Abstract
High-intensity focused ultrasound (HIFU) ablation is a minimally invasive treatment for benign gynecological diseases, though skin burns remain a potential complication. Understanding risk factors is critical for improving safety. In this study, we aimed to investigate the risk factors for skin burns in patients who underwent HIFU ablation. This retrospective study included 229 patients who underwent HIFU ablation for benign gynecological diseases. The medical records of the included patients were retrospectively analyzed to identify risk factors for skin burns. The main outcome measure was the identification of the risk factors contributing to skin burns following HIFU ablation. Among the 229 included patients, 6 experienced skin burns following HIFU ablation. Multivariate regression analysis identified several factors associated with the occurrence of skin burns, including treatment duration, sonication time, focusing depth, and water sac use (P < 0.05). The duration of treatment and degree of skin compression are key factors that contribute to skin burns (P < 0.05). Shortening treatment duration, incorporating intraoperative cooling intervals, and limiting both compression duration and intensity significantly reduce skin burn incidence and improve patient outcomes.
Similar content being viewed by others
Introduction
Uterine fibroids and adenomyosis are the most common benign uterine diseases in women and can lead to dysmenorrhea, menorrhagia, infertility, and other symptoms1,2,3. Surgical removal or radical hysterectomy is the most widely used treatment option4; however, surgical complications and the desire to preserve reproductive organs have prompted the medical community to search for a noninvasive, repeatable, safe, and effective treatment. High-intensity focused ultrasound (HIFU) was approved by the FDA for use in this field 20 years ago; however, it is not widely used and regarded by many as novel.
HIFU is an emerging noninvasive treatment with good tissue penetration ability that can induce coagulation necrosis in targeted lesions. This action is achieved through thermal and biological effects caused by the instant temperature increase from the energy generated by ultrasound waves focused on the target within the body5. By harnessing ultrasonic energy in vitro, HIFU can increase the instantaneous internal temperature of lesions in the body to over 60 °C to achieve lesion ablation. Previous studies have demonstrated the effectiveness of HIFU therapy6.
Although HIFU has been used for 20 years, its safety continues to attract attention7,8. Common complications associated with HIFU include skin heat injury, nerve injury, intestinal perforation, bladder perforation, and uterine perforation9. Skin heat injury is of particular concern because it has the highest incidence rate10. Abdominal wall scarring prior to HIFU treatment has been reported as a factor that impacts the occurrence of adverse events after HIFU treatment11; however, the relationship between skin scarring and skin heat damage remains a topic of debate. Therefore, we conducted a retrospective analysis of skin heat injury cases at our center to identify the leading risk factors for this injury.
Materials and methods
Patients
The protocol for this retrospective study was approved by the Ethics Clerk Association of Zhongshan City People’s Hospital (2024-057), and informed consent was obtained from each patient prior to surgery. All the methods described in this article were carried out in accordance with the relevant guidelines and regulations. Between June 2015 and January 2023, 685 patients were treated with HIFU; among them, 653 with uterine fibroids or adenomyosis were selected. Finally, 229 patients were included in the study (223 in the control group [group A] and 6 in the skin injury group [group B]) after screening based on the following criteria:
Inclusion criteria:
-
1.
Received HIFU treatment at our center between July 2015 and January 2023.
-
2.
The presence of deep solid tumors, such as uterine fibroids or adenomyosis.
-
3.
Treating physician has previous experience with burn complications.
Exclusion criteria:
-
1.
Did not undergo pre-HIFU magnetic resonance imaging (MRI).
HIFU ablation
HIFU treatment was performed using an ultrasound-guided Haifu system (JC200, Haifu Medical Technology Co., Ltd., Chongqing, China). Haifu Medical Technology operates at a frequency of 0.5–2 MHz, with an output energy above 400 W, focal sound above 10,000 W/cm2, and focusing depth of 50–200 mm. Before treatment, all patients were requested to sign the HIFU treatment consent form. Fentanyl and midazolam were administered to the patients before the procedure for analgesia and sedation, respectively. Patients were placed in a prone position on the HIFU treatment table to ensure that their skin was in full contact with degassed water, and a water balloon was placed between the transducer and the anterior abdominal wall to compress or push away the bowel. Treatment was initiated at the center of the lesion using point sonication. The sonication power ranged from 350 W to 400 W. During the procedure, the treatment area and sonication intensity were adjusted based on grayscale ultrasound changes and patient tolerance. During treatment, the patients were requested to lie still and report any discomfort, including burning of the skin or lower abdominal, leg, sciatic, or buttock pain12.
