Abstract
Cis-abienol, a naturally occurring labdanum diterpenoid, is believed to be an essential precursor of ambergris substances and is found on the surface of many plants. In this study, the strain Klebsiella oxytoca T2L was isolated from the soil of oriental tobacco and demonstrated effective degradation of cis-abienol. The strain was identified through morphological characteristics and 16 S rDNA sequence analysis. Under culture conditions of 30 °C, pH 7.0, 0.5 mg/mL ammonium sulfate, and 1 mg/mL cis-abienol—the degradation rate of cis-abienol by strain T2L reached 68.2% after 96 h. The influence of various carbon and nitrogen sources on the degradation effect of cis-abienol by T2L was examined. Additionally, the impact of pH and temperature on cell growth and cis-abienol degradation was also analyzed. GC-MS analysis indicated that the degradation of cis-abienol by strain T2L generated several potential aroma chemicals, including (+/-)-ambreinolide, sclareol, an analog of amberonne (isomer 3), and an analog of sclaral (sclareolide lactol), which resembled the composition of natural ambergris. This study provides a theoretical foundation for further research on the biodegradation of cis-abienol to high-value aroma compounds.
Similar content being viewed by others
Introduction
Cis-abienol, a naturally occurring labdanum diterpenoid1, has a molecular weight of 290.48. It is one of the exudates found on the surface of plants2; however, its contents are scarce and limited to a few plants. Initially discovered in the oil resin of Canadian fir, cis-abienol has been subsequently found in the essential oils of coniferous plants3,4, in the tuber of Compositaceae snow lily, and in the glandular hairs of tobacco5,6,7. Studies have demonstrated that cis-abienol possesses various biological functions, including insect resistance and disease resistance8,9,10, and it helps prevent other microbial infections11,12,13. For instance, cis-abienol has been used as an inducer to effectively control bacterial wilt in tomatoes and other plants14.
Despite its limited presence in plants, cis-abienol plays a crucial role in aroma properties15,16,17. It has been identified as an essential precursor of ambergris substances18,19,20, which are among the most precious spices in the world today. Ambergris is known for its lasting aroma, excellent harmonization, and strong fixative properties21,22. Currently, ambergris is primarily used as a tincture in the formulation of high-end and expensive perfumes, holding an indispensable position in fragrance creation. Among ambergris substitutes, ambrox23 and norambreinolide24 are prominent examples.
Although cis-abienol has been identified as a precursor for the chemical synthesis of ambergris-like substances, research on the degradation or transformation of cis-abienol into such products remains limited. Huang Tingting et al.25 summarized the metabolic regulation mechanism of labdanum diterpenoids and illustrated that cis-abienol could produce compounds such as sclareol, ambrox, and norambreinolide through redox reactions. Tadaharu Hieda et al.26 isolated a strain, JTS-131, capable of catalyzing cis-abienol degradation, yielding products such as (12Z)-labda-12,14-dien-18-01 and 12(Z)-labda-12,14-dien-18-oic acid and its methyl ester. Mohamad I. Farbood27 used two bacterial strains, Cryptococcus albidus ATCC20918 and Hyphozyma roseonigra ATCC20624, to transform sclareol into sclareolide and sclareol glycol. Similarly, S. A. Kouzi et al.28 investigated the microbial metabolism of sclareol using Bacillus cereus UI-1477, identifying seven metabolites.
In this study, a novel microbial strain capable of degrading cis-abienol as the sole carbon source to generate ambergris-like substances has been isolated. The degraded products were identified through GC-MS, and various growth parameters affecting the degradation rate of cis-abienol were optimized. This research provides a theoretical basis for the biodegradation of cis-abienol into valuable ambergris-like products.
Results
Isolation and identification of cis-abienol degrading bacteria
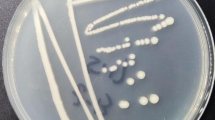
A strain capable of degrading cis-abienol was isolated using cis-abienol as the sole carbon source from the soil of aromatic tobacco. The strain was designated as T2L. Colonies of T2L were observed to form white, round spots with distinct transparent edges and a solid center. These colonies were opaque, lacked mycelia, and had a smooth appearance (Fig. 1A). Gram staining revealed that T2L was gram-positive (Fig. 1B). Scanning electron microscopy (15,000× magnification) showed that the strain consisted of short, rod-like cells measuring 7–9 μm in length and 18–20 μm in width. The cells lacked flagella and exhibited wrinkled surfaces (Fig. 1C).
