Abstract
Asthma is a chronic inflammatory respiratory disease that seriously affects patients’ quality of life. Although various pharmacological treatments are currently available, some patients still have poor responses, highlighting the urgent need for new intervention strategies. In recent years, stellate ganglion irradiation (SGI), as an emerging neuromodulation technique, has shown unique therapeutic advantages in various inflammatory diseases. However, the mechanism by which SGI alleviates asthma remains unclear. Silent information regulator 1 (SIRT1), an NAD+-dependent histone deacetylase, plays a key role in regulating cellular inflammatory responses by inhibiting NF-κB and other inflammatory signaling pathways. Given the involvement of SIRT1 and NF-κB imbalance in the pathogenesis of asthmatic airway inflammation, we hypothesize that SGI may alleviate asthmatic inflammation by activating SIRT1 and inhibiting NF-κB activation. To test this hypothesis, we used an ovalbumin (OVA)-induced mouse model of asthma to comprehensively evaluate the effects of SGI on asthma pathophysiology and explore the mediating role of the SIRT1-NF-κB pathway. We found that compared with the asthma group, SGI treatment significantly improved the general symptoms of mice, reduced airway hyperresponsiveness, alleviated inflammatory cell infiltration and cytokine levels in bronchoalveolar lavage fluid (BALF), and ameliorated lung histopathological changes. Mechanistic studies showed that SIRT1 expression was downregulated and the NF-κB pathway was activated in the lung tissues of asthmatic mice, while SGI treatment upregulated SIRT1 expression and inhibited NF-κB activity. In conclusion, this study reveals for the first time that SGI alleviates asthmatic airway inflammation through the SIRT1-NF-κB pathway, providing a new approach for the adjuvant treatment of asthma.
Similar content being viewed by others
Introduction
Epidemiology and pathogenesis of asthma
Asthma is a common chronic inflammatory respiratory disease characterized by a persistent increase in global prevalence1. According to statistics, the number of asthma patients in China has exceeded 30 million, bringing a heavy economic burden to individuals, families, and society2,3. The typical clinical manifestations of asthma include recurrent wheezing, dyspnea, chest tightness, and cough, which seriously affect patients’ quality of life4. Studies on the pathogenesis have shown that the infiltration of inflammatory cells, mainly eosinophils and Th2 lymphocytes, and the excessive secretion of cytokines such as IL-4, IL-5, and IL-13 are the core links driving chronic airway inflammation in asthma5,6. Inflammatory reactions further lead to airway hyperresponsiveness and airflow limitation, forming a vicious cycle7. Although glucocorticoids and other drugs have made some progress in controlling asthma symptoms, a considerable number of patients still present with refractory asthma, suggesting an urgent need to develop new intervention strategies8,9.
Principles and clinical applications of stellate ganglion irradiation (SGI)
The stellate ganglion is a cervical ganglion of the sympathetic chain that plays a key role in regulating the functions of multiple systems such as respiration, circulation, and immunity10. Stellate ganglion irradiation (SGI) is a new technique that selectively irradiates the stellate ganglion with infrared polarized light to exert therapeutic effects by regulating autonomic nerve function11,12. Studies have shown that SGI can effectively relieve pain, cardiovascular diseases, and other clinical symptoms13,14. In recent years, with the proposal of the neuro-immune regulation hypothesis, the application value of SGI in inflammatory diseases has gradually attracted attention15. Animal experiments have shown that SGI can improve inflammatory responses and pain behaviors in rats with arthritis16. Clinical studies have found that SGI treatment can significantly alleviate abdominal pain and diarrhea symptoms in patients with ulcerative colitis17. However, the effects and mechanisms of SGI in the treatment of asthma are still unclear and require further research.
Roles of SIRT1 and NF-κB signaling pathways in the pathogenesis of asthma
Silent information regulator 1 (SIRT1) is an NAD+-dependent histone deacetylase that regulates various physiological and pathological processes through epigenetic modifications18. Numerous studies have shown that SIRT1 plays a key role in anti-inflammatory, antioxidant, and immune tolerance induction19,20. In the respiratory system, SIRT1 can inhibit multiple inflammatory signaling pathways such as NF-κB and STAT3 to maintain airway homeostasis21,22. However, SIRT1 expression is significantly downregulated in asthma patients and animal models, suggesting that its deficiency may be an important cause of chronic airway inflammation23,24.
