Abstract
To evaluate the effectiveness, safety, and postoperative outcomes of endoscopic radiofrequency adenoidectomy in pediatric patients over seven years. This study was conducted at the University Hospitals between May 2017 and August 2024. This prospective observational case series included pediatric patients who underwent endoscopic radiofrequency adenoidectomy due to adenoid hypertrophy and related complications. Preoperative evaluation included clinical assessment, nasal endoscopy, and imaging. Intraoperative data, including blood loss and operative time, and postoperative outcomes, such as pain, bleeding, and halitosis, were recorded. The mean operative time was 15 min, and the mean blood loss was 31 ml (p = 0.0001). Postoperative pain decreased significantly over time (p = 0.0001). Primary bleeding occurred in 0.3% of cases. Halitosis affected 2% of patients during the first week but was reduced by the first month (p = 0.0001). Endoscopic radiofrequency adenoidectomy is a safe, efficient, and minimally invasive approach for managing adenoid hypertrophy in pediatric patients. It offers minimal blood loss, rapid recovery, and low complication rates.
Similar content being viewed by others
Introduction
Adenoidectomy is one of the most common procedures done on children, either by itself or frequently, with ventilation tube (grommet) insertion or tonsillectomy. Adenoids have a significant role in the immune response to various infections, especially in the first 3 years of age, and act as a reservoir of IgG and IgA in mucosal and submucosal portions. Hence, the most established evidence is on the impact of adenotonsillectomy as one entity, so there is no accurate literature on the immunological consequences of solitary adenoid removal1.
Children frequently exhibit adenoid hypertrophy, which is more prevalent. Hypertrophied adenoids cause many problems that impact well-being and interfere with children’s natural development. Airway obstruction, mouth breathing, adenoid faces, sleep apnea, hyponasal speech, rhinorrhea, and recurrent rhino sinusitis can all result from enlarged adenoids either through hypertrophy or by serving as a reservoir for respiratory infections2,3,4.
Recurrent Otitis media with effusion and consequent hearing loss, permanent facial growth features, and cardiopulmonary complications can follow chronic adenoid hypertrophy. All these conditions are indications of adenoidectomy with or without tonsillectomy5.
Many techniques are available for adenoidectomy; complete excision of adenoid tissue is the goal. Classic adenoidectomy done blindly using a sharp curette was the mainstay for a long time. However, the high recurrence rate and the higher incidence of complications such as bleeding and velopharyngeal insufficiency were major drawbacks. Some surgeons accomplished this with a laryngeal mirror, but visualization of the adenoid yielded a better result, so Endoscopic adenoidectomy offered a new effect6.
Endoscopic-assisted adenoidectomy uses different devices, including suction diathermy, microdebrider, coblation, radiofrequency, and laser. For example, intraoperative and postoperative blood loss are still the most common consequences of this treatment. Furthermore, postoperative pain, patient discontent, prolonged surgical time, residual adenoid tissue, and infection are some of the unavoidable problems of adenoidectomy. Because the adenoidectomy performance rate is growing, surgeons must select the best surgical strategy for their patients7,8,9,10.
This study presents our 7-year experience performing endoscopic radiofrequency-assisted adenoid volume reduction in our institute.
Patient and methods
Study design
The study is an observational descriptive prospective case series; this study was held in the Otolaryngology Department at the University Hospitals from May 2017 to August 2024. A total of 3450 cases underwent endoscopic adenoidectomy using our unique technique in this period. The Institutional Review Board of Kafr-Elsheikh University (KFS IRB) approved our study protocol with an approval number “KFSIRB200-201”. All parents or guardians of patients in our study signed informed written consent to use their data in our case series. All procedures performed in studies involving human participants followed the ethical standards of the institutional research committee and were conducted with the Helsinki Declaration.
Patient selection & evaluation
The initial evaluation was to examine the patients attending our tertiary center outpatient clinic with symptoms like nasal obstruction, snoring, sleep apnea, features of adenoid faces, and secretory otitis media through a detailed history obtained from parents.
