Abstract
Colorectal cancer (CRC) has one of the highest incidence and mortality rates of any cancer worldwide, and the recurrence and metastasis rates after surgery remain high. The search for simple and reliable prognostic markers is essential to improve patient prognosis. In this study, we investigated the predictive value of two easy-to-measure preoperative markers, platelet-to-albumin ratio (PAR) and cancer inflammation prognostic index (CIPI), in postoperative survival of patients with CRC. We retrospectively analyzed the clinical data of a total of 434 CRC patients from two hospitals. Data from one of the institutions were used as a training cohort. The predictive ability of both markers was assessed using subject work characteristics (ROC) curves, with optimal cutoff values of 11.69 and 6.85 for CIPI and PAR, respectively, and high CIPI and PAR were independently associated with poorer overall survival. We developed nomogram of overall survival (OS), performed internal and external validation, and showed superior prognostic accuracy of the nomogram models. Our results suggest that combined CIPI and PAR can be used as a valid and accessible prognostic tool that is expected to help develop personalized treatment strategies for CRC patients.
Similar content being viewed by others
Introduction
Colorectal cancer (CRC) is the third most common malignant tumor and the second leading cause of death in the world1. The incidence of CRC is further increasing due to changes in people’s lifestyles and dietary habits in recent years. Currently, the treatment of CRC still mainly relies on surgical methods, but the higher chance of recurrence and metastasis after surgery seriously affects the prognosis of CRC as well as the quality of life of patients2. In recent years, machine learning approaches have been increasingly applied to predict post-treatment complications and stratify high-risk patients across cancer types3,4,5. In gastric cancer, machine learning has facilitated the identification of novel immune regulators such as the COMMDs family, showing promise in immunotherapy response prediction6.
Inflammatory response is an important factor in initiating cancer and promoting its development, chronic stimuli and inflammatory factors can stimulate the development of many cancers, and a microenvironment composed of inflammatory cells can promote tumor proliferation, survival and migration7,8. Several studies have shown that inflammation-based prognostic markers such as neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), lymphocyte-to-monocyte ratio (LMR), and systemic immune-inflammatory index (SII), which are elevated, correlate with poor prognosis of patients with tumors9,10. And the preoperative CEA level has been widely used in clinical practice11,12. Nutritional indicators are also important factors that influence prognosis. A patient’s nutritional status affects the body’s immune ability, postoperative recovery time, and complication rate13common nutritional indicators such as albumin-related indicators have good predictive ability for the prognosis of cancer patients14,15. The discovery of biomarkers continues to play a key role in the prognosis and diagnosis of cancer. Relevant studies have already identified glutaminase as a promising diagnostic biomarker in breast cancer16. These biomarkers are widely used to assess the prognosis of cancer.
It has been shown that the preoperative cancer inflammation prognostic index: CIPI (= CEA (Carcinoembryonic Antigen) concentration (mg/L) × neutrophil count (109/L)/lymphocyte count (109/L)) and platelet-to-albumin ratio: PAR (= platelet count (109/L)/albumin (g/L)) have a good ability to predict the prognosis of CRC17,18,19. However there has been no study combining these two metrics to predict CRC prognosis. Therefore, in this study, we will combine preoperative CIPI and PAR to analyze the relationship between them and the pathological features of CRC, and analyze the predictive value of CIPI combined with PAR in postoperative CRC patients.
Patients and methods
Study population
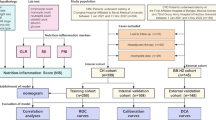
This study retrospectively selected 289 primary CRC patients who underwent radical surgery for CRC from January 2016 to December 2017 at the Affiliated Cancer Hospital of Xinjiang Medical University and 145 primary CRC patients who underwent radical surgery at the People’s Hospital of Bortala Mongolian Autonomous Prefecture. The inclusion criteria were (1) patients underwent radical surgery; (2) patients were confirmed to have primary CRC by postoperative histopathology; (3) blood laboratory indexes within 1 week before surgery were available; (4) patients were > 18 years old; and (5) patients’ clinical data were complete and reliable, and they were capable of completing follow-up. The exclusion criteria were (1) having non-primary CRC; (2) with other primary cancers; (3) patients with unresectable distant metastases; (4) patients with hematologic and autoimmune diseases; (5) with hematologic or autoimmune diseases; (6) patients with severe hepatic or renal insufficiency or diseases causing malnutrition; and (7) patients receiving parenteral nutritional support prior to surgical Treatment.
Patients in this study were followed up through clinical visits and telephone follow-ups. The follow-up frequency was every 3 months during the first two years after surgery and then every 6 months thereafter. If patients could not be contacted through telephone follow-up, survival data were obtained through the national cancer registry. The duration of follow-up for patients ranged from 60 months to 80 months, with a median follow-up duration of 70 months. The follow-up data were censored for patients who were lost to follow-up or those who survived without recurrence until the last follow-up date. The censoring process ensures that only patients with complete survival data were included in the analysis.
