Abstract
Yangyin Tongnao Granules (YYTN) is a traditional Chinese medicinal prescription that has been proposed to offer value as a treatment for ischemic stroke (IS). The mechanistic basis for its function, however, remains to be established. This study investigates the molecular mechanism of YYTN in treating IS through the HIF-1 signaling pathway. Two databases were accessed to determine the ingredients and targets of YYTN, while genes associated with IS were identified through GeneCards, DisGeNET, and Drugbank. Protein–protein interaction (PPI) analyses were conducted in Cytoscape. GO and KEGG analyses were performed in R4.2.3 and used to clarify the functions of particular genes and associated signaling pathways. Core targets associated with YYTN and IS were analyzed through molecular docking analyses. Corresponding analyses of associated genes and proteins of interest were then conducted in a rat model of cerebral ischemia using techniques including ELISAs, Western immunoblotting, immunohistochemistry and qPCR. YYTN was found to contain 150 active compounds, among which the core compounds were 3,9-di-O-methylnissolin, vallesiachotamine, and chrysotoxine. The most important targets of YYTN in the context of treating IS were identified as SRC, PIK3R1, and STAT3. The GO terms (biological process, cellular component, and molecular function) most closely associated with the action of YYTN were positive regulation of the MAPK cascade, membrane raft, and transmembrane receptor protein tyrosine kinase activity, respectively. The HIF-1 pathway was one of the top 15 most enriched KEGG pathways, with this pathway being associated with HIF-1α, VEGFA, and PAI-1. In molecular docking analyses, tested targets exhibited stable binding. Experimental analyses provided potential support for the ability of YYTN to exert beneficial therapeutic effects in IS through increases in HIF-1α levels in the brain tissue together upregulation of VEGFA and downregulation of PAI-1. These results provided support for the above network pharmacology analyses, confirming that YYTN is capable of impacting HIF-1α, VEGFA, and PAI-1 expression while also altering the activity of the HIF-1 signaling pathway. These results offer support for the network pharmacology results, demonstrating the ability of YYTN to exert therapeutic benefits in IS by modulating HIF-1α, VEGFA, and PAI-1 gene and protein expression.
Similar content being viewed by others
Introduction
Rates of stroke incidence have been rising throughout the world owing to economic changes, and these events have been affecting younger individuals1. Strokes are classified into cases of ischemic stroke (IS), cerebral hemorrhage stroke, and subarachnoid hemorrhage stroke. Of these, IS is the most common, with its incidence often being linked to the incidence of cardiovascular and cerebrovascular diseases2. As a treatment method for IS, tissue-type plasminogenactivator has a short therapeutic window and cannot provide benefits for patients who miss the optimal therapeutic window or experience sequelae3,4. There is thus a need to devise alternative therapeutic strategies.
Traditional Chinese medicine (TCM) practices have been extensively implemented for more than 2000 years across several Asian nations. Yangyin Tongnao Granules (YYTN) is a TCM prescription composed of six traditional medicines5: Shengdihuang (Rehmannia glutinosa Libosch.), Shihu (Dendrobium officinale Kimura et Migo), Gegen (Pueraria lobata (Willd.) Ohwi), Huangqi (Astragalus membranaceus (Fisch.) Bge.), Chuanxiong (Ligusticum chuanxiong Hort.) and Shuizhi (Hirudo nipponica Whitman). It had already been approved by the National Medical Products Administration of China in 2003 and the phase II/III clinical trials of YYTN have been executed for IS patients in the acute and convalescent phases. The fingerprint of YYTN extract was obtained by high-performance liquid chromatography (HPLC) previously6,7, which ensured the consistency and stability of the experimental formulation quality. Previous clinical research showed that YYTN could reduce patients’ scores of neurological impairment and improve their cognitive function8. Previous animal studies suggested that YYTN primarily exerts its therapeutic effects via stimulating neurogenesis, providing antioxidant benefits, suppressing inflammation, preventing the apoptotic death of neurons, and other mechanisms9. How this TCM Granules protects against IS at the mechanistic level, however, warrants further study.
Network pharmacology approaches through the construction of protein networks and enrichment analysis have emerged as a robust means of exploring the potential pharmacological and mechanistic basis for the effects of TCM preparations, helping to translate TCM practices into evidence-based medical systems. Such network pharmacology studies are widely used to screen for active TCM ingredients, facilitate drug repositioning, and clarify the complex mechanisms through which TCM preparations exert their effects10.
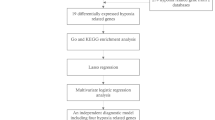
Building upon the pharmacological potential of YYTN in IS treatment, this study integrated a systematic YYTN-component-target-IS network analysis with molecular docking and experimental validation to comprehensively elucidate the underlying protective mechanisms of YYTN against IS-induced pathological consequences.
