Abstract
Human epidermal growth factor receptor 2 (HER2)-targeted therapies, including monoclonal antibodies (mAbs), small-molecule tyrosine kinase inhibitors (TKIs), and antibody-drug conjugates (ADCs), have significantly improved clinical outcomes in HER2-positive cancers. However, hepatotoxicity remains a major concern with these therapies, and a comprehensive analysis of the hepatotoxicity signals across different HER2-targeted drugs is lacking. We aim to conduct a comprehensive comparative analysis of hepatotoxicity risk related to various HER2-targeted agents. Real-world pharmacovigilance research was conducted on the United States Food and Drug Administration (FDA) Adverse Event Reporting System (FAERS) database from Q1 2012 to Q2 2024. Disproportionality analysis was used to assess the strength of hepatotoxicity signals for trastuzumab, pertuzumab, T-DM1, T-DXd, and TKIs. Reporting Odds Ratios (ROR) was calculated to identify significant hepatotoxicity signals. Moreover, the time to onset (TTO) of hepatotoxicity was also evaluated. A total of 2,986 hepatotoxicity cases concerning HER2-targeted agents were collected. The strongest hepatotoxicity signal was found with T-DM1, yielding an ROR of 6.00 (95% CI: 5.51–6.54), followed by trastuzumab with an ROR of 2.65 (95% CI: 2.52–2.79) and T-DXd with an ROR of 2.51 (95% CI: 2.25–2.80). TKIs showed an ROR of 2.36 (95% CI: 2.11–2.64), while pertuzumab demonstrated the lowest signal with an ROR of 1.73 (95% CI: 1.54–1.94). The liver toxicity of HER2-targeted drugs primarily manifests as elevated ALT, AST, and bilirubin levels. Additionally, T-DM1 can lead to hepatic coma and elevated ALP levels, while T-DXd may cause liver failure and jaundice. Besides, pertuzumab showed the shortest median TTO (21 days, interquartile range [IQR] 8–98), and trastuzumab as the longest (210 days, IQR 7-656). This study identifies significant hepatotoxicity signals associated with HER2-targeted therapies, highlights the need for regular and long-term assessment of liver enzymes and bilirubin monitoring. Despite the hepatotoxicity risks, HER2-targeted therapies remain vital in the treatment of HER2-positive cancers, requiring careful management to balance efficacy and safety.
Similar content being viewed by others
Introduction
Cancer represents a leading cause of mortality and a critical public health challenge that threatens both human survival and societal advancement. Based on Global Cancer Statistics 2022, breast cancer accounts for 11.6% of all cancer cases, followed by colorectal cancer at 9.6% and gastric cancer at 4.9%, collectively presenting a significant burden to global health systems1. HER2 (Human Epidermal Growth Factor Receptor 2) is a protein encoded by the ERBB2 gene, located on chromosome 17, that plays a crucial role in cell growth and differentiation2. In certain cancers, HER2 is overexpressed or amplified in approximately 20% of breast cancers, 20% of gastric cancers, and 5% of colorectal cancer, leading to aggressive tumor growth and poor prognosis3,4,5.
HER2-targeted therapies, including monoclonal antibodies (mAbs) (e.g., trastuzumab, pertuzumab), small-molecule tyrosine kinase inhibitors (TKIs) (e.g., lapatinib, neratinib, tucatinib), and antibody–drug conjugates (ADCs) (e.g., ado-trastuzumab emtansine [T-DM1] and trastuzumab deruxtecan [T-DXd]), have substantially improved clinical outcomes by inhibiting its signaling pathways and reducing tumor progression. For more than a decade, trastuzumab has been recommended as the standard of care for early-stage or metastatic HER2-positive breast cancer. In the pivotal CLEOPATRA phase III study, adding pertuzumab to trastuzumab and docetaxel therapy demonstrated a significant survival benefit, extending median overall survival by 15.7 months versus trastuzumab and docetaxel alone6. For HER2-positive advanced breast cancer, T-DM1, T-DXd, and TKIs are indicated as initial or subsequent treatment options following trastuzumab resistance. In the ToGA study7, HER2-positive gastric cancer patients were randomized to receive trastuzumab or placebo, with the trastuzumab arm demonstrating superior median overall survival (13.8 vs. 11.1 months), median progression-free survival (6.7 vs. 5.5 months), and objective response rate (47% vs. 35%). In addition, for patients with HER2-amplified metastatic colorectal cancer, approved treatment regimens include trastuzumab combined with either pertuzumab or lapatinib as therapeutic options. For example, in the phase 2 MyPathway study, treatment with pertuzumab plus trastuzumab demonstrated an objective response rate of 32% (95% CI 20–45)8.
Despite their efficacy, these agents are associated with adverse events (AEs) that may compromise treatment continuity and quality of life. In a Phase Ib study9, 63% of participants developed grade 3 or higher adverse events following T-DXd treatment. Frequent grade 3 or higher AEs (occurring in 5% or more of patients) included neutropenia, elevated AST levels, and diarrhea among others. Among the spectrum of toxicities observed with HER2-targeted drugs, hepatotoxicity is of particular clinical concern. Drug-induced liver injury (DILI) may range from transient liver enzyme elevations to fulminant hepatic failure, with potential implications for treatment modification or discontinuation10,11. Notably, the heterogeneity in the molecular design of ADCs—including differences in the cytotoxic payload (e.g., tubulin inhibitors vs. topoisomerase inhibitors) and linker chemistry—may result in distinct hepatotoxicity profiles.
