Abstract
To evaluate the synergistic effect of the Shugan Jieyu Capsule combined with Escitalopram in the treatment of senile depression. The study protocol has been registered in PROSPERO with registration number CRD42023440270. Eight databases, including PubMed, Embase, Cochrane Library, Web of Science, China National Knowledge Network (CNKI), China Biomedical Literature Database (CBM), VIP database, and Wanfang Database, were searched, respectively. Randomized controlled trial (RCTs) of the Shugan Jieyu Capsule combined with Escitalopram in the treatment of senile depression was included. The search time was from the listing of Shugan Jieyu Capsule (2009) to January 1, 2025. Outcome measures included Total effective rate, Hamilton Depression Scale (HAMD) score, Hamilton Anxiety Scale (HAMA) score, serum 5-hydroxytryptamine (5-HT), serum norepinephrine (NE), and Incidence of adverse reactions. The treatment period should be at least 6 weeks. Data extraction and methodological quality evaluation were carried out for the included literature, and statistical analysis was performed using RevMan 5.3 software. Finally, the Grading of Recommendations Assessment, Development and Evaluation (GRADE ) system was used to assess the quality of the evidence. A total of 8 RCTs were included in the study, with a total of 765 patients. The results of this meta-analysis showed that the combination of Shugan Jieyu Capsule and Escitalopram could improve Total effective rate [OR = 4.25, 95%CI(2.53, 7.14), P<0.00001] and reduce HAMD score in terms of clinical symptom indicators [SMD=-1.26, 95%CI(-1.45, -1.08), P<0.00001], and decrease HAMA score [SMD=-0.82, 95%CI(-1.05, -0.59), P<0.00001]. In terms of laboratory indexes, serum 5-HT levels could be increased in the combined group [MD = 31.92, 95%CI(28.34, 35.50), P < 0.00001]. In terms of safety, no serious adverse reactions were observed in both group, and the incidence of adverse reactions was lower in the combined group [OR = 0.62, 95%CI(0.42, 0.92), P = 0.02]. During the treatment period of 12 weeks, Shugan Jieyu Capsule combined with Escitalopram had a good effect in the treatment of senile depression, which could significantly reduce HAMD and HAMA scores of senile depression. In addition, the combined application of the two drugs can help to increase the level of serum 5-HT, without increasing the occurrence of adverse events, and has a good application prospect.
Similar content being viewed by others
Senile depression is one of the most common mental diseases in the elderly, which seriously affects the physical and mental health, social function, and quality of life of the elderly, and can also lead to suicidal behavior, with an estimated prevalence of 13.3%1,2. A survey based on the elderly population in China shows that the prevalence of Senile depression in China is 23.6%, and the prevalence of females is significantly higher than that of males, and it increases year by year with the increase of age3. The clinical manifestations of senile depression are atypical, often accompanied by somatization symptoms and cognitive impairment4, increasing the risk of chronic diseases. The treatment of Senile depression includes psychotherapy, physical therapy, and drug therapy. Antidepressants are still the primary treatment for depression in older adults5. The interaction between drugs, adverse reactions, comorbidities, compliance, and other factors should be considered when select drug therapy. Escitalopram has been widely used in the treatment of elderly patients with depression in recent years. Escitalopram is a highly selective serotonin reuptake inhibitor (SSRI) that significantly increases the level of 5-HT in the synaptic gap and plays a role in the treatment of depression6. However, the effect of the drug after use is relatively slow, especially in the early stage of the most critical treatment of depression, and its symptom improvement effect is not obvious7. In addition, this drug is prone to memory damage, hangover phenomenon, drug tolerance, dependence, and other risks, which are not conducive to long-term, large-scale use alone8,9.In the treatment of acute-stage of senile depression, Escitalopram is well tolerated, but there is no significant difference compared with placebo10. In view of the deficiency of escitalopram, how to develop a more reasonable and effective drug regimen for elderly patients with depression has become a hot spot in clinical research.
