Abstract
Reproductive characteristics, including age at menarche (AAM), age at natural menopause (ANM), and parity, may influence bone mineral density (BMD) in postmenopausal women through the duration of estrogen exposure and the regulation of calcium homeostasis. Current Fracture Risk Assessment Tool (FRAX) exclude these parameters, potentially limiting their accuracy in identifying high-risk individuals. This study aimed to investigate the associations between reproductive characteristics and the risk of osteoporosis, as well as the fracture probabilities derived from FRAX. This retrospective cross-sectional study included 417 postmenopausal women (aged 45–80 years) stratified by quantitative computed tomography (QCT)-measured lumbar volumetric bone mineral density (vBMD) into osteoporotic (n = 63, vBMD ≤ 80 mg/cm³) and non-osteoporotic (n = 354) groups. We collected demographic, reproductive, clinical, and radiological data, and calculated four categories of FRAX scores: 10-year probabilities of major osteoporotic fracture (MOF) and hip fracture (HF), with/without BMD integration. Binary logistic regression analyzed associations between reproductive characteristics and osteoporosis risk. Generalized linear models with Gamma distribution and log-link function evaluated independent associations between reproductive characteristics and FRAX scores. The osteoporotic group exhibited earlier ANM (48.0 ± 3.8 vs. 49.9 ± 4.2 years, p = 0.005) and higher parity (2.5 ± 1.4 vs. 1.7 ± 1.0, p < 0.001). Logistic regression revealed earlier ANM (OR = 0.831, 95% CI: 0.789–0.875) and increased parity (OR = 1.345, 95% CI: 1.075–1.683) as independent risk factors for osteoporosis (p < 0.05). Generalized linear models demonstrated that each 1-year delay in menopause decreased FRAX scores: BMD-integrated MOF and HF risks decreased by 2.1% and 4.0%, while non-BMD risks decreased by 2.7% and 4.4% (p < 0.001). Conversely, each additional parity increased FRAX scores: BMD-integrated MOF and HF risks increased by 5.9% and 12.8%, and non-BMD risks increased by 4.6% and 8.0% (p < 0.05). Age, ANM, and parity consistently affected all FRAX scores consistently (p < 0.05). Earlier ANM and higher parity are significantly associated with elevated osteoporosis risk and FRAX scores. Incorporating these reproductive variables into the FRAX algorithms may improve the accuracy of identifying high-risk individuals.
Similar content being viewed by others
Introduction
Osteoporosis is a systemic skeletal disorder characterized by reduced bone mineral density (BMD) and impaired bone microarchitecture, significantly increasing the risk of fractures1,2,3. As global populations age, the prevalence of osteoporosis and related fractures continues to rise. This condition not only severely compromises patients’ physical health and quality of life but also imposes a significant economic burden on healthcare systems, representing a critical public health challenge4. Postmenopausal women are at high risk for osteoporosis due to the abrupt reduction in estrogen levels following menopause5. The pathogenesis of postmenopausal osteoporosis involves multifactorial interactions, with estrogen deficiency serving as the central pathological mechanism6. Reproductive characteristics of postmenopausal women—including age at menarche (AAM), age at natural menopause (ANM), and parity—demonstrate potential associations with osteoporosis. These factors may independently affect BMD and fracture risk by regulating the estrogen exposure cycle and the homeostasis of calcium metabolism. However, the specific mechanisms of action and their clinical evaluation value remain to be fully elucidated7,8.
The Fracture Risk Assessment Tool (FRAX), an internationally recognized predictive model for osteoporotic fractures, calculates the 10-year fracture probability by analyzing clinical variables such as age, body mass index (BMI), and prior fracture history9. However, current FRAX algorithms do not incorporate female reproductive characteristics—including AAM, ANM, and parity—as independent risk parameters. While early menopause (< 45 years) is classified under secondary osteoporosis in FRAX, its influence is limited to risk calculations without bone mineral density (BMD) adjustments10,11. Notably, delayed menarche, early menopause, and multiparity may independently increase fracture risk through hormonal regulatory mechanisms, and the failure of FRAX to incorporate these factors may reduce the identification accuracy for high-risk populations.
