Abstract
In this study, a series of K2O/Zn-aluminate catalysts with varying Al/Zn ratios (0.5-4) were examined as heterogeneous base catalysts for the esterification of sunflower oil with methanol. The synthesized nanocatalysts were characterized using XRD, FESEM, EDX, and BET techniques. Furthermore, FTIR and H-NMR analyses were conducted to verify the successful transesterification reaction and biodiesel production. The characterization results the successful synthesis of the K2O/Zn-aluminate catalysts, demonstrating their favorable surface and structural properties for the esterification process. The reactor performance data under the conditions of 70 °C reaction temperature, 3 h reaction time, a methanol-to-oil ratio of 1:16, and a catalyst loading of 1% by weight revealed that the catalyst with an Al/Zn molar ratio of 4 exhibited the highest catalytic activity. This catalyst achieved a biodiesel production yield of 96% and a conversion efficiency of 94.6%. This yield is comparable to that of other synthesized samples. The enhanced catalytic performance can be attributed to the improved structural properties of the K2O/Zn-4Al2O4 samples. Furthermore, reusability tests were conducted to evaluate the practical viability of these catalysts. The K2O/Zn-4Al2O4 catalysts demonstrated the capacity to be recycled for four cycles without a substantial decline in catalytic activity, maintaining a yield that decreased from 96 to 90%. This stability highlights their potential as a sustainable alternative for biodiesel production.
Similar content being viewed by others
Introduction
One of the most pressing global challenges today is ensuring a sustainable energy supply, as the depletion of fossil fuel reserves, rising fuel costs, and increasing environmental concerns have driven researchers to seek renewable energy alternatives. It is essential to identify renewable substitutes that are non-toxic, environmentally friendly, and produce lower emissions of aromatic hydrocarbons, carbon monoxide, nitrogen oxides (NOx), and sulfur oxides (SOx)1. In response to these demands, biofuel technology has emerged as a large-scale solution widely adopted by both advanced and developing countries to address energy challenges2. This technology harnesses the energy stored in organic and natural materials, primarily carbon-based, to produce energy and fuel3. Among biofuels, biodiesel stands out as the most significant, functioning similarly to diesel in combustion engines, thus eliminating the need for new engine designs4.Moreover, due to its elevated flash point exceeding 130℃, it exhibits a high ignition value, making it widely recognized as an outstanding alternative energy source5. Biodiesel consists of fatty acid monoalkyl esters derived from renewable natural sources like vegetable oils or animal fats, microalgae, and waste products like grease using a catalyst. There are several methods for producing biodiesel, including pyrolysis, microemulsion, and transesterification. Among these, transesterification is the most effective biodiesel production method6. This process involves a catalyzed chemical reaction between oil and an alcohol, resulting in fatty acid alkyl esters, or biodiesel formation. The transesterification method offers several advantages over other methods. It preserves oxygen atoms in the biodiesel’s chemical structure, unlike the pyrolysis method, which removes them. Additionally, transesterification is cost-effective and has higher conversion efficiency7. Catalysts play a crucial role in chemical reactions, as they increase the reaction rate and facilitate the conversion of reactants into products. This is particularly true for the biodiesel production process, which also relies on catalysts8. The catalysts used in biodiesel production are categorized into homogeneous and heterogeneous types. Heterogeneous catalysts, in particular, offer significant advantages. Their solid form allows for straightforward separation from the reaction mixture, enabling efficient recovery and minimizing waste. This reusability not only streamlines the production process but also supports eco-friendly practices by reducing energy consumption and minimizing environmental impact, and lowers labor and overall production costs9,10,11. Among the different types of catalysts, heterogeneous alkaline catalysts have garnered significant attention due to their catalytic efficiency, reusability, and higher production yields in shorter times. However, heterogeneous catalysts often face challenges related to penetration and accessibility of reactants to the active sites. To overcome this limitation, supports are employed. The support material, which can be porous, provides a high surface area that enhances the dispersion of the active catalytic species. This increased surface area is crucial because it maximizes the exposure of active sites to the reactants, thereby improving the efficiency of the catalytic process12. Additionally, the support material provides structural stability to the catalyst, preventing agglomeration and minimizing the leaching of active components into the reaction mixture13. Numerous studies have been conducted to develop supported catalysts. Fitriana et al.14 utilized K2O/Natural