NF-κB is a key transcription factor that regulates inflammatory responses and consists of two subunits, p50 and p6525. Under normal conditions, NF-κB binds to the inhibitory protein IκB and exists in an inactive state in the cytoplasm26. When stimulated by inflammatory factors, IκB is phosphorylated and degraded, and NF-κB translocates into the nucleus to initiate the transcription of inflammatory factors such as IL-6, IL-8, and TNF-α27. Studies have found that the NF-κB pathway is abnormally activated in asthma patients, and specific inhibition of NF-κB can significantly improve airway inflammation and hyperresponsiveness in asthmatic mice28,29. Interestingly, SIRT1 can inhibit NF-κB activity through deacetylation modification30. Therefore, the imbalance of the SIRT1-NF-κB pathway may be a core link in asthmatic airway inflammation.
Innovative hypothesis of this study
In summary, SIRT1 deficiency and NF-κB activation play key roles in the pathogenesis of asthma, while SGI can alleviate various inflammatory diseases through neuro-immune regulation. In view of this, we propose the following innovative hypothesis: SGI may alleviate airway inflammation in asthmatic mice by upregulating SIRT1 and inhibiting NF-κB activation. To test this hypothesis, this study will systematically evaluate the effects of SGI on pathophysiological changes in asthmatic mice and explore the mediating role of the SIRT1-NF-κB pathway. This study is expected to provide new ideas for the adjuvant treatment of asthma and reveal new mechanisms of SGI intervention in inflammatory diseases.
Materials and methods
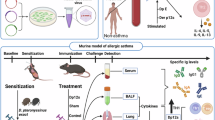
Experimental animals and grouping
Six-week-old SPF-grade female BALB/c mice weighing 18–20 g were selected for this experiment and provided by Beijing Vital River Laboratory Animal Technology Co., Ltd. [Animal License No.: SCXK (Beijing) 2016-0006]. The animals were housed in the SPF-grade animal room of the Plastic Surgery Hospital of the Chinese Academy of Medical Sciences, with a temperature of 22–25 °C, relative humidity of 40-60%, 12-hour light/dark alternation, and free access to food and water.
The mice were randomly divided into the following four groups, with six mice in each group:
-
(1)
Normal control group (Control): healthy mice, no treatment;
-
(2)
Asthma model group (OVA): OVA sensitization and challenge to establish an asthma model;
-
(3)
SGI treatment group (OVA + SGI): asthma model + SGI treatment (1000 mW/cm², every other day, a total of 6 times);
-
(4)
Sham operation group (OVA + Sham): asthma model + SGI sham operation (laser not turned on).
All animal experimental protocols were approved by the Animal Ethics Committee of the Plastic Surgery Hospital of the Chinese Academy of Medical Sciences (Approval No.: CMBSIACUC-2022001). The experimental process strictly followed relevant regulations such as the “Regulations on the Administration of Experimental Animals” and the “Guidelines for the Ethical Review of Experimental Animal Welfare”.
Establishment of asthma model
OVA sensitization
1 mg of OVA (Grade V, Sigma) was dissolved in 0.2 ml of sterile physiological saline and mixed with an equal volume of aluminum hydroxide adjuvant (Alum, Thermo Fisher) to prepare a sensitizer with a final concentration of 2 mg/ml. On days 0, 7, and 14 of the experiment, 0.2 ml of the sensitizer was injected intraperitoneally, a total of 3 times.
OVA challenge
On days 21–24 of the experiment, 20 µl of 2% OVA solution was administered intranasally to the mice once a day for a total of 4 times by direct nasal instillation. Mice in the normal control group and sham operation group received an equal volume of sterile physiological saline intranasally.
SGI treatment plan
SGI treatment was started on the 25th day of the experiment. Referring to the method in the literature31, the mice were fixed and SGI treatment was performed using a ZZIR-ID infrared polarized light therapy instrument (Beijing Zhuangzhi Technology). The emission probe was placed vertically on the skin of the right neck, located at the carotid artery pulsation point (the midpoint between the lower edge of the sternocleidomastoid muscle and the skull), and irradiated with a laser with a power density of 1000 mW/cm² for 15 min each time, every other day, for a total of 6 times. This regimen was selected as a starting point based on our team’s broader experience with this irradiation modality in mice, considering tolerability and potential efficacy. Mice in the sham operation group were subjected to the same body position and probe placement method, but the laser was not turned on.
Experimental observation indicators and detection methods
Histopathological examination of lung tissue
The left middle lobe of the lung was taken, fixed with 4% paraformaldehyde, embedded in paraffin, and continuously sectioned. HE staining, PAS staining, and Masson staining were performed to observe the infiltration of inflammatory cells, goblet cell hyperplasia, mucus secretion, and collagen deposition in the airway wall under a light microscope, and semi-quantitative scoring was performed according to the methods in the literature32,33.