Morphological characteristics of the screened strain T2L. (A) Colony morphology. (B) Gram staining results. (C) Scanning electron microscopy results.
Following purification, the strain was identified by 16 S rDNA technique, revealing a sequence length of 1,404 bp. BLAST homology analysis showed that the 16 S rDNA sequence of T2L shared 99.93% similarity with Klebsiella oxytoca M0572 and Klebsiella sp. HNDS8, and 99.79% similarity with Klebsiella pasteurii SPARK836C1. Phylogenetic analysis indicated that T2L formed a distinct species lineage within the K. oxytoca cluster. Although closely related, T2L occupied a separate branch in the phylogenetic tree, leading to its identification as K. oxytoca (Fig. 2).
Phylogenetic tree construction using MEGA software based on the 16 S rDNA sequence.
The growth and degradation conditions of T2L in a medium containing cis-abienol
After screening for the cis-abienol-degrading strain, T2L was inoculated into a medium containing cis-abienol at 1 mg/mL as the sole carbon source. Strain T2L exhibited rapid growth during the first 48 h, entering the logarithmic phase with an OD600 of 1.9. After 48 h, the strain reached the stationary phase, with bacterial content remaining stable. Concurrently, the cis-abienol content in the medium decreased, dropping sharply between 24 and 48 h. By 72 h, the relative content of cis-abienol had stabilized at 49.4%. After 96 h, the degradation rate slowed further, resulting in a relative cis-abienol content of 48.6% (Fig. 3A). The negative control used was the same medium containing cis-abienol at 1 mg/mL concentrations without T2L. The results indicated that cis-abienol remained stable throughout the entire fermentation process (Fig. S1).
To assess the effect of cis-abienol concentration on its degradation process, experiments were performed with various initial concentrations of cis-abienol. Specifically, at an initial concentration of 1 mg/mL, the degradation rate was found to be the highest, achieving 54.6%. At an initial concentration of 8 mg/mL, the degradation rate decreased significantly to 29.7% (Fig. 3B). Interestingly, at 0.5 mg/mL, the degradation rate was slightly lower than at 1 mg/mL, reaching 42.3%. Multiple repeats confirmed these results, indicating a consistent trend.
Growth conditions and degradation rate of cis-abienol by T2L. (A) Cell growth of T2L in medium containing 1 mg/mL cis-abienol. (B) Degradation rates of cis-abienol at different initial concentrations.
Analysis of possible metabolites formed from cis-abienol during fermentation with the strain T2L
GC-MS analysis was performed to identify the degradation products of cis-abienol by strain T2L during fermentation. Several chemicals were detected as possible degradation products when T2L utilized cis-abienol as the sole carbon source (Fig. 4). After 24 h, a new compound 6, potentially identified as (3R)-3,7-dihydroxy-9,11-eremophiladien-8-one 3-acetate, was detected and its concentration increased to 66.6 µg/mL by 96 h. At 48 h, a compound 7, potentially identified as 2,5,5,8a-tetramethyl-4-methylene-4a,5,6,7,8,8a-hexahydro-4 H-chromene was detected, reaching 30 µg/mL by 96 h. Another two possible compounds 8 and 5, (+/-)-ambreinolide and 15-isobutyl-(13.alpha.H)-isocopalane, were detected after 72 h, with concentrations increasing to 10.9 µg/mL and 23.1 µg/mL, respectively. Minor amounts of manoyl oxide, manool, and ambrial were also detected after 96 h according to the GC-MS analysis (Fig. 4; Table 1).
