Abstract
To evaluate the diagnostic value of plasma D-dimer combined with transvaginal ultrasonography for endometrial carcinoma in postmenopausal women, addressing the increasing incidence in China. This retrospective study analyzed 129 postmenopausal women (July 2021–January 2023) undergoing hysteroscopy, including 61 with endometrial carcinoma (mean age 56.5 ± 6.8 years) and 68 with benign lesions (60.2 ± 7.7 years). We assessed D-dimer levels, ultrasonographic parameters (endometrial thickness, uterine cavity occupation, blood flow signals), and postmenopausal bleeding. Diagnostic performance was evaluated using ROC curves. Multivariate analysis identified three independent predictors: intracavitary blood flow (OR 60.44, 95% CI 8.25–442.98, p < 0.001), postmenopausal bleeding (OR 5.84, 95% CI 1.82–18.75, p = 0.003), and elevated D-dimer (OR 3.12, 95% CI 1.06–9.22, p = 0.04). The comprehensive model achieved superior diagnostic performance (AUC = 0.920, sensitivity = 88.5%, specificity = 85.3%) compared to any individual parameter. Plasma D-dimer combined with transvaginal ultrasonography and clinical symptoms provides an effective screening approach for postmenopausal endometrial carcinoma, serving as a valuable risk assessment strategy for identifying candidates requiring definitive histopathological examination.
Similar content being viewed by others
Introduction
Endometrial carcinoma is a prevalent gynecologic malignancy in developed countries such as Europe and the United States. In China, the incidence of endometrial carcinoma has increased significantly in recent years, ranking as the second most common malignancy after cervical cancer1. Approximately 80% of endometrial carcinoma cases occur in postmenopausal women2. Changes in the coagulation and fibrinolysis systems exist in malignant tumors3. Plasma D-dimer is an important biomarker of fibrinolysis, and an imbalance in the coagulation system is closely related to tumorigenesis4. Huang et al. demonstrated that an increase in plasma D-dimer levels on the first day after surgery can predict high-risk factors for endometrial carcinoma recurrence, such as higher grade, advanced stage, deeper myometrial invasion, lymphatic vascular space infiltration, lymph node metastasis, and cervical involvement5. Transvaginal ultrasound employs high-frequency probes to observe the site of the lesion, echo, depth of infiltration, blood flow, etc., which can improve the effectiveness of diagnosis. For endometrial carcinoma, this imaging method aids in clinical staging by providing detailed visualization of the lesion and surrounding tissues, including the blood flow patterns within and around the lesion6,7. It can also clearly display the depth of the lesion infiltrating muscle layer, providing data reference for the formulation of clinical treatment plan. Ultrasonography has been widely used in clinical practice because of its simplicity, low cost, painlessness, non-invasiveness, and high patient acceptability8. Color ultrasound typically predicts benign and malignant endometrial lesions by endometrial thickness. However, this presents challenges without a clear critical threshold value2. Currently, studies on the correlation between D-dimer levels and transvaginal ultrasonography in endometrial lesions are limited. Therefore, this study aims to assess the diagnostic value of the preoperative detection of peripheral venous plasma D-dimer levels combined with vaginal ultrasound in postmenopausal women with endometrial carcinoma to provide references for the early detection, diagnosis, and treatment of postmenopausal endometrial carcinoma.
Methods
Populations
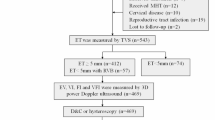
This retrospective study analyzed 129 postmenopausal women who underwent hysteroscopy (from July 2021 to January 2023) for indications including vaginal bleeding, asymptomatic endometrial thickening (≥ 5 mm), intracavitary masses, or prior pathological follow-up. Patients were stratified into endometrial carcinoma (n = 61, mean age 56.5 ± 6.8 years) and benign lesions (n = 68, mean age 60.2 ± 7.7 years) based on histopathology confirmed by gynecologic pathologists with ≥ 8 years of experience.
Inclusion criteria required confirmed menopause status, complete medical records, no history reproductive malignancies, and absence of autoimmune or hematological disorders. Exclusion criteria excluded case with incomplete data or concurrent genital tumors.
Univariate analysis was used to compare age, D-dimer levels, bleeding status, and ultrasonographic parameters. Significant variables (p < 0.05) were subsequently analyzed using multivariate logistic regression. Receiver operating characteristic (ROC) curves were constructed to evaluate diagnostic performance, with the area under the curve (AUC) quantifying accuracy.