Analysis of bronchoalveolar lavage fluid (BALF)
After lung function measurement, 1 ml of cold PBS was slowly injected into the lungs through the tracheal intubation, and the lavage fluid was aspirated after 3 times of washing to obtain BALF. 100 µl of BALF was taken, and an equal volume of trypan blue staining solution was added. The total number of cells was counted under a blood cell counting plate. 200 µl of BALF was taken and centrifuged at 1500 rpm for 5 min. The sediment was smeared and stained with Wright-Giemsa to count the number of eosinophils, lymphocytes, and other inflammatory cells under a light microscope. The remaining BALF was centrifuged at 3000 rpm for 10 min, and the supernatant was collected and stored at -80 °C for ELISA detection.
ELISA detection of cytokines in BALF
The levels of IL-4, IL-5, IL-13, IL-17, IFN-γ, and IL-10 in BALF were measured using ELISA kits (R&D Systems) according to the manufacturer’s instructions.
Real-time fluorescence quantitative PCR analysis
Total RNA was extracted from the right middle lobe of the lung tissue using the Trizol method, and the concentration and purity were determined using a NanoDrop spectrophotometer. 1 µg of RNA was taken, and Oligo(dT)18 primers and reverse transcriptase were added. After incubation at 42 °C for 60 min, cDNA was synthesized. SYBR Green method was used for qPCR amplification to detect the mRNA expression of SIRT1, NF-κB p65, IL-6, IL-8, TNF-α, IL-4, IL-5, IL-13, Mucin, Collagen, and α-SMA. The primer sequences are shown in (Table 1). β-actin was used as an internal reference. The reaction conditions were: 95 °C pre-denaturation for 5 min; 95 °C for 10 s, 60 °C for 30 s, a total of 40 cycles. The 2-ΔΔCt method was used to calculate the relative expression of the target genes.
Western blot analysis
Total protein was extracted from the right lung tissue, and the concentration was determined by the BCA method. 40 µg of protein was loaded. The proteins were separated by 10% SDS-PAGE gel electrophoresis, transferred to a PVDF membrane, and blocked with 5% skim milk. Primary antibodies (1:500-1:1000) against SIRT1, phosphorylated NF-κB p65 (p-NF-κB p65), phosphorylated IκBα (p-IκBα), and IκBα were added and incubated overnight at 4 °C. After washing with TBST, horseradish peroxidase-labeled secondary antibody (1:5000) was added, and the bands were visualized using ECL luminescent substrate. The band images were acquired using a ChemiDoc MP imaging system, and the gray values were analyzed using ImageJ software.
Statistical analysis
SPSS 23.0 software was used for statistical analysis. Measurement data are expressed as mean ± standard deviation (x ± s), and comparisons between groups were performed using one-way analysis of variance, and pairwise comparisons were performed using LSD-t test. Count data are expressed as percentage (%), and the χ2 test was used. P < 0.05 was considered statistically significant.
Ethics statement
All animal experiments were conducted in accordance with the ARRIVE guidelines and were approved by the Institutional Animal Care and Use Committee (IACUC) of the Plastic Surgery Hospital, Chinese Academy of Medical Sciences (Approval Number: No.2024003114). The study protocol was designed to minimize animal suffering and ensure compliance with international standards for the ethical use of animals in research.
Results
SGI improves airway hyperresponsiveness in asthmatic mice
Compared with the control group, OVA-sensitized mice exhibited a significant increase in airway resistance (Rrs) in response to increasing doses of methacholine (0–48 mg/mL) (P < 0.0001), demonstrating enhanced airway hyperresponsiveness (Fig. 1). The SGI-treated group showed significantly reduced airway responsiveness compared to the OVA group (P < 0.0001), with a rightward shift in the dose-response curve, indicating that SGI treatment effectively attenuates airway hyperresponsiveness in the asthmatic mouse model.
Rrs dose-response curves of each group of mice after MCH inhalation. Compared with the control group, #P < 0.05; compared with the asthma group, ****P < 0.0001.
SGI alleviates airway inflammation in asthmatic mice
SGI reduces inflammatory cell infiltration in BALF
Compared with the control group, the total cell number, eosinophil percentage, and lymphocyte percentage in BALF of asthmatic mice were significantly increased (P < 0.01). After SGI treatment, the number of various inflammatory cells was significantly lower than that in the asthma group (P < 0.05), indicating that SGI can alleviate inflammatory cell infiltration in the bronchial lumen (Fig. 2).
The total cell number and percentages of inflammatory cells in BALF of each group of mice. Compared with the OVA group, ****P < 0.001.
SGI inhibits cytokine secretion in BALF
ELISA results(Fig. 3) showed that compared with the control group, the levels of Th2 cytokines IL-4, IL-5, and IL-13 in BALF of OVA-induced asthmatic mice were significantly increased (P < 0.0001). After SGI treatment, the levels of these Th2 cytokines were markedly reduced compared to the OVA group (P < 0.0001), while no significant difference was observed between OVA and OVA + Sham groups, suggesting that SGI effectively suppresses Th2 cytokine production in BALF of asthmatic mice.
