Abstract
Multisensory integration (MSI) deficits in patients with Parkinson’s Disease (PD) have recently attracted growing interest in both clinical and research realms, MSI being crucial in maintaining well-coordinated responses to external stimuli. In this framework, we exploited the Rubber Hand Illusion (RHI), an experimental procedure that induces healthy individuals to embody a fake hand by integrating multisensory body-related cues, to address potential alterations of the embodiment phenomenon in PD. Specifically, we leveraged a visuo-tactile (VT; Experiment 1) and a visuo-motor (VM; Experiment 2) RHI version and applied it to 32 PD patients and age-matched controls. Classical RHI measures (i.e., ownership and agency questionnaires, proprioceptive drift) were collected. Questionnaires revealed typical VT- and VM-RHI patterns in patients, although diminished as compared to controls. Crucially, proprioceptive drift showed an abolishment of VM- (but not VT-) RHI effect in patients, advocating for a channel-specific embodiment abnormality that emerged only when the motor system, involved in the pathology, was challenged by the RHI. Interestingly, such a deficit emerged specifically when proprioceptive drift, engaging a motor response, was considered as a measure. We speculatively interpret this finding in light of the neural degeneration of basal ganglia in PD, being these structures engaged in visuo-motor integration.
Similar content being viewed by others
Introduction
Integrating multiple inputs from different sensory signals (i.e., multisensory integration) is a fundamental ability that promotes survival, allowing well-coordinated responses to external stimuli. Thereby, multisensory integration has been evaluated in different pathological contexts, especially in those affecting motor interactions with the environment, such as Parkinson’s Disease (PD).
PD is a progressive neurodegenerative condition associated with the neural degeneration of dopaminergic nuclei in the basal ganglia, that results in a multivariate pattern of motor symptoms ranging from bradykinesia, resting tremor, and rigidity1. Besides these evident motor deficits, other more subtle symptoms have been described, among which sensory and multisensory integration impairments have recently gained attention both in clinical and research fields. Concerning unisensory processing, capitalising on reaching tasks, a decrease in proprioceptive accuracy has been reported in PD2,3. Note that, according to clinical studies investigating these proprioceptive deficits, both position sense (i.e., the ability to perceive the body position in space) and kinaesthesia (i.e., the perception of the body movement) might be impaired in these patients4. Coherently, it has been largely shown that PD is associated with proprioceptive deficits affecting multiple functions5, especially when visual feedback is not allowed6. Previous studies highlighted that inadequate proprioceptive information has deleterious effects on postural orientation in these patients7,8,9,10. For instance, when performing a task that does not allow to compensate with vision for unreliable proprioception, subjects with Parkinson’s Disease show decrements in postural adjustments in response to external perturbations11,12,13,14. Furthermore, larger temporal binding windows have been described during temporal discrimination tasks, suggesting abnormal processing of not only unisensory15,16,17,18,19 but also multisensory20 stimulations in PD. As concerns multisensory processing, by leveraging pointing21 and gait control tasks22, a growing body of literature has also highlighted an evident impairment in integrating visual and kinesthesic inputs, especially when patients produce voluntary movements towards a seen target or during balance control while walking. In line with these results, it has been recently proposed that the visuo-motor integration deficit might underlie a critical issue for patients with PD, namely the high risk of falling, and should therefore be considered in the rehabilitation program22.
Given the prominent relevance that multisensory integration deficits have been afforded within the study of PD, here, we aimed to investigate whether the embodiment phenomenon, which heavily relies on the integration of stimuli from different sensory modalities, is altered in these patients. Thus, we leveraged both the classical visuo-tactile Rubber Hand Illusion paradigm [VT-RHI; Experiment 1;23 and, considering the motor dysfunction that characterises PD, a visuo-motor version of the procedure [VM-RHI; Experiment 224.
During the VT-RHI23, participants observe touches occurring on a fake (visible) hand while perceiving synchronous touches on their own (hidden) hand. This visuo-tactile stimulation adheres to a fundamental principle of multisensory integration: the temporal rule, which states that optimal integration occurs when stimuli are synchronous25. Accordingly, the synchronous visuo-tactile stimulations applied during the RHI are integrated as if they belonged to the same source, i.e., the own hand26,27,28,29. In other words, as a consequence of visuo-tactile integration, a body ownership alteration is observed, so that participants feel as if the fake hand were their own (here referred to as the “embodiment” phenomenon). Moreover, the synchronous procedure causes a proprioceptive re-localization of the own hand, which is felt as shifted towards the fake hand (i.e., proprioceptive drift). Similarly, in the VM-RHI24, visuo-motor inputs generated during the synchronous movement of the participant’s (hidden) hand and the (visible) fake one are integrated as if they originated from the same source, by remapping the felt motion onto the observed fake hand. As in the VT-RHI, a proprioceptive drift towards the fake hand is observed, altogether with the body ownership alteration. Note that, in the VM-RHI, besides such alteration, another modulation occurs. Specifically, the embodiment phenomenon also involves the sensation of motor control over the fake hand’s movements, reflecting an alteration of the sense of agency24.
In both paradigms, a typically employed control condition consists in touching (VT-RHI) or moving (VM-RHI) the fake hand in anti-phase with the participant’s hand (i.e., asynchronous condition)23,24. Since, in this condition, the temporal rule of multisensory integration is violated, the visual input is not integrated with the tactile (VT-RHI) or the motor (VM-RHI) one, thus not eliciting the illusion. This control condition specifically manipulates a “bottom-up” constraint of embodiment by tackling the temporal rule of multisensory integration. In other control conditions, violating “top-down” embodiment constraints, the fake hand’s position is manipulated (e.g., by placing it in allocentric perspective) so that its body-incongruent posture prevents the body ownership alteration25. The employment of adequate control conditions is crucial to exclude confounding effects, since, for instance, proprioceptive drift is known to be heavily influenced by visual capture of proprioception30.
Thereby, in both visuo-tactile and visuo-motor procedures, the RHI effect is measured when significantly greater proprioceptive drift and feeling of embodiment are observed in synchronous as compared to asynchronous conditions23,24,31.
In the present study, by systematically employing these similar RHI procedures, we aimed to elucidate whether embodiment processes, being a consequence of visuo-tactile (Experiment 1) or visuo-motor (Experiment 2) integration, are altered in PD (see Fig. 1). Specifically, we investigated such possible alterations relying on the classical RHI measures, namely ownership and agency questionnaires, as well as proprioceptive drift.
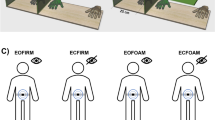
Experimental procedures. A represents the experimental procedure for each experimental condition. Before each condition, 10 proprioceptive judgements were collected by asking participants to point to the tested hand position, while being blindfolded. Then, 3 min of RHI was performed. Finally, the proprioceptive judgement procedure was repeated, and questionnaires were administered. In separate trials, during the RHI procedure, different VT- and VM-RHI conditions were performed. Note that, besides 16 age-matched controls, 32 PD patients were recruited. Specifically, 16 right and 16 left exordium PD patients were tested on both their right and left hands in order to account for possible exordium side-dependent confounds. B shows a schematic representation of the visuo-tactile RHI conditions performed in Experiment 1, while C shows those of the visuo-motor RHI performed in Experiment 2.
