Abstract
CanAssist Breast (CAB), an immunohistochemistry (IHC) and artificial intelligence-based prognostic test, was developed on Hormone receptor-positive (HR +), HER2/neu-negative (HER2-) breast tumors from Indian patients and validated in retrospective global studies. CAB combines the expression of five protein biomarkers with three clinical parameters to segregate patients as low-risk (LR) or high-risk (HR) for distant recurrence. CAB has been in clinical use in South Asia, UAE, Turkey, and Iran for the last 8 years on > 7000 Early breast cancer (EBC) patients. Here we showcase for the first time, the real-world data on the usefulness of CAB to prognosticate across different clinicopathological parameters, histological types, and impact on treatment planning by analysing CAB usage in 5926 patients diagnosed from mid-2016 to 2024. Overall, CAB stratified 72% of patients as LR and 28% as HR for distant recurrence. Interestingly, CAB showed meaningful differences in HR proportions across different histological types; 19% and 29% in mucinous versus mixed mucinous, while 26% and 50% in papillary and micropapillary carcinomas, respectively. In the intermediate Ki67 group, CAB segregated 77% of patients as LR and 23% as HR. In conclusion, CAB is a first-of-its-kind prognostic test that serves as a cost-effective, suitable alternative to Western prognostic tests.
Similar content being viewed by others
Introduction
Breast cancer is the second most diagnosed cancer globally, constituting 11.6% of all cancer cases. However, among women, the incidence of breast cancer is about 23.8% with a 15.4% mortality rate as per the GLOBOCAN 2022 estimates1. In India, the incidence of breast cancer is on an upward trend, with the breast being the leading site of cancer among Indian females, constituting 28.8% of all cancers, with approximately 216,108 cases reported in 2022. The cumulative risk of developing breast cancer over a lifetime of 74 years for females was one in 29. As per the ICD (International Classification of Diseases)−10th edition, there is an increase of 12–13% in breast cancer cases every 5 years from 2015–20252. While the occurrence of breast cancer is on the rise, effective breast cancer screening protocols have resulted in increased early-stage diagnosis with a 15% reduction in overall breast cancer mortality3.
The most common subtype of breast cancer is hormone receptor-positive, HER2/neu-negative (HR +/HER2-), comprising 70% of all breast cancers4. The standard of care for this subtype involves a combination of surgery, radiation therapy, adjuvant endocrine therapy, and adjuvant chemotherapy5,6. The mainstay treatment for this subtype, especially in early stages, is adjuvant endocrine therapy; however, depending on the aggressive nature of the tumor, adjuvant chemotherapy can be recommended. ER +/HER2- early-stage breast cancer (EBC) is known to have a prolonged risk of recurrence even after 5 years of endocrine therapy5,7, making it crucial to identify patients who will benefit from adjuvant chemotherapy to reduce their risk of recurrence. Also, identifying patients who do not require chemotherapy would spare them from the burden of unnecessary treatment.
Management of HR +/HER2-, EBC is aided by prognostic signatures (protein/genes) that provide complementary information to traditional prognostic factors (age, tumor size, lymph node status, tumor grade, and estrogen/progesterone receptor status, and HER2)8,9. Multigene tests such as Oncotype DX, MammaPrint, EndoPredict, and Prosigna are widely available and provide prognostic information in HR +/HER2- EBC10,11,12,13. The gene signatures used by these prognostic tests majorly focus on proliferation, estrogen and progesterone receptor and HER2 receptor pathways and do not take into account non-proliferative pathways such as epithelial-to-mesenchymal transition (EMT), mesenchymal- to- epithelial transition (MET), invasion, and drug resistance which are major drivers of tumor cell dissemination and metastasis14. Apart from these multi-marker tests, there are free equations and online tools in practice that solely focus on clinicopathological factors to predict the risk of recurrence15,16,17.
The above-discussed prognostic tests have been majorly validated in the Western population, limiting their validation in Asian patients18,19. The incidence and mortality rates of breast cancer vary across different races, ethnicities, and geographical regions. Although there has been an improvement in breast cancer survival over the years, disparities in clinical outcomes persist20. Subgroup analysis of the TAILORx population showed ethnic differences among black and non-black women, with worse clinical outcomes observed in black women despite similar recurrence scores and systemic therapy18. Studies focusing on the prognostication of MammaPrint on Korean and Japanese patients revealed differences in proportions of low and high-risk patients compared to European patients21. Though these tests are useful for predicting prognosis in breast cancer patients, it is beneficial to use markers that are validated across various ethnicities22 to improve the utility of prognostic tests. Due to their high cost, these tests cause significant financial toxicity to the patient, at least in many Asian countries where most healthcare expenditure is out of pocket23.