Zeolite as a catalyst in the transesterification of waste frying oil at 65 °C, with an alcohol-to-oil molar ratio of 8:1 and 2.5 wt% catalyst. Natural zeolite was employed to create a heterogeneous catalyst that can be easily separated from the reaction medium. This catalyst demonstrated high efficiency, achieving a 95% yield of methyl ester biodiesel. Additionally, the catalyst was recovered and reused four times, maintaining excellent performance. Kusmiyati et al.15 synthesized an alkaline KOH-impregnated eggshell nanocatalyst for producing biodiesel from Reutealis Trisperma Oil. Calcium oxide, known as one of the best catalysts for biodiesel production, was derived from eggshells, which contain 94 wt% CaCO3, making them a cost-effective natural source. The resulting catalyst exhibited high activity, achieving a 94% biodiesel yield in the transesterification reaction. Narowska et al.16 reported the synthesis of KOH/AC catalysts for the transesterification of corn oil. The incorporation of carbon allowed the catalyst to be reused in the production process and prevented soap formation. In their study, with a methanol-to-oil molar ratio of 3:1, temperature of 62.5 °C, and 0.75 wt% catalyst, a biodiesel yield of 92% was achieved. Helmi et al.17 assessed the potential of a KOH/Al2O3 catalyst, prepared via the impregnation method, for the esterification of Amygdalus scoparia oil to produce biodiesel. Al2O3 was used as a support for the catalyst. The highest biodiesel yield, 94%, was achieved with a methanol-to-oil ratio of 10:1, and a catalyst weight of 1.6 wt/v%, at room temperature over 2.5 h. Kazemifard et al.18 proposed using a KOH/Fe3O4@Al2O3 magnetic nanocatalyst for biodiesel production. The magnetic properties of this catalyst facilitate easy separation from the reaction environment. With its porous structure, the catalyst achieved a 98.8% conversion rate for biodiesel production from canola oil. Additionally, the catalyst maintained over 92% efficiency after being used five times.
Among various supports, spinel aluminate, with the general formula MAl2O4 (where M represents a divalent metal ion such as magnesium, zinc, or copper), is notable for addressing the disadvantages of heterogeneous catalysts. MAl2O4, with its spinel structure, offers high chemical, mechanical, and thermal stability, as well as strong magnetization, making it an effective catalytic support. Recently, ZnAl2O4 spinel has garnered significant attention as a catalyst support in biodiesel production. There are several methods for synthesizing ZnAl2O4 nanostructures, including co-precipitation19, sol-gel20, combustion21, and hydrothermal22. In addition to these, mechanochemical synthesis stands out as one of the simplest advanced methods for producing ZnAl2O4. It involves the release of metal-bound water through mechanical force, followed by precipitation induced by an alkaline agent. This solvent-free method can be conducted at ambient pressure and temperature, offering a short synthesis time and high productivity23. In our previous study24, we supported K2O catalysts on various M-aluminate spinel nanocatalysts (M = Mg, Zn, Cu) prepared via a mechanochemical method and evaluated their catalytic performance in biodiesel production. This study constituted the first reported use of this method to develop a suitable support for esterification reactions. The results indicated that the highest conversion and yield in biodiesel production were achieved using K2O/Zn-aluminate. Building on this work, we designed a K2O-impregnated ZnAl2O4 nanocatalyst to determine the optimum Al/Zn molar ratio for biodiesel production from sunflower oil for the first time.
Materials and methods
Materials
Analytical-grade chemicals from Merck company were used in this study. Sunflower oil was sourced from a local market. Other chemicals employed in the study included Zinc (II) nitrate (Zn (NO3)2.6H2O), Aluminum (III) nitrate (Al (NO3)3.6H2O), Ammonium bicarbonate (NH4HCO3), Potassium hydroxide (KOH), and Methanol (CH3OH).
Synthesis of parent Zn-Al2O4
The mechanochemical method was employed to synthesize Zn-aluminate support. In this approach, nitrate precursors were mixed in varying molar ratios of Al/M = 0.5–4. Ammonium bicarbonate was subsequently added as a precipitation agent, 2 moles for each mole of Zn nanomaterial and 3 moles for each mole of Al nanomaterial, resulting in the formation of metal carbonates through the reactions shown below. All the materials were then ground for 30 min using a mortar and pestle to facilitate the release of functional groups, such as -OH, until the powders gradually formed a slurry.
.
The slurry was then placed in an oven at 100 °C for 24 h to remove the water from its structure. Finally, the materials were calcined at 600 °C for 4 h.
Synthesis of K2O/Zn-Al2O4
To synthesize K2O-impregnated ZnAl2O4, 1 g of potassium hydroxide and 2 g of each support (with different Al/Zn ratios) were dissolved in 50 mL of distilled water and stirred for 2 h at ambient conditions. The mixture was then dried in an oven at 100 °C for 12 h, followed by calcination at 600 °C for 3 h. The prepared catalysts are labeled as K2O/aZn-bAl2O4, where b/a represents the Al/Zn molar ratio of the Zn-aluminate support.”