Dual-energy X-ray absorptiometry (DEXA), recognized as the gold standard for osteoporosis diagnosis, assesses the risk through two-dimensional areal bone mineral density (aBMD, g/cm²). However, the results are susceptible to interference from spinal degeneration, fat tissue distribution, and other confounding factors. In contrast, quantitative computed tomography (QCT) measures three-dimensional volumetric BMD (vBMD, mg/cm³), enabling precise differentiation of trabecular and cortical bone while remaining unaffected by spinal degeneration or adipose distribution. This technique provides a more reliable technical method for BMD evaluation12,13. Building on the advantages of QCT, this study stratified postmenopausal women into two groups based on QCT-vBMD. For the first time, we systematically investigated the associations between reproductive characteristics and osteoporosis risk, as well as Fracture Risk Assessment Tool (FRAX) scores. Our findings aim to identify independent risk factors unaccounted for in conventional models and to provide a theoretical foundation for enhancing the clinical utility of osteoporosis risk assessment tools.
Materials and methods
Study population
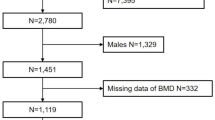
We retrospectively analyzed the clinical and imaging data of postmenopausal women who underwent osteoporosis screening at our hospital between January and October 2024. Participants were stratified into two groups based on QCT-measured L1-L2 volumetric bone mineral density (vBMD), according to the diagnostic thresholds established by the American College of Radiology (ACR)14,15: the osteoporotic group (vBMD ≤ 80 mg/cm³) and the non-osteoporotic group (vBMD > 80 mg/cm³), the latter including individuals with osteopenia and normal bone mass. Inclusion criteria required: (1) natural menopause aged 45–80 years; (2) postmenopausal duration ≥ 1 year; (3) completed DXA and QCT assessments; (4) complete reproductive histories and clinical data for Fracture Risk Assessment Tool (FRAX) scoring; (5) no history of hormone replacement therapy (HRT) or medications that affect bone metabolism. Exclusion criteria included: (1) age < 45 years or non-natural menopause; (2) secondary osteoporosis comorbidities (for example, hyperthyroidism, Cushing’s syndrome); (3) spinal deformities or surgical history; (4) incomplete data or suboptimal imaging quality; (5) severe cardiovascular/cerebrovascular diseases or malignancies. The flowchart of patient screening is shown in Fig. 1.
Flowchart of patient selection in the study.
Clinical data
A standardized osteoporosis questionnaire was administered by trained staff. Prior to the survey, participants were informed about the study’s purpose and procedures, and written informed consent was obtained. The survey instrument, which was completed independently by all participants, included the following: demographic characteristics: (1) demographic parameters (age, height, weight; with BMI calculated from these measures); reproductive characteristics (AAM, ANM, and parity); clinical information: (prior fractures, parental history of hip fractures, comorbidities, medication use, current smoking status, and alcohol consumption). ANM was defined as the cessation of menstruation for ≥ 12 consecutive months without surgical intervention (for example, hysterectomy or bilateral oophorectomy) or iatrogenic causes (for example, chemotherapy) and parity was defined as the number of pregnancies reaching 28 weeks of gestation or longer. Completed questionnaires were collected and securely stored by designated personnel.
BMD measurement
This study utilized a GE Medical Systems Revolution Apex scanner in conjunction with the QCT Pro software program (Mindways Software, Inc., Austin, TX, USA) and the European Spine Phantom for vBMD measurements. Scanning sequences with QCT Pro identifiers were configured using the following parameters: a fixed tube voltage of 120 kVp, a scan field of view (SFOV) of 500 mm, a tube current of 59 mA, and both slice thickness and spacing set to 1.25 mm. Following image acquisition, QCT Pro-series datasets were manually selected and transferred to the Mindways QCT workstation for data archiving and analytical processing. Volumetric BMD was automatically calculated using the “3D spine exam analysis” function, in which regions of interest (ROIs) were manually delineated at the mid-vertebral level of L1-L2 to maximize trabecular bone inclusion while excluding cortical bone and the posterior venous plexus. Areal bone mineral density (aBMD) of the femoral neck was assessed using dual-energy X-ray absorptiometry (DEXA; Prodigy Advance, General Electric Company, USA). All QCT and DEXA data were reviewed by radiologists to exclude invalid data resulting from spinal degeneration, metal artifacts, or inadequate scan coverage.