To the best of our knowledge, only a limited number of studies have investigated embodiment deficits in PD using the RHI paradigm, and exclusively in its visuo-tactile version32,33,34. When synchronous and asynchronous VT-RHI were delivered to PD patients, a diminished effect was found as compared to controls32,33,34. More precisely, patients showed a reduced difference between RHI conditions, due to both a decreased RHI effect in the synchronous condition32 and an increased effect in the asynchronous control one32,33,34. The authors interpreted the latter result in light of the extended multisensory temporal binding windows in PD20,33, which rendered the two RHI conditions essentially similar for the patients. Finally, an overall increase in proprioceptive drift was observed, irrespective of the VT-RHI condition. The authors explained this finding as potentially influenced by a proprioceptive confound, since position sense alterations are often observed in PD patients. Against this background, to collect a reliable proprioceptive drift, we recruited PD patients with no proprioceptive deficit. Furthermore, we inserted a 1s delay between the stimulation on the fake and the participant’s hand in the control conditions, to ensure that patients could perceive the asynchrony between tactile or motor sensations and the visual ones.
Based on the classical RHI literature, as well as the multisensory integration deficits reported in PD, we formulated the following predictions. If embodiment, heavily depending on multisensory integration phenomena, is altered in PD, we predicted a decrease of the RHI effect in the synchronous condition in the PD group as compared to the control one, possibly leading to the abolishment of the typically observed difference between synchronous and asynchronous conditions. Otherwise, the typical pattern should emerge both in PD patients and controls. Secondly, if such an embodiment deficit depends on the specific sensory modalities involved in the integration processes inducing the illusion, we predicted that the typical pattern would be abolished in only one of the two procedures, i.e., possibly the VM-RHI, considering the prevalence of visuo-motor integration deficits in PD.
Results
VT-RHI results (Experiment 1)
To compare the VT-RHI effect between PD patients and controls, we ran 2 × 2 repeated-measures ANOVAs with condition (synchronous, asynchronous) as within-subject factor and group (controls, PD patients) as between-subject factor, both for the ownership score (i.e., mean rating to the real items minus mean rating to the control items of the ownership questionnaires) and the proprioceptive drift (i.e., mean proprioceptive judgement collected at the end of the RHI procedure minus mean proprioceptive judgement collected at the beginning of the RHI procedure).
As regards ownership score, significant main effects of group (F1,46=4.99; p = 0.03; η2p = 0.1) and condition (F1,46=75.7; p = 0.00000000003; η2p = 0.62) were found, indicating greater ownership score, respectively, in the controls’ group as compared to the patients’ one and in the synchronous condition than in the asynchronous one. Moreover, we found a significant condition x group interaction (F1,46=5.26; p = 0.03; η2p = 0.1; see Fig. 2A). Duncan post hoc tests exploring the interaction revealed a significantly greater ownership score in the synchronous as compared to the asynchronous condition in both groups (controls’ group: p = 0.00006; patients’ group: p = 0.0001), in line with the significant main effect of condition. More interestingly, a significantly greater ownership score was observed in the synchronous condition of the controls’ group as compared to that of the patients’ group (p = 0.002), possibly explaining the interaction effect. No difference between groups emerged in the asynchronous condition (p = 0.60). For completeness, we also report the remaining comparisons: asynchronous controls vs. synchronous PD patients: p = 0.003, synchronous controls vs. asynchronous PD patients: p = 0.00005.
Concerning proprioceptive drift, results showed only a significant main effect of condition (F1,46=15.88; p = 0.0002; η2p = 0.26; see Fig. 2B), indicating greater proprioceptive drift in the synchronous than in the asynchronous condition irrespective of the group. No other significant effect was found (main effect of group: F1,46=0.22; p = 0.64; η2p = 0.005; condition x group interaction: F1,46=0.14; p = 0.71; η2p = 0.003).
All in all, our results showed the presence of a VT-RHI effect (synchronous > asynchronous) both in patients’ and controls’ groups. Moreover, PD patients showed a diminished effect compared to the controls’ group in the synchronous condition.
Experiment 1 (VT-RHI) results. The figure shows the plot of the Condition x Group interaction, concerning ownership questionnaire (A) and proprioceptive drift measures (B). In all boxplots, the whiskers represent the minimum and the maximum value, the limits of the box represent the first and the third quartile, the median is depicted by the line that divides the box into two parts, while dots represent individual participants. Black asterisks indicate significant post-hoc comparisons exploring the interaction (**p < 0.005; ***p < 0.0005). Grey asterisks represent the significant main effect of Condition (***p < 0.0005; ******p < 0.0000005).
VM-RHI results (Experiment 2)
In the VM-RHI analyses, besides ownership score and proprioceptive drift, we also computed an agency score (i.e., mean rating to the real items minus mean rating to the control items of the agency questionnaires). To compare the VM-RHI effect between PD patients and controls, we ran 2 × 2 repeated-measures ANOVAs, one for each RHI measure (i.e., ownership score, agency score, and proprioceptive drift) with condition (synchronous, asynchronous) as within-subject factor and group (controls, PD patients) as between-subject factor.
As regards the ownership questionnaire, a significant main effect of condition was found (F1,46=60.06, p = 0.0000000007, η2p = 0.57), indicating a higher ownership score in the synchronous condition than in the asynchronous one. Interestingly, a significant condition x group interaction emerged (F1,46=7.81, p = 0.008, η2p = 0.15; see Fig. 3A). Duncan post hoc tests exploring the interaction showed a significantly greater ownership score in the synchronous as compared to the asynchronous condition in both groups (controls’ group: p = 0.00006; patients’ group: p = 0.001), in line with the significant main effect of condition. Crucially, a significantly higher ownership score was observed in the synchronous condition of the controls’ group as compared to the same condition of the patients’ group (p = 0.01), possibly explaining the interaction effect. No difference between the two asynchronous conditions was found (p = 0.46). For completeness, we also report the remaining comparisons: asynchronous controls vs. synchronous PD patients: p = 0.0008, synchronous controls vs. asynchronous PD patients: p = 0.00005. No significant main effect of group was found (F1,46=1.21; p = 0.28; η2p = 0.03).