Given the limitations of clinical trials in detecting rare or delayed hepatotoxic events, post-marketing surveillance using large-scale databases is essential. The U.S. Food and Drug Administration’s Adverse Event Reporting System (FAERS) offers an invaluable resource for real-world safety assessments. This system plays a crucial role in identifying potential safety concerns, evaluating the risk-benefit profiles of medications, and informing regulatory decisions. Using systematic data-mining techniques and signal detection approaches, including disproportionality analysis, we can detect and evaluate variations in adverse drug reactions. For post-marketing study, an Italian pharmacovigilance study suggests that a higher frequency of cardiac disorders, palmar-plantar erythrodysesthesia (PPE) syndrome, thrombocytopenia, and infusion-related reactions was noticed for HER2-positive breast cancer treatments12. One study revealed that T-DXd shows higher rates of interstitial lung disease/pneumonia and myelosuppression compared to T-DM113. Another study has identified a relationship between drug-induced liver injury and specific antibody-drug conjugates, including T-DXd and enfortumab vedotin14. Furthermore, previous research has investigated adverse reactions associated with HER2-targeted agents, though it primarily examined their impact on ocular toxicity15. Though prior research has investigated various adverse reactions to HER2-targeted agents, a thorough and detailed analysis of liver toxicity patterns and characteristics remains lacking in the literature.
Based on the growing use of HER2-targeted agents, conducting a thorough real-world safety monitoring study is essential to assess the liver-related AEs of these medications. To our knowledge, this is the first pharmacovigilance analysis regarding HER2-targeted agents associated hepatotoxicity using FAERS real-world big data. Base on the FAERS database, we aim to conduct a comprehensive comparative analysis of hepatotoxicity risk related to various HER2-targeted drugs, offering healthcare providers valuable real-world data to optimize the benefit-risk assessment of these therapeutic options.
Methods
Data source and study design
The analysis utilized the FDA Adverse Event Reporting System (FAERS), a comprehensive database containing spontaneous adverse event reports submitted by healthcare providers, consumers, and drug manufacturers16,17. FAERS serves as a key resource for pharmacovigilance, enabling large-scale safety monitoring for marketed drugs, collecting over 20 million reports from patients, healthcare professionals, and pharmaceutical companies across the USA, Europe, and Asia. The dataset includes detailed information on drug exposure, patient demographics, adverse event descriptions, and patient outcomes18,19. Our analysis was based on quarterly data from FAERS, covering reports from Q1 2012 to Q2 2024. The extracted datasets included DEMO (demographic and administrative information), DRUG (drug details), RPSR (report sources), REAC (coded adverse events), INDI (indications for drug administration), THER (therapy start and end dates for reported drugs) and OUTC (patient outcomes). These datasets were linked through a primary identification number (PRIMARYID).
We included all FAERS reports involving the following HER2-targeted therapies: (1) Monoclonal antibodies (mAbs): Trastuzumab and pertuzumab; (2) Antibody-drug conjugates (ADCs): Ado-trastuzumab emtansine (T-DM1) and trastuzumab deruxtecan (T-DXd); (3) Tyrosine kinase inhibitors (TKIs): Lapatinib, neratinib, tucatinib. The Product Information or Summary of Product Characteristics (SmPCs) of HER2-targeted therapies in this study is shown in Supplementary Table S1, which presents the generic name, brand name, earliest time to market, indications, and safety profile of the drug.
Data mining and signal detection
Medical preferred terms (PTs) can be organized into broader high-level group terms (HLGTs) and system organ classes (SOCs), or grouped using Standardized MedDRA Queries (SMQs) for particular medical conditions. In the REAC table, adverse events are coded using the MedDRA v25.0, represented by PTs18,19. For signal detection, the drug was considered the primary suspect. In our study, adverse events related to hepatotoxicity were identified and searched the database using PTs from the custom liver event terms and MedDRA v25.0, including terms such as hepatic enzyme increased, acute hepatic failure, and drug-induced liver injury. Finally, hepatotoxicity cases were classified into three categories based on previous studies: hepatocellular injury (26 PTs), cholestatic injury (16 PTs), and hepatic failure (6 PTs)14,20,21. A full list of PTs combined for the custom liver event terms is provided in Supplement table S2. Reports involving HER2-targeted drugs were extracted by searching for both brand and generic names. To minimize bias, reports with missing drug information or unclear adverse event descriptions were excluded. Additionally, reports where the indication for use was not cancer-related were removed to focus on the target patient population.
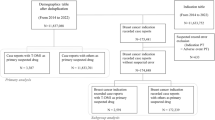
We specifically focused on cases where HER2-targeted agents were designated as Primary Suspect (PS), enhancing the precision and strength of our potential safety signals. The systematic data cleaning workflow is visually presented in Fig. 1. A single imputation was performed for missing values in event date, age, sex, and reporter country. For duplicate case versions, the most complete and recent data were retained. A two-step de-duplication was conducted: identical cases (based on case ID, follow-up status, event date, age, sex, country, drug names, and outcomes) were consolidated, keeping the latest version. Demographic data from DEMOyyQq were then linked to DRUG and REAC tables via primaryid. Only cases with HER2-targeted agents as the PS were included. Data were standardized using Observational Health Data Sciences and Informatics (OHDSI) Vocabulary 5.0 and MedDRA.
The time to onset (TTO) was defined as the duration between the initiation of anti-HER2 agents and the occurrence of hepatotoxicity events. Clarifying the onset time of AEs is essential for the proper use of drugs, as it allows healthcare professionals, including physicians and pharmacists, to implement timely interventions, thereby preventing the development and aggravation of AEs. The onset time was calculated based on the formula “(date of AE onset) - (start date of drug administration) + 0.5” as a general principle22. To illustrate the TTO of hepatotoxicity, we presented the median number of days and the corresponding interquartile range (IQR) encompassing the first quartile to the third quartile.
After standardized data processing, the analysis identified 48,258 total adverse events reports for HER2-targeted agents, with 2,986 being drug-induced liver injury cases, comprising 1,501 for trastuzumab, 301 for pertuzumab, 547 for T-DM1, 325 for T-DXd and 312 for TKIs.
