Abstract
Early stages of age-related macular degeneration (AMD) can lead to a number of visual function deficits, but the patient relevance of these deficits is largely unknown. We therefore investigated how bilateral visual function domains affected by age-related macular degeneration (AMD) are associated with patient-reports. Using data from the cross-sectional part of the MACUSTAR study with 245 individuals with AMD (34 early AMD, 168 intermediate (i) AMD, 43 late AMD), the Vision Impairment in Low Luminance (VILL) questionnaire (subscales: reading, VILL_R; mobility, VILL_M; emotional well-being, VILL_E) and visual function assessments from both eyes (best-corrected and low-luminance visual acuity, BCVA, LLVA; Moorfields acuity, MA; contrast sensitivity, CS) were included. Associations between VILL and visual function data (better and worse eyes defined based on BCVA) were investigated using age- and sex-adjusted regression models. In the overall sample, VILL_R and VILL_M were associated with all functional tests across eyes (p ≤ 0.0389), while VILL_E was associated with MA and CS (p ≤ 0.0302). Regression estimates for BCVA, LLVA, MA and CS in the better-seeing eyes were -2.70, -1.84, -1.83 and 1.08 (VILL_R); -2.71, -1.87, -1.90 and 1.88 (VILL_M), and -0.25, -0.22, -2.15 and 1.57 (VILL_E). In iAMD, CS and MA in the worse-seeing eye were associated with two VILL subscales, respectively (VILL_R and VILL_M; VILL_M and VILL_E, respectively; p ≤ 0.0395), while BCVA and LLVA in the worse-seeing eye were both associated with one VILL subscale (VILL_M; p ≤ 0.0317). CS in the better eye was associated with VILL_M (p = 0.0454). Thus, patient-reported outcomes are associated with visual function assessments in both eyes in people with AMD. Contrast vision seems particularly patient-relevant in iAMD. Our results further support the construct validity of the VILL questionnaire.
Similar content being viewed by others
Introduction
Age-related macular degeneration (AMD) is a common global cause of blindness and is associated with vision impairment, falls, loss of independence and depression1,2. Although treatments are currently available only for late AMD – i.e. the disease stage when such outcomes occur3,4 – reductions in visual function and vision-related quality of life already appear in earlier AMD stages5,6. Visual domains affected in early and intermediate AMD include night vision, contrast vision, and dark adaptation, which are rarely assessed in routine patient care and large-scale clinical trials. Also, multiple visual function assessments are time- and cost-intensive and difficult to implement in trials. Patient-reported outcome measures (PROMs), on the other hand, are easy to obtain on site or remotely, standardized, and measure concepts relevant to patients, which is increasingly requested by regulatory authorities7.
The Vision Impairment in Low Luminance (VILL) questionnaire has recently been introduced and validated to assess vision-related quality of life in AMD6,8,9. It overcomes disadvantages of other previously used instruments. The VILL was developed in accordance with the requirements of the United States Food and Drug Administration (FDA) guideline for PROM development10 and has been shown to be internally consistent, test–retest reliable, inter-mode reliable, content valid, and able to differentiate between different AMD stages6,8,9.
Several visual function assessments including best-corrected and low-luminance visual acuity, low luminance deficit, contrast sensitivity, Moorfields acuity, and rod intercept time from adaptometry11,12,13,14,15 are associated with vision-related quality of life and patient-reported symptoms of AMD. However, most of these data were obtained from visual function assessments of only one eye, which limits the ecological (i. e., ‘real-life’) external validity of these analyses.
Despite increasing recognition of the importance of patient-reported outcomes in AMD, evidence supporting the construct validity of newer, specific instruments like the VILL questionnaire remains limited. In particular, associations between bilateral functional vision measures and patient-reported difficulties under low luminance have not been thoroughly investigated, leaving a critical gap in validating such tools for use in clinical trials. We thus analyzed the association between vision-related quality of life, as assessed by the VILL questionnaire, and visual function in both eyes in the MACUSTAR study, a multi-centre European cohort study on intermediate AMD and neighbouring disease stages.
Methods
Participants
We included participants of the cross-sectional part of the MACUSTAR study. The MACUSTAR study is a multi-centre cohort study investigating functional, structural and patient-reported outcomes in intermediate AMD. It is conducted at 20 study sites across seven European countries, from which 18 sites recruited participants for the cross-sectional part referred to in this report16,17.