Characterization methods
The crystallographic structure of the synthesized samples was analyzed using a Bourevestnik XRD (model DRON-8, Russia). The morphology was examined through scanning electron microscopy (FESEM) on a TESCAN-MIRA3 (Czech Republic). The elemental distribution was examined through the utilization of energy-dispersive Xray (EDX) analysis. The surface area and pore size distribution of the samples were measured using nitrogen adsorption/desorption in the relative pressure range of 0.05 to 0.99 with a Belsorp mini II instrument. The structure of biodiesel, including proton-containing compounds, was verified by Proton Nuclear Magnetic Resonance (H-NMR, VARIAN model INOVA 500 MHz). In addition, Fourier transform infrared spectroscopy (FTIR, PerkinElmer IR spectrophotometer version 10.03.06, USA) was used to verify and identify the functional groups of produced biodiesels.
Activity tests
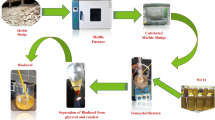
The transesterification reactions were performed in a flask placed in a water bath and connected to a condenser. Initially, a mixture of methanol and catalyst was introduced into the reaction vessel and heated at 70 °C for 30 min, followed by the addition of sunflower oil. The fatty acid composition of sunflower oil is presented in Table 1. The transesterification process was carried out under specific conditions: a methanol-to-oil molar ratio of 16:1, 1 wt% catalyst, a reaction temperature of 70 °C, and a reaction time of 3 h. Upon completion, the resulting mixture was transferred to a decanter, where two distinct phases quickly formed. Due to glycerol’s higher density relative to methyl ester, the upper phase contained methyl ester (biodiesel), while the lower phase comprised glycerol and catalyst. The glycerol was separated from the biodiesel phase, which was then washed multiple times with hot water to remove impurities, resulting in clear-colored biodiesel. Each test was conducted in triplicate, and the results were averaged. After each reaction, the catalyst was recovered from the lower phase of the decanter, washed several times with methanol and water, and centrifuged. The cleaned catalyst was dried in an oven at 80 °C and reused for subsequent esterification reactions in additional cycles.
Results and discussion
Nanocatalysts characterization
XRD analysis
The XRD patterns of the synthesized catalysts with varying Al/Zn ratios are presented in Fig. 1. The characteristic peaks at 2θ values of 31.88°, 36.40°, 56.69°, and 68° correspond to the (220), (311), (511), and (440) planes of Zn-Al₂O₄ spinel metal oxides25. Additional peaks observed at 2θ = 32.87°, 34.55°, 47.69°, and 64.02° are attributed to ZnO, which is predominant at low Al/Zn ratios due to the excess availability of Zn metal. This excess Zn, combined with its limited solubility in the Zn-Al₂O₄ lattice, results in the formation of ZnO phases. Furthermore, as the Al/Zn molar ratio increases, the intensity of the diffraction peaks diminishes, indicating reduced crystallinity. This behavior can be attributed to lattice distortions caused by the difference in ionic radii between Zn²⁺ and Al³⁺, which disrupt the crystal structure.
XRD patterns of (a): K2O/2Zn-Al2O4, (b): K2O/Zn-Al2O4, (c): K2O/Zn-2Al2O4, and (d): K2O/Zn-4Al2O4.
FESEM analysis
The FESEM images of the synthesized K₂O/Zn-Al₂O₄ with varying Al/Zn ratios are shown in Fig. 2. As noted in our previous study24 and corroborated by some research, our synthesized Zn-aluminate exhibits a rod-like shape26 with all images showing spherical K₂O nanoparticles on the Zn-aluminate rods. As the Al/Zn ratio increases, smaller particles are formed, likely due to the difference in ionic radii between Al³⁺ (approximately 0.53 Å) and Zn²⁺ (approximately 0.74 Å). The incorporation of more Al³⁺ ions into the Zn-Al₂O₄ lattice results in a more compact and tightly packed structure, attributed to the smaller ionic radius of Al³⁺. This structural compactness restricts the growth of individual particles, leading to smaller particle sizes. Furthermore, higher Al/Zn ratios contribute to increased surface roughness due to the formation of less crystalline phases, as corroborated by the XRD results.
FESEM images of (a): K2O/2Zn-Al2O4, (b): K2O/Zn-Al2O4, (c): K2O/Zn-2Al2O4, and (d): K2O/Zn-4Al2O4.
