Abstract
Breast cancer is one of the most common malignant tumors in women worldwide. Inflammation plays an important role in the occurrence and development of breast cancer. This study aimed to evaluate the association between inflammatory markers and breast cancer prevalence and explore their potential biomarker value. Using data from the NHANES, approximately 20,000 participants were analyzed to assess the association between six systemic inflammatory markers and breast cancer prevalence. Statistical methods including multivariable logistic regression, subgroup analysis, and interaction testing were employed. ROC curves were used to evaluate and compare their diagnostic capabilities. This study included 19,734 participants. We observed a significant positive correlation between platelet-to-lymphocyte ratio (PLR) and breast cancer prevalence (OR = 1.35; 95% CI:1.09, 1.67), with PLR demonstrating good predictive performance for breast cancer. Additionally, monocyte-to-lymphocyte ratio (MLR), neutrophil-to-lymphocyte ratio (NLR), systemic immune-inflammation index (SII), systemic inflammation response index(SIRI) and aggregate index of systemic inflammation(AISI) were also found to be positively associated with breast cancer prevalence. Subgroup analysis and interaction tests revealed that the association between PLR and BC did not differ significantly among population groups. ROC curve analysis indicated that PLR (AUC = 0.59; 95%CI: 0.56, 0.63) outperformed other inflammatory markers in predicting BC. Systemic inflammatory markers, especially PLR, are significantly associated with BC prevalence and demonstrate potential as biomarkers for early detectionn. People with elevated inflammation markers should pay close attention to the latent prevalence of BC.
Similar content being viewed by others
Introduction
Among the most common malignant tumors worldwide, breast cancer ranks second in terms of cancer incidence in 2022. It represents a significant contributor to cancer-related mortality among women, accounting for approximately one-sixth of all female cancer deaths1. Countries with higher Human Development Index (HDI) tend to have higher breast cancer incidence rates, reflecting the prevalence of risk factors related to reproductive and lifestyle patterns. These risk factors include early menarche, late menopause, higher age at first childbirth, fewer children, less breastfeeding, hormone replacement therapy, oral contraceptive use, alcohol consumption, obesity, and lack of physical activity2. Given the scarcity of definitive modifiable risk factors, establishing a primary prevention strategy for breast cancer remains a significant challenge3,4.
During tumor initiation and development, inflammation plays a crucial role5,6. Tumor-infiltrating lymphocytes are important biomarkers for cancer stratification and can also serve as prognostic indicators7,8,9. Inflammation can be assessed using various biochemical or hematological markers10, and some inflammatory markers have been shown to be associated with cancer risk. These markers include the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), lymphocyte-to-monocyte ratio (LMR), and systemic immune-inflammation index (SII)11. Some studies have observed associations between these indicators and cancer risk and mortality, but most research has focused on using these markers as prognostic indicators, and the results have been inconsistent12. Furthermore, the evidence regarding their association with breast cancer risk is also insufficient13,14.
The National Health and Nutrition Examination Survey (NHANES) data were used to better understand the association between inflammatory markers and breast cancer prevalence. Based on a cohort of approximately 20,000 participants, six systemic inflammatory markers were analyzed to determine how they relate to breast cancer prevalence, as well as their potential as biomarkers for the early detection of breast cancer.
Materials and methods
Data source
In the United States, the NHANES survey is used to measure health and nutrition status of the non-institutionalized population15. To obtain a representative sample, it uses a stratified, multistage sampling design. A biennial survey is conducted by NHANES with approval from NCHS’ Ethics Review Board. Each participant consented to participating in the study. For more information, you can visit their website at www.cdc.gov/nchs/nhanes/.
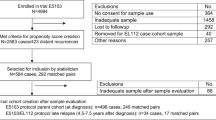
We included participants from the NHANES surveys conducted between 2007 and 2016. After a series of screening steps, we excluded individuals who were minors, those with missing demographic data, missing information on smoking and alcohol consumption status, missing data on diabetes and hypertension status, missing inflammatory markers, missing cancer data, or those with cancers other than breast cancer. Ultimately, 19,734 participants were included in the study, among whom 312 were identified as breast cancer patients(Fig. 1).