Shugan Jieyu Capsule is the first Chinese patent medicine approved for the treatment of mild and moderate Uniphase depression. It can improve the symptoms of depression caused by liver stagnation and spleen deficiency, and its curative effect mainly shows that it can improve depression, anxiety, insomnia, and other aspects, with fewer adverse reactions and good compliance11. Shugan Jieyu Capsule is innovative in TCM theory of fire causing depression, clearing liver fire, tonifying spleen and kidney, and playing the function of soothing liver and depression, strengthening the spleen, and calming the mind, which can significantly improve the clinical symptoms of low mood, decreased interest, difficulty in falling asleep, irritability and so on in mild and moderate monophasic depression patients with liver-depression and spleen-deficiency syndrome12. The number of clinical studies on the treatment of senile depression with the combination of Shugan Jieyu Capsule have gradually increased, indicating that the combination of Shugan Jieyu Capsule has gradually become a focus of clinical research in recent years.
In this study, clinical RCTs on the intervention of Shugan Jieyu Capsule combined with Escitalopram in senile depression were screened from multiple databases, aiming to evaluate the clinical value of the combined treatment of the two drugs in Senile depression and provide new evidence for further research on the synergistic and antagonistic effects of integrated Chinese and Western medicine in clinical application.
Data and methods
The study was developed in accordance with the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA 2020)13. The protocol was registered on PROSPERO (CRD42023440270).
Inclusion criteria
-
(1)
Participants: Participants meet the clear diagnostic criteria for depression (e.g., the Chinese Diagnostic Classification System and diagnostic criteria in 200114 or consistent with the Tenth Revision of the International Statistical Classification of Diseases and Related Health Problems in 198815 or The American Psychiatric Association’s Diagnostic Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) in 201316); Age ≥ 60 years old, regardless of ethnicity or gender;
-
(2)
Intervention: The trial group was treated with Shugan Jieyu Capsule combined with Escitalopram. The treatment period should be at least 6 weeks.
-
(3)
Control: The control group was treated with Escitalopram alone. The treatment period should be at least 6 weeks.
-
(4)
Outcome: Outcome indicators included clinical symptom assessment, such as Total effective rate, Hamilton Depression Scale (HAMD) score, and Hamilton Anxiety Scale (HAMA) score. Laboratory indicators such as serum 5-hydroxytryptamine (5-HT), and Serum norepinephrine (NE); Incidence of adverse reactions.
-
(5)
Study type: Randomized controlled trials.
Exclusion criteria
-
(1)
The study objects were animal, pharmacokinetic and other non-clinical efficacy evaluation literature.
-
(2)
Literature that does not adequately describe interventions;
-
(3)
The efficacy evaluation index is not standardized;
-
(4)
The relevant data could not be extracted from the research literature.
Prognostic criteria
Total effective rate was determined according to the reduction rate of HAMD scale score before and after treatment. The definition of “Total effective rate” is the percentage of the sum of the number of basically cured cases, the number of significant effective cases and the number of effective cases in the total number of cases. This criterion has been widely applied in international research17.
-
(1)
Cure: the reduction rate of more than 75% before and after treatment;
-
(2)
Significant effect: the reduction rate before and after treatment was more than 50%;
-
(3)
Effective: the reduction rate before and after treatment is more than 25%;
-
(4)
Ineffective: the reduction rate before and after treatment is less than 25%.
Total effective rate=(cured cases + significant effective cases + effective cases)/total cases ×100%.
Literature search strategy
A literature search was carried out according to the principle of evidence-based medicine search strategy, searching CNKI, China Biomedical Literature Database, Chinese Science and Technology Journal full-text database, Wanfang database, Pubmed, Elsevier, CochraneLibrary, and other databases. The search time was from the listing of Shugan Jieyu Capsule (2009) to January 1, 2025. The combination of subject words and free words is adopted. The search strategy for all databases was shown in supplementary file 1.
Evaluation methods
Literature screening and extraction
Two researchers independently screened literature and extracted literature data, including title, author, year, intervention measures, intervention time, outcome indicators, etc. Disagreement after cross-checking was determined by mutual discussion or adjudication by a third-party investigator.