289 patients from the Affiliated Cancer Hospital of Xinjiang Medical University were used as a training cohort to construct the nomogram. 145 patients from People’s Hospital of Bortala Mongolian Autonomous Prefecture were used as a validation cohort to validate the nomogram. The study was authorized by the Ethics Committee of Xinjiang Medical University Cancer Hospital (Approval No. K-2024056) and the Ethics Committee of People’s Hospital of Bortala Mongolian Autonomous Prefecture (Approval No. LLSH20241221), in accordance with the Declaration of Helsinki, and informed consent was obtained from all participants and/or their legal guardians.
Data collection
The following information was collected from the electronic medical record system of Xinjiang Medical University Cancer Hospital and People’s Hospital of Bortala Mongolian Autonomous Prefecture: including basic information (age, gender, height, weight, smoking, drinking history), hematological parameters (platelets, lymphocytes, neutrophils, albumin, CEA, CA-199 (Carbohydrate Antigen 19 − 9)) within 1 week before surgery, postoperative pathology (degree of tumor differentiation, vascular cancer embolism, nerve invasion, TNM staging) and follow-up information (survival outcome, survival time).
Calculated from hematological indices: CIPI = CEA concentration (mg/L) × neutrophil count (109/L) / lymphocyte count (109/L), PAR = platelet count (109/L) / albumin (g/L).
Statistical analysis
In this study, SPSS (v29.0) and R programming language (v4.4.1) were applied to statistically analyze the collected data, and for baseline clinical data, the categorical variables were expressed as counts (percentage, %), qualitative data were tested using the χ2 test or Fisher’s exact test, quantitative data were tested using the t-test or analysis of variance (ANOVA), and stratified data were analyzed using the Wilcoxon test. General clinical data and pathological characteristics of the groups were compared and analyzed. Subject work characteristics (ROC) curve analysis was used to determine the optimal critical values for CIPI and PAR. The area under the ROC curve (AUC) was calculated to assess the predictive ability of CIPI and PAR. Survival curves for CIPI and PAR were plotted separately using the Kaplan-Meier method, and survival differences were compared using the Log-Rank test. Risk factors for overall survival (OS) were determined by Cox proportional risk regression analysis and were expressed as risk ratios (HR) with 95% confidence intervals (CI). Based on the results of Cox regression analysis, a nomogram model was constructed using the R software package “rms” to predict the prognosis of CRC patients, and ROC curves and calibration curves were used to evaluate the discriminative and predictive power of the nomogramand to validate the nomogram internally and externally. In the statistical analysis of this study p < 0.05 was considered statistically significant.
Results
Clinicopathological characteristics of patients
In this study, 289 patients with primary CRC at Xinjiang Medical University Cancer Hospital from January 2016 to December 2017 were retrospectively selected as the training cohort, and 145 patients with primary CRC at Bozhou People’s Hospital in Xinjiang Uygur Autonomous Region were selected as the validation cohort. The demographic and clinical factors of the training and validation cohorts were basically the same. Among them, 252 (58.1%) were male with the median age being 62 years, and tumor TNM stages I, II, III, and IV accounted for 20.5, 45.2, 31.6, and 2.7%, respectively. The majority of patients (71.2%) had moderately differentiated histologic type, followed by undifferentiated/lowly differentiated (24.9%) and highly differentiated (3.9%). Neuroinvasive and vascular cancerous embolisms were found in 65 (15%) and 67 (15.4%) patients respectively. Patients with high CA-199 accounted for 10.4% of all the patients. Additional baseline information is shown in (Table 1).
Optimal cutoff values for CIPI and PAR in the training cohort
We assessed the accuracy of CIPI and PAR in predicting the prognosis of patients by performing ROC analysis on the OS of the training cohort, which had an area under the curve (AUC) of 0.753 and 0.646, with an optimal cutoff value of 11.69 and 6.85, respectively (Fig. 1), and based on the cutoff value, CIPI ≥ 11.69 and < 11.69 were defined as H -CIPI, L-CIPI, and define PAR ≥ 6.85, < 6.85 as H-PAR, L-PAR, respectively, based on the cutoff values.
ROC curves for CIPI and PAR in the training cohort.
Subgroup analysis in the training cohort
Among our subgroup analyses, we found that an increased risk of death was independently associated with both the high CIPI group compared to the low CIPI group, and the high PAG group compared to the low PAG group, as shown in (Fig. 2A,B).
Stratified analysis in CRC patients. CIPI (A) and PAG (B).
Relationship of preoperative CIPI and PAR to clinicopathologic features in the training cohort
The differences between preoperative CIPI and PAR were statistically significant in terms of N staging (p = 0.012, p = < 0.001) and TNM staging (p = 0.020, p < 0.001). In addition, the differences in CIPI were statistically significant for nerve invasion (p = 0.002), vascular cancer embolism (p = 0.014), and CA-199 (p = < 0.001), and the differences in PAR were statistically significant for gender (p = 0.009), age (p = 0.002), and smoking (p = 0.030). However, there was no statistically significant difference between CIPI and PAR in terms of BMI, alcohol consumption, T-staging, and histologic staging, as shown in (Table 2).
