Abstract
Our study aimed to explore the calcification patterns of culprit lesions associated with artery-to-artery embolism (AAE) infarction in patients with vertebrobasilar artery atherosclerosis, using vessel wall magnetic resonance imaging (VW-MRI) and computed tomography (CT). We retrospectively analyzed data from patients undergoing VW-MRI and CT scans at two institutions. Patients with ≥ 50% vertebrobasilar atherosclerotic stenosis were categorized based on their stroke mechanisms as AAE (+) and AAE (-). Calcification profiles of the culprit lesion, including the morphology, location, quantity, and VW-MRI characteristics were compared among stroke patients. Multivariate logistic regression model was used to evaluate the relationship between vertebrobasilar artery calcification characteristics and the presence of AAE (+). Among the 151 stroke patients included, 56 (37.09%) were classified as AAE (+), while 95 (62.91%) as AAE (-). Multivariate regression analysis revealed the odds ratio of AAE (+) was 2.69 and 2.15 for the presence and count of spotty calcification, 3.33 for intimal-predominant calcification, and 3.65 for multiple calcifications. Calcification characteristics observed at the culprit lesion were associated with posterior circulation infarction in patients exhibiting AAE infarction. Understanding the relationship between intracranial artery calcification and stroke mechanisms may help clinicians improve stroke prevention and treatment.
Similar content being viewed by others
Introduction
Intracranial atherosclerotic disease is a significant cause of ischemic stroke globally, responsible for 40% of strokes in Asian populations. Ischemic strokes occur in the posterior circulation in approximately 20–30% of scenarios1. Intracranial atherosclerotic stroke in the posterior circulation could be caused by various mechanisms, including artery-to-artery embolism (AAE), branch artery occlusion (BAO) and hemodynamic impairment (HI)2. Among all the mechanisms, AAE, indicated by the presence of multiple infarctions, has been associated with a higher risk of stroke recurrence3. Moreover, apart from being alone, AAE commonly coexisted with HI, displaying a mutually interactive pathophysiology4. Understanding the characteristics of intracranial atherosclerosis that potentially contribute to AAE can aid in improving the secondary prevention and treatment of ischemic stroke.
There are several studies that substantiated the association between extracranial artery calcification and thromboembolic disease5,6. Although the underlying mechanism remains elusive, it is suggested that the risk of plaque rupture and erosion was elevated, potentially culminating in thrombus formation and subsequent embolic occurrences7. Intracranial artery calcification could not only reflect intracranial atherosclerotic burden but also cause hemodynamic impairment8. Recently, specific types of intracranial calcifications, such as spotty calcification and intimal predominant calcification, have been reported to be associated with an increased risk of downstream stroke and plaque instability. So far, there exists a scarcity of data regarding the role of calcified plaques localized at the culprit lesion within intracranial arteries, as well as their contribution to stroke pathogenesis.
High-resolution vessel wall magnetic resonance imaging (VW-MRI) enables direct assessment of intracranial atherosclerosis, facilitating the identification of various stroke mechanisms9. Nevertheless, MRI exhibits constrained resolution capabilities in delineating the interface between calcification and concomitant plaque components. CT remains the gold standard for visualizing and characterizing calcifications. The integration of VW-MRI and CT enables a comprehensive evaluation of intracranial atherosclerosis, including its morphology and composition. Based on this background, we aimed to investigate the characteristics of culprit lesions attributed to the mechanism of AAE (+) in patients with vertebrobasilar artery atherosclerosis using multi-modality imaging methods.
Results
Patient characteristics
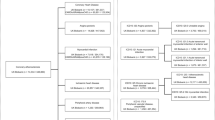
The flow chart of patient selection is shown in Fig. 1. Among the 151 stroke patients in the study, the most common stroke mechanisms were BAO (32.45%) and HI (27.15%), followed by AAE (17.88%), and a mixed mechanism of AAE + HI (17.88%). Only 5 (3.31%) patients were classified as BAO + HI and 2 (0.66%) patients as AAE + BAO + HI. None were classified as AAE + BAO. In total, 56 patients had an AAE (+) mechanism, and 95 patients had AAE (-) mechanism. The median interval between symptom onset and VW-MRI was 20 days (ranging from 10 to 28 days). Clinical characteristics of the study population are summarized in Table 1. Among the risk factors, hypertension was more prevalent in the AAE (+) group than in the AAE (-) group (96.4% vs. 87.4%, p = 0.083). There were no significant differences in medications and laboratory data between the two groups.