Network pharmacology
YYTN ingredient and target selection
Chemical compounds found in YYTN were identified with the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP, http://tcmspw.com/tcmsp.php) and Traditional Chinese Medicines Integrated Database (TCMID, http://www.megabionet.org/tcmid/). Active ingredients were retained if they exhibited an oral bioavailability (OB) ≥ 30% and a drug-likeness (DL) ≥ 0.1811,12. These were then explored in TCMID as well as PubChem (https://pubchem.ncbi.nlm.nih.gov/) for canonical SMILES analyses, selected with the five principles of class drugs (Lipinski’s Rule of Five) in SwissADME (http://www.swissadme.ch/), and introduced into SwissTargetPrediction (http://www.swisstargetprediction.ch) to determine the associated target candidates.
IS-related target selection
IS-related target genes were obtained from Genecards (https://www.genecards.org/), Drugbank (https://www.drugbank.ca/), and DisGeNET (https://www.disgenet.org/) using the keywords “cerebral ischemic stroke” and “cerebral infarction”. Targets in Genecards were screened based on relevance levels exceeding the median. Targets from Drugbank were introduced into UniProt (https://www.uniprot.org/) as a means of obtaining standardized gene names. The results from the three databases were then pooled and deduplicated.
Network construction
YYTN- and IS-related target genes were assessed in Venny2.1.0 (http://bioinfogp.cnb.csic.es/tools/venny/) to determine the intersecting overlapping genes. A YYTN-ingredient-target-IS network was then developed in Cytoscape3.7.2 to gain a more intuitive understanding of the relationships between YYTN and IS.
Protein–protein interaction (PPI) analyses
STRING (https://string-db.org/) was employed to assess interactions among genes and common targets associated with YYTN and IS. The established network was visualized in Cytoscape3.7.2 and subjected to topological analysis.
Functional enrichment analyses
Target genes in the PPI network were analyzed using ClusterProfiler13, org.Hs.eg.db, enrichplot, and ggplot2 in R4.2.3. This approach was used to perform GO analyses of molecular function (MF), biological process (BP), and cellular component (CC) terms. KEGG analyses14,15,16 were also used to explore the biological pathways through which YYTN was predicted to function, focusing on signaling pathways enriched for potential targets. P < 0.05 was deemed statistically significant.
Molecular docking
To expand on the network pharmacology predictions, molecular docking analyses implemented using AutoDock14 were conducted for the four drug compound associated with YYTN with the highest degree values and the four targets with the highest degree values in the PPI network. Good binding was represented by a binding energy < − 5 kcal/mol15. Docking and binding patterns were ultimately generated with Pymol.
Experimental materials and methods
YYTN preparation
YYTN is a national innovative drug and has obtained clinical research approval, approval number: 2003L00206. YYTN comprising six Chinese traditional medicines (Rehmannia glutinosa Libosch.; Dendrobium officinale Kimura et Migo; Pueraria lobata (Willd.) Ohwi; astragalus membranaceus (Fisch.) Bge.; Ligusticum chuanxiong Hort.; Hirudo nipponica Whitman) was provided by Shandong Buchang Pharmaceuticals Co.,Ltd and the batch number was 200602. There was 5.5 g in each package, and it was prepared with sterile water when used. The active compounds in YYTN specimen had been identified by high-performance liquid chromatography (HPLC) with an injection volume of 20 μL, and fow velocity was set at 0.8 mL/min in the previous experiment5. This ensured the reliability of the sample.
Animal
A total of 24 Sprague–Dawley rats (3–4 months old, 250 ± 20 g) from Shanghai SLAC Laboratory Animal Co. LTD were housed in a controlled environment (22 ± 1 °C, 50% ± 10% humidity, 12 h light/dark cycle), with animals being acclimatized for 1 week under routine care. The Animal Experiment Center of Zhejiang Chinese Medical University approved all animal studies (IACUC-20220606-05), which were consistent with the criteria of the Chinese Medical Ethics Committee.
