Abstract
Sharks often modify their behaviour to obtain food from humans. Food provisioning can lead to behavioural conditioning, which can alter resource use patterns and interactions within and between species. We used acoustic telemetry and dietary biomarkers (fatty acids and stable isotopes) to quantify dusky (Carcharhinus obscurus; n = 18) and Galapagos (C. galapagensis; n = 16) shark associations with fishing piers at Norfolk Island, Australia between 2021 and 2024. Individuals visited the piers, on average, once every four days (~ 3 h daily), with dusky sharks usually arriving in the early afternoon and staying until dusk while Galapagos sharks arrived at dusk and left in the early morning. Despite this temporal partitioning, the diets of both species were indistinguishable from each other and showed only limited contribution of discarded fish; however, fish scraps were more common in the diet of sharks with high residency to the piers. These findings indicate that although incidental feeding contributes to the presence of sharks at the piers, it has likely not markedly altered natural foraging behaviour with regular visits to the piers only conducted intermittently by a subset of the tagged population to supplement diet. Understanding how sharks use the piers in response to human activities will help assess current waste disposal practices at Norfolk Island and ongoing monitoring needs.
Similar content being viewed by others
Introduction
The introduction of food to animals in natural environments by humans (often derived from disparate ecosystems), and the unintended ecological, social, and economic consequences, are a major concern for managers and impacted stakeholders1. Food provisioning, where animals obtain food from humans either directly or indirectly (e.g. incidental feeding), is common in the aquatic realm, especially when recreational fishers clean their catch and discard remains back in the water2. Sharks and rays commonly scavenge in areas of incidental feeding where fishing catch is cleaned due to the higher net energy gains attainable relative to natural conditions3.
Associations with fishing activities and consumption of discards can have a range of behavioural, physiological, and environmental effects on elasmobranchs. For example, natural movements may be altered to follow fishing vessels4; site fidelity and residency may increase around provisioning sites5,6; shifts from solitary to aggregatory behaviour may occur7,8; activity patterns may change9,10; species assemblages and composition may shift11,12,13; and anticipatory behaviour that persists for long periods after positive reinforcement is stopped may develop14. The effects from associations with food provisioning activities can be cumulative and develop rapidly7,14,15, vary across species, age groups, sex classes, and locations, and in some instances have had cascading effects such as increased disease transmission, inbreeding due to unnatural aggregations, modified dominance structure, and changes in predator-prey relationships8,16. In contrast, studies on sharks have also found that food provisioning can be relatively harmless with no observed effect on site fidelity17, behaviour and diet18, activity19, nutritional condition20 or daily energy budget21. Ultimately, as with any foraging behaviour, the benefits of foraging on human-associated food (e.g. reduced search time) need to outweigh the costs (e.g. mortality risk) to be viable long-term for the individual and, consequently, its population (e.g.7).
Fishing is a popular activity in Oceania, with approximately 37 million people in this region engaging in fishing activities22. A fraction of this contingent is found on Norfolk Island, a small island group located ~ 1,400 km from the Australian mainland. The remote and small-island characteristics of Norfolk Island limit local food production and transport from mainland Australia; accordingly, fishing in the productive waters surrounding the islands is an important activity and source of food for many of the ~ 2,000 inhabitants. With limited waste disposal options, it is common practice to clean fish (i.e. scale, gut, fillet) immediately upon return at one of only two piers where boats can be launched and retrieved (Cascade and Kingston piers; Fig. 1). Boats return from fishing at various times, but the small-scale commercial fishers can spend the entire day fishing further offshore, often returning in late afternoon and cleaning until dusk or later. Carcasses of the cleaned fish are discarded directly into confined waters adjacent to the piers. As a result, dusky (Carcharhinus obscurus) and Galapagos (C. galapagensis) sharks are frequently seen scavenging on the fish scraps near the piers. Previous research has shown that juvenile dusky (< 2.8 m total length [TL]23; 50% length-at-maturity ~ 2.7 m TL and 2.8 m TL for males and females, respectively24) and Galapagos (< 2.0 m TL23; minimum length-at-maturity > 2.0 m and > 2.1 m TL for males and females, respectively25) sharks remain year-round at Norfolk Island primarily being detected at the piers or adjacent sites nearby23. Similar to other locations2,26 the predictable aggregation of sharks at the piers has led to these areas becoming a local tourist attraction.
The aggregation of sharks at the piers, presumably due to the discarding of fish waste, has been occurring for over 30 years27. Fishing activity at Norfolk Island occurs throughout the year with consistent effort28 (M. Scott pers. obs.). The impact of this human-derived food source on the space use, behaviour, and diet of the two species commonly observed scavenging at the piers, the dusky shark and the Galapagos shark, is unknown. The main goal of this study was to explore space use and diet differences of these sympatric species at the piers in the context of small-scale niche partitioning and impact of human activities. Specifically, the objectives were to (1) evaluate diel patterns of occurrence between species, (2) investigate patterns of co-occurrence and any associated density-dependent relationships, and (3) quantify spatial and trophic overlap. Findings will contribute to local management considerations in relation to incidental shark feeding activities and will build on ecological knowledge of how provisioning affects the long-term behaviour of sympatric shark species.
Results
Movements of dusky and Galapagos sharks
The acoustic telemetry data from 18 dusky (mean TL ± standard error: 2.28 ± 0.07 m; Table 1) and 16 Galapagos (1.66 ± 0.05 m; Table 1) sharks, detected between February 2021 and April 2024 (Fig. 2), were analysed. One dusky shark was not analysed because it was only detected at the piers on one day prior to a period of no receiver coverage at Cascade Pier due to a lost receiver (see Methods). This individual was detected at different sites around the island but primarily at Simons and Archway (sites adjacent to Cascade Pier; Fig. S1). One dusky shark and two Galapagos sharks were also removed from analysis because they were never detected at the piers. These three individuals were only detected at a few sites, all of which were adjacent to the piers they were released at (the dusky shark also overlapped with the period of no receiver coverage at Cascade Pier; Fig. S1). Finally, 14 dusky sharks and 14 Galapagos sharks were never detected at any receiver following tagging and could not be analysed (see23 for additional information).
The mean detection periods of the analysed sharks were 436 ± 63 days (dusky sharks; Table 1) and 413 ± 82 days (Galapagos sharks; Table 1), and the mean residency (i.e. proportion of days an individual was detected within its detection period) at either pier was 0.26 ± 0.06 (dusky sharks; Table 1) and 0.22 ± 0.02 days (Galapagos sharks; Table 1).
Diel presence at piers
Models exploring the presence of sharks across diel categories were completed separately for the different species and tagging piers because the interaction between diel category, pier, and species were included in the best performing global model (R2 conditional = 0.52; Table S2). The models that included the interaction between diel periods and TL were the best performing models across species and piers, except for dusky sharks at Cascade Pier, for which diel period was the only fixed effect included in the top model (Table 2). Diel periods better explained presence in Galapagos sharks (R2 conditional = 0.66 (Kingston) and 0.69 (Cascade); Table 2) compared to dusky sharks (R2 conditional = 0.39 (Kingston) and 0.47 (Cascade); Table 2). For dusky sharks at Cascade Pier, morning and afternoon were the most frequently visited periods compared to dusk and night, whereas at Kingston Pier, afternoon periods were most used followed by dusk and night (Fig. 3). For Galapagos sharks, night was the most frequently visited period compared to morning, afternoon, and dusk at both piers. Here, our results are not standardised relative to diel period duration (i.e. dusk is shorter and night is longer than other periods resulting in different temporal availability of being detected) because we were interested in providing an absolute likelihood of presence during each period (Fig. 3). When diel period durations were standardised, dusky sharks were more likely to be present in the afternoon and at dusk, while Galapagos sharks were present later, beginning at dusk and throughout the night (Fig. 4). On days when individuals were present at the piers, they typically spent 3.8 ± 0.1 and 1.9 ± 0.1 (dusky shark) and 3.1 ± 0.1 and 2.0 ± 0.1 (Galapagos shark) hours at Cascade and Kingston piers, respectively.
There was a positive relationship between dusky shark use of Kingston Pier and shark size for morning and afternoon periods where an increase in TL from 1.8 to 2.8 m was predicted to triple and double the likelihood of presence, respectively (Fig. 5). Subsequently, there was a negative relationship between dusky shark use at Kingston Pier and shark size for dusk and night (Fig. 5). However, random effects (i.e. animal ID) explained more variation than fixed effects for dusky shark models, indicating a high amount of inter-individual variation. For Galapagos sharks, there was a marginal decrease in use of piers at night by larger individuals at both sites, although larger individuals increased use of piers during the afternoon, especially at Cascade Pier. (Fig. 5).
Relative to other sites, Cascade Pier had the highest use by dusky sharks during morning, afternoon, and dusk; at night they were more common at Simons, the receiver site adjacent to Cascade (Fig. S2). Detections at Kingston were more common than at adjacent sites (i.e. Surf and Nepean) except during morning at Surf (Fig. S2). For Galapagos sharks, Nepean was a highly visited location, with greater occurrence than Kingston Pier during morning, afternoon, and night (Fig. S3). The only diel period when Galapagos sharks were more likely to be at one of the piers compared to other sites was at dusk (Fig. S3).
