Abstract
Organic acidemias (OADs) are a group of congenital metabolic disorders whose incidence, disease spectrum, and genetic profiles differ greatly across countries. This study aimed to determine the characteristics of OADs in Quanzhou, China. A total of 693,797 newborns were screened for OADs from 2014 to 2023, and the acylcarnitine and genetic profiles of patients with OADs were analysed. Sixty-nine patients were confirmed to have OADs, with an overall incidence of 1/10,055 newborns. Seven types of OADs were identified, of which 18 were 2-methylbutyryl-CoA dehydrogenase deficiency (MBAD), 18 were 3-methylcrotonyl-CoA carboxylase deficiency (3-MCCD), 13 were glutaric acidemia type 1 (GA-1), nine were isobutyryl-CoA dehydrogenase deficiency (IBDD), five were isovaleric acidemia (IVA), four were methylmalonic acidemia (MMA), and two were propionic academia (PA). All but one of the patients with MBAD had elevated isovalerylcarnitine levels and corresponding ratios during screening. All patients with GA-1 had elevated glutarylcarnitine levels and corresponding ratios during screening, except for one with a low free carnitine level. The remaining patients presented with elevated acylcarnitine levels during screening and recall. Several variant hotspots were identified in the ACADSB, MCCC1, MCCC2, GCDH, ACAD8, and IVD. The overall incidence of OADs in the study population was 1/10,055 newborns, with MBAD, 3-MCCD, and GA-1 being the three most common. The acylcarnitine profiles and genetic features of most OADs have been elucidated. Our findings provide useful information for newborn screening, genetic diagnosis, and the prevention of OADs.
Similar content being viewed by others
Introduction
Organic acidemias (OADs) are a group of congenital metabolic disorders that have been detected since the introduction of analytical gas chromatography techniques in the 1960s1. Most OADs are caused by defects in the mitochondrial breakdown of CoA-activated carbonic acids, such as isovaleryl-CoA, methylmalonyl-CoA, propionyl-CoA, and glutaryl-CoA. A few of these toxic metabolites cause acute and chronic organ dysfunction with high energy requirements by inducing mitochondrial dysfunction, energy damage, and oxidative stress. The clinical presentation of OADs can vary greatly with respect to age of onset, organ involvement, and symptom severity2. Patients with OADs can present with acute metabolic collapse during the neonatal period or later in childhood, with unexplained developmental delays accompanied by recurrent episodes of metabolic decompensation. However, many individuals identified by newborn screening (NBS) remain asymptomatic throughout their lives3.
NBS for OADs has been implemented in many countries, and with the widespread application of tandem mass spectrometry (MS/MS) for NBS, an increasing number of patients with OADs have been identified. Early detection using NBS allows for early intervention and better outcomes, and potentially acute life-threatening events can be prevented. However, data on the incidence, disease spectrum, and genetic profiles of OADs are heterogeneous. The number of screened newborns and patients with OAD-positive reported in many studies is relatively limited. NBS using MS/MS was initiated in Quanzhou, China, in January 2014. This study was designed to present ten years of experience with NBS for OADs in nearly 700,000 newborns to aid in screening, diagnosis, and prevention.
Methods
Participants
Quanzhou is a prefecture-level city in southeast China. All newborns were born in Quanzhou, China, and screened at the NBS Center of Quanzhou Maternity and Children’s Hospital. Between January 2014 and December 2023, 693,797 newborns (90.5% of total births) were screened for inherited metabolic disorders using MS/MS. Newborns with positive results indicative of OADs and those who were genetically diagnosed were included. This study was approved by the Ethics Committee of the Quanzhou Maternity and Children’s Hospital (reference number: 2024-IRB-107). Written informed consent was obtained from the parents of newborns before screening.
NBS using MS/MS
Sample collection and testing were conducted as described in our previous articles4,5. Briefly, neonatal heel prick blood samples were collected and spotted on Whatman 903 filter paper (Guthrie card) between 72 h and 7 days after delivery. The dried blood spot (DBS) samples were delivered by cold chain transportation to the NBS laboratory within 3 days and pre-processed using NeoBase™ non-derivatised MS/MS reagent kits (PerkinElmer, Turku, Finland) according to the manufacturer’s protocol. The acylcarnitine concentration in DBS was quantitatively determined using an AQCUTY TQD tandem mass spectrometer (Waters, Milford, MA, USA). The cut-off values were set at the 99.5th (0.05th ) percentile in our laboratory and were gradually modified with an increase in screened numbers and positive patients. Newborns with MS/MS results that exceeded the cut-off values indicative of OADs were referred for repeat screening within one week. The re-examined positive samples were transferred to specialists for genetic analysis and diagnosis.