Animal modeling
A modified Zea Longa middle cerebral artery occlusion (MCAO) model16 was established as it provides advantages including the absence of craniotomy, a lack of systemic effects, constant ischemia, and the ability to accurately control the duration of ischemia and reperfusion17. Briefly, rats were anesthetized (intramuscular atropine [0.04 mg/kg] and intraperitoneal Shutai 50 [40 mg/kg]) and a midline neck incision was used for exposure of the common carotid artery (CCA), external carotid artery (ECA), and internal carotid artery (ICA), followed by the insertion of a 0.28 mm polylysine-coated nylon monofilament from the CCA into the ICA until reaching the middle cerebral artery at a depth of 18 ± 2 mm. Following ischemic induction for 1 h, this filament was removed. MCAO modeling success was assessed based on the presence of Horner syndrome in the right eye, circling to the left, and inextensible contralateral paws of the forelimb. Based on the comparison of body surface area in rats and humans, the dose of YYTN selected for use in this study was 1.65 g/kg5. The animals were randomized into various groups (n = 6 /group): control, MCAO model, YYTN treatment (1.65 g/kg), and Edaravone treatment (6.3 mg/kg) groups. The animals in the YYTN and Edaravone groups were given their first dose after 24 hours of ischemia-reperfusion, followed by daily gavage of the appropriate compound for 2 weeks.
ELISAs
Rats were fasted for 12 h following the final treatment, deeply anesthetized, and blood samples were obtained via cardiac puncture. After centrifugation (4500 rpm, 12 min, 4 °C), the sera were retained and stored at − 20 °C. Serum HIF-1α and PAI-1 levels were analyzed with commercial ELISA kits (Jiangsu Enzyme Label Biotechnology Co., LTD., Jiangsu, China).
Western immunoblotting
Samples of homogenized and lysed brain tissue from ratsin each group were centrifuged (12,000 rpm, 5 min, 4 °C) and then analyzed via BCA (Solarbio, pc0020). Samples were then separated via 5% SDS-PAGE, transferred to blots, and probed with primary antibodies specific for β-actin (affinity, AF7018), HIF-1α (28b) (SantaCruz, sc-13515), and VEGFA [VG-1] (abcam, ab1316) and secondary antibodies (Anti-rabbit IgG, HRP-linked (CST, 7074). ECL reagent was used for band development, with protein bands then being analyzed with an ECL chemiluminescence instrument (Clinx Science Instruments Co., Ltd., 610020-9Q) and the ChemiCapture software.
Immunohistochemistry (IHC)
Paraffin-embedded brain sections were deparaffinized, treated with citric acid buffer (pH 6.0, MDL, MD911411) for antigen retrieval, and decolorized using PBS (pH 7.4, MDL, MD911705), treated with 3% hydrogen peroxide for blocking endogenous peroxidase activity, followed by blocking with 3% BSA. They were then probed with primary and secondary antibodies (anti-rabbit IgG, HRP-linked antibody (CST, 7074)) were added for reaction, followed by DAB color development (Servicebio, G1211) and hematoxylin staining. Samples were imaged with an upright fluorescence microscope (Leica, DM3000) and analyzed to clarify how YYTN affects HIF-1α and VEGFA.
qPCR
Trizol was used to isolate total RNA from rat brain tissues, followed by the dissolution of this RNA in 40 µL of DEPC water and the measurement of RNA concentrations. These samples were then used for first-strand cDNA synthesis, followed by qPCR analysis using SYBR Green Master Mix in a PCR instrument (Eppendorf, Mastercycler) with the settings: 95 °C for 10 min; 40 cycles of 95 °C for 15 s, 60 °C for 60 s. The 2−ΔΔCT method was used to determine relative expression, with GAPDH as a normalization control. Utilized primers are shown in Table S1.
Data analyses
Results were analyzed and visualized with SPSS 26.0 and GraphPad Prism 8. If data were normally distributed with homogenous variance, they were analyzed via one-way ANOVAs with Tukey’s test. Data are presented as means ± standard deviation (\(\overline{x }\)± s), with P < 0.05 as the significance threshold.
Results
YYTN target protein identification
A total of 27 compounds in YYTN in the TCMSP database met the criteria for inclusion, including 16, 4, and 7 respectively belonging to Huangqi, Gegen, and Chuanxiong. In addition, 445 compounds in YYTN in the TCMID database met the criteria for inclusion, including 70 belonging to Huangqi, 51 belonging to Gegen, 182 belonging to Chuanxiong, 49 belonging to Shengdihuang, 58 belonging to Shihu, and 35 belonging to Shuizhi. After these two datasets were merged and deduplicated, the remaining ingredients were introduced into PubChem for queries of Canonical SMILES. Based on the three established SwissADME criteria of gateway absorption (high), Lipinski (yes), and Veber (yes), 150 compounds were identified (Table S2). Following the removal of duplicates, 856 predicted protein targets of these compounds were identified with the online SwissTarget Prediction tool at a probability threshold ≥ 0.05 (Table S3).
IS-Related target selection
In total, 3276, 687, and 82 IS-related targets were identified from GeneCards, DisGeNET, and Drugbank, of which 2,428 targets were retained after deduplication (Table S4).