Co-occurrence of dusky and Galapagos sharks
The maximum number of individuals detected during the same hourly period were 6 (5 dusky and 1 Galapagos, as well as 1 dusky and 5 Galapagos sharks) and 7 (2 dusky and 5 Galapagos sharks) at Cascade and Kingston piers, respectively. There were 55 and 78 ‘Dusky-Dusky’ dyads (individual pairings with overlapping detection periods across all receiver sites), 50 and 84 ‘Galapagos-Galapagos’ dyads, and 118 and 177 ‘Dusky-Galapagos’ dyads at Cascade and Kingston piers, respectively (Table 3). The mean (± se) hourly simple ratio index (SRI; number of hourly periods during overlapping detection periods in which both individuals of a dyad were present at a pier relative to the number of hourly periods at least one individual was present) for ‘Dusky-Dusky’, ‘Galapagos-Galapagos’, and ‘Dusky-Galapagos’ dyads was 0.08 ± 0.01, 0.06 ± 0.01, and 0.02 ± 0.01, and 0.04 ± 0.01, 0.04 ± 0.01 and 0.02 ± 0.01 at Cascade and Kingston piers, respectively (Table 3). These hourly SRI values equated to being co-detected (during at least one hourly period) an average of 14%, 14%, and 5% and 6%, 7%, and 3% of days for ‘Dusky-Dusky’, ‘Galapagos-Galapagos’, and ‘Dusky-Galapagos’ dyads at Cascade and Kingston piers, respectively (Table 3). While the hourly SRI values were low (i.e. SRI is a scale between 0 and 1), for each species combination, the observed probability of dyad co-occurrence was higher than the expected probability based on the frequency that each individual randomly occurred at the piers each hour (Fig. S4). Hourly co-occurrences of dusky and Galapagos sharks were typically distributed from 12:00 to midnight at Cascade Pier and 17:00 to 21:00 at Kingston Pier (Fig. 4). Hourly co-occurrences varied in structure at Cascade and Kingston piers but were broadly distinguished by species (Fig. 6). Modularity (Q), the degree to which a network can be divided into distinct and relatively independent clusters/communities (low Q ~ one interconnected community vs. high Q ~ distinct communities) was relatively high at both sites (QCascade = 0.52; QKingston = 0.40) indicating the likelihood of separate interconnected communities that were either spatially or temporally stratified. Three distinct communities, based on modularity, were identified for both Cascade and Kingston piers. At Cascade Pier, dusky sharks consisted of 100%, 78%, and 31% of sharks in the different communities (Fig. 6). At Kingston Pier, dusky sharks consisted of 91%, 42%, and 0% of sharks in the different communities (Fig. 6). Two communities (one at Cascade and one at Kingston), independent of species, were also distinguished by tagging date (i.e. individuals stopped being detected before other individuals were tagged; (Fig. S5). Modularity permutation tests indicated that the observed networks were non-random (PCascade = 0.018; PKingston < 0.001).
The activity spaces of dusky and Galapagos sharks during hourly periods when both species were detected were similar, with Cascade and the adjacent Simon site being the primary areas used (i.e. UD50%) by individuals released at Cascade Pier, and Kingston and the adjacent Surf and Nepean sites being the primary areas used by individuals released at Kingston Pier (Fig. S6). Nevertheless, dusky sharks (UD50/95%: 0.93/8.61 km2 [Cascade Pier]; 1.01/8.90 km2 [Kingston Pier]) typically used more locations than Galapagos sharks (UD50/95%: 0.62/4.12 km2 [Cascade Pier]; 0.79/5.11 km2 [Kingston Pier], Fig. S6).
Density-dependent relationships
The models testing whether monthly residency (the monthly number of days an individual was detected at a pier) was influenced by the monthly number of sharks present or proportion of dusky sharks present indicated that there was an interaction between number of sharks and species at Cascade Pier, with Galapagos sharks being more resident when the total number of sharks increased (P = 0.032, slope 0.759; (Fig. S7). At Kingston Pier, the relationship between number of days present each month and species differed (P = 0.001, slope = 5.75); specifically, Galapagos sharks were more resident than dusky sharks (Fig. S7). The proportion of dusky sharks when multiple individuals co-occurred (in hourly periods) at Cascade Pier was positively associated with the number of co-occurring individuals within that aggregation indicating that Galapagos sharks were less likely to be detected as the size of aggregations at the piers increased (P = 0.036, slope = 0.085; Fig. S8). This relationship was not significant at Kingston Pier (P = 0.168, slope = 0.024).
Movements of sharks between piers
Out of 11,680 (dusky shark; n = 18 individuals) and 13,991 (Galapagos shark; n = 16 individuals) detections (when both sites were active), there were 104 (dusky shark; n = 8 individuals) and 73 (Galapagos shark; n = 7 individuals) instances of movements between sites, with 75 (dusky shark; n = 7 individuals) and 60 (Galapagos shark; n = 6 individuals) instances occurring within a period shorter than 7 days and none during a period of less than four hours (Fig. S9). Variation existed in the number of cross-pier movements made by each individual with two dusky sharks (Male – 2.15 m TL; Female – 2.30 m TL) making > 28 one-way movements between sites, and one Galapagos shark (Female – 1.52 m TL) making 35 movements (Fig. S10). For sharks that were detected at both sites, they were more often detected at their site of release with dusky sharks released at Cascade and Kingston piers detected at these sites ~ 69% and ~ 37% of all detections (across all receivers), and Galapagos sharks released at Kingston and Cascade piers detected at these sites ~ 67% and ~ 53% of all detections, respectively (Fig. S11).
Diet of dusky and Galapagos sharks
A total of 21 muscle, 19 plasma, and 21 red blood cell samples from dusky (1.80 TL ± 0.06 m) and Galapagos sharks (1.71 TL ± 0.08 m) were analysed for fatty acids and δ13C and δ15N (Table 4). An insufficient number of males were sampled across species to include sex as a factor. Of the 67 different fatty acids detected within the muscle, plasma, or red blood cells, 20 contributed 0.3% or greater (Table S3). Neither fatty acid or stable isotope profiles differed between species across all three tissue types (all p(MC) > 0.05) (Table 5).
Dietary niche
Dispersion in fatty acid and isotopic spaces for each tissue type did not differ between species (all PERMDISP p > 0.05, Table 6). Standard ellipse areas representing niche size revealed minor and inconsistent differences between the niches of the two species (Table 6). Dusky shark muscle and plasma fatty acid and isotope niches were larger than those of Galapagos sharks, while Galapagos sharks had comparatively larger niches from red blood cells (Table 6; Fig. 7). Species’ proportional niche overlap was consistently ~ 0.5 for all fatty acid niches and muscle isotope niches, but was marginally higher for plasma isotope niches at 0.69 (Table 6). Red blood cell isotope niches only overlapped 0.27, driven by two Galapagos sharks with lower δ13C values, and two dusky sharks with low δ15N values relative to other individuals (Fig. 7F). These four samples could not be identified as true outliers due to the small sample size, thus were not removed from the analysis. While the proportional calculated niche overlap averaged 0.53, the proportion of samples that fell inside the shared niche space averaged 0.94.
Diet reconstruction
The Bayesian mixing model indicated dusky and Galapagos sharks fed primarily on Nannygai (Centroberyx affinis) (group B) followed by amberjack (Seriola dumerili) and yellowfin tuna (group C) (Fig. 8). Species often discarded at the piers, including redthroat emperor (Lethrinus miniatus) and yellowtail kingfish, contributed the lowest proportion, averaging 12.5%. The proportional contribution of both the discard spp. and group B were highly variable (Fig. 8), indicating substantial individual variation in diet. The muscle fatty acid profiles discernibly shifted with increasing days detected at the piers (CAP p = 0.013, Fig. 9), with sharks detected for > 50 days having comparatively higher levels of 20:5n3 (EPA) and 22:6n3 (DHA) potentially from consuming the discarded fish skin and liver (Fig. 9). The mean residency index (number of days individuals detected at the piers relative to the number of days between release and last detection) of these four individuals was 0.25 (i.e. detected at piers on 25% of days). Additionally, the individual with the largest CAP discrimination (i.e. detected for 266 days) made the most movements between piers (35 one-way movements).
Discussion
Offshore islands and seamounts are often productive areas with high levels of endemism and biodiversity29,30. The remote nature of these bathymetric structures also limits human disturbances — one of the main factors affecting shark populations31,32. As such, understanding how humans alter the behaviour of aquatic animals at remote sites, even at small scales, is integral for resource management under global changes. The incidental feeding of dusky and Galapagos sharks at Norfolk Island is an example of small-scale provisioning with only a few operators fishing for subsistence or commercial purposes. Still, fishing excursions occur regularly throughout the year and are only limited by unsuitable weather conditions (e.g. strong winds) due to the use of small boats and lack of a protected harbour. Catches, consisting mostly of redthroat emperor28,33, are substantial, often taking hours for fish to be cleaned from early afternoon through to dark. This results in a reliable and somewhat predictable source of food for sharks — fish frames. Recording boating and fish cleaning activity was beyond the scope of this study, limiting our ability to directly match shark behaviour to incidental feeding events. Nevertheless, sharks at Cascade and Kingston piers, often numbering more than 20 individuals at a time (M. Scott pers. obs.), are readily observed during fish cleaning. Therefore, it is highly likely that human activity has shaped the space use and foraging activities of a subset of dusky and Galapagos sharks around Norfolk Island either directly (e.g. feeding on scraps) or indirectly (e.g. anticipatory behaviour or foraging on other species also attracted to the feeding). Still, it appears that dusky and Galapagos sharks only use the area intermittently during opportunistic foraging excursions as part of their home range and are not exclusively reliant on discards. Overall, this study provides new information about how two closely related shark species with similar diet and movements partition use of feeding sites at different times of day.