Effects of SGI on Th2 cytokine levels in BALF. The concentrations of IL-4, IL-5, and IL-13 in BALF were measured by ELISA. Data are presented as mean ± SEM. ****P < 0.0001; ns, not significant.
SGI improves lung histopathological changes
HE staining showed a large number of inflammatory cell infiltrations around the bronchi and in the alveolar septa, airway epithelial shedding, and smooth muscle hyperplasia in asthmatic mice. PAS staining revealed goblet cell hyperplasia and the formation of mucus plugs in the lumen. After SGI treatment, the above pathological changes were significantly alleviated (P < 0.05) (Fig. 4).
HE staining (A) and PAS staining (B) of lung tissues from each group of mice. Scale bar = 50 μm.
SGI inhibits NF-κB activation by upregulating SIRT1
Effects of SGI on SIRT1 and NF-κB expression
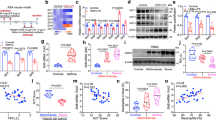
qRT-PCR results showed that compared with the control group, SIRT1 mRNA expression was downregulated in the lung tissues of asthmatic mice, while NF-κB p65 mRNA levels were significantly upregulated. After SGI treatment, SIRT1 expression was significantly increased (P < 0.001) and NF-κB p65 mRNA was markedly reduced (P < 0.0001) compared to the OVA group (Fig. 5A).
Western blot further confirmed these findings at the protein level. SIRT1 protein expression was downregulated in the asthma group compared to control (P < 0.001). SGI treatment significantly reversed this decrease in SIRT1 protein (P < 0.01). Phospho-NF-κB p65 (p-NF-κB p65) levels were elevated in asthmatic mice (P < 0.01), indicating activation of the NF-κB inflammatory pathway. Importantly, SGI treatment attenuated this p-NF-κB p65 increase (P < 0.05). While total NF-κB p65 was unchanged, the ratio of p-IκBα to total IκBα was increased in the OVA group (P < 0.001), further indicating NF-κB activation. Again, SGI administration significantly lowered this ratio (P < 0.0001) (Fig. 5B).
Effects of SGI on the mRNA (A) and protein (B) expression of SIRT1, NF-κB p65, and related inflammatory factors in lung tissues of asthmatic mice. *P < 0.05; **P < 0.01;***P < 0.001;****P < 0.0001.
Discussion
Asthma is a chronic respiratory disease that seriously endangers human health. Although glucocorticoids and other drugs play an important role in controlling asthma symptoms, a considerable number of patients still present with refractory asthma, suggesting an urgent need to develop new treatment strategies34,35. In recent years, with the proposal of the neuro-immune regulation hypothesis, the role of autonomic nervous system imbalance in asthma and other inflammatory diseases has gradually attracted attention36. Studies have shown that sympathetic excitation can activate β2-adrenergic receptors, promote Th2 cell differentiation, and aggravate airway inflammation37. Blocking sympathetic nerves can significantly alleviate airway hyperresponsiveness and inflammatory responses in asthmatic mice38, suggesting that regulating autonomic nerve function may be a new target for the treatment of asthma.
Effects of SGI on pathophysiological changes in asthmatic mice
The stellate ganglion is a cervical ganglion of the sympathetic chain that dominates important organs such as the heart, lungs, and head and face10. Clinically, as a safe and effective minimally invasive interventional technique, SGI can selectively regulate the function of the stellate ganglion and has been widely used in the fields of pain management, cardiovascular diseases, and other fields13,14. Recent studies have found that SGI also shows unique advantages in alleviating inflammatory responses. Animal experiments have shown that SGI can effectively improve the survival rate and lung inflammation of mice with acute lung injury induced by endotoxin39. Clinical studies have found that SGI combined with conventional treatment can significantly improve lung function and quality of life in patients with chronic obstructive pulmonary disease40. However, whether SGI can alleviate asthma is still unclear.
In this study, we successfully established an asthmatic mouse model using the method of OVA sensitization and challenge. Compared with the control group, asthmatic mice showed obvious asthma-like symptoms, enhanced airway hyperresponsiveness, increased levels of inflammatory cells and cytokines in BALF, and aggravated lung histopathological changes, which is consistent with the reports in the literature41,42. On this basis, we first explored the effects of SGI on the pathophysiological changes of asthmatic mice. The results showed that compared with the asthma group, SGI treatment not only improved the airway hyperresponsiveness of mice but also alleviated the inflammatory responses in BALF and lung tissues. Combining the above results, we preliminarily confirmed that SGI can effectively alleviate airway inflammation in asthmatic mice and has good therapeutic prospects.