Flowchart of the sample selection from NHANES 2007–2016.
The definition of inflammatory markers and the assessment of BC
Blood cell counts of the participants were measured using an automated blood analyzer, providing counts of lymphocytes, neutrophils, monocytes, and platelets in the participants’ fasting venous blood samples collected in the morning. The counts were reported in units of 1000 cells/µL. From these counts, we calculated six inflammatory markers: PLR (platelet-to-lymphocyte ratio), NLR (neutrophil-to-lymphocyte ratio), MLR (monocyte-to-lymphocyte ratio), SII (systemic immune-inflammation index), SIRI (systemic inflammation response index), and AISI (aggregate index of systemic inflammation). \(\:\text{P}\text{L}\text{R}=\text{p}\text{l}\text{a}\text{t}\text{e}\text{l}\text{e}\text{t}\:\text{c}\text{o}\text{u}\text{n}\text{t}/\text{l}\text{y}\text{m}\text{p}\text{h}\text{o}\text{c}\text{y}\text{t}\text{e}\:\text{c}\text{o}\text{u}\text{n}\text{t}\), \(\:\text{N}\text{L}\text{R}=\text{n}\text{e}\text{u}\text{t}\text{r}\text{o}\text{p}\text{h}\text{i}\text{l}\:\text{c}\text{o}\text{u}\text{n}\text{t}/\text{l}\text{y}\text{m}\text{p}\text{h}\text{o}\text{c}\text{y}\text{t}\text{e}\:\text{c}\text{o}\text{u}\text{n}\text{t}\), \(\:\text{M}\text{L}\text{R}=\text{m}\text{o}\text{n}\text{o}\text{c}\text{y}\text{t}\text{e}\:\text{c}\text{o}\text{u}\text{n}\text{t}/\text{l}\text{y}\text{m}\text{p}\text{h}\text{o}\text{c}\text{y}\text{t}\text{e}\:\text{c}\text{o}\text{u}\text{n}\text{t}\)16. SII = platelet count * NLR17. SIRI = monocyte count *NLR18. AISI = neutrophil count * platelet count * monocyte count / lymphocyte count19.
The assessment of breast cancer was conducted by asking participants the following questions: “Have you ever been told by a doctor or other health professional that you had cancer or a malignancy of any kind?” and “What kind of cancer was it?” Participants who reported breast cancer were included in the study.
Covariates
The covariates used in our study were used to control for confounding variables, including a range of demographic indicators such as gender, age, race, education level, marital status, and the ratio of family poverty income (PIR). These data were collected through standardized questionnaires during household interviews. Additionally, measurements of weight and height were taken during physical examinations, and subsequently, body mass index (BMI) was calculated. Overweight was defined as 24 < BMI ≤ 30, and obesity was defined as BMI > 30. We also included information on participants’ smoking status, alcohol consumption, and important medical conditions and ailments, namely diabetes and hypertension. Participants who answered affirmatively to the question “Had at least 12 alcohol drinks/1 yr?” were defined as having an alcohol consumption status. Similarly, participants who answered affirmatively to the question “Smoked at least 100 cigarettes in life” were defined as having a smoking status20. Information on diabetes and hypertension was retrieved using questionnaire items. Hypertension was defined based on the response to the question “Ever told you had high blood pressure?” Participants who answered affirmatively were considered to have hypertension. Answers to the question “Doctor told you have diabetes” were used to define diabetes. Participants who answered affirmatively were considered to have diabetes.