The cis-abienol utilized in the experiments was obtained commercially and contained various impurities as determined by GC-MS analysis, including amberonne (an isomer of cis-abienol), sclareol analog, sclaral, and n-nonenylsuccinic anhydride. During fermentation, the concentrations of amberonne analog (compound 3), sclaral (compound 2), and n-nonenylsuccinic anhydride (compound 1) increased to 40.7, 28.7, and 42.4 µg/mL, representing 10.3-, 14.35-, and 10.6-fold increases, respectively, compared to the initial levels. In contrast, the concentration of sclareol analog (compound 4) remained relatively unchanged.
GC-MS analysis of fermentation products of T2L using cis-abienol as the sole carbon source. 1: n-Nonenylsuccinic anhydride; 2: Sclaral (sclareolide lactol); 3: Amberonne (isomer 3) analog; 4: Sclareol analog; 5: 15-Isobutyl-(13.alpha.H)-isocopalane; 6: (3R)-3,7-Dihydroxy-9,11-eremophiladien-8-one, 3-acetate; 7: 2,5,5,8a-Tetramethyl-4-methylene-4a,5,6,7,8,8a-hexahydro-4 H-chromene; 8: (+/-)-Ambreinolide. The peak ion profile of the compounds on MS was supplemented in Fig. S4.
The influence of different sources of carbon and nitrogen on cis-abienol degradation
To determine the effects of various carbon sources on the degradation of cis-abienol by strain T2L, several carbon sources at a concentration of 1 mg/mL were added to the medium separately alongside cis-abienol. The results indicated that the addition of other carbon sources was detrimental to cis-abienol degradation. Glucose, sucrose, and maltose significantly inhibited the growth of T2L, with OD600 values decreasing by 46.34%, 53.65%, and 43.81%, respectively. Similarly, β-cyclodextrin reduced cell growth in the inorganic medium by 27.97%. Moreover, the degradation rate of cis-abienol was also negatively affected by the addition of these carbon sources. The degradation rates decreased significantly by 90.1% with glucose, 85.15% with sucrose, 77.3% with maltose, and 84.32% with β-cyclodextrin (Fig. 5A). These findings suggest that the addition of alternative carbon sources is not beneficial for either cell growth or cis-abienol degradation by T2L in an inorganic medium.
Following the analysis of carbon sources, the effects of different nitrogen sources on cis-abienol degradation were also examined. The inorganic medium initially contained 0.5 mg/mL of ammonium sulfate as the nitrogen source. To evaluate the influence of other nitrogen sources, 0.5 mg/mL of urea, peptone, yeast powder, sodium nitrate, or potassium nitrate was used to replace ammonium sulfate. The results showed that all these nitrogen sources significantly affected both cell growth and cis-abienol degradation. The addition of urea, peptone, yeast powder, and sodium nitrate caused reductions in cell growth and cis-abienol degradation of more than 50%. Potassium nitrate also had a significant inhibitory effect, reducing cell growth and degradation by 59.32% and 77.32%, respectively (Fig. 5B). Among all tested nitrogen sources, ammonium sulfate yielded the best results for cell growth and cis-abienol degradation.
The effect of carbon and nitrogen sources on the degradation rate of cis-abienol by T2L. (A) Effect of different carbon sources. (B) Effect of different nitrogen sources. (C) Effect of ammonium sulfate concentration.
Since ammonium sulfate was the most effective nitrogen source for T2L in degrading cis-abienol, the optimal concentration of ammonium sulfate for cis-abienol degradation was investigated. Ammonium sulfate concentrations ranging from 0.5 to 8 mg/mL were tested. The peak degradation rate of cis-abienol was achieved at 61.3% when the concentration of ammonium sulfate was 6 mg/mL. However, at a concentration of 8 mg/mL, the degradation rate sharply decreased to 16.2% (Fig. 5C). The optimal concentration of ammonium sulfate was therefore determined to be 6 mg/mL.