Flowchart of the selection process of HER2-targeted agents related hepatotoxicity reports.
Statistical analysis
A case/non-case disproportionality analysis approach, which is a commonly established technique for detecting potential safety signals in pharmacovigilance studies, was utilized in this research. The disproportionality analysis was conducted using a 2 × 2 contingency table, where cases were defined as reports of HER2-targeted therapy associated with hepatotoxicity, and non-cases were all other reports involving these therapies23,24.
Adverse event of interest (Y) | Other adverse events | Total | |
|---|---|---|---|
Using drug of interest (X) | a | b | (a + b) |
Using other drugs | c | d | (c + d) |
Total | (a + c) | (b + d) | (a + b + c + d) |
The conditions for detecting a signal of disproportionate reporting (SDR) are defined below.
The formula for calculating the ROR is:
The 95% CI for ROR can be calculated using the following formula:
exp: Exponential function; ln: Natural logarithm; 1.96: Z-score corresponding to a 95% confidence level.
The Reporting Odds Ratio (ROR) was a key metrics, and calculated for each drug to assess the likelihood of hepatotoxicity reports compared to other drugs in the database25,26. A signal was deemed statistically significant when the lower bound of the 95% confidence interval (CI) for ROR was greater than 1, with more than 3 reported cases.
Categorical data were expressed as frequencies and percentages, while continuous variables were presented as means and standard deviations. The statistics and analysis of data was performed using R (version 4.4.2) and SPSS (version 25.0). Load different packages, R is mainly used for data analysis and graphing. Employing Chi-square tests, Mann-Whitney U test, Kruskal-Wallis test or Fisher’s exact method, the SPSS compares the differences between different drugs. The tests were deemed statistically significant with a significance threshold of p < 0.05.
Results
Demographic analysis and reporting characteristics
DILI cases totaling 2,986 were connected to 2,266 individual patients through PRIMARYID identifiers, and these patients’ clinical features were subsequently analyzed. Table 1 provides an overview of the clinical demographic analysis and reporting characteristics for DILI cases associated with the HER2-targeted agents. Female patients predominantly experienced hepatotoxicity from HER2-targeted therapies, representing over 90% of cases across all six drug types, with TKIs showing the highest rate at 99.1% (n = 221) and trastuzumab deruxtecan the lowest at 90.4% (n = 227). Among patients experiencing hepatotoxicity, those weighing between 50 and 100 kg showed the highest incidence, with all six drugs having occurrence rates exceeding 30%. In terms of occupation, physician was the primary source of hepatotoxicity reports, with health practitioner and consumers being the next most frequent reporters. In the TKIs group, 26.0% (n = 58) of reports were submitted by consumers, which was the highest among all drug groups. The United States, China and Japan constituted the largest proportion of reported countries.
Among all serious adverse reactions, death is the most critical outcome measure. In reports of deaths related to HER2-targeted drugs, T-DXd accounted for the highest proportion of deaths due to hepatotoxicity (22.7%, with 57 deaths out of 251 reports), followed by TKIs (18.4%, with 41 deaths out of 223 reports), then T-DM1 (11.1%, with 43 deaths out of 387 reports). The lowest proportions of death reports were for trastuzumab (9.2%, with 106 deaths out of 1,151 reports) and pertuzumab (9.2%, with 23 deaths out of 250 reports).
Based on the total number of AE reports, Table 1 presents the top three indications for HER2-targeted drugs. These agents are primarily used for breast cancer treatment, accounting for 75–95% of cases, with the highest proportion seen in pertuzumab (231 cases, 92.40%) and the lowest in T-DXd (191 cases, 76.10%). The second most common indication is gastric cancer, including trastuzumab (47 cases, 4.08%) and T-DXd (26 cases, 10.36%). In addition, trastuzumab is also used for colorectal cancer (5 cases, 0.43%), while T-DXd is used for non-small cell lung cancer (10 cases, 3.98%).
Spectrum of hepatotoxicity signals in HER2-targeted agents
The hepatotoxicity signals for several HER2-targeted drugs are presented in Table 2. The hepatotoxicity signals were analyzed based on ROR and 95% CIs. In addition, using trastuzumab as a reference, we conducted statistical comparisons between different drugs employing chi-square tests and Fisher’s exact method. In general, hepatotoxicity signals were detected in all five groups of HER2-targeted drugs, but there were differences in signal intensity. The strongest hepatotoxicity signal was found with T-DM1, yielding an ROR of 6.00 (95% CI: 5.51–6.54), followed by trastuzumab with an ROR of 2.65 (95% CI: 2.52–2.79) and T-DXd with an ROR of 2.51 (95% CI: 2.25–2.80). TKIs showed an ROR of 2.36 (95% CI: 2.11–2.64), while pertuzumab demonstrated the lowest signal with an ROR of 1.73 (95% CI: 1.54–1.94).
