Abstract
Previous studies have described the association between calf circumference (CC) and chronic kidney disease (CKD). We aim to evaluate the associations between CC and cardiovascular and all-cause mortality in patients with CKD stages 3–5. Data on CKD were sourced from the National Health and Nutritional Examination Survey (NHANES) 1999–2004. The population was stratified into three groups based on their CC tertile. Kaplan-Meier method with log-rank tests for significance was used for survival analysis. Weighted Cox proportional hazards regression models were employed to estimate the hazard ratios (HRs) for cardiovascular and all-cause mortality. The potential nonlinear relationship between CC and mortality was assessed using restricted cubic spline (RCS) models. Subgroup and sensitivity analyses were conducted to strengthen the results. A total of 1166 patients were eventually included in this study. After a mean follow-up of 127.78 months, a total of 922 all-cause deaths were recorded, with 515 of them attributed to cardiovascular diseases. The Kaplan-Meier curve indicated a significant difference in overall survival between the three groups (log-rank test, P < 0.0001). Compared to the CC > 38.5 group, participants in the CC < 35.0 group had HR of 2.05 (1.44, 2.93) for all-cause mortality and 1.58 (0.75, 3.33) for cardiovascular mortality, respectively. We observed a significant nonlinear relationship between CC and cardiovascular and all-cause mortality (P-nonlinear < 0.05). Subgroup analysis further validated our results and demonstrated that the impact of CC on prognosis varies according to distinct characteristics. Sensitivity analyses yielded similar results for both all-cause and cardiovascular mortality. A reduced CC is correlated with a poorer prognosis in CKD stages 3–5 patients, suggesting its potential utility as an innovative prognostic marker.
Similar content being viewed by others
Introduction
Chronic Kidney Disease (CKD) stands as a significant health issue globally, exerting a notable impact on morbidity and mortality. The Global Burden of Disease study reveals that CKD accounted for 1.2 million deaths in 20171. CKD not only leads to disability and shortened life expectancy but also emerges as a crucial risk factor for cardiovascular diseases, necessitating prompt attention from the healthcare community2. CKD patients, especially those with end stage kidney disease (ESKD), are prone to Protein-Energy Wasting (PEW) and muscle wasting. This is primarily due to metabolic alterations in CKD patients, such as metabolic acidosis, secondary hyperparathyroidism, vitamin D deficiency, chronic low-grade inflammation, and anemia, which lead to increased protein degradation, decreased protein synthesis, or both3,4. Previous studies found that low muscle mass is a predictive factor for muscle weakness, decreased quality of life, and increased risks of hospitalization and mortality among patients with CKD, especially those undergoing dialysis5,6. Therefore, assessing and managing muscle quality may help control the adverse consequences of CKD in clinical practice.
The muscle quality of patients is often assessed through anthropometric measurements such as calf circumference (CC), arm circumference, and thigh circumference7. Compared to imaging techniques like Computed Tomography (CT), Magnetic Resonance Imaging (MRI), and Ultrasound (US), anthropometric measurements are highly suitable for clinical use due to their portability, relative affordability, and ease of execution8,9. CC measurements are considered a simple and inexpensive method for examining muscle mass and have been used in research as a simplified means of assessing the presence of sarcopenia10. A study found that CC has a superior ability to identify sarcopenia in hemodialysis (HD) patients compared to the modified creatinine index and standardized assessment tools such as the SARC-F score and SARC-CalF score11. Another study found that, among participants aged 75 and older, the strongest association with the estimated glomerular filtration rate (eGFR) and the highest likelihood of CKD were identified using CC measurements12. These studies suggest that CC may be a potential marker of CKD in addition to being a surrogate for nutritional status.