Statistical analysis
Due to the complex multistage sampling design adopted by NHANES, as recommended by the Centers for Disease Control and Prevention (CDC), continuity variables were expressed as means with standard errors, while categorical variables were expressed as percentages. Additionally, due to the skewed distribution of the inflammatory marker data mentioned above, These inflammatory markers were log2-transformed for subsequent analysis. Three models were constructed using multivariable logistic regression to examine the relationship between these inflammatory markers and BC. Model 1 was unadjusted, Model 2 adjusted for gender, age, education level, marital status, race, and PIR, while Model 3 further adjusted for BMI, smoking status, alcohol consumption status, diabetes status, and hypertension status based on Model 2. Sensitivity analyses were performed by categorizing these inflammatory markers into tertiles to assess the stability of the results. Smooth curve fitting and GAM were utilized to explore the linear relationship between these inflammatory markers and BC. Threshold effect analysis was performed by comparing segmented regression models with a single-line model using the log-likelihood ratio test. To examine the stability of the correlation between these inflammatory markers and BC, subgroup analyses were conducted using stratified multivariable logistic regression models to assess the potential heterogeneity between different subgroups. These analyses were stratified by age, BMI, smoking status, alcohol consumption, hypertension, and diabetes, which were considered potential effect modifiers. Finally, receiver operating characteristic (ROC) curves were employed to evaluate the predictive ability of these inflammatory markers for BC. Statistical analyses were conducted using the Empower software and R software.
Results
Participant demographics
In our analysis, a total of 19,734 participants were included, including 312 patients with breast cancer. Among them, the average age of participants who did not report breast cancer was 47.60 ± 17.17 years, while the average age of participants who reported breast cancer was 67.33 ± 10.91 years (Table 1). Our study found that compared to participants without breast cancer, those with breast cancer had higher MLR, NLR, PLR, SII, SIRI, and AISI. Additionally, there were significant differences between the cancer and non-cancer groups in terms of age, PIR, lymphocyte count, gender, race, alcohol consumption, diabetes, and hypertension.
Association of inflammatory markers with BC
For the inflammatory markers, according to our survey results (Table 2), in the unadjusted, partially adjusted, and fully adjusted models, these six inflammatory markers were positively associated with BC. Subsequently, we categorized the inflammatory markers into tertiles, and participants in the highest tertiles of MLR (OR = 1.40; 95%CI:1.03, 1.89), NLR (OR = 1.39; 95%CI:1.03, 1.88), PLR (OR = 1.39; 95%CI:1.04, 1.86), SII (OR = 1.41; 95%CI:1.05, 1.90), SIRI (OR = 1.53; 95%CI:1.13, 2.07), and AISI (OR = 1.47; 95%CI:1.09, 1.99) showed higher rates of BC.
Through GAM and smooth curve fitting, we found a non-linear relationship between PLR and BC (Fig. 2), with a breakpoint (K) at 7.71 (Table 3). No significant non-linear relationships were found between other inflammatory markers and BC. In the standard linear model, SII (OR = 1.21; 95%CI 1.05, 1.40) showed linearity, that is, a linear relationship with breast cancer prevalence.
Smooth curve fitting for PLR and other inflammatory markers with BC. (A) PLR and BC; (B) MLR and BC; (C) AISI and BCB; (D) NLR and BC; (E) SII and BC; (F) SIRI and BC.
Subgroup analysis
In all groups stratified by alcohol consumption status, a positive correlation between PLR and BC was observed. Additionally, significant associations were observed in the overweight, ≥ 60 years old, non-smoking, hypertensive, and non-diabetic populations (Fig. 3). Through interaction testing, we found that diabetic status significantly altered the association between MLR and PLR and BC, while no significant associations with BC were found in subgroups stratified by BMI, age, smoking status, alcohol consumption status, and hypertension status. Furthermore, the associations between NLR, SII, SIRI, AISI, and BC remained unaffected in each subgroup.
Subgroup analysis for the associations of NLR and other inflammatory markers with BC. (A) MLR and BC; (B) NLR and BC; (C) PLR and BC; (D) SII and BC; (E) SIRI and BC; (F) AISI and BC.
The predictive value of inflammatory markers
To compare the predictive ability of each inflammatory marker for BC, we compared their AUC values (Fig. 4). Our analysis found that the platelet-to-lymphocyte ratio (PLR) shows a modest association with breast cancer prevalence (OR = 1.35; 95% CI:1.09, 1.67), suggesting a positive correlation between elevated PLR and breast cancer. However, with an AUC of 0.59(95%CI: 0.56, 0.63), sensitivity of 0.38, and specificity of 0.78, PLR demonstrates limited predictive performance as a standalone biomarker. The low sensitivity in particular indicates that PLR would miss a substantial proportion of breast cancer cases (62%). While the specificity is reasonable, suggesting potential utility in ruling in disease when elevated, the overall discriminatory ability is weak. These findings suggest PLR may have some value when considered alongside other established prevalence factors or biomarkers, but its independent predictive capability appears insufficient for clinical application as a screening or diagnostic tool. We found that PLR had better predictive performance for BC (AUC = 0.59; 95%CI: 0.56, 0.63) compared to other inflammatory markers (Table 4), and the differences in AUC values between PLR and SII, SIRI, and AISI were statistically significant.