Correlation of CIPI and PAR with survival in the training cohort
The median OS of all patients in the training cohort was 67 months. We could find that high CIPI in the Kaplan-Meier curve was associated with shorter OS and the survival difference was statistically significant (P < 0.001). High PAR was associated with shorter OS and the survival difference was also statistically significant (P < 0.001) as shown in (Fig. 3A,B).
Kaplan-Meier curves in the CRC. CIPI (A) and PAR (B) .
To further explore the predictive value of combined CIPI and PAR in CRC, we performed a combined analysis of CIPI and PAR. The median OS of patients with high CIPI-high PAR was lower than that of the high CIPI-low PAR group, the low CIPI-high PAR group, and the low CIPI-low PAR group. This difference in survival was statistically significant (P < 0.0001), as shown in (Fig. 4).
Kaplan-Meier survival curves for combined CIPI and PAR in the training cohort.
Independent risk factors for OS in the training cohort
We performed univariate and multifactorial analyses for the clinicopathologic characteristics of CRC patients in the training cohort, as shown in (Table 3). Univariate analyses demonstrated that preoperative CIPI, preoperative PAR, T stage, N stage, TNM stage, and vascular cancer embolism were significantly associated with OS in CRC patients (< 0.05). In COX multifactorial analysis, preoperative CIPI, preoperative PAR, age, N stage and TNM stage were independent risk factors affecting OS in CRC patients. Patients with high preoperative CIPI had lower OS than those with low CIPI (HR = 5.69,95% CI [3.22 to 10.05] P < 0.001), patients with high preoperative PAR had lower OS (HR = 2.63,95% CI [1.65 to 4.19] P < 0.001), and CRC patients with age > 60 had poorer OS than younger patients (HR = 0.54,95% CI [0.34 to 0.86] P = 0.010), and CRC patients with N-stages N1 and N2 had lower OS (HR = 2.24,95% CI [1.36 to 3.69] P=-0.001), (HR = 4.13,95% CI [2.50 to 6.81] P < 0.001), CRC patients with TNM stage II, III, and IV had worse OS than those with low TNM stage (HR = 0.24,95% CI [0.14 to 0.41] P < 0.001), (HR = 0.36,95% CI [0.23 to 0.58] P < 0.001), (HR = 2.09, 95% CI [0.94to 4.66] P < 0.001).
Construction and validation of nomogram for the training cohort
Based on the results of the above COX regression analysis, we included those clinicopathological characteristics with P < 0.05 in the multifactorial analysis in the nomogram model to further analyze the impact of these independent risk factors on patient prognosis. According to the nomogram model, we found that preoperative CIPI, N stage and TNM stage had a greater impact on patients’ OS, and the prognosis was worse for patients with high preoperative CIPI as well as high N stage and high TNM stage. High PAG had a negative impact on patients’ OS. In addition, we noticed an inconsistency between the nomogram and the multivariate Cox regression results regarding age. While Cox analysis showed that younger age was associated with better survival, the nomogram appeared to predict worse outcomes for patients under 45 years. This discrepancy is likely due to the small sample size in the < 45-year group (n = 40) and the presence of more aggressive tumor features in some younger patients. We have interpreted this finding cautiously and acknowledged it as a limitation of the predictive model. As shown in (Fig. 5).
Nomogram prediction model including CIPI, PAG, age, N staging, TNM staging.
We then performed an internal validation of the nomogram model. The validation was carried out by the ROC curve, and its AUC values for 1, 3, and 5 years were 0.940, 0.879, and 0.847, respectively, as shown in (Fig. 6A). We also applied the calibration curves (Fig. 7) and the DCA decision curves (Fig. 8A–C) of the column-line graph model for 1, 3, and 5 years to validate and evaluate the predictive ability of the column-line graph model. It can be seen that the prediction results of the calibration curves are highly consistent with the actual results, which indicates that the nomogram model we constructed has good predictive ability.
1-, 3-, and 5-year ROC curves based on nomogram for the training cohort (A) and validation cohort (B).
Calibration curves for 1-, 3- and 5-year nomogram.
1-, 3-, and 5-year DCA calibration curves for the training cohort (A–C) and the validation cohort (D–F) .
Finally we used the validation cohort data as external validation. The validation was performed by the ROC curve, whose AUC values for 1, 3, and 5 years were 0.862, 0.827, and 0.787, respectively, as shown in (Fig. 6B). In addition, the DCA decision curves (Fig. 8D–F) also indicated that the prediction model had good predictive ability. The results of internal validation and external validation showed that the column-line graph model has good predictive ability for predicting the overall survival of CRC patients.