FRAX score calculation
The 10-year risk of major osteoporotic fractures (MOF) and hip fractures (HF) was calculated using the FRAX® online tool (Asia-China version, accessed at: frax.shef.ac.uk). The input parameters included age, weight, height, history of prior fractures, parental history of hip fracture, smoking status, glucocorticoid use, rheumatoid arthritis, secondary osteoporosis, daily alcohol intake, with femoral neck aBMD included as an optional variable. Four types of scores were generated: MOF risk with BMD (FRAX-MOF-BMD), HF risk with BMD (FRAX-HF-BMD), and their counterparts without BMD data (FRAX-MOF and FRAX-HF). FRAX scores were calculated independently by two researchers, and consistency verification showed an intraclass correlation coefficient (ICC) > 0.96.
Statistical analysis
Statistical analyses were conducted using SPSS 26.0 (IBM Corp.) and R 4.3.1 (R Foundation). Continuous variables were expressed as mean ± standard deviation (SD) or median [interquartile range (IQR)]. Normality of data distribution was assessed using the Shapiro-Wilk test. For normally distributed data, group comparisons were performed using the independent samples t-test; otherwise, the Mann-Whitney U test was applied. Categorical variables were presented as frequencies and percentages. Group comparisons were conducted using Chi-square tests. Binary logistic regression models examined associations between reproductive characteristics (AAM, ANM, parity) and osteoporosis status, adjusting for chronological age. Model calibration was verified through Hosmer-Lemeshow testing. Non-parametric correlations between vBMD, FRAX scores, and reproductive variables were evaluated using Spearman’s ρ. For FRAX score comparisons, participants were stratified into high- and low-risk groups based on median FRAX scores. Group differences in continuous variables were evaluated using the t-test. The associations between reproductive characteristics and FRAX scores were analyzed using generalized linear models (GLM) with a Gamma distribution and log-link function. Multicollinearity was assessed using the variance inflation factor (VIF). The p value < 0.05 was considered to indicate statistical significance.
Results
A total of 417 patients participated in this study, comprising 63 individuals in the osteoporotic group and 354 individuals in the non-osteoporotic group. The osteoporotic group demonstrated a significantly older age (68.8 ± 6.3 vs. 61.9 ± 6.19 years, p < 0.001), a later AAM (15.7 ± 1.8 vs. 14.6 ± 1.7 years, p < 0.001), earlier ANM (48.0 ± 3.8 vs. 49.9 ± 4.2 years, p = 0.005), and higher parity (2.5 ± 1.4 vs. 1.7 ± 1.0, p < 0.001) compared to the non-osteoporotic group. Lumbar vBMD measurements at L1-L2 showed marked reduction in the osteoporotic group (64.98 ± 13.12 vs. 119.48 ± 26.29 mg/cm³; p < 0.001). The osteoporotic group exhibited a significantly higher prevalence of fragility fractures compared to the non-osteoporotic group (p = 0.016). No statistically significant difference in BMI (p = 0.146). (Table 1)
Independent risk factors for osteoporosis
The results of the binary logistic regression analysis are presented in Table 2. Age (OR = 1.078, 95% CI: 1.038–1.119, p < 0.001), ANM (OR = 0.831, 95% CI: 0.789–0.875, p < 0.001) and parity (OR = 1.345, 95% CI: 1.075–1.683, p = 0.01) were identified as independent risk factors for osteoporosis. The Hosmer-Lemeshow test indicated good model calibration (χ² =11.143, p = 0.194).
Correlation analysis
Spearman’s ρ analysis revealed statistically significant but weak correlations among vBMD, FRAX scores, and reproductive characteristics. The vBMD showed a negative correlation with AAM (r = −0.148, p < 0.01) and parity (r = −0.265, p < 0.01), but a positive correlation with ANM (r = 0.195, p < 0.01). Similarly, FRAX scores in all categories were negatively correlated with ANM and positively correlated with parity (p < 0.01), as illustrated in Fig. 2.
Correlation analysis of QCT-VBMD and FRAX scores with reproductive characteristics. (*p < 0.05, **p < 0.01) AAM, age at menarche; ANM, age at natural menopause; FRAX-MOF-BMD and FRAX-HF-BMD,10-year probabilities of major osteoporotic fracture (MOF) and hip fracture (HF) with femoral neck BMD integration; FRAX-MOF and FRAX-HF: 10-year probabilities of MOF and HF without BMD integration;.