Drug-compound-target-disease network
Venny2.1.0 was used to compare these lists of YYTN-related and IS-related target proteins, ultimately yielding a list of 434 candidate IS-related treatment targets of YYTN (Fig. 1).
Intersection genes of YYTN and IS.
PPI network establishment
The STRING database was used to generate a PPI network (species: Homo sapiens) followed by visualization with Cytoscape3.7.2 in Fig. 2. Topological analyses were conducted to determine the sizing and color of targets based on degree values, while edge thickness corresponded to the thickness of edge combined scores. The resultant network had 434 nodes, 1,604 edges, 7.39 average node degree, P-value of PPI enrichment < 1.0 exp (− 16). The IS-related targets of YYTN included SRC, PIK3R1, STAT3, MAPK3, VEGFA, HIF and so on. PPI network analysis prioritized hub proteins (PIK3R1/PI3K-AKTpathway, MAPK3/MAPK pathway, VEGFA/VEGF pathway, HIF1A/HIF-1 pathway) as mechanistic candidates.
PPI Network diagram of YYTN and IS.
Functional enrichment analyses
GO and KEGG analyses were next conducted for the targets identified above, revealing 3,123, 147, and 286 GO-BP, CC, and MF terms, respectively. Based on the adjusted statistical significance P-values, the top 10 most significant terms for each category were plotted (Fig. 3).
GO-BP/CC/MF enrichment analyses of IS-related targets (Top 10). (Gene counts for individual terms are shown on the x-axis. Pathway names are shown on the y-axis. Adjusted P-values are presented with the color scale.)
The three most highly ranked BP terms were: positive regulation of MAPK cascade (GO: 0043410), response to xenobiotic stimulus (GO: 0009410), and response to oxygen levels (GO: 0070482). The response to oxygen levels entry in the bubble plot had a high enrichment factor and low P-value, providing key clues for understanding the molecular mechanism of YYTN therapy for hypoxia related diseases. The three most highly ranked CC terms were: membrane raft (GO: 0045121), membrane microdomain (GO: 0098857), and synaptic membrane (GO: 0097060). This suggested that YYTN may treat IS by altering the localization of signaling proteins in membrane rafts and regulating the expression of synaptic membrane-associated proteins. The three most highly ranked MF terms were: transmembrane receptor protein tyrosine kinase activity (GO: 0004714), transmembrane receptor protein kinase activity (GO: 0019199), and protein tyrosine kinase activity (GO: 0004713). They indicated that YYTN therapy for IS was likely associated with the tyrosine kinase family, such as VEGFA.
A total of 186 KEGG pathways were found, and the top 15 terms showing the highest statistical significance (adjusted P-value < 0.05) were shown in Fig. 4. Among them, the HIF-1 signaling pathway (hsa04066, P-value = 3.61 exp(-26)) was a key regulatory pathway for cellular hypoxia response. Combining GO analysis and KEGG analysis, YYTN treatment for IS may be related to compensatory responses under hypoxic conditions. The HIF-1 signaling pathway map18,19,20 was plotted (Fig. 5), and the red rectangles represent the genes or proteins enriched in this study. The key genes or proteins enriched in this pathway included STAT3, PI3K, HIF-1α and downstream VEGF and PAI-1, which could guide the selection of key proteins and genes in experiments.
Dot plot of the pathways of IS in KEGG (Top 15). (The enrichment gene ratio in each pathway is shown on x-axis. The name of each pathway is shown on the y-axis. The gene count according with each term are presented with the size of the dot. Adjusted P-values are presented with the color of the scale.)
The enrichment results included in the HIF-1 signaling pathway map.
Molecular docking analyses
Molecular docking was used to validate the feasibility of network pharmacology predictions at the structural level of molecular interactions. 3D structures of the four drug compounds (3,9-di-O-methylnissolin, chrysotoxine, erianin and vallesiachotamine) were from the PubChem database. The PBD ID of the four targets (SRC, STAT3, PIK3R1 and MAPK3) were 7NG7, 6NJS, 4JPS, 4QTB from the PDB database. After downloading 3D structures for these compounds from the PubChem database, PyMol was used to remove heteroatoms and water molecules. Molecular docking was performed with AutoDock vina20,21,22, using the position of the original ligand as the docking site. Docking scores were used to select optimal conformations. The good binding activity (< − 5 kcal/mol) between the compounds and the target protein were presented in Table 1. The four optimal binding conformations between the compounds and the target protein (including hydrogen bonds and amino acid residues) were shown in Fig. 6.
Schematic diagram of the optimal conformation for molecular docking.