The influence of pH and temperature on cell growth of T2L and cis-abienol degradation
Further experiments were performed to evaluate the impact of different temperatures and pH levels on the growth of strain T2L and its capacity to degrade cis-abienol. The T2L strain was cultured in a medium containing 1 mg/mL of cis-abienol at temperatures of 20 °C, 25 °C, 30 °C, 35 °C and 40 °C, all with a constant rotation speed of 150 rpm. As shown in Fig. 6A, the highest cell OD600 value of approximately 2.0 was achieved at 30 °C, corresponding to the highest degradation rate of cis-abienol at 51.4%. Both cell growth and cis-abienol degradation declined as temperatures increased beyond 30 °C. These results identify 30 °C as the optimal temperature for T2L to degrade cis-abienol in the inorganic medium.
The effect of different temperatures and pH on the degradation of cis-abienol by strain T2L. (A) Effect of temperature. (B) Effect of pH.
The impact of pH levels ranging from 5 to 9 on the growth and degradation rate of cis-abienol by strain T2L was also investigated. Initially, the stability of cis-abienol was examined in a medium with varying pH levels as a negative control. The findings revealed that cis-abienol is stable at pH 5 and pH 9 (Fig. S2). Subsequently, the pH value of the fermentation medium was tested, which contained strain T2L, cis-abienol, glucose as the carbon source, and different nitrogen sources like ammonium sulfate or peptone. It was observed that the pH value remained consistent throughout the entire experiment (Fig. S3). The results showed that the optimal pH for cis-abienol degradation was 7.0, achieving a degradation rate of approximately 52.9%. Furthermore, the optimal pH for cell growth was also found to be 7.0. Deviations from this pH value, either below or above 7.0, negatively affected both cell growth and the degradation rate of cis-abienol. Interestingly, at a pH of 9.0, the degradation rate of cis-abienol reached 28.82%, although it remained lower compared to the results at pH 7.0 (Fig. 6B) .
Discussion
In this study, the cis-abienol-degrading strain T2L was successfully isolated and identified using cell morphology, cell size analysis, and 16 S rDNA sequencing. BLAST homology analysis through the NCBI GenBank database confirmed that strain T2L is a member of K. oxytoca. Previous studies have shown that species within this genus can degrade nitrobenzene and 4-chloronitrobenzene and improve the salt-alkali tolerance of corn seedlings, highlighting their potential applications in agriculture and environmental management29,30. In the present study, K. oxytoca T2L degraded 68.2% of cis-abienol (1 mg/mL) within 96 h, producing several substances, including labdanum and ambergris-like compounds, thereby confirming its potential as a cis-abienol-degrading strain.
The results displayed that ammonia sulfate was much better than other nitrogen sources, including organic nitrogen and certain inorganic nitrogens. It has been reported that certain nitrogen in ammonium salts can be directly absorbed and utilized by bacterium, while nitrogen in nitrate or organic nitrogen needs to be converted to ammonia prior to absorption31. Additionally, we speculate that sulfate might be a beneficial substance for some enzymes in strain T2L.
It has been reported by Tingting Huang et al.25 that cis-abienol can be transformed into sclareol. In our experiment, however, a potential sclareol analog was detected using GC-MS in comparison to the standard sclareol. The concentration of this analog initially decreased and then increased after 72 h. This could be attributed to the similarity between cis-abienol and the sclareol analog. The detected chemical sclareol analog is present in commercial cis-abienol. We speculate that the sclareol analog was initially consumed by the cell and subsequently increased, potentially due to the biodegradation of cis-abienol by strain K. oxytoca T2L. Furthermore, after 96 h, possible chemicals such as (+/-)-ambreinolide, sclaral (sclareolide lactol) and amberonne (isomer 3) analog were detected. Studies suggest that cis-abienol32 and sclareol33 can be transformed into nor-ambreinolide34, with sclareol being the more probable precursor35. Combining the chemical reactions from cis-abienol to ambergris-like chemicals with published articles, it is speculated that K. oxytoca T2L transforms cis-abienol into a sclareol analog, which is then converted into (+/-)-ambreinolide, sclaral, and ambrial. For the amberonne (isomer 3) analog, no relevant studies are available and its specific degradation pathway remains unclear. Other major products such as (3R)-3,7-dihydroxy-9,11-eremophiladien-8-one,3-acetate and n-nonenylsuccinic anhydride are presumed to be metabolic byproducts generated during the utilization of cis-abienol by the strain. The predicted degradation pathway of cis-abienol by strain T2L is summarized in Fig. 7.