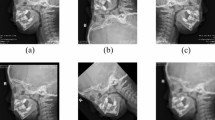
An ENT physician conducted a thorough clinical examination using anterior nasal endoscopy with a 0-degree endoscope, where adenoid grading was done according to the scale of Clemens et al.. Patients were asked to x-ray the nasopharynx lateral skull view with the mouth opened and closed, and the adenoid-nasopharyngeal ratio was calculated according to Fujioka et al.11,12.
We obtained a Computerized Tomography scan for older patients to exclude nasopharyngeal masses or anatomical abnormalities. Laboratory testing, including CBC, prothrombin time, partial thromboplastin time, international nationalized ratio, and chest X-ray, was performed on all participants before the surgery as a standard protocol of our hospital.
Surgical technique
All the operations were done by a senior ENT surgeon under general anesthesia using endotracheal tubes of suitable size according to age. The patient lay in a supine position with the head in a flat plane. The mouth was opened using a Boyle Davis mouth gag supported over the table with a Maguaran plate for holding a Draffin bipod stand. The soft palate was elevated using two blunt-tip catheters of 8 French size introduced through each nasal cavity into the oral cavity to be tied gently near the upper lip for easy access to the nasopharynx without injuring the uvula. Figure 1.
The nasopharynx by endoscopic view trans orally after elevating the soft palate at the beginning of the procedure.
The nasopharynx was visualized using a 45-degree Endoscope (Karl Storz Hopkins® II endoscope, 18 cm length, and 4 mm diameter) and Storz IMAGE 1 SPIES HD Camera introduced transorally. We used the CURIS® 4 MHz Radio-frequency Generator _ Sutter_ USA with curved bipolar blade for adenoid ablation (Sutter, Freiburg, Germany) in (RaVoR™) Radiofrequency Volume Reduction mode with power 25 Watts and “auto-stop” feature turned on to stop the current when the tissue reached the maximum ablation13.
We used a curved blade to perform adenoid ablation, which was carried out starting from the low nasopharynx and then continued from side to side till reaching the vomer edge with caution not to injure the tubal fold on each side. Vigorous saline washing and suction were used to protect the sub-mucosal layer and Eustachian tube from collateral heat and remove the remaining cauterized tissue debris that causes infection and halitosis. Figures 2 and 3.
The beginning of the operation with an endoscope and the curved blade of Sutter radiofrequency, with the soft palate retracted by two catheters.
The ablation of adenoid tissue and surrounding structures (eustachian tube opening ).
For hemostasis, we used the coagulation mode set at the power of 30 Watts to secure any bleeding points; the surgeon used a gauze soaked in hydrogen peroxide at 20% concentration diluted with normal saline, and then the nasopharynx was visualized for a final look. Figure 414.
The end of the operation with ablated adenoid tissue after finishing the hemostasis.
The duration of setup and operation was calculated. The amount of blood loss during the operation was measured using the suction vacuum flask, which was added to the weight difference between the gauze used for hemostasis before and after application. The incidence of primary and secondary (in more than 24 h) was calculated. The pain was evaluated using the pain VAS score by asking the child (or their parent) to point to one of the ten faces drawn, ranging from a smiling face representing no pain (0 on a scale) to a sad crying face representing severe pain (10 on a scale)15,16. Other complaints, like halitosis, were recorded through parents’ notice and reporting.
Postoperative follow-up
Patients were discharged on the same day as the operation if there were no complications. We followed up with our patients for complications such as pain and bleeding recurrence. We also recorded the average procedure time, amount of blood loss, and rate of secondary bleeding.
Results
The study included 3450 patients who underwent endoscopic radiofrequency adenoidectomy. However, only 1200 continued the study period; the remaining either dropped out or refused to continue follow-up. The mean age was 6 years, ranging from 2 to 15 years. A higher proportion of patients were male (54%) compared to females (46%) (p = 0.012). The mean operative time was 14 min, with the mean blood loss during surgery being 31 mL (p = 0.0001) (Table 1).