Through molecular docking results and the HIF-1 signaling pathway map (Fig. 5), it was found that the binding energy of STAT3 and PI3K was low. However, HIF-1 was a common downstream indicator of these two targets23,24, and the enrichment of GO and KEGG confirmed hypoxia related processes and activation of the HIF-1 pathway. VEGF and PAI-1 were downstream indicators associated with angiogenesis in HIF-1 pathway25,26. Therefore, the experimental validation prioritized investigating the mechanism by which YYTN ameliorates cerebral ischemia–reperfusion injury through HIF-1 pathway modulation to promote angiogenesis.
Analyses of serum HIF-1α and PAI-1 expression
In ELISAs, serum HIF-1α and PAI-1 levels were measured in different groups of rats (Fig. 7). Significantly higher serum HIF-1α and PAI-1 levels were observed in the Model group relative to control rats (P < 0.05 and P < 0.01, respectively), HIF-1α levels in the YYTN and Edaravone groups were markedly reduced relative to the Model group (P < 0.05). PAI-1 levels were also significantly reduced in the YYTN group relative to the Model group (P < 0.05), with a more marked reduction seen in the Edaravone group (P < 0.01).
Serum HIF-1α and PAI-1 expression (\(\overline{x }\)±s, n = 6). (Meaning of capital letters in abscissa: (A) Control; (B) Model; (C) YYTN; (D) Edaravone).
Analyses of brain HIF-1α and VEGFA protein expression
Next, Western immunoblotting (WB) was used to evaluate HIF-1α and VEGFA levels in the brains of rats in different groups (Fig. 8). HIF-1α and VEGFA levels in the Model group were markedly raised relative to those in the control group (P < 0.05 and P < 0.01, respectively), while the levels of these proteins in the YYTN and Edaravone groups were significantly elevated (P < 0.01). HIF-1α levels in the Edaravone group were markedly raised relative to the YYTN group (P < 0.05), while the VEGFA levels showed no change (P > 0.05).
Brain HIF-1α and VEGFA protein expression in WB (\(\overline{x }\)±s, n = 3). ((a) Protein expression cut bands of HIF-1α and VEGFA; (b) histogram of HIF-1α and VEGFA) (meaning of capital letters in abscissa: A: Control; B: Model; C: YYTN; D: Edaravone).
IHC staining revealed a significant increase in the average optical density (AOD) of HIF-1α and VEGFA in the Model group compared with the controls (Fig. 9) (P < 0.05). The AOD for HIF-1α and VEGFA in the YYTN group was markedly raised relative to the Model group (P < 0.05), with an even more significant increase in the Edaravone group (P < 0.01).
Brain HIF-1α and VEGFA protein expression in IHC (\(\overline{x }\)±s, n = 3). ((a) Staining sections of rat brain tissue with HIF-1α under vertical fluorescent microscope at different magnification and histograms of HIF-1α immunoreactive products AOD in brain tissue; (b) staining sections of rat brain tissue with VEGFA under vertical fluorescent microscope at different magnificationand histograms of HIF-1α immunoreactive products AOD in brain tissue.) (Meaning of capital letters in abscissa: A: Control; B: Model; C: YYTN; D: Edaravone;)
Analyses of brain HIF-1α and VEGFA mRNA levels in qPCR
Significant increases in the mRNA levels of HIF-1α and VEGFA expression were evident in the Model group relative to the controls (Fig. 10) (P < 0.05). No differences in HIF-1α expression were observed when comparing the YYTN and Model groups (P > 0.05), while these levels were markedly raised in the Edaravone treatment group (P < 0.05). VEGFA expression decreased markedly relative to the Model group in response to both YYTN and Edaravone treatment (P < 0.05).
Histogram of brain HIF-1α and VEGFA gene expression in qPCR (\(\overline{x }\)±s, n = 3). (Meaning of capital letters in abscissa: A: Control; B: Model; C: YYTN; D: Edaravone;)
Discussion
IS occurs as a result of the disruption of arterial blood flow in the brain and consequent hypoxia-related ischemic necrosis in the surrounding brain tissue. This article explored the mechanism of YYTN treating IS through the hypoxia signaling pathway—HIF signaling pathway combining network pharmacology and experimental verification. In this study, YYTN, which consists of 6 traditional TCMs, was subjected to network pharmacology analyses, leading to the identification of 150 bioactive compounds within these medicines. The key ingredients in Huangqi include astragaloside, which can reportedly help prevent and treat cerebrovascular disease by protecting nerve cells and preventing apoptotic death27,28. The key ingredients in Chuanxiong include tetramethylpyrazine, which can prevent or treat IS through vascular expansion, neuroprotection, and antioxidant effects29,30. The key ingredients in Shihu include dendrobine, which can lower blood pressure to prevent IS31,32. Daidzein, derived from Gegen, can contribute to improved neurological function scores and to significant reductions in blood fibrinogen levels in cerebral infarction patients30,33.