A total of 45 potential safety signals at the PT levels were identified (Fig. 2). Trastuzumab accounted for more PT signals (34), whereas T-DXd had fewer PT signals (24). The primary hepatotoxicities of trastuzumab include hyperbilirubinaemia (ROR: 7.11, 95% CI: 5.65–8.94), hepatic function abnormal (ROR: 5.75, 95% CI: 5.03–6.57), and transaminases increased (ROR: 5.07, 95% CI: 4.25–6.05). Pertuzumab’s main hepatotoxicities are blood bilirubin abnormal (ROR: 7.56, 95% CI: 2.43–23.5), hepatic function abnormal (ROR: 6.49, 95% CI: 5.18–8.13), and transaminases increased (ROR: 3.47, 95% CI: 2.36–5.09). For T-DM1, the major hepatotoxicities are blood bilirubin unconjugated increased (ROR: 24.17, 95% CI: 6.01–97.16), blood bilirubin increased (ROR: 12.67, 95% CI: 9.42–17.04), coma hepatic (ROR: 12.17, 95% CI: 1.71–86.74), aspartate aminotransferase increased (ROR: 11.89, 95% CI: 9.47–14.93), and blood alkaline phosphatase increased (ROR: 11.16, 95% CI: 7.88–15.8). The primary hepatotoxicities of T-DXd are coma hepatic (ROR: 9.93, 95% CI: 1.39–71.03), hepatic failure (ROR: 7.17, 95% CI: 5.28–9.76), blood bilirubin increased (ROR: 7.02, 95% CI: 5.03–9.79), and jaundice cholestatic (ROR: 5.4, 95% CI: 2.02–14.41). For TKIs, the main hepatotoxicities are blood bilirubin abnormal (ROR: 15.57, 95% CI: 7.76–31.26), hyperbilirubinaemia (ROR: 7.94, 95% CI: 5.64–11.18), and blood bilirubin increased (ROR: 6.88, 95% CI: 5.44–8.7). Based on the hepatotoxicity spectrum findings, HER2-targeted drugs should be monitored for transaminases and bilirubin levels. For T-DM1, attention should be given to coma hepatic and blood alkaline phosphatase increased, while for T-DXd, hepatic failure and jaundice cholestatic should be closely monitored. TKIs should especially focus on monitoring bilirubin levels.
Heatmap showing the associations between HER2-targeted agents and hepatotoxicity at PT levels.
Analysis of the time interval of the occurrence of AEs
At onset, a total of 3,612 hepatotoxic adverse events (AEs) linked to HER2-targeted agents were reported. Figure 3 displays the Kaplan-Meier curves of AE onset times for different HER2-targeted agents, with a P-value less than 0.0001 following the Kruskal-Wallis test, indicating a significant difference in AE onset times among these agents. The risk table at the lower part of Fig. 4 shows the number of individuals followed up at each time point.
Figure 4 illustrates the TTO of hepatotoxicity for various HER2-targeted agents, providing calculated median times and interquartile ranges (IQR). Pertuzumab showed the shortest median onset time (21, IQR 8–98), followed by T-DXd (63, IQR 6–329), TKIs (72, IQR 7–182), T-DM1 (165, IQR 7–672), and trastuzumab as the longest (210, IQR 7–656). Using the Kruskal-Wallis test, we identified a significant difference in median onset times among the five HER2-targeted drugs (p = 0.000), and further analysis with the Mann-Whitney U test revealed statistical significance between each pair of drugs, all with p < 0.05.
Kaplan-Meier curves and risk tables for AE reports of the hepatotoxicity associated with HER2-targeted agents.
TTO of hepatotoxicity for the HER2-targeted agents. Each boxplot represents the distribution of onset times for hepatotoxicity associated with each drug. The horizontal line in the middle of the box represents the median. The medians vary for different drugs. For example, it is 210 for Trastuzumab and 21 for Pertuzumab, etc. The numbers in parentheses are the first quartile (Q1) and the third quartile (Q3) respectively. For instance, for Trastuzumab, it is (7, 658). The two ends of the box are the first quartile (Q1) and the third quartile (Q3) respectively. The whiskers extending from the box indicate the distribution range of the data. The blue dots inside the box represent the mean (average) of the data.
Discussion
In previous studies, hepatotoxicity associated with HER2-targeted drugs in clinical practice was rarely mentioned12,27,28. Hepatotoxicity, or drug-induced liver injury, represents one of the most frequent medication-related adverse events. With the rapid expansion of cancer-targeted therapies leading to an increase in hepatotoxicity reports, drug-induced liver injury remains a major concern in oncology clinical practice. These hepatic complications can significantly impact treatment decisions and patient outcomes, with serious cases potentially progressing to acute liver failure and mortality. Based on pathogenesis, drug-induced hepatotoxicity can be classified into intrinsic, idiosyncratic, and indirect types29. Intrinsic toxicity results from the inherent toxic effects of a drug or its metabolites on the liver, idiosyncratic toxicity arises from metabolic or immune-mediated mechanisms, and indirect toxicity occurs when a drug alters pre-existing liver conditions or the immune status of the patient, indirectly leading to liver injury30. In this study, HER2-targeted drugs primarily caused indirect hepatotoxicity, typically with a delayed latency of several months. The clinical phenotypes are generally categorized into hepatocellular injury, cholestatic injury, and mixed injury types.
In this study, we performed a comprehensive comparative analysis of hepatotoxicity risk related to various HER2-targeted agents, including mAbs (e.g., trastuzumab, pertuzumab), ADCs (e.g., T-DM1, T-DXd), and TKIs (e.g., lapatinib, neratinib, tucatinib). Our findings offer valuable insights into the hepatotoxicity profiles of these drugs, emphasizing notable differences in both the seriousness of hepatotoxicity and the timing to onset across the different agents. In the following sections, we will elaborate on the characteristics, mechanisms of action, time to onset, and classification of hepatotoxicity-related PTs associated with different HER2-targeted drugs.