In summary, 586 participants with intermediate AMD and 133 control participants with bilateral early AMD (n = 34), late AMD (n = 43) or no AMD (n = 56) were recruited for the MACUSTAR study from early 2018. The disease categorization of AMD followed the Beckman classification18. Comprehensive six- to twelve-monthly assessments including visual function testing, multimodal imaging and administration of PROMs were performed in all participants with intermediate AMD or early AMD within the longitudinal part of MACUSTAR.
Within the cross-sectional part of the MACUSTAR study, 168 participants with bilateral intermediate AMD and all control participants attended an additional retest visit scheduled 2 weeks (± 1 week) after the baseline visit. Acquisition of functional, structural and patient-reported data followed procedure manuals specifically developed for the study and all functional data underwent a formal, pre-specified review procedure on a semi-annual basis to ensure that the multi-centre setting of MACUSTAR does not compromise data quality. Imaging data were graded by a central reading centre (GRADE Reading Center, University Hospital Bonn, Germany)16,17.
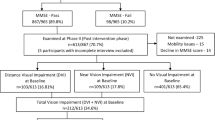
We included only participants with AMD in our analysis and thus excluded healthy controls (Supplementary Figure). We also excluded participants who only participated in the longitudinal part, where disease stage was allowed to be asymmetric.
The study protocol and additional information on the conduct of the MACUSTAR study have been published previously16,17 and can be found on the study website www.MACUSTAR.eu. Ethics committees at all participating study sites approved the conduct of the MACUSTAR study and it followed the tenets of the Declaration of Helsinki. All participants provided written informed consent. The clinicaltrials.gov registration identifier is NCT03349801.
Vision Impairment in Low Luminance (VILL) questionnaire
The VILL questionnaire is a 33-item PROM assessing vision-related quality of life in individuals with AMD, specifically designed for future use in intermediate AMD clinical trials. Its item pool was developed with extensive patient input (in-depth interviews, focus group discussions, cognitive debriefs in people with AMD) and it focuses on the visual difficulties in low-luminance and low-contrast situations which is frequently reported by people with AMD8. An initial 37 item-version of the VILL was reduced to a final set of 33 items based on the MACUSTAR data6,8. The VILL includes items from the three independent subscales “reading and accessing information” (17 items, “reading”), “mobility and safety (12 items, “mobility”) and “emotional well-being” (4 items, “emotional”). The response scale of the VILL includes four response options per item and a separate response option ”not applicable”. The VILL is scored through Rasch models which have been established and psychometrically validated in the MACUSTAR cohort previously6,8.
The VILL has been shown to be an internally consistent, test–retest reliable, content and construct valid PROM which yields similar scores across modes of administration (interview administration, self-administration via paper and electronic self-administration)6,9. Within MACUSTAR, the VILL was administered in a standardized manner, using a PROM administration manual by the clinical site personnel in the respective local language6,17.
Visual function assessments
Visual function assessments implemented in the MACUSTAR study include best-corrected (BCVA) and low-luminance visual acuity (LLVA), Moorfields acuity (MA), Pelli-Robson chart contrast sensitivity (PR-CS), mesopic and scotopic fundus-controlled perimetry (microperimetry; S-MAIA, CenterVue, Padova, Italy) as well as dark adaptometry (AdaptDx, Maculogix, Pennsylvania, USA). The assessments available from both eyes were selected as main read-outs for in this study, i.e. BCVA [logMAR], LLVA [logMAR], MA [logMAR] and PR-CS [logCS], which are all chart-based. In a supplemental analysis, average threshold from mesopic and scotpic microperimetry (mAT and sAT [dB]) as well as rod intercept time from dark adaptometry (RIT [min]) were also considered. However, mAT, sATm, and RIT were only available for one eye, which was not necessarily the better eye (i.e., the eye with higher BCVA). Reading performance was also assessed in the MACUSTAR study but not considered in our analysis due to the performance-based nature of reading speed assessments, contrasting the other visual function metrics assessed.
All functional testing procedures were conducted by specifically trained and tested staff that followed specific testing protocols as reported previously5,17,19. Visual function testing was conducted separately on both eyes in the MACUSTAR study. Following the study protocol, both eyes had the same AMD stage. Previous research supports that visual function of both eyes impacts vision-related quality of life20,21,22,23, which was the rationale for including only the visual function assessments that were available from both eyes in our main analysis. In a supplemental analysis, data from only the study eyes were considered, which were selected based on better visual acuity at screening if both eyes were considered eligible.
