Abstract
Behavioral ecologists frequently focus on a single modality of wildlife response to disturbance, which can limit inference because different antipredator responses reflect various aspects of predation risk management. We investigated intrapopulation variation in tolerance to human-associated risk within a grizzly bear-ecotourism system in Nuxalk Territory, considering behavior results in tandem with spatial data from genetically tagged individuals. Whereas our behavioral analysis revealed no effects of ecotourism on alertness, our central measure of tolerance, we observed variation in space-use among individuals (n = 80). Only 12, primarily females (n = 10), showed preference for the area of highest ecotourism activity; others showed little to no use of the area, despite its close proximity and high resource abundance. These patterns suggest that behavioral data may have been biased towards individuals tolerant enough to coexist with ecotourism. Examining behavior of ostensibly tolerant ecotourism individuals alone would have overlooked intrapopulation variation in space-use at broader spatial scales. A comprehensive assessment that simultaneously draws upon both spatial and behavioral dimensions may therefore provide richer insight into coexistence than either lens alone. More broadly, coexistence dynamics in this and other systems might exclude some individuals within populations that are not tolerant enough to participate.
Similar content being viewed by others
Introduction
Although researchers commonly examine only one behavioral dimension, responses by wildlife to risk can manifest in multiple ways among individuals (e.g., in habitat use, flight initiation distance, vigilance behavior). Some individuals may spatially avoid habitats with pronounced human-associated risk, whereas others may tolerate the presence of risky stimuli by responding behaviorally or physiologically1. Owing to a broad predatory niche and high levels of exploitation over millennia2,3, humans are commonly perceived as risky stimuli—predators—by wildlife4. Even seemingly benign or low-level forms of human activity and infrastructure can modify wildlife behavior, displacing them in space or time5,6,7,8. Given such variation in antipredator responses to diverse human cues, a multi-lens perspective that examines more than one dimension of response may provide richer insight into our understanding of wildlife tolerance, especially in scenarios in which human-wildlife coexistence is desired.
Placing emphasis on only one modality of response effectively limits the conclusions that can be drawn. Specifically, whether individuals observed to invest in anti-predator behaviors (e.g., vigilance) represent the only individuals affected is unknown without examining if there was prior spatial segregation of the population; spatial segregation of animals may have already occurred based on their willingness to be in the presence of risk in the first place9, biasing behavior results to individuals willing to accept the mere presence of risk. Similarly, whether animals avoid disturbance spatially may not provide a comprehensive indication of the extent to which animals are disturbed without considering additional measures of behavior10. A comprehensive assessment that simultaneously draws on both spatial and behavioral dimensions can therefore provide more insight than either lens alone. Indeed, antipredator behaviors are not always expected to manifest consistently, which justifies an approach for considering more than one behavioral response11.
Variation within species in tolerance related to human-dominated spaces can manifest in different ways owing to diverse drivers. On one hand, differences in risk perception among age-sex classes and individuals can elicit spatial segregation in a heterogenous environment according to the level of risk different animals perceive12. For example, Wilkinson et al.13 found that individual coyotes (Canis latrans) in urban environments displayed differing levels of boldness toward humans; some were more likely to engage in conflicts or occupy densely populated areas. On the other hand, individuals might not sort spatially based on perceived risk from humans but rather based on perceived protection. Prugh et al.14 found that mesopredators shifted into human-modified landscapes to avoid larger and dangerous apex predators, despite the increased risk of human-caused mortality. Similarly, individuals or the groups they compose (i.e., family groups) might perceive humans as ‘shields’ that indirectly provide protection from greater perceived risk from conspecifics15,16. For example, the presence of ecotourists can provide a perceived shield from infanticidal adult male grizzly bears for other age-sex classes (i.e. sub-adults, females with young;16,17,18. Consequently, understanding the extent to which human activity might influence intrapopulation variation in space-use and behavior may increase our understanding of variation in tolerance within populations and its underlying processes.
Wildlife ecotourism is one context that can present myriad influences across a range of taxa globally19 and commonly creates scenarios in which human disturbance varies spatially and behavioral trade-offs are relevant. For example, several case studies have demonstrated that grizzly bears show varied anti-predator responses towards humans [see20 for review], including diverse responses to ecotourism [see21 for review]. Recreational activity by people can displace bears from preferred habitat, increase energetic costs, and reduce nutritional intake22,23. In other cases, bears may habituate to people over time to reduce the detrimental effects of a stress response24. Ecotourism sites also tend to be resource rich, offering abundant natural foods [e.g.,17], or even provisioned foods by ecotourism operators [e.g.,25]. Accordingly, some individuals subject to ecotourism may engage in trade-offs; specifically, they may be unwilling to avoid people at the cost of forgoing resources10, but instead may employ behavioral strategies to reduce perceived risks. Huveneers et al.26, for example, found considerable individual behavioral variation in fine-scale spatial distribution among white sharks (Carcharodon carcharias) in response to cage-diving tourism; some individuals spent more than 60% of their time within 60 m of cage-diving tourists and tethered bait, while others were mostly detected further than 120 m. Despite the valuable inference provided from this body of work, how wildlife individuals might vary both in their presence—and for those present, their behavior—at ecotourism sites is less understood. Such insight requires knowledge of space-use by individuals within, and beyond, areas of ecotourism, as well as detailed behavioral data at ecotourism sites.
Here, we examine whether grizzly bears show intrapopulation variation in behavioral tolerance to ecotourism in the Atnarko River Corridor, which is located in unceded Nuxalk Territory in coastal British Columbia, by quantifying their behavioral states. Concurrently, we collected space-use data for genetically tagged grizzly bears, which we considered to supplement and inform insight from behavior observations. Our hypotheses consider how the population might vary in how individuals or the age-sex classes they constitute perceive humans (as risky or benign), and modulate their behavioral tolerance accordingly. We consider our spatial data as context with which to consider the results from our behavioral analysis, with the objective of providing richer insight into bear activity than the behavior dataset could alone. Specifically, the objective of collecting two datasets simultaneously–and considering them collectively–was to evaluate the possibility that results from the behavioral analysis might be biased towards individuals that spatially co-occur with high ecotourism activity.
The Atnarko River provides bears access to aggregations of spawning salmon (Oncorhynchus spp.) during hyperphagia—a critical period to amass large amounts of energy before denning. The high ecotourism activity areas in particular (‘land- and boat-based’ area, below) host readily-accessible spawning aggregations throughout the study area17. Wildlife ecotourism in this area thus provides a system in which non-lethal human stimuli are present, trade-offs related to foraging under the perceived risk of predation (by humans and male bears) are relevant, and potential grizzly bear responses in the form of anti-predator behavior are measurable.