Data collection
A total of 129 postmenopausal populations who underwent hysteroscopy from July 2021 to January 2023 were collected as the study objects, Clinical and pathological data of the populations, including the presence or absence of postmenopausal vaginal bleeding, D-dimer levels, uterine occupation by transvaginal ultrasonography, intrauterine blood flow signal, and pathological type, were collected.
Transvaginal ultrasonography
A Voluson E8 ultrasonic diagnostic instrument (US GE S8) with a probe frequency of 5–9 MHz was used for all Populations. Transvaginal ultrasonography assessment included multiple parameters: (1) endometrial thickness measured in the longitudinal plane of the uterus; (2) color Doppler flow evaluation of intrauterine blood flow signals classified as absent, minimal, moderate, or marked; (3) identification of intracavitary masses including their size, border characteristics (regular/irregular), and echogenicity (hyperechoic/hypoechoic/mixed); and (4) assessment of myometrial invasion depth when suspected. While we did not employ the formal IOTA (International Ovarian Tumor Analysis) scoring system, we evaluated similar morphological features including lesion borders, echogenicity, and vascular patterns. Blood flow impedance was assessed using pulsatility index (PI) and resistance index (RI) when appropriate. All measurements were performed in accordance with the guidelines of the International Endometrial Tumor Analysis (IETA) group9.
Enzyme-linked immunosorbent assay (ELISA)
Peripheral venous blood samples (2 mL) were collected from all participants, and plasma D-dimer levels were quantified using the Multiskan SkyHigh automated ELISA system.
Hysteroscopy
An OLYMPUS rigid hysteroscope with a 3 mm outer diameter, 30° angle, and 4.5 mm outer sheath was used for the examination. The uterus was dilated by continuous perfusion with normal saline and the operation was performed under illumination with a cold light source. After the hysteroscope passed through the vagina, the cervix, anterior and posterior walls of the uterus, uterine fundus, both sides of the uterine horn, and left and right sides of the uterus were systematically examined by rotating the hysteroscope lens. The complete uterine cavity and cervical canal were observed during mirror exit. Hysteroscopies were performed based on standard clinical indications, primarily postmenopausal bleeding and abnormal ultrasound findings, following current practice guidelines.
Pathological diagnosis
All pathological diagnoses were made by experienced pathologists with over 8 years of experience at the level of associate chief physician or higher.
Statistical analysis
Statistical analysis was performed using SPSS version 25.0 (IBM Corp., Armonk, NY, USA). Categorical data are presented as counts and percentages and were compared using the χ2 test or Fisher’s exact test as appropriate. Continuous variables are presented as mean ± standard deviation or median (interquartile range) based on their distribution, which was assessed using the Shapiro–Wilk test. Between-group comparisons were conducted using the independent t-test for normally distributed data and the Mann–Whitney U test for non-normally distributed data. Receiver operating characteristic (ROC) curves were generated to analyze the diagnostic value of D-dimer, ultrasound parameters, and clinical symptoms. Area under the curve (AUC), sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were calculated. The optimal cut-off values were determined using the Youden index (sensitivity + specificity − 1). Multivariable logistic regression analysis was performed to identify independent predictors of endometrial carcinoma. A p value < 0.05 was considered statistically significant for all analyses.
Results
Univariate analysis revealed significant differences between endometrial carcinoma (n = 61) and benign lesions (n = 68): carcinoma cases demonstrated higher D-dimer levels (1.13 ± 0.98 vs. 0.61 ± 0.49 μg/mL, p = 0.001), thicker endometrium (11.71 ± 6.03 vs. 6.97 ± 5.12 mm, p = 0.002), and younger age (56.5 ± 6.8 vs. 60.2 ± 7.7 years, p = 0.005). Postmenopausal bleeding predominated in carcinoma (78.95% vs. 21.05%, p = 0.001), while uterine cavity occupation was more frequent in benign lesions (66.18% vs. 33.82%, p = 0.001). Malignancies showed enhanced intracavitary blood flow (88.89% vs. 11.11%, p = 0.003) (Table 1).
Multivariate analysis identified three independent predictors: intracavitary blood flow (OR 60.44, p < 0.001), postmenopausal bleeding (OR 5.84, p = 0.003), and elevated D-dimer (OR 3.12, p = 0.04). Uterine occupation negatively correlated with malignancy (OR 0.14, p = 0.023). Age and endometrial thickness lacked independent predictive value (p > 0.05) (Table 2).