N2 Adsorption-desorption analysis
The nitrogen adsorption/desorption analysis results for K₂O/Zn-Al₂O₄ with varying Al/Zn ratios are summarized in Table 2. The data reveal that increasing the Al/Zn molar ratio leads to a rise in both surface area and pore volume. Specifically, at an Al/Zn ratio of 4, the K₂O/Zn-4Al₂O₄ nanocatalyst exhibited the most favorable pore characteristics, with a surface area of 9.38 m²/g and a pore volume of 0.0335 m³/g. This improvement in pore properties is likely due to the optimal arrangement and distribution of aluminum and zinc within the catalyst structure, enabling greater accessibility to active sites and enhancing reactivity. Moreover, the increased surface area facilitates improved interactions with reactants, contributing to superior catalytic performance27.
Figure 3 presents the nitrogen adsorption/desorption isotherms for the samples under investigation. All synthesized samples exhibit Type III isotherms, characteristic of materials with a broad pore size distribution. The limited surface area and low pore volume of the samples contribute to a predominance of macroporous structures. As a result, this leads to either the absence of hysteresis or, for the first three samples, only minimal changes in adsorption capacity with increasing pressure. Variations in pore size, particularly within the macropore range, are further supported by the BJH pore size distribution analysis presented in the inset of Fig. 3.
Nitrogen adsorption − desorption isotherm and BJH pore size distribution curves (internal) of samples.
Catalytic tests
To verify that the upper phase separated in the decanter, during experimental tests, is biodiesel, FTIR analysis was performed. Figure 4 displays the FTIR spectra of biodiesels produced using K₂O/Zn-Al₂O₄ with varying Al/Zn ratios. The peak at 730 cm⁻¹ indicates the presence of the aromatic alkyne group -CH₂28. The peaks at 2912 and 2834 cm⁻¹ correspond to the symmetric and antisymmetric stretching vibrations of C-H in CH₂ and CH₃ groups, respectively29, while the bending vibrations are observed at 1437 cm⁻¹ and 1134 cm⁻¹, respectively30. The prominent peak at 1746 cm⁻¹ confirms the presence of C = O stretching vibrations characteristic of carbonyl groups in esters. Additionally, the absence of a peak at 3200 cm⁻¹, which corresponds to the OH group, indicates that the biodiesel is free from diglycerides, unreacted glycerin, and methanol31. Therefore, these spectra confirm the formation of biodiesel by displaying functional groups characteristic of biodiesel across all these catalysts.
FTIR spectra of produced biodiesel using K₂O/Zn-Al₂O₄ with varying Al/Zn ratios.
The biodiesel production yield was determined using the following equation:
Figure 5a illustrates the biodiesel yield over K₂O/Zn-Al₂O₄ with varying Al/Zn ratios in the esterification process. Catalytic tests were performed three times, and the average results were reported. It is evident that increasing the Al/Zn ratio initially decreases the biodiesel yield. However, for the K₂O/Zn-4Al₂O₄ sample, the biodiesel yield significantly increases, achieving an impressive yield of 96%, surpassing all other supported catalysts. This enhanced activity can be attributed to the sample’s superior pore characteristics and substantially increased surface area, which provide better accessibility to active sites. Furthermore, the synergistic interactions between Al and Zn at this specific ratio may enhance key phenomena, such as electron transfer or the formation of unique reaction intermediates, thereby facilitating a more efficient transesterification process. Another contributing factor could be that, at an Al/Zn ratio of 4, the catalyst achieves an optimal balance of basic and acidic properties, which enhances its catalytic performance and leads to the observed higher yield.
Biodiesel yield (a), H-NMR spectra of produced biodiesel over K2O/Zn-4Al2O4 (b).
Furthermore, to calculate the conversion of triglycerides to methyl esters, nuclear magnetic resonance (H-NMR) spectroscopy data were employed, with Eq. 4 used for analysis.
To clarify what A1 and A2 represent, the H-NMR analysis results of the produced biodiesel and its spectra are first discussed. As shown in Fig. 5b, the absorption peak of the proton attached to the methyl ester, observed in the range of 3.6–3.7 ppm32 serves as the primary standard peak for verifying biodiesel. The second dominant peak, attributed to hydrogen atoms in methylene (-CH₂-) groups of hydrocarbons, appears within the range of 2.2–2.3 ppm33. Additionally, the peak in the range of 5.3–5.37 ppm corresponds to the hydrogens of the saturated alkene esters group. After confirming the successful formation of biodiesel through H-NMR analysis, the A1 and A2 parameters in the conversion relation can now be introduced. A1 and A2 represent the integrals of the signals related to the H-NMR spectrum of the proton in methyl ester and methylene, respectively. Thus, based on Fig. 5b and Eq. 4, the K₂O/Zn-4Al₂O₄ catalyst, identified as the optimum sample, achieved a 94.6% conversion of triglycerides to methyl esters.