ROC curves and the AUC values of the six inflammatory markers in diagnosing BC.
Discussion
This cross-sectional study involving 19,734 participants, we explored the relationship between systemic inflammatory markers and BC. We found a positive correlation between PLR and the BC prevalence, with PLR exhibiting better predictive performance for BC. Additionally, MLR, NLR, SII, SIRI, and AISI were also positively associated with BC prevalence. In subgroup analyses and interaction testing, we found no significant differences in the associations between these inflammatory markers and BC across various subgroup populations. These findings provide new insights into the role of inflammatory markers in the pathogenesis of BC and suggest the potential value of these markers in the early detection of BC.
Previous studies have reported an association between PLR and BC. A study results shows that patients with persistently high PLR after treatment had poorer prognosis21. Another study in a Japanese population suggested that a high PLR was linked to adverse outcomes in BC and that combining it with tumor-infiltrating lymphocytes (TIL) could more accurately predict outcomes in triple-negative breast cancer patients22. Meta-analysis results indicated that a high PLR might be associated with a lower rate of complete remission in BC patients receiving neoadjuvant chemotherapy23. Another meta-analysis including 39 studies totaling 17,079 BC patients showed that an elevated PLR was linked to poor overall survival and high prevalence of recurrence in breast cancer patients24. Previous studies have mainly concentrated on the correlation between PLR and the prognosis of BC patients. Our study further explored the correlation between PLR and the prevalence of BC. Additionally, we conducted synchronous analysis and comparison of PLR with other inflammatory markers, and finally, we applied ROC analysis to assess the predictive performance of these inflammatory markers for BC, which is the innovation of our study.
Other types of inflammatory markers have also received increasing attention for their potential diagnostic and prognostic value in BC. A retrospective study evaluated the relationship between SII and axillary lymph node metastasis in BC, with results indicating that high levels of SII were a prevalence factor for metastasis25. A study using data from the UK Biobank explored the association between systemic inflammation due to unhealthy lifestyle and BC prevalence, finding that higher levels of C-reactive protein (CRP), SII, and the ratio of CRP to albumin were associated with increased BC prevalence, consistent with our findings26. However, their results indicated a negative correlation between MLR and BC prevalence, contrary to our findings, which may be attributed to differences in study populations and regions, warranting further prospective research to validate our results. Satu et al. investigated the prognostic impact of MLR and NLR on BC and their predictive role in the efficacy of trastuzumab treatment, demonstrating that trastuzumab could overcome the adverse prognostic impact of high MLR and NLR27. Regarding SIRI, a study evaluated its prognostic value in operable BC patients, showing a significant reduction in overall survival in patients with high SIRI28, consistent with our findings, as elevated MLR, NLR, and SIRI were significantly associated with increased BC incidence.
Our main finding is the positive association between PLR and breast cancer, with an odds ratio of 1.35, suggesting that higher PLR values are associated with increased odds of breast cancer. Therefore, individuals with high PLR levels should be vigilant about the potential prevalence of BC. This association between PLR and BC may be related to the functions of platelets and lymphocytes. Platelets can induce cancer cell plasticity through interactions with cancer cells, thereby promoting tumor progression and metastasis, while tumors can regulate platelet function through intra-tumoral platelet aggregation29,30,31,32. Lymphocytes, on the other hand, exert anti-tumor effects by reducing tumor growth through cytotoxicity and inducing cell apoptosis33,34. Therefore, the increase in PLR may contribute to the initiation and progression of tumors in the body.