SIRT1-NF-κB pathway: a new mechanism for SGI to intervene in asthma
Although SGI has shown good therapeutic effects in various inflammatory diseases, its mechanism of action has not been fully elucidated. Previous studies have shown that the autonomic nervous system can regulate the body’s immune response through the neuro-immune axis43. Norepinephrine released by sympathetic nerve endings can bind to β2 receptors on the surface of immune cells, activate the PKA-CREB pathway, and promote the expression of Th2 cell cytokines44. Severing sympathetic nerves can significantly inhibit allergen-induced Th2 inflammatory responses45. In addition, sympathetic hyperactivity can also cause mast cell degranulation and release inflammatory mediators such as histamine and leukotrienes, exacerbating airway inflammation46. However, the above studies mainly focused on classical neuro-immune regulatory pathways and lacked in-depth exploration of key molecular mechanisms.
Our study found that SIRT1 expression was significantly downregulated and the NF-κB pathway was abnormally activated in the lung tissues of asthmatic mice, suggesting that the imbalance between the two is involved in the pathogenesis of asthmatic airway inflammation. qRT-PCR and Western blot results also confirmed that SGI can activate SIRT1 and inhibit the expression of NF-κB downstream inflammatory factors. Combining the above results, we speculate that the SIRT1-NF-κB pathway may be a key link for SGI to exert anti-inflammatory effects. Related studies have shown that SIRT1, as an NAD+-dependent histone deacetylase, can directly bind to and deacetylate the p65 subunit of NF-κB, thereby blocking its transcriptional activity47,48. The new mechanism revealed in this study that SGI activates SIRT1 and subsequently inhibits NF-κB activity not only enriches the understanding of the anti-inflammatory effects of SGI but also provides new ideas for the adjuvant treatment of asthma.
Limitations of this study and future prospects
This study used animal models to preliminarily explore the effects and mechanisms of SGI in alleviating asthmatic airway inflammation, which has good innovative significance. However, there are still some limitations.
First, this study mainly observed the effects of early intervention with SGI, while asthma often requires long-term management. Therefore, it is necessary to conduct studies with longer durations to evaluate the impact of SGI on chronic pathological changes such as airway remodeling, including aspects like fibrosis and smooth muscle hypertrophy.
Second, in addition to the SIRT1-NF-κB pathway, SGI may also exert effects through other signaling pathways, such as JAK-STAT pathway, inflammasome activation, or further interactions with pathways like STAT347,48,49 and Nrf250, which need to be further explored.
Third, this study preliminarily confirmed the therapeutic effect of SGI on asthmatic mouse models, but its efficacy and safety in clinical applications need to be verified by large-sample, multicenter randomized controlled trials.
Fourth, while our sham operation controlled for handling and procedural stress, future studies could incorporate more rigorous sham controls, such as irradiation of a non-relevant anatomical site or using a non-therapeutic wavelength with similar thermal properties, to definitively rule out non-specific effects of the irradiation procedure itself.
Fifth, while SGI is applied locally to the stellate ganglion, the potential for systemic effects mediated by SIRT1 activation, including unintended metabolic or broader immunological consequences, was not assessed. Future studies should consider evaluating such systemic impacts to better understand the overall physiological response to SGI.
Sixth, while airway hyperresponsiveness (AHR) was assessed via Rrs, the inclusion of other lung function parameters, such as compliance, tidal volume, or measures analogous to FEV in humans (though challenging in mice), could offer a more comprehensive evaluation of SGI’s respiratory effects in future studies.
Despite this, the exploration of the mechanism of SGI’s anti-asthmatic effects in this study is expected to provide new candidate targets for the adjuvant treatment of asthma. Future research can be conducted in the following aspects: First, further clarify the causal relationship between SIRT1 and NF-κB in the anti-inflammatory effects of SGI. This can be achieved by employing SIRT1-specific agonists (e.g., resveratrol, as mentioned) or inhibitors, or by utilizing SIRT1 knockout/transgenic mice in the asthma model alongside SGI treatment to determine if the therapeutic effects of SGI are abolished or diminished in the absence of SIRT1 signaling. Second, explore the effects of SGI on regulatory immune cells such as Treg and Breg and the peripheral tolerance mechanisms mediated by them. Third, develop drugs targeting SIRT1, such as resveratrol51, and evaluate their therapeutic effects on asthma and other inflammatory diseases. Fourth, conduct well-designed clinical trials to evaluate the efficacy, safety, and optimal parameters of SGI in asthmatic patients, considering different asthma phenotypes and severity.