We made several predictions about intrapopulation variation in behavior based on theory relating to tolerance, predation risk, and human shielding. Specifically, owing to risk-foraging trade-offs under predation risk theory, we considered multiple hypotheses. We hypothesized that grizzly bears tolerant enough to spatially co-occur with humans where salmon is plentiful could offset such potential risk by investing in anti-predator behavior. Accordingly, we predicted a positive association between alertness and ecotourism metrics. On the other hand, the relatively predictable tourist activity could be perceived as benign, eliciting little responses. Specifically, aligning with the human shield hypotheses, female with young would be less likely to be alert with greater ecotourism intensity. Additionally, because our behavioral state data comprise females, females with young, and subadults (see Methods), we also considered the presence of other bears as risky stimuli, predicting a positive association between alertness and other bear presence. Finally, we predicted a positive association between salmon biomass and alertness, reasoning that when food resources are limited, allocating energy away from fishing would be perceived as more costly than allocating energy to anti-predator behaviors because foragers with lower energy reserves will accept greater risk than foragers that have adequate energy reserves27. Our results may help identify individuals and contexts that are potentially more sensitive to the influences of ecotourism (e.g., boat-based versus land-based ecotourism) and thereby provide evidence-based guidance for consideration by ecotourism management and more broadly in the context of coexistence.
Methods
Study area
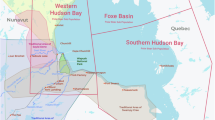
The study occurred in the Atnarko River corridor (hereafter ‘Atnarko’; Fig. 1A) in Tweedsmuir Provincial Park, British Columbia, which is located in the unceded Territory of Nuxalk First Nation. Genetic sampling and behavioral observations occurred concurrently with a companion study using an array of camera traps in the same area [see17 for details].
(A) Atnarko River Corridor hair snag and behavior sites. Inset A1: Pacific Coast of British Columbia, Canada, with red frame denoting Inset A2; Inset A2: Bella Coola Valley and surrounding tributaries, with red frame denoting A3; Inset A3: Atnarko River Corridor study area, including behavior sites and hair snag sites, where hair snag sites are categorized by spatial treatment. (B) Heatmap illustrates the spatial detection histories of each individual grizzly bear. Each column represents an individual (data were subset to include individuals that were detected at least twice; n = 80 individuals out of original 118). Each row represents a spatial treatment, and the colour ramp represents the proportion of detections per individual that occurred in each spatial treatment. We identified three clusters (three identified as optimal number of clusters via elbow method in k-means clustering), which cluster individuals with similar spatial detection histories (B1, B2, and B3). That is, individuals clustered together were most similar to each other in their space-use. Annotated values below each column denote detection frequencies.
The abundance and activity of people in the park vary spatially during the salmon spawning season. We defined ‘spatial treatments’ according to variation in bear viewing and other human activity: a ‘no tours’ reference area furthest upstream, a ‘land-based tour’ area in the middle, and a ‘land- and boat-based tour’ area furthest downstream17. Activities in the ‘land- and boat-based tour’ area, which have the potential to elicit perceived risk by bears, are varied but include camping, swimming, angling, hiking, paddling, picnicking, mushroom picking, and bear viewing (on land and aboard drift boats). Owing to a well-established industry of bear-viewing infrastructure and drift boat tours, the ‘land- and boat-based tour area’ (Fig. 1A) hosts on average ~ 2 orders of magnitude more people compared with the ‘no tours’ area of the corridor ~ 35 km upstream (average weekly visitors to ‘land- and boat-based tour’ area was 1,363 across years, compared with 11 in the ‘no-tours’ area and 14 in the ‘land-based tour’ area17.
We collected behavioral state data from two sites within the ‘land- and boat-based’ area, which varied in their management of human behavior and activity (Fig. 1A). One site was the Belarko bear viewing platform (‘Belarko’), a purpose-built bear viewing platform staffed by BC Parks and the Nuxalk Nation that included an electric fence and a regulated entry and exit system. The second site was BC Parks Fisheries Pool campground (‘Fisheries Pool’), an area designed for camping and recreation, including swimming, picnicking, and angling. Although signage to regulate the behavior of campers and day-users is posted in prominent locations at Fisheries Pool, bear viewing is self-pursued by the public at their own risk. Ecotourism boats were also present in the river at and between these two ‘land- and boat-based’ sites. Up to 11 commercially operated ‘drifts’ (slow-pace rafting-based bear viewing with up to six guests each) were permitted per day. Drift boats launched ~ 200 m upstream from Belarko and hauled out at Fisheries Pool.
Field methods
Space-use data collection using genetic tagging
We non-invasively sampled bears to identify how individuals used the Atnarko corridor. Specifically, we deployed passive, non-baited hair snags28,29 to genetically identify individuals using microsatellite data30,31. Snags were set of barbed wire at ~ 0.5 m height32 every ~ 0.5–2 km along the Atnarko river across bear trails or riverside banks where bears were commonly observed. We monitored hair snag sites for 317 days over three years during the late summer and autumn salmon runs (late July to early November; n = 28 sites in 2019 for 113 days; n = 29 sites in 2020 for 101 days; and n = 28 sites in 2021 for 103 days). In 2021, there were 7 snags in the ‘land- and boat-based’ area (~ 4 km river network distance), 13 snags in the ‘land-based’ area (~ 22 km river network distance), and 8 in the ‘no tours’ area (~ 9 km river network distance) (Fig. 1A). Across years, crews revisited hair snag sites and sampled hair on average every ~ 9 days (\(\bar{x}\) = 8.6 days in 2019, \(\bar{x}\) = 9.0 days in 2020, and \(\bar{x}\) = 8.8 days in 2021), collecting 1,806 samples. We subsampled high-quality samples (with abundant guard hair; n = 713), identifying 118 individual bears (73 female and 45 male) over the three years. Research was approved by University of Victoria’s Animal Care Committees (2019-004).
Behavioral state data collection using observational approach
Sampling methods
We collected behavioral state data from early August to late October, 2019–2021. The first sampling sessions (i.e., time blocks awaiting bears to be present and if bears were present, observation periods of collecting behavior data; defined below) of the season occurred after all hair snags had been installed. Sampling sessions occurred continuously from ~ 2 weeks before to ~ 2 weeks after bear viewing season (bear viewing season occurred August 15-October 15), and during diurnal and crepuscular periods. We defined an ‘observation period’ as the time during which a bear was present during a sampling session. All observation periods occurred between 0600 and 2100 h. The two sites were sampled on a rotating schedule of ~ 4-hour blocks to capture different photic regimes and periods of human activity. In total, we recorded 35 h and 58 min of bear behavior during observation periods; 18 h and 46 min at Belarko and 17 h and 12 min at Fisheries Pool.
We collected behavioral state data by video-recording bear behavior for the full duration of the observation period. We recorded behavior with a Nikon® camera (Z6 body, 200–500 mm lens). We recorded behavior continuously, and behaviors were later coded from the video recordings (see ‘Coding behavioral states’). We collected measures for ecotourism and behavioral state data simultaneously. While recording bear behavior, we also counted boats and people on land at ~ two-minute intervals (herein ‘counts’). Sometimes there were observation periods when counts were more challenging at those intervals (for example, if people were walking around, coming and going, or there were many people and boats), which resulted in longer times between counts. We therefore excluded observation periods if counts were on average more than 5 min apart within an observation period (n = 9). We also excluded observation periods where only one count occurred (n = 2) because we were interested in examining potential lead effects (see Analysis), which require more than one count per observation period. These steps resulted in a mean of 2.5 min between counts across 107 observation periods. The mean number of counts per observation period was 9.0, with a range of 2 to 33 and a median of 7.