Both dusky and Galapagos sharks were commonly detected at the piers, and often more frequently than any other receiver site, highlighting the relative importance of these areas. Indeed, after accounting for the receiver that was lost at Cascade Pier, every individual that was detected following tagging visited the piers, except for two. Cascade and Kingston piers are small, relatively enclosed areas without unique habitat compared to other sites studied. Thus, sharks appear to regularly visit these areas, the only sites on the island where fish are cleaned, for human-derived food through incidental feeding. Nevertheless, these areas were not used every day by individuals and when they were it was only for a few hours, corroborating the overall low dietary contribution of fish discards and suggesting the piers are only ephemeral scavenging areas. Furthermore, rates of co-occurrence were often low and many individuals of both species were never detected at the piers following tagging, suggesting that the area only consistently attracts a subset of both populations. Diel differences between dusky and Galapagos sharks were pronounced with dusky sharks consistently visiting the piers a few hours earlier than Galapagos sharks. Yet, these temporal differences in occurrence at the piers did not result in detectable dietary differences between species, further supporting limited contribution of discards to the diet given that dusky sharks associated more strongly with periods when scraps were discarded (i.e. afternoon/dusk) compared to Galapagos sharks.
Diel use of the piers by dusky sharks varied considerably by individual, which was not surprising given the highly mobile nature of dusky sharks34,35,36. At both sites, the highest probability of occurrence for dusky sharks was in the afternoon. Diel spatial patterns of dusky sharks appear to be variable across pelagic continental shelf regions; for example, individuals spent more time at the surface during dawn compared to dusk in South Australia37 and no differences were observed in time spent at surface between day and night in the Gulf of Mexico38,39. A dusky shark tracked at the Revillagigedo Archipelago, a group of four volcanic islands > 350 km offshore from Mexico, showed a high propensity for diurnal activity near one of the islands, rarely being detected after 18:0040. We observed a similar diel shift in space use with detections at Cascade Pier decreasing during the night as dusky sharks moved to the deeper site at Simons. Given the propensity for fishers to return and clean their catch from midday until the end of day, the use of Cascade and Kingston piers aligns with availability of fishing discards.
Diel differences in Galapagos shark occurrence were consistent across piers with an increasing likelihood of presence as the day progressed and peaking at night. Diel patterns of Galapagos sharks have been associated with shifts in habitat and depth use, but the specific patterns often vary41,42,43. Similar to this study (although across different life history stages), Galapagos sharks that were acoustically tracked throughout the Eastern Tropical Pacific, mainly at the Revillagigedo Archipelago, were more commonly detected during the night compared to the day — receiver locations, however, differed as they were not within embayments but adjacent to steep bathymetric features44. Furthermore, at Lord Howe Island, Australia, Galapagos sharks often occupied shallower depths at night compared to daytime indicative of diel vertical migration45. Behavioural modification of Galapagos sharks in response to fish cleaning activity was also identified at Lord Howe, with higher residency and smaller activity spaces around cleaning sites45. Presumably, the use of Cascade and Kingston piers at night was foraging-related as part of nocturnal activity (e.g. scavenging for left-over fish discards or foraging on other organisms attracted to the discards). The Nepean receiver site, located southeast of Kingston Pier, also had high relative occurrences that peaked at night, which may be another common foraging site due to the diverse habitat and strong currents that pass between Norfolk and Nepean Island46 (C. Huveneers pers. obs.).
Despite temporal partitioning, the lack of detectable differences in the diet of these two species suggests similar natural diets or equal scavenging opportunities. Globally, both species are primarily piscivores, consuming carangids, scombroids, and clupeids25,49,50. The mixing models revealed that at Norfolk Island, the sharks mostly consume reef fish such as nannygai Centroberyx sp., followed by offshore tuna. While the teleost species commonly discarded at the piers (redthroat emperor and yellowtail kingfish) contributed minimally to both species’ diets, we cannot assess whether these prey sources are distinct from the natural diet of the population. The scraps discarded at the piers may be the same species or trophic groups as these sharks consume naturally, preventing the detection of provisioning-induced differences in diet between species, or at the population level. Furthermore, Norfolk Island fishers report frequent depredation by sharks (S. Greenwood pers. com.), such that some proportion of the contribution of redthroat emperor and yellowtail kingfish may originate from depredation instead of consuming fish scraps at the piers. Although the contribution of discards to the population could not be fully assessed, the fatty acid profiles of individuals suggest that discarded fish contribute measurably to the diet of certain individuals. The shift in fatty acid profiles with increasing residency at the piers, in conjunction with the high variability in the diet contribution of discarded species (up to 65%), suggest resident individuals capitalize on this nutritionally distinct food source and have learnt to specialise on foraging at the piers opportunistically. Individuals frequently detected at the piers contained more essential fatty acids (EFAs) 20:5n3 and 22:6n3, which occur in higher percentages in the discarded liver and skin of many fishes compared to muscle (e.g. southern bluefin tuna Thunnus maccoyii [Henkens et al., unpublished data] and madeiran sardinella Sardinella maderensis51. Essential fatty acids 20:5n3 and 22:6n3 are also key micronutrients supporting and indicating vertebrate health52,53. Their comparatively high levels in the sharks frequenting the piers, owing to supplementing natural feeding with lipid- and EFA-rich discards, likely benefits the growth, fecundity, and physiological function of the pier-associated individuals53,54,55.
The relatively high use of piers by both species, in concert with indistinguishable natural diets, raises the topic of niche partitioning at Norfolk Island. Niche partitioning constitutes the segregation of resources in space or time enabling the coexistence among species that would otherwise compete over the same resources47. Several findings in this study suggest the possibility of spatiotemporal niche partitioning between dusky and Galapagos sharks, with the most compelling being the difference in diel patterns. Dusky sharks were associated with periods when fish cleaning was more frequent (i.e. afternoon to dusk), while Galapagos sharks increasingly visited the piers primarily from dusk onwards. This shift in use of the piers may have resulted from competition or dominance since dusky sharks were, on average, 0.62 m bigger than Galapagos sharks. Dusky sharks have similarly been observed co-existing with sandbar sharks in shallow waters of the Mediterranean Sea with diel shifts to preferred habitat suggested to be mediated by competition in favour of the larger dusky sharks48. The size of individuals may have also led to preferential within-species use of the piers, as demonstrated by the higher occurrence of larger dusky (i.e. Kingston Pier) and Galapagos (i.e. Cascade and Kingston piers) sharks during the afternoon. The sharks in this study were likely all immature (except one male dusky shark — ID5643923,25), which may explain the limited strength of size effects on presence. We also found preliminary evidence for density-dependent competitive pressures, supporting dusky sharks as being the more dominant species, at least at Cascade Pier where an increase in co-occurring individuals was associated with a higher proportion of dusky sharks. Without sampling the entire populations at the piers and noting that a species-specific diel shift in use of the piers may mirror density-dependent interactions, it is not known if these patterns are more broadly representative; nevertheless, density-dependent competition, both within and between species, can shape spatial separation of sharks56,57.
Spatiotemporal niche partitioning, resulting from competition or other means, was not inflexible. Co-occurrence networks showed that individuals often overlapped in the use of the piers independent of species. Similarly, activity spaces of both species, composed of co-occurring hourly detections, strongly overlapped, with Cascade and Kingston piers being primary sites used by both species. Furthermore, dusky and Galapagos sharks are one of the few shark species that hybridise in different parts of the world including Norfolk Island58 (C. Huveneers pers. obs.). Therefore, while co-occurrences within species are expected to be more common (e.g. based on species-specific diel preferences and SRI values), segregation between species was not absolute and differences in diel use of piers may have instead been a result of inherent population-level diel behaviour (e.g. nocturnal behaviour of Galapagos sharks) as opposed to direct competition or avoidance. While co-occurrence across all species combinations were low (mean hourly SRI < 0.1), our observed frequency of dyad co-occurrence was typically greater than expected suggesting that SRI values still represented a net positive affinity for dyads to co-occur even between species. Furthermore, detectability at the piers was expected to be low given the restricted area and density of sharks using it (e.g. higher rate of signal collisions). Likewise, we did not incorporate environmental variability (e.g. strong winds reducing fishing activity) because of the expected impact on detectability at the piers. Whether diel differences in space use between species has been initiated by exclusionary processes over generations or independently through distinct, but malleable, diel activity modes is not known. In other regions of the world, both species appear not to be strictly confined to specific diel periods of foraging activity and sharks have been shown to alter natural foraging behaviour in response to peak periods of direct feeding (e.g.9). Thus, it is reasonable to conclude that it is not beneficial for Galapagos sharks to forage concomitantly with dusky sharks at the piers during peak periods of fish cleaning, either due to sub-optimal foraging conditions (i.e. density-dependence or competition) or risks to health (i.e. aggression). Despite several sharks intermittently using both piers, the lack of short-term (< 4 h) movements between them suggests that exploiting both piers during the same day is not energetically profitable and occurs across greater temporal periods in relation to factors such as foraging conditions, home range plasticity, environmental variability, and demographic patterns.