Conclusion
In conclusion, this study first confirmed that SGI can effectively improve the airway inflammatory response in asthmatic mice and alleviate airway hyperresponsiveness and pathological changes. Mechanistic studies showed that SGI can upregulate SIRT1, inhibit NF-κB pathway-mediated inflammatory factor expression, and revealed a new mechanism for SGI to intervene in asthma. This study not only deepens the understanding of the anti-inflammatory effects of SGI but also provides new ideas for the adjuvant treatment of asthma. In the future, it is hoped that through in-depth mechanistic research, optimization of administration plans, and rigorous safety assessments, SGI can be transformed into a safe and effective new method for the prevention and treatment of asthma.
Data availability
Data supporting the findings of this study are available in Zenodo under the DOI: https://doi.org/10.5281/zenodo.14671155.
References
Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention. http://www.ginasthma.org (2024).
Lin, J. et al. Prevalence and risk factors of asthma in Mainland china: the CARE study. Respir. Med. 137, 48–54. https://doi.org/10.1016/j.rmed.2018.02.010 (2018).
Huang, K. et al. Prevalence, risk factors, and management of asthma in china: a National cross-sectional study. Lancet 394 (10196), 407–418. https://doi.org/10.1016/S0140-6736(19)31147-X (2019).
Papi, A., Brightling, C., Pedersen, S. E., Reddel, H. K. Asthma. Lancet 391 (10122), 783–800. https://doi.org/10.1016/S0140-6736(17)33311-1 (2018).
Lambrecht, B. N., Hammad, H. & Fahy, J. V. The cytokines of asthma. Immunity 50 (4), 975–991. https://doi.org/10.1016/j.immuni.2019.03.018 (2019).
Gour, N. & Wills-Karp, M. IL-4 and IL-13 signaling in allergic airway disease. Cytokine 75 (1), 68–78. https://doi.org/10.1016/j.cyto.2015.05.014 (2015).
Wenzel, S. Severe asthma in adults. Am. J. Respir Crit. Care Med. 172 (2), 149–160. https://doi.org/10.1164/rccm.200409-1181PP (2005).
Porsbjerg, C. & Menzies-Gow, A. Co-morbidities in severe asthma: clinical impact and management. Respirology 22 (4), 651–661. https://doi.org/10.1111/resp.13026 (2017).
Fitzpatrick, A. M. & Moore, W. C. Severe asthma Phenotypes - How should they guide evaluation and treatment?? J. Allergy Clin. Immunol. Pract. 5 (4), 901–908. https://doi.org/10.1016/j.jaip.2017.05.015 (2017).
Sletten, D. M., Nickander, K. K. & Low, P. A. Postural tachycardia syndrome. Auton. Neurosci. 145 (1–2), 73–77. https://doi.org/10.1016/j.autneu.2008.03.004 (2009).
Singh, H. & Rajarathinam, M. Stellate ganglion block beyond chronic pain: A literature review on its application in painful and non-painful conditions. J. Anaesthesiol. Clin. Pharmacol. 40 (2), 185–191. https://doi.org/10.4103/joacp.joacp_304_22 (2024).
Patel, R. A., Condrey, J. M., George, R. M., Wolf, B. J. & Wilson, S. H. Stellate ganglion block catheters for refractory electrical storm: a retrospective cohort and care pathway. Reg. Anesth. Pain Med. 48 (5), 224–228. https://doi.org/10.1136/rapm-2022-104172 (2023).
Piraccini, E., Munakomi, S. & Chang, K. V. Stellate Ganglion Blocks (StatPearls. StatPearls Publishing, 2022).
Lee, Y. S., Wie, C., Pew, S. & Kling, J. M. Stellate ganglion block as a treatment for vasomotor symptoms: clinical application. Cleve Clin. J. Med. 89 (3), 147–153. https://doi.org/10.3949/ccjm.89a.21032 (2022). Published 2022 Mar 1.
Lipov, E. & Candido, K. Efficacy and safety of stellate ganglion block in chronic ulcerative colitis. World J. Gastroenterol. 23 (32), 5925–5932. https://doi.org/10.3748/wjg.v23.i32.5925 (2017).
Huh, Y., Ji, R. R., Chen, G. & Neuroinflammation Bone marrow stem cells, and chronic pain. Front. Immunol. 8, 1014. https://doi.org/10.3389/fimmu.2017.01014 (2017).
Zhao, H. Y., Yang, G. T., Sun, N. N., Kong, Y. & Liu, Y. F. Efficacy and safety of stellate ganglion block in chronic ulcerative colitis. World J. Gastroenterol. 23 (3), 533–539. https://doi.org/10.3748/wjg.v23.i3.533 (2017).
Yang, Y. et al. Regulation of SIRT1 and its roles in inflammation. Front. Immunol. 13, 831168. https://doi.org/10.3389/fimmu.2022.831168 (2022). Published 2022 Mar 11.