To address the above unmet needs, a cost-effective indigenous prognostic test, CanAssist Breast (CAB), was developed. It is a proteomic-based test that helps predict the risk of distant cancer recurrence within five years of diagnosis in EBC patients with HR +/HER2- tumors. CAB was developed on Indian patient tumors to ensure Asian patients’ tumor biology is well represented. Using immunohistochemistry (IHC) and a machine learning algorithm, CAB combines the expression of five protein biomarkers with three clinical parameters to arrive at a risk score to segregate patients into ‘low-risk’(LR) or ‘high-risk’(HR) for distant breast cancer recurrence. CAB LR patients can benefit from avoiding chemotherapy, while HR patients may require chemotherapy and additional therapies24. The rationale for selecting CAB biomarkers (CD44, N‐Cadherin, pan‐Cadherin, ABCC4, and ABCC11) was due to their crucial role in diverse signaling pathways that are known to regulate cancer metastasis and drug resistance. These biomarkers are involved in key processes such as EMT, loss of cell–cell adhesion, MET, and drug resistance, which play an essential role in promoting metastasis and recurrence25,26. Unlike the multigene tests currently available, CAB is one of its kind, which takes tumor biology into account and examines the expression of proteins involved in the hallmarks of cancer24.
The clinical utility of CAB has been established by retrospective validation in India, the USA, and Europe (Spain, Germany, Austria, and Italy), demonstrating an acceptable performance of 5-year distant recurrence risk predictions across multiple ethnicities and in a prospective randomised completed TEAM trial in the Netherlands25,27,28,29,30. CAB has been in clinical use since mid-2016 in India31 and in select countries internationally on approximately > 7000 EBC patients.
In this manuscript, we evaluate the real-world data generated post-use of CAB in clinical settings. The objectives of this study are to i) understand the baseline patient demographics and tumor characteristics ii) to assess the impact of CAB risk stratification across clinicopathological parameters iii) to demonstrate the clinical use of CAB in male patients, different breast cancer histological types, and multifocal/multicentric tumors, and iv) to evaluate the influence of CAB on treatment decisions by the clinicians.
Methods
Ethics declarations
This prospective data analysis study was performed in accordance with the Declaration of Helsinki, and ethical approval was granted by the Sri Venkateshwara Hospital Ethics Committee (ECR/298/Inst/KA/2013/RR-19). The current anonymised study involves the analysis of the existing patient data obtained for CAB testing as per the patients’ written informed consent given in the test requisition form. All the patient identifiers were de-identified to maintain patient anonymity.
Patient selection
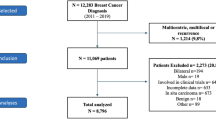
The current study cohort comprised 5926 patients with HR +/HER2- EBC for whom CAB was prescribed by a treating clinician during the period mid-2016 to July 2024. Surgically resected formalin-fixed paraffin-embedded tumor specimens, along with their reports, were sent to us as part of CAB testing. These patients belong to different regions of India and nearby Asian countries. All clinical details and ER, PR, HER2, and Ki67 status of the patients were obtained from the histopathology and IHC reports provided to us.
CanAssist Breast test performance and follow-up
The assessment of tumor blocks prior to IHC staining and detailed IHC staining protocol for the five CAB biomarkers, grading, and risk score generation were performed as described in our previous studies32,33. Information regarding the treatment (chemotherapy and hormonal therapy) given was obtained as part of follow-up with patient’s consent.
Statistical analyses
Descriptive statistics were used to evaluate the clinicopathological parameters of the study cohort. Statistical graphs and figures were prepared using GraphPad Prism 10 software version 10.3.1 (509) and Microsoft Excel. Significance was evaluated with Chi-squared test using MedCalc software version (22.032–64-bit). Treatment compliance was calculated as a ratio of patients complying with the treatment recommended, expressed as a percentage.
