Abstract
Honey bee colonies adapt their foraging behaviours to the availability of floral resources to meet their nutritional needs. However, it is unknown if the nutritional quality of stored or supplemented pollen can influence the floral choices of bees during commercial crop pollination. The foraging behaviour of bees from 40 colonies was studied during avocado pollination in southern Western Australia. A pollen database of the orchard was built and used to assess the floral preference of the bees. Pollen collectors and nectar foragers showed different foraging behaviour as indicated by their Dominance Candidate Index (DCI). The foraging choices were partially affected by the type of supplemented pollen that consisted of agricultural and forest species. Aside from nutritional cues, floral source abundance and attractiveness played a role in influencing the foraging behaviour for pollen and nectar. Both pollen and nectar foragers chose a sub-set of flowers available at the avocado orchard. Foraging bees respond strongly to various environmental cues that potentially underlie energetic investment and food reward principals.
Similar content being viewed by others
Introduction
Honey bee colonies are continually trying to optimise their nutritional requirements with resources available from the environment. The quality and quantity of pollen and nectar play a crucial role in their survival. Bees gather nectar to meet their needs for carbohydrates, and pollen to meet their need for protein, fat, vitamins and minerals1. The availability and the seasonality of flower resource influences the gathering of pollen and nectar, and affects colony growth, reproduction and behabiour2.
The quantity and quality of stored resources and feedback from nestmates provide information to foragers about the nutritional status of the colony and foraging is adjusted accordingly. For example, low pollen stores inside colonies stimulate bees to forage for more pollen3. Nectar collection on the other hand seems to be largely regulated by floral nectar availability1,4. Cues about food availability are also provided during trophallactic food exchange between colony members whereby the quality and quantity of food existing in the hive is communicated and passed on to other foragers through pheromones and dance language5,6.
The type of flower that bees select to collect pollen and/ or nectar from depends on various floral cues such as visual stimuli (morphology and colour)7,8 odour9,10 floral source abundance11 and the distance from the hive12,13. Recent studies indicate that honey bees can detect the nutritional quality of food sources and may be able to selectively forage for missing nutrients14,15,16. However, to what degree honey bees can modify their foraging behaviour based on nutritional quality remains largely unknown.
In commercial settings, where honey bees are used for crop pollination, the availability and quality of floral resources are largely dictated by colony stores from previous sites, floral resources provided by the crop and potentially other resources available in forage distance surrounding the orchard. While honey bees need to be able to rapidly adjust to changes in their environment to survive, it is unknown to what extent the foraging behaviour of honey bees is influenced by floral cues and by the nutritional quality of stored resources when shifted to a different location.
One such example is the pollination of avocado crops in Western Australia (WA). During the winter months beekeepers use apiary sites along the coastal heathlands north of Perth, or nearby canola crops, to build up the colonies’ strength in preparation for pollination services. The honey and pollen stores from these sites, may vary in floral origin, composition and nutritional quality. If pollen is in short supply to grow colonies to full strength, beekeepers may need to supplementary feed colonies before commercial pollination events. Supplementary feed may be provided by feeding irradiated bee-collected pollen such as that from marri (Corymbia calophylla), as practised in WA or other commercial artificial feeds17,18. Excess amounts of honey may be extracted before providing hives for avocado pollination services to stimulate bees to preferentially collect nectar, supporting the successful cross-pollination of avocado flowers19.
Here, we hypothesised that honey bees would display different foraging behaviour depending on the quality and floral diversity of the food stores obtained from previous sites after they were shifted to an agricultural setting for pollination services in an avocado orchard. The effect of four different feed sources on the foraging behaviour of bees collecting pollen and bees collecting nectar was investigated. Nectar collectors directly influence the pollination of the avocado crop, and pollen collection by honey bees is crucial in supporting the nutritional needs of colonies whilst in the avocado orchard. A plant and pollen database of the orchard was built and used to assess the preference of honey bees for different floral sources.
Methodology
Experimental design
The methods for colony setup and feed source preparation are described in detail in Kratz et al.20. Briefly, 40 bee packages (800 g of bees) were obtained from a commercial beekeeper on a canola (Brassica napus) site near Geraldton 6530 (Western Australia). Colonies were then established at a site near Oldbury 6121 (Western Australia) on four feed treatments (marri, jarrah, clover and canola) over six weeks before being moved to an avocado orchard near Busselton 6280 (Western Australia) for a further 6-week period. Avocado pollination is unique because each flower opens twice, first as a functional female and the second time as a functional male, separated by one overnight period, known as synchronous dichogamy21,22. For successful cross-pollination honey bees need to visit both types of flowers during a small overlap of 1–3 h a day, either in the morning or afternoon19. Nectar collectors are crucial for avocado pollination as they visit both male and female flowers.
The 40 experimental hives were placed in the centre of the 221.9 ha avocado orchard near Busselton (WA); 0.4–0.6 km from the edge of the orchard north to east and 0.7–1.3 km south to west (Fig. S1). The orchard was mainly bound by farmland with a stretch of bushland traversing the centre and a national park located within flying distance to the south of the orchard. The hives assigned to the various feed treatments were randomly organised in a single row separated by 1.5 m.
Four different feed sources (150 g patties) consisting of both honey and pollen were prepared. Each feed source was given to 10 colonies, replaced weekly and feed consumption was measured. Two of the feed sources, Brassica napus (canola) and Trifolium repens (white clover), were naturally available to honey bees during the winter months near a coastal heathland site prior to avocado pollination. The other two feed sources were from Corymbia calophylla (marri) and Eucalyptus marginata (jarrah), both forest species endemic to WA. Marri is often offered as a supplementary pollen by commercial beekeepers during pollen shortages because of its recognised high protein content.
The first cycle of brood was reared on the assigned feed source prior to being placed at the orchard. To be able to test the effects of assigned feed treatments a pollen trap was mounted to each colony to restrict bees from using pollen they collected from their environment throughout the experimental period (Fig. S2).
Floral diversity was analysed by identifying the pollen in the food patties, the corbicular pollen (pollen baskets) of pollen collectors and the pollen on nectar foragers. The corbicular pollen and the pollen on bodies of nectar foragers was analysed weekly over the peak flowering period from the end of October to early November (weeks 1–3) during avocado pollination.
Floral diversity of feed sources
To identify and quantify the pollen types in the patties, approximately 3 g of each of the prepared pollen patties (patty preparation is described in detail in Kratz et al.20, were placed into 50mL centrifuge tubes. Warm (~ 40 °C) distilled water (20–30mL) was added to the tubes, and the tubes were agitated until the pollen was dispersed. The samples were then sieved into a small flask through a 120 μm nylon mesh to remove extraneous material (e.g. bee parts) and returned to the 50 mL tubes. Samples were rinsed–centrifuged in hot distilled water several times, the supernatant discarded, and residue retained. The residues were then acetolysed as per Erdtman (1960)23 and slides prepared as follows. Approximately 5mL of water was added to the acetolysed pellet, and a small drop (approx. 0.1mL) of the mixture was pipetted into a weak solution of polyvinyl acetate (PVA) on a glass microscope coverslip. When dry, the coverslip was inverted onto a small drop of the permanent mounting medium Eukitt© on a glass microscope slide and left to dry for several days.