Peng, Z., Zhang, W., Qiao, J. & He, B. Melatonin attenuates airway inflammation via SIRT1 dependent Inhibition of NLRP3 inflammasome and IL-1β in rats with COPD. Int. Immunopharmacol. 62, 23–28. https://doi.org/10.1016/j.intimp.2018.06.033 (2018).
Fu, C. et al. Activation of SIRT1 ameliorates LPS-induced lung injury in mice via decreasing endothelial tight junction permeability. Acta Pharmacol. Sin. 40 (5), 630–641. https://doi.org/10.1038/s41401-018-0045-3 (2019).
Zhang, Y. & Zeng, Y. Curcumin reduces inflammation in knee osteoarthritis rats through blocking TLR4 /MyD88/NF-κB signal pathway. Drug Dev. Res. 80 (3), 353–359. https://doi.org/10.1002/ddr.21509 (2019).
Kim, D. H. et al. SIRT1 activation by Resveratrol ameliorates cisplatin-induced renal injury through deacetylation of p53. Am. J. Physiol. Ren. Physiol. 301 (2), F427–F435. https://doi.org/10.1152/ajprenal.00258.2010 (2011).
Chen, J. et al. SIRT1 protects against microglia-dependent amyloid-β toxicity through inhibiting NF-κB signaling. J. Biol. Chem. 280 (48), 40364–40374. https://doi.org/10.1074/jbc.M509329200 (2005).
Tang, L. et al. Suppression of Sirtuin-1 increases IL-6 expression by activation of the Akt pathway during allergic asthma. Cell. Physiol. Biochem. 43 (5), 1950–1960. https://doi.org/10.1159/000484119 (2017).
Kim, S. R. et al. Involvement of Sirtuin 1 in airway inflammation and hyperresponsiveness of allergic airway disease. J. Allergy Clin. Immunol. 125 (2), 449–460. https://doi.org/10.1016/j.jaci.2009.08.009 (2010).
Li, K., Lv, G. & Pan, L. Sirt1 alleviates LPS induced inflammation of periodontal ligament fibroblasts via downregulation of TLR4. Int. J. Biol. Macromol. 119, 249–254. https://doi.org/10.1016/j.ijbiomac.2018.07.099 (2018).
Nakamura, K. et al. Sirtuin 1 attenuates inflammation and hepatocellular damage in liver transplant ischemia/reperfusion: from mouse to human. Liver Transpl. 23 (10), 1282–1293. https://doi.org/10.1002/lt.24821 (2017).
Yao, H. et al. SIRT1 protects against emphysema via FOXO3-mediated reduction of premature senescence in mice. J. Clin. Invest. 122 (6), 2032–2045. https://doi.org/10.1172/JCI60132 (2012).
Ichikawa, T. et al. Sirtuin 1 activator SRT1720 suppresses inflammation in an ovalbumin-induced mouse model of asthma. Respirology 18 (2), 332–339. https://doi.org/10.1111/j.1440-1843.2012.02284.x (2013).
Yang, S. R. et al. Sirtuin regulates cigarette smoke-induced Proinflammatory mediator release via RelA/p65 NF-κB in macrophages in vitro and in rat lungs in vivo: implications for chronic inflammation and aging. Am. J. Physiol. Lung Cell. Mol. Physiol. 292 (2), L567–L576. https://doi.org/10.1152/ajplung.00308.2006 (2007).
Wang, W. et al. Stellate ganglion block attenuates chronic stress induced depression in rats. PLoS One. 12 (8), e0183995. https://doi.org/10.1371/journal.pone.0183995 (2017). Published 2017 Aug 31.
Khodir, A. E., Ghoneim, H. A., Rahim, M. A. & Suddek, G. M. Montelukast reduces sepsis-induced lung and renal injury in rats. Can. J. Physiol. Pharmacol. 92 (10), 839–847. https://doi.org/10.1139/cjpp-2014-0191 (2014).
Daan de Boer, J. et al. Lipopolysaccharide inhibits Th2 lung inflammation induced by house dust mite allergens in mice. Am. J. Respir Cell. Mol. Biol. 48 (3), 382–389. https://doi.org/10.1165/rcmb.2012-0331OC (2013).
von Mutius, E. & Smits, H. H. Primary prevention of asthma: from risk and protective factors to targeted strategies for prevention. Lancet 396 (10254), 854–866. https://doi.org/10.1016/S0140-6736(20)31861-4 (2020).
Peters, M. C. et al. A transcriptomic method to determine airway immune dysfunction in T2-High and T2-Low asthma. Am. J. Respir Crit. Care Med. 199 (4), 465–477. https://doi.org/10.1164/rccm.201807-1291OC (2019).
Ramirez, J. M. The integrative role of the sigh in psychology, physiology, pathology, and neurobiology. Prog Brain Res. 209, 91–129. https://doi.org/10.1016/B978-0-444-63274-6.00006-0 (2014).