Genetic analysis and diagnosis
Genetic testing and data analysis were performed by Hangzhou Biosan Clinical Laboratory Co., Ltd. (Hangzhou, Zhejiang, China). Targeted next-generation sequencing (NGS) was conducted on suspected patients using a sequencing panel of 94 genes associated with inborn metabolic errors as previously described4,5. All possible disease-causing variants detected using NGS were validated using Sanger sequencing. Individuals with abnormal acylcarnitine profiles and biallelic pathogenic/likely pathogenic variants of disease-causing genes relevant to OADs were considered OAD-positive. Individuals with one or no disease-causing variants were excluded.
Results
NBS and disease spectrum of OADs
Among the 693,797 screened newborns, 3961 (0.57%) newborns had positive NBS results indicative of OADs; 3822 (96.5%) were successfully recalled, while 432 (11.3%) still had positive MS/MS results. After genetic analysis and differential diagnosis, 69 patients were confirmed to have OADs, meaning that the positive predictive value (PPV) was 1.8%. The overall incidence of OADs was 1/10,055 newborns.
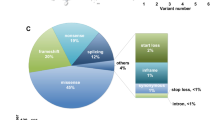
Seven types of OADs were identified: 18 were 2-methylbutyryl-CoA dehydrogenase deficiency (MBAD, OMIM #600301), 18 were 3-methylcrotonyl-CoA carboxylase deficiency (3-MCCD, OMIM #210210), 13 were glutaric acidemia type 1 (GA-1, OMIM #231670), nine were isobutyryl-CoA dehydrogenase deficiency (IBDD, OMIM #611283), five were isovaleric acidemia (IVA, OMIM #243500), four were methylmalonic acidemia (MMA, OMIM #609058), and two were propionic academia (PA, OMIM #606054). MBAD (1/38,544 newborns, 26.09%), 3-MCCD (1/38,544 newborns, 26.09%), and GA-1 (1/53,369 newborns, 18.84%) were the three most common OADs. The distribution and incidence of OADs are shown in Table 1.
Acylcarnitine profile analysis
All but one patient with MBAD had mildly or moderately elevated isovalerylcarnitine (C5) levels and corresponding ratios during NBS. The initial NBS result showed that patient No. 6 only had a low free carnitine (C0) level, which was suspected to be a primary carnitine deficiency; however, elevated C5 levels and corresponding ratios were observed during the recall review. Mean C5 concentrations during NBS and recall were 0.52 ± 0.22 and 0.74 ± 0.35 µmol/L (cut-off value: 0.03–0.35 µmol/L), respectively. All patients with 3-MCCD had moderately to markedly elevated 3-hydroxyisovalerylcarnitine (C5OH) levels and corresponding ratios. Mean C5OH concentrations during NBS and recall were 4.98 ± 3.60 and 7.02 ± 6.02 µmol/L (cut-off value: 0.07–0.5 µmol/L), respectively. In addition, two patients had low C0 levels during NBS, and nine patients (Nos. 1, 9–14, 17, and 18) had low C0 levels after recall. All patients with GA-1 had moderately to markedly elevated glutarylcarnitine (C5DC) levels and corresponding ratios during NBS, except for one patient (No. 7) with a low C0 level. Mean C5DC concentrations during NBS and recall were 2.27 ± 1.04 and 2.06 ± 1.22 µmol/L (cut-off value: 0.03–0.3 µmol/L), respectively. The C5DC level of one patient (No. 3) fell into the normal range during recall, while three patients had low C0 levels. The remaining patients presented with elevated acylcarnitine profiles during NBS and recall (Table 2) .