Regarding the agency questionnaire, we found a significant main effect of condition (F1,46=93.98, p = 0.000000000001, η2p = 0.67), revealing a greater agency score in the synchronous condition as compared to the asynchronous one. Importantly, a significant condition x group interaction emerged (F1,46=18.76; p = 0.00008; η2p = 0.29; see Fig. 3B). Duncan post hoc tests exploring the interaction showed, alongside a significant difference between synchronous and asynchronous conditions within each group (controls’ group: p = 0.00006; patients’ group: p = 0.0005), a significantly higher agency score in the synchronous condition of the controls’ than the patients’ group (p = 0.0004). Moreover, we found a significantly higher agency score in the asynchronous conditions of the patients’ as compared to the controls’ group (p = 0.01). For completeness, we also report the remaining comparisons: asynchronous controls vs. synchronous PD patients: p = 0.00005, synchronous controls vs. asynchronous PD patients: p = 0.00005. No significant main effect of group was found (F1,46=0.84; p = 0.37; η2p = 0.02).
Concerning proprioceptive drift, a significant main effect of condition was found (F1,46=21.99, p = 0.00003, η2p = 0.32), thus indicating that proprioceptive drift was greater in the synchronous than in the asynchronous condition. Crucially, a significant condition x group interaction was found (F1,46=7.53, p = 0.009, η2p = 0.14; see Fig. 3C). Duncan post hoc tests exploring the interaction revealed a significant difference between synchronous and asynchronous conditions only in the controls’ group (p = 0.00007) and not in the patients’ one (p = 0.20). Coherently, significantly greater proprioceptive drift was found in the synchronous condition of the controls’ group as compared to that of the patients’ group (p = 0.002). No difference between the two asynchronous conditions was found (p = 0.92). For completeness, we also report the remaining comparisons: asynchronous controls vs. synchronous PD patients: p = 0.32, synchronous controls vs. asynchronous PD patients: p = 0.0001. Finally, although no significant group effect was found (F1,46=3.92; p = 0.054; η2p = 0.08), results showed a trend for significance, suggesting overall greater proprioceptive drift in controls as compared to PD patients.
To sum up, our questionnaire results showed a VM-RHI effect (synchronous > asynchronous) in both groups. However, such an effect was diminished in the patients’ group. Moreover, a significantly decreased proprioceptive drift in the synchronous condition as compared to controls highlighted the abolishment of the VM-RHI effect in PD patients.
Experiment 2 (VM-RHI) results. The figure shows the plot of the Condition x Group interaction, concerning ownership (A), agency (B) questionnaires and proprioceptive drift measures (C). In all boxplots, the whiskers represent the minimum and the maximum value, the limits of the box represent the first and the third quartile, the median is depicted by the line that divides the box into two parts, while dots represent individual participants. Black asterisks indicate significant post-hoc comparisons exploring the interaction (*p < 0.05; **p < 0.005; ***p < 0.0005). Grey asterisks represent the significant main effect of Condition (****p < 0.00005; ******p < 0.0000005).
Between-experiments analysis
Given that proprioceptive drift analyses revealed a different pattern between VT- and VM-RHI procedures (see above), we performed a post-hoc analysis to draw a direct comparison. As regards the synchronous condition, the results showed a trend for statistical significance (t31 = 1.99; p = 0.056), with greater drift values in the VT- as compared to the VM-RHI. As regards the asynchronous condition, no significant difference (nor any trend) was observed (t31 = 0.28; p = 0.78). Although they should be interpreted with caution since they emerge from data acquired in different sessions, these results seem to support those of the analyses conducted on the data acquired within each session.
Correlational analyses between visuo-motor integration and motor deficit
To verify whether the extent of the motor impairment predicted the RHI deficit, we computed a VM-RHI measure (calculated as synchronous - asynchronous, on proprioceptive drift values) for each patient and ran a Pearson correlation between these values and the UPDRS score. No significant effect was found (r = 0.19; p = 0.29), suggesting that, at least in our sample, there was no linear relationship between the level of motor impairment and visuo-motor integration. Note that, in light of our small sample, we cannot exclude such linear relationship could emerge by testing a larger patient group.
Discussion
In the present study, we exploited the visuo-tactile [VT-; Experiment 1;23 and visuo-motor [VM-; Experiment 2;24 RHI to investigate whether the embodiment phenomenon, relying respectively on visuo-tactile or visuo-motor integration processes, is altered in patients with Parkinson’s Disease (PD). Our findings pinpoint distinct results in RHI measures, namely (ownership and agency) questionnaires and proprioceptive drift.
Concerning the ownership questionnaire, in the PD patients group, we found the typical RHI effects in both the VT- and the VM-RHI, with a greater body ownership alteration in the synchronous as compared to the asynchronous conditions. However, in both experiments, the illusory feeling experienced by PD patients in the synchronous condition was significantly decreased as compared to that experienced by controls, as explicitly reported by the patients, both when visuo-tactile and visuo-motor integration were involved in the illusion induction.
Previous works have already applied the VT-RHI to PD patients, highlighting similar results. Besides reporting significant differences between RHI conditions in the ownership questionnaire33,34, some studies found such a pattern to be reduced in the PD group as compared to controls32. In line with our results, Waldmann and colleagues reported attenuated embodiment during the synchronous condition in patients as compared to controls32. Moreover, these studies also observed an enhanced effect in the asynchronous condition32,33,34. The latter result was interpreted in light of a reduced temporal discrimination ability in these patients. Indeed, previous works already showed enlarged temporal binding windows both for unisensory15,16,17,18 and multisensory20 stimuli, possibly preventing patients from detecting the delay between visual and tactile stimulation in the asynchronous VT-RHI condition. If patients perceived visuo-tactile stimuli as essentially synchronous also in the control condition, it is not surprising that they experienced embodiment also during asynchronous VT-RHI. Based on this evidence, in our study, we introduced a clear delay between the tactile and visual stimuli during the asynchronous condition15,16,20. In this way, we were able to isolate the confound of temporal discrimination abnormality, which represents a higher-level deficit that affects both unisensory and multisensory processing. Therefore, our findings contribute to the evidence provided by previous studies32,33,34, suggesting that when asynchrony is detectable by PD patients, they exhibit the typical VT-RHI pattern on the ownership questionnaire, albeit reduced compared to controls, due to diminished performance in the synchronous condition. Furthermore, to the best of our knowledge, no previous study has applied the VM-RHI to PD patients. Our VM-RHI findings align with those of the VT-RHI, indicating a decreased RHI effect, as reported by our patients.
Our results of the VM-RHI also reveal interesting new findings concerning illusion-related modulations in the sense of agency. In line with the typical VM-RHI effect35, the control sample reported greater agency towards the fake hand’s movement in the synchronous as compared to the asynchronous condition. Such a significant difference was also found in the PD sample, in line with the ownership questionnaire’s results. However, it was reduced in PD patients as compared to controls, due to both a decrease in the synchronous and an increase in the asynchronous conditions.