Ultrasonographic evaluation in postmenopausal women initially revealed normal endometrial morphology, characterized by a thin, homogeneous endometrium (< 4 mm in thickness) (Fig. 1A). Subsequent differentiation of malignant lesions was based on pathological features including endometrial thickening (≥ 4 mm), heterogeneous echogenicity, and myometrial invasion (Fig. 1B,C), which correlated with prominent vascular signals on Doppler imaging. When combined with elevated D-dimer levels, these sonographic parameters demonstrated superior diagnostic accuracy (AUC = 0.920, 95% CI 0.87–0.96) compared to individual biomarkers (D-dimer AUC = 0.710; abnormal bleeding AUC = 0.781).
Transvaginal ultrasonographic features of normal endometrium and endometrial carcinoma. (A) Normal postmenopausal endometrium showing a thin, homogeneous endometrial line with well-defined borders and no detectable blood flow. (B) Endometrial carcinoma displaying abnormal thickening with heterogeneous echogenicity. Color Doppler imaging demonstrates increased vascularity with multiple areas of blood flow signals (red and blue areas) within the endometrial tissue. (C) Advanced endometrial carcinoma with irregular borders and myometrial invasion. Note the disruption of the endometrial–myometrial junction and the presence of blood flow signals extending into the superficial myometrium.
ROC analysis confirmed optimal performance of the comprehensive model (sensitivity = 88.5%, specificity = 85.3%), outperforming isolated markers (Table 3). This non-invasive strategy enhances preoperative risk stratification but requires histopathological confirmation.
Discussion
Endometrial carcinoma is the sixth most common malignant disease in women and most common gynecological malignancy in high- and middle-income countries. In 2020, 417,000 new cases and 97,000 deaths were reported worldwide10. With the continuous increase in life expectancy, the incidence of endometrial carcinoma continues to increase annually. It is projected that by 2040, the worldwide incidence of endometrial carcinoma is expected to increase by > 50%11. Hysteroscopy and diagnostic curettage pathology are the gold standards for the diagnosis of endometrial carcinoma. Hysteroscopy can directly observe the presence of lesions in the uterine cavity and cervical canal, as well as the size and location of the lesions, and biopsy can be performed under direct vision, which is more accurate for the diagnosis of focal endometrial carcinoma and assessment of cervical involvement12,13. However, its clinical application is relatively limited owing to risks associated with surgery and anesthesia, trauma, relatively high price, and possible risk of tumor spread13. Additionally, for postmenopausal populations, blind invasive examinations such as curettage or hysteroscopy may increase the difficulty of surgery owing to multiple factors, and postoperative complications are higher than those in premenopausal populations14. Hysteroscopy in asymptomatic women has a 3.6–13.6% chance of causing serious complications15. Papakonstantinou et al. found that endometrial biopsy in postmenopausal populations is more painful and less acceptable than that in premenopausal populations16. Cervical atrophy in postmenopausal populations leads to greater difficulty during surgery, which increases the risk of uterine infection, bleeding, and perforation17. Commonly used clinical serum tumor markers for endometrial carcinoma include CAl25, LDH, D-dimer, and NLR18,19,20,21. However, because a single index can also increase in malignant tumors, such as ovarian cancer, cervical cancer, and some benign diseases, its clinical application value is relatively limited22. This study concluded that transvaginal ultrasound combined with plasma D-dimer examination can make a preliminary diagnosis of postmenopausal populations more clearly, and transvaginal ultrasonography indicating uterine occupation and uterine blood signal combined with postmenopausal vaginal bleeding has a high value in the diagnosis of endometrial carcinoma, which can reduce the trauma and pain of populations.
This study found that plasma D-dimer levels combined with transvaginal ultrasonography parameters and postmenopausal vaginal bleeding symptoms serve as a valuable screening tool for endometrial carcinoma. ROC analysis showed our comprehensive approach achieved an AUC of 0.920, outperforming individual parameters and providing clinicians with a more precise diagnostic tool.
D-dimer levels are elevated in postmenopausal endometrial carcinoma and correlate with clinical stage5. Our study confirmed its diagnostic value (AUC = 0.710, sensitivity = 0.557, specificity = 0.838) at the 0.860 μg/mL cut-off. However, its moderate sensitivity would result in numerous false negatives, and other gynecological malignancies also exhibit elevated D-dimer levels. D-dimer promotes cancer cell adhesion, invasion, and migration, while enhancing vascular endothelial growth factor expression5, explaining both its association with endometrial carcinoma and its imperfect specificity.