Flowchart of patient selection process. AAE artery-to-artery embolism, BAO branch artery occlusion, HI hemodynamic impairment, VW-MRI vessel wall magnetci resonance imaging.
Comparison of VW-MRI variables at culprit lesions
A total of 70 (46.36%) plaques were in the BA and 81 (53.64%) in the VA. The prevalence of AAE (+) was significantly higher in the VA compared to the BA (66.07% vs. 46.32%, p = 0.028). Stenosis degree was similar between the AAE (+) and the AAE (-) groups (p = 0.434). Among patients with culprit lesions in the VA, the degree of stenosis in contralateral vertebral lesions did not exhibit a statistically significant difference between the AAE (+) and AAE (-) groups (median 0, interquartile range [IQR] 0-50.08% vs. 33.51%, 0-74.59%, p = 0.106). No statistically significant differences were found in surface irregularity, T1 hyperintensity, PL, RI, signal heterogeneity (CVpre and CVpost) and CE% between the two groups (all p > 0.05) (Table 2). However, compared with the AAE (-) group, plaques in the AAE (+) group had a larger MinWT (median 1.10, IQR 0.78–1.54 vs. 0.87, 0.68–1.35, p = 0.043) and marginally larger NWI (median 91.55, IQR 86.27–93.49 vs. 89.44, 83.34–92.77, p = 0.072).
Comparison of calcification characteristics at culprit lesions
In comparing calcification characteristics, the AAE (+) group demonstrated a higher prevalence of spotty calcification than the AAE (-) group. Notably, the number of spotty calcifications at the culprit lesion in the AAE (+) group was, on average, 2.5 times as many as the AAE (-) group (p = 0.002). As shown in Fig. 2, a trend was found in the spatial distribution pattern of spotty calcification that AAE + HI and AAE had significantly larger amounts of spotty calcifications compared with AAE (-) (e.g., HI and BAO). The AAE (+) group also demonstrated a significantly higher presence of intimal predominant calcification and multiple calcifications than the AAE (-) group, with no difference in the presence of IEL predominant calcification. The total volume of calcification at the culprit lesion, including both calcified and non-calcified plaques at all evaluated locations, was also larger in the AAE (+) group, although there were large individual variations within each group [AAE (+) vs. AAE (-): 33.50 ± 56.66 vs. 13.55 ± 38.73, p < 0.001] (Table 2).
Frequency of spotty calcification, intimal or IEL predominant calcification, and single or multiple calcifications across various stroke mechanisms. There was a trend of higher prevalence of spotty calcification, intimal predominant calcification and multiple calcifications in cases of AAE + HI and AAE alone. AAE artery-to-artery embolism, BAO branch artery occlusion, HI hemodynamic impairment.
Relationship between calcification characteristics and AAE (+) infarction
Univariable and multivariable logistic regression models are shown in Table 3. Logistic regression analysis demonstrated that spotty calcification, intimal predominant calcification, and multiple calcifications were associated with an increased risk of AAE (+) infarction (odds ratio [OR] of 2.69, 3.33, and 3.65, respectively) when adjusted for clinical risk factors and plaque variables separately (Figs. 3 and 4). The total number of spotty calcifications from the culprit lesion was also positively associated with AAE (+) infarction (OR of 2.15).
Representative case of AAE (+) stroke in a 61-year-old male patient. DWI (A) demonstrated multiple scattered infarcts seen at the left occipital lobe (arrowhead, restricted diffusion). Post-contrast vessel wall MRI acquired in the coronal plane (B) demonstrated the most significant stenosis with wall thickening at the proximal basilar artery. Pre-contrast (C) and post-contrast (D) vessel wall MRI images show an eccentric plaque at the corresponding site in the axial images(arrow). Axial (E) and coronal (F) CT images reveal the presence of spotty and intimal predominant calcification (arrow) in the corresponding image slices.
Representative case of a 67-year-old male patient presenting with hemodynamic impairment infarction. Diffusion-weighted imaging (A) demonstrated territory infarcts of the left cerebellum. Post-contrast vessel wall MRI in coronal view (B) revealed diffuse wall thickening in the V4 segment of the left vertebral artery. Pre-contrast (C) and post-contrast (D) vessel wall MRI images showed the presence of a plaque in the left vertebral artery (arrow). Axial (E) and coronal (F) CT images demonstrated the presence of IEL predominant calcification (arrow) in the corresponding image slices.