Results
Description of the cohort
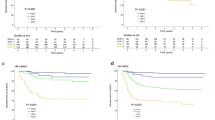
99% of the entire 5926 patient study cohort was constituted by females and 1% by male breast cancer patients. 78% of women were older than 48 years, while 22% were 48 years of age or younger. CAB segregated 72% (n = 4290) as LR and 28% (n = 1636) as HR. Interestingly, CAB risk proportions (LR: HR) between the female (72:28) and male patients (71:29) were similar (P = 0.87) (Fig. 1). The majority of patients (64%) had T2 tumors, followed by T1 tumors (34%) and T3 tumors (2%). Regarding node status, 82% of patients had node-negative (N0), and 18% of patients had node-positive (N1) disease. Patients with grade 2 (G2) tumors constituted the majority (65%) of the cohort (Table 1).
CAB risk stratification in male and female patients. Graph representing the comparison of CAB low-risk and high-risk proportions in female and male patients. Chi squared test was used to calculate P values on MedCalc software.
Performance of CAB in breast cancer histological types
Of the study cohort consisting of 5926 patients, 83.1% patients had invasive ductal carcinomas (IDC), 6.5% had invasive lobular carcinomas (ILC), 5.8% had mucinous carcinomas, 2.4% had papillary carcinomas, 0.5% mixed-papillary carcinomas, 0.3% micro-papillary carcinomas and 1.3% patients were grouped as ‘others’ respectively as depicted (Fig. 2a). Others include special types such as invasive tubular, cribriform, medullary carcinomas, etc.
Histological types of breast cancer evaluated by CAB. (a) Pie chart depicting the relative proportions of various histological types of breast cancer. (b) Comparison of CAB risk proportions in different breast cancer histological types is shown in the graph. The blue colored box indicates the risk proportions in mucinous and mixed mucinous carcinomas. Magenta colored box indicates the risk proportions in papillary and micropapillary carcinomas. Significance among the histological types was analysed using a comparison of proportions on MedCalc software.
Risk stratification by CAB was assessed in different breast cancer histological types. CAB LR: HR proportions were observed to be 72:28 for IDC, 76:24 for ILC, 81:19 for mucinous carcinomas, 71:29 for mixed mucinous carcinomas, 74:26 for papillary carcinomas, and 50:50 for micropapillary carcinomas. Comparison of CAB prognostication among mucinous and mixed mucinous carcinomas revealed a significant difference in LR proportions (81:71) and HR proportions (19:29) (P = 0.041). Similar analysis between papillary and micropapillary carcinomas showed a significant difference in LR proportions (74:50) and HR proportions (26:50) (P = 0.027) (Fig. 2b).
Distribution and CAB risk stratification in Ki67 subgroups
Out of the 5926-patient cohort, Ki67 data was available only for 4160 (70%) patients. As per the latest international Ki67 working group (IKWG) guidelines, patients were categorised into low Ki67 (≤ 5%), intermediate Ki67 (6–29%), and high Ki67 (≥ 30%) subgroups based on their percentage Ki67 expression. We observed that the majority (63%) of these patients expressed intermediate levels of Ki67, followed by 29% of patients who expressed high levels of Ki67, and only 8% of patients expressed low levels of Ki67 (Fig. 3a).
Use of CAB in different Ki67 subgroups. (a) Pie chart illustrating the percentage of patients in different Ki67 subgroups (subgroupings done as per the IKWG guidelines). (b) Graph showing CAB risk stratification in Low Ki67 (≤ 5%), Intermediate Ki67 (6–29%), and High Ki67 (≥ 30%) subgroups. Chi squared test was used to calculate the P values on MedCalc software.
CAB stratified patients expressing low, intermediate, and high Ki67 with LR: HR proportions of 81:19 (P < 0.0001), 77:23 (P < 0.0001), and 60:40 (P = 0.045) significantly. Notably, CAB was able to stratify 19% of patients as HR in the low Ki67 subgroup and 60% as LR in the high Ki67 subgroup (Fig. 3b).
Performance of CAB in Multifocal/Multicentric tumors
Of the 70 patients with multifocal/multicentric (MF/MC) tumors, CAB stratified 66% (n = 46) into LR and 34% (n = 24) into HR categories when the first CAB was performed on the clinically higher-risk tumor (Tumor1). No additional CAB testing was required for CAB HR patients. However, a second CAB was performed for CAB LR patients on the clinically lower-risk tumors (Tumor 2). CAB was able to re-stratify 85% (n = 39) into LR and 15% (n = 7) into HR categories (Fig. 4a,4b).
Usefulness of CAB in Multifocal/Multicentric tumors. (a) Flow chart representation of recommended protocol to evaluate the intertumoral prognostication in multifocal/multicentric tumors by CAB. Tumor 1 represents clinically high-risk tumor; Tumor 2 represents clinically low risk tumor. (b) Re-stratification of CAB low risk category when second CAB was performed on Tumor 2.