Conclusion
A main concern of wildlife provisioning is that it can alter natural paths of resource utilisation, which in turn can impact individual, population, or ecosystem health59,60. Associating with fishing vessels or specific locations, or changing behaviour in response to provisioning is commonly reported for sharks9,61,62. Several studies have shown marginal effects of wildlife tourism affecting natural behaviours or fitness14,20,63,64, while others have raised potential concerns associated with behavioural modification negatively impacting competitive interactions13, energy use65, residency patterns5, and food uptake66. It is evident that behavioural modification has occurred for a subpopulation of sharks at Norfolk Island. Nevertheless, given the history of cleaning fish at the piers and the periodic-only use of the piers across all individuals tracked — notwithstanding the individuals that were never detected following tagging — incidental feeding at its current level appears to only supplement natural diet and not replace it. Whether the incidental feeding has resulted in shifts that affect the ecosystem more broadly (e.g. altering energy pathways, reducing competition at other sites, shifting prey species’ composition near the piers) cannot be commented on without ongoing research in combination with monitored restrictions on discarding activities.
From a management perspective, there are few alternate options to dispose of fish discards at the piers without removing that source of energy from the ocean altogether (e.g. composting). Furthermore, the activity adds to the tourist experience, which has socio-economic importance on an island where tourism is the main industry. Human safety also does not appear to be affected, at least while incidental feeding has been ongoing; no incidents have been reported despite a surf break being located only a few hundred meters away from Kingston Pier, near a receiver site that regularly detects both species. Still, while the relatively small-scale practice of discarding fish scraps at the piers appears to be innocuous, it is important for ongoing monitoring of the interactions between sharks and fish cleaning activities to ensure that associations are maintained at relatively low levels and local management has the capacity to change regulations as needed.
Methods
Study area
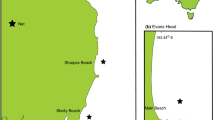
Norfolk Island (29°02’05.0"S, 167°57’23.7"E; Fig. 1) is a small (~ 35 km2) remote island in the southwestern Pacific Ocean. Maximum depths within a few kilometres from land reach ~ 50 m with areas directly adjacent to land typically < 20 m. There is a unique mix of temperate and tropical fish and invertebrate species, including several different species of demersal and pelagic sharks67. Norfolk Island is a designated Important Shark and Ray Area (ISRA) due to its role supporting dusky, Galapagos, and tiger (Galeocerdo cuvier) sharks68. Fishing is a common activity throughout the year with redthroat emperor being the most commonly targeted and consumed species. Less common catch includes rock cod (Epinephelus spp.), trevally (Pseudocaranx spp.), yellowtail kingfish (Seriola lalandi), yellowfin tuna (Thunnus albacares), and other mesopredatory fish28,33. From 2006 to 2009, the average monthly catch of fish by weight at Norfolk Island was estimated to be 847 kg, with no seasonal patterns observed in catch rates or fishing activity28.
Cascade and Kingston piers (Fig. 1), constructed with steel piles, rubble, and concrete, are the only two locations to launch and retrieve boats (using a crane) due the exposed nature of Norfolk Island to waves and swell. Fish cleaning takes place at these locations using wooden tables. At Kingston, the water is only accessible on the northern side of the pier (there is exposed limestone reef on the south side) where it is mostly enclosed by shallow waters (< 5 m depth) and high-relief rock formations that are exposed at low-tide. Habitat at Kingston Pier consists mainly of sand and hardbottom/rock partly covered in coral and macroalgae69. A mix of sand and rock boulders make up the main habitat around the pier at Cascade with adjacent waters < 10 m deep. Strong surge exists at both sites depending on prevailing winds and swell. Secondary study locations (i.e., receiver sites away from the piers — see below) consist of a mix of hardbottom, rock, coral, and unconsolidated gravel and sand69 in deeper waters (~ 15–20 m deep).
Receiver array
Acoustic receivers (VR2Ws, innovasea.com) were deployed at Kingston and Cascade Piers in February 2021 (Fig. 1). Receivers were affixed to square wooden pier pylons directly next to sections of the pier where fishing boats launch/retrieve and discard waste. They were strategically placed on the inner margin of the pylons to restrict detections further from shore. An array comprising an additional 14 receivers were deployed around Norfolk Island (Fig. 1) across the same period, with annual servicing and data downloads between February and April. The last receiver download for this study was in April 2024. Wave action from a significant weather event damaged the pylon at Cascade in March 2024, resulting in the receiver being lost (no detection history beyond March 2023 was available).
Shark tagging and tissue collection
Between February 2021–2023, 34 dusky and 32 Galapagos sharks were caught and tagged at Kingston (11 dusky/17 Galapagos) and Cascade Piers (20 dusky/13 Galapagos). Sharks were caught at the piers via a baited handline consisting of an 8–10 mm polypropylene/polyethylene blend rope and circle hooks (size 13/0–16/0), before being landed on the lower steps of the pier for tagging. Once landed and secured, sharks were inverted to initiate tonic immobility70, and a ~ 4 cm incision was made where each shark was internally tagged with a V16 acoustic transmitter (Innovasea; random interval of 80–140 s; 3,650 days expected battery life). Incisions were sealed using Gauge 2 Polyglycolic Acid absorbable sutures (silverglide.com.au). Following acoustic tagging, individuals were also tagged in the musculature at the base of the first dorsal fin with an external identification tag (hallprint.com) to avoid double-tagging if recaptured. Sharks were measured and sex was recorded when possible.
In February 2021, muscle samples (∼2 g) were collected from the ventral musculature along the margin of the anal fin using an 8 mm biopsy punch. Whole blood samples (∼4 ml) were collected from the caudal vein using a heparin-lined 5 ml syringe with hypodermic needle (1.6 × 40 mm), placed into a 10 ml lithium heparin vial and placed in a cooler. Whole blood was spun within 3 h of collection to separate into red blood cells and plasma components using a portable centrifuge at 3000 rpm for 3 min. Red blood cells and plasma layers were pipetted into separate 2 ml tubes and frozen at − 4℃ immediately until samples could be transported to the laboratory. Muscle samples (~ 5 g) were also collected from nine teleost species caught by fishers around Norfolk Island (Table S1). Sample collection occurred at both Kingston and Cascade piers, and the tissue was taken from along the vertebrae following filleting. Teleost samples were also frozen at − 4℃ until they could be transported to the laboratory alongside the shark samples. All samples were then stored at −20℃ until freeze-drying using a Christ Beta 2–8 freeze dryer at −96℃ at 0.0015 atm for 72 h. Muscle samples were then ground into a fine powder with a mortar and pestle for subsequent fatty acid and stable isotope analysis.
Activities involving animal sampling were approved under the Australian Government’s ‘Environment Protection and Biodiversity Conservation Regulations 2000’ and ‘Access to Biological Resources in a Commonwealth Area for Non-Commercial Purposes’ issued to Dr Lauren Meyer (Flinders University) under Permit number AU-COM2021-503. All work conducted with animals was approved by James Cook University Animals Ethics Committee (A2864) and adhered to institutional animal care and use guidelines and regulations. Authors also complied with ARRIVE guidelines.
Biochemical analysis
Fatty acids
Approximately 0.025 g of dried sample was directly transmethylated following Parrish et al.71 detailed in Burke et al.72. Fatty acid methyl esters (FAMEs) were isolated and extracted from samples by soaking in 10:1:1 methanol: dichloromethane: hydrochloric acid solution at 80℃ for 2 h, cooled, and then the solution separated into two layers by adding 1 mL of Milli-Q water and 1.8 mL of a 4:1 hexane: dichloromethane solution and centrifuged. The top layer containing the FAMEs was transferred to a 2 mL vial for identification and quantification using gas chromatography mass spectrophotometry (GCMS) analysis. A blank and an internal standard (C4-C24, Sigma-Aldrich, USA) was added for identification of FAMEs at both the start and end of each run of 60 samples. Individual sample chromatograms were analysed and processed using MassHunter Qualitative Analysis software and the NIST database of fatty acid compounds. Individual fatty acid values were reported as a percentage of the total fatty acids per sample. Fatty acids contributing < 0.1% across all samples were removed, leaving 29 fatty acids for subsequent statistical analyses.