Postoperative pain significantly improved over time. The mean pain VAS score on the first postoperative day was 5, reduced to 3 in the first week, and 1 by the first month. All the patients did the test alone without their parents’ help, except 1.5% (2 years old). This progressive reduction in pain was statistically significant (p = 0.0001) (Table 2).
Primary bleeding occurred in 0.3% of cases (3 patients), while secondary bleeding was noted in 0.1% (one patient). Most patients did not experience postoperative bleeding; the differences were statistically significant (p = 0.0001) (Table 2).
Halitosis was a typical early complication observed in 2% of patients during the first postoperative week. By the end of the first month, the incidence decreased to 1.1%, showing a significant resolution over time (p = 0.0001) (Table 2).
After one year of follow-up, only 1.8% of the patients had regrowth of adenoid tissue by endoscopic and x-ray images. Of all the 1200 patients who had adenoidectomy, only six patients had a revision adenoidectomy after 1 year, and all of them were under the age of 5 years at the time of the first surgery. (Table 3)
Discussion
Our seven years of endoscopic radiofrequency adenoid ablation experience show many benefits; however, some drawbacks will occur. The findings are consistent with the increasing use of endoscopic methods within modern otolaryngology. This study’s large sample size is one of its advantages since it offers solid evidence of the endoscopic radiofrequency technique’s safety and effectiveness. The method’s effectiveness is further demonstrated by thoroughly assessing intraoperative parameters, such as blood loss and length. Its ability to enhance patient happiness and recovery is shown by postoperative outcomes, such as low rates of subsequent bleeding and minimal discomfort reported by patients.
Precision tissue ablation with minor thermal damage was made possible using cutting-edge tools like the CURIS® 4 MHz radiofrequency generator. The low rates of complications, such as primary bleeding (0.3%) and secondary bleeding (0.1%), which contrast nicely with more conventional techniques like suction diathermy or sharp curettage, are especially indicative of this.
Traditional adenoidectomy techniques like curettage have been the cornerstone of surgical management for decades; however, these methods often present limitations in achieving complete adenoid tissue removal, particularly in areas difficult to access, such as the superior nasopharynx or around the Eustachian tube. This incomplete removal can result in adenoid regrowth and recurrence of symptoms, necessitating revision surgery in some cases. In this context, endoscopic adenoidectomy has become a significant advancement, offering improved visualization and precision in adenoid tissue removal. Using rigid or flexible endoscopes, the endoscopic approach allows surgeons to visualize the nasopharynx directly and precisely target adenoid tissue, minimizing the risk of injury to surrounding structures, such as the torus tubarius and the pharyngeal constrictor muscles. Moreover, radiofrequency volume reduction in conjunction with endoscopic adenoidectomy has further refined the procedure, providing a controlled and effective tissue ablation with minimal thermal damage to adjacent tissues17,18.
Endoscopic adenoidectomy provides better visualization, enabling complete and safer removal of adenoid tissue, resulting in less postoperative pain and lower recurrence. Mirror adenoidectomy is quicker and uses cheaper tools but carries a higher risk of residual tissue and complications due to limited visualization. The choice of technique may depend on the surgeon’s expertise, equipment availability, and case specifics. However, endoscopic methods are generally regarded as superior in efficacy and safety. Using the mirror is an indirect, less magnified view, shorter operative time (average 14 min), slightly higher pain scores reported, higher chance of residual adenoid tissue due to indirect visualization, potentially leading to recurrence or symptoms. Regarding the teaching and precision, the mirror is less optimal due to the limited visualization. On the other hand, endoscope with camera and monitor is direct, magnified, and clear view of surgical field, operation time is slightly longer (average 18 min) due to setup time and detailed resection, blood loss is comparable to mirror method; no significant difference but lower postoperative pain scores reported, likely due to precise technique and less collateral damage with more complete and accurate removal under direct vision results in fewer residuals and lower recurrence rates and safer due to direct vision; reduces risk of injury to eustachian tube and surrounding structures, superior teaching tool; allows precise dissection and complete resection19,20,21,22.