Verma, M. et al. Experimental asthma persists in IL-33 receptor knockout mice because of the emergence of thymic stromal Lymphopoietin-Driven IL-9 + and IL-13 + Type 2 innate lymphoid cell subpopulations. J. Allergy Clin. Immunol. 142 (3), 793–803e8. https://doi.org/10.1016/j.jaci.2017.10.020 (2018).
Barnes, P. J. Targeting cytokines to treat asthma and chronic obstructive pulmonary disease. Nat. Rev. Immunol. 18 (7), 454–466. https://doi.org/10.1038/s41577-018-0006-6 (2018).
Herz, J., Filiano, A. J., Smith, A., Yogev, N. & Kipnis, J. Myeloid cells in the central nervous system. Immunity 46 (6), 943–956. https://doi.org/10.1016/j.immuni.2017.06.007 (2017).
Zuo, H., Cattani-Cavalieri, I., Musheshe, N., Nikolaev, V. O. & Schmidt, M. Phosphodiesterases as therapeutic targets for respiratory diseases. Pharmacol. Ther. 197, 225–242. https://doi.org/10.1016/j.pharmthera.2019.02.002 (2019).
Shen, H. H. et al. A causative relationship exists between eosinophils and the development of allergic pulmonary pathologies in the mouse. J. Immunol. 170 (6), 3296–3305. https://doi.org/10.4049/jimmunol.170.6.3296 (2003).
Xu, Y. et al. Regulation of endothelial intracellular adenosine via adenosine kinase epigenetically modulates vascular inflammation. Nat. Commun. 8 (1), 943. https://doi.org/10.1038/s41467-017-00986-7 (2017). Published 2017 Oct 16.
Rosas-Ballina, M. et al. Acetylcholine-Synthesizing T cells relay neural signals in a vagus nerve circuit. Science 334 (6052), 98–101. https://doi.org/10.1126/science.1209985 (2011).
Pavlov, V. A., Chavan, S. S. & Tracey, K. J. Molecular and functional neuroscience in immunity. Annu. Rev. Immunol. 36, 783–812. https://doi.org/10.1146/annurev-immunol-042617-053158 (2018).
Bellinger, D. L. et al. Sympathetic modulation of immunity: relevance to disease. Cell. Immunol. 252 (1–2), 27–56. https://doi.org/10.1016/j.cellimm.2007.09.005 (2008).
Procaccini, C., Pucino, V., De Rosa, V., Marone, G. & Matarese, G. Neuro-endocrine networks controlling immune system in health and disease. Front. Immunol. 5, 143. https://doi.org/10.3389/fimmu.2014.00143 (2014). Published 2014 Apr 7.
Yang, T. et al. Genetic abrogation of adenosine A3 receptor prevents uninephrectomy and high Salt-Induced hypertension. J. Am. Heart Assoc. 5 (7), e003868. https://doi.org/10.1161/JAHA.116.003868 (2016).
Muraoka, S., Kusunoki, N., Takahashi, H., Tsuchiya, K. & Kawai, S. Leptin stimulates interleukin-6 production via janus kinase 2/signal transducer and activator of transcription 3 in rheumatoid synovial fibroblasts. Clin. Exp. Rheumatol. 31 (4), 589–595 (2013).
Ansari, M. Y., Ahmad, N. & Haqqi, T. M. Oxidative stress and inflammation in osteoarthritis pathogenesis: role of polyphenols. Biomed. Pharmacother. 129, 110452. https://doi.org/10.1016/j.biopha.2020.110452 (2020).
Jung, S. M. et al. The Interleukin 33/ST2 axis in patients with primary Sjögren syndrome: expression in serum and salivary glands, and the clinical association. J. Rheumatol. 42 (2), 264–271. https://doi.org/10.3899/jrheum.140234 (2015).
Cicero, A. F. G., Ruscica, M. & Banach, M. Resveratrol and cognitive decline: a clinician perspective. Arch. Med. Sci. 15 (4), 936–943. https://doi.org/10.5114/aoms.2019.85463 (2019).
Author information
Authors and Affiliations
Contributions
K.Z. and H.Z. contributed equally to this work and share first authorship. K.Z., H.Z., and Y.L. designed and performed the experiments, analyzed the data, and prepared the figures. Y.Z. and J.Z. assisted with animal model establishment and histological analysis. Q.W. contributed to the molecular biology experiments and data interpretation. D.Y. conceived and supervised the study, provided funding, and revised the manuscript. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhao, K., Zhang, H., Liu, Y. et al. Stellate ganglion irradiation alleviates airway inflammation in asthmatic mice via activating SIRT1 signaling pathway. Sci Rep 15, 28092 (2025). https://doi.org/10.1038/s41598-025-12901-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-12901-y