Statistical analysis
To investigate the relationship between the VILL subscale person measures and the above-mentioned visual function parameters of both eyes in all participants with AMD, we fitted linear regression models with VILL person measures as dependent variables and the respective functional parameters as independent variables, controlling for age, sex, and AMD stage. Next, we fitted linear regression models using the same approach in the intermediate AMD subgroup, which is the main target population of the MACUSTAR study. All analyses were conducted using R (version 4.3.0, R Core Team, Vienna, Austria). P-values < 0.05 were considered statistically significant.
Results
We included 245 participants with AMD in our analysis (early AMD, n = 34, 79% female, mean age 72 ± 6 years; intermediate AMD, n = 168, 65% female, mean age 71 ± 8 years; late AMD, n = 43, 64% female, mean age 72 ± 7 years). Mean logMAR BCVA was 0.15 ± 0.32 for study eyes and 0.20 ± 0.36 for fellow eyes (Table 1).
Vision-related quality of life and visual function across AMD stages
Absolute Pearson correlation coefficients between VILL subscale scores and chart-based visual function parameters from both eyes in all 245 participants with AMD ranged between 0.50 and 0.67 for the VILL reading subscale, between 0.48 and 0.58 for the mobility subscale and between 0.28 and 0.36 for the emotional subscale (Fig. 1). When introducing these variables into a regression model adjusted for age, sex and AMD stage, all visual function parameters were significantly associated with the VILL reading, mobility, or emotional subscale scores (Table 2). Specifically, BCVA, LLVA, MA and PR-CS were associated with VILL-Reading and VILL-Mobility in both the better and the worse eye. VILL-Emotional was associated with MA and PR-CS in the worse-seeing eye.
Correlation heatmap of VILL subscale scores (displayed in rows) and visual function testing results from both eyes (columns) across AMD stages (n = 245). BCVA, best-corrected visual acuity; BE, better eye; LLVA, low-luminance visual acuity; MAT, Moorfields acuity test; PR-CS, Pelli-Robson contrast sensitivity; WE, worse eye.
Vision-related quality of life and visual function in intermediate AMD
In the subgroup of 168 participants with intermediate AMD, BCVA, LLVA, MAT and PR-CS were associated with VILL mobility subscale scores in the worse-seeing eye when controlling for age and sex (Table 3). PR-CS was also significantly associated with patient-reports in the better eye (i.e., the VILL mobility subscale) and with the VILL reading subscale, while MAT was associated with the VILL emotional well-being subscale (Table 3). Notably, effect sizes were generally larger in models that included visual function data from the worse eye compared to the better eye.
In the MACUSTAR study, microperimetry and dark adaptometry were performed only in the study eye. Therefore, we additionally evaluated associations between VILL scores and unilateral visual function in the study eye (Supplementary Table). This analysis revealed significant associations between the VILL reading and emotional well-being subscales and average thresholds on microperimetry.
Discussion
Our findings show that patient-reported vision impairment in low luminance, as measured by the VILL questionnaire, is generally associated with bilateral visual function deficits across all AMD stages – and specifically in intermediate AMD. This further supports the construct validity of the VILL as a tool for assessing visual functioning and vision-related quality of life in AMD. Additionally, our results underscore contrast sensitivity as a key factor contributing to everyday visual difficulties, particularly in individuals with intermediate AMD.
This is the first study to investigate the association between visual function measures and patient-reported low-luminance vision impairment in individuals with intermediate AMD using the VILL questionnaire. These findings are clinically and regulatorily relevant, as they support the use of the VILL as a potential endpoint for assessing functional vision and vision-related quality of life in this patient population. Other patient-reported instruments assessing general vision-related quality of life, as well as those specifically targeting mesopic and scotopic visual functioning, have previously been compared to visual function measurements11,24,25,26. The Low Luminance Questionnaire (LLQ) was associated with dark adaptation, as reported by Owsley and colleagues27. This was consistent with a later study by another group, in which rod intercept time from dark adaptometry was highly associated with LLQ scores, alongside BCVA and contrast sensitivity, which were linked to some of the subdomains28. In a more recent paper, LLVA and low luminance deficit – defined as the difference between BCVA and LLVA – were significantly associated with LLQ composite scores, whereas BCVA, rod intercept from dark adaptation, and microperimetry thresholds were not13. Wu and coleagues used the 10-item Night Vision Questionnaire (NVQ)29 in 100 individuals with bilateral intermediate AMD and found that the PROM scores were not significantly associated with BCVA, LLVA, or retinal sensitivity measured by microperimetry12. However, the low luminance deficit was significantly greater in individuals reporting night vision difficulties in their study.