Stable isotope analysis
Lipids and urea were removed from shark and prey samples following a modified Bligh et al.73 protocol, where samples were soaked in 10 mL of 2:1 dichloromethane: methanol solution for 24 h, filtered through 8 μm filter paper, scraped into a 5 mL Eppendorf tube and dried at 60 °C for 48 h. Approximately 1.50 mg (± 10%) of dried tissue was weighed into a 8 × 5 mm tin capsule for quantification. Samples were analysed for δ13C and δ15N using Isoprime GC5 continuous flow Isotope Ratio Mass Spectrometer with vario ISOTOPE cube elemental analyser (Elementar Australia Pty Ltd) at Flinders University. Five blank tin capsules and seven of each standard (L-glutamic acid USGS40 [δ13C = −26.39‰, δ15N = −4.52‰], marine collagen peptides [δ13C = −20.80‰, δ15N = + 5.55‰], and enriched caffeine [δ13C = −25.90‰, δ15N = + 80.70‰] from Sercon Ltd, UK). were run throughout each batch of 60 samples to measure analytical precision (δ13C = 0.18, δ15N = 0.07) and correct for isotopic drift.
Stable isotope results are reported in conventional delta notation as.
\(\delta {\text{X}}=[{\text{Rsample}}/{\text{Rstandard}}\, - \,{\text{1}}] * {\text{1}}000\delta {\text{X}}\,=\,{\text{Rsample}}/{\text{Rstandard}}\, - \,{\text{1}} * {\text{1}}000\)
where X represents 15N or 13C, and R is the ratio of 15N/14N or 13C/12C in the sample, following the international standards of Pee Dee Belemnite for CO2, and atmospheric nitrogen for N2.
Data analysis
Detection data filtering
Analysis of movement data was carried in the R Foundation for Statistical Computing Platform (version 4.3.0). For all analyses, only detections following a 48-hour period post-tagging were considered to reduce the likelihood of including abnormal behaviour resulting from capture and tagging. Additionally, individuals that were only detected at either Cascade or Kingston piers for a period < 15 days (i.e. during the period of 48 h after tagging to the last day detected) were noted, but removed from analyses because they were not informative to the objectives of this study. False detections, specifically false-positive detections caused by signal interference or ghost detections74, were investigated by identifying any consecutive detections (of the same transmitter) equating to travel speeds > 5 ms-1 75 after allowing a 15-min buffer for receiver-clock disparities between receivers. We did not set a cutoff for minimum hourly or daily number of detections because false-positive detections were expected to be very low (e.g. stationary sync tags integrated within some receivers were never detected outside their detection range over the course of a year) and because we expected the ability to detect transmitters at the piers to be relatively low (e.g. shallow, restricted horizontal range, high surge activity), and did not want to risk removing a true detection. Finally, detections were explored to ensure that they represented living animals, as opposed to shed tags or dead animals by removing any animal that had consistent multi-month detections at singular receiver sites75.
Diel presence at piers
We were first interested in investigating the relative occurrence of dusky and Galapagos sharks at Cascade and Kingston piers during different diel periods. We only included days when an individual was present at either pier since days of complete absence were not informative for this specific analysis. Diel categories were divided into the following categories using the suncalc R package77: dawn (end of night to end of sunrise), morning (end of sunrise to solar noon), afternoon (solar noon to golden hour), dusk (golden hour to dusk), and night (dusk to end night). Dawn was afterwards combined with the morning category because there were few detections during the dawn period (i.e. <5%), which hindered model performance. An individual was considered present within a diel category on days when it was detected at least once (during that diel category). Comparisons of presence across diel categories was investigated using a generalised linear mixed effects model approach with a binomial distribution (lme4 R package78. First, we tested for differences in presence across diel categories, pier location (i.e. Cascade vs. Kingston), and species (as an interaction) to determine if separate analyses were warranted per pier and species. This global model (and all others described below) was evaluated using model selection from the MuMIn R package79 with AICc values < 2 used to inform the most parsimonious models. Additionally, marginal and conditional R2 values (from top performing models) were used to evaluate how well fixed effects (i.e. marginal R2) and both random and fixed effects (conditional R2) explained presence. Differences in presence across diel periods were tested separately for each pier location and species following the analysis described above (i.e. differences between location and species within the global model). This approach also simplified interpretation of presence at each site (e.g. as opposed to multiple interactions), as well as accounted for the different receiver deployment periods between Cascade and Kingston piers. We also included total length (TL, m) as a continuous factor (and its interaction with diel category) to explore size-related behavioural changes across diel periods. In all models, animal ID was selected as a random effect and each diel period was nested within each respective date. The relative use of other receiver sites around Norfolk Island was also explored across the different diel periods to evaluate additional site preferences.
Co-occurrence of dusky and Galapagos sharks
A social network approach was used to quantify the frequency of co-occurrences among species and individuals at the piers. Here, co-occurrence was defined as an hourly period (on the same day) when a dyad (i.e. two individuals present at Norfolk Island at the same time) was detected at either Cascade or Kingston piers. Adjacency matrices were created for Cascade and Kingston piers separately using the simple ratio index (SRI) calculated with the Asnipe R package80. Here, SRI was the number of hourly periods during overlapping detection periods in which both individuals of a dyad were present at a pier relative to the number of hourly periods at least one individual was present. The SRI represents the strength of association between two individuals in a dyad. The modularity of each site’s network was calculated to measure the strength of divisions of a network (i.e. high modularity ~ dense connections between nodes within modules and sparse connections between modules; low modularity ~ no modular structure). We used the ‘fast greedy’ modularity-optimization method81 to identify different communities within the network, independent of species. A randomised swap approach was used to test whether the observed modularity differed from a random assortment of networks, whereby co-occurrences were deemed non-random (i.e. p < 0.025) when the modularity of the observed network was greater than 97.5% of permuted networks82. We evaluated whether SRI values were greater or less than expected for each species combination by comparing the difference between observed probability of co-occurrence (i.e. number of hours co-occurring at piers relative to number of shared days monitored) and expected probability of co-occurrence (i.e. product of each species’ number of hours detected at the piers relative to number of shared days monitored). Observed values greater than expected would suggest that SRI values (independent of their absolute value) represent a positive affinity for dyads to co-occur.
Additionally, to assess spatial variation in co-occurrence patterns between species, activity spaces of each species were calculated during hourly periods when both species were detected at any site around the island. The goal of this approach was to determine if species used different areas during overlapping periods of known location. Activity spaces were quantified using kernel density estimation within the adehabitatHR R package83. Centres of activity (COAs84 at 1-hr intervals were used as individual locations, as opposed to raw detections, to help account for movements between receivers. Centres of activity that were estimated on-land were reassigned to the nearest shore and any land area was removed from activity space estimates. A smoothing parameter (h) of 300 was used to estimate 50% and 95% utilisation distributions (UDs) based on successive sensitivity explorations to match UD size estimates (at one receiver) with predicted detection ranges (50% detection efficiency ~ 250 m; J.K. Matley, pers. obs.).
Density-dependent relationships
We investigated density-dependent relationships based on the potential for antagonistic interactions or access to food to change as the number of sharks using the piers increased. It is important to note that not all individuals (of either species) that visit the piers were tagged, therefore, this investigation was limited to only the tagged population and may differ from population-wide relationships. Our first approach was to test whether monthly residency (i.e. number of days an individual was detected at a pier) was influenced by the monthly number of sharks or proportion of dusky sharks also detected at that pier during the same month. We used a linear mixed effects model (lme4 R package78 to test the interaction between species and number of sharks, as well as species and proportion of dusky sharks relative to residency, with ID set as a random variable; model terms with P-values < 0.05 were considered significantly different from slope = 0. We also investigated whether the proportion of dusky sharks in co-occurring aggregations (hourly) changed as the total number of individuals (in the aggregation) increased using a linear regression model. The number of dusky and Galapagos sharks (i.e. for the proportional dusky shark calculations), were standardised for comparison by dividing the number of each species by the total number detected that month. The overall goal of this approach was to test whether Galapagos sharks were less likely to be detected in dense aggregations of dusky sharks.
Movements of sharks between piers
Finally, the number of times that individuals made movements between piers was calculated to investigate whether individuals readily used both sites. For individuals that were detected at both sites, we also quantified the proportion of detections that occurred at each receiver site to elucidate site preferences relative to release site. Note that cross-pier movements were only summarised prior to February 2023 (when both Kingston and Cascade receivers were present).
Diet of dusky and Galapagos sharks
Multivariate analyses were carried out using PRIMER v.7/PERMANOVA + software85. Fatty acid and stable isotope profiles from all three shark tissue types (muscle, plasma, and red blood cells) were analysed separately given well-established physiological differences between tissues and the resulting differences in stable isotope and fatty acid profiles52. Different levels of transformation were explored via shade plots86, and for fatty acid profiles a square-root transformation was selected to avoid over-emphasis of extreme values. No transformations were applied to the stable isotope data. Resemblance matrices were calculated using Euclidean distance measures between samples. A permutational analysis of variance (PERMANOVA) with Monte Carlo simulations (denoted as p(MC)) on the unrestricted raw values was used to test whether stable isotope or fatty acid profiles differed by species. Canonical analysis of principal coordinates (CAP)87 was used to test if shark fatty acid profiles differed with time spent at piers (days detected at either Kingston or Cascade) as a continuous covariate.