Postmenopausal vaginal bleeding demonstrated significant diagnostic value (AUC = 0.781, sensitivity = 0.738, specificity = 0.824), emerging as the best-performing individual parameter. This aligns with clinical experience, as vaginal bleeding is the most common initial symptom of endometrial carcinoma. Nevertheless, approximately 26.2% of patients may not present with this symptom in early stages, necessitating additional diagnostic indicators.
Our analysis of transvaginal ultrasonography parameters revealed that uterine cavity occupation showed limited value (AUC = 0.358), likely because early endometrial carcinoma often lacks characteristic ultrasonographic features. Blood flow signals performed better (AUC = 0.675) with high specificity (0.956) despite lower sensitivity (0.393). While intrauterine blood flow signals are associated with endometrial carcinoma, they are not exclusive to malignancy. Benign conditions, particularly endometrial polyps and submucosal fibroids, can present with increased vascularity, though typically with different vascular patterns—malignant lesions often show chaotic, irregular vessels with lower impedance, whereas benign lesions exhibit more organized patterns with higher impedance. This differentiation remains challenging, underscoring the necessity of interpreting Doppler findings within a broader clinical context.
Although guidelines typically use 4–5 mm as the threshold for normal postmenopausal endometrial thickness, this value may not be universally appropriate23,24. Factors including abnormal uterine position, obesity, fibroids, and previous surgery can affect measurements25. Clinical assessment should therefore consider not only endometrial thickness but also echo patterns, blood flow signals, and uterine cavity occupation.
The most significant finding was the superior performance of our comprehensive approach. Combining D-dimer with vaginal bleeding symptoms increased the AUC to 0.856, and further integration of ultrasonography parameters achieved an AUC of 0.920 (sensitivity = 0.885, specificity = 0.853). This improvement confirms these parameters’ complementary nature, with each capturing different disease aspects: D-dimer reflects coagulation activation, vaginal bleeding represents clinical manifestation, and ultrasonography provides anatomical and hemodynamic information. As shown in Fig. 2A–C, this comprehensive method reduces both false positive and false negative results. While sensitivity remained stable at 0.885, specificity improved from 0.721 to 0.853, indicating that ultrasonography parameters primarily enhanced the specificity of detection.
Receiver operating characteristic (ROC) curve analysis for predicting endometrial carcinoma in postmenopausal women. (A) ROC curves for individual predictive parameters. Blue line represents postmenopausal vaginal bleeding (symptom); light green line represents plasma D-dimer levels; green line represents uterine cavity occupation detected by transvaginal ultrasonography; and red line represents intrauterine blood flow signals assessed by color Doppler ultrasonography. The diagonal reference line (black dashed) represents a test with no discriminatory ability (AUC = 0.5). (B) ROC curve for the combination of postmenopausal vaginal bleeding and plasma D-dimer levels, demonstrating improved diagnostic performance compared with individual parameters. (C) ROC curve for the comprehensive combined diagnostic approach incorporating all parameters (postmenopausal vaginal bleeding, plasma D-dimer levels, uterine cavity occupation, and intrauterine blood flow signals), showing superior diagnostic accuracy with optimal sensitivity and specificity values. This combined approach provides the most robust predictive model for endometrial carcinoma. AUC area under the curve.
These findings have important clinical implications. For postmenopausal women, especially those with vaginal bleeding, comprehensive assessment significantly improves diagnostic accuracy. For those without obvious bleeding but with elevated D-dimer or abnormal ultrasonography findings, this approach may facilitate early detection, crucial for improved prognosis. We recommend incorporating this screening tool particularly for postmenopausal women with bleeding symptoms. Patients with D-dimer levels exceeding 0.860 μg/mL, postmenopausal bleeding, and abnormal ultrasonography findings should be prioritized for histopathological examination.
It is important to emphasize that while our combined model demonstrated excellent performance, it should not replace histopathological examination, which remains the gold standard for definitive diagnosis. Rather, our approach serves as a risk assessment and screening tool to identify patients who would most benefit from more invasive diagnostic procedures, potentially improving diagnostic efficiency while ensuring no cases are missed due to reliance on any single parameter.
Our study has several limitations, including relatively limited sample size, potential selection bias inherent to retrospective design, inability to assess all factors influencing D-dimer levels, operator-dependent ultrasonography interpretation, and lack of direct comparison with histological examination. Future research should focus on prospective validation, exploration of additional biomarkers, evaluation of cost-effectiveness in various clinical settings, and integration into existing guidelines.