Discussion
The incidence of CRC has been increasing with changes in diet and lifestyle, and CRC has become an important public health problem, seriously affecting people’s quality of life and longevity. Despite the trend of younger CRC incidence, patients are still predominantly elderly20. Due to the highly invasive nature of CRC and the general poor physical condition of the elderly, poor postoperative prognosis of CRC patients often occurs21. Therefore, it is important to explore effective prognostic factors.
In this study, we collected clinical information from a total of 434 patients in two hospitals and investigated the relationship between preoperative CIPI and PAR and the postoperative prognosis of CRC patients. Through statistical analysis we found that high preoperative CIPI and high PAR were associated with poorer CRC prognosis, and that preoperative CIPI and PAR were independent risk factors for OS in CRC patients. We also predicted the prognosis of CRC patients by constructing a nomogram, and evaluated the predictive ability of the nomogram by external validation.
Some studies have shown that chronic inflammatory response is an important factor in promoting cancer development, especially in the tumor microenvironment (TME) where the presence of inflammatory cells stimulates the proliferation of cancer cells22,23. The tumor microenvironment (TME) consists of a variety of cell populations including cancer cells, immune cells, etc., which are involved in all the processes of tumorigenesis, proliferation, and migration through direct contact or cytokine exchange24,25. In addition to host immune and metabolic status, intratumoral heterogeneity—including that driven by microbiota diversity—has been shown to shape tumor development and therapeutic outcomes26. Recent evidence has shown that radiotherapy, particularly high-LET modalities, can modulate the tumor immune microenvironment and enhance response to immunotherapy in certain malignancies27. Inflammatory mediators and cellular effectors of chronic inflammatory response are important components in promoting tumorigenesis and development. Inflammatory response can enhance the proliferative ability of tumor cells through the transcription factor NF-kappaB28 on the other hand, inflammatory factors can be involved in the process of tumor angiogenesis, tumor infiltration and metastasis by inhibiting the anti-tumor effect of p5329.
In recent years the role of inflammatory cells and factors in tumorigenesis and development has received increasing attention, including neutrophils, lymphocytes and monocytes, which are important factors involved in angiogenesis, invasion and metastasis in the process of tumorigenesis and development30,31. Preoperative CIPI (= CEA concentration (mg/L) × neutrophil count (109/L)/ Lymphocyte count (109/L)) where neutrophils are the most abundant immune cells in the peripheral blood and bone marrow, and they are the most predominant cells in tumor inflammation. Neutrophils can promote tumorigenesis through the release of reactive oxygen species (ROS), reactive nitrogen species (RNS), or proteases32. Neutrophils can also promote tumor proliferation through the suppression of other cells in the immune system, such as CD8 + T lymphocytes can be inhibited by arginase 1 (ARG1) released by neutrophils33. During tumor progression, neutrophils promote tumor metastasis by secreting MMP9, neutrophil elastase (NE), and IL-1B34. Lymphocyte cells are important players in immune defense and cancer control, for example, CD4+ T promotes the production of other immune cells (e.g., macrophages, B-cells, and cytotoxic CD8+ T-cells) and creates the appropriate conditions for them to work, whereas CD8 memory T-cells have been demonstrated to mediate tumor regression, leading to a long-lasting antitumor effect35,36 and this particular sustained response is thought to be based on the T-cells’ ability to act as potent effectors and subsequently generate long-lived memory37,38. It has also been shown that high expression of tumor-infiltrating lymphocytes is associated with a favorable cancer prognosis39. Similarly, immune-related subtypes such as those associated with efferocytosis have been linked to distinct patterns of immune infiltration and prognosis in glioblastoma40,41,42supporting the idea that inflammatory-based scores like CIPI may also reflect immune status in CRC.
Platelet-tumor interactions promote hematogenous metastatic properties of tumors. After tumor cells reach the bloodstream, platelets are activated to form an environment suitable for tumor metastasis43. Platelets can protect tumor cells by wrapping around them to form microthrombi, thereby using them as a physical barrier against blood shear and to avoid being killed by immune cells. Activation of platelets by cancer cells may also promote metastasis and cancer progression by forming neutrophil extracellular traps (NETs) that “hide” the cancer cells44,45. In addition, platelets are a major source of vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), and basic fibroblast growth factor (bFGF), each of which promotes intra-tumor angiogenesis and thus tumor growth46,47.
Along with the progression of cancer, cancer patients often suffer from malnutrition due to excessive consumption and insufficient intake, which is often manifested as anemia, decreased albumin, weight loss, etc48. especially in elderly patients49. Compared with other cancers, gastrointestinal cancer patients are more likely to suffer from malnutrition, which is due to the poor digestion and absorption function of the gastrointestinal tract or intestinal obstruction caused by cancer, and it is difficult for patients to obtain sufficient nutrition through daily meals50,51. About 35% of CRC patients will be moderately to severely malnourished prior to surgery, and their length of hospitalization and gastrointestinal recovery time will be longer52. Malnourished patients have an increased incidence of postoperative infections due to immunosuppression, and malnutrition also leads to lower serum albumin levels and higher anastomotic fistula rates53. Therefore, the present study included albumin in the assessment to develop an effective predictor of prognosis in CRC.