Slides were examined and pollen photographed using a transmitted light biological microscope (OLYMPUS-BX51, Olympus Corporation) with a digital camera attachment (OLYMPUS-P71, Olympus Corporation). At least 200 grains were counted and identified24. The contrast and background of photomicrographs was adjusted in Photoshop (Adobe) which did not change the pollen structure. All pollen grains were identified to the species level by comparing pollen types with the personal pollen databases of Dr Lynne Milne, pollen collected from the field and the Western Australian Herbarium. Pollen grains that could not be identified to the species level were reported to the family or genus level.
Pollen percentages were calculated from the total pollen counts for each individual sample. A dominant floral source was classified as dominant when pollen abundance was greater than 45%25,26.
Sample collection
Collection of pollen from traps
Corbicular pollen was collected weekly from pollen traps mounted at the colony entrances, stored at – 20 °C and processed at a later date in the laboratory. The pollen samples were cleaned of any debris, dried at 60 °C for 24 h (until no weight change was observed) and weighed.
The weight of dry pollen collected for each colony was expressed as a daily average (g/day), as the number of days between sampling times varied slightly between weeks.
To take into account the influence of colony size on the amount of pollen collected by bees, brood count of individual colonies was used as an estimate of colony size. Colony size was determined by the number of brood cells per colony every second week from digital photographs and counted using the desktop software ”Beestly” (available from www.cyency.com). The amount of dry pollen (g/day) was then expressed as per 100 brood cells (g/day/100 brood cells).
Collection of nectar collectors
Five nectar foraging bees were collected at the entrance of each colony in both the morning (10am − 12pm) and the afternoon (2–5pm). Individual colonies were sampled randomly each week to minimise any bias in the order of sampling by generating a random list of colony numbers. Each bee was placed into a 5 mL tube containing 3 mL of 70% ethanol using tweezers (washed between sample collection). Foraging honey bees returning to hives that carried corbicular pollen pellets were not collected. A total of 400 bees were collected each week (five bees in the morning and five bees in the afternoon from each of the 40 colonies).
Database for reference pollen
Samples of potential target plants of foraging honey bees were collected throughout the pollination period to build a pollen reference database. Permission to collect plant samples was granted from the owner of the avocado orchard. Plant samples including flowers were collected from the immediate proximity of the colonies and from within the orchard by walking along rows of avocado trees, and by driving around the border of the property. A total of 25 specimens were collected and dried by pressing them between sheets of paper. Plant specimens were identified by a botanist at the Western Australian Herbarium and represented 16 families. If there was not enough pollen from a collected plant specimen additional pollen was obtained from the Western Australian Herbarium collection. The pollen of all plants was acetolysed and permanent slides were prepared. Pollen from all species was photographed and morphologically differentiated by Dr L. Milne to form a pollen reference database (Table S1, 2 and Plate S1-8). Pollen from Corymbia calophylla (marri) and Eucalyptus marginata (jarrah) were identified according to an existing pollen database of Dr L. Milne, no plant specimens were collected. No voucher specimens were deposited at the Australian Herbarium from this study.
Pollen identification
Different methods were used for the identification of pollen sampled from the corbicular basket and from the nectar collectors. Corbicular pollen was acetolysed following Erdtman23 for ease of pollen identification to the species level. Avocado pollen, unlike pollen of other plant families, has an exine consisting only of a thin coherent layer ornamented with spinules that is destroyed by acetolysis27,28. The destruction of avocado pollen was not of concern because only minimal amounts of avocado pollen (< 2%) was found in fresh pollen samples and, bees that only collect pollen usually do not contribute to the pollination of avocado flowers29.
Pollen of nectar collectors was identified from fresh pollen samples to keep avocado pollen intact, as nectar collectors perform the pollination of avocado flowers and it was of interest to quantify the amount of avocado pollen these bees carried on their bodies.
Any pollen that could not be identified to species level was instead identified to the family or genus level (e.g. ‘Myrtaceae spp and Eucalyptus spp) or placed in a morphological type (e.g. tricolporate).
Identification of corbicular pollen
A subsample of 2 g of fresh pollen was used for the palynology of corbicular pollen collected from traps. The samples were prepared as described for the floral diversity of the patties and identified from the reference samples collected from the orchard. Pollen that did not match any of the reference samples was identified from the database of Dr L. Milne. Very few pollen grains (< 0.5%) that could not be identified were classified as ‘Not formally identified’ (NFI) and were not included in the dataset.
Identification of pollen from nectar foragers
One drop of Triton X - detergent was added to each of the 1200 5 mL tubes containing nectar collectors in ethanol and were vortexed for 90 s to wash the pollen from individual bees30. The liquid content of the 5 mL tubes was poured into 15mL centrifuge tubes. The 5 mL tube and the bee were then rinsed twice with 1mL of 70% ethanol into the 15mL centrifuge tube to remove any remaining pollen.
The 15mL centrifuge tubes were then centrifuged at 3000 rpm for 10 min. The supernatant was aspirated. The remaining ethanol was removed without any pollen loss using a stream of air gently blown over the samples for approximately 30 min until the samples were dry.
The pollen pellet was resuspended in 30µL of distilled water and vortexed for 30s. A 10 µL subsample was immediately placed into a Kova glasstic slide haemocytometer (Hycor Biomedical Inc., California, USA). The number of pollen grains was counted in all grids representing a volume of 0.9µL31 under 400 x magnification using a light microscope (Olympus BH-2, Japan). The total number of grains per bee were determined by multiplying the number of pollen grains counted on the haemocytometer by a dilution factor of 33.3.
Pollen types representing more than 5% of the total pollen across the feed sources were identified to the family and/or genus/species level by comparison with fresh pollen samples collected from other plants in the avocado orchard. Avocado pollen was collected from flowers in the orchard.
No pollen grains were detected in three samples from the nectar foraging bees: two from week 1 of the jarrah feed source in the afternoon and the canola feed source in the morning, and one bee from week 2 of the clover feed. Those samples were removed from the dataset as the bees may have been water-collecting bees. One morning sample from week 3 of the clover feed source was lost during sample preparation.
Data visualisation and classification
Pollen types that represented more than 5% of the total pollen collected were considered to contribute to the nutrition of honey bees and were statistically analysed (Fig. 1).
Photomicrographs of prominent pollen types (> 5%) of reference flowers identified to the species level. (A–D) Persea americana (avocado) not acetolysed. (A, B) same grain, high and median focus, (C) median focus, (D) larger hydrated grain. (E, F) Hypochaeris radicata, high and median focus respectively. (G, H) Arctotheca calendula, median and high focus respectively. (I, J) Eucalyptus marginata (jarrah) showing range of morphology. (K–M) Trifolium repens (clover). (K, L) median and high focus of equatorial view, (M) high focus of polar view. Scale bars 10 μm (= magnification x 1000).