Quality analysis of biodiesel
To assess the quality of the biodiesel produced, key properties such as density and viscosity were evaluated against established standards. The kinematic viscosity of the biodiesel was measured at 4.8 mm²/s at 38 °C, which aligns with the ASTM D4704 standard that specifies a range of 1.9 to 6 mm²/s at 40 °C. Furthermore, the density of the biodiesel, determined using a pycnometer, was found to be 0.87 g/cm³, falling within the acceptable range of 0.86 to 0.9 g/cm³ as per ASTM D1480. These findings confirm that the properties of the produced biodiesel meet the required standards.
Esterification mechanism catalyzed by K2O/Zn-aluminate
The mechanistic pathway of the transesterification reaction facilitated by K2O/Zn-aluminate catalyst is illustrated in Fig. 6. This process commences with the adsorption of methanol onto the catalyst surface, which enables the generation of methoxide ions (CH3O−) that function as nucleophiles. Concurrently, the catalyst undergoes protonation due to ion exchange, forming a protonated species. In the next phase of the reaction, the nucleophilic methoxide ions (CH3O−) initiate a nucleophilic attack on the electrophilic carbonyl carbon of the triglyceride molecules, leading to the formation of a tetrahedral intermediate. This intermediate subsequently undergoes a rearrangement process, culminating in the production of fatty acid esters and diglyceride anions. The final step involves the protonation of these diglyceride anions by the protonated catalyst. Following this, the catalyst is regenerated and prepared to commence another cycle by interacting with methanol, thus generating a blend of fatty acid alkyl esters and glycerol. This ongoing catalytic cycle ensures a highly efficient synthesis of biodiesel34.
Esterification mechanism catalyzed by K2O/Zn-aluminate.
Deactivation study
Reusability of nanocatalysts
The study investigated the reusability of the K₂O/Zn-4Al₂O₄ catalyst in the transesterification process under the same conditions as the initial test: methanol to oil ratio of 1:16, reaction time of 3 h, temperature of 70 °C, and a catalyst amount of 1 wt%. The results shown in Fig. 7 indicate that the K₂O/Zn-4Al₂O₄ catalyst maintained an acceptable yield up to the fourth reuse without a significant drop in biodiesel yield.
Common factors contributing to catalyst deactivation encompass the deposition of byproducts or secondary products on the catalyst surface, which blocks active sites and impedes reaction progress35. Additionally, leaching of active materials during recycling and washing processes can lead to a loss of catalytic components, further reducing performance36. Aggregation or sintering of the metal active phase also diminishes the accessible surface area, thereby lowering activity37. Other contributing factors include structural degradation of the catalyst framework and mechanical stresses like crushing, all of which collectively compromise catalyst stability and efficiency.
Reusability of K2O/Zn-4Al2O4 sample for four esterification reaction cycles.
Spent nanocatalysts characterizations
To examine the factors contributing to the slight decrease in catalyst activity observed in this study, XRD, FESEM, and EDAX analyses were conducted on the used samples. The XRD results for both fresh and used K2O/Zn-4Al2O4 catalysts, as presented in Fig. 8, indicate a significant alteration in the crystalline structure of the catalyst after four cycles of reuse. This transformation is attributed to the degradation of the catalyst’s surface and structural properties, likely resulting from the cumulative effects of repeated exposure to reaction conditions and the potential leaching of active components.
XRD analysis of K2O/Zn-4Al2O4 catalyst (a): fresh, and (b): after 4 cycles.
Figure 9 compares the FESEM images of the fresh and spent K₂O/Zn-4Al₂O₄ catalysts. The structure of the spent sample is completely different from the fresh one. The spent K₂O/Zn-4Al₂O₄ catalyst exhibits an agglomerated structure, in contrast to the rod-shaped structure of the fresh sample. This indicates that the K₂O/Zn-4Al₂O₄ structures degraded during the regeneration and reuse processes.
FESEM analysis of K2O/Zn-4Al2O4 catalyst (a): fresh, and (b): after 4 cycles.