Assessment of bias risk of included studies
The evaluation was carried out according to the RCT bias risk assessment tool provided by Cochrane Handbook 5.3, which included randomized sequence generation, allocation hiding, blinding of researchers and subjects, blinded evaluation of research outcomes, the integrity of outcome data, selective reporting of research results, and other possible biases. The risk of bias for each item was divided into three levels: “Low risk of bias”, “Unclear risk of bias” and “High risk of bias”. Disagreements were resolved by discussion or referred to a third reviewer.
Statistical methods
Meta-analysis was performed using RevMan5.3 software provided by Cochrane Collaboration. The odds ratio (OR) was used as the efficacy statistic for count data (binary variables). For measurement data, mean difference (MD) was used if the measurement method was the same as the expression. If the measurement scales were different or the outcome variables were highly inconsistent, the standardized mean difference (SMD) combined with I2 was used to evaluate the degree of heterogeneity. When P ≥ 0.1 and I2 < 50%, the statistical heterogeneity of each study was small, and the fixed effect model was used. Otherwise, a random-effects model was used for meta-analysis. Before performing a meta-analysis, determine whether clinical heterogeneity exists in the included studies. Potential sources of heterogeneity (e.g. course of treatment, dose, etc.) were discussed in detail. Finally, the factors leading to heterogeneity were explored by subgroup analysis. The test level of the meta-analysis was set as α = 0.05. If the included studies could not be synthesized statistically, descriptive analysis was used. Finally, the Grading of Recommendations Assessment, Development and Evaluation (GRADE) system was used to assess the quality of the evidence.
Results
Results of the literature search
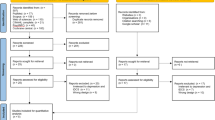
As of January 1, 2025, a total of 52 studies were retrieved, and 32 duplicates were excluded. After reading the title and abstract of the literature, 8 studies that did not meet the requirements were excluded, including 3 studies without relevant intervention measures, 2 studies without relevant research objects, and 3 studies without RCT test. After reading the full paper, 4 studies did not meet the requirements, 1 study without relevant intervention measures, 1 study without relevant research objects, and 2 studies without an RCT test. Finally, a total of 8 pieces of literature11,12,13,14,15,16,17,18 were included in this study, with a total of 765 cases, including 383 cases in the combined treatment group and 382 cases in the control group. All 8 studies were published in Chinese journals. The retrieval and screening process is shown in Fig. 1. The basic characteristics of the included studies are shown in Table 1.
Flow diagram of the study selection process.
Risk of bias assessment results of included studies
Random grouping was mentioned in all 8 studies18,19,20,21,22,23,24,25, among which 418,20,21,24 were grouped by random number table, while 419,22,23,25 did not give a specific description of the random method. 8 studies18,19,20,21,22,23,24,25 did not mention the specific method of allocation hiding; Eight studies18,19,20,21,22,23,24,25 did not implement the blind method, and eight studies18,19,20,21,22,23,24,25 did not report relevant information about clinical trial registration, so it is impossible to determine whether there was selective reporting. The bias risk analysis of the included studies is shown in Fig. 2, and the bias risk summary is shown in Fig. 3.
Analysis of risk of bias in clinical studies.
Summary of the risk of bias in clinical studies.
Results of meta-analysis
Total effective rate
A total of seven studies assessed Total effective rate. There was no significant heterogeneity among the included studies (I2 = 0%, P = 0.90),, so the fixed-effect model was used for analysis. The results showed that there was a statistically significant difference in Total effective rate between the two groups (OR = 4.25, 95%CI(2.53, 7.14), P < 0.00001), suggesting that compared with Escitalopram alone, Shugan Jieyu Capsule combined with Escitalopram could improve Total effective rate in the treatment of senile depression. The forest map is shown in Fig. 4.
Meta-analysis forest plot of the total effective rate.