Conclusions
Plasma D-dimer proves to be a helpful diagnostic tool for endometrial carcinoma in postmenopausal women, with its utility enhanced when combined with specific transvaginal ultrasonography parameters (including endometrial thickness, presence of uterine cavity occupation, and color Doppler blood flow signals) and clinical symptoms (AUC = 0.920). Importantly, this approach should be considered a risk assessment strategy to identify candidates for definitive histopathological examination, which remains the diagnostic gold standard, rather than a standalone diagnostic method. Our findings address the specific objective of evaluating this multi-parameter approach’s diagnostic performance in postmenopausal women. We acknowledge the limitations of our current data and recommend further large-scale prospective studies to validate these findings. The scope of our study was limited to diagnosis assessment, and did not extend to early detection methods or treatment protocols.
Data availability
The raw data supporting the conclusions of this article will be made available by Xiaoli Guo, without undue reservation. Data is provided in Supplementary material.
Abbreviations
- AUC:
-
Area under the curve
- ROC:
-
Receiver operating characteristic
- OR:
-
Odds ratio
- CI:
-
Confidence interval
- ELISA:
-
Enzyme-linked immunosorbent assay
- PPV:
-
Positive predictive value
- NPV:
-
Negative predictive value
- IETA:
-
International Endometrial Tumor Analysis
- PI:
-
Pulsatility index
- RI:
-
Resistance index
- LDH:
-
Lactate dehydrogenase
- NLR:
-
Neutrophil-to-lymphocyte ratio
- CA125:
-
Cancer antigen 125
- IOTA:
-
International Ovarian Tumor Analysis
References
Han, B. et al. Cancer incidence and mortality in China, 2022. J. Natl. Cancer Cent. 4, 47–53. https://doi.org/10.1016/j.jncc.2024.01.006 (2024).
Liu, W. & Bai, W. Association of endometrial thickness with lesions in postmenopausal asymptomatic women: Risk factors and diagnostic thresholds. BMC Womens Health 25, 105. https://doi.org/10.1186/s12905-025-03641-2 (2025).
Marchetti, M. & Falanga, A. Hemostatic biomarkers in occult cancer and cancer risk prediction. Thromb. Res. 191(Suppl 1), S37-s42. https://doi.org/10.1016/s0049-3848(20)30395-9 (2020).
Wu, J. et al. Clinical significance of plasma D-dimer in ovarian cancer: A meta-analysis. Medicine (Baltimore) 96, e7062. https://doi.org/10.1097/md.0000000000007062 (2017).
Huang, Y. J. & Li, B. L. The significance of plasma D-dimer level in predicting high risk factors of endometrial cancer. Transl. Cancer Res. 9, 7688–7696. https://doi.org/10.21037/tcr.2020.04.22 (2020).
Wong, M., Amin, T., Thanatsis, N., Naftalin, J. & Jurkovic, D. A prospective comparison of the diagnostic accuracies of ultrasound and magnetic resonance imaging in preoperative staging of endometrial cancer. J. Gynecol. Oncol. 33, e22. https://doi.org/10.3802/jgo.2022.33.e22 (2022).
Spagnol, G. et al. Three-dimensional transvaginal ultrasound vs magnetic resonance imaging for preoperative staging of deep myometrial and cervical invasion in patients with endometrial cancer: Systematic review and meta-analysis. Ultrasound Obstet. Gynecol. 60, 604–611. https://doi.org/10.1002/uog.24967 (2022).
Rei, M., Bernardes, J. F. & Costa, A. Ultrasound in endometrial cancer: Evaluating the impact of pre-surgical staging. Oncol. Rev. 19, 1446850. https://doi.org/10.3389/or.2025.1446850 (2025).
Epstein, E. et al. Ultrasound characteristics of endometrial cancer as defined by International Endometrial Tumor Analysis (IETA) consensus nomenclature: Prospective multicenter study. Ultrasound Obstet. Gynecol. 51, 818–828. https://doi.org/10.1002/uog.18909 (2018).
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249. https://doi.org/10.3322/caac.21660 (2021).
Koskas, M., Amant, F., Mirza, M. R. & Creutzberg, C. L. Cancer of the corpus uteri: 2021 update. Int. J. Gynaecol. Obstet. 155(Suppl 1), 45–60. https://doi.org/10.1002/ijgo.13866 (2021).