We attributed behavioral states to each ecotourism metric. We herein refer to these as ‘scan samples’33 and each served as a case (i.e., datum) in our analysis (below). Whereas other behavioral sampling approaches examine all occurrences of certain behavioral events of interest (e.g., focal sampling33, our approach of extracting behavioral states from video recordings at the same timestamp at which ecotourism metrics were recorded supported precise alignment in time between measures of human activity and bear behavior.
Selecting a bear for observation
The observation period (and video recording) began when a bear entered our view. If a bear went out of view but returned within ten minutes, we considered the two portions a single observation period. If we were observing a family group, we collected behavior data from the mother. We determined bear sex by direct observation of genitals, including vulva hair, urination patterns, or presence of young, or by opportunistically consulting local ecotourism guides in the field if they were present, based on their knowledge of the individual. Additionally, given that grizzly bears are sexually dimorphic34, age-class and sex may be identified from size and body shape, provided a clear view of the bear’s profile and before extreme weight gain in the mid to late fall. We used features in Timelapse Image Analyzer (https://timelapse.ucalgary.ca/) to aid in identifying bear sex (e.g., playback speed, magnification tool).
We could discern some but not all individual bears, enabling a minimum estimate of numbers. We used physiognomic features, such as patterns of scars or wounds, hair coverage on outer ear, and hair pattern on muzzle35 to attempt to identify individuals. Despite this approach, we could not confidently identify all bears visually. Nonetheless, after subsampling (details below), we could estimate individual identifications for 81% of observation periods in 2019 (10 individuals [4 adult female, 4 female with young, and 2 subadult] across 27 of 33 observation periods), 65% of observation periods in 2020 (9 individuals [4 adult female, 1 female with young, and 4 subadult] across 21 of 32 observation periods), and 82% of observation periods in 2021 (18 individuals [8 adult female, 10 female with young] across 37 of 45 observation periods). Owing to changes that can occur across years36,37,38, separated by nine months of no observations, we did not attempt to match individuals among years. Accordingly, we could not consider individual identification (i.e. as a random factor) in our analyses, representing a constraint imposed by data limitations.
Coding behavioral States
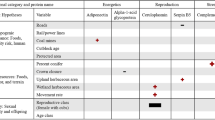
We collected data for three behaviors: ‘alertness’, ‘fishing’, and ‘other’ (Table 1 and Supplementary Information, Figure S1). Because detection of a stimulus is the outcome of vigilance39, we identified alertness as more appropriate than vigilance to examine bears that appeared to be monitoring stimuli from people and other bears. That is, people seldom go undetected by bears in an ecotourism context; therefore, vigilance may not accurately describe monitoring of stimuli in every case. We note, however, that subtle and overt alertness can include routine and induced vigilance, respectively (Table 1)40. We had originally used an ethogram to classify grizzly behavior, which included a comprehensive potential scope of behaviors we originally expected be observed (e.g., non-agonistic conspecific behaviors, locomotion [walking, swimming], non-locomotory postures [sitting, lying down]). However, behaviors other than Fishing, Alertness, and Locomotion seldom occurred. We suspect this is attributed to the time of year and hyperphagic state of grizzlies. Accordingly, we grouped all non-alertness and non-fishing behaviors into ‘Other’. Such an approach also allowed us to focus on the two dominant behavioral states, which were also the two most closely related to our hypotheses. Behavioral states were then attributed to counts for ecotourism metrics by timestamp (see Sampling methods above). We tested for inter- and intra-observer agreement in behavioral states between the two behavior coders. Inter- and intra-observer precision test results were 88.57% and 93.55% agreement, respectively.
Analysis
Individual space-use from genetic tagging
We created a visualization to illustrate how individuals showed different spatial detection histories. We defined one spatial detection as any detection of a genetically tagged individual per revisit (revisits occurred on average every 9 days; see Methods) to a hair-snag site (i.e., if there was more than one hair sample from an individual within a revisit, it was defined as one detection). Spatial detection histories are a function of the proportion of detections per individual that occurred in one or more spatial treatments. Limited detection data precluded any detailed spatial statistical approach. Instead, we examined detection histories of each individual bear by constructing a clustered heatmap with R package ‘pheatmap’ (version 1.0.12; R Core Team, 2018). Specifically, we used a hierarchical clustering algorithm to categorize individuals based on the similarity of their spatial detection histories, whereby we created a matrix of spatial detection histories with individuals as columns and spatial treatments as rows. Our matrix was used as input to the ‘pheatmap’ function, which applied hierarchical clustering to both rows and columns to visualize patterns of similarity in spatial detection histories among individuals. To determine the optimal number of clusters, we employed the ‘elbow’ method to identify the point at which the total intra-cluster variation, or total within-cluster sum of squares (WSS), was minimized (Supplementary Information; Figure S2). We used the ‘fviz_nbclust’ function from the ‘factoextra’ package (version 1.0.7) to plot WSS against different numbers of clusters, and selected the ‘elbow point’ as the optimal number.
Behavioral states
We used a multinomial model to examine the potential effects of ecotourism and environmental variables on the probability of a behavioral state (Table 1) being ‘alertness’ or ‘fishing’ with ‘other’ behavior as the reference category. Due to small sample sizes at upstream sites (230 hours at upstream sites across years, yielding only 9 observation periods), we analyzed behavioral state data exclusively from bears that visited ecotourism sites in the ‘land-and-boat-based’ area, which hosted considerable human visitation variation both within and across observation periods (Figures S3-S6). Our case was a scan sample, defined by a behavioral state (see Methods)33. After subsampling, described below, scan samples totalled 936 across years; 233 in 2019; 374 in 2020; and 329 in 2021) across 107 observation periods, which summed to 35 hours and 58 minutes of time observing bear behavior.
We excluded males due limited data for this sex. Although we initially included all age-sex classes (adult males, adult females, subadults, and females with young), low samples sizes for males resulted in convergence problems with all models that included age-sex class when adult males were included. As such, we excluded the limited data on adult males (n = 4 observation periods) from our analysis.
We constructed a set of a priori candidate models that considered combinations of multiple environmental- and ecotourism-related variables (i.e., ‘environmental models’, ‘ecotourism models’, and ‘lead ecotourism models’; Table 2). We compared each model set against null and environmental-only models. Models included weekly salmon biomass (see Field et al.17 for comprehensive methodological details for estimating salmon biomass), presence/absence of other bears, number of people viewing bears from the shore, number of boats, and bear age-sex class (i.e., female with young, adult female, and subadult). We included salmon biomass and other bear presence in each model (all ‘environmental’ and ‘ecotourism’ models, and excluding the null model), reasoning that behavioral states would always be influenced by food availability and presence of conspecifics, as related to predation risk41. In ‘ecotourism’ models (Table 2), we considered people visiting Fisheries Pool and Belarko who were viewing bears from shore (herein ‘visitors’) and/or the number of boats, reasoning that one or both measures of human activity may affect behavioral states, also related to risk perception4,17. Additionally, in ‘lead ecotour’ models, we considered lead effects of boats and visitors (i.e., boats t + 1 and boats t + 2, which represent the counts of these measures in the following 1 and 2 scans, respectively), reasoning that bear behavior might not only be influenced by human activity in ‘real time’ but also before a boat is visible to observers, potentially heard or smelled by bears in the minutes prior. Finally, in ‘ecotourism’ models we considered an interaction between boats and visitors, reasoning that the number of boats might be important, but only when visitors were also high in numbers. We nested observation periods (i.e., multiple scans covering the duration over which a bear was present) within site and year, and included observation periods as a random effect in all models. As noted, owing to uncertainty in visually identifying individuals, we did not include individual as a random effect.