Intergroup FRAX score comparisons
Comparisons of FRAX scores between the osteoporotic and non-osteoporotic groups were conducted using the nonparametric Mann-Whitney U test. All four types of FRAX scores in the osteoporotic group—regardless of whether BMD data were included—were significantly higher than those in the non-osteoporotic group (Table 3): FRAX-MOF-BMD (4.9 [3.5–6.3] vs. 3.2 [2.5–4.8]), FRAX-HF-BMD (1.2 [0.8–2.2] vs. 0.5 [0.2–1.0]), FRAX-MOF (6.9 [5.2–9.9] vs. 4.0 [3.0–6.4]), and FRAX-HF (2.9 [1.7–4.3] vs. 1.0 [0.5–2.1]), with all p < 0.001.
Comparison of clinical and reproductive characteristics across FRAX score subgroups
Participants were stratified into high- and low-risk groups based on the median FRAX score (Table 4). The high-risk group exhibited a significantly older age (for example, FRAX-MOF-BMD: 65.4 ± 6.5 vs. 60.4 ± 5.9 years; p < 0.001) and lower vBMD (for example, FRAX-MOF-BMD: 99.26 ± 26.86 vs. 123.52 ± 31.28 mg/cm³; p < 0.001). Earlier ANM was observed in most high-risk subgroups (p < 0.05), except for FRAX-MOF-BMD (p = 0.054). Parity was consistently higher in high-risk groups across all FRAX categories (for example, FRAX-MOF-BMD: 2.1 ± 1.3 vs. 1.6 ± 1.0; p < 0.001). These findings indicate that an elevated FRAX score is associated with advanced age, earlier AAM, higher parity, and reduced bone density.
FRAX score predictors
Associations between reproductive characteristics and FRAX scores were analyzed using generalized linear models (GLM) with Gamma distribution and log-link function, with the results summarized in Table 5. IRR (Incidence Rate Ratio) reflects the multiplicative effect of a one-unit increase in the predictor variable on the outcome. In the FRAX-MOF-BMD and FRAX-HF-BMD models, each one-year increase in age was associated with a 3.1% and 7.4% increase in FRAX outputs, respectively. Each one-year delay in ANM was linked to a 2.1% and 4.0% decrease, while each additional parity was associated with a 5.9% and 12.8% increase. In the FRAX-MOF and FRAX-HF models (without BMD data), each one-year increase in age corresponded to 5.0% and 10.7% increase in FRAX outputs, respectively. Each one-year delay in ANM was associated with a 2.7% and 4.4% decrease, and each additional parity was linked to a 4.6% and 8.0% increase. All four models demonstrated independent effects of age, ANM, and parity on FRAX outputs. Age exhibited significant positive associations in all models (p < 0.001), with notably stronger effects in BMD-unadjusted models (for example, FRAX-HF IRR = 1.107), aligning with the FRAX algorithm’s design to upweight clinical indicators when BMD data are absent. ANM showed significant negative associations across all models (p < 0.001). Parity significantly increased FRAX outputs in all models (p < 0.05), with the largest effect observed in the FRAX-HF-BMD model, which indicated a 12.8% increase per additional pregnancy.
Discussion
This study demonstrated that women with an earlier age at natural menopause (ANM) and higher parity exhibited significantly elevated osteoporosis risk and FRAX scores. Both vBMD and FRAX scores were significantly associated with reproductive characteristics. The age at menarche (AAM) and ANM influence bone metabolism by regulating the duration of estrogen exposure. Delayed menarche shortens the promotive effect of adolescent estrogen on bone mass accumulation16, leading to reduced peak bone mass and increased susceptibility to osteoporosis. Early menopause results in the premature termination of estrogen’s protective effects, the decline in estrogen levels after menopause weakens its protective role on bone tissue, resulting in significantly increased bone loss during the first few postmenopausal years17,18. Mechanistically, estrogen binds to ERα/ERβ receptors on the osteoblasts and osteocytes, downregulating the expression of receptor activator of nuclear factor κB ligand (RANKL), inhibiting osteoclast differentiation and activation, and reducing bone resorption. It also accelerates bone matrix mineralization by directly stimulating osteoblast proliferation and collagen synthesis, as well as upregulating factors such as transforming growth factor-β6,19,20. Additionally, estrogen enhances calcium transport efficiency in intestinal epithelial cells and tubular calcium reabsorption in renal tubules, thereby maintaining systemic calcium homeostasis and mitigating skeletal calcium mobilization21,22. In this study, the osteoporotic group exhibited a later AAM compared to the non-osteoporotic group (15.7 ± 1.8 years vs. 14.6 ± 1.7 years, p < 0.001). However, multivariate analysis revealed no statistically significant association between AAM and osteoporosis risk (p = 0.081). This discrepancy may stem from limitations in sample size, yet the observed intergroup differences and correlation analyses still hold clinical relevance.