Strain T2L effectively degrades cis-abienol, producing several ambergris-like products, thus providing a valuable microbial resource for studying ambergris synthesis and cis-abienol degradation. Products such as (+/-)-ambreinolide, ambrial, and amberonne (isomer 3) share similarities with natural ambergris and exhibit an amber-like aroma, making them key intermediates in the synthesis of ambrox36,37,38. Sclaral (sclareolide lactol), presumed to be an oxidation product of sclareol, is not currently available in domestic or international markets.
Prediction of the chemicals involved in the cis-abienol degradation process using strain T2L.
Conclusion
K. oxytoca T2L was identified as a strain capable of effectively degrading cis-abienol. The strain degraded 68.2% of 1 mg/mL cis-abienol within 96 h. Optimal growth and degradation conditions for K. oxytoca T2L were determined to be 30 °C, pH 7, 6 mg/mL ammonium sulfate, and 1 mg/mL cis-abienol. Gas chromatography-mass spectrometry (GC-MS) analysis of the degradation products revealed the formation of several aroma compounds, including (3R)-3,7-dihydroxy-9,11-eremophiladien-8-one,3-acetate, n-nonenylsuccinic anhydride, amberonne (isomer 3), (+/-)-ambreinolide, ambrial, sclaral (sclareolide lactol), and sclareol analog. This research provides a theoretical basis for the biodegradation of cis-abienol into valuable aroma compounds.
Materials and methods
Isolation of cis-abienol-degrading bacteria and the culture medium
Soil samples were collected from oriental tobacco fields in Zhengzhou, Henan Province, China, and transported to the laboratory for bacterial isolation. A 1 g soil sample was added to 100 mL sterile water in a conical flask under aseptic conditions. The mixture was soaked and shaken for approximately 1 h. From this suspension, 1% of the bacterial source was enriched in Luria–Bertani (LB) medium at 150 rpm/min and 30 °C for 6 h. Once the medium became slightly turbid, indicating enrichment, 1% of the enriched culture (diluted from 10⁻¹ to 10⁻⁶) was spread onto selective plates containing cis-abienol as the sole carbon source39.
The composition of the selective inorganic medium was as follows: K2HPO4 (1 g/L), MgSO4·7 H₂O (0.5 g/L), FeSO4·7 H₂O (0.005 g/L), NaCl (0.5 g/L), KH₂PO4 (0.65 g/L), MnSO4 (0.001 g/L), (NH4)2SO4 (0.5 g/L), CaCl2·2 H₂O (0.1 g/L), and Na2MoO4·2H2O (0.005 g/L). To screen for cis-abienol-degrading strains, 1 mg/mL cis-abienol was added to the medium. Plates were incubated at 30 °C for 1–2 days. Colonies appearing on the plates were isolated and cultured in an LB medium. Isolated strains were preserved in 60% (w/v) glycerol at − 80 °C. The fermentation medium used to evaluate cis-abienol degradation consisted of a selective inorganic medium with variable carbon or nitrogen sources, depending on experimental requirements.
Molecular identification of T2L strain by 16 S rDNA and morphology observation by scanning electron microscopy
Colony morphology and cell shape were examined using high-resolution cold-field emission scanning electron microscopy (Hitachi, Regulus 8100). Genomic DNA was extracted using a bacterial genomic DNA rapid extraction kit (TIANamp Bacteria DNA Kit, TIANGEN BIOTECH, Beijing, China). The 16S rDNA was amplified using the primer pairs 5’-AGAGTTTGATCCTGGCTCAG-3’ and 5’-GGTTACCTTGTTACGACTT-3’, synthesized by Personalbio Technology (Shanghai) Co., Ltd. The PCR reaction yielded a 1,424 bp fragment, which was purified and sequenced by Personalbio Technology (Shanghai) Co., Ltd.
Phylogenetic analysis was conducted using MEGA 5.1 software, with a phylogenetic tree constructed based on the 16 S rDNA sequence. The neighbor-joining method was used, with 1,000 bootstrap replicates.