Trophic niche space
A permutational analysis of multidimensional dispersion (PERMDISP) was performed on the multivariate resemblance matrix to calculate the dispersion among species as a measure of niche, and test for differences between species’ niche widths. To quantify and visualize niche space, standard ellipse areas containing 95% of the data were calculated for both dusky and Galapagos sharks using stable isotope and fatty acid data separately using the SIMMR package in R88. This package was selected due to its robustness to small sample sizes owing to a probabilistic method of calculating niche space89. Two-dimensional ellipses were calculated on the untransformed isotope data (δ13C and δ15N) to measure isotopic niche space. Fatty acid niche space was calculated following Roberts et al.90 whereby ordinations were extracted from the nMDS on Euclidean distances of square-root transformed fatty acid data. These ordinations were then used to create bivariate fatty acid ellipses.
Diet reconstruction
As muscle δ13C and δ15N did not differ between dusky and Galapagos sharks (p(MC) = 0.608), diet reconstruction using a Bayesian mixing model in SIMMR R package88 was performed on the two shark species combined. The δ13C and δ15N values of potential prey were taken from fish muscle collected from Norfolk Island in February 2021 (Table S1). To avoid exceeding the recommended maximum of prey sources91, prey items were grouped into four groups following isotopic value similarities. Samson fish were removed from the mixing model as they were deemed unlikely prey given their high δ15N values (Table S1). The “discard spp.” (Table S1) group included the commonly caught redthroat emperor and yellowtail kingfish, and the isotopically similar tomato cod (Epinephelus rivulatus), all of which had high δ15N values (e.g. ~ 15‰), and similar intermediate δ13C values (e.g. ~ -17‰). Wrasses and trevally were combined into “Group A” given their similar means and overlapping standard deviations, and were about half a trophic level below the “discard spp.” group. “Group B” was distinguished by intermediate δ13C and δ15N values and consisted only of nannygai as an indicator of this portion of the food web. The remaining two species of tuna both shared low δ15N and were grouped into “Group C”. The mixing model estimating diet from muscle tissue used an established shark- and tissue-specific trophic enrichment factor (TEF) (δ13C − 0.9‰, δ15N – 2.3‰)49. After correcting for TEFs (to compare shark isotope values with likely prey sources), four sharks fell outside expected values and were removed from the subsequent analysis because diet could not be reliably estimated. The convergence of the mixing model was evaluated using R-hat values, which equalled 1.
Study area at Norfolk Island, Australia with photo insets showing the receiver locations (yellow stars) at Cascade and Kingston piers. Black dots on map indicate receiver sites. All receivers were deployed between February 2021 and April 2024, except Chute and Fish Bowl (deployed in March 2023) and Cascade (receiver was lost between March 2023 and April 2024). Google Earth Pro (https://earth.google.com/web/) version 7.3.6.10201 (Image © 2025 Airbus) was used to create satellite imagery.
Detections of individual dusky and Galapagos sharks at Cascade (red) and Kingston (blue) piers. The ‘x’ symbol indicates the date of tagging and the circle and square symbols represent detections from individuals tagged at Cascade or Kingston, respectively. The vertical red line marks the last day of detections available at Cascade Pier before the receiver was lost. The vertical grey stripes highlight approximate periods when Norfolk Island experienced cyclones or other extreme weather events.
Predicted (+ 1 confidence interval) proportion of presence of dusky and Galapagos sharks at Cascade and Kingston piers across diel periods. Note that dusk is shorter and night is longer than other periods resulting in different times that detections can occur. Fit of best performing (full) model: Galapagos sharks (R2 conditional = 0.66 (Kingston) and 0.69 (Cascade); dusky sharks (R2 conditional = 0.39 (Kingston) and 0.47 (Cascade).
Proportional hourly occurrence of dusky and Galapagos sharks at Cascade and Kingston piers, calculated from the hourly proportion that each individual was present for each species as well as when both dusky and Galapagos sharks occurred during the same hour. Each species combination was scaled to 1 for comparison (i.e. not absolute estimates of presence).
Predicted (± 1 confidence interval) proportion of presence of dusky and Galapagos sharks at Cascade and Kingston piers across shark total lengths and diel periods. Fit of best performing (full) model: Galapagos sharks (R2 conditional = 0.66 (Kingston) and 0.69 (Cascade); dusky sharks (R2 conditional = 0.39 (Kingston) and 0.47 (Cascade).
Hourly co-occurrence networks of individual dusky (red) and Galapagos (blue) sharks. The width of edges represents the strength of associations between individuals (i.e. the number of co-occurrences) and the size of nodes is scaled to the detection period of each individual (number of days between first and last detection). Communities, based on fast-greedy modularity optimisation, are distinguished by the coloured polygons. Modularity at Cascade and Kingston piers was 0.52 and 0.4, respectively.
Bivariate plots of dusky (red) and Galapagos shark (blue) fatty acid niche space calculated using nMDS ordinations of fatty acids (left column), and isotopic niche space calculated using δ13C and δ15N values (right column) with probabilistic standard ellipse areas containing 95% of the data. Muscle (A, D), plasma (B, E), and red blood cells (C, F) plotted separately.
(A) Isotopic biplot of dusky (o) and Galapagos shark (+) muscle, with mean (± SD) prey species; muscle isotope values are corrected for comparison using trophic enrichment factors (mean (± SD) Δ15N of 2.3 ± 0.2‰ and Δ13C of 0.9 ± 0.3‰ from Hussey et al.49). (B) Estimated proportional contributions of each prey group to the diet of the dusky and Galapagos sharks (combined). Species commonly filleted at the piers, whereby frames and skin are discarded, are indicated as “discard spp.” in green.
Canonical Analysis of Principal Coordinates (CAP) of dusky and Galapagos shark muscle fatty acid profiles plotted against days detected at Norfolk Island piers (Cascade and Kingston combined). Inset – Principal Coordinate Analysis (PCO) of dusky and Galapagos shark muscle fatty acids with vector overlays showing fatty acids with correlation values > 0.8.
Data availability
Acoustic telemetry data is available at https://animaltracking.aodn.org.au/. Biochemical data is available at https://open.flinders.edu.au/articles/dataset/Resource_use_of_two_sympatric_sharks_in_response_to_fishing_activities_at_a_remote_island_-_fatty_acid_and_stable_isotope_data/28693385?file=53314109.
Change history
18 October 2025
The original online version of this Article was revised: The Acknowledgements section in the original version of this Article was incomplete. It now reads: We thank Mitch Graham, Luke Fitzpartrick, Dean Fitzpatrick, Philip Reid, Frances Murray, Russell Gueho, Jens Neiser, Elizabeth Dinsdale, Michael Doane, Lisa Hoopes, Ryan Hesse, Emma Kerr, Natalie Klinard, Laura Ryan, Olivia Seeger, and Suki for their help with fieldwork and logistics. Funding and support was provided by the Australian Government through the Our Marine Parks Grants Program (4-HANQ6UL), Neiser Foundation, Georgia Aquarium, Biopixel Oceans Foundation, Big Wave TV, and Norfolk Island Regional Council. Data were sourced from Australia’s Integrated Marine Observing System (IMOS) (animaltracking.aodn.org.au) – IMOS is enabled by the National Collaborative Research Infrastructure Strategy (NCRIS). Additional equipment and support were also provided by the IMOS Animal Tracking Facility.
References
Mitchell, J. D. et al. Shark depredation: future directions in research and management. Rev. Fish. Biol. Fish. 33, 475–499 (2023).
Pini-Fitzsimmons, J., Knott, N. A. & Brown, C. Effects of food provisioning on site use in the short-tail Stingray Bathytoshia brevicaudata. Mar. Ecol. Prog Ser. 600, 99–110 (2018).
Svane, I., Roberts, S. & Saunders, T. Fate and consumption of discarded by-catch in the Spencer Gulf Prawn fishery, South Australia. Fish. Res. 90, 158–169 (2008).
Bicknell, A. W., Oro, D., Camphuysen, K. & Votier, S. C. Potential consequences of discard reform for seabird communities. J. Appl. Ecol. 50, 649–658 (2013).
Mourier, J., Claudet, J. & Planes, S. Human-induced shifts in habitat use and behaviour of a marine predator: the effects of bait provisioning in the Blacktip reef shark. Anim. Conserv. 24, 230–238 (2021).
Pini-Fitzsimmons, J., Knott, N. A. & Brown, C. Recreational fishery discard practices influence use of tidal estuary by a large marine mesopredator. Mar. Freshw. Res. 74, 320–334 (2023).
Semeniuk, C. A. & Rothley, K. D. Costs of group-living for a normally solitary forager: effects of provisioning tourism on Southern stingrays Dasyatis Americana. Mar. Ecol. Prog Ser. 357, 271–282 (2008).
Corcoran, M. J. et al. Supplemental feeding for ecotourism reverses diel activity and alters movement patterns and Spatial distribution of the Southern stingray, Dasyatis Americana. PLoS ONE. 8, e59235 (2013).
Fitzpatrick, R., Abrantes, K. G., Seymour, J. & Barnett, A. Variation in depth of whitetip reef sharks: does provisioning ecotourism change their behaviour? Coral Reefs. 30, 569–577 (2011).
Barnett, A., Payne, N. L., Semmens, J. M. & Fitzpatrick, R. Ecotourism increases the field metabolic rate of whitetip reef sharks. Biol. Conserv. 199, 132–136 (2016).
