (2023).
Goldenson, N. I. et al. Abuse liability assessment of the JUUL system in two nicotine concentrations compared to combustible cigarette, nicotine gum and comparator electronic nicotine delivery system. Drug Alcohol Depend. 217, 108441 (2020).
Phillips-Waller, A., Przulj, D., Smith, K. M., Pesola, F. & Hajek, P. Nicotine delivery and user reactions to Juul EU (20 mg/ml) compared with Juul US (59 mg/ml), cigarettes and other e-cigarette products. Psychopharmacol. (Berl). 238, 825–831 (2021).
Talih, S. et al. Comparison of design characteristics and toxicant emissions from vuse solo and Alto electronic nicotine delivery systems. Tob. Control. e-pub ahead of print (20230418). https://doi.org/10.1136/tc-2022-057711 (2023).
Chang, J. T. et al. Characteristics and patterns of cigarette smoking and vaping by past-year smokers who reported using ENDS to help quit smoking in the past year: Findings from the 2018–2019 Tobacco Use Supplement to the Current Population Survey. Nicotine Tob Res ; e-pub ahead of print 20220817; (2022). https://doi.org/10.1093/ntr/ntac199
Acknowledgements
The authors acknowledge GL Prasad (Prasad Scientific Consulting LLC) for support in preparing the manuscript.
Funding
This study was funded by RAI Services Company, a wholly owned subsidiary of Reynolds American Inc.’s (“RAI”), which is an indirect, wholly owned subsidiary of British American Tobacco p.l.c. The funder had the following involvement in the study: study design, interpretation of data, the writing of this manuscript, and the decision to submit for publication.
Author information
Authors and Affiliations
Contributions
Conceptualization: K.P.; Methodology: P.M., and K.P.; Investigation: P.M., K.G.J., and S.A.B.; Formal Analysis: S.A. and T.J.; Resources: K.P.; Writing, review & Editing: K.G.J., B.M.K., M.N.K., P.M., T.J., S.A., S.A.B., J.D., and K.P.; Supervision: K.P.; Project Administration: K.P. and J.D. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors are employees of RAI Services Company, a wholly owned subsidiary of RAI., which is an indirect, wholly owned subsidiary of British American Tobacco plc. RAI through its other subsidiary companies manufactures and markets Vuse Alto products.
Ethics approval
Study 1 (ClinicalTrials.gov identifier; date: NCT 04094363; 18/09/2019) was approved by IntegReview IRB (Austin, TX; IORG0007169), and Study 2 (ClinicalTrials.gov identifier; date: NCT04751461; 12/02/2021) was approved by Advarra IRB (Columbia, MD; IORG0000635). Informed consent was obtained from all individual participants included in the studies.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Prevette, K., Jordan, K.G., Keyser, B.M. et al. Randomized clinical studies assessing the pharmacokinetics and subjective effects of vuse alto electronic nicotine delivery systems. Sci Rep 15, 21711 (2025). https://doi.org/10.1038/s41598-025-06716-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-06716-0