Previous studies have reported that a decrease in CC may be associated with higher mortality. Costa Pereira et al.13 indicated that BMI-adjusted CC was inversely associated with mortality serving as a significant predictor of mortality in older patients with high BMI. A study involving 19,735 participants revealed that both all-cause and cardiovascular mortality rates were inversely correlated with CC measurements in the general population14. Rodrigues et al.15found that CC predicts falls in older adults on HD. Ozawa et al.16 demonstrated that shorter CC is associated with an increased risk of osteoporosis and lower bone mineral density (BMD) in HD patients, which elevates the risks of fractures and mortality. However, a recent study found that the CC trajectory was not related to all-cause death and the number of all-cause hospitalizations among HD patients17. Furthermore, studies examining the correlation between CC and kidney disease prognosis have predominantly concentrated on dialysis patients15,16, with the relationship between CC and the prognosis of general CKD patients still remaining unknown. Thus, we used data from the 1999–2004 National Health and Nutrition Examination Survey (NHANES) to determine the association between CC and all-cause and cardiovascular mortality in CKD.
Methods
Data source and study population
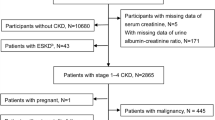
The data for the study were sourced from NHANES, a continuous cross-sectional research initiative in the United States. Employing a stratified, multistage probability sampling methodology, NHANES assesses the health and nutritional profiles of the U.S. population. This comprehensive dataset encompasses demographic characteristics, dietary conditions, clinical examination, laboratory analyses, and participant questionnaires, all of which are subsequently released to the public. Approval for the research protocols was granted by the Ethics Review Board of the National Center for Health Statistics, and all participants gave their written informed consent. Utilizing consistent CC measurements from examination data conducted between 1999 and 2004, we established an observational cohort of CKD patients. Participants under the age of 18 and those with missing data on CC, serum creatinine levels, or survival status were excluded from the analysis. The included participants were subsequently categorized into three distinct groups based on the tertiles of CC. A comprehensive illustration of the participant selection procedure is presented in Fig. 1.
Flow chart of the study population selection. eGFR, estimated glomerular filtration rate.
Definition of CKD
The eGFR was determined utilizing the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation18, which is predicated on baseline serum creatinine concentrations. CKD stages 3–5 was diagnosed when eGFR values were less than 60 mL/min/1.73 m².
Measurement of CC
All examinees underwent body measurements conducted by a trained examiner at the mobile examination center (MEC). CC was measured using the standard methodology, which involved sliding the tape measure up and down the calf to determine the widest point and then recording the value.
Mortality ascertainment and definition
This study assessed all-cause and cardiovascular mortality as primary outcomes. Mortality data, collected up to December 31, 2019, were sourced from the NHANES Public-Use Linked Mortality File, which is integrated with the National Death Index (NDI). The underlying causes of death were determined using the International Statistical Classification of Diseases and Related Health Problems, 10th Revision (ICD-10). Cardiovascular mortality was defined as deaths resulting from heart diseases (codes I00-I09, I11, I13, I20-I51) and cerebrovascular diseases (codes I60-I69), in accordance with the ICD-10 classification. The duration of follow-up for each participant was measured from the date of their examination at the NHANES MEC to the last known date they were alive or censored from the mortality file.
Covariates assessment
The research included a variety of factors such as demographic characteristics, physical examination results, and comorbidity status. Ethnicity was divided into Mexican American, non-Hispanic Black, non-Hispanic White, and other Hispanic groups. Family income relative to the poverty line was segmented into three levels: ≤1.30, 1.30 to 3.50, and > 3.50, with higher ratios reflecting improved economic standing. Smoking background was categorized based on self-reported consumption of at least 100 cigarettes in a lifetime and current status into nonsmokers, former smokers, and current smokers. Alcohol consumption was classified into abstainers, light or moderate alcohol consumption drinkers (up to one drink per day for women, 1–2 for men), and heavy drinkers (more than two drinks per day for women, more than three for men). Educational level was grouped into less than high school, completed high school, or college education or higher. BMI was computed from weight in kilograms over height squared in meters and grouped into < 25.0, 25.0 to 30, and ≥ 30.0 kg/m². Hypertension was defined according to the 2017 American College of Cardiology guidelines, which include the use of baseline antihypertensive medication, a systolic blood pressure of ≥ 130 mmHg, and/or a diastolic blood pressure of ≥ 80 mmHg. Dyslipidemia was diagnosed based on a physician’s assessment, the use of hypolipidemic medication, or biochemical indicators such as triglycerides ≥ 150 mg/dL or HDL cholesterol < 40 mg/dL, in accordance with the National Cholesterol Education Program Adult Treatment Panel III criteria. Diabetes mellitus was identified through medical history or the presence of elevated HbA1c levels (≥ 6.5%) or fasting blood glucose levels (≥ 126 mg/dL).