We evaluated the performance of our models. All models had an over-dispersion parameter below 1.1542. We centered continuous predictors and scaled (divided by 2 SDs), back-transforming to raw data in figures. We calculated a variance inflation factor for each co-variate in all models to test for collinearity; all values were below 2, other than the multiple variables related to boat presence (t, t + 1, t + 2), which were highly collinear. Accordingly, we only considered one time period’s measure of boats and visitors in a single model. We assessed the relative performance of models by comparing AIC values, focusing inference on top performing models (i.e., those with Δ AIC < 2). All statistical analyses were performed using R (version 4.3.2, R Foundation for Statistical Computing), and the study area map (Fig. 1A) was created with QGIS Desktop 3.38.1.
Results
Individual space-use from genetic tagging
We identified 118 grizzly bears (73 female and 45 male) along the Atnarko river. Of those 118 bears, 80 (68%) were detected at least twice. We subset those individuals with multiple detections, which resulted in a dataset of 53 females and 27 males. Across years, individuals were detected on average 4.3 times (range = 2–22; SD = 4). Forty-four of these 80 (55%; 31 female and 13 male) were detected in more than one year, and thirteen (16%; 9 females and 4 males) were detected in all three years. We pooled data across years to assess any patterns of space-use among the three spatial treatments by these multiple-detected individuals.
Hierarchical clustering identified three clusters of bears based on their similarities in spatial detection histories (Fig. 1B; with clusters delineated as B1, B2, and B3). Each cluster comprised individuals that were most similar to each other in their space-use across years. Cluster B1 comprised 12 individuals (10 female and 2 male), all of which were detected within the ‘land- and boat-based’ area at high proportions of detections. Cluster B2 comprised 31 individuals (19 female and 12 male), all of which were detected in high proportions within the ‘land-based’ area. Cluster B3 comprised 37 individuals (24 female and 13 male), all of which were detected in high proportions within the ‘no-tour’ area.
Cluster B1, which showed spatial preference for the ecotourism area, comprised individuals that were the most spatially constrained (i.e., contained the highest within-cluster proportion of individuals (75%) that were detected exclusively in one spatial treatment). The second most spatially constrained cluster was B3 (67% of individuals were detected exclusively in the ‘no tour’ area), and the least spatially constrained cluster was B2 (45% of individuals were detected exclusively in the ‘land-based’ area). In these B2 and B3 clusters, a higher proportion of individuals crossed boundaries (at a higher frequency), but mostly did so only into the adjacent land-based or reference, and not the ecotourism-dominated ‘land- and boat-based’ area.
Behavioral States
Inference from our top model suggested that only salmon biomass influenced behavioral states (Fig. 2A). Specifically, the only evident pattern was a strong negative association (P = 0.002) between salmon biomass and the probability of alertness (Fig. 2B); the top model also included whether other bears and boats (t + 2) were present, but neither influenced the probability of alertness. A similar and similarly ranked model (Δ AIC = 0.002) likewise identified only a strong negative association between salmon biomass and the probability of alertness (Supplementary Information; Table S1; Figure S7). There were no associations between any co-variate in the top models and the probability that a behavioral state was fishing (Supplementary Information; Table S1).
Top multinomial model (Δ AIC = 0.0) predicting ‘fishing’, ‘alertness’, and ‘other’ behavioral states among grizzly bears subject to ecotourism in the Atnarko River Corridor, Nuxalk Territory, British Columbia (2019–2021). (A) Parameter coefficients and CIs for fixed effects, centered and scaled by two standard deviations. Red dots represent parameter coefficients and black lines span 95% CIs. (B) Probability that a bear was alert as a function of salmon biomass. Figure is backtransformed to raw data.
Discussion
To understand how coexistence might manifest, we assessed whether and how a grizzly bear population might show intrapopulation variation in tolerance to ecotourism activity through the consideration of not only a behavioral analysis but also visualization of space-use as informed by our genetics data. Our behavioral observations that occurred exclusively in the ‘land- and boat-based’ area considered how ecotourism intensity (number of visitors, number of boats) and environmental variables (presence of other bears, salmon biomass) might affect fishing and alertness behavior. Our findings revealed no evidence for an effect of ecotourism metrics on grizzly behavior. Instead, we found a negative association between salmon biomass and alertness, a pattern opposite to predictions based on predation risk theory. Our companion spatial data revealed only a small subset of individuals (n = 12 of 80 individuals detected at least twice) showed spatial histories that favoured this area of pronounced human activity, despite the high mobility of grizzlies and the abundant salmon resources present at ecotourism sites. This pattern suggests that the population could include distinct subgroups that vary in their space-use among treatments (though, see limitations below). Most of these individuals (n = 10 of 12) were female (Fig. 1B). While the smaller sample size in this area raises the possibility that this female bias may be a sampling artifact (see limitations below), the pattern is also consistent with previous support for a human shield effect in the same study area, as assessed with remote camera data17.
Among those bears that use the land- and boat-based area, human activity did not influence the behavioral metrics we recorded. Contrary to our prediction that emerged from predation risk theory, whereby bears tolerant enough to spatially co-occur with humans would offset risk by investing more in alertness behavior when human activity was higher, we found no such evidence. Instead, the bears we observed in the ‘land- and boat-based’ area during daylight and crepuscular hours demonstrated two dimensions of tolerance: they were present, and they did not respond behaviorally to ecotourism. Nisbet43 defined tolerance as “the intensity of disturbance that an individual tolerates without responding in a defined way” (p. 315), and tolerance can be attributed to animals when they permit close proximity by humans without overtly responding or fleeing44. Our ‘non-response’ (no relationship between alertness and either ecotourism metric) result suggests behavioral tolerance among those individuals that do not avoid the ecotourism area. We infer that the stimuli at ecotourism sites were perceived as non-novel, predictable, specific, and low risk. However, we note that remote camera data previously suggested that bears in the same area and periods responded in their activity rates to the number of visitors at a weekly scale; specifically, bear activity decreased with higher visitor numbers/week17. Responses of bears to ecotourism thus appear to be measurement- and scale-dependent.