Our PLR subgroup analysis suggests that alcohol consumption may lead to an increased prevalence of BC. This could be associated with the effects of alcohol on estrogen and its receptors, as well as the production of acetaldehyde or reactive oxygen species. Additionally, alcohol can activate vascular endothelial growth factor to stimulate tumor growth35,36. A study found that the prevalence of breast cancer was higher in the heavy alcohol drinking group compared to the non-drinking group37. Furthermore, through interaction testing, we found no significant association with breast cancer in subgroups stratified by BMI, age, smoking status, alcohol consumption status, and hypertension status.
Inflammation’s role in the onset and progression of tumors has garnered significant attention38. Research indicates that inflammation may serve as a bridge between cancer and its primary risk factors39. These risk factors such as smoking, alcohol consumption, ultraviolet radiation, high blood sugar, etc., ultimately lead to localized or systemic inflammation activation, causing genomic instability in cells, promoting the malignant transformation of normal cells, and inflammation can continue to drive the proliferation of malignant cells40,41,42,43. In cancer metastasis, inflammation also plays a crucial role. Various inflammatory mediators such as IL−6, IL−8, TNF-α are closely associated with epithelial-mesenchymal transition (EMT), and inflammation and EMT interact to form a positive feedback loop44. Studies suggest that the onset and progression of BC may be closely associated with chronic inflammation. The IL−6/STAT3 signaling pathway can upregulate the expression of anti-apoptotic proteins while downregulating the expression of E-cadherin, thereby inhibiting cell apoptosis and promoting cell migration45,46. The PTEN/PI3K/Akt signaling pathway plays a significant role in regulating cell growth, proliferation, apoptosis, etc., so any changes in this pathway’s components can have an impact on tumor induction47,48.
While this study provides important insights, it also has several limitations. First, the association of these inflammatory markers with breast cancer may be driven by reverse causality because the NHANES data have a cross-sectional design and cannot be used to determine causality. The NHANES data may be subject to survival bias, that is, patients with breast cancer who survived longer were more likely to be included in the survey. This may affect the generalisability of the findings, so future studies should adopt a prospective design to reduce the impact of survival bias. In addition, we relied on questionnaire and self-reported data, which may have led to information bias. Despite adjustment for various confounding variables, potential unadjusted confounders may still exist. Future studies should use a prospective cohort design to control for the influence of confounding factors on the association of these inflammatory markers with breast cancer as much as possible, and further verify the predictive value of these inflammatory markers in different populations. In addition, in interaction and subgroup analyses, since multiple testing may lead to false positive risks, more accurate multiple comparisons should be used in subsequent studies to reduce false positives.
Conclusion
In conclusion, systemic inflammatory markers, especially PLR, are significantly associated with breast cancer prevalence. These findings offer new insights and potential biomarkers for early detection and prevention of breast cancer. Future research should further explore the mechanisms of these markers and validate their clinical utility in larger and more diverse populations.
Data availability
Publicly available datasets were analyzed in this study. This data can be found at: https://wwwn.cdc.gov/nchs/nhanes/default.aspx.
References
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74, 229–263 (2024).
Sung, H. et al. Global Cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249 (2021).
Arnold, M. et al. Current and future burden of breast cancer: global statistics for 2020 and 2040. Breast 66, 15–23 (2022).
The benefits. And harms of breast cancer screening: an independent review. Lancet 380, 1778–1786 (2012).
Mantovani, A., Allavena, P., Sica, A. & Balkwill, F. Cancer-related inflammation. Nature 454, 436–444 (2008).
Diakos, C. I., Charles, K. A., McMillan, D. C. & Clarke, S. J. Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 15, e493–503 (2014).
Zhang, D. et al. Scoring system for Tumor-Infiltrating lymphocytes and its prognostic value for gastric Cancer. Front. Immunol. 10, 71 (2019).
Donnem, T. et al. Strategies for clinical implementation of TNM-Immunoscore in resected nonsmall-cell lung cancer. Ann. Oncol. 27, 225–232 (2016).
Salgado, R. et al. The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: recommendations by an international TILs working group 2014. Ann. Oncol. 26, 259–271 (2015).
Sylman, J. L. et al. The predictive value of Inflammation-Related peripheral blood measurements in Cancer staging and prognosis. Front. Oncol. 8, 78 (2018).