Association between T1 hyperintensity and calcification
Univariable logistic regression demonstrated no statistically significant association between T1 hyperintensity and the presence or count of spotty calcification (OR, 1.09; 95% confidence interval [CI], 0.52–2.31; p = 0.815 and OR, 1.14; 95% CI, 0.78–1.66; p = 0.509), intimal predominant calcification (OR, 1.64; 95% CI, 0.73–3.69; p = 0.236), or multiple calcifications (OR, 1.76; 95% CI, 0.79–3.92; p = 0.169).
Subgroup analysis of AAE (+)
Patients with AAE (+) were divided into 3 groups: AAE alone (n = 27), AAE + HI (n = 27), and AAE + BAO + HI (n = 2). The AAE + BAO + HI group did not achieve statistical power and was therefore excluded from further analysis. The subgroup analysis between AAE alone and AAE + HI is shown in Table 4. The AAE + HI group demonstrated significantly larger PL and higher CVpre than the AAE alone group in the VW-MRI assessments. All the calcification characteristics were similar between the group of AAE alone and AAE + HI.
Inter-reader reliability of VW-MRI
Inter-reader agreement for qualitative analysis (presence of T1 hyperintensity and surface irregularity) were good to excellent (κ: 0.70 to 0.94). Inter-reader reliability (ICC) for quantitative measurements were good to excellent (MaxWT, 0.87; MinWT, 0.85; LA, 0.70; OWA, 0.96).
Discussion
In this study, we investigated the association between calcification patterns at the culprit lesion and AAE infarction in the vertebrobasilar artery. We found that spotty calcification, intimal predominant calcification, and multiple calcifications were independently associated with a higher risk of having caused AAE infarction, both before and after conducting the multivariable analysis. Subgroup analysis did not reveal significant differences in calcification characteristics between AAE alone and AAE + HI. Our study indicated that the morphology, location, and quantity of calcification may contribute to the occurrence of AAE infarction.
The role of calcification at the culprit lesion in the intracranial artery is rarely reported. Wu et al. reported that intracranial internal carotid artery calcification was associated with downstream embolic stroke, but its effect on the culprit’s vessels is poorly understood10. This is mainly because MRI has limited spatial resolution for detecting intracranial artery calcification with variable signal characteristics. Our study entailed a comparative image analysis of CT and VW-MRI scans, aimed at assessing calcification within atherosclerotic plaques and elucidating the associated stroke mechanisms. Comprehensive evaluations of calcification are particularly feasible in the straight vertebrobasilar artery. Furthermore, the combined application of VW-MRI has enhanced physicians’ accuracy in identifying the specific underlying cause of stroke. For instance, BAO could be identified by the presentation of plaque at the beginning of a penetrating artery11. Non-atherosclerotic etiologies of hemodynamic insufficiency, such as dissection, dysplasia, and embolism, can be excluded using VW-MRI12,13.
The impact of calcification on atherosclerotic plaques remains a subject of debate. Some researchers proposed that calcification contributes to plaque stabilization14whereas others contend that it serves as a marker for increased risk of thromboembolic disease15. Our research reported a correlation between spotty calcification, which signifies the small but discernible calcification unit on CT imaging, and AAE infarction. Despite reports associating spotty calcification with plaque instability in coronary, carotid, and intracranial arteries16,17the underlying mechanisms remained poorly understood. Pathological analysis revealed a significant correlation between the presence of small, dispersed calcifications and the occurrence of plaque rupture18. Notably, these calcifications were significantly smaller in size compared to the spotty calcifications observed on CT imaging. Positron emission tomography provided evidence that spotty calcification indicated an active atherosclerotic process19. Subsequently, inflamed endothelial activity can intensify thrombus formation, resulting in the occurrence of downstream micro-emboli.
To our knowledge, our research represents the initial investigation of the association between the localization of intracranial artery calcification and AAE within the intracranial circulation. Biomechanical factors may contribute to the relationship between superficial calcification and symptomatic manifestation in the carotid artery20. Our study extends to the possibility that the focal and dot-like patterns evident in the superficial intimal layer may elevate wall shear stress within the intracranial artery. In contrast, calcification with deep and circumferential patterns correlate with reduced wall shear stress, and do not entail an elevated risk of embolic stroke. Calcification within the IEL and medial layers of the vessel wall could contribute to decreased arterial compliance and distensibility, ultimately resulting in arterial stiffening.