GO enrichment analysis-derived BP terms highlighted the important role of MAPK signaling in stroke, suggesting that drugs and genes associated with this pathway may be relevant to the alleviation of ischemic brain injury34. With respect to CC terms, membrane lipid rafts are very important for survival signaling in neurons35. Among MF terms, protein tyrosine phosphatase 1B-induced microglia activation can lead to the worsening of neuroinflammation and aggravated brain injury36. Based on network pharmacology and KEGG enrichment analyses, various key proteins were identified including HIF-1α, VEGFA, and PAI-1, which are related to the HIF-1 signaling pathway, as has been reported previously. HIF-1 is a key regulator of hypoxic activity, influencing the expression of more than 700 target genes associated with both adaptive and damaging processes37. HIF-1 can regulate both VEGF and PAI-138. Network pharmacology can search for the main active ingredients of traditional Chinese medicine formulas and their mechanisms of action in treating diseases39. However, network pharmacology also has limitations, as its predictions depend on the comprehensiveness and accuracy of the reference database. For example, owing to the overly large chemical formula of hirudin, which is the primary ingredient in Shuizhi, its structure could not be determined such that it was omitted from these analyses. Inaccurate or blank data in these databases may affect the reliability of the results. Therefore, other methods such as experiments are needed to validate these findings40.
After completing the above network pharmacology and bioinformatics analyses, experimental validation was performed through a series of Western blotting, IHC, and qPCR analyses demonstrating that YYTN was able to treat IS through the enhancement of brain HIF expression. Edaravone is a potent antioxidant that has been proposed as an effective component of stroke care in Asian nations41, and it was thus selected here as a positive control drug. The upregulation of HIF-1α can reduce neuronal apoptosis and improve systemic IS resistance42. YYTN-based IS treatment was associated with increased VEGFA protein levels in the brains of MCAO model rats as determined through IHC and Western blotting. Higher levels of VEGFA have been demonstrated to facilitate collateral circulation, contributing to better angiogenesis and post-IS recovery43. Astragaloside IV can reportedly improve VEGF levels and markedly bolster HIF/VEGF pathway activity28. Under YYTN and edaravone treatment in qPCR, however, brain VEGFA levels decreased. Significant reductions in VEGFA expression levels have been reported during cerebral ischemia44. Alternative splicing can also give rise to multiple subtypes of VEGFA45. Additional studies will be necessary to overcome the limitations of the present analyses. ELISAs performed herein revealed that serum PAI-1 levels declined in response to YYTN and edaravone treatment. PAI-1 deficiency can reportedly lead to reductions in infarct size and improved recovery of cerebral blood flow46. However, acute IS patients reportedly exhibit higher PAI-1 levels, and no differences have been reported among cerebral infarction clinical subgroups47. PAI-1 may thus be an effective biomarker of IS incidence but a poor indicator of stroke length or severity.
Conclusion
In conclusion, the network pharmacology analyses performed herein offer new insights into the multi-component multi-target nature of YYTN-mediated IS treatment. Molecular docking results provided further support for these network pharmacology predictions, while animal model experiments confirmed the efficacy of YYTN as an IS treatment capable of improving cerebral arterial blood flow interruptions and protecting against hypoxic-ischemic necrosis in the brain through the regulation of HIF-1α and downstream targets including VEGFA and PAI-1. These insights into the therapeutic effects of YYTN in a model of IS can serve as a foundation for efforts to develop new treatments for IS. In the future, in-depth studies will be performed to validate and expand on these findings at the cellular level.
Data availability
Data is provided within the manuscript or supplementary information files. If necessary, additional data can be provided by the corresponding author.
References
Collaborators, G. S. Global, regional, and national burden of stroke and its risk factors, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 20, 795–820 (2021).
Hu, X., Silva, T. M. D., Chen, J. & Faraci, F. M. Cerebral vascular disease and neurovascular injury in ischemic stroke. Circ. Res. 120, 449–471 (2017).
Li, T. Clinical curative effect of r-tPA intravenous thrombolysis at different time windows in treatment of acute cerebral infarction. World Latest Med. Inf. 18, 7+24 (2018).
Meschia, J. F. et al. Guidelines for the primary prevention of stroke: A statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 45, 3754–3832 (2014).
Wang, Y. et al. Yangyin Tongnao Granules enhance neurogenesis in the peri-infarct area and upregulate brain-derived neurotrophic factor and vascular endothelial growth factor after focal cerebral ischemic infarction in rats. Mol. Biol. Rep. 46, 3817–3826 (2019).