Alertness behavior varied solely as a function of salmon biomass, whereby contrary to our prediction, grizzlies were less likely to be alert when salmon availability was high. Should this be representative of patterns elsewhere in grizzly bears, this result suggests that the fitness benefits from increased food intake are greater than gains from increased alertness. We speculate that such a pattern could arise in a species that undergoes hyperphagia. This is a life history strategy marked by periods of extraordinarily intense food consumption made possible by the ability to store readily the body reserves extra calories provide45. Notably among females, the consumption of salmon positively correlates with multiple measures of over-winter survival and reproductive success45. Thus, we speculate that investing in anti-predator behavior could reduce time spent foraging at times when food is hyper-abundant and easily-acquired, thus imposing potential fitness costs46. This interpretation, however, is tempered by the reality that we observed no variation in the presence of the fishing behavioral state over the range of salmon biomass. Data and theory from other contexts, however, provide insight. Notably, Repasky47 found support for a negative relationship between vigilance and food patch profitability in Sage Sparrows (Amphispiza belli) and Black-throated Sparrows (A. bilineata), suggesting that wintering birds near starvation are confronted with starvation versus predation trade-offs. Moreover, McNamara and Houston48considered a model in which an animal can control both its probability of starvation and its probability of predation, whereby increases in food requirements will cause an animal to increase their predation risk to avoid starvation. Although as large-bodied omnivores grizzlies are at low risk of starvation, we suspect a similar trade-off might be relevant to another fitness measure (reproduction). Finally, we note that the subordinate age-sex classes, which comprise our behavioral data, are seemingly tolerant to real-time measures of human activity (number of visitors and boats), perhaps predisposing them to allocate only modest time to alertness when salmon are abundant during hyperphagia.
While the extent to which spatial variation in genetically tagged individuals relates to ecotourism activity compared with other environmental factors is unknown, spatial patterns provide context with which to consider results from our behavior analysis. We differentiated the spatial behavior of genetically marked individuals by examining their space-use, finding three distinct subgroups that comprised individuals with minimal intra-cluster variation in their spatial detection histories (Fig. 1B; with clusters delineated as B1, B2, and B3; see also Figure S8). Individuals that composed cluster B1 were the most spatially constrained; 75% of these individuals were never detected outside of the ‘land- and boat-based’ area, which represents the smallest portion of the study area (~ 4 km river network distance). By contrast, the least spatially constrained individuals composed cluster B2, whereby only 45% of individuals were detected exclusively in the ‘land-based’ area (~ 22 km river network distance), and the remaining 55% traversed spatial treatments, with most individuals entering the adjacent reference area upstream that hosts very limited human activity. 67% of individuals that composed cluster B3 were never detected downstream from the ‘no-tour’ area, which hosts ~ 2 orders of magnitude less people than the ‘land- and boat-based area’17. These findings suggest the possibility of fine-scale spatial variation, but the extent to which this spatial variation relates to ecotourism activity compared with other environmental factors is unknown (alternative hypotheses might relate to resource availability, conspecific competition, or home range patterns; see limitations below). We note, however, that ecotourism sites occur near the mouth of the Atnarko River, where salmon congregations are dense and most available to fishing bears17. Accordingly, we find an explanation that individuals are likely detected where resources are most accessible plausible but not entirely comprehensive in that by itself it cannot exhaustively discount the possibility that risk-adverse individuals will forgo resource acquisition to avoid human-associated risk.
Our spatial data allowed us to consider the possibility that the behavioral tolerance we infer from our behavior analysis is attributed to individuals that will spatially tolerate ecotourism presence in the first place. Given the high mobility of grizzly bears, and considering the relatively small study area in the context of their home ranges (i.e., river network distance of ~ 35 km, which falls well within potential travel distances inside the home range of male and female grizzly bears in the valley49, our findings suggest the possibility of spatial segregation among individuals aligning with the spatial heterogeneity of risk, as seen in other grizzly bear systems50. Finnegan et al.51 demonstrated support for the Resource Dispersion Hypothesis (RDH) in the summer, a season of increased resource availability, finding that bears increased their space-use to access dispersed and patchy resources during the salmon spawning season. They noted that bears likely adhered to the RDH as they moved between patches to gain more diverse nutritional resources. Moreover, Leacock et al.52 found that collared Kodiak brown bears selected for greater stream length during the sockeye salmon (Oncorhynchus nerka) spawning season, reflecting relative ease of travel alongside streams and rivers and the fact that bears often move along streams to exploit stretches with high salmon concentrations. We therefore consider the possibility that in a case where individuals are spatially segregated according to risk perception, behavior data may be biased to ecotourism-tolerant individuals. Indeed, some individuals were seldom or never detected in the areas with highest human activity, and others were more flexible in their space-use.
Although our spatial data do not directly test human shield hypotheses, sex ratios also differed among spatial treatments, consistent with the possibility of human shielding. The proportion of male detections increased with distance from the area with highest human activity. Field et al.17 found that camera trap detections were more likely to be male at night and when visitor numbers were low, suggesting a human shield effect in the same study area. This suggests a possibility that males detected by our hair snags in the ‘land- and boat-based’ were captured at night, which would align with our infrequent observations of males during dawk to dusk observations in our behavioral data set (details in Methods) and other studies18. Although data limitations prevented detailed consideration, we also consider the possibility that individual variation in space-use could be mediated by personality type, whereby bold individuals use areas with higher human disturbance and shy individuals use areas with reduced disturbance53.
Learned experiences of animals through repeated exposure to a stimulus can shape tolerance to human activity, such as we observed, and has mixed implications for the management of wildlife ecotourism. On one hand, tolerance that is shaped by habituation-like processes can allow animals to better tolerate ecotourism activities19. On the other hand, exposure to benign human activity and associated tolerance has potential to create ecological traps if tolerant individuals later encounter humans that present a real threat54,55. Following this logic, reduced reaction to stimuli perceived as neither aversive nor beneficial (i.e., habituation44 could predispose individuals to become ensnared in conflict with humans. Not all human individuals pose the same risk to wildlife, posing a cognitive challenge in risk-perception. However, in this same population of bears, Field et al.31 found that grizzly bears that were exposed to ecotourism were less likely than predicted by chance to encounter subsequent conflict in communities downstream of ecotourism sites. Although this result might arise from well-fed ecotourism bears not needing to acquire human foods (predisposing them to conflict), it is also possible that grizzlies might be as equipped to recognize different sources and levels of risk. African elephants (Loxodonta africana) can reliably make functionally relevant distinctions between human subcategories that differ in the level of threat they pose56. Levey et al.57 similarly found behavioral flexibility and a keen awareness in Northern Mockingbirds (Mimus polyglottos) of different levels of threat posed at the finer scale of individual people.
Managers might also consider our results, while acknowledging the limitations to inference. Specifically, in the apparent absence of behavioral responses, physiological responses may be concealed. Ditmer et al.58 observed physiological responses (elevated heart rates) of American black bears (Ursus americanus) to unmanned aerial vehicles, despite a lack of observed behavioral changes. Charuvi et al.1 simultaneously measured behavioral and physiological responses during human approaches to incubating Masked Lapwings (Vanellus miles), finding that reduced flight initiation distance (generally interpreted as a sign of tolerance) was associated with greater physiological costs through longer durations of elevated heart rate. Moreover, Sganzerla et al.59 found that studies that measured physiological responses were more likely to detect disturbances than behavioral studies. Future studies might therefore add another dimension of inquiry by incorporating physiological measures of stress as indicators of tolerance in addition to spatial and behavioral measures. More broadly, how such variation in response to disturbance affects demographic parameters such as survival and reproduction would also broaden the scope of conclusions that can be drawn from impact case studies10.