Statistical analysis
The NHANES sample design allows for the integration of multiple survey cycles, thereby enhancing the sample sizes and expanding the range of analytical options. Following the Analytic Guidelines of the National Center for Health Statistics (https://www.cdc.gov/nchs/nhanes/analyticguidelines.htm), we used weights of 2/3* NHANES examination weights for the 4-year survey cycle (WTMEC4YR) for 1999–2002 and 1/3*WTMEC2YR for 2003–2004 to combine three cycles of data, ensuring that our findings accurately reflected the broader U.S. population. We computed descriptive statistics within each group, relying on mean (SD) for continuous data and proportions for categorical data. Survival trends were explored using the Kaplan-Meier method, with significance determined via log-rank tests. Log-log plots and Schoenfeld residual test were used to verify the proportional hazards assumption. Weighted multivariable Cox proportional hazards regression models were employed to assess the hazard ratios (HRs) for both all-cause and cardiovascular mortality, taking into account potential confounding factors. The analysis incorporated four distinct adjustment models. Model 1 served as the primary model without any adjustments, while Model 2 adjusted for age, sex, and race. Model 3 extended the adjustments of Model 2 to include alcohol intake, smoking status, BMI, education level, and economic situation. Model 4 further adjusted for comorbidity conditions, in addition to the variables in Model 3. To detect potential non-linear relationships between the CC and both all-cause and cardiovascular mortality, restricted cubic spline (RCS) fitting curves with 4 knots were applied to the adjusted Cox proportional hazards models. Furthermore, we carried out subgroup analyses to explore potential modifiers and confirm the findings. Due to significant sex differences in CC, sensitivity analyses were conducted using sex-specific tertiles. All statistical analyses were performed using R software version 4.4.1 (https://www.r-project.org/), with statistical significance determined at a significance level of P < 0.05.
Results
Patient characteristics
A total of 1166 patients, comprising 540 males and 626 females, were ultimately enrolled in this study, as illustrated in Fig. 1. The average age of the participants was 71.1 years. Based on the CC tertile points of 35 and 38.5, the patients were divided into three groups with 373, 400, and 393 individuals, respectively. The baseline characteristics of these patients are summarized in Table 1.
Kaplan-Meier survival curve analysis
After a mean follow-up of 127.78 months, a total of 922 all-cause deaths were recorded, with 515 of these deaths being attributed to cardiovascular diseases. Among these participants, 339, 323, and 260 deaths occurred in three groups. The Kaplan-Meier curves, illustrated in Fig. 2, depict the survival probabilities for each group. A log-rank test was performed to compare the survival probabilities among the three groups, and the results indicated significant differences (P < 0.0001) for both all-cause mortality and cardiovascular mortality. The log-log survival plot (Figure S1) exhibited intersecting Kaplan-Meier curves during the early phase (< 30 months), likely attributable to small sample size and high censoring rates, which compromised the stability of survival estimates. In the later phase (> 30 months), the curves maintained parallel trajectories, suggesting consistent hazard ratios over time. Schoenfeld residual tests yielded non-significant p-values (0.86 for all-cause mortality and 0.57 for cardiovascular mortality), confirming that the proportional hazards assumption of the Cox regression model was not violated.
Kaplan–Meier curves of the survival rate with calf circumference tertile groups. A, All-cause mortality; B, Cardiovascular mortality.