When patients develop skin burns, those with first- and second-degree burns receive follow-up treatment in the outpatient department—including treatment with burn ointment—and return to the hospital weekly to check the wound. Antibiotics are provided if necessary to prevent infection. Patients with deep second- or third-degree burns may require surgery to remove the burned skin.
Evaluation content
We analyzed the data of the two groups of patients based on the following factors: epidemiological factors (age and body mass index [BMI]), focusing depth, uterine flexion, thickness of the subcutaneous adipose tissue, presence of abdominal scars, and technical parameters of surgery (sonication time, ablation ratio, treatment time, total energy and use of water sacs). All imaging features were recorded during pretreatment MRI identification.
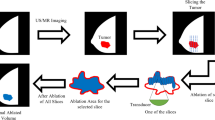
The focusing depth refers to the maximum depth of focus of the HIFU therapy instrument in the body (Figure 1–1). The distance between the deepest part of the lesion and the skin on the largest surface of the lesion in the MRI images was measured (Figure 1-2). Due to the need to prevent heat effects on the surrounding tissues during treatment, the focusing depth was 1 cm from the deepest depth of the lesion. Therefore, the maximum depth was calculated as the measured distance in Figure 1-2 minus 1 cm (Fig. 1).
Schematic diagram illustrating HIFU focusing depth(1–1), measurement of the distance between the deepest part of the lesion and the skin(1–2).
Temporal progression of third-degree burn healing in patient 1. Day 1: Cutaneous pallor and ischemic changes at treatment site. Day 4: Erythema with blistering at treatment site. Day 14: Complete resolution. Histopathology of excised skin specimen: Epidermal necrosis with dermal collagen coagulation, and thrombosed microvessels.
Sonication time refers to the exact ablation time of the lesion, measured in seconds. The ablation ratio is the ratio of ablation time (s) to cooling time (s). Sonication-to-cooling ratio is an instrument mode that is selected by the treating physician—multiple ratios are available. Option 1 represents the 1:3 mode, where the working mode of the therapeutic instrument is to output treatment energy for 1 s and stop for 3 s; option 2 represents the 1:2 mode, where the working mode is to output treatment energy for 1 s and stop for 2 s; and option 3 represents the 2:3 mode, where the working mode is to output treatment energy for 2 s and stop for 3 s. Treatment time refers to the time from the start to the end of ablation therapy measured in minutes. Total energy (J) refers to the total amount of ultrasound capacity used during the treatment.
Outcomes
The main outcome measure was to identify the risk factors that contribute to skin burns after high-intensity focused ultrasound ablation. These factors were thoroughly analyzed to help create a prevention plan for skin injury.
Statistical analysis
A generalized linear logistic regression model was used to evaluate the effects of various factors on skin burn occurrence. In univariate and multivariate analyses of the selected variables, the results are expressed as odds ratios and 95% confidence intervals. Statistical significance was set at P < 0.05. SPSSAU software (SPSS AU, Beijing Green Silk Technology Co., Ltd.) was used for the data analysis.
Results
A total of 229 patients were included in this study, of whom 6 had skin injuries and 4 had third-degree burns requiring dermal resection. The resulting damage is shown in Fig. 2. The other two patients had shallow second-degree burns that recovered after conservative treatment. The control group consisted of 223 patients. Regression analysis was performed for the two groups of patients using 10 variables. Table 1 shows the general profiles of each variable in the two patient groups and the results of the regression analysis.
The mean age of group A (control group) was 40.25 ± 6.30 years and that of group B (skin injury group) was 41.50 ± 5.17 years. The mean BMI of group A was 22.79 ± 3.43 kg/m2 and that of group B was 23.85 ± 3.65 kg/m2. The mean thickness of abdominal wall fat was 16.89 ± 8.01 mm in group A and 21.41 ± 8.98 mm in group B. The mean total energy was 255271.75 ± 174606.91 J in group A and 448266.67 ± 342760.06 J in group B. As shown in Table 1, there were no significant differences between the two groups in terms of age, BMI, total energy, and abdominal wall fat thickness.