We also offer some system-specific recommendations for consideration by those managing ecotourism in the Atnarko corridor. Given the potential spatial segregation of individuals according to the risk they tolerate, a cautious approach might consider limiting the number of new ecotourism sites in the future. These areas might currently serve as refuges for less tolerant bears. Other management interventions from a complementary study17 in this system can also be considered. These identified the role of context specificity in shaping another dimension of behavior (activity levels, as estimated by detection rates on remote cameras), and illustrated the roles of human visitation, salmon biomass, and their interaction underlying observed patterns. This insight allowed for predictions of how different prescriptions of visitor numbers would modify bear activity under various salmon abundance scenarios17.
There are several limitations to our approach. For example, the strength of inferences related to space-use by bears with lower detection frequencies is weaker than those we detected more often. To address this, we assessed whether the optimal number of clusters changed as we subsampled our data to include only those individuals that were detected using higher thresholds for ‘minimum detection number’ (3 and 4), finding the optimal number and general spatial characteristics of clusters was unchanged (Figures S10-S13). Additionally, an unequal number of hair snags per treatment might bias the number of individuals detected, whereby more snags might detect more individuals. We note, however, that in some cases more individuals were detected in areas with fewer snags and shorter river network distances. Specifically, more individuals were detected in the ‘no tour’ area (n = 62 out of 118) than in the ‘land- and boat-based’ area (n = 59 out of 118), despite fewer snags and shorter river network distance in the ‘no tour’ area (8 snags in the ‘no tour’ [~ 9 km river network distance] and 13 snags in the ‘land-based’ area [~ 22 km river network distance] in 2021). We emphasize that these data alone cannot directly link tolerance to ecotourism, but they are suggestive that river segment length might not affect detection numbers. River and habitat characteristic data would be important for future analyses to predict captures. Indeed, the lack of additional information about other ecological factors (e.g., conspecific competition for optimal fishing locations; home range centers) limits the inferences we can make about whether space-use is directly linked to individual tolerance.
The detectability of bears via hair snags may also vary by individual, presenting limits to interpretations of tolerance. Detection probabilities in non-invasive genetic studies can be influenced by behavioral responses, such as increased or decreased likelihood of recapture following an initial detection [e.g.,60], as well as by individual variability in detection rates61 and seasonal effects62. In the absence of bait and with the use of a non-invasive sampling technique, however, we considered detection heterogeneity minimal, and that the likelihood of detecting an individual was not substantially affected by prior detections63. Moreover, using paired camera and hair snags, Wold et al.64 found that most bears (81%) that approached hair snags made contact with the snag, indicating that this technique likely provides a representative sample. Despite these considerations, we acknowledge that telemetry data would afford a more in-depth analysis of space-use. On the other hand, we also note that individuals that researchers can capture (and subsequently collar) might reflect a non-random sample of the population. An additional consideration related to detectability is that spatial data may include samples that are biased to those individuals with home ranges that include or are centered on the river valley bottom, where probability of detection is a decreasing function of distance from home range center65. However, the river network distance of our study area falls well within potential travel distances inside the home range of both male and female grizzly bears in coastal temperate rainforests49, especially considering the relatively easy movement along riparian routes. Nonetheless, a telemetry sampling approach to examine fine-scale spatial and temporal movement would glean richer information about tolerance. Our community partners (which included Fisheries and Wildlife Managers from the Nuxalk First Nation and BC Parks), however, preferred a minimally-invasive approach (e.g., avoidance of capturing and collaring bears). Accordingly, we opted for hair-snagging. Although we forewent fine-scale spatial information, the method still yielded data on the detection of specific individuals over space and time.
An additional limitation is our lack of behavioral state data from sites without ecotourism. We suspect, however, that we were only able to collect such few data in the area related to the fact that researcher presence was novel, unpredictable and potentially perceived as risky for bears in the 'land-based’ and especially ‘no tours’ reference areas with lower human activity. Despite the lack of behavioral state data, we know bears were present, as evidenced by our genetically-informed spatial data described above, as well as camera trap data17. Nonetheless, given these limitations, we note that we could not attribute measures of tolerance to individuals based on their space-use alone. Instead, we drew on our spatial data to create a visualization tool to illustrate a possibility that not all bears in the corridor are using the ecotourism area. Doing so, however, illuminates a key point: behavioral studies focused on ecotourism areas may be collecting data solely from individuals that tolerate human presence.
Our work has broader implications for management and human-wildlife coexistence. Investigating more than one modality of wildlife response to disturbance broadens the scope of conclusions that can be drawn because different antipredator responses reflect different aspects of predation risk management11. Such an approach can provide richer insight in managing for human-wildlife coexistence. Specifically, when our behavioral and spatial data were considered in tandem, our work suggests the possibility that behavior data are collected from individuals tolerant enough to coexist with people, which might be overlooked if we examined only the behavioral states of those individuals that were present. Put another way, considering only the absence of association between behavioral states and ecotourism metrics in isolation from spatial data could suggest a grizzly bear population tolerant of ecotourism. While this may be the case, our work highlights the possibility that coexistence in this and perhaps many systems could be a process that excludes some individuals within wildlife populations that are not tolerant enough to participate. Accordingly, knowledge about the modality of response by wildlife can help managers identify effective and ecologically appropriate management strategies to foster coexistence. More broadly, research does not often distinguish between whether individuals decrease behavioral response towards humans because they are tolerant, or because shyer individuals move away from disturbed areas66. Our conclusions support Bedjer et al.’s9 argument that placing emphasis on only one modality of response effectively limits the conclusions that can be drawn from the investigation. Specifically, our work contributes to this concept by highlighting that in addition to behavioral observation data, concurrent spatial inquiry can provide richer insight into the extent to which animals might be disturbed.
Data availability
Data analyzed during the study and associated code are uploaded to the following GitHub repository: https://github.com/ACSLabUVic/atnarko-behaviour-analysis.
References
Charuvi, A. et al. A physiological cost to behavioral tolerance. Behav. Processes. 181, 104250 (2020).
Darimont, C. T., Fox, C. H., Bryan, H. M. & Reimchen, T. E. The unique ecology of human predators. Science 349 (6250), 858–860 (2015).
Darimont, C. T. et al. Humanity’s diverse predatory niche and its ecological consequences. Commun. Biol. 6 (1), 609 (2023).
Frid, A. & Dill, L. Human-caused disturbance stimuli as a form of predation risk. Conserv. Ecol. 6(1). (2002).
Gaynor, K. M., Brown, J. S., Middleton, A. D., Power, M. E. & Brashares, J. S. Landscapes of fear: Spatial patterns of risk perception and response. Trends Ecol. Evol. 34 (4), 355–368 (2019).
Tucker, M. A. et al. Moving in the anthropocene: global reductions in terrestrial mammalian movements. Science 359 (6374), 466–469. https://doi.org/10.1126/science.aam9712 (2018).
Zanette, L. Y. & Clinchy, M. Ecology and neurobiology of fear in free-living wildlife. Annu. Rev. Ecol. Evol. Syst. 51, 297–318 (2020).