The “Dominance Candidate Index (DCi)”32 was calculated for each sample according to the following equation: DCi = (average relative abundance + relative frequency)/2.
The DCi takes into account the relative abundance (%) as well as the frequency (count) at which a specific pollen type was collected by bees, providing a DCi value between 0 and 1. The use of frequency rather than relative frequency means that both the amount of pollen collected and the number of bees collecting a particular pollen type are accounted for, and therefore DCi gives a more accurate representation of the foraging behaviour of the colony. For foragers collecting corbicular pollen, the DCi was calculated at the colony level for each feed source, with the relative abundance of individual species being obtained from a collective pollen sample of the whole colony (obtained from pollen traps). The frequency of a pollen type was defined by the number of times this pollen type was collected by each colony (10 per feed source). The frequency of pollen on nectar foragers was calculated for each individual bee, from five replicate bees collected for each of the time periods (morning and afternoon).
Statistical analysis
All statistical analyses were performed in R, version 3.6.233. All datasets were checked for normality and equal distribution of variance, and analysed by Linear Mixed Effects Models using the package “nlme”33. Feed Source and Week were included as fixed factors in each model. Colony (hive) was included as a random factor. Week was included as a random factor and nested within Colony to account for the repeated sampling of bee-collected pollen from the same colonies.
Model: lme (Y) <- Feed Source x Week, random = ~ 1|Colony/Week, data = XX
Patty consumption adjusted by colony size, reported in Kratz et al.20 was included as a covariate in the statistical analysis of the quantity of corbicular pollen (not adjusted for colony size). The model was applied to the peak flowering period at the avocado orchard (weeks 1–3). A square root transformation was used to normalise the data. Only significant interaction terms were included in the model.
One pollen sample, for the analysis of the pollen diversity of corbicular pollen, from the clover feed source in week 3 was too small to analyse for its diversity and was not included in the results.
For the pollen diversity of nectar foragers and total pollen count, Time of day (morning or afternoon) were included as fixed factors in the model. The dataset for the pollen count on nectar foragers was log transformed to achieve a normal distribution.
All data are presented as mean ± sem and only significant interaction terms p < 0.05% are reported. A post-hoc using Tukey’s pairwise comparisons were used to test for within factor effects from the R package ‘lsmeans’ and are reported in the supplementary material.
Results
Pollen diversity
Feed sources
Across all four patty types 16 pollen types were present, two of which have not been formally identified. The other pollen types belonged to eight different plant families: Amaranthaceae, Asteraceae, Brassicaceae, Casuarinaceae, Fabaceae, Myrtaceae, Poaceae and Proteaceae (Table 1). Fabaceae and Myrtaceae were the most represented plant families.
The floral contribution of the target species (marri, jarrah, clover and canola) in the patties ranged between 29.4 and 93.0% (Table 1), with clover being the most represented (93.0%), followed by canola (88.4%), marri (71.2%), and Jarrah being the least represented (29.4%). Only one or two floral species made up between 90.6% and 99.2% of the pollen types in any of the patties (Table 1).
Corbicular pollen
Pollen types present at more than 5% comprised 90.2–97.6% of the total pollen counted on the pollen foraging bees and represented five pollen types across the different feed sources. Three plant families, Asteraceae (species: Arctotheca calendula and Hypochaeris radicata), Fabaceae (species: Trifolium repens) and Myrtaceae (species: Eucalyptus marginata and Myrtaceae spp), were dominant across all feed sources (Fig. 1). Avocado pollen was one of the least represented pollen types (< 0.5% of the total pollen collected) across feed sources (marri, jarrah, clover and canola) and not present at the beginning of the peak flowering period (week 1). Unknown pollen types represented 1.15–2.12% across feed sources and weeks.
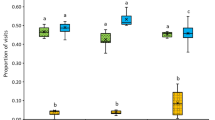
There was an interaction between Species and Feed source (F = 16.6, df = 12, p < 0.0001), Species and Week (F = 161.3, df = 8, p < 0.0001), and Feed source and Week (F = 5.4, df = 6, p < 0.0001), as well as a main effect of Species (F = 1513.8, df = 4, p < 0.0001 ), Food source (F = 14.0, df = 3, p < 0.0001) and Week (F = 13.7, df = 2, p < 0.0001) on the DCi of corbicular pollen. The DCi of T. repens (clover) was significantly greater compared to A. calendula, E. marginata, H. radicata and Myrtaceae spp, representing the dominant pollen type for relative abundance and frequency of collection. (Fig. 2A, Table S3). Arctotheca calendula and Myrtaceae spp had the second highest DCis, and were about half the DCi of T. repens, followed by E. marginata and H. radicata (Fig. 2A, Table S3), which were about 55% and 65% smaller than the DCi of T. repens respectively.
The dominance candidate index (DCi) of pollen species of (A) corbicular pollen and (B) pollen found on nectar collecting honey bees. Ac: Arctotheca calendula, Hr: Hypochaeris radicata, Em: Eucalyptus marginata, Mspp: Myrtaceae spp, Tr: Trifolium repens, Tspp: Trifolium spp and Pa: Persea americana. Crosses indicate the mean, bars indicate the median and the whiskers show the minimum and maximum values. Different letters show a significant difference at p < 0.05 from Tukey’s Comparison of Means.
Nectar foragers
Pollen types present at more than 5% comprised 80.0–96.4% of the total pollen counted on nectar foraging bees. The pollen grains represented four plant families: Asteraceae (Arctotheca calendula and Hypochaeris radicata), Fabaceae (Trifolium repens and Trifolium spp), Lauraceae (Persea americana) and Myrtaceae (Eucalyptus marginata and Myrtaceae spp).
There was a significant interaction between Species and Week (F = 210.3, df = 10, p < 0.0001), Species and Feed source (F = 6, df = 15, p < 0.0001), Species and Time of day (F = 91.3, df = 5, p < 0.0001), and Time of day and Week (F = 10.3, df = 2, p < 0.0001) on the DCi. There was a main effect of Species (F = 1448.8, df = 5, p < 0.0001) and Week (F = 18.6, df = 2, p < 0.0001), but not for Feed source (F = 1.1, df = 3, p = 0.37) or Time of day (F = 0.02, df = 1, p = 0.88) on the DCi of nectar foragers (Fig. 2B). The DCis of A. calendula, Myrtaceae spp and P. americana were similar between each other and greater compared to T. repens, Trifolium spp and H. radicata, which also differ significantly between each other (Fig. 2B, Table S8).
Abundance of individual pollen types
Trifolium repens (clover)
Corbicular pollen
There was no interaction between Feed source and Week (F = 1.038, df = 6, p = 0.41) and no main effect of Feed source (F = 1.45, df = 3, p = 0.24) on the DCi of T. repens, but there was an effect of Week (F = 4.6, df = 2, p = 0.013 (Fig. 3A). The DCi was lowest in week 1 compared to week 2 and week 3 (Fig. 3B, Table S5).