Treatment compliance by clinicians
Of the total 5926 patients, we were able to obtain information on treatment received only for 3075 patients, which constitutes 52% of the cohort. 74% (n = 2279) were stratified by CAB as LR and 26% (n = 796) as HR. 95% (n = 2166) of CAB LR patients did not receive chemotherapy as suggested, leading to 95% treatment compliance in the LR category. In contrast, 79% (n = 625) of CAB HR patients received chemotherapy, resulting in 79% treatment compliance in the HR category (Table 2).
We performed a similar analysis in N0/N1 subgroups and patients aged ≤ 48 years and > 48 years. For CAB LR patients, a good compliance of 96% was observed for the N0 subgroup and 90% for the N1 subgroup. Regarding age, in CAB LR patients, good compliance of 92% was observed in patients aged ≤ 48 years and 96% in patients aged > 48 years (Table 2).
Discussion
Prognostic and predictive tests, in addition to standard clinical parameters (TNM and grade), are of great importance in planning treatment to anticipate the best clinical outcomes for each EBC patient with HR +/HER2- disease. Increasingly, tumor biology and microenvironment are being considered as major factors in determining clinical outcomes34. Amongst all the prognostic tests, CAB is the only multi-protein test that uses IHC coupled with an AI-based algorithm, developed and validated on Indian and global breast cancer patients. The current study was conducted to analyse how CAB is being utilised in the real world. CAB has been in clinical use since 2016 on more than 7000 patients and has seen a gradual increase in its usage in India and internationally year on year. While genomic prognostic tests have shown their utility, this is the first publication of the usefulness of CAB for patients across multiple clinicopathological parameters, breast cancer histological types, and in treatment planning.
In this study, the majority of patients were women older than 48 years with T2, N0, and G2 tumors, and this is in line with our previous study31. This use of CAB primarily for patients with a slightly better prognosis is understandable, as CAB is the new kid on the block in the area of breast cancer prognosis. Interestingly, we observe that CAB prognostication correlated with clinically higher risk parameters such as N1 and grade 3 (G3) tumors for e.g. CAB segregated 56% of patients with N1 and 75% with G3 disease as HR patients who can benefit from chemotherapy as against 21% and 5% HR patients from N0 and grade 1 (G1) tumors. Interestingly, CAB also segregated 44% and 25% of patients as LR in N1 and G3 subgroups, showcasing its ability to find LR patients in clinically higher-risk tumors, thereby helping them avoid aggressive treatments. Although rare, breast cancer is also observed in men, comprising only 1% of all breast cancers. Unfortunately, male breast cancer has high mortality due to late diagnosis35. Interestingly, the risk proportions (LR: HR) of male (71:29) were similar to that of female patients (72:28), demonstrating the robustness of CAB across genders, thereby highlighting its clinical use in male patients and showcasing that the underlying tumor biology that CAB analyses remain similar in males or females.
Invasive breast carcinomas are categorised by their morphological subtypes. Breast carcinomas exhibiting a distinct histological pattern in more than 90% of the tumors are classified as a pure special tumor type, such as lobular, mucinous, papillary, micropapillary, tubular, cribriform, and medullary carcinomas36. In agreement with published reports, our cohort has IDC as the most commonly occurring histological type, followed by ILC37. We observed comparable CAB LR: HR proportions among IDC, ILC, mucinous, and papillary histological types.
However, we noted interesting observations in CAB risk proportions of micropapillary and mixed mucinous carcinomas. Interestingly, CAB has stratified 50% of micropapillary patients as HR in line with their aggressive clinical behavior, especially compared with papillary carcinomas, which are known to be not so aggressive. Studies show that micropapillary carcinomas are known to exhibit aggressive clinical features such as increased tumor size, higher grade, and lymph node metastasis due to their higher tendency of lymphovascular invasion38. Overexpression of a cell adhesion molecule, N- cadherin, was observed in micropapillary type compared to non-micropapillary type, indicating its role in promoting tumor invasion and metastasis, which in turn contributes to their aggressive nature39. CD44 is a transmembrane glycoprotein with multiple roles in tumor progression. A couple of studies indicate that enhanced lymphovascular invasion in micropapillary type is observed due to the expression of CD44-positive and CD24-negative phenotypes on micropapillary tumor cells. Changes in the expression of these molecules are known to contribute to the stem cell characteristics of tumor cells38,40,41. It is noteworthy that CD44 and N-cadherin are a part of five biomarkers evaluated by CAB, proving the usefulness of CAB in assessing the underlying tumor biology of micropapillary carcinomas.