Sytsma, M. L. T., Lewis, T., Gardner, B. & Prugh, L. R. Low levels of outdoor recreation alter wildlife behaviour. People Nat. 4, 1547–1559 (2022).
Bejder, L., Samuels, A. M. Y., Whitehead, H., Finn, H. & Allen, S. Impact assessment research: use and misuse of habituation, sensitisation and tolerance in describing wildlife responses to anthropogenic stimuli. Mar. Ecol. Prog. Ser. 395, 177–185 (2009).
Gill, J. A., Norris, K. & Sutherland, W. J. Why behavioral responses May not reflect the population consequences of human disturbance. Biol. Conserv. 97 (2), 265–268 (2001).
Uchida, K. & Blumstein, D. T. Habituation or sensitization? Long-term responses of yellow-bellied marmots to human disturbance. Behav. Ecol. 32 (4), 668–678 (2021).
Čapkun-Huot, C., Blumstein, D. T., Garant, D., Sol, D. & Réale, D. Towards a unified framework for studying behavioral tolerance to anthropogenic disturbance. (2023).
Wilkinson, C. E., Quinn, N., Eng, C. & Schell, C. J. Environmental health and societal wealth predict movement patterns of an urban carnivore. Ecol. Lett., 28(2), e70088. (2025).
Prugh, L. R. et al. Fear of large carnivores amplifies human-caused mortality for mesopredators. Science 380 (6646), 754–758 (2023).
Berger, J. Fear, human shields and the redistribution of prey and predators in protected areas. Biol. Lett. 3 (6), 620–623 (2007).
Steyaert, S. M. et al. Human shields mediate sexual conflict in a top predator. Proc. Royal Soc. B: Biol. Sci. 283 (1833), 20160906 (2016).
Field, K. A. et al. Influence of ecotourism on Grizzly bear activity depends on salmon abundance in the Atnarko river corridor, nuxalk territory. Conserv. Sci. Pract. 6 (4), e13097 (2024).
Nevin, O. T. & Gilbert, B. K. Perceived risk, displacement and refuging in brown bears: positive impacts of ecotourism? Biol. Conserv. 121 (4), 611–622. https://doi.org/10.1016/j.biocon.2004.06.011 (2005).
Blumstein, D. T., Geffroy, B., Samia, D. S. M. & Bessa, E. (eds). Ecotourism’s Promise and Peril: A Biological Evaluation (1st ed. 2017.). Springer International Publishing. (2017). https://doi.org/10.1007/978-3-319-58331-0.
Mattson, D. Roads, Residences, and Grizzly Bears: Effects of Human Infrastructure on Brown and Grizzly Bears. (2024). https://doi.org/10.13140/RG.2.2.27234.54720.
Penteriani, V. et al. Consequences of brown bear viewing tourism: A review. Biol. Conserv. 206, 169–180. https://doi.org/10.1016/j.biocon.2016.12.035 (2017).
Fortin, J. K. et al. Impacts of human recreation on brown bears (Ursus arctos): A review and new management tool. PloS One. 11 (1), e0141983 (2016).
Dyck, M. G. & Baydack, R. K. Vigilance behavior of Polar bears (Ursus maritimus) in the context of wildlife-viewing activities at churchill, manitoba, Canada. Biol. Conserv. 116 (3), 343–350. https://doi.org/10.1016/S0006-3207(03)00204-0 (2004).
Wheat, R. E. & Wilmers, C. C. Habituation reverses fear-based ecological effects in brown bears (Ursus arctos). Ecosphere 7(7) (2016).
Maréchal, L., Semple, S., Majolo, B. & MacLarnon, A. Assessing the effects of tourist provisioning on the health of wild barbary macaques in Morocco. PloS One. 11 (5), e0155920 (2016).
Huveneers, C. et al. The effects of cage-diving activities on the fine-scale swimming behavior and space use of white sharks. Mar. Biol. 160, 2863–2875 (2013).
Clark, C. W. Antipredator behavior and the asset-protection principle. Behav. Ecol. 5 (2), 159–170. https://doi.org/10.1093/beheco/5.2.159 (1994).
Proctor, M. et al. Ecological investigations of Grizzly bears in Canada using DNA from hair, 1995–2005: A review of methods and progress. Ursus 21 (2), 169–188. https://doi.org/10.2192/1537-6176-21.2.169 (2010).
Woods, J. G. et al. Genetic Tagging of Free-Ranging Black and Brown Bears. Wildlife Soc. Bull. 27(3), 616–627 (1999).
Paetkau, D. The optimal number of markers in genetic capture-mark–recapture studies. J. Wildl. Manag. 68 (3), 449–452 (2004).
Field, K. A., Moody, J. E., Levi, T. & Darimont, C. T. Grizzly bears detected at ecotourism sites are less likely than predicted by chance to encounter conflict. Can. J. Zool. (2025).
Quinn, T. P., Wirsing, A. J. & Proctor, M. Optimal barbed wire height for brown bear hair sample collection. Ursus 2022, 33e2. https://doi.org/10.2192/URSUS-D-21-00007.1 (2022).
Altmann, J. Observational study of behavior: sampling methods. Behavior 49 (3–4), 227–266 (1974).
McDonough, T. J. & Christ, A. M. Geographic variation in size, growth, and sexual dimorphism of Alaska brown bears, ursus Arctos. J. Mammal. 93 (3), 686–697 (2012).
Prop, J., Staverløkk, A. & Moe, B. Identifying individual Polar bears at safe distances: A test with captive animals. PLoS One, 15(2), e0228991. (2020).
Hilderbrand, G. V. et al. The importance of meat, particularly salmon, to body size, population productivity, and conservation of North American brown bears. Can. J. Zool. 77 (1), 132–138. https://doi.org/10.1139/z98-195 (1999).
Kingsley, M. C. S., Nagy, J. A. & Reynolds, H. V. Growth in length and weight of Northern brown bears: differences between sexes and populations. Can. J. Zool. 66 (4), 981–986. https://doi.org/10.1139/z88-145 (1988).
Clapham, M., Miller, E., Nguyen, M. & Darimont, C. T. Automated facial recognition for wildlife that lack unique markings: A deep learning approach for brown bears. Ecol. Evol. 10 (23), 12883–12892. https://doi.org/10.1002/ece3.6840 (2020).
Beauchamp, G. Animal Vigilance: Monitoring Predators and Competitors (Academic, 2015).
Blanchard, P. & Fritz, H. Induced or routine vigilance while foraging. Oikos 116 (10), 1603–1608. https://doi.org/10.1111/j.0030-1299.2007.15799.x (2007).
Lima, S. L. & Dill, L. M. Behavioral decisions made under the risk of predation: a review and prospectus. Can. J. Zool. 68 (4), 619–640 (1990).
McCullagh, P. & Nelder, J. A. Generalized Linear Models 2nd edn (Chapman & Hall, 1989).
Nisbet, I. C. Disturbance, habituation, and management of waterbird colonies. Waterbirds, 312–332. (2000).