The mean sonication time was 610.18 ± 363.37 s in group A and 1128.17 ± 872.94 s in group B. The mean treatment time was 66.79 ± 34.33 min in group A and 95.33 ± 54.36 min in group B. The mean focusing depth was 91.04 ± 20.68 mm in group A and 86.84 ± 26.90 mm in group B. According to the results presented in Table 1(Fig. 3), the influence of these three variables on the differences between the two groups was significant.
Multivariate regression analysis of skin burn predictors. With 95% confidence intervals (CI).
In terms of the ablation ratio, uterine flexion and abdominal scarring did not affect the outcomes of the two groups. Only the use of a water sac showed a significant effect in both groups.
Discussion
Treatment of skin burns
HIFU is widely used for the conservative treatment of benign gynecological diseases13. Several literature reviews have demonstrated the effectiveness and safety of this treatment14. However, a small number of patients still experience complications, with skin burn being common15,16,17.
According to Zanganeh et al.18, skin grafting is more effective than conservative treatment for deep skin burns above grade II. Additionally, for minor deep burns exceeding second-degree severity, surgical removal of necrotic skin is more effective19,20. Figure 2 shows the healing process in one patient with a third-degree burn. The burned area on the patient’s skin measured approximately 4 × 6 cm. Postoperative day 1 clinical imaging revealed concurrent thermal injury and mechanical compression injury at the probe-skin interface. For such extensive skin burns, the risk of infection and scarring associated with conservative treatment is high. Therefore, we performed an excision of the burned skin, a stage I skin lesion excision, and a suture 5 days after the burn. The sutures were removed smoothly 1 week after surgery, and the wound healed well. We performed the same procedure for the other three patients with third-degree burns, all of which healed well. The other two patients had second-degree burns that were approximately 1 cm in size. Conservative treatment was adopted, and the wounds healed well (Fig. 4).
Patients(2 and 3) with second-degree burns.
In our experience, skin burns after HIFU treatment are often associated with crush injuries from the ultrasonic probe and skin excision should be performed as soon as possible for burns in large areas (diameter greater than 2 cm). This treatment can reduce the incidence of skin infection and scarring.
Causes of skin burn
Although HIFU is not widely used, several studies have shown its safety21,22. However, no clear and unified definition for HIFU complications exists. According to a review published by Kociuba et al.23, complications of HIFU exposure include pain, such as abdominal, lower back, and leg pain; vaginal discharge; vaginal bleeding; skin burns; uterine infection; bladder perforation; and intestinal perforation. Among them, the incidences of pain, vaginal discharge, and vaginal bleeding were higher; however, these were minor adverse events. The incidences of skin burns, bladder perforation, and intestinal perforation are low; however, these are major adverse events24,25. Among the major adverse events, the incidence of skin burns was the highest.
As shown in Table 1, patient age, ablation ratio, BMI, uterine location, total energy, and abdominal fat thickness did not correlate with the occurrence of skin burns. The finding that age and BMI were not correlated is consistent with the results of the study by Kociuba et al.26, whereas other factors were not investigated in this study. It is also worth noting that this study found no correlation between the presence of scarring on the abdominal wall and occurrence of skin burns, which is contrary to the findings of Xiong et al.2. In this study, six patients had skin burns, of whom only three had abdominal scars. Xiong et al. noted that patients with skin scars were more likely to have skin burns; however, this factor is not necessarily related to the occurrence of skin burns.
According to the results of this study, sonication time, treatment time, focusing depth, and water sac use are related to the occurrence of skin burns. The longer the sonication and treatment times, the greater the skin’s exposure to ultrasound, which increases the likelihood of skin burns. According to analysis results, there is no correlation between total ultrasonic energy and occurrence of skin burns. It appears that skin burns are more likely to occur with longer treatment times using the same total ultrasonic energy; this suggests that the severity of skin burns is linked to the duration of skin squeezing. The Pennes bioheat equation establishes the fundamental relationship between thermal injury development and exposure parameters (power, duration, and energy deposition)27. However, contemporary research by Tasnim et al.28 demonstrates that under high-intensity focused ultrasound (HIFU) conditions, transient thermal wave behavior supersedes classical Pennes predictions. This non-Fourier heat transfer phenomenon is characterized by the thermal relaxation time (τ), where shorter τ values significantly increase skin damage susceptibility due to insufficient thermal diffusion.