Hamilton depression scale (HAMD) score
A total of six studies assessed HAMD score. There was no significant heterogeneity among the included studies (I2 = 0%, P = 0.52), so fixed effects model was used for analysis. The results showed that there was a statistically significant difference in HAMD score between the two groups (SMD=−1.26, 95%CI(−1.45, −1.08), P<0.00001), suggesting that Shugan Jieyu Capsule combined with Escitalopram could reduce the HAMD score of Senile Depression. Subgroup analyses showed that after 6 or 8 weeks of treatment, compared with Escitalopram alone, Shugan Jieyu Capsule combined with Escitalopram could reduce the HAMD score of senile depression(SMD=−0.91, 95%CI(−1.44, −0.38), P = 0.0008) (SMD=−1.31, 95%CI(−1.51, −1.12), P<0.00001).The forest map is shown in Fig. 5.
Forest plot of HAMD score meta-analysis.
Hamilton anxiety scale (HAMA) score
A total of four studies assessed HAMA score. There was no significant heterogeneity among the included studies (I2 = 4%, P = 0.37), so fixed effects model was used for analysis. The results showed that there was a statistically significant difference in HAMA score between the two groups (SMD=−0.82, 95%CI(−1.05, −0.59), P<0.00001), suggesting that Shugan Jieyu Capsule combined with Escitalopram could reduce the HAMA score of Senile Depression. Subgroup analyses showed that after 6 of treatment, there was no significant difference in HAMA score between Shugan Jieyu Capsule combined with Escitalopram and Escitalopram alone (SMD=−0.49, 95%CI(−1.00, −0.03), P = 0.06). However, after 8 weeks of treatment, compared with Escitalopram alone, Shugan Jieyu Capsule combined with Escitalopram could reduce the HAMD score of senile depression(SMD=−0.90, 95%CI(−1.15, −0.65), P<0.00001).The forest map is shown in Fig. 6.
Forest plot of HAMA meta-analysis.
Serum 5-hydroxytryptamine (5-HT)
A total of two studies reported serum 5-HT. There was no significant heterogeneity among the included studies (I2 = 30%, P = 0.23), so fixed effects model was used for analysis. The results showed that there was a statistically significant difference in serum 5-HT between the two groups (MD = 31.92, 95%CI(28.34, 35.50), P<0.00001), suggesting that Shugan Jieyu Capsule combined with Escitalopram could increase serum 5-HT levels in senile depression patients compared with Escitalopram alone. The forest map is shown in Fig. 7.
Forest plot of Serum 5-HT meta-analysis.
Incidence of adverse reactions
A total of seven studies reported the incidence of adverse reactions after treatment. The incidence of adverse reactions was 15.87%(53/334) in the trial group and 23.12%(77/333) in the control group. There was no significant heterogeneity among the included studies (I2 = 32%, P = 0.18), so fixed effects model was used for analysis. The results showed that the incidence of adverse reactions after treatment in the trial group was lower than that in the control group, and the difference was statistically significant [OR = 0.62, 95%CI(0.42, 0.92), P = 0.02], indicating that the incidence of adverse reactions after treatment with Shugan Jieyu Capsule combined with Escitalopram was lower than that with Escitalopram alone. The forest map is shown in Fig. 8. Statistics of adverse events are shown in Table 2.
Forest plot of Incidence of adverse reactions meta-analysis.
Publication bias and sensitivity analysis
Publication bias could not be assessed because fewer than 10 studies were included in the combined outcome measures. Sensitivity analysis was performed on Total effective rate, HAMD score, HAMA score, 5-HT, and Incidence of adverse reactions of the included studies, and some of them were excluded one by one. The results showed that there was no statistically significant difference in the heterogeneity of the studies, indicating that the results of this study were stable.
Narrative description
Zhu19 reported that after 8 weeks of treatment, the serum NE level in the combined group was 4.61 ± 0.26ng/L, which was higher than that in the western medicine group of 3.08 ± 0.18ng/L, with statistical significance (P < 0.05). Bao18 reported that after 8 weeks of treatment, the serum NE level of the combined group was 0.22 ± 0.03ng/ml, which was lower than that of the western medicine group (0.43 ± 0.11ng/ml), and the difference was statistically significant (P < 0.05). We found that Bao[18] reported the course of the patient’s disease and mentioned the determination of serum NE levels by fluorescence spectrometry, while Zhu[19] did not mention the above information. Therefore, different course of disease and different methods of serum index determination may have some influence on the results.