Genetic analysis
Eight distinct ACADSB variants were identified in patients with MBAD. The most common variant was c.1165 A > G (p.M389V) with an allele frequency of 50%, followed by c.275 C > G (p.S92*) (16.67%) and c.923G > A (p.C308Y) (11.11%). Ten MCCC1 variants and seven MCCC2 variants were identified in patients with 3-MCCD; 50% had MCCC1 variants, and 50% had MCCC2 variants. The most frequent MCCC1 variant was c.1331G > A (p.R444H), with an allele frequency of 31.25%. The most frequent MCCC2 variant was c.351_353delTGG (p.G118del) (50%), followed by c.1103delG (p.G368Vfs*70) (16.67%). Eleven different GCDH variants were detected in patients with GA-1, with c.1244–2 A > C (57.69%) being the most common variant. The two most common ACAD8 variants were c.286G > A (p.G96S) (44.44%) and c.444G > T (p.P148P) (22.22%). Eight different IVD variants were detected in patients with IVA, with c.1208 A > G (p.Y403C) (30%) being the most common variant. Five MMUT variants and one MMAA variant were identified in patients with MMA, while two PCCB variants and one deletion were detected in patients with PA (Supplementary File 1).
Discussion
The incidence of OADs detected using MS/MS varies widely among countries, and it is estimated to be 1/6565 in Singapore6, 1/22,000 in Japan7, 1/13,532 in Australia8, and 1/16,300 in the United States9. OAD incidence varies greatly throughout China, and it is higher in northern China due to the frequent detection of MMA. This study summarised ten years of NBS data for OADs in Quanzhou, indicating that the incidence was 1/10,055 newborns; this was similar to that reported in southern cities such as Shanghai (1/10,139)10 and Jiangsu (1/12,465)11 but much lower than that reported in northern cities such as Jining (1/3956)12 and Xi’an (1/5220)13.
Although MS/MS-based NBS has been performed in China for many years, the overall PPV of most laboratories is generally low10, possibly because of the lack of further adoption of second-tier tests. The PPV of OADs in this study was 1.8%, implying a false positive rate of 98.2%, which poses a great challenge and financial burden to the healthcare system. We have not observed any false-negative patients, but there may be false negatives that have occurred without our attention, as they can occur if the cut-off value is not set properly or if a comprehensive genetic diagnosis is not performed.
The most common OAD in this study was MBAD, which may be attributed to the recurrent c.1165 A > G (p.M389V) mutation observed in the selected population. Previous studies have shown that this disorder has a particularly high incidence among the Hmong ethnic population14. However, most patients with MBAD detected through NBS are asymptomatic, and their inclusion in NBS panels remains controversial15. Similar to previous studies, no clinical symptoms were found in our patients.
Examining the acylcarnitine profiles of 18 patients with MBAD, one patient had a normal C5 level, and six patients (Nos. 7, 8, 11, 13, 15, and 18) had levels near the cut-off during NBS, indicating that these patients were easily missed by NBS. Genetically, c.1165 A > G (p.M389V) is a founder mutation observed in Hmong patients. Half of the patients in this study carried this variant, suggesting that it is a founder mutation in the southern Chinese population. In addition, c.275 C > G (p.S92*) and c.923G > A (p.C308Y) were relatively common in this cohort.
3-MCCD is the most common OAD in many countries, with an incidence ranging from 1/2400 to 43,00016,17,18. However, there is debate regarding whether 3-MCCD should be included in NBS panels because of its benign course. As of 2015, the Israeli Ministry of Health has stopped national NBS for 3-MCCD16. Although two patients in this cohort had clinical manifestations, it remains uncertain whether they were even related to 3-MCCD. Currently, all NBS centres in China retain 3-MCCD in their NBS programs. Our data showed that 3-MCCD had a high incidence in Quanzhou (1/38,544), which is close to that reported in Shanghai (1/42,003)10 and Jiangsu province (1/38,286)19 and higher than that reported in Zhejiang province (1/83,068)20. In addition to the typical elevation of C5OH in 3-MCCD, a few patients (11.1%) had secondary carnitine deficiency during NBS, and half of all patients had secondary carnitine deficiency during recall. Most patients with secondary carnitine deficiency (77.8%, 7/9) had mutations in MCCC2. In China, previous studies have shown that c.639 + 2T > A and c.1144_1147delinsTTTT (p.K382-K383delinsF*) are the most common mutations in MCCC1 and MCCC2, respectively10,19,20; in contrast to previous studies, these two mutations were rarely detected in our patients. Our data showed that c.1331G > A (p.R444H) and c.351_353delTGG (p.G118del) were the most common mutations in MCCC1 and MCCC2, respectively, suggesting that the MCCC1 and MCCC2 mutational spectra vary significantly across regions.