Relationship between CC and cardiovascular and all-cause mortality
Table 2 displays the result of unadjusted and multivariable-adjusted Cox models, based on baseline CC as both a continuous and categorical variable. In the fully adjusted model, for every 1 unit reduce in CC as a continuous variable, there was a increased HR of 1.08 (1.03, 1.13) for all-cause mortality and 1.04 (0.93, 1.17) for cardiovascular mortality. When CC was analyzed as a categorical variable, compared to the CC > 38.5 group(used as the reference), participants in the 35.0 < CC < 38.5 group had an HR of 1.55 (1.20, 2.00) for all-cause mortality and 1.28 (0.72, 2.28) for cardiovascular mortality. Similarly, participants in the CC < 35.0 group had an HR of 2.05 (1.44, 2.93) for all-cause mortality and 1.58 (0.75, 3.33) for cardiovascular mortality. Adjustment for potential confounders had a minor impact on the association between baseline CC and both all-cause and cardiovascular mortality.
RCS analysis
Consistent with the Cox regression results, the risk both for all-cause and cardiovascular death decreased with the increase of CC value. We observed a significant nonlinear relationship between CC and cardiovascular mortality (P-nonlinear = 0.0032) and all-cause mortality (P-nonlinear = 0.0112) with the cut-point value at 36.66. The risk of death decreases rapidly before reaching the cut-point but slows down after surpassing it. (Fig. 3)
The weighted restricted cubic splines for associations of calf circumference with all-cause and cardiovascular mortality. (A) All-cause mortality; (B) Cardiovascular mortality.
Subgroup and sensitivity analyses
Figures 4 and 5 present the correlation between CC and the risk of long-term all-cause and cardiovascular mortality among various subgroups. The outcomes largely mirror our preliminary findings, with the exception of a few subgroups where statistical significance was not achieved. The p-value for the interaction term suggests the absence of significant interaction between CC and other variables in the model. The results indicated several significant associations between participant characteristics and the risk of all-cause and cardiovascular mortality. Participants under the age of 65 exhibit a markedly increased risk in comparison to their older counterparts. Females demonstrated a significantly increased risk compared to males across both mortality outcomes. Mexican American individuals display a reduced risk compared to Other Hispanic individuals. Socioeconomic status, represented by the family income to poverty ratio, suggests that higher income is associated with a decreased risk of mortality. Nonsmokers and former smokers generally have a lower risk of mortality compared to current smokers. Patients with lower BMI have a highest risk of mortality. Furthermore, a higher education level is linked to a reduced risk of all-cause mortality. Regarding health conditions, both diabetes, hypertension areassociated with a higher risk of mortality but dyslipidemia is related to a lower risk of mortality. Patients with lower eGFR are associated with a higher risk of mortality. Furthermore, sensitivity analyses yielded similar results for both all-cause and cardiovascular mortality (Table S1). These findings underscore the complex interplay of demographic, socioeconomic, behavioral, and health factors in determining the role of CC in the prognosis of the CKD population.
Forest plot of subgroup analyses for all-cause mortality. Subgroup analysis employed full model adjustment, which accounts for potential confounding variables (e.g., age, sex, race, alcohol intake, smoking status, BMI, ratio of family income to poverty, education level, diabetes, hypertension, dyslipidemia) alongside subgroup-specific categorical variables. HR, hazard ratio; BMI, body mass index.
Forest plot of subgroup analyses cardiovascular mortality. Subgroup analysis employed full model adjustment, which accounts for potential confounding variables (e.g., age, sex, race, alcohol intake, smoking status, BMI, ratio of family income to poverty, education level, diabetes, hypertension, dyslipidemia) alongside subgroup-specific categorical variables. HR, hazard ratio; BMI, body mass index.
Discussion
This study demonstrated the association between lower CC and higher risk of all-cause and cardiovascular mortality among a community-based CKD population in the United States. Using multivariate Cox regression and RCS analysis, we found that CC was a reliable predictor of prognosis in CKD stages 3–5 patients. The relationship between CC and the risk of all-cause and cardiovascular mortality exhibited a nonlinear pattern, with individuals possessing a CC below 36.66 cm facing a notably heightened risk of death compared to those with larger calf sizes. Besides, Subgroup and sensitivity analyses further corroborated the robustness of our findings.