Pure mucinous carcinomas are known to be slow growing with a lower tendency of lymph node metastasis and have a good prognosis, and in line with this, CAB stratified 81% of patients as LR. However, compared to mucinous carcinomas, ‘mixed mucinous’ carcinomas have a mixture of mucinous areas and infiltrating ductal epithelial components, which makes them behave more like IDC with respect to prognosis and treatment42. This is reflected in CAB-based risk stratification, where significantly higher proportions (29%) of HR cases are observed in mixed mucinous carcinoma, and its LR: HR proportions are similar to IDC, indicating once again the prognostic utility of CAB in this histological type. Both these observations are especially worth mentioning as CAB relies only on five biomarkers plus tumor size, grade, and node status, and does not consider the histological type or ER expression in risk categorization. This shows that the underlying tumor biology seen by five CAB biomarkers is reflective of aggressiveness based on histological types as well.
High levels of Ki67 are known to be associated with poor prognosis in HR +/HER2- EBC43. Here, we have shown that prognostication by CAB has some correlation with levels of Ki67 -for e.g., in ≤ 5% Ki67, CAB segregated 81% of patients as LR, and in ≥ 30% Ki67, CAB segregated 40% as CAB HR. Interestingly, CAB found HR patients in low Ki67 and vice versa, indicating the usefulness of CAB-based risk stratification. More importantly, we noticed that the majority (63%) of the CAB prescriptions fell in intermediate Ki67 (6–29%), and as per IKWG guidelines, a prognostic test is recommended for such patients44. CAB successfully segregated these patients into 77% LR and 23% HR, thus showcasing the utility of CAB in treatment planning over Ki67, which is in line with our previously published retrospective data of superior performance of CAB in different Ki67 subgroups45.
Multifocal/Multicentric breast tumors are characterised by the presence of two or more distinct foci within the same quadrant or across different quadrants of the same breast46. These tumors are aggressive with a worse prognosis due to a higher rate of lymph node metastasis47. Evaluation of all tumor foci is important in the estimation of risk of recurrence and metastasis, which would aid in more effective therapy and prognosis48. In patients with MF/MC tumors, we typically perform first CAB on a ‘clinically higher risk’ tumor (larger/higher grade/node positive) if that tumor is CAB HR, then treatment is planned accordingly, but if CAB LR then CAB is recommended on the next larger/higher grade tumor. Interestingly, we observed that out of all CAB LR patients, in 15% of patients, the second CAB performed showed HR disease. This change in the risk category indicates the usefulness of CAB to assess ‘intertumoral prognostication’ and emphasizes the necessity of testing the second tumor (i.e., clinically lower-risk tumor) for effective treatment planning. Increased EMT is often observed in MF/MC tumors49, and since CAB biomarkers play a key role in EMT, we think CAB is able to assess the tumor biology of MF/MC tumors effectively.
Evaluating the adherence to CAB treatment recommendations by clinicians is critical to understanding the impact of CAB in clinical settings. Overall, we see higher treatment compliance in the CAB LR category (95%) than in the CAB HR category (79%). Interestingly, the CAB LR compliance goes up to 96% in ‘clinically lower risk’ patients who are either older or with N0 disease, and conversely reduces to 90% in ‘clinically higher risk’ patients who are younger or with N1 disease. Additionally, in CAB HR patients, the highest treatment compliance of 92% was seen in young patients. All of these observations indicate that the underlying ‘clinical risk’ category in the clinician’s mind, perhaps, does have an impact on treatment compliance. We hope in the future, clinicians’ trust in CAB for treating ‘clinically higher risk’ patients will increase. This data on the treatment planning of patients using CAB strengthens the utility and emphasizes the confidence of clinicians in CAB.