Figure 10 presents the EDX spectra of both the fresh and spent K2O/Zn-4Al2O4 catalysts. As evident from the figure, the peak intensities corresponding to Zn, O, and K decreased, while that of Al increased after four cycles of use. This alteration is likely attributed to the washing of the catalyst and potential leaching of the active phase.
EDX analysis of K2O/Zn-4Al2O4 catalyst (a): fresh, and (b): after 4 cycles.
Comparison with the published studies
Table 3 compares the activity of various spinel aluminate-based catalysts in the esterification process, highlighting the promising catalytic efficiency and reusability of K₂O/Zn-4Al₂O₄ catalysts as heterogeneous base catalysts for biodiesel production and other value-added products.
Future prospects
As biodiesel technology remains in its early stages of development, achieving practical utilization requires addressing several critical aspects in future studies. Subsequently, we discuss a number of these key elements:
-
Catalyst Synthesis: Efficient separation of catalysts from reaction mixtures is a critical factor influencing both catalytic performance and operational costs. The integration of magnetite or analogous materials into catalyst design facilitates recovery processes, thereby reducing costs and promoting sustainability. Additionally, the leaching of active components during reaction cycles poses a significant challenge to maintaining catalyst efficiency. To address this, advanced synthesis methods, such as encapsulation or the use of more stable support materials, are necessary to ensure the structural integrity and activity of catalysts over multiple cycles.
-
Catalytic Test: Mass transfer limitations in biodiesel production are critical factors that can significantly impact reaction efficiency, biodiesel yield, and process scalability. Optimizing mixing process through advanced technologies like static mixers, ultrasonic cavitation, or enhanced reactor designs can significantly improve mass transfer between reactants, thereby increasing catalytic efficiency.
-
Scale-up Potential: Conducting comprehensive scale-up studies is crucial for evaluating the economic viability and feasibility of biodiesel production at an industrial scale. Such analyses can identify challenges associated with transitioning from laboratory-scale processes to commercial applications.
These considerations aim to refine biodiesel production processes, ensuring improved efficiency, sustainability, and scalability while aligning with green chemistry principles.
Conclusion
The study evaluated the catalytic efficiency of a series of K2O/Zn-aluminate catalysts with different Al/Zn ratios by examining their ability to facilitate the esterification reaction, a key step in biodiesel production. The catalysts were prepared using the mechanochemical-wet impregnation method. The esterification of sunflower oil was conducted at a reaction temperature of 70 °C, with a reaction time of 3 h, methanol-to-oil ratio of 1:16, and a catalyst amount of 1 wt%. The activity tests showed that the K2O/Zn-4Al2O4 catalyst exhibited the highest performance, converting over 94.6% of sunflower oil into biodiesel, with an overall biodiesel yield reaching 96%. Importantly, the K2O/Zn-4Al2O4 nanocatalyst demonstrated excellent stability, allowing for 4 times reusing with only a slight reduction in conversion efficiency. Finally, this study identified key considerations that should be addressed in future research to enable practical implementation.
Data availability
The datasets used and analyzed during the current study are available from the corresponding author uponreasonable request.
References
Buasri, A. et al. Green synthesis of metal oxides (CaO-K2O) catalyst using golden Apple snail shell and cultivated banana Peel for production of biofuel from non-edible Jatropha Curcas oil (JCO) via a central composite design (CCD). J. Saudi Chem. Soc. 28, 101836 (2024).
Abdelrahman, A. A. & Abo El-Khair, M. A. Advanced biodiesel production: feedstocks, technologies, catalysts, challenges, and environmental impacts. J. Environ. Chem. Eng. 13, 114966 (2025).
Nath, S. Biotechnology and biofuels: paving the way towards a sustainable and equitable energy for the future. Discover Energy. 4, 8 (2024).
Ge, J. C., Yoon, S. K. & Choi, N. J. Using Canola Oil Biodiesel as an Alternative Fuel in Diesel Engines: A Review. Applied Sciences vol. 7 Preprint at (2017). https://doi.org/10.3390/app7090881
Loryuenyong, V., Rohing, S., Singhanam, P., Kamkang, H. & Buasri, A. Artificial Neural Network and Response Surface Methodology for Predicting and Maximizing Biodiesel Production from Waste Oil with KI/CaO/Al2O3 Catalyst in a Fixed Bed Reactor. Chempluschem 89, e202400117 (2024).
Vilas Bôas, R. N. & Mendes, M. F. A review of biodiesel production from non-edible Raw materials using the transesterification process with a focus on influence of feedstock composition and free fatty acids. J. Chil. Chem. Soc. 67, 5433–5444 (2022).