Muscle wasting is a common complication in CKD patients, especially among those undergoing dialysis, where the frequency of muscle atrophy is higher19,20. Anthropometric indices serve as crucial parameters for understanding basic health status and the overall nutritional condition of the human body19. Research indicates that CC has emerged as a significant indicator for assessing nutritional risk21, muscle mass22, BMD23, and sarcopenia24. Previous studies using the NHANES cohort have similarly demonstrated that lower CC is associated with higher risks of all-cause and cardiovascular mortality, which aligns with our findings in CKD stages 3–5 patients25,26. Tsai and Chang27conducted a prospective analysis of the relationship between CC and mortality risk among 4,191 elderly individuals, finding that CC is a more effective predictor of long-term mortality risk compared to BMI. Rodrigues et al.28 indicated that CC has been shown to predict adverse clinical outcomes in elderly HD patients. Another study has found that non-invasive testing for sarcopenia, using a simple screening test that includes age, grip strength, and CC, can predict future cardiovascular events in CKD patients, which is in line with our study29. These studies suggested CC is a significant prediction indicator of CKD prognosis. However, one research finding failed to observe a link between decreased CC and clinical events in HD patients17. The difference between our study and this study may be explained by the potential presence of fluid retention and edema in HD patients throughout their illness progression.
Establishing cutoff values for CC measurements holds crucial importance not only in directing diagnosis and treatment strategies but also in assessing the efficacy of therapeutic interventions with optimal precision. A universal cutoff for low CC is lacking. Most studies define it as < 34 cm for men and < 33 cm for women30. A meta-analysis showed that lower CC was associated with an increased risk of mortality across the cutoff values31. Additionally, a CC value below 31 cm predicts declines in physical function and reduced survival rates among elderly individuals; this threshold has been adopted as the standard assessment cutoff by the European Working Group on Sarcopenia in Older People 2 (EWGSOP2)32,33. The elevated cutoff of 36.66 cm identified in our study—compared to thresholds established in general and elderly populations—reflects accelerated muscle vulnerability due to CKD-specific pathophysiology. PEW, chronic inflammation, and metabolic acidosis amplify sarcopenia progression in CKD, precipitating mortality risk at less severe muscle depletion than in healthy individuals34,35. Consequently, adopting this cutoff (36.66 cm) would improve risk stratification by enabling earlier nutritional support in CKD patients.
The mechanisms linking CC to CKD adverse outcomes are multifaceted. Given that CKD patients are susceptible to PEW and metabolic disturbances, these conditions lead to muscle wasting and a reduction in CC, which further increases the risk of mortality3. Additionally, research has demonstrated that, in contrast to the abdominal fat pool, the limb fat pool exhibits greater passivity and can store fatty acids over extended periods, preserving its protective qualities14,36. Moreover, an augmentation in muscle tissue content is correlated with an enhancement in metabolic status, whereas a deficiency in muscle mass may compromise this protective effect, intensifying the incidence of adverse outcomes in CKD patients ultimately37,38. The increase in cardiovascular events associated with CC in CKD patients may be linked to decreased cytokine expression from skeletal muscle. Studies indicate that elevated muscle mass offers protection against left ventricular remodeling and dysfunction, as well as acute kidney injury, whereas the decline in muscle mass and function in CKD elevates the risk of cardiovascular events29,39.
We found that the relationship between CC and the risks of all-cause mortality and cardiovascular is more significant in CKD patients who are younger than 65 years, females, and with a BMI below 25 kg/m². CC may exhibit a positive association with a decreased frailty index and enhanced functional performance in older individuals33. Elderly patients with CKD, particularly those undergoing dialysis, are susceptible to malnutrition16. However, we found that patients younger than 65 years had a higher mortality risk, potentially attributable to more severe PEW in this younger cohort, which may trigger mitochondrial dysfunction and needs further investigation. Research conducted by Cao et al.40 found that middle-aged and older women exhibit greater susceptibility to sarcopenia than men, as estrogen deficiency may impair anabolic sensitivity and accelerate muscle protein catabolism41. BMI is another crucial health indicator. Multiple studies indicate that older adults with lower BMI face higher risks of sarcopenia42,43, which in line with our study. This may be attributable to nutritional resource depletion and chronic inflammation in low-BMI individuals, whereas obesity confers protective metabolic reserves44. The above findings suggest that CC should be regularly assessed in specific populations to monitor nutritional status, and corresponding nutritional intervention measures, such as diet adjustment, increased protein intake, and appropriate physical activity, should be taken to reduce the risks of cardiovascular and mortality.