In Low/middle-income countries, including India, the economic burden due to breast cancer treatment is high50. Greater than 50% of patients from low-income households devote more than 20% of their annual household budget to cancer treatment of 1 family member51. Limited studies are available that represent the economic burden of breast cancer in India. However, a study conducted in Punjab households revealed that more than 60% of breast cancer treatment costs were contributed by medicines and hospitalization23.. The treatment cost for breast cancer in India in private set up including surgery, radiation, and examinations, ranges between US$6700- US$8000. The cost of chemotherapy (six rounds) with tailored treatment is estimated to be higher than US$27,00052. Such exorbitant treatment costs can be avoided by the use of a prognostic test. A recent study conducted by Bapna et al. at a cancer care center in Rajasthan compared the costs of using the CAB and ODX tests. The findings revealed that using ODX was 5.8 times more expensive than CAB, with patients saving up to USD 3,477 (INR 278,161) by using CAB, further emphasizing its cost-effectiveness53. In the current study, we observe that CAB stratified 72% of patients as LR, indicating these LR patients can avoid chemotherapy and minimise the financial burden associated with it. In our study cohort, 95% of patients are treated in private hospitals, and since CAB stratifies the majority of these patients as LR use of CAB is leading to a reduction in financial and physiological toxicity.
The strength of this study lies in its large sample size and a well-represented study population. However, a limitation is the lack of clinical outcomes for all patients at the end of 5-year follow-up. Since all 5926 patients will complete 5 years in 2029, we will analyse the entire data and publish with clinical outcomes in 2029. As of now, only 583 patients have completed 5 years since diagnosis, and we have follow-up of 305 patients. While we understand this is a very small number of the total and hence not added in the results in this manuscript, we have observed that the current NPV in the LR category is > 95% as predicted. The follow-up of these patients is ongoing. We hope that this additional data will strengthen the usage of CAB in treatment planning and provide valuable insights into its prognostic utility in real-world settings.
Conclusion
In summary, CAB is the first of its kind prognostic test developed on Indian patients to ensure Asian patients’ tumor biology is well represented. CAB is effectively stratifying HR +/HER2- EBC patients across different clinicopathological parameters routinely seen. We observed an increase in CAB prescriptions over the years in India and other countries. This demonstrates a gradual acceptance of CAB by clinicians and their trust in its effectiveness and utility. CAB usage has helped clinicians to tailor treatment for the patients. Coupled with worldwide validation, it presents as a cost-effective, suitable alternative to Western prognostic tests for patients in Asia, Africa, and the Middle East.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- CAB:
-
CanAssist Breast
- HR + :
-
Hormone receptor-positive
- HER2-:
-
Human epidermal growth factor receptor 2 negative
- LR:
-
Low risk
- HR:
-
High risk
- IHC:
-
Immunohistochemistry
- EBC:
-
Early breast cancer
- EMT:
-
Epithelial-to-Mesenchymal transition
- MET:
-
Mesenchymal-to-Epithelial transition
- IDC:
-
Invasive ductal carcinoma
- ILC:
-
Invasive lobular carcinoma
- IKWG:
-
International Ki67 working group
- MF/MC:
-
Multifocal/Multicentric
References
Bray, F. et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 74, 229–263 (2024).
Sathishkumar, K., Chaturvedi, M., Das, P., Stephen, S. & Mathur, P. Cancer incidence estimates for 2022 & projection for 2025- result from national cancer registry programme. India. Ind. J. Med. Res. 156, 598–607 (2022).
Mittra, I. et al. Effect of screening by clinical breast examination on breast cancer incidence and mortality after 20 years: Prospective, cluster randomised controlled trial in Mumbai. BMJ 372, 256 (2021).
National Cancer Institute: Surveillance, Epidemiology, and End Results. Cancer Stat Facts: female breast cancer. https://seer.cancer.gov/statfacts/html/breastsubtypes.html. Accessed on (2024).
Huppert, L. A., Gumusay, O., Idossa, D. & Rugo, H. S. Systemic therapy for hormone receptor-positive/human epidermal growth factor receptor 2-negative early stage and metastatic breast cancer. CA Cancer J. Clin. 73, 480–515 (2023).
Walsh, E. M., Smith, K. L. & Stearns, V. Management of hormone receptor-positive, HER2-negative early breast cancer. Semin. Oncol. 47, 187–200 (2020).
Pan, H. et al. 20-year risks of breast-cancer recurrence after stopping endocrine therapy at 5 years. N. Engl. J. Med. 377(19), 1836–1846 (2017).
Cianfrocca, M. & Goldstein, L. J. Prognostic and predictive factors in early-stage breast cancer. Oncologist 9, 606–616 (2004).
Gaß, P. et al. Factors influencing decision-making for or against adjuvant and neoadjuvant chemotherapy in postmenopausal hormone receptor-positive breast cancer patients in the EvAluate-TM study. Breast Care. 11, 315–322 (2016).