Blumstein, D. T. Habituation and sensitization: new thoughts about old ideas. Anim. Behav. 120, 255–262 (2016).
Hilderbrand, G. V., Schwartz, C. C., Robbins, C. T. & Hanley, T. A. Effect of hibernation and reproductive status on body mass and condition of coastal brown bears. J. Wildl. Manag. 64 (1), 178. https://doi.org/10.2307/3802988 (2000).
Lima, S. L. Vigilance while feeding and its relation to the risk of predation. J. Theor. Biol. 124 (3), 303–316 (1987).
Repasky, R. R. Using vigilance behavior to test whether predation promotes habitat partitioning. Ecol. (Durham). 77 (6), 1880–1887. https://doi.org/10.2307/2265791 (1996).
McNamara, J. M. & Houston, A. I. Starvation and predation as factors limiting population size. Ecology 68 (5), 1515–1519 (1987).
MacHutchon, A. G., Himmer, S. & Bryden, C. A. Khutzeymateen Valley Grizzly Bear Study: Final Report (Ministry of Forests Research Program, 1993). Wildlife Report No. R-25.
Wielgus, R. B. & Bunnell, F. L. Sexual segregation and female Grizzly bear avoidance of males. J. Wildl. Manag. 405–413. (1994).
Finnegan, S. P., Svoboda, N. J., Fowler, N. L., Schooler, S. L. & Belant, J. L. Variable intraspecific space use supports optimality in an apex predator. Sci. Rep. 11 (1), 21115 (2021).
Leacock, W. B., Smith, K. T. & Deacy, W. W. Travel specific resource selection by female Kodiak brown bears during the Sockeye salmon spawning season. Mov. Ecol. 12 (1), 1–13 (2024).
Samia, D. S. M., Nakagawa, S., Nomura, F., Rangel, T. F. & Blumstein, D. T. Increased tolerance to humans among disturbed wildlife. Nat. Commun. 6 (1), 8877–8877. https://doi.org/10.1038/ncomms9877 (2015).
Geffroy, B., Samia, D. S. M., Bessa, E. & Blumstein, D. T. How Nature-Based tourism might increase prey vulnerability to predators. Trends Ecol. Evol. 30 (12), 755–765. https://doi.org/10.1016/j.tree.2015.09.010 (2015).
Darimont, C. T. & Shukla, I. When the enemy of an enemy is no friend. Science 380 (6646), 691–692. https://doi.org/10.1126/science.adh9166 (2023).
McComb, K., Shannon, G., Sayialel, K. N. & Moss, C. Elephants can determine ethnicity, gender, and age from acoustic cues in human voices. Proc. Natl. Acad. Sci. - PNAS. 111 (14), 5433–5438. https://doi.org/10.1073/pnas.1321543111 (2014).
Levey, D. J., Londoño, G. A., Ungvari-Martin, J., Hiersoux, M. R., Jankowski, J.E., Poulsen, J. R., & Robinson, S. K. Urban mockingbirds quickly learn to identify individual humans. Proc. Natl. Acad. Sci. 106(22), 8959–8962 (2009).
Ditmer, M. A., Vincent, J. B., Werden, L. K., Tanner, J. C., Laske, T. G., Iaizzo,P. A., & Fieberg, J. R. Bears show a physiological but limited behavioral response to unmanned aerial vehicles. Curr. Biol. 25(17), 2278–2283 (2015).
Sganzerla, F., Scillitani, L., Brivio, F. & Grignolio, S. The effects of recreational activities on wild mammals. Global Ecol. Conserv. e03485. (2025).
Zarnoch, S. J. Simulation of effects of learned trap response on three estimators of population size. J. Wildl. Manag. 474–483. (1979).
Boulanger, J. et al. Multiple data sources improve DNA-based mark–recapture population estimates of Grizzly bears. Ecol. Appl. 18 (3), 577–589 (2008).
Boulanger, J., Stenhouse, G. & Munro, R. Sources of heterogeneity bias when DNA mark-recapture sampling methods are applied to Grizzly bear (Ursus arctos) populations. J. Mammal. 85 (4), 618–624 (2004).
Wirsing, A. J. et al. Alaskan brown bears (Ursus arctos) aggregate and display fidelity to foraging neighborhoods while preying on Pacific salmon along small streams. Ecol. Evol. 8 (17), 9048–9061 (2018).
Wold, K., Wirsing, A. J. & Quinn, T. P. Do brown bears Ursus Arctos avoid barbed wires deployed to obtain hair samples? A videographic assessment. Wildl. Biology. 2020 (1), 1–6 (2020).
Efford, M. G. & Mowat, G. Compensatory heterogeneity in spatially explicit capture–recapture data. Ecology 95 (5), 1341–1348. https://doi.org/10.1890/13-1497.1 (2014).
Martin, J. G. & Réale, D. Temperament, risk assessment and habituation to novelty in Eastern chipmunks, Tamias striatus. Anim. Behav. 75 (1), 309–318 (2008).
Acknowledgements
This work was conducted in the unceded Territory of the Nuxalk First Nation. Thank you to the directors and managers from Nuxalk Stewardship Office at the time of the project (Wally Webber, Sam Pascoe and Rhonda Morton) and BC Parks (Steve Hodgson, Lori Homstol, Sam McKoy and Rae Busse) for continuous support throughout the years. A special acknowledgement to the Nuxalk Stataltmc (Hereditary Chiefs) supporting ongoing bear work following the signing of the Grizzly Bear Protocol on 12 November 2011 at Snuxyaltwa’s (Deric Snow’s) Potlatch. The work would not have been possible if it were not for contributions from Nuxalk Fisheries and Wildlife staff (Kelly Milton, Shanti Tallio-Milton, Ron Schooner, and Quentin Hans), including sharing of in-depth knowledge of the river to guide our study design. Thank you to Nuxalk Coastal Guardian Watchmen for in-field assistance. Thank you to field research assistants (Andrew Sheriff, Emina Ida, Sean Murray, and Nina Sylvester) and volunteers of the Raincoast Applied Conservation Science Lab. Thank you to ecotour operators, Snootli Hatchery crews, and Department of Fisheries and Oceans crews for working alongside us during the data collection stages of the project. Thank you to Belarko Wildlife Viewing Platform staff, BC Parks Rangers, and many community members who housed and worked with us throughout the project.
Author information
Authors and Affiliations
Contributions
Conceptualization: KAF, JEM, DAC, PCP, CTDData curation: KAF, PBK, MCData analysis: KAF, CTD Funding acquisition: KAF, CTD Investigation: KAF, JEM, CTD Methodology: KAF, JEM, MC, TL, CTD Project administration: KAF, JEM, CTD Resources: JEM, CTD Supervision: JEM, DAC, PCP, CTD Visualization: KAF Writing – original draft: KAF, CTDWriting – review & editing: KAF, JEM, MC, DAC, PBK, TL, PCP, CTD.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Field, K.A., Moody, J.E., Clapham, M. et al. Integrating spatial and behavioral data provides comprehensive assessment of grizzly bear-ecotourism coexistence in Nuxalk Territory. Sci Rep 15, 30339 (2025). https://doi.org/10.1038/s41598-025-14625-5
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-14625-5