The present study identified a significant association between increased parity and elevated risks of osteoporosis, as well as higher FRAX scores. Existing evidence highlights that pregnancy and lactation impose substantial physiological demands for calcium due to fetal and neonatal skeletal development. A full-term singleton pregnancy requires approximately 30 g of calcium, with 80% of this amount transferred during the third trimester (300–400 mg/day). In cases of inadequate dietary calcium intake, the maternal skeleton acts as a calcium reservoir, releasing calcium ions through upregulated bone resorption pathways, which leads to progressive bone loss23,24. During lactation, estrogen levels drop further (to < 50 pg/mL), exacerbating bone resorption by removing the physiological inhibition of the ERα signaling pathway in bone tissue25. Multiple pregnancies may induce cumulative bone loss and reduce peak bone mass reserves through the following mechanisms: Although elevated levels of 1,25-dihydroxyvitamin D3 during pregnancy enhance intestinal calcium absorption efficiency, this effect is partially offset by the placenta’s selective barrier to vitamin D metabolites. Concurrently, the sharp decline in estrogen levels during lactation upregulates macrophage colony-stimulating factor (M-CSF) expression by 2.5-fold in the bone marrow microenvironment, promoting the recruitment of osteoclast precursors and accelerating bone resorption26. Syncytiotrophoblasts in placental villi initiate the secretion of parathyroid hormone-related peptide (PTHrP) during the first trimester, with maternal serum levels progressively increase throughout gestation and reaching maximal concentrations near term. PTHrP activates the receptor activator of nuclear factor-κB ligand (RANKL)/osteoprotegerin (OPG) axis in osteoblasts, stimulating osteoclast differentiation and activity while suppressing osteoblast-mediated bone formation. This dual action disrupts the coupling of bone resorption and formation, leading to a net loss of bone mass27. A single pregnancy typically results in a 3–5% reduction in BMD, which is partially restored during a 6–12-month postnatal recovery period under adequate nutritional conditions. However, repeated pregnancies (≥ 3) with short inter-pregnancy intervals (< 24 months) may repeatedly interrupt bone remodeling cycles, thereby preventing the complete restoration of bone mass and resulting in cumulative skeletal depletion28,29. In summary, the mechanisms linking parity to bone health are multifactorial and interdependent, necessitating further basic and clinical research to clarify these complex interactions.
Notably, no statistically significant difference in BMI was observed between the osteoporotic and non-osteoporotic groups when stratified by QCT-measured vBMD (22.70 ± 2.93 vs. 23.27 ± 2.84 kg/m², p = 0.146). This finding may be attributed to QCT’s technical superiority: its three-dimensional phantom-calibrated measurements directly quantify trabecular bone vBMD in vertebral bodies, minimizing measurement biases from skeletal size, adipose distribution, or osteophyte formation—biases inherent in DEXA measurements. Adipose tissue may exert dual effects on bone metabolism: mechanical loading from increased body weight could enhance bone density, whereas obesity-related proinflammatory cytokines (for example, IL-6, TNF-α) may promote bone resorption and increase fracture risk30. In contrast, lean body mass—a measure of skeletal muscle content—has been identified as an independent protective factor for bone density, as supported by longitudinal clinical data31,32.
Reproductive characteristics, including ANM and parity, influence BMD through estrogen-dependent pathways and the calcium metabolic demands during pregnancy. These factors indirectly or directly modulate fracture risk, as quantified by FRAX scores. Although the current FRAX tool categorizes early menopause (< 45 years) as a secondary osteoporosis risk factor, this classification only affects models without BMD input and fails to account for the independent contributions of parity or menopause timing to fracture risk, which may lead to an underestimation of fracture risk in women with high parity or premature menopause. To optimize risk assessment, the FRAX tool could consider incorporating ANM and parity as independent variables or covariates to reflect their direct effects on BMD and fracture risk.