GA-1 is a rare inherited neurometabolic disorder that has been included in many national NBS programs with an estimated worldwide incidence of 1/90,000–120,000 newborns21. Our data indicates that the vast majority of patients have obvious clinical symptoms, and NBS for GA-1 is of great significance. The estimated incidence in Quanzhou population was 1/53,369, which is higher than that reported in Zhejiang (1/221,053)22 and Jiangsu provinces (1/89,335)11. Certain patients with low excretion can be easily overlooked, and several false-negative cases with normal C5DC levels have been reported23,24. Two situations related to low C0 levels must be considered : (a) those babies whose mothers are carnitine deficient and (b) when low C0 levels are secondary to inherited metabolic disorders. Both situations can lead to insufficient carnitine levels due to the conjugation to acyl residues, and must be considered because if an organic acidemia or fatty acid oxidation deficiency exist, biomarkers will not be present and the possibility of obtaining a false negative result. One patient with normal C5DC levels was identified in this study; this patient was unexpectedly detected because of persistently low C0 levels. Additionally, four patients (30.8%, 4/13) were observed to have secondary carnitine deficiency during recall. Several GCDH pathogenic variants have been identified in specific ethnic groups; c.1204 C > T (p.R402W) is highly prevalent in Europe and in the Chinese population25. Consistent with previous studies, c.1204 C > T (p.R402W), with an allelic frequency of 57.69%, was the most common variant in this study. The recurrence of this variant in the selected population suggests a possible founder effect.
As evidence suggests that IBDD is a biochemical phenotype with a benign clinical course, consideration may be given to the removal of this disorder from NBS programs or retaining it while informing parents of its benign nature and that it does not require treatment26. In either case, C4 can be retained as a valuable marker for the differential diagnosis of glutaric academic type II. Nine patients with IBDD with elevated C4 levels were identified in our population, with an incidence of 1/77,089, which is similar to that reported in Zhejiang Province (1/62,599)27 and lower than that reported in Ningbo City (1/30,300)28. Consistent with most published cases, all patients in this study turned out healthy and had no clinical characteristics. Our previous study reported that c.286G > A (p.G96S) is a potential hotspot variant in the Chinese population5. With an increasing number of patients with IBDD being detected using NBS, subsequent studies have confirmed that this variant is most prevalent in Chinese patients27,28. This study also confirmed previous findings that c.286G > A (p.G96S) is the most common ACAD8 variant, followed by c.444G > T (p.P148P) (22.22%).
The remaining three types of OADs (IVA, MMA, and PA) had relatively low incidences, each of which was less than 1/100,000. Although both IVA and PA are relatively rare in the Chinese population, MMA is the most common OAD in northern China12,29. The incidence of MMA in this study was significantly lower than that reported in northern China, indicating that MMA is rare in southern China. However, all three of these diseases can cause severe clinical manifestations. Our data indicate that despite the implementation of NBS, the prognosis for patients with severe clinical symptoms is very poor. Especially for MA and PA, severe patients often present obvious clinical manifestations before undergoing NBS. The genetic profiles of these diseases in the study population are poorly understood because their detection rates are relatively low. c.1208 A > G (p.Y403C) was observed in the IVD with an allele frequency of 30%, which coincided with a previous study showing that c.1208 A > G (p.Y403C) is a hotspot variant in the Chinese population30. The genetic characteristics of MMA and PA should be further clarified by collecting additional screening samples and patients.
This study had several limitations. First, all patients were diagnosed using targeted NGS, so disease-causing pathogenic variants in regulatory regions or deep introns cannot be detected. Additionally, a small number of newborns with positive NBS results may remain underdiagnosed due to the lack of further confirmatory analysis. Finally, the PPV in this study was much lower than that of developed countries9,31, emphasising the importance of introducing second-tier tests to improve performance.
Conclusions
In total, 69 patients with seven different types of OADs were identified during the 10-year study period. The overall incidence of OADs in the study population was 1/10,055 newborns, with MBAD, 3-MCCD, and GA-1 being the three most common OADs. The acylcarnitine profiles and genetic features of most OADs were elucidated. Several variant hotspots were identified in ACADSB, MCCC1, MCCC2, GCDH, ACAD8, and IVD. Our findings provided useful information for NBS, genetic diagnosis, and prevention of OADs.
Data availability
Data included in article/supp. Material/referenced in article. The datasets used and analyzed in this study are available from the corresponding author upon reasonable request.