Hu, B. et al. Systemic immune-inflammation index predicts prognosis of patients after curative resection for hepatocellular carcinoma. Clin. Cancer Res. 20, 6212–6222 (2014).
Li, X., Zhang, S., Lu, J., Li, C. & Li, N. The prognostic value of systemic immune-inflammation index in surgical esophageal cancer patients: an updated meta-analysis. Front. Surg. 9, 922595 (2022).
Nøst, T. H. et al. Systemic inflammation markers and cancer incidence in the UK biobank. Eur. J. Epidemiol. 36, 841–848 (2021).
Fest, J. et al. The systemic immune-inflammation index is associated with an increased risk of incident cancer-A population-based cohort study. Int. J. Cancer. 146, 692–698 (2020).
Zipf, G. et al. National health and nutrition examination survey: plan and operations, 1999–2010. Vital Health Stat. 1, 1–37 (2013).
Bala, M. M. & Bala, K. A. Bone mineral density and complete blood count ratios in children and adolescents with obesity. Eur. Rev. Med. Pharmacol. Sci. 26, 249–256 (2022).
Zhou, D. et al. Calculated inflammatory markers derived from complete blood count results, along with routine laboratory and clinical data, predict treatment failure of acute peritonitis in chronic peritoneal dialysis patients. Ren. Fail. 45, 2179856 (2023).
Wang, R. H. et al. The clinical value of neutrophil-to-lymphocyte ratio (NLR), systemic immune-inflammation index (SII), platelet-to-lymphocyte ratio (PLR) and systemic inflammation response index (SIRI) for predicting the occurrence and severity of pneumonia in patients with intracerebral hemorrhage. Front. Immunol. 14, 1115031 (2023).
Xiu, J. et al. The aggregate index of systemic inflammation (AISI): a novel predictor for hypertension. Front. Cardiovasc. Med. 10, 1163900 (2023).
Qiu, Z. et al. Associations of serum carotenoids with risk of cardiovascular mortality among individuals with type 2 diabetes: results from NHANES. Diabetes Care. 45, 1453–1461 (2022).
Kim, J. Y. et al. Dynamic changes of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio predicts breast cancer prognosis. BMC Cancer. 20, 1206 (2020).
Onagi, H. et al. High platelet-to-lymphocyte ratios in triple-negative breast cancer associates with immunosuppressive status of TILs. Breast Cancer Res. 24, 67 (2022).
Qi, X. et al. Prognostic significance of platelet-to-lymphocyte ratio (PLR) in patients with breast cancer treated with neoadjuvant chemotherapy: a meta-analysis. BMJ Open. 13, e074874 (2023).
Guo, W. et al. Prognostic value of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio for breast cancer patients: an updated meta-analysis of 17079 individuals. Cancer Med. 8, 4135–4148 (2019).
Tong, L. et al. High levels of SII and PIV are the risk factors of axillary lymph node metastases in breast cancer: A retrospective study. Int. J. Gen. Med. 16, 2211–2218 (2023).
Zhang, Y. et al. Healthy lifestyles, systemic inflammation and breast cancer risk: a mediation analysis. BMC Cancer. 24, 208 (2024).
Tiainen, S., Rilla, K., Hämäläinen, K., Oikari, S. & Auvinen, P. The prognostic and predictive role of the neutrophil-to-lymphocyte ratio and the monocyte-to-lymphocyte ratio in early breast cancer, especially in the HER2 + subtype. Breast Cancer Res. Treat. 185, 63–72 (2021).
Wang, L. et al. Prognostic value of the systemic inflammation response index (SIRI) before and after surgery in operable breast cancer patients. Cancer Biomark. 28, 537–547 (2020).
Braun, A., Anders, H. J., Gudermann, T. & Mammadova-Bach, E. Platelet-Cancer interplay: molecular mechanisms and new therapeutic avenues. Front. Oncol. 11, 665534 (2021).
Stoiber, D. & Assinger, A. Platelet-leukocyte interplay in cancer development and progression. Cells 9 (2020).