Certainty of evidence
Table 3 presents the certainty assessment based on the GRADE analysis. The certainty of evidence regarding Total effective rate, HAMD score, HAMA score, 5-HT and Incidence of adverse reactions outcomes has been deemed very low. This is primarily due to the high risk of bias observed in most of the studies included. Furthermore, the small number of participants contributes to the imprecision in the reported effects, which further weakens the evidence.
Note: (i) Risk of bias in studies: downgraded by one level if some concerns and two levels if high-risk of bias; (ii) Indirectness: considered low due to eligibility criteria; (iii) Risk of publication bias: not assessed, as all comparison had < 10 studies available; downgrade one level if Egger’s test < 0.05; (iv) Inconsistency: downgraded by one level when the impact of statistical heterogeneity (I2) was moderate (> 25%) and by two levels when high (> 75%); (v) Imprecision: downgraded by one level when < 800 participants were available for a comparison or if there was no clear direction of the effects; accumulation of both resulted in downgrading by two levels. GRADE: Grading of Recommendations Assessment, Development and Evaluation; PSS: pooled sample size.
Discussion
Significance and value of this study
To the best of our knowledge, our study is the first systematic review using RCTs of Shugan Jieyu Capsule Combined with Escitalopram in the treatment of senile depression. We use a detailed and stable search strategy that can span multiple databases. A sensitive analysis shows that most of our results are stable. This study included 8 RCTs of the Shugan Jieyu Capsule combined with Escitalopram in the treatment of senile depression. The results of the meta-analysis showed that Shugan Jieyu Capsule combined with Escitalopram had a good effect in the treatment of senile depression, which could significantly reduce the HAMD score and HAMD score of senile depression. In addition, the combined application of the two drugs can help to increase the level of serum 5-HT. In the included studies, adverse reactions such as increased body mass, dry mouth and dizziness occurred in the trial group, and the incidence of adverse reactions was 15.87%(53/334). In the control group, body mass increase, dry mouth, dizziness, appetite increase, nausea, vomiting, constipation and other adverse reactions occurred after taking the drug, and the incidence of adverse reactions was 23.12%(77/333). However, the symptoms of the patients were mild, did not affect the course of treatment, and were relieved by themselves without special treatment. At the same time, there was a statistically significant difference in the incidence of adverse reactions between the two groups (P < 0.05), suggesting that the treatment of Shugan Jieyu Capsule combined with escitalopram has fewer adverse reactions, high tolerance in elderly patients, and is safe and reliable.
Limitations of this study
Although this meta-analysis shows that Shugan Jieyu Capsule combined with Escitalopram demonstrate advantages in improving the symptoms of depression in the elderly, this conclusion must be viewed with caution. The currently included RCTs generally have methodological flaws. The four included studies19,22,23,25 did not specifically elaborate on the randomization method. None of the eight studies mentioned specific methods of assigning concealment, nor did they report information about clinical trial registration, so it is impossible to determine whether there was selective reporting. Based on the GRADE analysis, the certainty of evidence regarding Total effective rate, HAMD score, HAMA score, 5-HT and Incidence of adverse reactions outcomes has been deemed very low. This is primarily due to the high risk of bias observed in most of the studies included. Furthermore, the small number of participants contributes to the imprecision in the reported effects, which further weakens the evidence. In summary, although this study included randomized controlled trials, which are considered the gold standard for evaluating treatment effects, significant risks of bias and inconsistencies in the study cannot be ignored.
Therefore, it is highly necessary to carry out strictly designed and large-scale randomized controlled trials in future research. Large-scale RCTs can significantly improve statistical power, thereby more accurately evaluating the effect of intervention measures. In the case of a small sample size, even if the intervention measures have a real effect, they may not be detected due to excessive random errors. Large-scale RCTs, on the other hand, can reduce random errors and improve the accuracy of effect estimation by increasing the sample size. Large-scale RCTs usually have stricter quality control measures and more complete research designs, thereby being able to reduce the occurrence of various biases. For example, implementing allocation hiding through the central random system and the sealed opaque envelope method can prevent researchers from generating subjective bias during the grouping process. The implementation of blinding through double-blind or triple-blind designs can reduce the occurrence of measurement bias and implementation bias. Furthermore, multi-center RCTs can include patients from different regions, different populations, and different clinical practice environments, thereby improving the external validity and universality of the results.