Depth of focus is a key factor affecting the pressure exerted by the treatment probe on the skin. During treatment, patients with a deep focus must squeeze their skin by raising the probe to reach the required depth. Squeezing the skin for a long time can easily cause crush injuries29, which are the leading cause of severe burns in patients (Fig. 2). The patient in Fig. 2 is the only patient who did not use a water sac. The shape of the probe was clearly observed in the patient’s skin scars on the second day. Direct compression of the skin using an ultrasonic probe is a major cause of burns. A water sac placed between the therapeutic probe and the patient’s skin acts as a buffer, thereby reducing skin compression. This is consistent with the findings of this study that water sac use is associated with the development of skin burns. In addition, Liu et al.30 proposed inserting a Foley catheter into the rectum to assist in HIFU therapy, which could bring the compression lesion forward to reduce the focus, thereby reducing the occurrence of skin burns. Further prospective studies are required to investigate its feasibility.
Prevention of skin burn
Based on our study, we identified two modifiable risk factors—treatment duration and skin compression—and propose corresponding prevention strategies. First, for deep-seated lesions, mandatory water sac utilization is essential to distribute interfacial pressure, combined with periodic probe elevation at clinically appropriate intervals to restore perfusion and facilitate heat dissipation. Second, implement a fractionated ablation protocol for extensive deep lesions. Third, leverage preoperative interventions: 3-month neoadjuvant therapy with GnRH agonists/mifepristone achieves lesion volume reduction prior to HIFU, thereby reducing complication rates without compromising therapeutic efficacy.
Limitations
This study has several limitations that warrant consideration. As a retrospective analysis, potential selection bias may exist despite consecutive patient enrollment. Additionally, incomplete documentation in medical records (e.g., unstandardized pressure measurements) constrained mechanistic analysis. The small burn cohort (*n*=6) reflects the low incidence of this complication but reduces statistical power; nevertheless, multivariate regression demonstrated robust significance (F = 6.750, P = 0.000)31,32. Future prospective studies should:
-
1.
Implement standardized pressure monitoring.
-
2.
Include thermographic mapping.
-
3.
Enlarge cohorts through multi-center collaboration to validate our risk mitigation framework.
Conclusions
Multivariate analysis assessed epidemiological factors (age and body mass index [BMI]), focusing depth, uterine flexion, thickness of the subcutaneous adipose tissue, presence of abdominal scars, and technical parameters (sonication time, ablation ratio, treatment time, total energy, and water sac use). Critically, only treatment duration and water sac omission demonstrated statistically significant associations with skin injury; all other variables showed non-significant associations (P > 0.05).
Treatment duration and degree of skin compression are key factors contributing to skin burns. Prolonged treatment duration and increased skin compression (both duration and intensity) significantly elevate the risk of thermal injury. Clinically, we recommend: (1) Fractional ablation for lesions too large for single-session treatment; (2) Combined with periodic probe elevation at clinically appropriate intervals to restore perfusion and facilitate heat dissipation. These evidence-based protocols optimize safety while retaining HIFU’s non-invasive benefits for benign gynecological diseases.
Data availability
The data supporting the findings of this study are available upon request from the corresponding author. The data are not publicly available because of privacy or ethical restrictions.
References
Stewart, E. A., Cookson, C. L., Gandolfo, R. A. & Schulze-Rath, R. Epidemiology of uterine fibroids: a systematic review. BJOG 124, 1501–1512. https://doi.org/10.1111/1471-0528.14640 (2017).
Stewart, E. A. et al. Uterine fibroids. Nat. Rev. Dis. Primers. 2, 16043. https://doi.org/10.1038/nrdp.2016.43 (2016).
Taran, F. A., Stewart, E. A., Brucker, S. & Adenomyosis Epidemiology, risk factors, clinical phenotype and surgical and interventional alternatives to hysterectomy. Geburtshilfe Frauenheilkd. 73, 92431. https://doi.org/10.1055/s-0033-1350840 (2013).