Previous evidence revealed a link between the difficulty of this clinical population in initiating and controlling voluntary movements and abnormal results in classical explicit and implicit measures of agency36,37,38. It has been proposed that agency disturbances in these patients might be associated with a failure to weigh internal and external cues to give rise to agency experiences, with an exaggerated reliance on external cues and a reduced precision of internal cues. This theoretical account is in line with the evidence that Parkinson’s patients show abnormal self-agency when their actual movements are precluded from view38. Our data suggest an altered sense of agency in patients when they observed the fake hand moving, while their real hand was hidden. However, it is important to highlight that, here, PD patients showed diminished VT-RHI effect compared to controls, as revealed by the ownership questionnaire’s results. In other words, since the fake hand’s embodiment was decreased, the actions performed by that hand were not considered as controlled by themselves to the same extent as controls, in the synchronous condition. Thus, here, we cannot fully disentangle whether the observed abnormality in the agency scores was explained by the fact that their moving real hand was precluded from view, thus inducing an over-reliance on the external visual cues (i.e., the fake hand’s movements) to attribute agency, or by the reduced ownership alteration. Since the design of the present study does not allow for disentangling these explanations, future studies should more thoroughly investigate this issue.
Crucially, the results of the proprioceptive drift lead to highly interesting results, showing distinct patterns of RHI effects in PD when either visuo-tactile or visuo-motor integration was tackled to induce the illusion.
As concerns Experiment 1, PD patients revealed greater proprioceptive drift in the synchronous as compared to the asynchronous VT-RHI, i.e., the classical RHI effect [see34 for similar results]. Coherently, no difference between patients and controls was found, advocating for similar RHI effects in PD patients and controls when visuo-tactile integration was involved.
Previous works observed greater proprioceptive drift in PD patients as compared to controls, independently from the VT-RHI condition32,33,34. This effect was ascribed to a detrimental reliance afforded to proprioception in PD patients, who frequently show proprioceptive impairment. Being no longer able to rely on deficient proprioception, the estimates of the own hand position might have been captured by the seen fake hand during the procedure [i.e., visual capture of proprioception; see e.g.,39 resulting in higher proprioceptive drift values irrespective of the VT-RHI condition. Coherently, our patients with no position sense loss could rely on both vision and proprioception to localize the hand in space, revealing proprioceptive drift only when the illusion emerged.
Interestingly, the results of Experiment 2 showed an abolishment of the VM-RHI effect in PD patients on proprioceptive drift. Indeed, this measure showed no difference between synchronous and asynchronous conditions in our patients. Coherently, a between-group difference was observed, with decreased drift in PD patients as compared to controls, in the synchronous condition. In other words, proprioceptive drift results suggest that, when visuo-motor integration was tackled to induce the illusion, the RHI effect was abolished in PD patients.
In line with these results, Chen and colleagues recently showed visuo-motor integration impairment in patients with PD40. Specifically, in their task, participants used a joystick to control a moving visually-displayed target to keep it centered on a monitor through their hand movement. The results highlighted significantly decreased precision in PD patients as compared to healthy controls. The authors suggested that the patients’ poor performance was related to deficient visuo-motor integration, rather than simply originating from lower-level motor deficits. Coherently, our correlational analyses revealed that the visuo-motor integration deficit during the VM-RHI did not linearly increase with the level of motor impairment. This finding suggests that, at least in our patients, the primary motor deficit per se could not explain the integration deficit observed with the proprioceptive drift measure. Although, given the modest sample size, our negative correlational findings should be interpreted with caution, they seem to suggest that the impairment shown by our patients is not directly related to low-level motor disabilities, but rather they could pertain to abnormal visuo-motor integration mechanisms.
In line with this interpretation, it has been recently proposed that the visuo-motor integration deficit in PD patients might be related to basal ganglia dysfunction21,41. It is well-known that basal ganglia play a pivotal role in the integration of visual and proprioceptive inputs21,42,43. Previous literature showed that the neuronal response within the basal ganglia is greater when multiple-modalities simultaneous stimulation is applied, as compared to the additive response to single-modality stimulations, suggesting that the basal ganglia may be not only a node in a relay network for sensory perception and motor output but also a crucial hub for multisensory integration43. Thus, being basal ganglia’s functionality deeply affected by the degeneration of dopaminergic neurons of the substantia nigra in PD44, it seems reasonable to hypothesize that the visuo-motor RHI deficit observed here might stem from basal ganglia hypo functioning. However, while the link between basal ganglia dysfunction and embodiment deficits emerging from impaired visuo-motor integration in PD seems plausible, this remains a purely speculative interpretation that requires confirmation through dedicated neuroimaging studies.
Another issue to be discussed is the absence of exordium-side-related effects on right and left hand manipulations. This negative result seems to suggest that, at least in our sample, the bodily origin of the pathology did not interact with embodiment effects. Our findings may therefore pinpoint that exordium-side does not imply a specific involvement of a hemi-body on the multisensory integration processes allowing the RHI. However, we must acknowledge a limitation of our study, consisting of our sample not being tested at the pathology exordium, but in a chronic phase (see pathology onset in Table 1). Future studies could deepen this issue by addressing whether a more pronounced VM-RHI deficit is present in the exordium side at earlier stages of the pathology. Furthermore, it may be interesting to investigate how dopamine intake, which typically increases in later stages of the disease, might modulate the effects observed here in patients who were all tested in the on-medication state.
Finally, following a theoretical framework suggesting that, under typical conditions, the RHI can modulate both pain perception45 and pain-related brain activity46, future studies should investigate whether not only multisensory integration deficits, but also less effective endogenous pain control mechanisms, may underlie the channel-specific alteration of the VM-RHI effect observed here in PD patients.
Altogether, the results of Experiments 1 and 2 suggested that, when ownership and agency questionnaires were analysed, the RHI effect was still present in PD patients both when visuo-tactile and visuo-motor integration processes were tackled to elicit the illusion. However, this effect was reduced as compared to controls. Conversely, when a sensorimotor measure was considered (i.e., proprioceptive drift), a channel-specific deficit emerged in the PD group, involving only the visuo-motor RHI effect. The dissociation between the two measures’ results is in line with the common notion that RHI questionnaires and proprioceptive drift do not always go hand-in-hand47. Moreover, it seems particularly interesting that a prominent embodiment alteration emerged when the measure under consideration involved not only the RHI procedure that leverages visuo-motor integration, but also a response engaging sensorimotor channels, which is undoubtedly involved in the pathology.
Conclusion
Overall, our questionnaires’ results revealed the presence of a typical visuo-tactile and visuo-motor RHI pattern in PD patients, although diminished as compared to controls. Crucially, proprioceptive drift’s results showed the abolishment of visuo-motor (but not visuo-tactile) RHI effect in PD patients, advocating for a channel-specific abnormality of embodiment that emerged only when the visuo-motor integration, involved in the pathology, was challenged by the RHI procedure. Interestingly, such a channel-specific deficit emerged only when proprioceptive drift was considered, as it engages a sensorimotor response. These findings may highlight the importance of developing ad-hoc rehabilitative programs in PD, targeting, besides primary motor deficits, multisensory integration phenomena and body representations.