The dominance candidate index (DCi) of plant species visited by pollen collectors from colonies (N = 40) fed on marri, jarrah, clover and canola feed sources over a three week period (1–3). Plant species include: (A, B) Trifolium repens; (C, D) Myrtaceae spp; (E, F) Arctotheca calendula; and (G, H) Hypochaeris radicata. Crosses indicate the mean, bars indicate the median and the whiskers show the minimum and maximum values. Different letters show a significant difference at p < 0.05 from Tukey’s Comparison of Means.
Nectar foragers
There was an interaction between Feed source and Time of day (F = 11.3, df = 3, p < 0.0001) and between Time of the day and Week (F = 3.1, df = 2, p = 0.026), but not between Feed source and Week (F = 0.34, df = 6, p = 0.91) on the DCi (Fig. 2) on the DCi of nectar foragers. There was a main effect of Feed source (F = 3.2, df = 3, p = 0.036) and Week (F = 34.7, df = 2, p < 0.0001) but not for Time of day (F = 3.1, df = 1, p = 0.81) on the DCi of nectar foragers. Bees fed on the clover feed source collected significantly less T. repens pollen compared to bees fed on jarrah, and marginally less compared to bees fed on marri, but not compared to those on the canola feed (Fig. 4A, Table S9). The DCi in week 3 was lower compared to week 1 and week 2 (Fig. 4B, Table S10). Time of day had no effect on the DCi (Fig. 4C).
The dominance candidate index (DCi) of pollen on bodies of nectar foragers from colonies (N = 40) fed on marri, jarrah, clover and canola feed sources over a three-week period (1–3). Foragers were collected in the morning (am) and afternoon (pm). Plant species include: (A–C) Trifolium repens; (D–F) Myrtaceae spp; (G–I) Arctotheca calendula; and (J–L) Hypochaeris radicata. Crosses indicate the mean, bars indicate the median and the whiskers show the minimum and maximum values. Different letters show a significant difference at p < 0.05 from Tukey’s Comparison of Means.
Myrtaceae spp
Corbicular pollen
There was an interaction between Feed source and Week (F = 23.6, df = 6, p < 0.0001) and a main effect of Feed source (F = 15.5, df = 3, p < 0.0001) and Week (F = 265.8, df = 2, p < 0.0001) on the DCi of ‘Myrtaceae spp’. Colonies fed on marri and canola had similar DCis, which were greater than for both jarrah and clover feeds (Fig. 3C, Table S4). The DCis of jarrah and clover did not differ from each other. The DCi significantly decreased from weeks 1–2 and week 2–3 (Fig. 3D, Table S5).
Nectar foragers
There was a significant Time of day (F = 4.0, df = 1, p = 0.046) and Week (F = 23.9, df = 2, p < 0.0001) effect, as well as an interaction between Feed source and Time of day (F = 11.6, df = 3, p < 0.0001) and Time of day and Week on the DCi (F = 14.6, df = 2, p < 0.0001). There was no effect of Feed source (F = 0.24, df = 3, p = 0.87) on the DCi of unidentifiable Myrtaceae species grouped into ‘Myrtaceae spp’ (Fig. 4D). The DCi of Myrtaceae spp was greater in the afternoon compared to the morning (Fig. 4F, Table S11). The DCi of week 1 was greater compared to week 2 and 3 (Fig. 4E, Table S10).
Arctotheca calendula (capeweed)
Corbicular pollen
There was an interaction between Feed source and Week (F = 146.6, df = 6, p < 0.0001) and a main effect of Feed source (F = 88.7, df = 3, p < 0.0001) and Week (F = 134.3, df = 2, p < 0.0001) on the DCi of A. calendula. The DCi of colonies fed on canola and jarrah were greater compared to those fed on marri (Fig. 3E, Table S4). The DCi of canola and jarrah were also greater compared to those fed on clover (Fig. 3E, Table S4). The DCi of colonies fed on marri and clover were similar and did not significantly differ from each other). (Fig. 3E). The DCi of A. calendula increased significantly in week 3 compared to weeks 1 and 2 (Fig. 3F, Table S5).
Nectar foragers
There was a significant interaction between Feed source and Time of day (F = 17.5, df = 3, p < 0.0001) and a Time of day and Week interaction in the DCi for A. calendula (F = 14.2, df = 2, p < 0.0001). There was no effect of Feed source (F = 1.3, df = 3, p = 0.3) and Week (F = 0.3, df = 2, p = 0.73) on the DCi for A. calendula (Fig. 4G,H). There was an effect of Time of day (F = 259.9, df = 1, p < 0.0001) with the DCi significantly greater in the mornings for bees collecting nectar (Fig. 4I, Table S11).
Hypochaeris radicata (flatweed)
Corbicular pollen
There was an interaction between Feed source and Week (F = 24.2, df = 6, p < 0.0001) as well as a main effect of Feed source (F = 67.9, df = 3, p < 0.0001) and Week (F = 3119.6, df = 2, p < 0.0001) on the DCi of H. radicata. The DCi of H. radicata was highest for canola and jarrah feeds followed by that of clover feeds, and the lowest for marri feeds, the latter two of which differed between each other (Fig. 3G, Table S4). The DCi of H. radicata significantly increased across the weeks from week 1 to week 2 and week 2 to week 3 (Fig. 3H, Table S5).
Nectar foragers
There was a significant interaction between Feed source and Time of day (F = 13.1, df = 3, p < 0.0001) as well as Time of day and Week (F = 18.4, df = 2, p < 0.0001) (Fig. 4K,L). There was no effect of Feed source (F = 1.0, df = 3, p = 0.4) and Week (F = 1.3, df = 2, p = 0.29) on the DCi of H. radicata (Fig. 4J). There was an effect of Time of day (F = 195.9, df = 1, p < 0.0001) and the DCi was significantly greater in the mornings for bees collecting nectar (Fig. 5, Table S11).
The dominance candidate index (DCi) of plant species visited either by pollen collectors or nectar foragers from colonies (N = 40) fed on marri, jarrah, clover and canola feed sources over a three-week period (1–3). Nectar foragers were collected in the morning (am) and afternoon (pm). Plant species include: (A, B) Eucalyptus marginata; (C–E) Trifolium spp; (E–H) Persea americana. Crosses indicate the mean, bars indicate the median and the whiskers show the minimum and maximum values. Different letters show a significant difference at p < 0.05 from Tukey’s Comparison of Means.
The following pollen types were either present in corbicular pollen or on nectar foragers.
Eucalyptus marginata (jarrah)
Corbicular pollen
There was an interaction between Feed source and Week (F = 14.03, df = 6, p < 0.0001), and a main effect of Feed source (16.04, df = 3, p < 0.0001) and Week (F = 689.83, df = 2, p < 0.0001) on the DCi of E. marginata (Fig. 5). The DCi of E. marginata was lower in colonies that were fed on jarrah (E. marginata) compared to those fed on marri, clover and canola (Fig. 5A, Table S4). The DCi significantly decreased from weeks 1–2 and 2–3 (Fig. 5B, Table S5).