Shen, Y., Yang, W., Liu, J. & Zhang, Y. Minimally invasive approaches for the early detection of endometrial cancer. Mol. Cancer 22, 53. https://doi.org/10.1186/s12943-023-01757-3 (2023).
Di Spiezio Sardo, A. et al. Endometrial biopsy under direct hysteroscopic visualisation versus blind endometrial sampling for the diagnosis of endometrial hyperplasia and cancer: Systematic review and meta-analysis. Facts Views Vis. Obgyn. 14, 103–110. https://doi.org/10.52054/fvvo.14.2.023 (2022).
Wolfman, W. et al. Guideline No. 451: Asymptomatic endometrial thickening in postmenopausal women. J. Obstet. Gynaecol. Can. 46, 102591. https://doi.org/10.1016/j.jogc.2024.102591 (2024).
Li, Z. & Li, L. Risk of malignancies among asymptomatic postmenopausal women with thickened endometrium: A cohort study. Medicine (Baltimore) 98, e14464. https://doi.org/10.1097/md.0000000000014464 (2019).
Papakonstantinou, E. & Adonakis, G. Management of pre-, peri-, and post-menopausal abnormal uterine bleeding: When to perform endometrial sampling?. Int. J. Gynaecol. Obstet. 158, 252–259. https://doi.org/10.1002/ijgo.13988 (2022).
Vitale, S. G. et al. Uterine cervical stenosis: From classification to advances in management. Overcoming the obstacles to access the uterine cavity. Arch. Gynecol. Obstet. 309, 755–764. https://doi.org/10.1007/s00404-023-07126-1 (2024).
He, S. M. et al. Determination of CA-125 levels in the serum, cervical and vaginal secretions, and endometrium in Chinese women with precancerous disease or endometrial cancer. Med. Sci. Monit. 17, 618–625. https://doi.org/10.12659/msm.882046 (2011).
Qin, L. The predictive value of NLR, PLR and MLR in the differential diagnosis of benign uterine diseases and endometrial malignant tumors. Discov. Oncol. 15, 91. https://doi.org/10.1007/s12672-024-00956-8 (2024).
Tian, Y., Chen, Y. Y. & Han, A. L. MiR-1271 inhibits cell proliferation and metastasis by targeting LDHA in endometrial cancer. Eur. Rev. Med. Pharmacol. Sci. 23, 5648–5656. https://doi.org/10.26355/eurrev_201907_18300 (2019).
Seebacher, V. et al. The prognostic value of plasma fibrinogen levels in patients with endometrial cancer: A multi-centre trial. Br. J. Cancer 102, 952–956. https://doi.org/10.1038/sj.bjc.6605547 (2010).
Yang, Q. et al. Value of ultrasound and magnetic resonance imaging combined with tumor markers in the diagnosis of ovarian tumors. World J. Clin. Cases 11, 7553–7561. https://doi.org/10.12998/wjcc.v11.i31.7553 (2023).
Giannella, L. et al. A risk-scoring model for the prediction of endometrial cancer among symptomatic postmenopausal women with endometrial thickness > 4 mm. Biomed. Res. Int. 2014, 130569. https://doi.org/10.1155/2014/130569 (2014).
Wong, M., Amin, T., Thanatsis, N., Foo, X. & Jurkovic, D. Efficacy of transrectal ultrasound in assessing endometrium of postmenopausal women with axial uterus. Ultrasound Obstet. Gynecol. 60, 414–419. https://doi.org/10.1002/uog.24835 (2022).
Maclean, A. et al. Human uterine biopsy: Research value and common pitfalls. Int. J. Reprod. Med. 2020, 9275360. https://doi.org/10.1155/2020/9275360 (2020).
Acknowledgements
We would like to thank Editage (www.editage.cn) for English language editing.
Funding
The authors declare that no funds, grants, or other support was received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
XG and YC designed, drafted, and revised the paper and analyzed the data. JD supervised and submitted the paper for publication. Each author contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All methods of this study were carried out in accordance with relevant guidelines and regulations. This study was approved by the Ethics Committee of Dongyang People’s Hospital of Wenzhou Medical University (approval number: DRY-2024-YX-062).
Informed consent
Obtain informed consent from all subjects and/or their legal guardians in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Guo, X., Cheng, Y. & Du, J. Clinical value of D-dimer combined with transvaginal ultrasonography in the postmenopausal population with endometrial carcinoma. Sci Rep 15, 27447 (2025). https://doi.org/10.1038/s41598-025-12980-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-12980-x