Meyer, C. G., Dale, J. J., Papastamatiou, Y. P., Whitney, N. M. & Holland, K. N. Seasonal cycles and long-term trends in abundance and species composition of sharks associated with cage diving ecotourism activities in Hawaii. Environ. Conserv. 36, 104–111 (2009).
Clarke, C. R., Lea, J. S. E. & Ormond, R. F. Changing relative abundance and behaviour of silky and grey reef sharks baited over 12 years on a red sea reef. Mar. Freshw. Res. 64, 909–919 (2013).
Brunnschweiler, J. M., Abrantes, K. G. & Barnett, A. Long-term changes in species composition and relative abundances of sharks at a provisioning site. PLoS One. 9, e86682 (2014).
Heinrich, D. et al. Short-term impacts of daily feeding on the residency, distribution and energy expenditure of sharks. Anim. Behav. 172, 55–71 (2021).
Milazzo, M., Anastasi, I. & Willis, T. J. Recreational fish feeding affects coastal fish behavior and increases frequency of predation on damselfish Chromis chromis nests. Mar. Ecol. Prog Ser. 310, 165–172 (2006).
Clua, E., Buray, N., Legendre, P., Mourier, J. & Planes, S. Behavioural response of sicklefin lemon sharks Negaprion acutidens to underwater feeding for ecotourism purposes. Mar. Ecol. Prog Ser. 414, 257–266. https://doi.org/10.3354/meps08746 (2010).
Séguigne, C., Bègue, M., Meyer, C., Mourier, J. & Clua, É. Provisioning ecotourism does not increase tiger shark site fidelity. Sci. Rep. 13, 7785 (2023).
Abrantes, K. G., Brunnschweiler, J. M. & Barnett, A. You are what you eat: examining the effects of provisioning tourism on shark diets. Biol. Conserv. 224, 300–308 (2018).
Hammerschlag, N., Gutowsky, L. F. G., Gallagher, A. J., Matich, P. & Cooke, S. J. Diel habitat use patterns of a marine apex predator (tiger shark, Galeocerdo cuvier) at a high use area exposed to dive tourism. J. Exp. Mar. Biol. Ecol. 495, 24–34 (2017).
Meyer, L., Pethybridge, H., Beckmann, C., Bruce, B. & Huveneers, C. The impact of wildlife tourism on the foraging ecology and nutritional condition of an apex predator. Tourism Manage. 75, 206–215 (2019).
Gooden, A., Clarke, T. M., Meyer, L. & Huveneers, C. Wildlife tourism has little energetic impact on the world’s largest predatory shark. Anim. Behav. 207, 247–265 (2024).
Arlinghaus, R., Tillner, R. & Bork, M. Explaining participation rates in recreational fishing across industrialised countries. Fish. Manage. Ecol. 22, 45–55 (2015).
Matley, J. K. et al. Comparative space use of sympatric sharks at a small remote Island in the South Pacific ocean. Ecol. Evol. 15, e71534 (2025).
Geraghty, P. T., Macbeth, W. G. & Williamson, J. E. Aspects of the reproductive biology of dusky, spinner and sandbar sharks (Family Carcharhinidae) from the Tasman sea. Mar. Freshw. Res. 67, 513–525 (2015).
Wetherbee, B. M., Crow, G. L. & Lowe, C. G. Biology of the Galapagos shark, Carcharhinus galapagensis, in hawai’i. Environ. Biol. Fishes. 45, 299–310 (1996).
Newsome, D., Lewis, A. & Moncrieff, D. Impacts and risks associated with developing, but unsupervised, Stingray tourism at Hamelin bay, Western Australia. Int. J. Tourism Res. 6, 305–323 (2004).
Tofts, B. Shark Tales and Yarns from Norfolk Island (National Library of Australia, 1993).
AFMA (Australian Fisheries Management Authority). Norfolk Island Inshore Fishery Data Summary 2006–2009 (Australian Fisheries Management Authority, 2010).
de Richer, B., Koslow, J. A. & Poore, G. Diversity and endemism of the benthic seamount fauna in the Southwest Pacific. Nature 405, 944–947 (2000).
Williams, A., Althaus, F., Clark, M. R. & Gowlett-Holmes, K. Composition and distribution of deep-sea benthic invertebrate megafauna on the Lord Howe rise and Norfolk ridge, Southwest Pacific ocean. Deep Sea Res. Part. II. 58, 948–958 (2011).
Field, I. C., Meekan, M. G., Buckworth, R. C. & Bradshaw, C. J. Susceptibility of sharks, rays and chimaeras to global extinction. Adv. Mar. Biol. 56, 275–363 (2009).
Dulvy, N. K. et al. Challenges and priorities in shark and ray conservation. Curr. Biol. 27, R565–R572 (2017).
Anderson, A. & White, P. Approaching the prehistory of Norfolk Island. Records-Australian Museum. 53, 1–10 (2001).
Braccini, M., de Lestang, S. & McAuley, R. Dusky sharks (Carcharhinus obscurus) undertake large-scale migrations between tropical and temperate ecosystems. Can. J. Fish. Aquat. Sci. 75, 1525–1533 (2018).
Bartes, S. et al. Conventional tagging of sharks in Western australia: the main commercial species exhibit contrasting movement patterns. Mar. Freshw. Res. 72, 1643–1656 (2021).
Huveneers, C. et al. Continental-scale network reveals cross-jurisdictional movements of sympatric sharks with implications for assessment and management. Front. Mar. Sci. 8, 697175 (2021).
Rogers, P. J., Huveneers, C., Goldsworthy, S. D., Mitchell, J. G. & Seuront, L. Broad-scale movements and pelagic habitat of the Dusky shark Carcharhinus obscurus off Southern Australia determined using pop‐up satellite archival tags. Fish. Oceanogr. 22, 102–112 (2013).
Carlson, J. K. & Gulak, S. Habitat use and movement patterns of oceanic whitetip, Bigeye thresher and Dusky sharks based on archival satellite tags. Collect. Vol Sci. Pap ICCAT. 68, 1922–1932 (2012).
Hoffmayer, E. R. et al. Habitat, movements and environmental preferences of Dusky sharks, Carcharhinus obscurus, in the Northern Gulf of Mexico. Mar. Biol. 161, 911–924 (2014).
Klimley, A. P. et al. Studies of the movement ecology of sharks justify the existence and expansion of marine protected areas in the Eastern Pacific ocean. Environ. Biol. Fishes. 105, 2133–2153 (2022).
Lowe, C. G., Wetherbee, B. M. & Meyer, C. G. Using acoustic telemetry monitoring techniques to quantify movement patterns and site fidelity of sharks and giant trevally around French frigate shoals and Midway Atoll. Atoll Res. Bull (2006).
Papastamatiou, Y. P., Meyer, C. G., Kosaki, R. K., Wallsgrove, N. J. & Popp, B. N. Movements and foraging of predators associated with mesophotic coral reefs and their potential for linking ecological habitats. Mar. Ecol. Prog Ser. 521, 155–170 (2015).
Morales, N. A. et al. Residential movements of top predators in chile’s most isolated marine protected area: implications for the conservation of the Galapagos shark, Carcharhinus galapagensis, and the Yellowtail amberjack, Seriola Lalandi. Aquat. Conserv. Mar. Freshw. Ecosyst. 31, 340–355 (2021).
Lara-Lizardi, F. et al. Shark movements in the Revillagigedo Archipelago and connectivity with the Eastern Tropical Pacific. bioRxiv 2020-03 (2020).
Mitchell, J. D., Camilieri-Asch, V., Jaine, F. R., Peddemors, V. M. & Langlois, T. J. Galapagos shark movement patterns and interactions with fishing vessels in the marine parks surrounding Lord Howe Island. Final report to Parks Australia. Canberra (2021).
Pendoley, K. Commonwealth of Australia nearshore marine habitat mapping of the Norfolk Marine Park. Report to the Commonwealth of Australia (2022).
Chesson, P. Mechanisms of maintenance of species diversity. Annu. Rev. Ecol. Syst. 31, 343–366 (2000).
Barash, A. et al. Depth partitioning and diel movement of two large carcharhinid sharks in extremely shallow waters. Fishes 8, 85 (2023).
Hussey, N. E., Brush, J., McCarthy, I. D. & Fisk, A. T. δ15N and δ13C diet–tissue discrimination factors for large sharks under semi-controlled conditions. Comp. Biochem. Physiol. Mol. Integr. Physiol. 155, 445–453 (2010).
Papastamatiou, Y. P., Wetherbee, B. M., Lowe, C. G. & Crow, G. L. Distribution and diet of four species of carcharhinid shark in the Hawaiian islands: evidence for resource partitioning and competitive exclusion. Mar. Ecol. Prog Ser. 320, 239–251 (2006).
Njinkoué, J. M., Barnathan, G., Miralles, J., Gaydou, E. M. & Samb, A. Lipids and fatty acids in muscle, liver and skin of three edible fish from the Senegalese coast: Sardinella maderensis, Sardinella aurita and Cephalopholis taeniops. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 131, 395–402 (2002).