This study employed QCT technology, which addresses measurement biases in DEXA caused by two-dimensional projection errors, spinal degeneration, and adipose tissue interference, thereby enhancing the accuracy of BMD measurement and group stratification. For the first time, this study systematically investigates the associations between reproductive characteristics, postmenopausal osteoporosis, and FRAX scores, analyzing not only FRAX scores calculated with BMD data but also those derived without BMD input. The findings suggest that reproductive characteristics should be considered in clinical assessments of osteoporosis risk, with particular attention to multiparous women and those with an early ANM. The study underscores the unquantified independent risk value of reproductive characteristics in traditional FRAX models, offering a new perspective for optimizing the FRAX. Several limitations warrant consideration: the retrospective design restricts causal inference; confounding factors such as dietary calcium intake and physical activity were not controlled; recall bias may exist in the questionnaire data; and the single-center sample introduces potential selection bias, affecting the generalizability of the results. Future studies could employ multicenter prospective cohort designs combined with dynamic monitoring of bone metabolic markers to further validate the long-term effects of reproductive characteristics on osteoporosis.
Conclusion
In summary, our findings indicate that in postmenopausal women, an earlier age at natural menopause and higher parity are significantly associated with elevated osteoporosis risk and FRAX scores. These results underscore the clinical relevance of incorporating reproductive characteristics into osteoporosis risk assessments. Incorporating these reproductive variables into the FRAX algorithms may improve the accuracy of identifying high-risk individuals in future clinical applications.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- BMD:
-
Bone mineral density
- AAM:
-
Age at menarche
- ANM:
-
Age at natural menopause
- QCT:
-
Quantitative computed tomography
- DEXA:
-
Dual-energy X-ray absorptiometry
- vBMD:
-
Volumetric bone mineral density
- aBMD:
-
Areal bone mineral density
- FRAX:
-
Fracture risk assessment tool
References
Compston, J. E., McClung, M. R. & Leslie, W. D. Osteoporos. Lancet ;393:364–376. (2019).
Anam, A. K. & Insogna, K. Update on osteoporosis screening and management. Med. Clin. North Am. 105, 1117–1134 (2021).
Lane, N. E. Epidemiology, etiology, and diagnosis of osteoporosis. Am. J. Obstet. Gynecol. 194, S3–11 (2006).
Zhang, X. et al. (ed Roggen, I.) The prevalence and treatment rate trends of osteoporosis in postmenopausal women. PLoS ONE 18 e0290289 (2023).
Lorentzon, M. et al. Osteoporosis and fractures in women: the burden of disease. Climacteric 25, 4–10 (2022).
Fischer, V. & Haffner-Luntzer, M. Interaction between bone and immune cells: implications for postmenopausal osteoporosis. Semin. Cell Dev. Biol. 123, 14–21 (2022).
Yang, Y., Wang, S. & Cong, H. Association between parity and bone mineral density in postmenopausal women. BMC Womens Health. 22, 87 (2022).
He, Y. et al. Menarche age exceed 17 years and menopausal age smaller than 48 years May affect prevalence of osteoporosis for Chinese women. Arch. Osteoporos. 16, 123 (2021).
Vandenput, L. et al. Update of the fracture risk prediction tool FRAX: A systematic review of potential cohorts and analysis plan. Osteoporos. Int. 33, 2103–2136 (2022).
Wang, Y., Yu, S., Hsu, C., Tsai, C. & Cheng, T. Underestimated fracture risk in postmenopausal women—application of the hybrid intervention threshold. Osteoporos. Int. 31, 475–483 (2020).
Schini, M. et al. An overview of the use of the fracture risk assessment tool (FRAX) in osteoporosis. J. Endocrinol. Invest. 47, 501–511 (2024).
Messina, C. et al. DXA beyond bone mineral density and the REMS technique: new insights for current radiologists practice. Radiol. Med. 129, 1224–1240 (2024).
Link, T. M. & Majumdar, S. Osteoporosis imaging. Radiol. Clin. North Am. 41, 813–839 (2003).
Koweek, R. & Heller, S. L. Patient-friendly summary of the ACR appropriateness criteria: osteoporosis and bone mineral density. J. Am. Coll. Radiol. 19, e55 (2022).
Ward, R. J. et al. ACR appropriateness criteria ® osteoporosis and bone mineral density. J. Am. Coll. Radiol. 14, S189–202 (2017).
Wang, J. et al. Urban–rural differences in bone mineral density and its association with reproductive and menstrual factors among older women. Calcif Tissue Int. 106, 637–645 (2020).