Paik, S. et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N. Engl. J. Med. 351, 2817–2826 (2004).
van de Vijver, M. et al. A gene-expression signature as a predictor of survival in breast cancer. N. Engl. J. Med. 347, 1999–2009 (2002).
Filipits, M. et al. A new molecular predictor of distant recurrence in ER-positive, HER2-negative breast cancer adds independent information to conventional clinical risk factors. Clin. Cancer Res. 17, 6012–6020 (2011).
Chia, S. K. et al. A 50-gene intrinsic subtype test for prognosis and prediction of benefit from adjuvant tamoxifen. Clin. Cancer Res. 18, 4465–4472 (2012).
Karagiannis, G. S., Goswami, S., Jones, J. G., Oktay, M. H. & Condeelis, J. S. Signatures of breast cancer metastasis at a glance. J. Cell Sci. 129, 1751–1758 (2016).
Cuzick, J. et al. Prognostic value of a combined estrogen receptor, progesterone receptor, Ki-67, and human epidermal growth factor receptor 2 immunohistochemical score and comparison with the Genomic Health recurrence score in early breast cancer. J. Clin. Oncol. 29(32), 4273–4278 (2011).
Wishart, G. C. et al. PREDICT: A new UK prognostic model that predicts survival following surgery for invasive breast cancer. Breast Cancer Res. 12, R1 (2010).
Fong, Y. et al. The Nottingham Prognostic Index: Five- and ten-year data for all-cause survival within a screened population. Ann. R. Coll. Surg. Engl. 97(2), 137–139 (2015).
Albain, K. S. et al. Race, ethnicity, and clinical outcomes in hormone receptor-positive, HER2-negative, node-negative breast cancer in the randomised TAILORx trial. J. Natl. Cancer Inst. 113(4), 390–399 (2021).
Cardoso, F. et al. 70-gene signature as an aid to treatment decisions in early-stage breast cancer. N. Engl. J. Med. 375, 717–729 (2016).
Hirko, K. A. et al. The impact of race and ethnicity in breast cancer—disparities and implications for precision oncology. BMC Med. 20, 72 (2022).
Na, K. Y. et al. The 70-gene prognostic signature for Korean breast cancer patients. J. Breast Cancer. 14(1), 33–38 (2011).
Bhattacharyya, G. S. et al. Overview of breast cancer and implications of overtreatment of early-stage breast cancer: An Indian perspective. JCO Global. Oncol. 6, 789–798 (2020).
Jain, M. & Mukherjee, K. Economic burden of breast cancer to the households in Punjab India. Int. J. Med. Pub. Health. 6, 13–18 (2016).
Ramkumar, C. et al. Development of a novel proteomic risk-classifier for prognostication of patients with early-stage hormone receptor-positive breast cancer. Biomark. Insights. 13, 1–9 (2018).
Bakre, M. M. et al. Clinical validation of an immunohistochemistry-based CanAssist-Breast test for distant recurrence prediction in hormone receptor-positive breast cancer patients. Cancer Med. 8, 1755–1764 (2019).
Chaffer, C. L. & Weinberg, R. A. A perspective on cancer cell metastasis. Science 331, 1559 (2011).
Doval, D. C. et al. The usefulness of CanAssist breast in the assessment of recurrence risk in patients of ethnic Indian origin. Breast 59, 1–7 (2021).
Sengupta, A. K. et al. Comparison of breast cancer prognostic tests CanAssist breast and oncotype DX. Cancer Med. 9, 7810–7818 (2020).
Gunda, A. et al. A retrospective validation of CanAssist breast in European early-stage breast cancer patient cohort. Breast 63, 1–8 (2022).
Zhang, X. et al. Ten-year distant-recurrence risk prediction in breast cancer by CanAssist breast (CAB) in Dutch sub-cohort of the randomised TEAM trial. Breast Cancer Res. 25, 40 (2023).
Sankaran, S. et al. CanAssist breast impacting clinical treatment decisions in early-stage HR+ breast cancer patients: Indian scenario. Ind. J. Surg. Oncol. 12, 21–29 (2021).
Attuluri, A. K. et al. Analytical validation of CanAssist-Breast: An immunohistochemistry based prognostic test for hormone receptor positive breast cancer patients. BMC Cancer 19, 249 (2019).
Serkad, C. P. et al. Validation of CanAssist Breast immunohistochemistry biomarkers on an automated platform and its applicability in tissue microarray. Int. J. Clin. Exp. Pathol. 14, 1013–1021 (2021).