In our study, we observed a significant association between multiple calcifications and an elevated risk of AAE infarction. Histological studies indicated that intimal and IEL calcification frequently occurred simultaneously21. Consequently, the presence of multiple calcifications is more likely to indicate intimal atherosclerosis compared to single calcification. Furthermore, previous research suggested a correlation between specific calcification features and intraplaque hemorrhage22. Nevertheless, our findings failed to demonstrate a significant association between calcification characteristics and intraplaque hemorrhage (i.e., T1 hyperintensity). Histological variations between intracranial and carotid arteries may underlie this discrepancy23. The thick layer of external elastic lamina in the carotid artery encapsulates plaque components, while the thick layer of IEL in the intracranial artery separates atherosclerosis from non-intimal layer calcification.
In the previous studies, several plaque characteristics on VW-MRI were found to be associated with AAE. Wu et al. proposed that hyperintensity plaques observed in the middle cerebral artery on T1-weighted images were correlated with artery-to-artery embolic infarction24. Hou et al. observed a correlation between intraplaque enhancement and AAE within the vertebrobasilar artery25. The inconsistency with our results may be due to the inclusion criteria, as we included not only AAE but also other mixed mechanisms. Since AAE and hypoperfusion frequently coexist in clinical contexts, exhibiting interconnected pathophysiology, our inclusion criteria may have substantial clinical significance.
Interestingly, several differences in VW-MRI plaque measurements were observed in the comparison within AAE (+) subgroups, except for calcification characteristics. The mixed mechanism of AAE + HI demonstrated a larger plaque length than the AAE alone. The lesions caused by AAE + HI tend to be elongated rather than focal, indicating a more extensive involvement of the compromised endothelium. CVpre can reflect the heterogeneity of signal intensity within atherosclerotic plaques. Our findings indicated a correlation between increasing CVpre values, which may signify a more advanced and heterogeneous stage of atherosclerosis, and hemodynamic insufficiency. Future studies should be performed on the value of CVpre as a marker of clinical severity in patients with ischemic stroke.
The study had several limitations. Firstly, the inclusion of only patients with complete VW-MRI and CT examinations could introduce selection bias, which is inherent in retrospective studies. Secondly, as this is a cross-sectional study, our study is limited to examining the potential association between plaque characteristics and ischemic stroke at a specific point in time. Future longitudinal studies are necessary to ascertain the robustness of our findings. Thirdly, hypoperfusion is defined based on previously validated standards. Quantitative Magnetic Resonance Angiography is recommended to quantify the correlation between blood flow and plaque instability in future studies. Fourthly, the field of view was limited in several cases and therefore, not included in the study. Fifthly, our study was confined to the posterior circulation. Future investigations should be conducted in the anterior circulation, including the intracranial internal carotid artery and middle cerebral artery.
In conclusion, we have identified distinct characteristics in the lesions of patients with AAE compared to those without AAE, and between patients with AAE alone and those with AAE accompanied by HI. Different types of calcifications present at the culprit lesion site may provide valuable clues to understanding the etiologies of strokes affecting the vertebrobasilar artery.
Methods
Study patients
The local institutional review board at Shandong Provincial Hospital and Qilu Hospital granted approval, and informed consent was waived for this retrospective study. The research was conducted in accordance with the Declaration of Helsinki. From October 2018 and June 2024, all the patients who underwent VW-MRI and CT within 2 weeks at both institutions were reviewed. Only the patients with acute/subacute cerebral infarction in the territory of the vertebrobasilar artery (VBA) were included. Exclusion criteria consisted of (i) patients without substantial VBA stenosis (< 50%) with preceding VW-MRI26(ii) evidence of non-atherosclerotic intracranial vascular pathology (e.g., vasculitis, dissection, and reversible cerebral vasoconstriction syndrome), (iii) evidence of cardiac sources of emboli (e.g., atrial fibrillation and prosthetic cardiac valve), (iv) patients with a history of stent or treatment in the posterior circulation, and (v) inadequate image quality based on the vessel wall delineation and artifacts.