Abbreviations
- OADs:
-
Organic acidemias
- NBS:
-
Newborn screening
- MS/MS:
-
Tandem mass spectrometry
- DBS:
-
Dried blood spot
- PPV:
-
Positive predictive value
- MBAD:
-
2-Methylbutyryl-CoA dehydrogenase deficiency
- 3-MCCD:
-
3-Methylcrotonyl-CoA carboxylase deficiency
- GA-1:
-
Glutaric acidemia type 1
- IBDD:
-
Isobutyryl-CoA dehydrogenase deficiency
- IVA:
-
Isovaleric academia
- MMA:
-
Methylmalonic academia
- PA:
-
Propionic academia
- C5:
-
Isovalerylcarnitine
- C5OH:
-
3-Hydroxyisovalerylcarnitine
- C0:
-
Free carnitine
- C5DC:
-
Glutarylcarnitine
References
Dimitrov, B. et al. Organic acidurias: major gaps, new challenges, and a yet unfulfilled promise. J. Inherit. Metab. Dis. 44 (1), 9–21 (2021).
Heringer, J. et al. Impact of age at onset and newborn screening on outcome in organic acidurias. J. Inherit. Metab. Dis. 39 (3), 341–353 (2016).
Ramsay, J., Morton, J., Norris, M. & Kanungo, S. Organic acid disorders. Ann. Transl Med. 6 (24), 472 (2018).
Lin, Y. et al. Biochemical, clinical, and genetic characteristics of short/branched chain Acyl-CoA dehydrogenase deficiency in Chinese patients by newborn screening. Front. Genet. 10, 802 (2019).
Lin, Y. et al. Clinical, biochemical and genetic analysis of Chinese patients with isobutyryl-CoA dehydrogenase deficiency. Clin. Chim. Acta. 487, 133–138 (2018).
Lim, J. S. et al. Inborn error of metabolism (IEM) screening in Singapore by electrospray ionization-tandem mass spectrometry (ESI/MS/MS): an 8 year journey from pilot to current program. Mol. Genet. Metab. 113 (1–2), 53–61 (2014).
Shibata, N. et al. Diversity in the incidence and spectrum of organic acidemias, fatty acid oxidation disorders, and amino acid disorders in Asian countries: selective screening vs. expanded newborn screening. Mol. Genet. Metab. Rep. 16, 5–10 (2018).
Kasper, D. C. et al. The National Austrian newborn screening program - eight years experience with mass spectrometry. Past, present, and future goals. Wien Klin. Wochenschr. 122 (21–22), 607–613 (2010).
Frazier, D. M. et al. The tandem mass spectrometry newborn screening experience in North carolina: 1997–2005. J. Inherit. Metab. Dis. 29 (1), 76–85 (2006).
Hao, L. et al. Screening of 1.17 million newborns for inborn errors of metabolism using tandem mass spectrometry in shanghai, china: A 19-year report. Mol. Genet. Metab. 141 (1), 108098 (2024).
Yang, Y. et al. Application of Next-Generation sequencing following tandem mass spectrometry to expand newborn screening for inborn errors of metabolism: A multicenter study. Front. Genet. 10, 86 (2019).
Yang, C. et al. Newborn screening and diagnosis of inborn errors of metabolism: A 5-year study in an Eastern Chinese population. Clin. Chim. Acta. 502, 133–138 (2020).
Zhang, R. et al. Spectrum analysis of inborn errors of metabolism for expanded newborn screening in a Northwestern Chinese population. Sci. Rep. 11 (1), 2699 (2021).
Van Calcar, S. C. et al. Prevalence and mutation analysis of short/branched chain acyl-CoA dehydrogenase deficiency (SBCADD) detected on newborn screening in Wisconsin. Mol. Genet. Metab. 110 (1–2), 111–115 (2013).
Porta, F., Chiesa, N., Martinelli, D. & Spada, M. Clinical, biochemical, and molecular spectrum of short/branched-chain acyl-CoA dehydrogenase deficiency: two new cases and review of literature. J. Pediatr. Endocrinol. Metab. 32 (2), 101–108 (2019).
Rips, J. et al. Primary and maternal 3-methylcrotonyl-CoA carboxylase deficiency: insights from the Israel newborn screening program. J. Inherit. Metab. Dis. 39 (2), 211–217 (2016).
Thomsen, J. A., Lund, A. M., Olesen, J. H., Mohr, M. & Rasmussen, J. Is L-Carnitine supplementation beneficial in 3-Methylcrotonyl-CoA carboxylase deficiency?? JIMD Rep. 21, 79–88 (2015).