Our findings revealed T-DM1 had the strongest hepatotoxicity signal (ROR = 6.00, 95% CI: 5.51–6.54), indicating a robust association with liver-related adverse events, consistent with prior ADC toxicity reports. Previous research has documented HER2-targeting ADCs primarily cause elevated AST and ALT (18.75% and 18.33% incidence, with 2.98% and 2.19% grade ≥ 3)31. Comparative studies showed T-DXd had the highest AST elevation rate (31.89%), while T-DM1 led in grade 3 or more serious AST and ALT increases (3.95% and 3.42%)32. ADCs combine mAbs with cytotoxic agents via specific linkers, enabling targeted drug delivery to tumor cells, and overcoming the limitations of traditional chemotherapy33. T-DM1 links IgG1 to DM1 (microtubule inhibitor) via SMCC, while T-DXd uses GGFG to connect IgG1 with DXd (TOPO I inhibitor). In the hepatotoxicity profile analysis, we found that T-DM1 induced elevated bilirubin levels as a strong positive signal, with the ROR for unconjugated bilirubin elevated being 24.17 and for total bilirubin elevated being 12.67. Additionally, hepatic coma, elevated AST, and increased blood alkaline phosphatase also showed strong potential safety signals. T-DXd also exhibited similar adverse reaction signals, notably leading to hepatic failure (ROR: 7.17, 95% CI: 5.28–9.76). Due to the unique structure of ADCs, although both the antibody and linker may influence the toxicity of ADCs, the payload drug remains the primary determinant of ADC toxicity34. Hepatic damage caused by T-DM1 has already been included in the FDA black box warning. DM1, a cytotoxic agent derived from maytansine, is metabolized in the liver via cytochrome P450 3A4/5 (CYP3A4/5) pathways. Furthermore, potential mechanisms of hepatotoxicity caused by ADCs include: low-level expression of target antigens on normal cells, which may lead to specific toxicity; early cleavage of the linker between the antibody and drug in circulation, resulting in nonspecific drug release and off-target toxicity; nonspecific endocytosis, which may lead to ADC uptake by normal liver cells, causing cytotoxic effects; and receptor-mediated uptake, where the receptor recognizes the Fc region of the IgG backbone in the ADC, thereby mediating non-targeted internalization in normal cells35.
The current study showed significant hepatotoxicity signals for trastuzumab and pertuzumab, with RORs of 2.65 2.65 (95%CI: 2.52–2.79) and 1.73 (95%CI: 1.54–1.94) respectively, demonstrating better potential liver safety profiles than ADCs. Trastuzumab’s mechanism involves binding to HER2’s domain IV, affecting signaling through multiple pathways: preventing dimerization, causing receptor internalization/degradation, inhibiting PI3K-AKT signaling, and triggering antibody-dependent cellular cytotoxicity (ADCC)3,35,36. Pertuzumab differs by targeting HER2’s domain II, thereby blocking ligand-induced heterodimerization37,38. Hepatotoxicity may involve multiple mechanisms, including immune-mediated liver injury, drug metabolism and cytotoxicity, hepatic hemodynamic alterations, dose- and treatment duration-related effects, individual variability, and drug interactions39,40. Notably, the blood bilirubin abnormal associated with pertuzumab may represent a new potential safety signal. We reviewed the latest FDA drug label and relevant literature revealed no current mention of this association41. However, abnormal bilirubin levels may be associated with various factors, such as hepatic parenchymal injury and different hemolytic disorders42,43, highlighting the need for further research to clarify the causal relationship.
The analysis revealed a significant hepatotoxicity signal for TKIs with an ROR of 2.36 (95% CI: 2.11–2.64). While lower than T-DM1’s signal, this indicates noteworthy liver toxicity risk. Our study examined three HER2-targeting TKIs - lapatinib, neratinib, and tucatinib, which showed particular association with blood bilirubin abnormal (ROR = 15.57, 95% CI: 7.76–31.26). Lapatinib’s hepatotoxicity involves multiple pathways including apoptosis and DNA damage, with its N-dealkylated metabolite contributing to toxicity. CYP3A5 and CYP3A7 metabolism helps moderate lapatinib’s cytotoxic effects44,45. Network pharmacology research has identified several pathways through which TKIs may cause liver injury, including protein processing in the endoplasmic reticulum and retinol metabolism. The HSP70 protein family, particularly HSPA8 and HSPA1A, appears crucial in the endoplasmic reticulum stress response, with their decreased expression following TKI treatment potentially leading to liver cell damage46.
This study indicates a significant difference in the median onset time of hepatotoxicity among HER2-targeted drugs. Interestingly, both pertuzumab and trastuzumab are mAbs primarily used to treat HER2-positive breast cancer. However, in our study, pertuzumab had the shortest median time to hepatotoxicity onset (21, IQR 8–98), whereas trastuzumab had the longest median onset time (210, IQR 7–656). Pertuzumab’s smaller molecular size (~ 150 kDa vs. trastuzumab’s ~ 185 kDa) may result in faster hepatic clearance or altered tissue distribution, leading to higher liver exposure and metabolic stress37,47. Trastuzumab is predominantly degraded via lysosomal pathways in non-hepatic tissues, reducing direct hepatocyte burden. Moreover, pertuzumab’s optimized Fcγ receptor binding enhances antibody-dependent cellular cytotoxicity (ADCC) and macrophage activation (ADCP), potentially triggering inflammatory responses in the liver, such as Kupffer cell activation. In contrast, trastuzumab exhibits weaker immune effector functions, leading to milder hepatic immune activation37,48. The earlier hepatotoxicity of pertuzumab stems from its dual HER2/HER3 pathway disruption, enhanced immune activation, and pharmacokinetic profile, compounded by synergistic toxicity in combination therapies37,47. Early and regular liver function monitoring is essential during pertuzumab treatment.
Furthermore, we examined the influence of combination treatments by analyzing the top five reported concomitant drugs and conducting signal detection analysis (Supplementary Table S4). Overall, concomitant drugs increased potential hepatotoxicity signals across HER2-targeted therapies. With trastuzumab, docetaxel was most frequently co-administered (n = 535), but cyclophosphamide showed stronger hepatotoxicity association (n = 193, ROR = 4.34, 95% CI: 3.77–5.02). For pertuzumab, docetaxel was again most common (n = 130), while carboplatin demonstrated higher hepatotoxicity correlation (n = 64, ROR = 5.13, 95% CI: 3.98–6.54). With T-DM1, ondansetron showed notable liver toxicity signal enhancement (n = 14, ROR = 9.09, 95% CI: 5.29–15.44). For T-DXd, acetaminophen was associated with increased hepatotoxicity risk (n = 14, ROR = 7.72, 95% CI: 4.53–13.14). Among TKIs, zoledronic acid demonstrated significant potential to amplify hepatotoxicity signals (n = 20, ROR = 9.93, 95% CI: 6.30-15.48). On one hand, chemotherapeutic agents such as docetaxel and carboplatin, as mentioned above, inherently possess strong hepatotoxic potential. On the other hand, during combination therapy, certain drugs may modulate the production of reactive metabolites by inducing, inhibiting, or competing as substrates, particularly through interactions with the cytochrome P450 enzyme system, thereby influencing the individual risk of developing hepatotoxicity10,49,50.