MRI examination
MRI exams were performed on 3T MR imaging scanners (Ingenia, Philips Healthcare; MAGNETOM Prisma, Siemens Healthineers) with either an 8-channel or 16-channel head coil. VW-MRI was performed using T1-volumetric isotropic turbo spin echo acquisition (T1-VISTA) or T1-sampling perfection with application-optimized contrasts using different flip angle evolutions (T1-SPACE). The parameters of T1-VISTA were as follows: time of repetition (TR)/time of echo (TE), 425 ms/19 ms; field of view (FOV), 220 mm × 220 mm; matrix, 312 × 312; acquired spatial resolution: 0.7 × 0.7 × 1.1 mm; and scanning time, 6.1 min. The parameters of T1-SPACE were as follows: TR/TE, 590 ms/15 ms; FOV, 130 mm × 180 mm; matrix, 256 × 256; acquired spatial resolution: 0.9 × 0.9 × 0.9 mm; and scanning time, 5 min. A plaque was considered culprit if the only or most stenotic lesion within the VBA territory of acute/subacute cerebral infarction. The presence or absence of surface irregularity and T1 hyperintensity were recorded. Quantitative measurements of the plaque had been present in our previous study15: (1) lumen area (LA), outer wall area (OWA), wall area (WA: OWA-LA), and normal wall index (NWI: WA/OWA*100%); (2) maximum wall thickness (MaxWT), minimum wall thickness (MinWT), and eccentricity index [EI: (MaxWT-MinWT) / MaxWT]; (3) plaque length (PL); (4) remodeling index (RI) was calculated as the ratio of the WA at the maximal lumen narrowing site to that at the reference site; (5) contrast-enhancement ratio (CE%) was calculated as Spost/Spre*100%, where Spost and Spre were the normalized signal intensities on post- and pre-contrast VW-MRI images, respectively; and (6) coefficient of variation (CV) was calculated as SD/SI*100%, where SD and SI were the standard deviation and signal intensity of plaques.
CT acquisition
CT protocol was conducted on 128-slice dual-source CT scanners (SOMATOM Force, Siemens Healthineers; SOMATOM Definition Flash, Siemens Healthineers). The CT protocol included non-contrast CT (NCCT), CT angiography (CTA), and 4D multi-phase CTA acquisition. For NCCT, tube voltage was 120 kV, and tube current was 200 mA. For CTA, tube voltages were 100 kV and Sn140 kV, and tube current was 260–300 mA. For 4D multi-phase CTA, tube voltage was 80 kV, and tube current was 150 mA. All CT images were reconstructed with a section thickness of 0.5 mm and an interval of 0.7 mm. Images were evaluated using a bone CT window setting (width of 1500 HU and center of 300 HU). Two radiologists independently reviewed all CT images, and a consensus was reached after disagreements. VW-MRI images were analyzed in both axial and coronal planes to identify the culprit plaques. Multiple planar reconstructions facilitated the alignment of VW-MRI and CT images with anatomical structures. Reconstructions perpendicular to the flow direction were performed to demonstrate the coexistence of culprit lesions and calcification. CT images with contrast agents administered (i.e., CTA or CTP) were used to differentiate between the vessel wall and lumen, if available. The presence of hyperdensity with a CT attenuation of 130 Hounsfield units (HU) or greater was identified as calcification. The presence and count of spotty calcifications were identified, indicating the presence of minute calcium deposits (less than 3 mm in diameter) within a sub-90-degree arc17. The location of calcification was classified as either intimal or internal elastic lamina (IEL) predominant, using a validated grading system. Calcification volume was quantified using the Medical Imaging Interaction Toolkit and PyRadiomics, as previously published27.
Stroke mechanisms
Two neuroradiologists (with 12 and 5 years of experience in neuroradiology, respectively) independently analyzed diffusion-weighted imaging (DWI), VW-MRI and clinical data. If disagreements occurred between the readers, a final decision was made through consensus. Stroke mechanisms were categorized as the following: artery-to-artery embolism (AAE), branch artery occlusion (BAO), hemodynamic impairment (HI), and multiple mechanisms4.
-
(1)
AAE is defined as the presence of two or more scattered hyperintense areas on DWI, located distally to the stenosed vessel in the territory of the relevant artery.
-
(2)
BAO is defined as infarcts localized to an area adjacent to the stenosed vessel, resulting from the occlusion of the orifice of one or more perforators that supply the region.