Munroe, S., Meyer, L. & Heithaus, M. Dietary biomarkers in shark foraging and movement ecology. Shark Res.: Emerg. Technol. Appl. Field Lab. 1–24 (2018).
Parrish, C. C. Essential fatty acids in aquatic food webs. Lipids Aquat. Ecosyst. 309–326 (2009).
de Rangel, S. et al. Neonatal nutritional strategy of a viviparous elasmobranch with extremely low reproductive output. Mar. Ecol. Prog Ser. 638, 107–121 (2020).
Meyer, L. et al. The use of muscle lipids and fatty acids to assess shark diet and condition. J. Fish. Biol. 98, 566–571 (2021).
Papastamatiou, Y. P. et al. Spatial separation without territoriality in shark communities. Oikos 127, 767–779 (2018).
Sabando, M. A., Rieucau, G., Bradley, D., Caselle, J. E. & Papastamatiou, Y. P. Habitat-specific inter and intraspecific behavioral interactions among reef sharks. Oecologia 193, 371–376 (2020).
Pazmiño, D. A. et al. Introgressive hybridisation between two widespread sharks in the East Pacific region. Mol. Phylogenet Evol. 136, 119–127 (2019).
Meyer, L., Whitmarsh, S. K., Nichols, P. D., Revill, A. T. & Huveneers, C. The effects of wildlife tourism provisioning on non-target species. Biol. Conserv. 241, 108317 (2020).
Shutt, J. D. & Lees, A. C. Killing with kindness: does widespread generalised provisioning of wildlife help or hinder biodiversity conservation efforts? Biol. Conserv. 261, 109295 (2021).
Martin, K. L., Abel, D. C., Crane, D. P., Hammerschlag, N. & Burge, E. J. Blacktip shark Carcharhinus limbatus presence at fishing piers in South carolina: association and environmental drivers. J. Fish. Biol. 94, 469–480 (2019).
Mitchell, J. D., McLean, D. L., Collin, S. P. & Langlois, T. J. Shark depredation in commercial and recreational fisheries. Rev. Fish. Biol. Fish. 28, 715–748 (2018).
Jacoby, D. M. et al. Social network analysis reveals the subtle impacts of tourist provisioning on the social behavior of a generalist marine apex predator. Front. Mar. Sci. 8, 665726 (2021).
Heinrich, D. D., Dhellemmes, F., Guttridge, T. L., Brown, C. & Huveneers, C. No evidence of time–place learning in juvenile lemon sharks, Negaprion brevirostris. Anim. Behav. 203, 75–88 (2023).
Huveneers, C., Watanabe, Y. Y., Payne, N. L. & Semmens, J. M. Interacting with wildlife tourism increases activity of white sharks. Conserv. Physiol. 6, coy019 (2018).
Heim, V., Dhellemmes, F., Smukall, M. J., Gruber, S. H. & Guttridge, T. L. Effects of food provisioning on the daily ration and dive site use of great hammerhead sharks, Sphyrna Mokarran. Front Mar. Sci 8 (2021).
Francis, M. P. Checklist of the coastal fishes of Lord howe, norfolk, and kermadec islands, Southwest Pacific ocean. Pac. Sci. 47, 136–170 (1993).
IUCN, Norfolk Island, I. S. R. A. & Factsheet IUCN SSC Shark Specialist Group, Dubai (2024).
Pendoley, K. Nearshore marine habitat mapping of the Norfolk marine park. Report to Commonwealth of Australia. J60002 (2022).
Kessel, S. T. & Hussey, N. E. Tonic immobility as an anaesthetic for elasmobranchs during surgical implantation procedures. Can. J. Fish. Aquat. Sci. 72, 1287–1291 (2015).
Parrish, C. C., Nichols, P. D., Pethybridge, H. & Young, J. W. Direct determination of fatty acids in fish tissues: quantifying top predator trophic connections. Oecologia 177, 85–95 (2015).
Burke, P. J., Meyer, L., Raoult, V., Huveneers, C. & Williamson, J. E. Multi-disciplinary approach identifies pelagic nutrient linkage by sawsharks. Rev. Fish. Biol. Fish. 34, 1567–1584 (2024).
Bligh, E. G. & Dyer, W. J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Phys. 37, 911–917 (1959).
Simpfendorfer, C. A. et al. R. Ghosts in the data: false detections in VEMCO pulse position modulation acoustic telemetry monitoring equipment. Anim. Biotelemetry. 3, 1–10 (2015).
Matley, J. K. et al. Where giants roam: the importance of remote Islands and seamount corridors to adult tiger sharks in the South Pacific ocean. Mar. Environ. Res. 206, 107026 (2025).
Klinard, N. V. & Matley, J. K. Living until proven dead: addressing mortality in acoustic telemetry research. Rev. Fish. Biol. Fish. 30, 485–499 (2020).
Thieurmel, B. & Elmarhraoui, A. Compute Sun Position, Sunlight Phases, Moon Position and Lunar Phase. R package version 0.5.1, (2022). https://CRAN.R-project.org/package=suncalc
Bates, D., Maechler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Soft. 67, 1–48 (2015).
Bartoń, K. & MuMIn Multi-Model Inference. R package version 1.47.5. (2023). https://CRAN.R-project.org/package=MuMIn
Farine, D. R. Animal social network inference and permutations for ecologists in R using asnipe. Methods Ecol. Evol. 4, 1187–1194 (2013).
Clauset, A., Newman, M. E. & Moore, C. Finding community structure in very large networks. Phys. Rev. E. 70, 066111 (2004).
Findlay, R., Gennari, E., Cantor, M. & Tittensor, D. P. How solitary are white sharks: social interactions or just Spatial proximity? Behav. Ecol. Sociobiol. 70, 1735–1744 (2016).
Calenge, C. & adehabitatHR Home Range Estimation. R package version 0.4.21 (2006). https://CRAN.R-project.org/package=adehabitatHR
Simpfendorfer, C. A., Heupel, M. R. & Hueter, R. E. Estimation of short-term centers of activity from an array of omnidirectional hydrophones and its use in studying animal movements. Can. J. Fish. Aquat. Sci. 59, 23–32 (2002).
Clarke, K. R. & Gorley, R. N. PRIMER v7: User Manual/Tutorial. PRIMER-E, Plymouth (2015).
Clarke, K. R., Tweedley, J. R. & Valesini, F. J. Simple shade plots aid better long-term choices of data pre-treatment in multivariate assemblage studies. J. Mar. Biol. Assoc. UK. 94, 1–16 (2014).
Anderson, M. J. & Willis, T. J. Canonical analysis of principal coordinates: a useful method of constrained ordination for ecology. Ecology 84, 511–525 (2003).
Parnell, A. & Inger, R. Stable isotope mixing models in R with simmr. (2016). Available from: https://cran.r-project.org/web/packages/simmr/vignettes/simmr.html
Swanson, H. K. et al. D. A new probabilistic method for quantifying n-dimensional ecological niches and niche overlap. Ecology 96, 318–324 (2015).
Roberts, C. N. et al. Integrated fatty acid and stable isotope niches reveal trophic connections among sympatric large-bodied sharks. Aquat. Conserv. Mar. Freshw. Ecosyst. 34, e4251 (2024).
Phillips, D. L. et al. Best practices for use of stable isotope mixing models in food-web studies. Can. J. Zool. 92, 823–835 (2014).
Acknowledgements
We thank Mitch Graham, Luke Fitzpartrick, Dean Fitzpatrick, Philip Reid, Frances Murray, Russell Gueho, Jens Neiser, Elizabeth Dinsdale, Michael Doane, Lisa Hoopes, Ryan Hesse, Emma Kerr, Natalie Klinard, Laura Ryan, Olivia Seeger, and Suki for their help with fieldwork and logistics. Funding and support was provided by the Australian Government through the Our Marine Parks Grants Program (4-HANQ6UL), Neiser Foundation, Georgia Aquarium, Biopixel Oceans Foundation, Big Wave TV, and Norfolk Island Regional Council. Data were sourced from Australia’s Integrated Marine Observing System (IMOS) (animaltracking.aodn.org.au) – IMOS is enabled by the National Collaborative Research Infrastructure Strategy (NCRIS). Additional equipment and support were also provided by the IMOS Animal Tracking Facility.
Author information
Authors and Affiliations
Contributions
JKM: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Project administration; Supervision; Validation; Visualization; Writing – original draft; Writing – review & editing, LM: Conceptualization; Data curation; Funding acquisition; Formal analysis; Investigation; Methodology; Project administration; Supervision; Validation; Visualization; Writing – original draft; Writing – review & editing, CNR: Investigation; Methodology; Writing – review & editing, TMC: Investigation; Methodology; Writing – review & editing, MS: Investigation; Writing – review & editing, AB: Conceptualization; Funding acquisition; Investigation; Methodology; Project administration; Supervision; Writing – review & editing, CH: Conceptualization; Funding acquisition; Investigation; Methodology; Project administration; Supervision; Visualization; Writing – review & editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Matley, J.K., Meyer, L., Roberts, C.N. et al. Resource use of dusky and Galapagos sharks in response to fishing activities at a remote Pacific Island. Sci Rep 15, 31743 (2025). https://doi.org/10.1038/s41598-025-15677-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-15677-3