Yu, L. et al. Spectrum-effect relationship between HPLC fingerprints and antioxidant activity of Yangyin Tongnao prescription. J. Anal. Methods Chem. 2021, 6650366 (2021).
Jin, L. et al. Simultaneous optimization of the extraction process of Yangyin Yiqi Huoxue prescription with natural deep eutectic solvents for optimal extraction yield and antioxidant activity: A comparative study of two models. Phytomed. Int. J. Phytother. Phytopharmacol. 102, 154156 (2022).
Wang, Y. et al. Efficacy of Yangyin Yiqi Huoxue Granule () in treatment of ischemic stroke patients with Qi-Yin deficiency and blood stasis syndrome: A randomized, double-blind, multicenter, phase-2 clinical trial. Chin. J. Integr. Med. 27, 811–818 (2021).
Wang, Y. et al. Effects of Yangyin Tongnao Granules on the expression of Caspase-3, Bax, Bcl-2 mRNA in rats after cerebral ischemia/reperfusion injury. China J. Tradit. Chin. Med. Pharm. 33, 3875–3878 (2018).
Chen, J. & Chen, Q. Current situation and thinking of network pharmacology in the research of traditional Chinese medicine. Acta Univ. Tradit. Med. Sin. Pharmacol. Shanghai 35, 1–6 (2021).
Ru, J. et al. TCMSP: A database of systems pharmacology for drug discovery from herbal medicines. ChemInform 6, 13 (2014).
Xiao, G. et al. Network pharmacology analysis and experimental validation to explore the mechanism of Bushao Tiaozhi capsule (BSTZC) on hyperlipidemia. Sci. Rep. 12, 6992 (2022).
Wu, T. et al. clusterprofiler 4.0: A universal enrichment tool for interpreting omics data. Innovation (Cambridge) 2, 100141 (2021).
Delano, W. L. PyMOL: An Open-Source Molecular Graphics Tool. (2002).
Zhu, W. et al. The mechanism of triptolide in the treatment of connective tissue disease-related interstitial lung disease based on network pharmacology and molecular docking. Ann Med. 54, 541–552 (2022).
Longa, E. Z. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20, 84–91 (1989).
Xi, Y., Wang, H. & Lu, S. Research progress of focal ischemic stroke in production of animal models. Chin. J. Comp. Med. 33, 140–148 (2023).
Kanehisa, M. et al. KEGG: Biological systems database as a model of the real world. Nucleic Acids Res. 53, D672–D677 (2025).
Kanehisa, M. Toward understanding the origin and evolution of cellular organisms. Protein Sci. 28, 1947–1951 (2019).
Kanehisa, M. & Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28, 27–30 (2000).
Eberhardt, J. et al. AutoDock Vina 1.2.0: New docking methods, expanded force field, and python bindings. J. Chem. Inf. Model. 61, 3891–3898 (2021).
Trott, O. & Olson, A. J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 31, 455–461 (2010).
Jin, W. et al. Neuronal STAT3/HIF-1α/PTRF axis-mediated bioenergetic disturbance exacerbates cerebral ischemia-reperfusion injury via PLA2G4A. Theranostics. 12, 3196–3216 (2022).
Zhang, Z. et al. PI3K/Akt and HIF-1 signaling pathway in hypoxia-ischemia (review). Mol. Med. Rep. 18, 3547–3554 (2018).
Ni, H. et al. Cardamonin attenuates cerebral ischemia/reperfusion injury by activating the HIF-1α/VEGFA pathway. Phytother. Res. 36, 1736–1747 (2022).
Jung, S. Y. et al. Pyruvate promotes tumor angiogenesis through HIF-1-dependent PAI-1 expression. Int. J. Oncol. 38, 571–576 (2011).
Huang, X. et al. Effects of the combination of the main active components of astragalus and panax notoginseng on inflammation and apoptosis of nerve cell after cerebral ischemia-reperfusion. Chin. Tradit. Herb. Drugs 46, 2257–2264 (2015).
Liang, C. et al. Astragaloside IV regulates the HIF/VEGF/Notch signaling pathway through miRNA-210 to promote angiogenesis after ischemic stroke. Restor. Neurol. Neurosci. 38, 271–282 (2020).
Cao, G. et al. Research progress of traditional Chinese medicine treating ischemic stroke. Liaoning J. Tradit. Chin. Med. 46, 2666–2671 (2019).
Liao, S. et al. Tetramethylpyrazine reduces ischemic brain injury in rats. Neurosci. Lett. 372, 40–45 (2004).