Our study has some advantages. Firstly, it leverages the extensive and nationally representative sample of the NHANES database, ensuring robust statistical power and external validity for the research findings. Secondly, our study unveils the correlation between CC and both all-cause mortality and cardiovascular among patients with CKD. Secondly, we adhered to standardization principles, meticulously adjusted covariates to minimize bias, applied stratified and sensitivity analyses to deeply explore variable relationships, and established segmented effects and identified threshold values. However, there are also several limitations. Firstly, the data primarily depend on self-reporting, potentially introducing information bias, and the study cohort is largely comprised of young Americans, thereby restricting the global applicability of our findings. Secondly, our data collection was confined to baseline CC data, neglecting any temporal variations. Thirdly, as an observational study, reverse causality cannot be ruled out, where participants with lower baseline calf circumference may have had pre-existing health deterioration that contributed to both muscle loss and increased mortality. Lastly, we are unable to establish causality, which hampers our capacity to further evaluate associations. Despite these constraints, our study still adds value to the existing knowledge base, and there is a pressing need for more extensive data collection and research in the future to further validate and elucidate these relationships.
Conclusion
Our research revealed that a reduced CC is correlated with a poorer prognosis in CKD and CC may serve as an effective predictive tool for assessing the prognosis in patients with CKD stages 3–5 in the United States. Furthermore, the relationship between CC and the risk of all-cause and cardiovascular death exhibits nonlinear characteristics. Future large sample sizes and well-designed prospective studies should further investigate intervention measures targeting CC management, to improve the clinical prognosis of CKD patients.
Data availability
The data relevant to my research is stored in the publicly accessible repository on the NHANES website (https://wwwn.cdc.gov/nchs/nhanes/Default.aspx).
Abbreviations
- CKD:
-
Chronic kidney disease
- NHANES:
-
National Health and Nutritional Examination Survey
- RCS:
-
Restricted cubic spline
- HR:
-
Hazard ratio
- CI:
-
Confidence intervals
- eGFR:
-
Estimated glomerular filtration rate
- NDI:
-
National Death Index
- CKD-EPI:
-
Chronic Kidney Disease Epidemiology Collaboration
- BMI:
-
Body Mass Index
- CC:
-
Calf circumference
References
G.B.D.C.K.D. Collaboration Global, regional, and National burden of chronic kidney disease, 1990–2017: a systematic analysis for the global burden of disease study 2017. Lancet 395, 709–733 (2020).
Liyanage, T. et al. Worldwide access to treatment for end-stage kidney disease: a systematic review. Lancet 385, 1975–1982 (2015).
Sabatino, A. et al. Protein-energy wasting and nutritional supplementation in patients with end-stage renal disease on Hemodialysis. Clin. Nutr. 36, 663–671 (2017).
Bakinowska, E., Olejnik-Wojciechowska, J., Kielbowski, K., Skoryk, A. & Pawlik, A. Pathogenesis of sarcopenia in chronic kidney Disease-The role of inflammation, metabolic dysregulation, gut dysbiosis, and MicroRNA. Int. J. Mol. Sci. 25, 8474 (2024).
Martinson, M. et al. Associations of body size and body composition with functional ability and quality of life in Hemodialysis patients. Clin. J. Am. Soc. Nephrol. 9, 1082–1090 (2014).
Bichels, A. V. et al. Muscle mass assessed by computed tomography at the third lumbar vertebra predicts patient survival in chronic kidney disease. J. Ren. Nutr. 31, 342–350 (2021).
Carrero, J. J. et al. Screening for muscle wasting and dysfunction in patients with chronic kidney disease. Kidney Int. 90, 53–66 (2016).
Sabatino, A. et al. Making the invisible visible: imaging techniques for assessing muscle mass and muscle quality in chronic kidney disease. Clin. Kidney J. 17, sfae028(2024).