Quantification method for cis-abienol
Quantification of cis-abienol was performed using standard cis-abienol solutions with varying concentrations analyzed by ultra-performance liquid chromatography (UPLC). The UPLC conditions were as follows40,41,42: the chromatographic column was a WATERS XBridge C18 Column (4.6 × 250 mm, 5 μm). The mobile phase consisted of methanol (90%), water (8%), and acetic acid (2%). The mobile phase was filtered through a 0.22 μm organic microporous filter membrane, and bubbles were removed by ultrasonic agitation. The column temperature was maintained at 30 °C, and detection was performed at a wavelength of 237 nm using a diode-array detector. The mobile phase flow rate was set at 1 mL/min, and the injection volume was 5 µL.
GC-MS analysis of biodegradation products of cis-abienol
To analyze the biodegradation products of cis-abienol, 15 mL of fermentation solution was combined with an equal volume of analytical-grade dichloromethane. The mixture was subjected to vortex extraction at 2,500 rpm for 10 min and then centrifuged at 10,000 rpm for 10 min. The lower organic phase was collected, and the extraction steps were repeated three times. The combined organic phases were dried over anhydrous sodium sulfate and left to stand overnight. Subsequently, 100 µL of 6-dichlorotoluene internal standard solution was added, and the solution was concentrated to 1 mL at atmospheric pressure. The final solution was filtered through a 0.22 μm organic filter membrane and transferred to a chromatographic vial for analysis43.
GC conditions:
The GC column used was HP-5MS 5% PhenylMethylSilox (30 m × 0.25 mm × 0.25 μm). The sample inlet temperature was set at 280 °C. High-purity helium served as the carrier gas at a flow rate of 1 mL/min. The temperature program was as follows: Initial temperature of 50 °C, maintained for 3 min. Increase to 130 °C at 6 °C/min, held for 3 min. Further increase to 180 °C at 4 °C/min, held for 2 min. Final increase to 280 °C at 3 °C/min, maintained for 5 min. The flow split ratio was set at 5:1, and the injection volume was 1 µL. The GC-MS analysis was performed using full-spectrum scanning.
MS analysis conditions: Transfer line temperature: 280 °C. Ion source temperature: 230 °C. Quadrupole electrode temperature: 150 °C. Ionization mode: 70 eV electron impact. Reaction lag time: 7 min. Mass-to-charge ratio (m/z) scan range: 50 to 700.
Measurement of cell growth of T2L strain in medium with cis-abienol as the sole carbon source
A single colony was picked from the LB medium plate and inoculated into 3 mL of LB liquid medium. The culture was incubated overnight at 150 rpm and 30 °C. The seed culture was then transferred into 20 mL of fermentation medium with an initial OD600 of 0.1. The fermentation was carried out at 150 rpm and 30 °C. Bacterial growth (OD600) was measured using a UV-Visible Spectrophotometer (UV-1500, Shanghai Macylab Instrument Co., Ltd).
To evaluate the effect of pH on bacterial growth and cis-abienol degradation, the pH of the fermentation medium was adjusted to 5.0, 6.0, 7.0, 8.0, or 9.0 using 0.1 M HCl or 0.1 M NaOH solutions.
Data availability
All sequencing reads obtained in this study have been deposited in the GenBank database (www.ncbi.nlm.nih.gov/genbank/) under accession numbers PV023313. The datas generated or analyzed during the current study are included in this article. The datasets generated or analyzed during the current study are available from the corresponding author on reasonable request.
References
Severson, R., Johnson, A. & Jackson, D. Cuticular constituents of tobacco: factors affecting their production and their role in insect and disease resistance and smoke quality. Recent. Adv. Tob. Sci. 11, 105–174 (1985).
Chen, W. et al. Effects of light quality on photosynthetic characteristics, carotenoids and surface extract content of tobacco leaves. Acta Ecol. Sinica. 31, 6877–6885 (2011).
Jing, Y. et al. Progress in the analysis of tobacco flavouring substances. Chin. Tob. Sci. 2, 44–48 (2005).