Although the FAERS database used in this study has a large sample size and a broad coverage of the population, it still has certain limitations. FAERS is a spontaneous reporting system, which may include reporting errors, duplicate reports, and inconsistencies. Additionally, it is difficult to account for confounding factors such as dosage, treatment duration, comorbidities, and other factors that could influence the occurrence of hepatotoxicity. Due to data limitations, we cannot determine patients’ disease progression status or establish direct causality between disease state and liver toxicity development. These factors can affect the reliability and generalizability of the results. Moreover, the methodology utilized for case determination in the context of combination therapies poses additional complexities. When AEs occur in patients receiving multiple drugs, FAERS enables the designation of primary and secondary suspects. Nonetheless, disentangling the specific contribution of each component of a combination regimen to the reported AE proves challenging, particularly evident in anti-tumor regimens. Furthermore, the disproportionality analysis used in pharmacovigilance studies is difficult to make a causal inference. Subsequent studies are necessary to validate these assumptions. Nevertheless, our research logically and methodically assessed the potential hepatotoxicity risk of HER2-targeted agents.
Conclusion
In conclusion, our findings underscore the importance of pharmacovigilance and the utility of the FAERS database in detecting adverse events that are often underreported in clinical trials. For HER2-targeted therapies, hepatotoxicity monitoring is crucial, particularly with T-DM1. Regular assessment of liver enzymes and bilirubin is essential. T-DM1 requires specific vigilance for signs of hepatic coma and elevated alkaline phosphatase, while T-DXd necessitates close monitoring for liver failure and hepatocellular jaundice. With TKIs, careful attention to bilirubin levels is particularly important. In addition, the hepatotoxicity of HER2-targeted drugs requires long-term monitoring, as some delayed manifestations may even occur more than a year after treatment. Further evidence is necessary to validate these hypotheses and develop enhanced clinical safety protocols. The results highlight the need for ongoing safety monitoring, improved adverse event reporting, and deeper investigation into underlying mechanisms to optimize cancer treatment strategies.
Data availability
Data availability statementData associated with our study has been deposited into a publicly available repository (https://fis.fda.gov/extensions/FPD-QDE-FAERS/FPD-QDE-FAERS.html).
References
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74 (3), 229–263 (2024). 21834.
Chen, N. et al. HER2 targeted therapy in colorectal cancer: Current landscape and future directions. Biochem. Pharmacol. 223, 116101. https://doi.org/10.1016/j.bcp.2024.116101 (2024).
Oh, D. Y. & Bang, Y. J. HER2-targeted therapies—a role beyond breast cancer. Nat. Rev. Clin. Oncol. 17 (1), 33–48. https://doi.org/10.1038/s41571-019-0268-3 (2020).
Hsu, J. L. & Hung, M. C. The role of HER2, EGFR, and other receptor tyrosine kinases in breast cancer. Cancer Metastasis Rev. 35 (4), 575–588. https://doi.org/10.1007/s10555-016-9649-6 (2016).
Pous, A. et al. HER2-Positive gastric cancer: The role of immunotherapy and novel therapeutic strategies. Int. J. Mol. Sci. 24 (14). https://doi.org/10.3390/ijms241411403 (2023).
Baselga, J. et al. Pertuzumab plus trastuzumab plus docetaxel for metastatic breast cancer. N. Engl. J. Med. 366 (2), 109–119 (2012). https://doi.org/10.1056/NEJMoa1113216.
Bang, Y. J. et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): A phase 3, open-label, randomised controlled trial. Lancet. 376(9742), 687–97 (2010). https://doi.org/10.1016/S0140-6736(10)61121-X.
Meric-Bernstam, F. et al. Pertuzumab plus trastuzumab for HER2-amplified metastatic colorectal cancer (MyPathway): An updated report from a multicentre, open-label, phase 2a, multiple basket study. Lancet Oncol. 20 (4), 518–530 (2019).
Modi, S. et al. Antitumor activity and safety of trastuzumab Deruxtecan in patients with HER2-Low-Expressing advanced breast cancer: Results from a phase Ib study. J. Clin. Oncol. 38 (17), 1887–1896 (2020).
Mao, Y. et al. Chinese guideline for the diagnosis and treatment of drug-induced liver injury: An update. Hepatol. Int. 18 (2), 384–419. https://doi.org/10.1007/s12072-023-10633-7 (2024).
Fettiplace, A. et al. Review article: recommendations for detection, assessment and management of suspected drug-induced liver injury during clinical trials in oncology patients. Aliment. Pharmacol. Ther. 60 (10), 1293–1307 (2024). https://doi.org/10.1111/apt.18271
Barbieri, M. A. et al. Adverse Drug Reactions with HER2-Positive Breast Cancer Treatment: An Analysis from the Italian Pharmacovigilance Database. Drugs Real World Outcomes. ;9(1):91–107. (2022). Santoro, Paola Maria Cutroneo, Tindara Franchina, and Edoardo Spina have no conflicts of interest that are relevant to the content of this article. The research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. https://doi.org/10.1007/s40801-021-00278-z
Ma, P. et al. High risks adverse events associated with trastuzumab emtansine and trastuzumab Deruxtecan for the treatment of HER2-positive/mutated malignancies: A pharmacovigilance study based on the FAERS database. Expert Opin. Drug Saf. 2023 22(8), 685–696. https://doi.org/10.1080/14740338.2023.2204228.