-
(3)
HI is defined as single or multiple infarcts in border zone (or watershed) areas and associated with perfusion deficits distal to the severely stenosed or occluded vessel.
-
(4)
4) Multiple mechanisms are categorized when multiple potential mechanisms could have contributed to the stroke.
Statistical analysis
Data were analyzed using SPSS 29.0 (Chicago, IL, USA). Categorical data were reported as frequencies and continuous data were expressed as means ± standard deviations or medians and interquartile range (IQR). Continuous variables between two groups were compared using the independent-sample t-test for normal distribution or the Mann-Whitney U test for non-normal distribution. The chi-square test was utilized to compare categorical variables. Multivariable logistic regression analysis was used to adjust for variables with p-value < 0.2 in univariate analysis. Reliability of categorical variables was evaluated using Cohen’s kappa test, while inter-reader agreements for continuous variables were assessed using the intraclass correlation coefficient (ICC). A two-tailed p-value of < 0.05 was considered as statistically significant.
Data availability
Data are available from the corresponding authors on reasonable request.
References
Banerjee, G., Stone, S. P. & Werring, D. J. Posterior circulation ischaemic stroke. BMJ 361, k1185. https://doi.org/10.1136/bmj.k1185 (2018).
Salerno, A., Strambo, D., Nannoni, S., Dunet, V. & Michel, P. Patterns of ischemic posterior circulation strokes: A clinical, anatomical, and radiological review. Int. J. Stroke. 17, 714–722. https://doi.org/10.1177/17474930211046758 (2022).
Kamel, H. et al. Cryptogenic stroke and nonstenosing intracranial calcified atherosclerosis. J. Stroke Cerebrovasc. Dis. 26, 863–870. https://doi.org/10.1016/j.jstrokecerebrovasdis.2016.10.035 (2017).
Feng, X. et al. Stroke mechanisms in symptomatic intracranial atherosclerotic disease: classification and clinical implications. Stroke 50, 2692–2699. https://doi.org/10.1161/STROKEAHA.119.025732 (2019).
Ntaios, G. et al. Aortic arch atherosclerosis in patients with embolic stroke of undetermined source: an exploratory analysis of the NAVIGATE ESUS trial. Stroke 50, 3184–3190. https://doi.org/10.1161/STROKEAHA.119.025813 (2019).
Cai, X., Geng, Y. & Zhang, S. The relationship between aortic arch calcification and recurrent stroke in patients with embolic stroke of undetermined Source-A Case-Control study. Front. Neurol. 13, 863450. https://doi.org/10.3389/fneur.2022.863450 (2022).
Sugiyama, T. et al. Calcified plaques in patients with acute coronary syndromes. JACC Cardiovasc. Interv. 12, 531–540. https://doi.org/10.1016/j.jcin.2018.12.013 (2019).
Wu, X. et al. Impact of intracranial artery calcification on cerebral hemodynamic changes. Neuroradiology 60, 357–363. https://doi.org/10.1007/s00234-018-1988-2 (2018).
Wu, F. et al. Differential features of culprit intracranial atherosclerotic lesions: A Whole-Brain vessel wall imaging study in patients with acute ischemic stroke. J. Am. Heart Assoc. 7 https://doi.org/10.1161/JAHA.118.009705 (2018).
Wu, X. H., Chen, X. Y., Fan, Y. H., Leung, T. W. & Wong, K. S. High extent of intracranial carotid artery calcification is associated with downstream microemboli in stroke patients. J. Stroke Cerebrovasc. Dis. 26, 442–447. https://doi.org/10.1016/j.jstrokecerebrovasdis.2016.10.007 (2017).
Ryoo, S., Lee, M. J., Cha, J., Jeon, P. & Bang, O. Y. Differential vascular pathophysiologic types of intracranial atherosclerotic stroke: A High-Resolution wall magnetic resonance imaging study. Stroke 46, 2815–2821. https://doi.org/10.1161/STROKEAHA.115.010894 (2015).
Wasserman, B. A. et al. Vessel wall imaging features of spontaneous intracranial carotid artery dissection. Neurology 102, e209250. https://doi.org/10.1212/WNL.0000000000209250 (2024).
Mossa-Basha, M. et al. Added value of vessel wall magnetic resonance imaging for differentiation of nonocclusive intracranial vasculopathies. Stroke 48, 3026–3033. https://doi.org/10.1161/STROKEAHA.117.018227 (2017).