Trifolium spp
Nectar foragers
There was an interaction between Feed source and Time of the day (F = 5.6, df = 3, p = 0.0008) and Time of the day and Week (F = 75.4, df = 2, p < 0.0001), as well as a main effect of Week (F = 234.8, df = 2, p < 0.0001) and Time of day (F = 353.0, df = 1, p < 0.0001) on the DCi for Trifolium spp. There was no effect of Feed source (F = 0.5, df = 3, p = 0.71) on the DCi (Fig. 5C). The DCi significantly increased from week 1 to week 2 and from weeks 2–3 (Fig. 5D, Table S10). The DCi of pollen on nectar foragers was greater in the afternoon (Fig. 5E, Table S11).
Persea americana (avocado)
Nectar foragers
There was an interaction between Feed source and Time of day (F = 2.9, df = 3, p = 0.033) as well as Time of day and Week (F = 169.8, df = 2, p < 0.0001) on the DCi of P. americana. There was no effect of Feed source (F = 0.54, df = 3, p = 0.66) or Week (F = 0.8, df = 2, p = 0.47) on the DCi of P. americana (Fig. 5F,G). There was an effect of Time of day (F = 64.3, df = 1, p < 0.0001) with the DCi greater in the afternoons compared to mornings (Fig. 5H, Table S11).
Pollen quantity
Corbicular pollen
There was a significant effect of Week (F = 174.9, df = 2, p < 0.0001), Feed source (F = 4.8, df = 3, p = 0.0064), and Patty consumption (F = 7.3, df = 1, p = 0.0086) as well as an interaction between Week and Feed source (F = 2.7, df = 6, p = 0.021) and Feed source and Patty consumption (F = 3.6, df = 3, p = 0.018), included as a co-variate, on the amount of pollen collected by colonies (Fig. 6). Colonies collected more corbicular pollen in week 1 compared to week 2 and week 3 during peak flowering (Fig. 6, Table S7). The amount of corbicular pollen collected in weeks 2 and 3 did not differ from each other (Table S7). Colonies fed on canola collected significantly more pollen compared to colonies fed on clover), but not compared to those fed on marri or jarrah (Fig. 6, Table S8).
Weight (mean ± sem) of corbicular pollen (g/day) trapped throughout the avocado pollination period of colonies (N = 40) fed on marri (dashed line), jarrah (solid line), clover (dotted line) and canola (dashed and dotted line) feed sources. Colonies collected more corbicular pollen in week 1 compared to any of the other weeks during peak flowering (p < 0.0001). The amount of corbicular pollen collected in weeks 2 and 3 did not differ from each other (p = 0.328).
Nectar foragers
Nectar foragers fed on the jarrah feed source had the greatest amount of pollen on their bodies followed by those fed on clover, canola and marri, but the difference was not significant (F = 1.4, df = 3, p = 0.25). The amount of pollen on all bees across the feed sources increased from week 1–3 but was not significant (F = 0.6, df = 2, p = 0.53). Time of day had no significant effect (F = 0.7, df = 1, p = 0.4) on the amount of pollen collected but the bees foraging in the afternoon had slightly greater pollen counts. There was an interaction between Time of day and Week (F = 19.7, df = 2, p < 0.0001) (Fig. 7).
Total number of pollen grains (mean ± sem) on bodies of nectar foragers across all detected pollen types (including pollen < 5%), (A) across colonies (N = 40) fed on marri, jarrah, clover and canola, (B) Across week 1–3; (C) time of day (morning and afternoon).
Discussion
Our study showed that pollen collectors and nectar foragers, as indicated by their Dominance Candidate Index, had different foraging behaviours when they were placed on an avocado orchard for pollination services. The foraging choices made by bees were partially affected by the type of supplementary food sources provided within the hive, which could suggest that honey bees adjust their foraging behaviour according to the nutritional values of their food sources as suggested in recent studies14,15,16. The type of supplemented food had some influence on the foraging behaviour of pollen collectors but not on that of nectar foragers, the pollinators, throughout the peak flowering period of avocadoes. Aside from nutritional cues, floral source abundance and attractiveness could have influenced the foraging behaviour of both pollen and nectar foragers. Both pollen and nectar foragers chose only a few floral species, five and six respectively (representing more than 5%), out of an abundance of flowering plants at the avocado orchard.
Feed sources
The floral diversity of the assigned feed sources indicated that honey bees foraged mostly on one to two major floral species at the targeted apiary sites. These foraging choices most likely reflect the abundance of available forage rewards and were influenced by the distance honey bees needed to fly to collect either pollen or nectar.
At the coastal heathland and canola sites, bee-collected pollen comprised close to 90% of clover and canola pollen respectively. There are two possible explanations why honey bees collected almost pure pollen from flowers at these sites. If a crop such as canola, which is highly attractive to bees34,35 produces large amounts of pollen, honey bees are likely to forage shorter distances (Frisch 1967 & Visscher and Seeley 1982, referenced in Beekman & Ratnieks 2000)36especially during cooler months37. However, the abundance of a pollen source is important because if nearby floral sources are exhausted or limited, honey bees will start to forage further away38,39. Given that honey bees collected almost negligible amounts of pollen from native species at the coastal heathland site, it is likely that a pollen shortage existed. Coastal heathland sites can vary largely in floral diversity over the winter and spring months, and shorter periods of fewer yielding plants can exist40. Clover (T. repens), which the bees mostly collected pollen from at this apiary site, is often found on agricultural farmland. It is therefore likely that the bees extended their foraging trips to nearby farmland where clover can be found in paddocks, providing a greater nectar and pollen reward for them.
While we cannot comment on the absolute floral diversity of each site, species of the Myrtaceae family dominated the floral makeup of both the marri and jarrah feed sources. About 71% of the floral diversity of marri feed sources was made up of marri pollen, and about 29% of the jarrah feed source were identified as jarrah pollen, with most of the pollen (70%) being from an unknown Myrtaceae species. Jarrah forests have a variety of understorey growth40 including other myrtaceous species that flower at the same time. Palynological analysis suggests that there may also be jarrah hybrids in the forests (L. Milne pers.comm.) making it difficult to confidently assign some pollen grains to jarrah. If we consider the total pollen percentage of all Myrtaceae species in the jarrah feed source, almost 100% of the pollen assemblage was from the Myrtaceae family.
Pollen collectors
The amount of corbicular pollen collected by bees largely depends on pollen availability in the environment and on the presence of brood41,42. The increased availability of pollen during the first week of peak flowering at the avocado orchard is likely a reflection of pollen available in the environment, rather than a reflection of the crop since a negligent amount of avocado pollen (< 2%) was found in the traps. Some differences in the quantity of pollen collected by colonies fed on the various feed sources are explained by differences in the amount of brood as documented in Kratz et al.17. Nevertheless, some differences in pollen quantity collected by colonies may be explained by the type of pollen being collected, as the amount of pollen produced and its nutrient composition varies between plant species20,43.