Our analysis adds to these results by showing that chart-based visual function tests were indeed associated with vision-related quality of life across AMD stages, and specifically with visual functioning of the worse eye in intermediate AMD. In contrast to previous studies, we included MAT and PR-CS in addition to BCVA and LLVA. Notably, PR-CS was significantly associated with visual functioning in intermediate AMD (i.e., reading and accessing information, mobility and safety). The association between the VILL’s mobility subscale and PR-CS was significant for both the better and worse eyes. The particular importance of contrast vision is also supported by previous qualitative data8,30. In addition to chart-based visual function tests, unilateral mean sensitivity reduction on microperimetry was associated with the reading and emotional well-being subscale scores of the VILL questionnaire in our data, contrasting the findings of Thompson et al. in a smaller cohort with varying disease stages13. Dark adaptometry in the study eye, however, did not have significant associations with any of the VILL scores, which could be related to the different testing spot in MACUSTAR compared to other protocols27. Interpreting our data in the context of similar PROMs further supports the VILL’s construct validity and suggests that the VILL is a suitable tool for future clinical studies and drug trials in the context of AMD.
The results of our study may serve as a baseline interpretation of the minimally important difference (MID) for the VILL questionnaire, using visual function as an anchor. Regulators commonly accept a three-line change in BCVA as clinically meaningful7, which could be translated into patient-relevant differences of 0.41 logits in the VILL-Reading subscale, 0.55 logits in the VILL-Mobility subscale, and 0.72 logits in the VILL-Emotional subscale. Previous data support that visual function in the better-seeing eye – used for this estimation – is a stronger predictor of vision-related quality of life than visual function in the worse-seeing eye23. However, this interpretation of our results as MIDs is limited by the absence of longitudinal data and a patient-reported anchor variable.
Our main results are based on the analysis of both eyes, which is known to better reflect vision-related quality of life than monocular vision in the context of visual impairment20,21,22,23. The particular importance of contrast vision of both eyes for vision-related quality of life in intermediate AMD is strongly supported by our findings, where PR-CS in both better and worse eyes was associated with VILL mobility subscale scores. Similar studies to ours have stratified the ‘better’ and ‘worse’ eyes based on BCVA11,12,27,28,31, while the relative value of other AMD-affected function domains (e.g., contrast vision, dark adaptation) to patients remains unknown.
Strengths of our study include the large, multicentric and thoroughly phenotyped sample with the same disease stage in both eyes; a broad battery of visual function tests performed under highly standardized settings by experienced study teams; and the validated VILL questionnaire, which was specifically developed to be used in the context of AMD, using both qualitative and quantitative methods and is scored with Rasch models. Limitations include the lack of binocular visual function testing, which we addressed by separately analysing data from the eye with better and worse BCVA. Some assessments, including microperimetry and dark adaptometry were performed in only one eye, and were thus used for generating supplementary findings only. Lastly, we were unable to directly compare associations between the VILL and visual function assessments with other PROMs, as the VILL is the only ophthalmic PROM included in the MACUSTAR study.
In conclusion, our data show that vision-related quality of life in AMD, as measured by the VILL questionnaire, is associated with a variety of visual function assessments. In intermediate AMD, contrast vision – when considering both eyes – is particularly linked to visual functioning. Our findings support the validity of the VILL questionnaire as a patient-relevant measure for future studies.
Data availability
The dataset used for this study cannot be publicly shared to protect the privacy of study participants. The data are available from the MACUSTAR consortium upon reasonable request via e-mail (dataaccess@macustar.eu).
References
Wong, W. L. et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: A systematic review and meta-analysis. Lancet Glob. Health 2, e106–e116 (2014).
Taylor, D. J., Hobby, A. E., Binns, A. M. & Crabb, D. P. How does age-related macular degeneration affect real-world visual ability and quality of life?. A syst. Rev. BMJ Open 6, e011504 (2016).
Rosenfeld, P. J. et al. Ranibizumab for neovascular age-related macular degeneration. N. Engl. J. Med. 355, 1419–1431 (2006).
Liao, D. S. et al. Complement C3 inhibitor pegcetacoplan for geographic atrophy secondary to age-related macular degeneration: A Randomized phase 2 trial. Ophthalmology 127, 186–195 (2020).