Materials and methods
Participants
32 patients with Parkinson’s Disease (PD) were recruited at the San Camillo Hospital of Turin for the present study. Since PD originates on a specific hemi-body before spreading bilaterally, we included both right and left exordium PD patients in the pathological sample and tested both their right and left hands to control for possible effects depending on the exordium side. Thus, we recruited 16 right exordium (age mean ± sd = 66.75 ± 9.41, 7 women) and 16 left exordium (age mean ± sd = 70.53 ± 7.51, 5 women) patients. Cognitive impairment and sustained tremor, which would have made the RHI procedure impossible to perform, were considered as exclusion criteria. Patients underwent a detailed clinical assessment, consisting of a neuropsychological and a psychological general evaluation, as well as an extended physiotherapy evaluation. The neuropsychological and psychological evaluation comprised cognitive impairment assessment through the Montreal Cognitive Assessment test48, and anxiety and depression assessment through the Hospital Anxiety and Depression Scale49,50. The physiotherapy evaluation included the Unified Parkinson’s Disease Rating Scale 3 and 551,52, which is the most commonly used tool to follow the longitudinal course of Parkinson’s Disease, as well as the assessment of balance and risk of falling through the Berg Balance Scale53,54, and the assessment of upper- and lower-body camptocormia55. Furthermore, to avoid our results being confounded by a potential proprioceptive deficit (see also 5.2 Experimental Procedure), patients underwent the Contralateral Limb Matching Task [CLMT56,57,58 to assess the absence of kinesthesia alterations. Moreover, to assess whether position sense was preserved, prior to each experimental session, we asked patients and healthy participants (see below) to indicate the felt position of the own tested hand by pointing at the vertical position of the hand on a ruler while being blindfolded to assess their precision (Proprioceptive Judgement Pre; see 5.2 Experimental design and procedures as well as Preliminary analyses on Proprioceptive Precision in the Supplementary Materials for a detailed description). In both experimental sessions, patients were tested while being in an on-medication state. See Table 1 for a detailed description of our patients’ clinical profile.
Finally, a group of 16 healthy age-matched participants (age mean ± sd = 65.88 ± 5.34, 11 women) was recruited as a control group. Note that patients (prot.n° 0032276/20 approved by the Intercompany Ethics Committee A.O.U. Cittа’ Della Salute E Della Scienza - A.O. Mauriziano - A.S.L. Citta’ Di Torino, detachment ASLTO1, Turin) and controls (prot.n° 199278/20 approved by the Bioethics Committee of the University of Turin) gave informed consent before participating. All methods were performed in accordance with the relevant guidelines and regulations.
Experimental design and procedures
Patients and controls underwent 2 experiments in separate sessions on different days. Indeed, each session lasted approximately one hour, and performing both on the same day would have caused excessive fatigue. In each session, they performed either the VT-RHI (Experiment 1) or the VM-RHI (Experiment 2). The experiments’ order was counterbalanced among participants so that half of them performed the VT-RHI in the first session and the remaining half performed it in the second session. In each experiment, they underwent VT- or VM-RHI conditions performed on both their left and right hand in random order. Before each condition, participants were asked to stretch the tested hand and place it in the bottom section of a wooden box, hidden from view. First, they were blindfolded and asked to point at the position of their tested hand’s index finger for 10 consecutive trials (Proprioceptive Judgement Pre; Fig. 1A). The experimenter measured the distance between the real position (0) and each pointed location in cm. Note that, since this measurement reflects the participants’ perception of hand position, we necessarily recruited patients with intact proprioception to collect reliable proprioceptive judgements. Then, a human-shaped rubber hand was positioned on the top of the box (visible) and the proximal parts of the participants’ shoulders were covered by a black cloth to ensure they could not perceive the discontinuity between their body and the rubber hand. When this setting was prepared, the blindfold was removed, and the RHI procedure began. Depending on the experiment, for 3 min, the participant either received touches while observing the fake hand being touched (Experiment 1; VT-RHI; Fig. 1B) or observed the fake hand moving while the own hand moved (Experiment 2; VM-RHI; Fig. 1C), in synchronous and asynchronous conditions (see below and Figure S3 in the Supplementary Information for further details about each RHI procedure). At the end of the RHI procedure, 10 consecutive trials of proprioceptive judgement were repeated (Proprioceptive Judgement Post), and RHI questionnaires were collected. In both RHI versions, responses to three real (“I felt as if I were looking at my own hand”; “I felt as if the rubber hand were part of my body”; “I felt as if the rubber hand were my hand”) and three control (“I felt as if my hand were becoming rubbery”; “I felt as if I had more than a left/right hand”; “I felt as if the rubber hand were drifting towards my hand”) items of the ownership questionnaire59 were collected. Only in the VM-RHI (Experiment 2), we additionally collected responses to three real (“The rubber hand moved just like I wanted it to, as if it was obeying my will”; “I felt as if I was controlling the movements of the rubber hand”; “I felt as if I was causing the movement I saw”) and three control (“I felt as if the rubber hand was controlling my will”; “I felt as if the rubber hand was controlling my movements”; “It seemed as if the rubber hand had a will of its own”) items of an agency questionnaire, in which the feeling of being the agent of the fake hand’s movement was explored24. In both questionnaires, participants had to rate their agreement on a 7-point Likert scale from − 3 (“totally disagree”) to + 3 (“totally agree”) and the items’ order was randomised within each questionnaire.
Experiment 1: visuo-tactile rubber hand illusion (VT-RHI)
In Experiment 1, participants observed the fake hand being touched while receiving real touches on their hidden hand, either in synchrony (i.e., synchronous condition, eliciting visuo-tactile integration) or out of phase (i.e., asynchronous control condition, not eliciting visuo-tactile integration). Hence, in this RHI version, the integration of two sensory modalities was tackled to induce the illusion: visual (i.e., the observation of touch on the fake hand) and tactile (i.e., the feeling of touch on the real hand) inputs. Visuo-tactile stimulation was performed manually by the experimenter with two brushes. Since recent literature pointed out that patients with Parkinson’s Disease have difficulties in discriminating temporal asynchrony15,20,33, a 1s delay was introduced between the seen touch (on the fake hand) and the perceived touch (on the real hand) in the asynchronous condition. (See Fig. 1B)
Experiment 2: visuo-motor rubber hand illusion (VM-RHI)
In Experiment 2, participants observed a fake hand moving in either synchrony (i.e., synchronous condition, eliciting visuo-motor integration) or out of phase (i.e., asynchronous condition, not eliciting visuo-motor integration) with the own hand. The movement consisted of a tapping action, performed by the participant with the index finger at a one-movement-per-second speed (see Fig. 1C). Hence, in this RHI version, the integration of two modalities was tackled to induce the illusion: visual (i.e., the observation of the fake hand’s movement) and motor (i.e., the feeling of the real hand’s movement) inputs. Note that, while in the synchronous condition the participant’s hand was connected to the fake hand via a small stick allowing for synchronous movement, in the asynchronous condition the stick was disconnected, and the experimenter moved the rubber hand in anti-phase (see Figure S3 in the Supplementary Information for more details). Again, a 1s delay was introduced between seen (fake hand’s) and perceived (own hand’s) movement in the asynchronous condition. (See Fig. 1C)
Data analysis
All analyses were performed using Statistica Software (StatSoft, release 8).