The effect of shugan jieyu capsule combined with escitalopram on neurotransmitters levels may have potential advantages
The occurrence of depression is mainly due to the reduction of 5-HT, NE, and other neurotransmitters in the central nervous system. In old age, the functions of various organs of the body undergo degenerative changes and are often combined with a variety of chronic underlying diseases, poor tolerance to drugs, and slow metabolism, so the safety requirements for drugs are high26,27. Shugan Jieyu Capsule is a traditional Chinese medicine compound preparation composed of Hypericum trifoliata and Acanthopanax, and it is also the first traditional Chinese medicine compound new drug approved by National Medical Products Administration (NMPA) for the treatment of depression. By activating the transient receptor potential channel, the Shugan Jieyu Capsule inhibits the reuptake of 5-HT, dopamine (DA), and NE neurotransmitters, thereby reducing the reabsorption of neurotransmitters in the presynaptic membrane and simultaneously increasing the synthesis and release of neurotransmitters in the synaptic gap. At the same time, it was observed that the elevation of 5-HT and DA in brain tissue was more significant28,29. Previous studies have observed a significant decline of corticosterone levels and a significant increase of 5-HT levels in the plasma of chronic unpredictable mild stress-model rats after treatment with Shugan Jieyu Capsule. Shugan Jieyu Capsule also significantly reduced the adrenal index, to an extent significantly more than fluoxetine or Hypericum perforatum L (p < 0.05). These data showed that Shugan Jieyu Capsule can partially neutralize corticosterone overproduction and the hypothalamic-pituitary-adrenal axis dysregulation, which may be one of the mechanisms explaining antidepressant effects30. To some extent, this also supports our finding that Shugan Jieyu Capsule combined with Escitalopram can significantly increase serum 5-HT levels while improving depressive symptoms.
The improvement direction and future research suggestions of shugan jieyu capsule combined with escitalopram in the treatment of senile depression
In recent years, more and more scholars began to pay attention to the clinical effect of Shugan Jieyu Capsule in the treatment of depression. Huang X11 found that Shugan Jieyu Capsule has clinical advantages compared with non-Shugan Jieyu treatment in terms of response rate, HAMD, National Institutes of Health Stroke Scale (NIHSS), and Pittsburgh Sleep Quality Index (PSQI) scores in patients with neurological diseases. Zhang X31 found that the combination of soothing liver and relieving depression was effective in the treatment of adult major depressive disorder, and the effective rate and remission rate were significantly higher than that of venlafaxine alone. Wang L32 found that Shugan Jieyu Capsule alone or combined with other antidepressants was effective in treating postpartum depression. However, these studies did not pay attention to the image of Shugan Jieyu Capsule on elderly depressed people, and the outcome indicators did not involve experimental indicators such as serotonin. This study provides new evidence for the clinical application of Shugan Jieyu Capsule combined with Escitalopram.The results of this study showed that Shugan Jieyu Capsule combined with Escitalopram had a significant effect on clinical efficacy, and could reduce depression score and anxiety score. In addition, the combination of the two drugs can significantly increase the level of 5-HT and play a role in improving the efficacy of antidepressants.