In this study, we combined the preoperative CIPI and PAR to predict the postoperative prognosis of patients with CRC, combining CIPI and PAR may offer enhanced prognostic value due to their ability to capture distinct yet complementary aspects of tumor biology and host response. CIPI incorporates the NLR, a marker of systemic inflammation and immune imbalance, along with CEA, which reflects tumor burden. In contrast, PAR integrates platelet count—a surrogate for inflammatory and pro-thrombotic activity—with serum albumin, a marker of nutritional status and systemic resilience. Chronic inflammation fosters tumor progression through angiogenesis, immune evasion, and cytokine-driven proliferation, while hypoalbuminemia reflects malnutrition and impaired immunological defense. Together, these indices reflect both tumor aggressiveness and host vulnerability, offering a more comprehensive risk assessment. This mechanistic rationale supports the additive predictive power observed in our nomogram model, and the results of the study showed that combining these two indices can reliably predict the prognosis of CRC. Other inflammation- and immune-related biomarkers, such as IL1RN and PRRX1, have been reported to correlate with immune infiltration and prognosis in CRC54 reinforcing the relevance of inflammation-based predictors like CIPI.However, there are some limitations of this study. First, as a retrospective study, detailed information on postoperative adjuvant chemotherapy was partially missing or inconsistently documented across the two participating centers. Given that chemotherapy—particularly fluoropyrimidine-based regimens with or without oxaliplatin—is a standard treatment for stage III and selected high-risk stage II CRC, we acknowledge that its omission from the multivariate analysis may limit the comprehensiveness of our prognostic evaluation. However, to avoid introducing potential bias due to incomplete treatment data, we chose not to include this variable. We have highlighted this limitation and recommend that future prospective studies with standardized treatment documentation incorporate adjuvant therapy data to validate and extend our findings. Second, microsatellite instability (MSI), which serves as a key prognostic and predictive biomarker in CRC, especially in patients with localized disease, was not consistently documented in the electronic medical records from the two participating centers. Given that MSI status can influence both survival outcomes and chemotherapy responsiveness—particularly to fluoropyrimidine monotherapy—its absence in our dataset limits the ability to explore potential molecular interactions with inflammation- and nutrition-based markers such as CIPI and PAR. Future prospective investigations should incorporate standardized molecular testing, including MSI status, as demonstrated in recent radiomics studies on endometrial cancer55 non-invasive imaging-based models may help estimate MSI status and could be integrated with systemic markers like CIPI and PAR in future CRC prognostic frameworks. Third, only patients who underwent radical surgical resection were included in this study. As a result, our findings may not be applicable to patients with advanced disease who are deemed unsuitable for curative surgery and instead receive palliative treatment. Since surgical candidates generally have better baseline functional status and lower tumor burden, the prognostic implications of inflammation- and nutrition-based markers may differ in non-surgical populations. Therefore, caution should be exercised when extrapolating our results to patients managed non-operatively. Finally, we assessed the prognostic utility of CIPI and PAR based solely on preoperative measurements. Due to the retrospective nature of the study, we were unable to perform longitudinal analyses to capture dynamic changes in these indices over time, such as during postoperative recovery, recurrence, or adjuvant therapy. Future prospective studies with serial biomarker monitoring may provide deeper insights into the temporal prognostic relevance of these markers and further enhance the model’s clinical utility when applied in real-world, time-sensitive clinical decision-making.
Conclusion
This study emphasizes the importance of combining CIPI and PAR in the prognosis of CRC patients. Both metrics were independently correlated with low postoperative survival. The column-line graphical model constructed from these two metrics provides a cost-effective, convenient, and reliable tool for stratifying patients according to survival risk, thus providing potential guidance for personalized postoperative management and treatment strategies. However, the retrospective design of this study needs to be further validated by large-scale, prospective validation. Future studies should also explore the broad applicability of CIPI with PAR in patients receiving different treatment modalities or in advanced stages of the disease. By integrating inflammatory and nutritional markers, this study paves the way for the development of simplified, clinically applicable prognostic tools for CRC.
Data availability
All data generated or analysed during this study are included in this published article.
References
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74, 229–263. https://doi.org/10.3322/caac.21834 (2024).
Han, L. et al. Nomogram of conditional survival probability of long-term survival for metastatic colorectal cancer: A Real-World data retrospective cohort study from SEER database. Int. J. Surg. 92, 106013. https://doi.org/10.1016/j.ijsu.2021.106013 (2021).