Baek, J. H. et al. The protective effect of middle cerebral artery calcification on symptomatic middle cerebral artery infarction. Stroke 48, 3138–3141. https://doi.org/10.1161/STROKEAHA.117.017821 (2017).
Song, G. et al. Intimal predominant calcification is associated with plaque instability in the vertebrobasilar artery by vessel wall magnetic resonance imaging and computed tomography. Eur. J. Radiol. 168, 111132. https://doi.org/10.1016/j.ejrad.2023.111132 (2023).
Homssi, M. et al. Association between spotty calcification in nonstenosing extracranial carotid artery plaque and ipsilateral ischemic stroke. J. Am. Heart Assoc. 12, e028525. https://doi.org/10.1161/JAHA.122.028525 (2023).
Zhang, F. et al. Spotty calcium on cervicocerebral computed tomography angiography associates with increased risk of ischemic stroke. Stroke 50, 859–866. https://doi.org/10.1161/STROKEAHA.118.023273 (2019).
Shapiro, S. D. et al. Pathological correlates of brain arterial calcifications. Cardiovasc. Pathol. 38, 7–13. https://doi.org/10.1016/j.carpath.2018.09.003 (2019).
Vesey, A. T. et al. 18)F-Fluoride and (18)F-Fluorodeoxyglucose positron emission tomography after transient ischemic attack or minor ischemic stroke: Case-Control study. Circ. Cardiovasc. Imaging. 10, e004976. https://doi.org/10.1161/CIRCIMAGING.116.004976 (2017).
Zhongzhao, T. et al. How does juxtaluminal calcium affect critical mechanical conditions in carotid atherosclerotic plaque? An exploratory study. IEEE Trans. Biomed. Eng. 61, 35–40. https://doi.org/10.1109/TBME.2013.2275078 (2014).
O’Neill, W. C., Han, K. H., Schneider, T. M. & Hennigar, R. A. Prevalence of nonatheromatous lesions in peripheral arterial disease. Arterioscler. Thromb. Vasc Biol. 35, 439–447. https://doi.org/10.1161/ATVBAHA.114.304764 (2015).
Lin, R., Chen, S., Liu, G., Xue, Y. & Zhao, X. Association between carotid atherosclerotic plaque calcification and intraplaque hemorrhage: A magnetic resonance imaging study. Arterioscler. Thromb. Vasc. Biol. 37, 1228–1233. https://doi.org/10.1161/ATVBAHA.116.308360 (2017).
Yang, W. J., Wong, K. S. & Chen, X. Y. Intracranial atherosclerosis: from microscopy to High-Resolution magnetic resonance imaging. J. Stroke. 19, 249–260. https://doi.org/10.5853/jos.2016.01956 (2017).
Wu, F. et al. Hyperintense plaque on intracranial vessel wall magnetic resonance imaging as a predictor of Artery-to-Artery embolic infarction. Stroke 49, 905–911. https://doi.org/10.1161/STROKEAHA.117.020046 (2018).
Hou, Z. et al. Intraplaque enhancement is associated with Artery-to-Artery embolism in symptomatic vertebrobasilar atherosclerotic diseases. Front. Neurol. 12, 680827. https://doi.org/10.3389/fneur.2021.680827 (2021).
Qiao, Y. et al. Intracranial plaque enhancement in patients with cerebrovascular events on high-spatial-resolution MR images. Radiology 271, 534–542. https://doi.org/10.1148/radiol.13122812 (2014).
Yang, H. et al. Comparison of symptomatic vertebrobasilar plaques between patients with and without diabetes mellitus using computed tomographic angiography and vessel wall magnetic resonance imaging. Diab. Vasc. Dis. Res. 19, 14791641211073944. https://doi.org/10.1177/14791641211073944 (2022).
Author information
Authors and Affiliations
Contributions
H.Y. and B.L. developed the concept; H.Y., H.L., Q.Y., C.X., and L.L. performed the data analysis and wrote the manuscript; B.L. was responsible for research ethics and project management. All authors reviewed the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yang, H., Lu, H., Xue, C. et al. Association between vertebrobasilar artery calcification and artery-to-artery embolism in the posterior circulation. Sci Rep 15, 22689 (2025). https://doi.org/10.1038/s41598-025-07229-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-07229-6