Dunbar, H. M. P. et al. Repeatability and discriminatory power of chart-based visual function tests in individuals with age-related macular degeneration: A MACUSTAR study report. JAMA Ophthalmol. 140, 780–789 (2022).
Terheyden, J. H. et al. Disease-specific assessment of Vision Impairment in Low Luminance in age-related macular degeneration - a MACUSTAR study report. Br. J. Ophthalmol. https://doi.org/10.1136/bjophthalmol-2021-320848 (2022).
Csaky, K. G., Richman, E. A. & Ferris, F. L. Report from the NEI/FDA ophthalmic clinical trial design and endpoints symposium. Invest. Ophthalmol. Vis. Sci. 49, 479–489 (2008).
Pondorfer, S. G. et al. Development of the vision impairment in low luminance questionnaire. Transl. Vis. Sci. & Technol. 10, 5 (2021).
Terheyden, J. H. et al. Interviewer administration corresponds to self-administration of the Vision Impairment in Low Luminance (VILL) questionnaire. Transl. Vis. Sci. & Technol. 11, 21 (2022).
U.S. Department of Health and Human Services, Food and Drug Administration. Patient-Reported Outcome Measures: Use in Medical Product Development to Support Labeling Claims (2009).
Pondorfer, S. G. et al. Association of vision-related quality of life with visual function in age-related macular degeneration. Sci. Rep. 9, 15326 (2019).
Wu, Z., Guymer, R. H. & Finger, R. P. Low luminance deficit and night vision symptoms in intermediate age-related macular degeneration. Br. J. Ophthalmol. 100, 395–398 (2016).
Thompson, A. C. et al. Association of low luminance questionnaire with objective functional measures in early and intermediate age-related macular degeneration. Invest. Ophthalmol. Vis. Sci. 59, 289–297 (2018).
Ivers, R. Q., Mitchell, P. & Cumming, R. G. Visual function tests, eye disease and symptoms of visual disability: A population-based assessment. Clin. Experiment. Ophthalmol. 28, 41–47 (2000).
Scilley, K. et al. Early age-related maculopathy and self-reported visual difficulty in daily life2 2The authors have no commercial interests in any device or product mentioned in this article. Ophthalmology 109, 1235–1242 (2002).
Finger, R. P. et al. MACUSTAR: Development and clinical validation of functional, structural, and patient-reported endpoints in intermediate age-related macular degeneration. Ophthalmologica. J. Int. d’ophtalmologie. Int. J. Ophthalmol. Zeitschrift fur Augenheilkunde 241, 61–72 (2019).
Terheyden, J. H. et al. Clinical study protocol for a low-interventional study in intermediate age-related macular degeneration developing novel clinical endpoints for interventional clinical trials with a regulatory and patient access intention-MACUSTAR. Trials 21, 659 (2020).
Ferris, F. L. et al. Clinical classification of age-related macular degeneration. Ophthalmology 120, 844–851 (2013).
Higgins, B. E. et al. Test-retest variability and discriminatory power of measurements from microperimetry and dark adaptation assessment in people with intermediate age-related macular degeneration - A MACUSTAR study report. Transl. Vis. Sci. & Technol. 12, 19 (2023).
Nickels, S. et al. Vision-related quality of life considering both eyes: results from the German population-based Gutenberg Health Study (GHS). Health Qual. Life Outcomes 17, 98 (2019).
Finger, R. P. et al. Visual impairment as a function of visual acuity in both eyes and its impact on patient reported preferences. PLoS ONE 8, e81042 (2013).
Vu, H. T. V., Keeffe, J. E., McCarty, C. A. & Taylor, H. R. Impact of unilateral and bilateral vision loss on quality of life. Br. J. Ophthalmol. 89, 360–363 (2005).
Hirneiss, C. The impact of a better-seeing eye and a worse-seeing eye on vision-related quality of life. Clin. Ophthalmol. (Auckland, N.Z.) 8(1703), 1709 (2014).
McGuinness, M. B. et al. Association between patient-reported outcomes and time to late age-related macular degeneration in the laser intervention in early stages of age-related macular degeneration study. Ophthalmol. Retina 4, 881–888 (2020).
McGuinness, M. B. et al. Properties of the impact of vision impairment and night vision questionnaires among people with intermediate age-related macular degeneration. Transl. Vis. Sci. & Technol. 8, 3 (2019).