Future studies need to adopt more rigorous and scientific randomized controlled trial designs to ensure the objectivity and accuracy of the studies. The study period was extended to observe the long-term efficacy and safety of the combination therapy. Strict quality control was carried out on the included studies to ensure the authenticity and reliability of the data. Establish uniform data standards and regulatory mechanisms to better integrate and analyze research results. At the same time, subgroup analysis was conducted according to the different severity, course and concomitant diseases of elderly patients with depression, so as to explore a more optimal treatment plan, so as to more accurately evaluate the efficacy and safety of combination therapy. Although the current meta-analysis included a certain number of studies, the sample size still needs to be further expanded to improve the accuracy and reliability of the analysis. In addition to the improvement of depressive symptoms, attention should also be paid to changes in patients’ quality of life, social functioning, and so on. This allows for a more comprehensive assessment of the efficacy of combination therapy and the benefits to patients. In addition, the specific mechanism of action of Shugan Jieyu Capsule combined with Escitalopram in the treatment of senile depression was studied, including the changes of neurotransmitters and neural pathways. It is helpful to provide theoretical basis for further optimization of combination therapy. Future studies should cover older patients with depression in more regions and different cultures to verify the general applicability of the combination therapy. Through these improvements and suggestions, it can promote the development of Shugan Jieyu Capsule combined with Escitalopram in the treatment of senile depression to a higher quality direction, and provide a more solid evidence base for clinical decision-making.
Data availability
Data is provided within the manuscript or supplementary information files.
References
Van Damme, A. et al. Late-life depression: issues for the general practitioner. Int. J. Gen. Med. 11, 113–120 (2018).
Wang, Y. et al. Association between residual symptoms and social functioning in patients with depression. Compr. Psychiatry. 98, 152–164 (2020).
Li, D. et al. A meta-analysis of the prevalence of depressive symptoms in Chinese older adults. Arch. Gerontol. Geriatr. 1, 1–9 (2014).
Wu, Z. et al. Clinical symptoms and their relationship with cognitive impairment in elderly patients with depressive disorder. Front. Psychiatry. 13, 1009653 (2022).
Kverno, K. S. & Mangano, E. Treatment-Resistant depression: approaches to treatment. J. Psychosoc Nurs. Ment Health Serv. 9, 7–11 (2021).
Zhang, X. L., Han, Y. P. & Lian, Y. D. Analysis of curative effect of Fluoxetine and Escitalopram in the depression treatment based on clinical observation. Pak J. Pharm. Sci. 31, 1115–1118 (2018).
Munier, H. Pathogenesis and clinical manifestations of elderly patients with depression. Chin. J. Health Psychol. 5, 654–657 (2015).
Lu, J. et al. Elderly antidepressant research progress of the clinical application. J. West. China Pharm. 04, 435–439 (2018).
Moreno, M. L. et al. Cognitive changes after tDCS and Escitalopram treatment in major depressive disorder: results from the placebo-controlled ELECT-TDCS trial. J. Affect. Disord. 263, 344–352 (2020).
Bose, A. et al. Escitalopram in the acute treatment of depressed patients aged 60 years or older. Am. J. Geriatr. Psychiatry. 1, 14–20 (2008).
Huang, X., Song, T., Ding, Z., Wang, D. & Wang, C. Efficacy of Shugan Jieyu capsule for treatment of neurologic disorders combined with depression: A meta-analysis. Pak J. Pharm. Sci. 35 (5), 1445–1458 (2022).
Wang, C. et al. Research progress on clinical application and mechanism of action of Shugan Jieyu capsule based on the theory of Zang-deficiency and Qi-stagnation. Pharmacol. Clin. Chin. Materia Med. 04, 120–124 (2023).
Page, M. J. et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372, n71 (2021).
Chinese Classification and Diagnosis Criteria for Mental Disorders (3rd edition). Chinese Journal of Psychiatry. ;03:59–63. (2001).
Brämer, G. R. International statistical classification of diseases and related health problems. Tenth revision. World Health Stat. Q. 1, 32–36 (1988).
First, M. B. Diagnostic and statistical manual of mental disorders, 5th edition, and clinical utility. J Nerv Ment Dis. ;201(9):727-9. (2013).
Zimmerman, M. et al. Severity classification on the Hamilton depression rating scale. J. Affect. Disord. 150 (2), 384–388. https://doi.org/10.1016/j.jad.2013.04.028 (2013).
Bao, S. H. et al. Effect and mechanism of Shugan Jieyu capsule combined with Escitalopram in the treatment of senile depression with insomnia. J. Mod. Integr. Chin. Western Med. 30, 3385–3389 (2020).