Duquesnoy, E. et al. Combined analysis by GC (RI), GC-MS and C-13 NMR of the supercritical fluid extract of Abies alba twigs. Nat. Prod. Commun. 5, 1995–1998 (2010).
Gray, P. S. & Mills, J. S. The isolation of abienol from Canada balsam, the Oleoresin of Abiesbalsamea(L.) mill. J. Chem. Soc. 1, 5822–5825 (1964).
Miyzawa, M. & Tamura, N. Characteristic odor components in the essential oil from Yacóntubers (Polymnia Sonchifolia poepp. Et Endl). J. Essent. Oil Res. 20, 12–14 (2008).
Heemann, V. et al. Composition of the leaf surface gum ofsome Nicotiana species and Nicotiana tabacum cultivarsi. Phytochemistry 22, 133–135 (1983).
Kenedy, B. S. et al. Leaf surface chemicals from nicotianaaffecting germination of Peronospora tabacina (adam) sporangia. J. Chem. Ecol. 18, 1467–1479 (1992).
Kessler, A. Defensive Finction of herbivore-induced plant volatile emissions in nature. Science 291, 2141–2144 (2001).
Sun, Y. et al. Study on the effect and mechanism of Cis-abienol inducing Nicotiana tabacum resistance to bacterial leaf blight. Chin. Tob. Sci. 44, 66–73 (2023).
Steede, W. T. et al. The tobacco trichome exudate Z-abienol and its relationshipwith plant resistance to phytophthora nicotianae. Plant. Dis. 101, 1214–1221 (2017).
Cheng, S. S., Chung, M. I., Lin, C. Y., Wang, Y. N. & Chang, S. T. Phytochemicals from cunninghamia Konishii Hayata act as antifungal agents. J. Agric Food Chem. 60, 124–128 (2012).
Wiliam, T. S. et al. The tobacco trichome exudate Z-abienol and its relationship with plant resistance to Phytophthora nicotianae. Plant. Dis. 101, 1214–1221 (2017).
Seo, S. et al. Identification of natural diterpenes that inhibit bacterial wilt disease in tobacco, tomato and arabidopsis. Plant. Cell. Physiol. 53, 1432–1444 (2012).
Zhang, S. et al. Comparative analysis of leaf tissue structure, glandular hairs and secretion of different roasted tobacco varieties (lines). Agric. Sci. Technol. 21, 132–140 (2019).
Wang, G. et al. Development and application of SNP functional markers for key tobacco abietadienol synthesis gene NtCPS2. Mol. Plant. Breed. 18, 8178–8186 (2020).
Li, Y., Zhang, H. & Wei Yi, Cui, H. Comparative analysis of glandular hair morphology and secretion characteristics of typical roasted tobacco varieties. Zhongguo Yancao Xuebao. 23, 84–91 (2017).
Li, X., Kang, C. & Bin, J. Research progress on the degradation law of aromatic compounds in tobacco leaves. Anhui Nongye Kexue 50, 21–26 (2022).
Sallaud C. et al. Characterization of two genes for the biosynthesis of the Labdanum diterpene in tobacco (Nicotiana tabacum) glandular trichomes. Plant J. 72, 1–17 (2012).
Peng, G. & Bai, X. The degradation of diterpenoids and its application in tobacco flavor. Food Ind. 30, 52–54 (2009).
Annonis, D. P. Flower Oils and Floral Compounds in Perfumery (ed. Renjie, C.) 257 (Flavour Fragrance J, 1994).
Xie, J. Tobacco Flavor Technology: Principles and Applications (ed. Xie, J.) 376 (Chemical Industry, 2009).
Cheng, H. A brief history of Ambergris. Daziran Tansuo. 1, 72–77 (2022).
Wang Wenjun, C. & Dai Qianhuan. Research progress on the composition of Ambergris and the synthesis of ambrox. Youji Huaxue. 1, 167–172 (2001).
Huang, T., Wang, J. & Fu, Y. Research progress on regulation mechanism of diterpenoid metabolism in tobacco. Zhongguo Yancao Xuebao. 25, 105–110 (2019).