Radiofrequency volume reduction relies on delivering high-frequency alternating current to generate heat within the target tissue, causing cellular destruction through coagulative necrosis during the procedure and over the time the tissue is going under volume involution At the same time, coblation utilizes radiofrequency power to create a plasma field that breaks down tissue at a lower temperature, theoretically minimizing thermal damage to surrounding structures, and diathermy employs electrical current to cauterize and remove tissue. The precision afforded by radiofrequency volume reduction and coblation may contribute to reduced blood loss by enabling targeted tissue removal with concurrent coagulation of small blood vessels23,24.
Multiple randomized studies report that children undergoing Radiofrequency adenoidectomy experience significantly less postoperative pain than those undergoing curettage. Pain after radiofrequency adenoidectomy is typically mild, peaking within the first few days after surgery and returning to preoperative levels by day 7. Most children can manage discomfort with simple analgesics and resume normal eating quickly. Unlike that of a curettage adenoidectomy, patients suffer from more intense pain that needs a higher dosage of painkillers and persists for a few days longer. Radiofrequency techniques remove adenoid tissue at much lower temperatures (40–70 °C) than traditional electrosurgical (400–600 °C), minimizing collateral thermal injury to surrounding tissues, resulting in less postoperative pain and more rapid healing, however, in the case of curettage there is more damage to surrounding and prevertebral muscle fascia leading to more pain13,25,26,27,28.
The risk of hemorrhage is a critical safety concern in adenoidectomy, with the potential for both primary (immediate) and secondary (delayed) bleeding, where meticulous surgical techniques, adequate hemostasis, and appropriate postoperative care are essential to minimize this risk. While radiofrequency ablation and coblation offer the advantage of cauterizing blood vessels during tissue removal, potentially reducing the incidence of primary hemorrhage, secondary hemorrhage can occur due to infection, sloughing of necrotic tissue, or disruption of blood clots. The choice of technique should consider the overall value proposition, balancing the clinical benefits with the economic implications for the healthcare provider and the patient. In a controlled trial, it was observed that there was no significant difference between monopolar electrocautery and radiofrequency ablation regarding the return to regular activity and daily pain scores27,29.
Lowe et al. presented early (during initial stay/delayed discharge) and late postoperative hemorrhage rates (requiring readmission after discharge) between 0.07 and 0.3% and 0.07–0.2%, respectively, depending on the surgical technique used (suction diathermy and the traditional cold steel curette technique), Our mean blood loss of 31 mL significantly improved over the 50–100 mL reported using standard methods. In other radiofrequency studies, average blood loss during the operation was reported as 20–40 ml intraoperative,0–3% postoperative blood loss, while in traditional adenoidectomy methods, they reported 36-55 ml intraoperative, 5–15% postoperative blood loss30,31,32.
Numerous studies have documented improved results since the inception of endoscopic techniques. For instance, enhanced visualization and decreased recurrence rates have been linked to coblation and microdebrider-assisted adenoidectomy. These techniques, however, frequently call for extended operating times. According to research by Singh et al., the average operating time for coblation-assisted adenoidectomy was 20–30 min, which is longer than our results. It has also been noted that endoscopic adenoidectomy causes less blood loss than conventional methods. Additionally, postoperative pain scores showed quick relief during the first month, which aligns with results from studies using minimally invasive technologies33,34.
There are similarities and advancements between our results and those of other studies. For example, because of restricted visualization and challenges in guaranteeing complete tissue removal, traditional curettage-based adenoidectomy frequently reports higher rates of intraoperative blood loss, complications, and recurrence due to residual adenoid tissue.