Wang, S., Li, Z. & Xu, R. Human cancer and platelet interaction, a potential therapeutic target. Int J. Mol. Sci. 19 (2018).
Gresele, P., Malvestiti, M. & Momi, S. Anti-platelet treatments in cancer: basic and clinical research. Thromb. Res. 164 (Suppl 1), S106–S111 (2018).
Li, A. et al. Activating cGAS-STING pathway for the optimal effect of cancer immunotherapy. J. Hematol. Oncol. 12, 35 (2019).
Nelson, M. A., Ngamcherdtrakul, W., Luoh, S. W. & Yantasee, W. Prognostic and therapeutic role of tumor-infiltrating lymphocyte subtypes in breast cancer. Cancer Metastasis Rev. 40, 519–536 (2021).
Starek-Świechowicz, B., Budziszewska, B. & Starek, A. Alcohol and breast cancer. Pharmacol. Rep. 75, 69–84 (2023).
Olver, I. N. Prevention of breast cancer. Med. J. Aust. 205, 475–479 (2016).
Sánchez-Bayona, R. et al. Binge drinking and risk of breast cancer: Results from the SUN (‘Seguimiento Universidad de Navarra’) project. Nutrients 12 (2020).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Aggarwal, B. B., Vijayalekshmi, R. V. & Sung, B. Targeting inflammatory pathways for prevention and therapy of cancer: short-term friend, long-term foe. Clin. Cancer Res. 15, 425–430 (2009).
Takahashi, H., Ogata, H., Nishigaki, R., Broide, D. H. & Karin, M. Tobacco smoke promotes lung tumorigenesis by triggering IKKbeta- and JNK1-dependent inflammation. Cancer Cell. 17, 89–97 (2010).
Park, E. J. et al. Dietary and genetic obesity promote liver inflammation and tumorigenesis by enhancing IL-6 and TNF expression. Cell 140, 197–208 (2010).
Wroblewski, L. E., Peek, R. M. Jr. & Wilson, K. T. Helicobacter pylori and gastric cancer: factors that modulate disease risk. Clin. Microbiol. Rev. 23, 713–739 (2010).
Read, S. A. & Douglas, M. W. Virus induced inflammation and cancer development. Cancer Lett. 345, 174–181 (2014).
Suarez-Carmona, M., Lesage, J., Cataldo, D. & Gilles, C. EMT and inflammation: inseparable actors of cancer progression. Mol. Oncol. 11, 805–823 (2017).
Knüpfer, H. & Preiss, R. Significance of interleukin-6 (IL-6) in breast cancer (review). Breast Cancer Res. Treat. 102, 129–135 (2007).
Barclay, J. L., Anderson, S. T., Waters, M. J. & Curlewis, J. D. SOCS3 as a tumor suppressor in breast cancer cells, and its regulation by PRL. Int. J. Cancer. 124, 1756–1766 (2009).
Georgescu, M. M. PTEN tumor suppressor network in PI3K-Akt pathway control. Genes Cancer. 1, 1170–1177 (2010).
Bahiraee, A., Ebrahimi, R., Halabian, R., Aghabozorgi, A. S. & Amani, J. The role of inflammation and its related MicroRNAs in breast cancer: A narrative review. J. Cell. Physiol. 234, 19480–19493 (2019).
Funding
The study described was supported by grants from a Natural Science Research Project of Chongqing Three Gorges Medical College (XJ2022003805) and Chongqing Education Commission Science and Technology Research Program Project (KJQN202302715).
Author information
Authors and Affiliations
Contributions
Conceptualization, Si-Qi Zhang, Ran Li and Jia-feng Tang; Data curation, Xiang Li and Miao Miao Liu; Investigation, Miao Miao Liu; Methodology, Si-Qi Zhang and CuiTing Chen; Software, Si-Qi Zhang and CuiTing Chen; Visualization, Si-Qi Zhang and Ran Li; Writing – original draft, Si-Qi Zhang; Writing – review & editing, Jia-feng Tang and Xiang Li.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, S., Li, R., Chen, C. et al. The association between systemic inflammation markers and breast cancer. Sci Rep 16, 9564 (2026). https://doi.org/10.1038/s41598-025-10809-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-10809-1