Mandari, V. & Devarai, S. K. Biodiesel production using homogeneous, heterogeneous, and enzyme catalysts via transesterification and esterification reactions: a critical review. Bioenergy Res. 15, 935–961 (2022).
Alismaeel, Z. T., Al-Jadir, T. M., Albayati, T. M., Abbas, A. S. & Doyle, A. M. Modification of FAU zeolite as an active heterogeneous catalyst for biodiesel production and theoretical considerations for kinetic modeling. Adv. Powder Technol. 33, 103646 (2022).
Thangaraj, B., Solomon, P. R., Muniyandi, B., Ranganathan, S. & Lin, L. Catalysis in biodiesel production—a review. Clean. Energy. 3, 2–23 (2019).
Faruque, M. O., Razzak, S. A. & Hossain, M. M. Application of Heterogeneous Catalysts for Biodiesel Production from Microalgal Oil—A Review. Catalysts vol. 10 Preprint at (2020). https://doi.org/10.3390/catal10091025
Ruhul, A. M. et al. State of the Art of biodiesel production processes: a review of the heterogeneous catalyst. RSC Adv. 5, 101023–101044 (2015).
Lakhani, P., Bhanderi, D. & Modi, C. K. Support materials impact on green synthesis and sustainable processing via heterogeneous catalysis. Discover Catal. 1, 2 (2024).
Ahmad, H. & Hossain, M. K. Supported nanocatalysts: recent developments in microwave synthesis for application in heterogeneous catalysis. Mater. Adv. 3, 859–887 (2022).
Fitriana, N. et al. Synthesis of K2O/Zeolite catalysts by KOH impregnation for biodiesel production from waste frying oil. IOP Conf. Ser. Mater. Sci. Eng. 334, 12011 (2018).
Kusmiyati, K. et al. Biodiesel Production from Reutealis Trisperma Oil Using KOH Impregnated Eggshell as a Heterogeneous Catalyst. Energies vol. 12 Preprint at (2019). https://doi.org/10.3390/en12193714
Narowska, B., Kułażyński, M., Łukaszewicz, M. & Burchacka, E. Use of activated carbons as catalyst supports for biodiesel production. Renew. Energy. 135, 176–185 (2019).
Helmi, M., Hemmati, A. & Tahvildari, K. Biodiesel production from amygdalus scoparia using KOH/Al2O3 catalyst: optimization by response surface methodology. Iranica J. Energy Environ. 12, 34–44 (2021).
Kazemifard, S., Nayebzadeh, H., Saghatoleslami, N. & Safakish, E. Assessment the activity of magnetic KOH/Fe3O4@Al2O3 core–shell nanocatalyst in transesterification reaction: effect of fe/al ratio on structural and performance. Environ. Sci. Pollut. Res. 25, 32811–32821 (2018).
Mekprasart, W., Worasawat, S., Tangcharoen, T. & Pecharapa, W. Characterization and effect of calcination temperature on structural properties of spinel zinc aluminate synthesized via Co-precipitation process. Phys. Status Solidi C. 12, 624–627 (2015).
Belyaev, A. V. et al. Sol-gel synthesis and characterization of ZnAl2O4 powders for transparent ceramics. Ceram. Int. 45, 4835–4839 (2019).
Mirbagheri, S. A., Masoudpanah, S. M. & Alamolhoda, S. Structural and optical properties of ZnAl2O4 powders synthesized by solution combustion method: effects of mixture of fuels. Optik (Stuttg). 204, 164170 (2020).
Song, L., Liu, G. & Qu, Z. Cation rearrangement at tetrahedral sites in the Cu/ZnAl2O4 spinel enhancing CO2 hydrogenation to methanol. Appl. Catal. B: Environ. Energy. 362, 124742 (2025).
Sakhaei, Z. & Rezaei, M. Mechanochemical synthesis of ZnO.Al2O3 powders with various zn/al molar ratios and their applications in reverse water-gas shift reaction. Environ. Sci. Pollut. Res. 28, 13790–13799 (2021).
Mirshafiee, F. & Rezaei, M. Catalytic efficiency and reusability of K2O/M-aluminate (M = Mg, zn, Cu) nanocatalyst in the esterification of sunflower oil with methanol for biodiesel production. J. Mol. Liq. 426, 127333 (2025).
Macedo, H. et al. Characterization of ZnAl2O4 spinel obtained by hydrothermal and microwave assisted combustion method: A comparative study. Mat. Res. 20, (2017).
Cheng, B., Ouyang, Z., Tian, B., Xiao, Y. & Lei, S. Porous ZnAl2O4 spinel nanorods: high sensitivity humidity sensors. Ceram. Int. 39, 7379–7386 (2013).