Sun, C. et al. A pharmacovigilance study on drug-induced liver injury associated with antibody-drug conjugates (ADCs) based on the food and drug administration adverse event reporting system. Expert Opin. Drug Saf. 23 (8), 1049–1060 (2024).
Chen, H., Yang, G. & Ma, J. Ocular toxicity associated with anti-HER2 agents in breast cancer: A pharmacovigilance analysis using the FAERS database. Int. J. Cancer. 154 (9), 1616–1625 (2024). .34848. PubMed PMID: 38216995.
Fusaroli, M., Salvo, F., Begaud, B. & Disproportionality Analysis for DrUg Safety Signal Detection Using Individual Case Safety Reports in PharmacoVigilance (READUS-PV). The REporting of A : Explanation and Elaboration. Drug Saf. 47(6):585–599. Mahaux and Andrew Bate are full-time employees at GSK and own GSK restricted shares. Manfred Hauben was a full-time employee at Pfizer when the Delphi was conducted and owns stock/stock options in pharmaceutical companies that may manufacture/market drugs mentioned in this paper. Michele Fusaroli, Francesco Salvo, Bernard Begaud, Thamir M. AlShammari, Vera Battini, Andreas Brueckner, Carla Carnovale, Salvatore Crisafulli, Paola Maria Cutroneo, Charles Dolladille, Milou-Daniel Drici, Jean-Luc Faillie, Adam Goldman, Maria Teresa Herdeiro, Francois Montastruc, Yoshihiro Noguchi, G. Niklas Noren, Roberta Noseda, Igho J. Onakpoya, Antoine Pariente, Elisabetta Poluzzi, Myriam Salem, Daniele Sartori, Nhung T.H. Trinh, Marco Tuccori, Florence van Hunsel, Eugene van Puijenbroek, Emanuel Raschi, and Charles Khouri declare no conflicts of interest in relation to this research. (2024). https://doi.org/10.1007/s40264-024-01423-7
Fusaroli, M. et al. The Reporting of a Disproportionality Analysis for Drug Safety Signal Detection Using Individual Case Safety Reports in PharmacoVigilance (READUS-PV): Development and Statement. Drug Saf. ;47(6):575–584. (2024). Mahaux and Andrew Bate are full-time employees at GSK and own GSK restricted shares. Manfred Hauben was a full-time employee at Pfizer when the Delphi was conducted and owns stock/stock options in pharmaceutical companies that may manufacture/market drugs mentioned in this paper. The remaining authors declare no conflict of interest specific for this research. https://doi.org/10.1007/s40264-024-01421-9
Russo, G. et al. Renal disorders with oral tyrosine kinase inhibitors in metastatic colorectal cancer: An analysis from the FDA adverse event reporting system database. Biomedicines 11 (8). https://doi.org/10.3390/biomedicines11082311 (2023).
Barbieri, M. A. et al. Unveiling cardiovascular and respiratory toxicities with monoclonal antibodies in multiple myeloma: Disproportionality analysis from the FDA adverse event reporting system. Eur. J. Clin. Pharmacol. 81 (5), 755–770. https://doi.org/10.1007/s00228-025-03824-8 (2025).
Suzuki, A. et al. Comedications alter drug-induced liver injury reporting frequency: data mining in the WHO vigibase. Regul. Toxicol. Pharmacol. 72 (3), 481–490. https://doi.org/10.1016/j.yrtph.2015.05.004 (2015).
Wei, C. et al. A pharmacovigilance study of the association between tetracyclines and hepatotoxicity based on food and drug administration adverse event reporting system data. Int. J. Clin. Pharm. 44 (3), 709–716. https://doi.org/10.1007/s11096-022-01397-5 (2022).
Ando, G. et al. Evaluation of the expression time of Ganciclovir-Induced adverse events using JADER and FAERS. Biol. Pharm. Bull. 42 (11), 1799–1804. https://doi.org/10.1248/bpb.b19-00156 (2019).
Rothman, K. J., Lanes, S. & Sacks, S. T. The reporting odds ratio and its advantages over the proportional reporting ratio. Pharmacoepidemiol Drug Saf. 13 (8), 519–523 (2004).
Cutroneo, P. M. S. D. et al. Conducting and interpreting disproportionality analyses derived from spontaneous reporting systems. Front. Drug Saf. 2024;Regul 3, 1323057. https://doi.org/10.3389/fdsfr.2023.1323057
Fusaroli, M. et al. The evolving role of disproportionality analysis in pharmacovigilance. Expert Opin. Drug Saf. 23 (8), 981–994 (2024).
Almenoff, J. S. et al. Novel statistical tools for monitoring the safety of marketed drugs. Clin. Pharmacol. Ther. 82 (2), 157–166. https://doi.org/10.1038/sj.clpt.6100258 (2007).
Kuemmel, S. et al. Subcutaneous trastuzumab with Pertuzumab and docetaxel in HER2-positive metastatic breast cancer: Final analysis of metapher, a phase IIIb single-arm safety study. Breast Cancer Res. Treat. 187 (2), 467–476. https://doi.org/10.1007/s10549-021-06145-3 (2021).
Garcia-Alvarez, A., Papakonstantinou, A. & Oliveira, M. Brain metastases in HER2-Positive breast cancer: Current and novel treatment strategies. Cancers (Basel). 13 (12). https://doi.org/10.3390/cancers13122927 (2021).
Hoofnagle, J. H. & Bjornsson, E. S. Drug-Induced liver Injury—Types and phenotypes. N. Engl. J. Med. 381 (3), 264–273 (2019). https://doi.org/10.1056/NEJMra1816149.