Pollen collectors would be expected to select pollen sources according to their nutritional needs, as pollen provides bees with the bulk of their nutritional requirements1. There was some evidence that pollen collectors changed their foraging behaviour according to the food sources that were provided in this study. In particular, colonies that were fed on jarrah showed a lower DCi for jarrah pollen from the environment. Jarrah pollen is known to be of low-medium protein value (~ 19%), low in isoleucine (an essential amino acid for honey bees) and low in total fat content (0.59 ± 0.17%)18. Previous studies have demonstrated that honey bees regulate their food intake to balance protein to carbohydrate ratios in their diet44 and are able to discriminate between complementary nutrients such as amino acids14. Specific amino acids added to sucrose solution have either an attractive or deterrent effect on honey bees45,46,47 whether honey bees can detect amino acids in pollen is unknown. Honey bees may also be able to recruit nestmates to forage for diets that complement essential fatty acid deficiencies15and may be able to regulate their protein to lipid (P: L) intake similarly to bumblebees48,49,50,51 as recently reported in nurse-aged adult worker honey bees52. Interestingly, pollen collectors from colonies fed on jarrah showed an increased DCi for pollen sources of greater fat content; Arctotheca calendula with 3.4% fat content and Hypochaeris radicata with a total fat content of 5.26 to 11.2%53. Colonies supplemented with clover pollen, which is more than four times greater in fat content (2.7–3.8%54) than jarrah pollen, foraged for less A. calendula and H. radicata pollen. This preference for pollen of greater or lower fat content suggests that honey bees regulate their fat intake at the colony level. However, the trend of foraging for a higher total fat content was not observed in the colonies fed on marri and canola. Marri pollen is known to be low in fat content 0.85–0.93% (similar to jarrah)18 compared to canola with an average of 6%43 to 7.1%53 total fat content. The results therefore suggest that factors other than the composition of provided food sources were influencing the foraging behaviour of bees within the avocado orchard.
Aside from the effect of food storage and its nutritive quality, foraging choices of pollen collectors seemed to be influenced by a combination of floral source abundance and attractiveness. While avocado flowers were the most abundant floral source at the orchard, it has been shown that avocado flowers are less attractive as a pollen source compared to that from the families Brassicaceae (canola) and Fabaceae (e.g. clover)55. Indeed, pollen collectors, regardless of the type of food supplemented to the colony, collected large amounts of pollen from Trifolium repens (clover), which is highly attractive to bees and a good source of both pollen and nectar34,56. Pollen collectors must have extended their foraging range beyond the orchard to at least 0.5 to 1.5 km (Fig. S1) to collect clover pollen, since no clover was present within the orchard. It is not surprising that pollen collectors chose to collect from attractive and abundant pollen sources because the colonies were restricted by pollen traps throughout the experiment, and low pollen storage stimulates honey bees to collect more pollen3,57. Other floral cues that may have influenced foraging choices such as flower morphology and ease of pollen collection cannot be excluded and would need further investigation.
Nectar foragers
The foraging behaviour of nectar foragers was almost entirely driven by factors other than the type of supplemented feed source. This was first observed by all nectar foragers carrying similar quantities of pollen on their bodies. In only one instance the type of supplementary feed influenced the foraging behaviour of nectar foragers. Foragers from colonies fed on clover collected less clover nectar compared to all other feed sources. Generally, nectar foragers are less stimulated by the amount of stores inside the hive4and trophallaxis, the transfer of food between colony members, is likely what signals the need for nectar5,58. Because nectar is the main source of carbohydrates it is likely that other trace elements and compounds such as minerals and plant secondary metabolites contribute less to the nutritional needs of honey bees, and play a larger role in floral choice by attracting or deterring bees59,60. For instance, avocado nectar contains high concentrations of potassium and phosphorus, which have been found to deter bees during controlled experiments, whereas phenolic compounds extracted from avocado honey was attractive to honey bees60. Further studies are required to disentangle the importance of other components of nectar, other than its sugar content, that may contribute to nutritional cues.
Nectar foragers may have a more opportunistic approach to foraging than pollen collectors. Where pollen collectors foraged substantially outside of the orchard, the Dominance Candidate Index (DCi) for nectar foragers showed that they predominantly visited plant species that were present within the orchard. The three main plant types visited by nectar foragers were Persea americana (avocado), Arctotheca calendula and Myrtaceae spp, and with no difference in their DCi values. Yet, avocado trees were the dominant floral source in the orchard with about 1 million flowers per mature tree21. The similar DCi, despite the difference in flower abundance, suggests that the attractiveness of A. calendula and Myrtaceae spp may have played a greater role than flower abundance in choices by nectar collectors. It was surprising though that clover was not one of the commonly visited species for nectar collection, as honey bees have been found to preferentially collect pollen and nectar from species that produce both61.
Pollen and nectar collectors have previously been observed to exhibit different foraging behaviours in agricultural settings. Honey bees placed in a commercial crop of canola and sunflowers collected nectar predominantly from the crops, but also collected pollen from a wider diversity of surrounding plants including weeds in the crops62. Honey bees may have invested more energy in collecting pollen than collecting nectar. However, the type of forage (nectar or pollen collection) is largely driven by other factors such as its availability, which determines the foraging distance, as honey bees tightly regulate their energy investment according to pollen and nectar availability in meeting their nutritional needs13,63. Unless resources are not available close by or are scarce, then honey bees will forage more broadly11. Therefore, in the current study the proximity of nectar resources, including avocado nectar, may have outweighed the reduced attractiveness of avocado flowers60 instead of flying further to collect nectar from clover. However, the possibility that clover may have not produced much nectar, and therefore honey bees collected less of it, cannot be excluded. In fact, nectar production in clover has been linked to environmental conditions such as soil moisture and temperature64. It would be interesting to compare honey samples from colonies and nectar samples collected from flowers at the orchard over multiple years, to determine how seasonal variations in the availability of nectar sources affect the foraging behaviour of honey bees during the pollination period, as it cannot be excluded that pollen collectors may have collected nectar at the same time.
Overall foraging choices
Both pollen and nectar collectors chose only relatively few floral species for collecting nectar and pollen, displaying a selective foraging behaviour as previously observed in other diverse landscapes61,62,65. Floral species that pollen collectors visited were also visited by nectar foragers, and nectar foragers in addition collected nectar from other Trifolium species and avocado. Honey bees have previously been found to collect very little pollen (0.1 to 7%) from avocado flowers during the peak flowering period in the presence of an abundance of other flowering plant species55. The Dominance Candidate Index (DCi) represented by individual plant species differed between pollen collectors and nectar foragers. This may be linked to the nutritional value of the collected resource, but also to the energy investment to collect the resource.
In this study, we provide the first insights into how the foraging behaviour of both pollen and nectar collectors is influenced in a commercial setting. Future studies may want to relate foraging behaviour with fruit set across other crop species, where it will be important to consider the relationship between stocking rate (no. of hives/ha), colony makeup (size and amount of brood) and food source availability and quality.
Conclusion
Our study showed that supplemented feed sources can have the potential to influence the foraging behaviour of honey bees, however environmental cues including the availability of resources can have a more profound impact on foraging choices. Both the quantity and the number of bees carrying a pollen type can assist in determining the pollination efficiency of a crop. The pollination of avocado flowers is unique because of the male and female flower display that changes throughout the day. There is a need to study the effects of stored resources on bees’ foraging behaviour and the potential effects on pollination efficiency on other crops. Nevertheless, from this study, it became clear that pollen and nectar foragers respond more strongly to different environmental cues that potentially underlie energetic investment and food reward principles for meeting nutritional needs.