Halitosis is a common postoperative complaint following adenoidectomy, often attributed to tissue necrosis, bacterial colonization, or delayed healing. Our study reported halitosis in 2% of cases during the first postoperative week, reduced to 1.1% by the end of the first month. These rates are consistent with the expected prevalence of postoperative halitosis in endoscopic adenoidectomy but demonstrate a faster resolution compared to traditional techniques. Previous studies have reported variable rates of halitosis based on the surgical technique employed. Coblation-assisted adenoidectomy has been associated with halitosis rates as high as 25% in the first week due to thermal effects on surrounding tissues. On the other hand, traditional curettage methods report even higher rates, often exceeding 30%, likely because of incomplete tissue removal, suboptimal healing environments, and high incidence of injury and infections, contributing to foul odour. In our study, we used vigorous saline irrigation during the operation to decrease the thermal effect of radiofrequency power on the adenoid tissue, and we removed any debris left behind during the operation. Another factor that contributed to decreasing the rate of halitosis in our cases was using hydrogen peroxide at the end of every operation, which decreases the infection rate and has an instant effect of debridement during the operation. Using an endoscope decreased the need for more damage and added a better view to the surgical precision. Also, saline irrigation and hydrogen peroxide during and after the operation decreased the heat and damage effect, decreasing the halitosis rate. The early intervention with saline irrigation and antiseptics significantly improved recovery from halitosis in coblation-assisted adenoidectomy. Results from the literature align with our study’s postoperative outcomes, which included minimal halitosis and quick recovery from pain. The early intervention with saline irrigation and antiseptics significantly improved recovery from halitosis in coblation-assisted adenoidectomy. Results from the literature align with our study’s postoperative outcomes, which included minimal halitosis and quick recovery from pain17,35,36,37. The radiofrequency delivery parameters warrant careful evaluation of their impact on postoperative pain levels compared to other radiofrequency-based and non-radiofrequency-based techniques. Halitosis, or bad breath, can be a temporary consequence of adenoidectomy, arising from tissue debris and bacterial colonization in the surgical site38.
There are a few limitations to consider. First, because the study is a case series, it does not have a control group, making it difficult to directly compare with other surgical methods. Because each case was performed by a top surgeon at a single tertiary center, selection bias cannot be ruled out, which may have limited the findings. The results validate endoscopic radiofrequency ablation as a safe, effective procedure. However, more randomized controlled trials contrasting this approach with others, such as coblation and microdebrider, must prove its superiority definitively.
Conclusion
Our study concludes by demonstrating the effectiveness, safety, and patient outcomes of endoscopic radiofrequency adenoidectomy in pediatric patients. Adenoid volume reduction with radiofrequency is safe, less painful, and has a lower recurrence rate. It also points out areas that require more research and development.
Data availability
The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
References
İkincioğulları, A., Doğu, F., ikincioğulları, A., Eğin, Y. & Babacan, E. Is immune system influenced by adenotonsillectomy in children? Int. J. Pediatr. Otorhinolaryngol. 66, 251–257 (2002).
Evcimik, M. F., Dogru, M., Cirik, A. A. & Nepesov, M. I. Adenoid hypertrophy in children with allergic disease and influential factors. Int. J. Pediatr. Otorhinolaryngol. 79, 694–697 (2015).
Nistico, L. et al. Adenoid reservoir for pathogenic biofilm bacteria. J. Clin. Microbiol. 49, 1411–1420 (2011).
Schupper, A. J., Nation, J. & Pransky, S. Adenoidectomy in children: what is the evidence and what is its role?? Curr. Otorhinolaryngol. Rep. 6, 64–73 (2018).
Redline, S. et al. The childhood adenotonsillectomy trial (CHAT): rationale, design, and challenges of a randomized controlled trial evaluating a standard surgical procedure in a pediatric population. Sleep 34, 1509–1517 (2011).