Long, G. et al. Predictors of osteoporotic fracture in postmenopausal women: A meta-analysis. J. Orthop. Surg. Res. 18, 574 (2023).
Shieh, A. et al. Associations of age at menopause with postmenopausal bone mineral density and fracture risk in women. J. Clin. Endocrinol. Metab. 107, e561–e569 (2021).
Cheng, C-H., Chen, L-R. & Chen, K-H. Osteoporosis due to hormone imbalance: an overview of the effects of Estrogen deficiency and glucocorticoid overuse on bone turnover. Int. J. Mol. Sci. 23, 1376 (2022).
Ko, S-H. & Jung, Y. Energy metabolism changes and dysregulated lipid metabolism in postmenopausal women. Nutrients 13, 4556 (2021).
Wang, J., Wu, S., Zhang, Y., Yang, J. & Hu, Z. Gut microbiota and calcium balance. Front. Microbiol. 13, 1033933 (2022).
Merlotti, D. et al. Bone fragility in Gastrointestinal disorders. Int. J. Mol. Sci. 23, 2713 (2022).
Ó Breasail, M. et al. Pregnancy-related change in pQCT and bone biochemistry in a population with a habitually low calcium intake. J. Bone Miner. Res. 36, 1269–1280 (2020).
O’Brien, E. C., Geraghty, A. A., Kilbane, M. T., McKenna, M. J. & McAuliffe, F. M. Bone resorption and dietary calcium in pregnancy—a window to future maternal bone health. Osteoporos. Int. 32, 1803–1814 (2021).
Ferreira, L. B., Tirapeli, K. G., Silva, C. C. & Goldberg, T. B. L. Lumbar spine bone mineral density in women breastfeeding for a period of 4 to 6 months: systematic review and meta-analysis. Int. Breastfeed. J. 18, 68 (2023).
Hysaj, O. et al. Parathyroid hormone in pregnancy: vitamin D and other determinants. Nutrients 13, 360 (2021).
Augustine, M. et al. Bone mineral density during and after lactation: A comparison of African American and Caucasian women. Calcif Tissue Int. 113, 426–436 (2023).
Oboh, I., Coleman, C. & Cremona, A. The influence of lactation and its duration on bone mineral density in pregnancy and postpartum: A systematic review with meta-analysis. Clin. Nutr. ESPEN. 46, 121–132 (2021).
Cohen, A. Bone metabolism, bone mass, and bone structure during pregnancy and lactation. Endocrinol. Metab. Clin. North Am. 53, 453–470 (2024).
Boot, A. M., De Ridder, M. A. J., Van Der Sluis, I. M., Van Slobbe, I. & Krenning, E. P. De muinck Keizer-Schrama SMPF. Peak bone mineral density, lean body mass and fractures. Bone 46, 336–341 (2010).
Lu, L-J-W., Nayeem, F., Anderson, K. E., Grady, J. J. & Nagamani, M. Lean body mass, not Estrogen or progesterone, predicts peak bone mineral density in premenopausal women. J. Nutr. 139, 250–256 (2009).
Nguyen, H. G., Pham, M. T. D., Ho-Pham, L. T. & Nguyen, T. V. Lean mass and peak bone mineral density. Osteoporos. Sarcopenia. 6, 212–216 (2020).
Acknowledgements
We want to express our gratitude to every researcher in this study.
Funding
Lijun Cai is currently receiving grants from the Ningxia Science and Technology Benefit People Project (2022CMG03043).
Author information
Authors and Affiliations
Contributions
Y.Y. and L.C. conceived and designed the study. Y.Y. and R.C. collected the data. D.Z. and W.Q. calculated the FRAX scores. Y.Y. and W.Q. analyzed the data. F.C, Y.W. and R.M. designed the figures. Y.Y. and R.C. wrote the manuscript. All authors reviewed and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent for publication
Not applicable.
Ethics approval and consent to participate
The study was performed according to the Helsinki Declaration and approved from the Institutional Review Board of the People’s Hospital of Ningxia Hui Autonomous Region, Ningxia Medical University (2025-LL-048) where the experiment was performed.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
You, Y., Chang, R., Qiu, W. et al. Association of reproductive characteristics with osteoporosis and FRAX scores in Chinese postmenopausal women: a QCT-based cross-sectional study. Sci Rep 15, 24937 (2025). https://doi.org/10.1038/s41598-025-09387-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-09387-z