Emon, S. H., Hossain, M. I., Khanam, M. & Yi, D. K. Expanding horizons: Taking advantage of graphene’s surface area for advanced applications. App. Sci. 15(8), 4145 (2025).
Pasawan, M. et al. Ultrasonication assisted catalytic transesterification of ceiba pentandra (kapok) oil derived biodiesel using immobilized iron nanoparticles. Fuels 3, 113–131 (2022).
Ali, C. H. et al. Improved transesterification of waste cooking oil into biodiesel using calcined goat bone as a catalyst. Energy Sour. Part A Recover. Utilization Environ. Eff. 1–8 https://doi.org/10.1080/15567036.2018.1469691 (2018).
Devaraj, K. et al. Feasibility of biodiesel production from waste cooking oil: lab-scale to pilot-scale analysis. Environ. Sci. Pollut. Res. 27, 25828–25835 (2020).
Rafati, A., Tahvildari, K. & Nozari, M. Production of biodiesel by electrolysis method from waste cooking oil using heterogeneous MgO-NaOH nano catalyst. Energy Sour. Part A Recover. Utilization Environ. Eff. 41, 1062–1074 (2019).
Kalu-Uka, G. M. et al. Prospects for biodiesel production from macrotermes nigeriensis: process optimization and characterization of biodiesel properties. Biomass Bioenergy. 146, 105980 (2021).
Nejad, A. S. & Zahedi, A. R. Optimization of biodiesel production as a clean fuel for thermal power plants using renewable energy source. Renew. Energy. 119, 365–374 (2018).
Yadav, M. & Sharma, Y. C. Transesterification of used vegetable oil using BaAl2O4 spinel as heterogeneous base catalyst. Energy Convers. Manag. 198, 111795 (2019).
Mirshafiee, F. & Rezaei, M. Bifunctional CoNi/Fe3O4@GO catalyst for hydrogen generation through NaBH4 in different solvolytic environments: the effect of sequential metal introduction. Int. J. Hydrogen Energy. 68, 1108–1118 (2024).
Mirshafiee, F. & Rezaei, M. Enhancing hydrogen generation from sodium borohydride hydrolysis and the role of a Co/CuFe2O4 nanocatalyst in a continuous flow system. Sci. Rep. 14, 9659 (2024).
Mirshafiee, F. & Rezaei, M. Engineering of the ferrite-based support for enhanced performance of supported pt, pd, ru, and Rh catalysts in hydrogen generation from NaBH4 hydrolysis. Sci. Rep. 14, 20818 (2024).
Nayebzadeh, H., Ajamein, H., Zakizadeh, T. & Rahmanivahid, B. Preparation of mixed spinel catalyst support (CaxMg1-xAl2O4) reinforced by calcium oxide toward in the biodiesel production from vegetable oil. Int. J. Green. Energy. 21, 745–756 (2024).
Heydari, H., Nayebzadeh, H. & Shahraki, M. Effect of fuel molecules on properties and activity of koh/calcium aluminate nanocatalyst for biodiesel production. Int. J. Environ. Sci. Technol. 15, 1719–1730 (2018).
Liu, Q. et al. The effect of lanthanum doping on activity of Zn-Al spinel for transesterification. Appl. Catal. B. 136–137, 210–217 (2013).
Nayebzadeh, H., Saghatoleslami, N., Haghighi, M. & Tabasizadeh, M. Catalytic activity of KOH–CaO–Al2O3 nanocomposites in biodiesel production: impact of Preparation method. Int. J. Self Propag. High Temp. Synth. 28, 18–27 (2019).
Author information
Authors and Affiliations
Contributions
Faezeh Mirshafiee: Conceptualization, Formal analysis, Investigation, Methodology, Writing – original draft. Mehran Rezaei: Conceptualization, Project administration, Funding, Supervision, Writing –review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Mirshafiee, F., Rezaei, M. Optimizing Al/Zn ratios in K2O/Zn-Aluminate catalysts for enhanced biodiesel production efficiency. Sci Rep 15, 25293 (2025). https://doi.org/10.1038/s41598-025-11268-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-11268-4
Keywords
This article is cited by
-
Eco-Friendly Biodiesel Synthesis via Microbubble-Aided Transesterification and Leaf-Derived Green Carbon Catalyst
Korean Journal of Chemical Engineering (2026)
-
Facile fabrication of heteropolyacid/bimetallic Bi/Ce-MOFs hybrid catalyst for sustainable biodiesel synthesis
BMC Chemistry (2025)