Hieda, T. et al. Microbial transformation of the labdanum cis-Abienol and sclareol. Agric. Biol. Chem. 47, 243–250 (2014).
Wang, X., Zhang, X., Yao, Q., Hua, D. & Qin, J. Comparative proteomic analyses of hyphozyma Roseonigra ATCC 20624 in response to sclareol. Braz J. Microbiol. 50, 79–84 (2019).
Kouzi, S. A. & McChesney, J. D. Hydroxylation and glucoside conjugation in the microbial metabolism of the diterpene sclareol. Xenobiotica 21, 1311–1323 (1991).
Yang, J., Huang, H. & Ren, Y. Degradation of p-nitrobenzene and 4-chloronitrobenzene by Klebsiella oxytoca. Huanjing Gongcheng Xuebao. 7, 4607–4613 (2013).
Yang, L. et al. Mechanism of Klebsiella oxytoca improving saline-alkali tolerance of maize seedlings. Zhiwu Yingyang Yu Feiliao Xuebao. 27, 1044–1054 (2019).
Stenow, R., Robertson, E. K. & Whitehouse, M. J. Single cell dynamics and nitrogen transformations in the chain forming diatom Chaetoceros affinis. ISME J. 17, 2070–2078 (2023).
F. A. B. et al. ChemInform abstract: synthesis of nor-Ambreinolide from (+)-cis-Abienol. ChemInform 25, 41 (1994).
Barrero, F. A. et al. Degradation of the side chain of (–)-Sclareol: A very short synthesis of nor ‐Ambreinolide and ambrox. Synth. Commun. 34, 3631–3643 (2004).
Paul, W., Helmar, G. & HansDieter, A. Multigram-Scale total synthesis of (±)-Ambreinolide by a diastereoselective polyene cyclization. Bioorg. Med. Chem. Lett. 26, 6 (2022).
Wang, Y. et al. Research progress on the application of Ambergris aroma Raw materials in cigarette and synthesis of ambrox. Hecheng Huaxue. 26, 55–65 (2018).
Barrero, A. F. et al. Synthesis of ambrox from sclareol and(+)-Cis-Abienol. Tetrahedron 49, 10405–10412 (1993).
Bolster, M. G. The synthesis of (-)-Ambrox strarting from Labdanolic acid. Tetrahedron 57, 5657–5662 (2001).
Koyama, H., Kaku, Y. & Ohno, M. Synthesis of ambrox from L-Abietic acid. Tetrahedron Lett. 28, 2863–2866 (1987).
Huang, S. et al. Isolation and identification of cilantogenic bacteria degraded by castriene-4, 6-diol in tobacco leaves. Qinggong Xuebao. 32, 73–80 (2017).
Severson, R. F. et al. Quantitation of the major cuticular componentsfrom green leaf of different tobacco types. J. Agric. Food Chem. 32, 1023–1030 (1984).
Ding, L., Xie, F., Zhao, M., Xie, J. & Xu, G. Quantification of cis-abienol in Orientaltobacco leaves by LC. Chromatographia 66, 529–532 (2007).
Fu, Q. et al. Determination of cis-abienol in tobacco by ultra-high performance liquid chromatography. Yancao Keji. 53, 64–68 (2020).
Huang, S. et al. Screening and identification of cedratriene-4, 6-diol degrading bacteria and their application in reconstituted tobacco. J. Henan Agric. Sci. 47, 137–142 (2018).
Acknowledgements
This work was supported by Joint Development Project of Henan Tobacco Company (No.AW2023014).
Author information
Authors and Affiliations
Contributions
L.H. and X.Z. participated in the design of this study; X.Z., G.X., X.H., W.Q. and H.C. performed all the experiments; L.Z. and K.W. analyzed the data; L.H., X.Z. and S.H. wrote manuscript; all authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Han, L., Zhang, X., Xu, H. et al. Isolation and identification of the cis-abienol degrading strain Klebsiella oxytoca T2L and its aroma products. Sci Rep 15, 21824 (2025). https://doi.org/10.1038/s41598-025-07179-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-07179-z