Data availability
The datasets generated and/or analysed during the current study are not publicly available but are available from the corresponding author on reasonable request.
References
Wright, G. A., Nicolson, S. W. & Shafir, S. Nutritional physiology and ecology of honey bees. Annu. Rev. Entomol. 63, 327–344 (2018).
Brodschneider, R. & Crailsheim, K. Nutrition and health in honey bees. Apidologie 41, 278–294 (2010).
Fewell, J. H. & Winston, M. L. Colony state and regulation of pollen foraging in the honey bee, Apis mellifera L. Behav. Ecol. Sociobiol. 30, 387–393 (1992).
Fewell, J. H. & Winston, M. L. Regulation of nectar collection in relation to honey storage levels by honey bees, Apis mellifera. Behav. Ecol. 7, 286–291 (1996).
Crailsheim, K. Trophallactic interactions in the adult honeybee (Apis mellifera L). Apidologie 29, 97–112 (1998).
Frisch, K. The Dance Language and Orientation of Bees (Harvard University Press, 1967).
Backhaus, W. Color vision and color choice behavior of the honey bee. Apidologie 24, 309–331 (1993).
Free, J. B. Effect of flower shapes and nectar guides on the behaviour of foraging honeybees. J. Soc. Dye Colour. 37, 369–390 (1970).
Dobson, H. E. M. & Bergström, G. The ecology and evolution of pollen odors. Plant. Syst. Evol. 222, 63–87 (2000).
Butler, C. G. The importance of perfume in the discovery of food by the worker honeybee (Apis mellifera L). R Soc. 138, 403–413 (1951).
Seeley, T. D. Social foraging by honeybees: how colonies allocate foragers among patches of flowers. Behav. Ecol. Sociobiol. 19, 343–354 (1986).
Harano, K. & Nakamura, J. Nectar loads as fuel for collecting nectar and pollen in honeybees: adjustment by sugar concentration. J. Comp. Physiol. A. 202, 435–443 (2016).
Couvillon, M. J. et al. Honey bee foraging distance depends on month and forage type. Apidologie 46, 61–70 (2015).
Hendriksma, H. P. & Shafir, S. Honey bee foragers balance colony nutritional deficiencies. Behav. Ecol. Sociobiol. 70, 509–517 (2016).
Zarchin, S., Dag, A., Salomon, M., Hendriksma, H. P. & Shafir, S. Honey bees dance faster for pollen that complements colony essential fatty acid deficiency. Behav. Ecol. Sociobiol. 71, (2017).
Arenas, A. & Kohlmaier, M. G. Nectar source profitability influences individual foraging preferences for pollen and pollen-foraging activity of honeybee colonies. Behav. Ecol. Sociobiol. 73, (2019).
Kratz, M. The Effects of Nutrition on Honey Bee (Apis mellifera) Health and Colony Performance during Crop Pollination (University of Western Australia, 2022).
Manning, R. Pollen analysis of eucalypts in Western Australia. https://agrifutures.com.au/product/pollen-analysis-of-eucalypts-in-western-australia/ (2001).
Ish-Am, G. Avocado pollination: A review. In New Zealand and Australia Avocado Grower’s Conference 1–10. https://doi.org/10.1016/0921-5093(93)90309-3 (2005).
Kratz, M., Manning, R., Dods, K., Baer, B. & Blache, D. Nurse bees regulate the larval nutrition of developing workers (Apis mellifera) when feeding on various pollen types. J. Econ. Entomol. 117, 683–695 (2024).
Davenport, T. L. Avocado flowering. in Horticultural Reviews vol. 8 257–289 (AVI Publishing Co., Inc. Westport, CN, (1986).
Kubitzki, K. & Kurz, H. Synchronized dichogamy and dioecy in Neotropical Lauraceae. Plant. Syst. Evol. 147, 253–266 (1984).
Erdtman, G. The acetolysis method: a revised description. Sven Bot. Tidskr. 54, 341–350 (1960).
Lau, P., Bryant, V. & Rangel, J. Determining the minimum number of pollen grains needed for accurate honey bee (Apis mellifera) colony pollen pellet analysis. Palynology 42, 36–42 (2018).
von der Ohe, W., Oddo, L., Piana, M., Marlot, M. & Martin, P. Harmonized methods of melissopalynology. Apidologie 35, 18–25 (2004).
Baum, K. A., Rubink, W. L., Coulson, R. N. & Bryant, V. M. Pollen selection by feral honey bee (Hymenoptera: Apidae) colonies in a coastal prairie landscape. Environ. Entomol. 33, 727–739 (2004).
Serra Bonvehi, J., Ventura Coll, F. & Orantes Bermejo, J. F. Characterization of avocado honey (Persea Americana Mill.) produced in Southern Spain. Food Chem. 287, 214–221 (2019).
Van Der Merwe, J. J. M., Van Wyk, A. E. & Kok, P. D. F. Pollen types in the Lauraceae. Grana 29, 185–196 (1990).
Ish-Am, G. & Eisikowitch, D. The behaviour of honey bees (Apis mellifera) visiting avocado (Persea americana) flowers and their contribution to its pollination. J. Apic. Res. 32, 175–186 (1993).
Lach, L., Kratz, M. & Baer, B. Parasitized honey bees are less likely to forage and carry less pollen. J. Invertebr Pathol. 130, 64–71 (2015).
Manning, R., Sakai, H. & Eaton, L. Methods and modifications to enhance the abundance of pollen on forager honey bees (Apis mellifera L.) exiting from beehives: implications for contract pollination services. Aust J. Entomol. 49, 278–285 (2010).
Avolio, M. L. et al. Demystifying dominant species. New. Phytol. 223, 1106–1126 (2019).
Pinheiro, J., Bates, D., DebRoy, S., Sarkar, D. & R Core Team. Linear and Nonlinear Mixed Effects Models_. R package version 3.1–144. (2020).
Lepage, M. & Boch, R. Pollen lipids attractive to honey bees. Lipids 3, 530–534 (1968).
Kotowski, Z. Results of the investigations into nectar secretion and pollen production of new cultivars of rape (Brassica Napus L). Acta Hortic. 11, 127–129 (2001).
Beekman, M. & Ratnieks, F. L. W. Long-range foraging by the honey-bee, Apis mellifera L. Br. Ecol. Soc. 14, 490–496 (2000).
Couvillon, M. J., Schürch, R. & Ratnieks, F. L. W. Waggle dance distances as integrative indicators of seasonal foraging challenges. PLoS One. 9, 1–7 (2014).
Seeley, T. D. Honey bee foragers as sensory units of their colonies. Behav. Ecol. Sociobiol. 34, 51–62 (1994).
Seeley, T. D. The Wisdom of the Hive the Social Physiology of Honey Bee Colonies (Harvard University Press, 1995).