Zhu, Y. et al. Using machine learning to identify patients at high risk of developing low bone density or osteoporosis after gastrectomy: a 10-year multicenter retrospective analysis. J. Cancer Res. Clin. Oncol. 149, 17479–17493. https://doi.org/10.1007/s00432-023-05472-w (2023).
Zhang, J., Li, H., Tao, W., Zhou, J. & GseaVis An R package for enhanced visualization of gene set enrichment analysis in biomedicine. Med. Res. 1, 131–135. https://doi.org/10.1002/mdr2.70000 (2025).
Wang, Y., Jian, W., Yuan, Z., Guan, F. & Carlson, D. Deep learning with attention modules and residual transformations improves hepatocellular carcinoma (HCC) differentiation using multiphase CT. Precision Radiation Oncol. 9, 13–22. https://doi.org/10.1002/pro6.70003 (2025).
Wan, R. et al. Decoding gastric cancer: machine learning insights into the significance of COMMDs family in immunotherapy and diagnosis. J. Cancer. 15, 3580–3595. https://doi.org/10.7150/jca.94360 (2024).
Greten, F. R. & Grivennikov, S. I. Inflammation and cancer: triggers, mechanisms, and consequences. Immunity 51, 27–41. https://doi.org/10.1016/j.immuni.2019.06.025 (2019).
Song, B. et al. A novel Necroptosis-Related gene signature in skin cutaneous melanoma prognosis and tumor microenvironment. Front. Genet. 13, 917007. https://doi.org/10.3389/fgene.2022.917007 (2022).
Nøst, T. H. et al. Systemic inflammation markers and cancer incidence in the UK biobank. Eur. J. Epidemiol. 36, 841–848. https://doi.org/10.1007/s10654-021-00752-6 (2021).
Passardi, A. et al. Inflammatory indices as prognostic markers in metastatic colorectal cancer patients treated with chemotherapy plus bevacizumab. Ther. Adv. Med. Oncol. 15, 17588359231212184. https://doi.org/10.1177/17588359231212184 (2023).
Xie, H. L. et al. The prognostic value of the postoperative serum CEA levels/preoperative serum CEA levels ratio in colorectal cancer patients with high preoperative serum CEA levels. Cancer Manag Res. 11, 7499–7511. https://doi.org/10.2147/cmar.S213580 (2019).
Xie, H., Wei, L., Tang, S. & Gan, J. Serum albumin-carcinoembryonic antigen ratio as an effective clinical tool for predicting recurrence and overall survival in patients with rectal cancer. Front. Nutr. 11, 1521691. https://doi.org/10.3389/fnut.2024.1521691 (2024).
Narimatsu, H. & Yaguchi, Y. T. The role of diet and nutrition in cancer: prevention, treatment, and survival. Nutrients 14 https://doi.org/10.3390/nu14163329 (2022).
Li, J. et al. Preoperative albumin-to-globulin ratio and prognostic nutritional index predict the prognosis of colorectal cancer: a retrospective study. Sci. Rep. 13, 17272. https://doi.org/10.1038/s41598-023-43391-5 (2023).
Wei, L., Xie, H., Wang, Q., Tang, S. & Gan, J. The prognostic value of preoperative D-dimer to albumin ratio for overall survival and progression-free survival in colorectal cancer. Front. Physiol. 15, 1369855. https://doi.org/10.3389/fphys.2024.1369855 (2024).
Zhang, D. et al. GLS as a diagnostic biomarker in breast cancer: in-silico, in-situ, and in-vitro insights. Front. Oncol. 13, 1220038. https://doi.org/10.3389/fonc.2023.1220038 (2023).
Făgărășan, V. et al. Lymphocyte-to-monocyte, platelet-to-albumin and platelet-to-lymphocyte ratios as prognostic biomarkers for neoadjuvant treatment response in rectal cancer patients. Surg. Oncol. 56, 102126. https://doi.org/10.1016/j.suronc.2024.102126 (2024).
You, J. F. et al. Preoperative cancer inflammation prognostic index as a superior predictor of Short- and Long-Term outcomes in patients with stage I-III colorectal cancer after curative surgery. Cancers (Basel). 14. https://doi.org/10.3390/cancers14246232 (2022).
Xie, H. et al. The cancer inflammation prognostic index is a valuable biomarker for predicting the survival of patients with stage I-III colorectal cancer. Sci. Rep. 13, 18080. https://doi.org/10.1038/s41598-023-45550-0 (2023).
Shapiro, J. A. et al. Screening for colorectal cancer in the united states: correlates and time trends by type of test. Cancer Epidemiol. Biomarkers Prev. 30, 1554–1565. https://doi.org/10.1158/1055-9965.Epi-20-1809 (2021).
Ugolini, G., Ghignone, F., Zattoni, D., Veronese, G. & Montroni, I. Personalized surgical management of colorectal cancer in elderly population. World J. Gastroenterol. 20, 3762–3777. https://doi.org/10.3748/wjg.v20.i14.3762 (2014).