McGuinness, M. B., Fraser, R. G., Tan, R., Luu, C. D. & Guymer, R. H. Relationship between rod-mediated sensitivity, low-luminance visual acuity, and night vision questionnaire in age-related macular degeneration. Transl. Vis. Sci. & Technol. 9, 30 (2020).
Owsley, C., McGwin, G., Scilley, K. & Kallies, K. Development of a questionnaire to assess vision problems under low luminance in age-related maculopathy. Investig. Ophthalmol. & Vis. Sci. 47(528), 535 (2006).
Yazdanie, M. et al. Decreased visual function scores on a low luminance questionnaire is associated with impaired dark adaptation. Ophthalmology 124(1332), 1339 (2017).
Ying, G.-S., Maguire, M. G., Liu, C. & Antoszyk, A. N. Night vision symptoms and progression of age-related macular degeneration in the complications of age-related macular degeneration prevention trial. Ophthalmology 115, 1876–1882 (2008).
Taylor, D. J., Jones, L., Binns, A. M. & Crabb, D. P. “You’ve got dry macular degeneration, end of story”: A qualitative study into the experience of living with non-neovascular age-related macular degeneration. Eye (Lond.) 34, 461–473 (2020).
Bansback, N. et al. Determinants of health related quality of life and health state utility in patients with age related macular degeneration: the association of contrast sensitivity and visual acuity. Quality Life Res. : Int. J. Quality life Asp. Treat. Care Rehabilit. 16, 533–543 (2007).
Acknowledgements
The communication reflects the author’s view and neither IMI nor the European Union, EFPIA, or any Associated Partners are responsible for any use that may be made of the information contained therein.
Funding
Open Access funding enabled and organized by Projekt DEAL. This project has received funding from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreement No 116076. This Joint Undertaking receives support from the European Union’s Horizon 2020 research and innovation programme and EFPIA.
Author information
Authors and Affiliations
Consortia
Contributions
JHT, RPF and CB designed the study. Investigators of the MACUSTAR study and their team members collected the data. JHT, CB, HMPD, FGH, DPC, UFOL and RPF analyzed and interpreted the data, SP, NZ, AMB, MS, SL and MS authors interpreted the data. JHT, CB and RPF were main contributors in writing the manuscript. All authors read, substantively revised and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
JHT: Beyer (C, F, R), Novartis (F, R), Roche (F), Okko (R) CB: None HMPD: Boehringer Ingelheim (C) SP: Novartis (E) NZ: Novartis (E) AMB: Apparatus and method for retinal measurement; Patent number 9492081 (P) MSa: Heidelberg Engineering (F), Optos (F), Carl Zeiss Meditec (F), CenterVue (F) SL: Bayer (E) MSc: None FGH: Acucela (C, F), Allergan (F), Apellis (C, F), Bayer (C, F), Boehringer-Ingelheim (C), Bioeq/Formycon (C, F), CenterVue (F), Ellex (F), Roche/Genentech (C, F), Geuder (C, F), Graybug (C), Gyroscope (C), Heidelberg Engineering (C, F), IvericBio (C, F), Kanghong (C, F), LinBioscience (C), NightStarX (F), Novartis (C, F), Optos (F), Oxurion (C), Pixium Vision (C, F), Oxurion (C), Stealth BioTherapeutics (C), Zeiss (C, F) DPC: Apellis (C, F), Santen (F, R), Allergan/AbbVie (R), Thea (R), Janssen (S) UFOL: F.Hoffmann-La Roche. Ltd. (E) RPF: Bayer (C, F), Novartis (C, F), Roche/Genentech (C), Alimera (C), Böhringer-Ingelheim (C), Santhera (C), Ellex (C), Novartis (C, F), Zeiss (F), Heidelberg Engineering (F), CenterVue (F), Biogen (F).
Ethics
Ethical approval was obtained from the Institutional Review Boards of all involved centres and all participants gave written informed consent. The study protocol followed the principles of the Declaration of Helsinki.
Disclaimer
The communication reflects the author’s view and neither IMI nor the European Union, EFPIA, or any Associated Partners are responsible for any use that may be made of the information contained therein.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Terheyden, J.H., Behning, C., Dunbar, H.M.P. et al. Patient-reported vision impairment in low luminance relates to visual function in age-related macular degeneration: A MACUSTAR study report. Sci Rep 15, 35223 (2025). https://doi.org/10.1038/s41598-025-14553-4
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-14553-4