Smith, F. G. Honey Plants in Western Australia (Department of Agriculture and Food, 1969).
Tsuruda, J. M. & Page, R. E. The effects of young brood on the foraging behavior of two strains of honey bees (Apis mellifera). Behav. Ecol. Sociobiol. 64, 161–167 (2009).
Schmickl, T. & Crailsheim, K. Inner nest homeostasis in a changing environment with special emphasis on honey bee brood nursing and pollen supply. Apidologie Springer Verlag. 35, 249–263 (2004).
Somerville, D. Nutritional Value of Bee Collected Pollens. Rural Industries Research and Development Corporation (RIRDC, 2001).
Altaye, S. Z., Pirk, C. W. W., Crewe, R. M. & Nicolson, S. W. Convergence of carbohydrate-biased intake targets in caged worker honeybees fed different protein sources. J. Exp. Biol. 213, 3311–3318 (2010).
Hendriksma, H. P., Oxman, K. L. & Shafir, S. Amino acid and carbohydrate tradeoffs by honey bee nectar foragers and their implications for plant-pollinator interactions. J. Insect Physiol. 69, 56–64 (2014).
Carter, C., Shafir, S., Yehonatan, L., Palmer, R. G. & Thornburg, R. A novel role for proline in plant floral nectars. Naturwissenschaften 93, 72–79 (2006).
Kim, Y. S. & Smith, B. H. Effect of an amino acid on feeding preferences and learning behaviour in the honey bee, Apis mellifera. J. Insect Physiol. 46, 793–801 (2000).
Vaudo, A. D. et al. Bumble bees regulate their intake of essential protein and lipid pollen macronutrients. J. Exp. Biol. 219, 3962–3970 (2016).
Vaudo, A. D., Patch, H. M., Mortensen, D. A., Tooker, J. F. & Grozinger, C. M. Macronutrient ratios in pollen shape bumble bee (Bombus impatiens) foraging strategies and floral preferences. Proc. Natl. Acad. Sci. 113, (2016).
Vaudo, A. D. et al. Pollen protein: lipid macronutrient ratios May guide broad patterns of bee species floral preferences. Insects 11, (2020).
Kraus, S. et al. Bumblebees adjust protein and lipid collection rules to the presence of brood. Curr. Zool. 65, 437–446 (2019).
Stabler, D. et al. Regulation of dietary intake of protein and lipid by nurse-age adult worker honeybees. J. Exp. Biol. 224, 1–9 (2021).
Somerville, D. Lipid content of honey bee-collected pollen from south-east Australia. Aust J. Exp. Agric. 45, 1659–1661 (2005).
Roulston, T. H. & Cane, J. H. Pollen nutritional content and digestibility for animals. Plant. Syst. Evol. 222, 187–209 (2000).
Ish-Am, G. & Eisikowitch, D. Low attractiveness of avocado (Persea Americana Mill.) flowers to honeybees (Apis mellifera L.) limits fruit set in Israel. J. Hortic. Sci. Biotechnol. 73, 195–204 (1998).
Somerville, D. Honey and Pollen Flora of South-Eastern Australia (NSW Department of Primary Industries, 2019).
Keller, I., Fluri, P. & Imdorf, A. Pollen nutrition and colony development in honey bees - Part II. Bee World. 86, 27–34 (2005).
Seeley, T. D. Social foraging in honey bees: how nectar foragers assess their colony’s nutritional status. Behav. Ecol. Sociobiol. 24, 181–199 (1989).
Stevenson, P. C., Nicolson, S. W. & Wright, G. A. Plant secondary metabolites in nectar: impacts on pollinators and ecological functions. Funct. Ecol. 31, 65–75 (2017).
Afik, O., Dag, A., Kerem, Z. & Shafir, S. Analyses of avocado (Persea americana) nectar properties and their perception by honey bees (Apis mellifera). J. Chem. Ecol. 32, 1949–1963 (2006).
Aronne, G., Giovanetti, M., Guarracino, M. R. & De, V. Foraging rules of flower selection applied by colonies of apis mellifera: ranking and associations of floral sources. Funct. Ecol. 26, 1186–1196 (2012).
Requier, F. et al. Honey bee diet in intensive farmland habitats reveals an unexpectedly high flower richness and a major role of weeds. Ecol. Appl. 25, 881–890 (2015).
Pernal, S. F. & Currie, R. W. Discrimination and preferences for pollen-based cues by foraging honeybees, Apis mellifera L. Anim. Behav. 63, 369–390 (2002).
Johnson, L. H. Nectar secretion in clover. Effect of soil and climate on honey production. New. Zeal J. Agric. 73, 111–112 (1946).
de Vere, N. et al. Using DNA metabarcoding to investigate honey bee foraging reveals limited flower use despite high floral availability. Sci. Rep. 7, 1–11 (2017).
Western Australian Herbarium. FloraBase-the Western Australian flora. Dept. Biodivers. Conserv. Attract.https://florabase.dpaw.wa.gov.au/ (1998).
Acknowledgements
This research was supported by the following funding bodies and grants: An Australian Government Research Training Program (RTP) Scholarship provided to M.K., Australian Research Council grants (LP100100438, DP130100087, LP130100029, FT110100105) granted to K.D. on behalf of the ChemCentre as well as Western Australian Department of Primary Industry and Regional Development Grower Group Research and Development Grants (WAHRDG) Program — GGRD 2015-0028-AGSC to K.D., and by the Centre for Integrative Bee Research (CIBER) - Future Bees Fund to B.B on behalf of CIBER. Additional support was provided by the Beekeeping Industry Council of Western Australia (BICWA). We thank Kingsley Dixon for his feedback and helpful discussions. We particularly thank Stephen Davies (deceased), Mark Rhodes, Colin Fleay and Dave Leyland for their time, helpful discussions, access to sites and contributions of honey and pollen. Stephen Davies was key to planning and designing the field experiment. We thank Mark Rhodes for providing bee packages, Michael Bellman on behalf of Capilano/Wescobee, for financial assistance for pollen and honey, Ron Clark for assistance with sister queens for the field experiment. We acknowledge the support from Giorgos Lykogiannis and Eleftherios Pantelakis from ANEL – LP Pantelakis & Co. (Athens, Greece) who kindly supplied 40 pollen traps to the project (ex freight). We sincerely thank Neil Delroy on behalf of Jasper farms for site access and research support.
Author information
Authors and Affiliations
Contributions
M.K. and R.M. designed and conducted the experiments. M.K, R.M. and L.M. processed samples. L.M. produced the photographic plates. M.K. analysed all data. D.B. and B.B. supervised the work and assisted with data analysis. K.D. contributed to the diversity analysis of feeds. M.K wrote the draft paper with input from co-authors. All authors reviewed the manuscript and agreed to its submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kratz, M., Manning, R., Milne, L. et al. Environmental cues rather than quality of supplemented pollen drive the foraging behaviour of honey bees during avocado pollination. Sci Rep 15, 29650 (2025). https://doi.org/10.1038/s41598-025-13981-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-13981-6