Coussens, L. M. & Werb, Z. Inflammation and cancer. Nature 420, 860–867. https://doi.org/10.1038/nature01322 (2002).
Sohrab, S. S. et al. Chronic inflammation’s transformation to cancer: A nanotherapeutic paradigm. Molecules 28 https://doi.org/10.3390/molecules28114413 (2023).
Hibino, S. et al. Inflammation-induced tumorigenesis and metastasis. Int. J. Mol. Sci. 22 https://doi.org/10.3390/ijms22115421 (2021).
Zhang, P. et al. The integrated single-cell analysis developed an Immunogenic cell death signature to predict lung adenocarcinoma prognosis and immunotherapy. Aging (Albany NY). 15, 10305–10329. https://doi.org/10.18632/aging.205077 (2023).
Gao, Z. et al. Heterogeneity of intratumoral microbiota within the tumor microenvironment and relationship to tumor development. Med. Res. 1, 32–61. https://doi.org/10.1002/mdr2.70006 (2025).
Meng, K. & Lu, H. Clinical application of high-LET radiotherapy combined with immunotherapy in malignant tumors. Precision Radiation Oncol. 8, 42–46. https://doi.org/10.1002/pro6.1225 (2024).
Fan, Y., Mao, R., Yang, J. & NF-κB STAT3 signaling pathways collaboratively link inflammation to cancer. Protein Cell. 4, 176–185. https://doi.org/10.1007/s13238-013-2084-3 (2013).
Balkwill, F. & Mantovani, A. Inflammation and cancer: back to virchow?? Lancet 357, 539–545. https://doi.org/10.1016/s0140-6736(00)04046-0 (2001).
Chen, X. et al. Immune cell types and secreted factors contributing to Inflammation-to-Cancer transition and immune therapy response. Cell Rep. 26, 1965–1977.e1964 https://doi.org/10.1016/j.celrep.2019.01.080 (2019).
Quigley, D. A. & Kristensen, V. Predicting prognosis and therapeutic response from interactions between lymphocytes and tumor cells. Mol. Oncol. 9, 2054–2062. https://doi.org/10.1016/j.molonc.2015.10.003 (2015).
Antonio, N. et al. The wound inflammatory response exacerbates growth of pre-neoplastic cells and progression to cancer. Embo J. 34, 2219–2236. https://doi.org/10.15252/embj.201490147 (2015).
Fridlender, Z. G. et al. Polarization of tumor-associated neutrophil phenotype by TGF-beta: N1 versus N2 TAN. Cancer Cell. 16, 183–194. https://doi.org/10.1016/j.ccr.2009.06.017 (2009).
Wang, X., Qiu, L., Li, Z., Wang, X. Y. & Yi, H. Understanding the multifaceted role of neutrophils in cancer and autoimmune diseases. Front. Immunol. 9, 2456. https://doi.org/10.3389/fimmu.2018.02456 (2018).
Wei, S. C., Duffy, C. R. & Allison, J. P. Fundamental mechanisms of immune checkpoint Blockade therapy. Cancer Discov. 8, 1069–1086. https://doi.org/10.1158/2159-8290.Cd-18-0367 (2018).
Yuan, K. et al. A novel T-cell exhaustion-related feature can accurately predict the prognosis of OC patients. Front. Pharmacol. 14, 1192777. https://doi.org/10.3389/fphar.2023.1192777 (2023).
Reading, J. L. et al. The function and dysfunction of memory CD8(+) T cells in tumor immunity. Immunol. Rev. 283, 194–212. https://doi.org/10.1111/imr.12657 (2018).
Peng, G. et al. Identification and validation of neurotrophic factor-related genes signature in HNSCC to predict survival and immune landscapes. Front. Genet. 13, 1010044. https://doi.org/10.3389/fgene.2022.1010044 (2022).
Qin, M. et al. Tumor-infiltrating lymphocyte: features and prognosis of lymphocytes infiltration on colorectal cancer. Bioengineered 13, 14872–14888. https://doi.org/10.1080/21655979.2022.2162660 (2022).
Wang, Q. et al. Interplay of sphingolipid metabolism in predicting prognosis of GBM patients: towards precision immunotherapy. J. Cancer. 15, 275–292. https://doi.org/10.7150/jca.89338 (2024).
Zhao, S. et al. Interaction, immune infiltration characteristics and prognostic modeling of efferocytosis-related subtypes in glioblastoma. BMC Med. Genomics. 16, 248. https://doi.org/10.1186/s12920-023-01688-4 (2023).
Chi, H. et al. Unraveling the role of disulfidptosis-related LncRNAs in colon cancer: a prognostic indicator for immunotherapy response, chemotherapy sensitivity, and insights into cell death mechanisms. Front. Mol. Biosci. 10, 1254232. https://doi.org/10.3389/fmolb.2023.1254232 (2023).