Preliminary analysis
Proprioceptive precision. In addition to the clinical assessments used for the recruitment of patients without proprioceptive deficit, to provide further evidence of the absence of proprioceptive impairment in our patients, we compared the mean Proprioceptive Judgements Pre collected at the beginning of the first session between groups. Indeed, such a dependent variable represents a measure of proprioceptive precision for each tested hand, before any RHI procedure. In line with clinical evaluations, the results revealed no significant difference between the patients’ and the controls’ groups (see Supplementary Information for a detailed description of the analyses and results).
RHI measures. For each experiment, we calculated ownership score and proprioceptive drift in each condition, as a proxy of the integration of visuo-tactile (Experiment 1) and visuo-motor (Experiment 2) inputs. The ownership score was calculated, separately for each experimental condition, as the mean rating in real items minus the mean rating in control items60 so as to control for response bias61. Proprioceptive drift was calculated as the mean Proprioceptive Judgement Post minus the mean Proprioceptive Judgement Pre. Furthermore, for Experiment 2, we computed an agency score (i.e., mean rating in real items minus mean rating in control items), an additional measure considered as a proxy of the sensation of controlling the fake hand’s movements.
Before running the main analyses, for each experiment, we performed preliminary analyses within each sample (i.e., the controls’ and the PD patients’ groups). Specifically, in the controls’ group, we addressed possible interactions between the RHI effect [consisting of significant differences between synchronous and asynchronous conditions23,24,62, and the hand involved in the procedure (i.e., the right or left hand). Additionally, in the PD group, we sought to investigate whether VT- and VM-RHI effects were present and whether they interacted with the patients’ exordium side, possibly leading to differential results depending on the tested hand. These analyses revealed that, as expected, VT- and VM-RHI effects were observed in the controls’ group irrespective of the tested hand. The patients’ group analyses showed that the RHI effects did not interact with the tested hand or the exordium side in either VT- or VM-RHI (see Supplementary Information for a detailed description of the analyses and results). Therefore, to reduce the complexity of our design in the main analyses63, we averaged responses of the right and left hands, and considered right- and left-exordium patients as a single group.
VT-RHI (Experiment 1) and VM-RHI (Experiment 2) analyses
In each experiment, ownership score and proprioceptive drift values were entered in a 2 × 2 repeated measures ANOVA with condition (synchronous, asynchronous) as within-subject factor and group (controls, PD patients) as between-subject factor. Furthermore, in Experiment 2, the very same analysis was run on the averaged values of the agency score.
Between-experiments analysis
Given that proprioceptive drift analyses revealed a different pattern between VT- and VM-RHI procedures (see 2.1 and 2.2 in the 2 Results section), we performed a post-hoc analysis to draw a direct comparison. Synchronous and asynchronous values of the patients’ group were compared between procedures through two-tailed matched-pairs t-tests.
Correlational analyses between visuo-motor integration and motor deficit
To investigate the possible contribution of primary motor impairment in the visuo-motor RHI deficit exhibited by the patients, we performed a Pearson correlation between a VM-RHI measure and the UPDRS 3 and 5 score. More specifically, since Experiment 2 analyses on proprioceptive drift revealed an embodiment deficit in the patients’ group [see 2.2 VM-RHI results (Experiment 2)], we computed an index as the delta between the proprioceptive drift in synchronous and asynchronous conditions as VM-RHI measure, and we entered this index in the Pearson correlation.
Data availability
The study data are available as a share link to a Mendeley Repository(https://data.mendeley.com/preview/z2f5htn89g? a=ecf6893d-c445-48d6-8fe9-10ba1974ea54). With such share link, anyone can see and download the unpublished datasets for review purposes. The link will be publicly available upon acceptance of the manuscript.
References
Tanner, C. M. & Ostrem, J. L. Parkinson’s disease. N Engl. J. Med. 391 (5), 442–452. https://doi.org/10.1056/NEJMra2401857 (2024).
Patel, N., Jankovic, J. & Hallett, M. Sensory aspects of movement disorders. Lancet Neurol. 13 (1), 100–112. https://doi.org/10.1016/S1474-4422(13)70213-8 (2014).
Jobst, E. E., Melnick, M. E. & Byl, N. N. Sensory perception in Parkinson disease. Arch. Neurol. 54, 450–454. https://doi.org/10.1001/archneur.1997.00550160080020 (1997).
Konczak, J. et al. Proprioception and motor control in parkinson’s disease. J. Mot Behav. 41 (6), 543–552. https://doi.org/10.3200/35-09-002 (2009).
Fiorio, M. et al. Defective Temporal discrimination of passive movements in parkinson’s disease. Neurosci. Lett. 417 (3), 312–315. https://doi.org/10.1016/j.neulet.2007.02.050 (2007).
Halperin, O., Israeli-Korn, S., Yakubovich, S. & Hassin-Baer, S. Self-motion perception in Parkinson s disease. Eur. J. Neurosci. 53, 2376–2387. https://doi.org/10.1111/ejn.14716 (2020).
Zia, S., Cody, F. W. J. & O’Boyle, D. Identification of unilateral elbow-joint position is impaired by Parkinson s disease. Clin. Anat. 15, 23–31. https://doi.org/10.1002/ca.1087 (2002).
Keijsers, N. L. W., Admiraal, M. A., Cools, A. R., Bloem, B. R. & Gielen, C. C. A. M. Differential progression of proprioceptive and visual information processing deficits in parkinson’s disease. Eur. J. Neurosci. 21 (1), 239–248. https://doi.org/10.1111/j.1460-9568.2004.03840.x (2005).
Schulz-Schaeffer, W. J. Camptocormia in Parkinson’s disease: a muscle disease due to dysregulated proprioceptive polysynaptic reflex arch. Front. Aging Neurosci. 8(JUN), 1–4. https://doi.org/10.3389/fnagi.2016.00128 (2016).
José Luvizutto, G., Souza Silva Brito, T., de Moura Neto, E., de Souza & L. Aparecida Pascucci Sande Altered visual and proprioceptive Spatial perception in individuals with parkinson’s disease. Percept. Mot Skills. 127 (1), 98–112. https://doi.org/10.1177/0031512519880421 (2020).
Tagliabue, M., Ferrigno, G. & Horak, F. Effects of parkinson’s disease on proprioceptive control of posture and reaching while standing. Neuroscience 158 (4), 1206–1214. https://doi.org/10.1016/j.neuroscience.2008.12.007 (2009).