Bidaye, R., Vaid, N. & Desarda, K. Comparative analysis of conventional cold curettage versus endoscopic assisted coblation adenoidectomy. J. Laryngol Otol. 133, 294–299 (2019).
Spencer, D. J. & Jones, J. E. Complications of adenotonsillectomy in patients younger than 3 years. Arch. Otolaryngol. Head Neck Surg. 138, 335–339 (2012).
Smith, T. L. & Smith, J. M. Electrosurgery in otolaryngology-head and neck surgery: principles, advances, and complications. Laryngoscope 111, 769–780 (2001).
De Canto, L. Adenotonsillectomy complications: A Meta-analysis. Pediatrics 136, 702–718 (2015).
Cj, S., Dj, T., Ar, L. T. & Dm, A. Suction diathermy for adenoidectomy: complications and risk of recurrence. Int. J. Pediatr. Otorhinolaryngol. 71, 917–20 (2007). https://pubmed.ncbi.nlm.nih.gov/17399802/
Clemens, J., McMurray, J. S. & Willging, J. P. Electrocautery versus Curette adenoidectomy: comparison of postoperative results. Int. J. Pediatr. Otorhinolaryngol. 43, 115–122 (1998).
Fujioka, M., Young, L. W. & Girdany, B. R. Radiographic evaluation of adenoidal size in children: adenoidal-nasopharyngeal ratio. AJR Am. J. Roentgenol. 133, 401–404 (1979).
Elzayat, S., Elsherif, H. & Aouf, M. Trans-oral endoscopic assisted radio-frequency for adenoid ablation; A randomized prospective comparative clinical study. Auris Nasus Larynx. 48, 710–717 (2021).
Elzayat, S. et al. What is the most appropriate hemostatic material during pediatric adenoidectomy? A prospective comparative randomized double-blinded controlled study. Int. J. Pediatr. Otorhinolaryngol. 156, 111095 (2022).
Garra, G. et al. Validation of the Wong-Baker FACES pain rating scale in pediatric emergency department patients. Acad. Emerg. Med. 17, 50–54 (2010).
Khatri, A. & Kalra, N. A. Comparison of two pain scales in the assessment of dental pain in East Delhi children. Int. Sch. Res. Not. 247351 (2012).
Abo Elmagd, E. A., Khalifa, M. S., Abeskharoon, B. K. & El tahan, A. A. Comparative study between conventional adenoidectomy and adenoidectomy using micro-debrider. Egypt. J. Otolaryngol. 37, 56 (2021).
Huang, H. M., Chao, M. C., Chen, Y. L. & Hsiao H.-R. A combined method of conventional and endoscopic adenoidectomy. Laryngoscope 108, 1104–1106 (1998).
Makki, H., Abbas, F. & Radi, M. M. Trans-oral endoscopic radio frequency adenoidectomy vs. mirror monopolar adenoidectomy in children: A comparative study. J. Otol. Rhinol. (2019).
Juneja, R. et al. Endoscopic assisted powered adenoidectomy versus conventional adenoidectomy - a randomised controlled trial. J. Laryngol Otol. 133, 289–293 (2019).
Yanılmaz, M. & Akduman, D. A comparative study of two adenoidectomy technics for efficacy and safety: conventional curettage adenoidectomy versus endoscopic microdebrider adenoidectomy. Am. J. Otolaryngol. 44, 103807 (2023).
Datta, R. & Singh, V. Deshpal. Conventional versus endoscopic powered adenoidectomy: A comparative study. Med. J. Armed Forces India. 65, 308–312 (2009).
Thiagarajan, B. Coblation an overview. Otolaryngol. Onine J. 4, 1–6 (2014).
Vogt, K., Daine-Loza, I. & Sperga, M. Comparison of the thermal effects of coblation and radiofrequency waves in a Porcine turbinate model. Romanian J. Rhinol. 8, 157–164 (2018).