Schlesinger, M. Role of platelets and platelet receptors in cancer metastasis. J. Hematol. Oncol. 11, 125. https://doi.org/10.1186/s13045-018-0669-2 (2018).
Nieswandt, B., Hafner, M., Echtenacher, B. & Männel, D. N. Lysis of tumor cells by natural killer cells in mice is impeded by platelets. Cancer Res. 59, 1295–1300 (1999).
Palacios-Acedo, A. L. et al. Thrombo-Inflammation, and cancer: collaborating with the enemy. Front. Immunol. 10, 1805. https://doi.org/10.3389/fimmu.2019.01805 (2019). Platelets.
Verheul, H. M. et al. Platelet: transporter of vascular endothelial growth factor. Clin. Cancer Res. 3, 2187–2190 (1997).
Ferrara, N., Gerber, H. P. & LeCouter, J. The biology of VEGF and its receptors. Nat. Med. 9, 669–676. https://doi.org/10.1038/nm0603-669 (2003).
Arends, J. Malnutrition in cancer patients: causes, consequences and treatment options. Eur. J. Surg. Oncol. 50, 107074. https://doi.org/10.1016/j.ejso.2023.107074 (2024).
Tang, S. et al. The value of geriatric nutritional risk index in evaluating postoperative complication risk and Long-Term prognosis in elderly colorectal cancer patients. Cancer Manag Res. 12, 165–175. https://doi.org/10.2147/cmar.S234688 (2020).
Hu, W. H. et al. Preoperative malnutrition assessments as predictors of postoperative mortality and morbidity in colorectal cancer: an analysis of ACS-NSQIP. Nutr. J. 14, 91. https://doi.org/10.1186/s12937-015-0081-5 (2015).
Peng, J. et al. Single-cell RNA-seq highlights intra-tumoral heterogeneity and malignant progression in pancreatic ductal adenocarcinoma. Cell. Res. 29, 725–738. https://doi.org/10.1038/s41422-019-0195-y (2019).
Lohsiriwat, V. The influence of preoperative nutritional status on the outcomes of an enhanced recovery after surgery (ERAS) programme for colorectal cancer surgery. Tech. Coloproctol. 18, 1075–1080. https://doi.org/10.1007/s10151-014-1210-4 (2014).
Zhang, Z. et al. A nomogram to predict the risk of colorectal anastomotic leakage combining inflammatory-nutritional and abdominal aorta calcium index. Front. Surg. 9, 1008448. https://doi.org/10.3389/fsurg.2022.1008448 (2022).
Wang, Q. et al. IL1RN and PRRX1 as a prognostic biomarker correlated with immune infiltrates in colorectal cancer: Evidence from bioinformatic analysis. Int J Genomics 2723264 https://doi.org/10.1155/2022/2723264 (2022).
Li, Z., Su, Y., Cui, Y., Yin, Y. & Li, Z. Multi-sequence MRI-based clinical-radiomics models for the preoperative prediction of microsatellite instability-high status in endometrial cancer. Precision Radiation Oncol. 9, 43–53. https://doi.org/10.1002/pro6.70000 (2025).
Funding
The study was authorized by the Ethics Committee of Xinjiang Medical University Cancer Hospital (Approval No. K-2024056) and the Ethics Committee of People’s Hospital of Bortala Mongolian Autonomous Prefecture (Approval No. LLSH20241221).
Author information
Authors and Affiliations
Contributions
Zeliang Zhao and Yi Chen contributed to study concept and design. Kuan Wang, Kejin Li, Ziyi Zhang, Xiangyue Zeng and Boxiang Zhang collected clinical data. Kuan Wang, Zhimin Wu, Yipeng Pan and Lucy Yue Lau contributed to analyze the data. Yi Chen contributed to prepare the manuscript. Kuan Wang provided critical feedback on methods, and supervised the study. All authors approved the final version to be published.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wang, K., Li, K., Zhang, Z. et al. Combined preoperative platelet-albumin ratio and cancer inflammation prognostic index predicts prognosis in colorectal cancer: a retrospective study. Sci Rep 15, 29500 (2025). https://doi.org/10.1038/s41598-025-15309-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-15309-w
Keywords
This article is cited by
-
ACSL4 as a potential ferroptosis target in hepatocellular carcinoma: from mechanisms to implications
European Journal of Medical Research (2026)
-
DMBX1 expression in colon cancer and its impact on prognosis and the tumor microenvironment
BMC Medical Genomics (2026)
-
Comparison of the safety and efficacy of different neoadjuvant therapy cycles for locally advanced gastric cancer: a retrospective propensity score-matched cohort study
World Journal of Surgical Oncology (2026)
-
PLOD2 promotes proliferation, migration and invasion of colorectal cancer cells via PI3K-AKT-GSK3β signaling pathway
Scientific Reports (2026)
-
Single-cell and machine learning-based analysis of the molecular mechanism of Banxia Xiexin Decoction in the treatment of gastric cancer
Cytotechnology (2026)