Lam, C. et al. Analysis of cases of 3-methylcrotonyl coa carboxylase deficiency (3-MCCD) in the California newborn screening program reported in the state database. Mol. Genet. Metab. 110 (4), 477–483 (2013).
Wang, H. et al. 3-Methylcrotonyl-CoA carboxylase deficiency newborn screening in a population of 536,008: is routine screening necessary? J. Pediatr. Endocrinol. Metab. 32 (12), 1321–1326 (2019).
Cheng, Y. et al. Newborn screening for 3-methylcrotonyl-CoA carboxylase deficiency in Zhejiang province, China. Clin. Chim. Acta. 542, 117266 (2023).
Boy, N. et al. Recommendations for diagnosing and managing individuals with glutaric aciduria type 1: third revision. J. Inherit. Metab. Dis. 46 (3), 482–519 (2023).
Lin, Y. et al. Biochemical, molecular, and clinical features of patients with glutaric acidemia type 1 identified through large-scale newborn screening in Zhejiang province, China. Clin. Chim. Acta. 530, 113–118 (2022).
Spenger, J. et al. Glutaric aciduria type I missed by newborn screening: report of four cases from three families. Int J. Neonatal Screen 7(2). (2021).
Boy, N. et al. Newborn screening: A disease-changing intervention for glutaric aciduria type 1. Ann. Neurol. 83 (5), 970–979 (2018).
Marti-Masso, J. F. et al. Exome sequencing identifies GCDH (glutaryl-CoA dehydrogenase) mutations as a cause of a progressive form of early-onset generalized dystonia. Hum. Genet. 131 (3), 435–442 (2012).
Sadat, R. et al. Increased parental anxiety and a benign clinical course: infants identified with short-chain acyl-CoA dehydrogenase deficiency and isobutyryl-CoA dehydrogenase deficiency through newborn screening in Georgia. Mol. Genet. Metab. 129 (1), 20–25 (2020).
Feng, J. et al. Phenotype, genotype and long-term prognosis of 40 Chinese patients with isobutyryl-CoA dehydrogenase deficiency and a review of variant spectra in ACAD8. Orphanet J. Rare Dis. 16 (1), 392 (2021).
Zhuang, D. Y. et al. Identification of six novel variants of ACAD8 in Isobutyryl-CoA dehydrogenase deficiency with increased C4 carnitine using tandem mass spectrometry and NGS sequencing. Front. Genet. 12, 791869 (2021).
Han, B. et al. Clinical presentation, gene analysis and outcomes in young patients with early-treated combined methylmalonic acidemia and homocysteinemia (cblC type) in Shandong province, China. Brain Dev. 38 (5), 491–497 (2016).
Li, Y. et al. Eight novel mutations detected from eight Chinese patients with isovaleric acidemia. Clin. Chim. Acta. 498, 116–121 (2019).
Couce, M. L. et al. Evaluation and long-term follow-up of infants with inborn errors of metabolism identified in an expanded screening programme. Mol. Genet. Metab. 104 (4), 470–475 (2011).
Acknowledgements
We thank all the participants for their help and support. We would like to thank Editage (www.editage.cn) for English language editing.
Funding
This study was supported by Joint Funds for the Innovation of Science and Technology, Fujian province (Grant number: 2024Y9458) and the Quanzhou Municipal Science and Technology Plan Project (Grant number: 2025QZNG025).
Author information
Authors and Affiliations
Contributions
Yiming Lin: Conceived and designed the experiments; Performed the experiments; Wrote the paper. Chunmei Lin: Contributed reagents, materials, analysis tools or data; Zhenzhu Zheng: analysed and interpreted the data; Yanru Chen: analysed and interpreted the data; Faming Zheng: Contributed reagents, materials, analysis tools or data; Weihua Lin: analysed and interpreted the data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
This study was approved by the Ethical Committee of Quanzhou Maternity and Children’s Hospital and was performed in accordance with the Declaration of Helsinki. Written informed consent was obtained from the parents of all infants for collection of samples and publication of medical data.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Lin, Y., Lin, C., Zheng, Z. et al. Large-scale newborn screening for organic acidemias in Quanzhou, China: a 10-year retrospective observational study. Sci Rep 15, 30592 (2025). https://doi.org/10.1038/s41598-025-15625-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-15625-1