Hwang, S., Agada, P., Grill, S., Kiemel, T. & Jeka, J. J. A central processing sensory deficit with parkinson’s disease. Exp. Brain Res. 234 (8), 2369–2379. https://doi.org/10.1007/s00221-016-4642-4 (2016).
Vaugoyeau, M., Hakam, H. & Azulay, J. P. Proprioceptive impairment and postural orientation control in parkinson’s disease. Hum. Mov. Sci. 30 (2), 405–414. https://doi.org/10.1016/j.humov.2010.10.006 (2011).
Vaugoyeau, M., Cignetti, F., Eusebio, A. & Azulay, J. P. Subthalamic deep brain stimulation modulates proprioceptive integration in parkinson’s disease during a postural task. Neuroscience 437, 207–214. https://doi.org/10.1016/j.neuroscience.2020.04.028 (2020).
Conte, A. et al. Somatosensory Temporal discrimination in parkinson’s disease, dystonia and essential tremor: pathophysiological and clinical implications. Clin. Neurophysiol. 129 (9), 1849–1853. https://doi.org/10.1016/j.clinph.2018.05.024 (2018).
Di Biasio, F. et al. Does the cerebellum intervene in the abnormal somatosensory Temporal discrimination in parkinson’s disease? Park Relat. Disord. 21 (7), 789–792. https://doi.org/10.1016/j.parkreldis.2015.04.004 (2015).
Artieda, J., Pastor, M. A., Lacruz, F. & Obeso, J. A. Temporal discrimination is abnormal in parkinson’s disease. Brain 115 (1), 199–210. https://doi.org/10.1093/brain/115.1.199 (1992).
Pastor, M. A., Artieda, J., Jahanshahi, M. & Obeso, J. A. Time Estimation and reproduction is abnormal in parkinson’s disease. Brain 115 (1), 211–225. https://doi.org/10.1093/brain/115.1.211 (1992).
Avanzino, L., Fiorio, M. & Conte, A. Actual and illusory perception in Parkinson’s disease and dystonia: a narrative review. Front. Neurol. 9(JUL), 1–10. https://doi.org/10.3389/fneur.2018.00584 (2018).
Rostami, Z., Salari, M., Mahdavi, S. & Etemadifar, M. Abnormal multisensory temporal discrimination in Parkinson’s disease. Brain Res. 1834(October), 148901. https://doi.org/10.1016/j.brainres.2024.148901 (2024).
Adamovich, S. V., Berkinblit, M. B., Hening, W., Sage, J. & Poizner, H. The interaction of visual and proprioceptive inputs in pointing to actual and remembered targets in parkinson’s disease. Neuroscience 104 (4), 1027–1041. https://doi.org/10.1016/S0306-4522(01)00099-9 (2001).
Roytman, S. et al. Multisensory mechanisms of gait and balance in parkinson’s disease: an integrative review. Neural Regen Res. 20 (1), 82–92. https://doi.org/10.4103/nrr.nrr-d-23-01484 (2025).
Botvinick, M. & Cohen, J. Rubber hands ‘feel’ touch that eyes see. Nature 391(6669), 756. https://doi.org/10.1038/35784 (1998).
Kalckert, A. & Ehrsson, H. H. Moving a rubber hand that feels like your own: a dissociation of ownership and agency. Front. Hum. Neurosci. 6(March), 1–14 https://doi.org/10.3389/fnhum.2012.00040 (2012).
Blanke, O., Slater, M. & Serino, A. Behavioral, neural, and computational principles of bodily Self-Consciousness. Neuron 88 (1), 145–166. https://doi.org/10.1016/j.neuron.2015.09.029 (2015).
Rossi Sebastiano, A. et al. Diametrical modulation of tactile and visual perceptual thresholds during the rubber hand illusion: a predictive coding account. Psychol. Res. 0123456789. https://doi.org/10.1007/s00426-021-01608-0 (2021).
Rossi Sebastiano, A. et al. Balancing the senses: electrophysiological responses reveal the interplay between somatosensory and visual processing during body-related multisensory conflict. J. Neurosci. 44, e1397232024. https://doi.org/10.1523/jneurosci.1397-23.2024 (2024).
Zeller, D., Litvak, V., Friston, K. J. & Classen, J. Sensory processing and the rubber hand illusion-an evoked potentials study. J. Cogn. Neurosci. 1–10.https://doi.org/10.1162/jocn_a_00705 (2014).
Zeller, D., Friston, K. J. & Classen, J. Dynamic causal modeling of touch-evoked potentials in the rubber hand illusion. Neuroimage 138, 266–273. https://doi.org/10.1016/j.neuroimage.2016.05.065 (2016).
Holmes, N. P., Snijders, H. & Spence, C. Reaching with alien limbs: visual exposure to prosthetic hands in a mirror biases proprioception without accompanying illusions of ownership. Percept. Psychophys. 68 (4), 685–701. https://doi.org/10.3758/bf03208768 (2006).
Galigani, M., Fossataro, C., Gindri, P., Conson, M. & Garbarini, F. Monochannel preference in autism spectrum conditions revealed by a Non-Visual variant of rubber hand illusion. J. Autism Dev. Disord. 52 (10), 4252–4260. https://doi.org/10.1007/s10803-021-05299-9 (2022).
Waldmann, A., Volkmann, J. & Zeller, D. Parkinson’ s disease may reduce sensitivity to visual-tactile asynchrony irrespective of dopaminergic treatment: evidence from the rubber hand illusion. Park. Relat. Disord. 78(July), 100–104. https://doi.org/10.1016/j.parkreldis.2020.07.016 (2020).
Ding, C. et al. Parkinson ’s disease alters multisensory perception: insights from the rubber hand illusion. Neuropsychologia 97, 38–45. https://doi.org/10.1016/j.neuropsychologia.2017.01.031 (2017).
Ding, C. et al. Deep brain stimulation for parkinson’ s disease changes perception in the rubber hand illusion. Sci. Rep. (January), 1–10. https://doi.org/10.1038/s41598-018-31867-8 (2018).
Kalckert, A. & Henrik Ehrsson, H. Moving a rubber hand that feels like your own: a dissociation of ownership and agency. Front. Hum. Neurosci. 6(March), 1–14. https://doi.org/10.3389/fnhum.2012.00040 (2012).
Seghezzi, S., Convertino, L. & Zapparoli, L. Sense of agency disturbances in movement disorders: A comprehensive review. Conscious. Cogn. 96, 103228. https://doi.org/10.1016/j.concog.2021.103228 (2021).
Wolpe, N. et al. Sensory Attenuation in parkinson’s disease is related to disease severity and dopamine dose. Sci. Rep. 8 (1), 1–10. https://doi.org/10.1038/s41598-018-33678-3 (2018).
Saito, N. et al. Altered awareness of action in parkinson’s disease: evaluations by explicit and implicit measures. Sci. Rep. 7 (1), 1–9. https://doi.org/10.1038/s41598-017-08482-0 (2017).