Chalasani, N. P. et al. ACG clinical guideline: Diagnosis and management of idiosyncratic Drug-Induced liver injury. Am. J. Gastroenterol. 116 (5), 878–898 (2021). https://doi.org/10.14309/ajg.0000000000001259
Fu, Z. et al. Treatment-related adverse events associated with HER2-Targeted antibody-drug conjugates in clinical trials: A systematic review and meta-analysis. EClinicalMedicine 55, 101795. https://doi.org/10.1016/j.eclinm.2022.101795 (2023).
Yang, P. et al. Incidence of antibody-drug conjugate-related hepatotoxicity in breast cancer: A systematic review and meta-analysis. Ther. Adv. Drug Saf. 15, 20420986241304680 (2024). https://doi.org/10.1177/20420986241304680.
Tarantino, P. et al. Optimizing the safety of antibody-drug conjugates for patients with solid tumours. Nat. Rev. Clin. Oncol.. 20(8), 558–576. (2023). https://doi.org/10.1038/s41571-023-00783-w.
Dumontet, C. et al. Antibody-drug conjugates come of age in oncology. Nat. Rev. Drug Discov. 22 (8), 641–661. https://doi.org/10.1038/s41573-023-00709-2 (2023).
Yan, H. et al. Ado-Trastuzumab emtansine targets hepatocytes via human epidermal growth factor receptor 2 to induce hepatotoxicity. Mol. Cancer Ther. 15 (3), 480–490. https://doi.org/10.1158/1535-7163.MCT-15-0580 (2016).
Klapper, L. N. et al. Tumor-inhibitory antibodies to HER-2/ErbB-2 May act by recruiting c-Cbl and enhancing ubiquitination of HER-2. Cancer Res. 60 (13), 3384–3388 (2000).
Sakai, K. et al. Pertuzumab, a novel HER dimerization inhibitor, inhibits the growth of human lung cancer cells mediated by the HER3 signaling pathway. Cancer Sci. 98 (9), 1498–1503. https://doi.org/10.1111/j.1349-7006.2007.00553.x (2007).
Mendoza, N. et al. Inhibition of ligand-mediated HER2 activation in androgen-independent prostate cancer. Cancer Res. 62 (19), 5485–5488 (2002).
Jaeschke, H. et al. Mechanisms of hepatotoxicity. Toxicol. Sci. 65 (2), 166–176. https://doi.org/10.1093/toxsci/65.2.166 (2002).
Shehu, A. I., Ma, X. & Venkataramanan, R. Mechanisms of Drug-Induced hepatotoxicity. Clin. Liver Dis. 21 (1), 35–54. https://doi.org/10.1016/j.cld.2016.08 (2017).
Zou, S. P. et al. A disproportionality analysis of adverse events associated to Pertuzumab in the FDA adverse event reporting system (FAERS). BMC Pharmacol. Toxicol. 24 (1), 62. https://doi.org/10.1186/s40360-023-00702-w (2023).
Phillips, J. & Henderson, A. C. Hemolytic anemia: Evaluation and differential diagnosis. Am. Fam. Phys. 98 (6), 354–361 (2018).
Weaver, L. et al. Biliverdin reductase and bilirubin in hepatic disease. Am. J. Physiol. Gastrointest. Liver Physiol. 314 (6), G668–G676. https://doi.org/10.1152/ajpgi.00026.2018 (2018).
Chen, S. et al. The involvement of hepatic cytochrome P450s in the cytotoxicity of lapatinib. Toxicol. Sci. 197 (1), 69–78. https://doi.org/10.1093/toxsci/kfad099 (2023).
Vigano, M. et al. Hepatotoxicity of small molecule protein kinase inhibitors for cancer. Cancers (Basel). 15 (6). https://doi.org/10.3390/cancers15061766 (2023).
Tang, M., Wu, Z. E. & Li, F. Integrating network Pharmacology and drug side-effect data to explore mechanism of liver injury-induced by tyrosine kinase inhibitors. Comput. Biol. Med. 170, 108040 (2024).
Gharwan, H. & Groninger, H. Kinase inhibitors and monoclonal antibodies in oncology: Clinical implications. Nat. Rev. Clin. Oncol. 13(4), 209–27. (2016). https://doi.org/10.1038/nrclinonc.2015.213.
Wu, X. et al. Emerging insights into mechanisms of trastuzumab resistance in HER2-positive cancers. Int. Immunopharmacol. 122, 110602. https://doi.org/10.1016/j.intimp.2023.110602 (2023).
Onda, K., Honma, T. & Masuyama, K. Methotrexate-related adverse events and impact of concomitant treatment with folic acid and tumor necrosis factor-alpha inhibitors: An assessment using the FDA adverse event reporting system. Front. Pharmacol. 14, 1030832 (2023).
Honma, T., Onda, K. & Masuyama, K. Drug-drug interaction assessment based on a large-scale spontaneous reporting system for hepato- and renal-toxicity, and thrombocytopenia with concomitant low-dose methotrexate and analgesics use. BMC Pharmacol. Toxicol. 25 (1), 13. https://doi.org/10.1186/s40360-024-00738-6 (2024).
Funding
There is no sponsorship for this study.
Author information
Authors and Affiliations
Contributions
Guangyi Yu and Junli Dong were involved in the conception and design. Dan Chen and Yumin Zhong were involved in the analysis and interpretation of the data. The drafting of the paper was conducted by Guangyi Yu, and revising it critically for intellectual content. Junli Dong made the final approval of the version to be published. All authors agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Dong, J., Chen, D., Zhong, M. et al. Real-world pharmacovigilance assessment of hepatotoxicity risk with HER2-Targeted drugs using FAERS database analysis. Sci Rep 15, 28790 (2025). https://doi.org/10.1038/s41598-025-14064-2
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-14064-2