Shrestha, A., Shrestha, R., Sedhai, L. B. & Pandit, U. Adenomyosis at hysterectomy: prevalence, patient characteristics, clinical profile and histopathological findings. Kathmandu Univ. Med. J. (KUMJ). 10, 53–56. https://doi.org/10.3126/kumj.v10i1.6915 (2012).
Tsai, M. C., Chang, L. T. & Tam, K. W. Comparison of high-intensity focused ultrasound and conventional surgery for patients with uterine myomas: a systematic review and meta-analysis. J. Minim. Invasive Gynecol. 28, 1712–1724. https://doi.org/10.1016/j.jmig.2021.06.002 (2021).
Cheung, V. Y. T. High-intensity focused ultrasound therapy. Best Pract. Res. Clin. Obstet. Gynaecol. 46, 74–83. https://doi.org/10.1016/j.bpobgyn.2017.09.002 (2018).
Ji, Y. et al. High-intensity focused ultrasound (HIFU) treatment for uterine fibroids: a meta-analysis. Arch. Gynecol. Obstet. 296, 1181–1188. https://doi.org/10.1007/s00404-017-4548-9 (2017).
Zhou, C. Y., Xu, X. J. & He, J. Pregnancy outcomes and symptom improvement of patients with adenomyosis treated with high intensity focused ultrasound ablation (in Chinese). Zhonghua Fu Chan Ke Za Zhi. 51, 845–849. https://doi.org/10.3760/cma.j.issn.0529-567X.2016.11.009 (2016).
Chen, J. et al. Safety of ultrasound-guided ultrasound ablation for uterine fibroids and adenomyosis: a review of 9988 cases. Ultrason. Sonochem. 27, 671–676. https://doi.org/10.1016/j.ultsonch.2015.05.031 (2015).
Lee, J. Y. et al. Portable ultrasound-guided high-intensity focused ultrasound with functions for safe and rapid ablation: prospective clinical trial for uterine fibroids-short-term and long-term results. Eur. Radiol. 30, 1554–1563. https://doi.org/10.1007/s00330-019-06468-2 (2020).
Yin, N. et al. Factors influencing thermal injury to skin and abdominal wall structures in HIFU ablation of uterine fibroids. Int. J. Hyperth. 34, 1298–1303. https://doi.org/10.1080/02656736.2018.1433880 (2018).
Yan, L., Huang, H., Lin, J. & Yu, R. High-intensity focused ultrasound treatment for symptomatic uterine fibroids: a systematic review and meta-analysis. Int. J. Hyperth. 39, 230–238. https://doi.org/10.1080/02656736.2022.2029956 (2022).
Marques, A. L. S., Andres, M. P., Kho, R. M. & Abrão, M. S. Is high-intensity focused ultrasound effective for the treatment of adenomyosis? A systematic review and meta-analysis. J. Minim. Invasive Gynecol. 27, 332–343. https://doi.org/10.1016/j.jmig.2019.07.029 (2020).
Izadifar, Z., Izadifar, Z., Chapman, D. & Babyn, P. An introduction to high intensity focused ultrasound: systematic review on principles, devices, and clinical applications. J. Clin. Med. 9, 460. https://doi.org/10.3390/jcm9020460 (2020).
Xiong, Y. et al. Ultrasound-guided high-intensity focused ultrasound (USgHIFU) ablation for the treatment of patients with adenomyosis and prior abdominal surgical scars: a retrospective study. Int. J. Hyperth. 31, 777–783. https://doi.org/10.3109/02656736.2015.1071436 (2015).
Leon-Villapalos, J., Kaniorou-Larai, M. & Dziewulski, P. Full thickness abdominal burn following magnetic resonance guided focused ultrasound therapy. Burns 31, 1054–1055. https://doi.org/10.1016/j.burns.2005.04.019 (2005).
Liu, X. et al. Clinical predictors of long-term success in ultrasound-guided high-intensity focused ultrasound ablation treatment for adenomyosis: a retrospective study. Med. (Baltim). 95, e2443. https://doi.org/10.1097/MD.0000000000002443 (2016).
Zanganeh, M. et al. Skin grafting compared with Conservative treatment in patients with deep second-degree burn wounds of the trunk and buttocks. World J. Plast. Surg. 11, 81–85. https://doi.org/10.52547/wjps.11.1.81 (2022).