Carey, M., Crucianelli, L., Preston, C. & Fotopoulou, A. The effect of visual capture towards subjective embodiment within the full body illusion. Sci. Rep. 9 (1), 1–12. https://doi.org/10.1038/s41598-019-39168-4 (2019).
Chen, J. et al. Visuomotor control in patients with parkinson’ s disease. Neuropsychologia 80, 102–114. https://doi.org/10.1016/j.neuropsychologia.2015.10.036 (2016).
Scarpina, F. Defective tool embodiment in body representation of individuals affected by Parkinson’s disease : a preliminary study. 9(January), 1–11. https://doi.org/10.3389/fpsyg.2018.02489 (2019).
Nagy, A., Eördegh, G., Paróczy, Z., Márkus, Z. & Benedek, G. Multisensory integration in the basal ganglia. Eur. J. Neurosci. 24 (3), 917–924. https://doi.org/10.1111/j.1460-9568.2006.04942.x (2006).
Permezel, F., Alty, J., Harding, I. H. & Thyagarajan, D. Brain networks involved in sensory perception in parkinson’s disease: a scoping review. Brain Sci. 13(11). https://doi.org/10.3390/brainsci13111552 (2023).
Blandini, F., Nappi, G., Tassorelli, C. & Martignoni, E. Functional changes of the basal ganglia circuitry in parkinson’s disease. Prog Neurobiol. 62 (1), 63–88. https://doi.org/10.1016/S0301-0082(99)00067-2 (2000).
Mosch, B., Fuchs, X., Tu, T. & Diers, M. Time course of the rubber hand illusion–induced analgesia. Pain Rep. 0 (2), 1–7. http://dx.doi.org/10.1097/PR9.0000000000001252 (2025).
Pamplona, G. S. P. et al. Illusory body ownership affects the cortical response to vicarious somatosensation. (February), 312–328 (2022).
Rohde, M., Luca, M. & Ernst, M. O. The rubber hand illusion: feeling of ownership and proprioceptive drift do not go hand in hand. PLoS One 6(6). https://doi.org/10.1371/journal.pone.0021659 (2011).
Conti, S., Bonazzi, S., Laiacona, M., Masina, M. & Coralli, M. V. Montreal cognitive assessment (MoCA)-Italian version: regression based norms and equivalent scores. Neurol. Sci. 36 (2), 209–214. https://doi.org/10.1007/s10072-014-1921-3 (2015).
Zigmond, A. S. & Snalth, R. P. The hospital anxiety and depression scale. [revista en Internet]. Acta Psychiatr. Scand. 64(5): 361–370 (2014). 67(6), 361–370 (1983). Available: https://www.ncbi.nlm.nih.gov/pubmed/6880820
Costantini, M. et al. Detecting psychological distress in cancer patients: validity of the Italian version of the hospital anxiety and depression scale. Support Care Cancer. 7 (3), 121–127. https://doi.org/10.1007/s005200050241 (1999).
Goetz, C. G. et al. Movement disorder Society-Sponsored revision of the unified parkinson’s disease rating scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov. Disord. 23 (15), 2129–2170. https://doi.org/10.1002/mds.22340 (2008).
Ramsay, N. et al. Validation of a UPDRS-/MDS-UPDRS-based definition of functional dependency for parkinson’s disease. Park Relat. Disord. 76, 49–53. https://doi.org/10.1016/j.parkreldis.2020.05.034 (2020).
La Porta, F. et al. Is the Berg balance scale an internally valid and reliable measure of balance across different etiologies in neurorehabilitation? A revisited Rasch analysis study. Arch. Phys. Med. Rehabil. 93 (7), 1209–1216. https://doi.org/10.1016/j.apmr.2012.02.020 (2012).
Castro, D. D. I. et al. Validity and reliability of the psychometric properties of the 12-item Berg balance scale (BBS-12) in the Italian population with hip or knee prosthesis: A cross sectional study. Minerva Ortop. E Traumatol. 71 (3), 104–110. https://doi.org/10.23736/S0394-3410.20.03977-6 (2020).
Fasano, A. et al. Diagnostic criteria for camptocormia in parkinson’s disease: A consensus-based proposal. Park Relat. Disord. 53, 53–57. https://doi.org/10.1016/j.parkreldis.2018.04.033 (2018).
Goble, D. J. Proprioceptive acuity assessment via joint position matching: from basic science to general practice. Phys. Ther. 90(8), 1176–1184. https://doi.org/10.2522/ptj.20090399 (2010).
Lincoln, N. B. et al. The unreliability of sensory assessments. Clin. Rehabil. 5 (4), 273–282. https://doi.org/10.1177/026921559100500403 (1991).
Piriyaprasarth, P., Morris, M. E., Delany, C., Winter, A. & Finch, S. Trials needed to assess knee proprioception following stroke. Physiother. Res. Int. 14(1), 6–16. https://doi.org/10.1002/pri.405 (2009).
Longo, M. R., Schüür, F., Kammers, M. P. M., Tsakiris, M. & Haggard, P. What is embodiment? A psychometric approach. Cognition 107 (3), 978–998. https://doi.org/10.1016/j.cognition.2007.12.004 (2008).
Errante, A. et al. Shared body representation constraints in human and non-human primates behavior. Cortex 181, 179–193. https://doi.org/10.1016/j.cortex.2024.10.011 (2024).
Romano, D., Maravita, A. & Perugini, M. Psychometric properties of the embodiment scale for the rubber hand illusion and its relation with individual differences. Sci. Rep. 11 (1), 1–16. https://doi.org/10.1038/s41598-021-84595-x (2021).
Rossi Sebastiano, A. et al. Reach planning with someone else’s hand. Cortex 153, 207–219. https://doi.org/10.1016/j.cortex.2022.05.005 (2022).
Frost, J. Hypothesis Testing: An Intuitive Guide Making Data Driven Decisions 1st edn (Statistics by Jim Publishing, 2024).
Acknowledgements
We are grateful to all the patients and their families for allowing our research, as well as to G. Bonino for her contribution to data acquisition.
Funding
This work was partially supported by a grant from the University of Turin, Ricerca Locale (RiLo) obtained by FGarbarini, and partially by the European Union (Next Generation EU, Missione 4, Componente 2, Project N. J11J24001950006).
Author information
Authors and Affiliations
Contributions
FGe: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Visualization; Writing - original draft; Writing - review & editing; MR: Formal analysis; Visualization; Writing - original draft; Writing - review & editing; FPT, NE, CD, PG: Conceptualization; Investigation; Data curation; Writing - review & editing; FGa: Conceptualization; Writing - review & editing; Project administration; Supervision; Funding acquisition; ARS: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Visualization; Supervision; Writing - original draft; Writing - review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Genovese, F., Romeo, M., Terrenzio, F.P. et al. Distinct patterns of visuo-tactile and visuo-motor body-related integration in Parkinson’s disease. Sci Rep 15, 27923 (2025). https://doi.org/10.1038/s41598-025-08965-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-08965-5