Zhu, H. J. et al. Effect of Shugan Jieyu capsule combined with Escitalopram on elderly patients with physical diseases accompanied by anxiety and depression and its influence on treatment compliance. Int. J. Psychiatry. 01, 151–153 (2019).
He, G. L. Effect of Shugan Jieyu capsule combined with Escitalopram on comorbidities of depression and anxiety in elderly patients. Henan Med. Res. 08, 1444–1446 (2018).
Li, Q. et al. Study on Shugan Jieyu capsule combined with Escitalopram to improve anxiety and depression in elderly patients with physical diseases and treatment compliance. J. Chin. Med. 04, 970–973 (2018).
Ni, X. D. et al. Effects of Shugan Jieyu capsule assisted Escitalopram on cognitive function, serum TSH, IL-6, and TNF-α in elderly patients with phlegm-heat depression. J. Mod. Integr. Chin. Western Med. 18, 2035–2037 (2017).
Lin, S. Z. & Lian, H. T. Effect of Escitalopram combined with Shugan Jieyu capsule on senile depression with anxiety. Straits Pharm. 09, 134–135 (2016).
Wang, X. H. et al. Effect of Shugan Jieyu capsule combined with Escitalopram on clinical efficacy and treatment compliance in elderly patients with somatic diseases accompanied by anxiety and depression. Int. J. Psychiatry. 01, 119–122 (2016).
Zhang, D. Q. Study of Shugan Jieyu capsule combined with Escitalopram in the treatment of senile depression and anxiety comorbidities. World Combines Traditional Chin. Western Med. 05, 683–685 (2015).
Kautzky, A. et al. Age as a moderating factor of treatment resistance in depression. ;66(1):e35. (2023). https://doi.org/10.1192/j.eurpsy.2023.17
Edwards, N. et al. Prevalence of depression and anxiety in older people in low- and middle-income countries in africa, asia, and South america: A systematic review and meta-analysis. J. Affect. Disord. 325, 656–674 (2023).
Quan, W. et al. Effect of Shugan Jieyu capsule on the expression of TRPC6, P-CREB, and BDNF in CUMS mice. Northwest. J. Pharm. 3, 400–404 (2020).
Doron, R. et al. A novel herbal treatment reduces depressive-like behaviors and increases BDNF levels in the brain of stressed mice. Life Sci. 2, 151–157 (2014).
Tan, J. et al. Antidepressant Shugan Jieyu capsule alters gut microbiota and intestinal Microbiome function in rats with chronic unpredictable mild stress -Induced depression. Front. Pharmacol. 13, 828595. https://doi.org/10.3389/fphar.2022.828595 (2022).
Zhang, X. et al. Shugan Jieyu capsule for major depressive disorder (MDD) in adults: a systematic review. Aging Ment Health. 18 (8), 941–953 (2014).
Wang, L. et al. Efficacy and safety of Shugan Jieyu capsule alone or in combination with other antidepressants in the treatment of postpartum depression: A Meta-Analysis. Evid. Based Complement. Alternat Med. 2022, 5260235 (2022).
Funding
This study was supported by Sichuan Province Traditional Chinese Medicine Development Special Fund Support (No. T000011947436); Chengdu Science and Technology Bureau Project (No. 2022-YF05-01932-SN); Project of Science and Technology Department of Sichuan Province (No. 2022YFS0446).
Author information
Authors and Affiliations
Contributions
Xing Tang drafted the manuscript. Chengcheng Wang and Chunxia Yang conducted a literature search. Qinwan Huang and YeaYin Yen independently screened the potential studies and extracted the data. Congcong Yu and ShouJen Lan edited the manuscript. Congcong Yu arbitrated any disagreement and ensured that no errors occurred during the review. All authors critically reviewed, revised, and approved the subsequent and final version of the protocol.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
All authors agree that the article is free from conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Tang, X., Wang, C., Huang, Q. et al. A systematic review and meta-analysis of shugan jieyu capsule combined with escitalopram in the treatment of senile depression. Sci Rep 15, 23111 (2025). https://doi.org/10.1038/s41598-025-09113-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-09113-9