Dávalos-Yerovi, V. et al. Sarcopenia according to the revised European consensus on definition and diagnosis (EWGSOP2) criteria predicts hospitalizations and Long-Term mortality in rehabilitation patients with stable chronic obstructive pulmonary disease. J. Am. Med. Dir. Assoc. 20, 1047–1049 (2019).
Portero-McLellan, K. C. et al. The use of calf circumference measurement as an anthropometric tool to monitor nutritional status in elderly inpatients. J. Nutr. Health Aging. 14, 266–270 (2010).
Kakita, D. et al. Simplified discriminant parameters for sarcopenia among patients undergoing haemodialysis. J. Cachexia Sarcopeni. 13, 2898–2907 (2022).
Guligowska, A. et al. Association between kidney function, nutritional status and anthropometric measures in older people the screening for CKD among older people across Europe (SCOPE) study. Bmc Geriatr. 20, 366 (2020).
Costa Pereira, J. P. D. et al. Body mass index-adjusted calf circumference is associated with mortality in hospitalized older patients with excess weight. Nutrition 125, 112505 (2024).
Liu, J. J., Jin, X. S., Feng, Z. Y. & Huang, J. M. The association of central and extremity circumference with all-cause mortality and cardiovascular mortality: a cohort study. Front. Cardiovasc. Med. 10, 1251619 (2023).
Rodrigues, R. G. et al. Calf circumference predicts falls in older adults on Hemodialysis. J. Ren. Nutr. 33, 363–367 (2023).
Ozawa, M. et al. The implication of calf circumference and grip strength in osteoporosis and bone mineral density among Hemodialysis patients. Clin. Exp. Nephrol. 27, 365–373 (2023).
Yoshikoshi, S. et al. Effects of anthropometric changes on hospitalization and mortality among patients on Hemodialysis. J. Nephrol. 36, 1983–1990 (2023).
Levey, A. S. & Stevens, L. A. Estimating GFR using the CKD epidemiology collaboration (CKD-EPI) creatinine equation: more accurate GFR estimates, lower CKD prevalence estimates, and better risk predictions. Am. J. Kidney Dis. 55, 622–627 (2010).
Yunita, S. et al. Anthropometry indices and body composition in adolescent girls with anemia: A scoping review. J. Multidiscip Healthc. 17, 4131–4141 (2024).
Li, Y., Yang, Y. & Wang, J. Skeletal muscle mass and kidney function among Chinese older adults: a cross-sectional study. Ren. Fail. 46, 2377776(2024).
Maeda, K., Koga, T., Nasu, T., Takaki, M. & Akagi, J. Predictive accuracy of calf circumference measurements to detect decreased skeletal muscle mass and European society for clinical nutrition and Metabolism-Defined malnutrition in hospitalized older patients. Ann. Nutr. Metab. 71, 10–15 (2017).
Kiss, C. M. et al. Calf circumference as a surrogate indicator for detecting low muscle mass in hospitalized geriatric patients. Aging Clin. Exp. Res. 36, 25 (2024).
Singh, R. & Gupta, S. Relationship of calf circumference with bone mineral density and hip geometry: a hospital-based cross-sectional study. Arch. Osteoporos. 10, 17 (2015).
Chen, H. T., Ma, J. X., Liu, A. F., Cui, Y. H. & Ma, X. L. The association between sarcopenia and fracture in middle-aged and elderly people: A systematic review and meta-analysis of cohort studies. Injury 51, 804–811 (2020).
Wu, C. J. et al. Does the additional component of calf circumference refine metabolic syndrome in correlating with cardiovascular risk?? J. Clin. Endocrinol. Metab. 103, 1151–1160 (2018).
Abreo, A. P., Bailey, S. R. & Abreo, K. Associations between calf, thigh, and arm circumference and cardiovascular and all-cause mortality in NHANES 1999–2004. Nutr. Metab. Cardiovasc. Dis. 31, 1410–1415 (2021).