Singh, C. S. & Bhansali, S. L. A. A comparative study of cold knife adenotonsillectomy and coblation adenotonsillectomy. J. Evol. Med. Dent. Sci. -JEMDS. 4, 8818–8826 (2015).
Mösges, R., Hellmich, M., Allekotte, S., Albrecht, K. & Böhm, M. Hemorrhage rate after coblation tonsillectomy: a meta-analysis of published trials. Eur. Arch. Otorhinolaryngol. 268, 807–816 (2011).
Banhiran, W. et al. A randomized study of temperature-controlled versus bipolar radiofrequency for inferior turbinate reduction. Eur. Arch. Otorhinolaryngol. 272, 2877–2884 (2015).
Wadia, J. & Dabholkar, Y. Comparison of conventional curettage adenoidectomy versus endoscopic powered adenoidectomy: A randomised Single-Blind study. Indian J. Otolaryngol. Head Neck Surg. 74, 1044–1049 (2022).
Prussin, A. J. et al. Radiofrequency ablation vs electrocautery blinded randomized trial: impact on clinically meaningful outcomes. Otolaryngol. Neck Surg. 164, 1186–1192 (2021).
Lowe, D., Brown, P. & Yung, M. Adenoidectomy technique in the united Kingdom and postoperative hemorrhage. Otolaryngol. Neck Surg. 145, 314–318 (2011).
Nofal, A. A. B., Alnemr, M. A., Sweed, A. H. & Abdulmageed, A. Endoscopic evaluation after conventional adenoid curettage. Int. Arch. Otorhinolaryngol. 28, e487–e491 (2024).
Krishnakumar, N., Bashir, N. K. & Raj, G. Comparison of blood loss in endoscopic powered adenoidectomy and conventional curettage. Int. J. Otorhinolaryngol. Head Neck Surg. 5, 577–580 (2019).
Singh, J. & Bhardwaj, B. The comparison between microdebrider assisted adenoidectomy and coblation adenoidectomy: analyzing the intraoperative parameters and postoperative recovery. Indian J. Otolaryngol. Head Neck Surg. 72, 59–65 (2020).
Bhardwaj, B. & Singh, J. Comparative postoperative pain analysis between coblator assisted and bipolar diathermy tonsillectomy in paediatric patients. Indian J. Otolaryngol. Head Neck Surg. 71, 90–94 (2019).
Saibene, A. et al. Endoscopic adenoidectomy: a systematic analysis of outcomes and complications in 1006 patients. ACTA Otorhinolaryngol. Ital. 40, 64–71 (2020).
Brietzke, S. E. & Brigger, M. T. Adenoidectomy outcomes in pediatric rhinosinusitis: a meta-analysis. Int. J. Pediatr. Otorhinolaryngol. 72, 1541–1545 (2008).
Ali, A. A. A. E., Elsharnouby, M. K., Khalil, Y. A. E., Allah, R. N. E., Khalifa, M. & M. G. & A. E. Evaluation of endoscopic assisted Suction coagulation adenoidectomy versus traditional curettage technique. Egypt. J. Otolaryngol. 37, 122 (2021).
Soumya, S., Vissapragada, R., Le, J. & Ooi, E. H. Halitosis and pain post electrocautery adenoidectomy. Med. (Mex). 55, 312 (2019).
Author information
Authors and Affiliations
Contributions
SE; reference collection - HE; methodology – EC; revision data collection - AK; editing final draft – IG; review writing and final revision - MA; idea formulation, Statistical analysis, and final revision. The parents of the children signed formal consent for sharing in this research.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All procedures performed in studies involving human participants followed the ethical standards of the institutional research committee and were conducted with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Aouf, M., Elzayat, S., Covelli, E. et al. A 7-year experience in adenoidectomy with endoscopic radiofrequency volume reduction. Sci Rep 15, 34311 (2025). https://doi.org/10.1038/s41598-025-14437-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-14437-7