Li, X. Study on the effect and mechanism of Dendrobium nobile Lindl. alkaloids on ischemic stroke, Hefei University of Technology. (2022).
Li, Y. Research progress on pharmacological effects of Dendrobium officinale on cardiovascular diseases. Food and Nutr. China 28, 62–66 (2022).
Ahmed, T. et al. Daidzein and its effects on brain. Curr. Med. Chem. 24, 365–375 (2017).
Liu, C. et al. Metformin protects against pericyte apoptosis and promotes neurogenesis through suppressing JNK p38 MAPK signalling activation in ischemia/reperfusion injury. Neurosci. Lett. 783, 136708 (2022).
Wei, Z. et al. Caveolin-1 and MLRs: A potential target for neuronal growth and neuroplasticity after ischemic stroke. Int. J. Med. Sci. 16, 1492–1503 (2019).
Zhu, Y. et al. PTP1B inhibitor alleviates deleterious microglial activation and neuronal injury after ischemic stroke by modulating the ER stress-autophagy axis via PERK signaling in microglia. Aging 13, 3405–3427 (2021).
Dengler, V. L. et al. Transcriptional regulation by hypoxia inducible factors. Crit. Rev. Biochem. Mol. Biol. 49, 1 (2014).
Leu, T. et al. When the brain yearns for oxygen. Neurosignals 27, 50–61 (2019).
Lai, X. et al. Editorial: Network pharmacology and traditional medicine. Front Pharmacol. 11, 1194 (2020).
Li, S. et al. Suppressive effects of a Chinese herbal medicine qing-luo-yin extract on the angiogenesis of collagen-induced arthritis in rats. Am. J. Chin. Med. 31, 713–720 (2003).
Tang, Y., Meng, Q. & Yu, H. Effect of Edaravone on cognitive function in patients with acute cerebral infarction. J. Qiqihar Med. Coll. 31, 2726–2727 (2010).
Snigdha, S. et al. Caspase-3 activation as a bifurcation point between plasticity and cell death. Neurosci. Bull. 28, 14–24 (2012).
Xiang, Y. et al. Houshiheisan promotes angiogenesis via HIF-1α/VEGF and SDF-1/CXCR4 pathways: In vivo and in vitro. Biosci. Rep. 39, BSR20191006 (2019).
Liang, Y. et al. The study of chemical components in Qishiwei Zhenzhu pills and its anti-apoptotic mechanism in cerebral ischemic based on LC-MS and network pharmacology. J. Ethnopharmacol. 302, 115891 (2023).
Bates, D. O. et al. Detection of VEGF-A(xxx)b isoforms in human tissues. PLoS ONE 8, e68399 (2013).
Torrente, D. et al. Compartmentalized actions of the plasminogen activator inhibitors, PAI-1 and Nsp, in ischemic stroke. Transl. Stroke Res. 13, 801–815 (2022).
Lindgren, A. et al. Tissue plasminogen activator and plasminogen activator inhibitor-1 in stroke patients. Stroke: A J. Cereb. Circ. 27, 1066 (1996).
Funding
This study was partially funded by the National Natural Science Foundation of China Nos. 82330120, 81904083, Natural Science Foundation of Zhejiang Province Nos. LZ23H270002, and LY24H270007, Key Research and Development Program of Zhejiang Province No. 2024C03105, Zhejiang Province Traditional Chinese Medicine Science and Technology Plan Project No. 2023ZR011, China Postdoctoral Science Foundation No. 2023TQ0295, Zhejiang Province Postdoctoral Research Project Selection Funding (First Class Funding) No. ZJ2023021, Research Project of Zhejiang Chinese Medical University, No. 2024RCZXZK24.
Author information
Authors and Affiliations
Contributions
JYY wrote, revised the manuscript, and visualized network pharmacology and experimental data. QQC and XTD conducted experiments, investigate and analyzed experimental data. RJZ validated the experiment. JHY studied activity planning and execution management. WFJ proposed overall research objectives and project funding. HTW provided experimental resources and project funding. LY supervised the execution of the project and provided project funding. YYZ validated the experiment and reviewed the revision of the manuscript. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The Animal Experiment Center of Zhejiang Chinese Medical University scrupulously followed the regulations, conformed to the criteria outlined in ARRIVE guidelines, and strictly adhered to all rules related to animal handling. The approval number of animal ethical and welfare was IACUC-20220606–05.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yu, J., Chen, Q., Dou, X. et al. Network pharmacology analyses and corresponding validation of the mechanistic effects of Yangyin Tongnao Granules in ischemic stroke. Sci Rep 15, 28074 (2025). https://doi.org/10.1038/s41598-025-10292-8
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-10292-8