Elfawy, L. A. et al. Sustainable approach of functional biomaterials-tissue engineering for skin burn treatment: a comprehensive review. Pharmaceuticals 16, 701. https://doi.org/10.3390/ph16050701 (2023).
Tammam, B. M. H., Habotta, O. A., El-Khadragy, M., Abdel Moneim, A. E. & Abdalla, M. S. Therapeutic role of mesenchymal stem cells and platelet-rich plasma on skin burn healing and rejuvenation: a focus on Scar regulation, oxido-inflammatory stress and apoptotic mechanisms. Heliyon 9, e19452. https://doi.org/10.1016/j.heliyon.2023.e19452 (2023).
Verpalen, I. M. et al. The focused ultrasound Myoma outcome study (FUMOS); a retrospective cohort study on long-term outcomes of MR-HIFU therapy. Eur. Radiol. 30, 2473–2482. https://doi.org/10.1007/s00330-019-06641-7 (2020).
Quinn, S. D., Vedelago, J., Gedroyc, W. & Regan, L. Safety and five-year re-intervention following magnetic resonance-guided focused ultrasound (MRgFUS) for uterine fibroids. Eur. J. Obstet. Gynecol. Reprod. Biol. 182, 247–251. https://doi.org/10.1016/j.ejogrb.2014.09.039 (2014).
Kociuba, J. et al. Adverse events and complications after magnetic resonance-guided focused ultrasound (MRgFUS) therapy in uterine fibroids - a systematic review and future perspectives. Int. J. Hyperth. 40, 2174274. https://doi.org/10.1080/02656736.2023.2174274 (2023).
Khalilzadeh, O. et al. Proposal of a new adverse event classification by the society of interventional radiology standards of practice committee. J. Vasc Interv Radiol. 28, 1432–37e3. https://doi.org/10.1016/j.jvir.2017.06.019 (2017).
Zhang, X. et al. Effective ablation therapy of adenomyosis with ultrasound-guided high-intensity focused ultrasound. Int. J. Gynaecol. Obstet. 124, 207–211. https://doi.org/10.1016/j.ijgo.2013.08.022 (2014).
Kociuba, J. et al. Occurrence of adverse events after magnetic resonance-guided high-intensity focused ultrasound (MR-HIFU) therapy in symptomatic uterine fibroids-a retrospective case-control study. Int. J. Hyperth. 40, 2219436. https://doi.org/10.1080/02656736.2023.2219436 (2023).
Mehtali, J., Verde, J. & Essert, C. Heat: high-efficiency simulation for thermal ablation therapy. Int. J. CARS. 20, 1135–1143. https://doi.org/10.1007/s11548-025-03350-z (2025).
Tasnim, Z. J. & Nasrin, R. Thermal wave and pennes’ models of bioheat transfer in human skin: A transient comparative analysis. Heliyon 10 (21), e40109. https://doi.org/10.1016/j.heliyon.2024.e40109 (2024).
Sagi, A. et al. Combined thermal and crush injury to the hand and fingers. Burns 23, 176–181. https://doi.org/10.1016/s0305-4179(96)00116-7 (1997).
Liu, W. et al. Intrarectal Foley catheter-assisted high-intensity focused ultrasound ablation for benign uterine diseases beyond the treatment region. Int. J. Hyperth. 40, 2219435. https://doi.org/10.1080/02656736.2023.2219435 (2023).
Dao-de, S. Selection of the linear regression model according to the parameter Estimation. Wuhan Univ. J. Nat. Sci. 5, 400–405. https://doi.org/10.1007/BF02850764 (2000).
Schneider, A., Hommel, G. & Blettner, M. Linear regression analysis: part 14 of a series on evaluation of scientific publications. Dtsch. Arztebl Int. 107, 776–782. https://doi.org/10.3238/arztebl.2010.0776 (2010).
Acknowledgements
We thank Professor Su Yuanyuan for providing financial support.
Funding
This research did not receive any specific grants from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
WS: Conceptualization, methodology, data curation, writing—original draft preparation. HY: Writing—review and editing. LJ and YL performed HIFU. TY: Software validation.All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wu, S., Liu, J., Tang, Y. et al. Skin burns after high-intensity focused ultrasound ablation: a retrospective control study. Sci Rep 15, 28127 (2025). https://doi.org/10.1038/s41598-025-13436-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-13436-y