Tsai, A. C. & Chang, T. L. The effectiveness of BMI, calf circumference and mid-arm circumference in predicting subsequent mortality risk in elderly Taiwanese. Br. J. Nutr. 105, 275–281 (2011).
Rodrigues, J. et al. Nutritional status of older patients on hemodialysis: which nutritional markers can best predict clinical outcomes? Nutrition 65, 113–119 (2019).
Hanatani, S. et al. Non-invasive testing for sarcopenia predicts future cardiovascular events in patients with chronic kidney disease. Int. J. Cardiol. 268, 216–221 (2018).
Chen, L. K. et al. Arai, Asian working group for sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J. Am. Med. Dir. Assoc. 21, 300– (2020).
Wei, J. et al. The association between low calf circumference and mortality: a systematic review and meta-analysis. Eur. Geriatr. Med. 13, 597–609 (2022).
Cruz-Jentoft, A. J. et al. EWGSOP2, sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 48, 16–31 (2019).
Landi, F. et al. Calf circumference, frailty and physical performance among older adults living in the community. Clin. Nutr. 33, 539–544 (2014).
Carrero, J. J. et al. Etiology of the protein-energy wasting syndrome in chronic kidney disease: a consensus statement from the International Society of Renal Nutrition and Metabolism (ISRNM). J Ren Nutr 23 77–90. (2013).
Fahal, I. H. Uraemic sarcopenia: aetiology and implications. Nephrol. Dial Transpl. 29, 1655–1665 (2014).
Alser, M., Naja, K. & Elrayess, M. A. Mechanisms of body fat distribution and gluteal-femoral fat protection against metabolic disorders. Front. Nutr. 11, 1368966 (2024).
Srikanthan, P. & Karlamangla, A. S. Muscle mass index as a predictor of longevity in older adults. Am. J. Med. 127, 547–553 (2014).
Li, R. et al. Associations of muscle mass and strength with All-Cause mortality among US older adults. Med. Sci. Sport Exer. 50, 458–467 (2018).
Araki, S. et al. Akt1-mediated skeletal muscle growth attenuates cardiac dysfunction and remodeling after experimental myocardial infarction. Circ. Heart Fail. 5, 116–125 (2012).
Cao, M. et al. Association of calf circumference with osteoporosis and hip fracture in middle-aged and older adults: a secondary analysis. BMC Musculoskelet. Disord. 25, 1095 (2024).
Hansen, M. Female hormones: do they influence muscle and tendon protein metabolism? P Nutr. Soc. 77, 32–41 (2018).
Han, P. et al. Incidence, risk factors, and the protective effect of high body mass index against sarcopenia in Suburb-Dwelling elderly Chinese populations. J. Nutr. Health Aging. 20, 1056–1060 (2016).
Yu, R. B. et al. Incidence, reversibility, risk factors and the protective effect of high body mass index against sarcopenia in community-dwelling older Chinese adults. Geriatr. Gerontol. Int. 14, 15–28 (2014).
Lu, B. W. et al. Association between atherogenic index of plasma, body mass index, and sarcopenia: a cross-sectional and longitudinal analysis study based on older adults in China. Aging Clin. Exp. Res. 37, 122 (2025).
Funding
This work was supported by the Medical Science Research Project of Hebei (reference number: 20240054).
Author information
Authors and Affiliations
Contributions
Panpan Liu and Xudong Huang: Writing original draft; Yuzhe Li, Yunshuang Chen and Xinjun Yang: Methodology and conceptualization; Jinghua Wang, Yaling Bai, and Wei Zhao: Data curation and formal analysis; Yunlong Qin and Lihui Wang: Writing, reviewing, and editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Statement of ethics
The NCHS Research Ethics Review Board reviewed and approved NHANES, and all survey participants provided signed informed consent to participate. No further ethical approval and informed consent were required for secondary analyses of NHANES data, which are de-identified before release by NCHS are and remain anonymous during data analysis.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Liu, P., Huang, X., Li, Y. et al. The associations of calf circumference and cardiovascular and all-cause mortality among population with chronic kidney disease stages 3–5. Sci Rep 15, 26764 (2025). https://doi.org/10.1038/s41598-025-12753-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-12753-6