Cejudo, M. A. D., Torralvo, A. G., Cejuela, M., Pinelo, S. M. & Bofill, J. V. Targeting the tumor microenvironment in breast cancer: Prognostic and predictive significance and therapeutic opportunities. Int. J. Mol. Sci. 24, 16771 (2023).
Ottini, L. et al. Male breast cancer. Crit. Rev. Oncol. Hematol. 73, 141–155 (2010).
WHO Classification of Tumours Editorial Board. Breast tumours. WHO classification of tumours series. In 5th ed., 2 (Lyon (France): International Agency for Research on Cancer 2019).
Lobbezoo, D. et al. The role of histological subtype in hormone receptor positive metastatic breast cancer: Similar survival but different therapeutic approaches. Oncotarget 7(20), 29412–29419 (2016).
Verras, G. I. et al. Micropapillary breast carcinoma: From molecular pathogenesis to prognosis. Breast Cancer: Targ. Ther. 14, 41–61 (2022).
Nagi, C. et al. N-cadherin expression in breast cancer: Correlation with an aggressive histologic variant – invasive micropapillary carcinoma. Breast Cancer Res. Treat. 94, 225–235 (2005).
Li, W. et al. The clinicopathological significance of CD44+/CD24−/low and CD24+ tumor cells in invasive micropapillary carcinoma of the breast. Pathol. Res. Pract. 206(828), 834 (2010).
Badyal, R. K., Bal, A., Das, A. & Singh, G. Invasive micropapillary carcinoma of the breast: Immunophenotypic analysis and role of cell adhesion molecules (CD44 and E-Cadherin) in nodal metastasis. Appl. Immunohistochem. Mol. Morphol. 24, 151–158 (2016).
Skotnicki, P.et al. Pure and Mixed mucinous carcinoma of the breast: A comparison of clinical outcomes and treatment results. Breast J. 1–6 (2016).
Ma, Q., Liu, Y. B., She, T. & Liu, X. L. The Role of Ki-67 in HR+/HER2- breast cancer: A real-world study of 956 patients. Breast Cancer: Targ. Ther. 16, 117–126 (2024).
Nielsen, T.O. et al. Assessment of Ki67 in breast cancer: Updated recommendations from the international Ki67 in breast cancer working group. JNCI J. Natl. Cancer Inst.113(7), (2021).
Eshwaraiah, M.S, Gunda, A., Kanakasetty, G.B.& Bakre, M.M. The usefulness of CanAssist breast over Ki67 in breast cancer recurrence risk assessment. Cancer Med. 1–10 (2023).
Andea, A.A. et al. Pathologic analysis of tumor size and lymph node status in multifocal/multicentric breast carcinoma. Cancer. 94(5) (2002).
Lynch, S. P. et al. Multifocality and multicentricity in breast cancer and survival outcomes. Ann Oncol. 23(3063), 3069 (2012).
Lang, Z. et al. Multifocal and multicentric breast Carcinoma: A Significantly more aggressive tumor than unifocal breast cancer. Anticancer Res. 37, 4593–4598 (2017).
Weissenbacher, T. et al. Multicentric and multifocal versus unifocal breast cancer: Differences in the expression of E-cadherin suggest differences in tumor biology. BMC Cancer 13, 361 (2013).
Wadasadawala, T. et al. 2024 Out-of-pocket payment and financial risk protection for breast cancer treatment: A prospective study from India. Lancet Reg. Health Southeast Asia. 24 100346
Rajpal, S., Kumar, A.& Joe, W. Economic burden of cancer in India: Evidence from cross-sectional nationally representative household survey PLoS ONE. 13(2) (2014).
Barathe, P. C. et al. Cost of breast cancer diagnosis and treatment in India: A scoping review protocol. BMJ Open 12, e057008 (2022).
Bapna, A., Samar, A. K., Gupta, T. & Agarwal, P. Impact of cost of Oncotype DX versus CanAssist Breast, an Indian prognostic test, in the management of node-negative early breast cancer in a single centre in India. JCO Glob. Oncol. 9, 14–14 (2023).
Acknowledgements
We thank Mr. Manjunatha G, Mrs. Prathima R, Mr. Harishkumar N, Mr. Dinesh Babu P and Ms. Riya Jain from OncoStem Diagnostics, Bangalore, for helping us with IHC experimentation, histopathological work and obtaining the follow up data.
Author information
Authors and Affiliations
Contributions
Conception and study design: MB. Acquisition, analysis and interpretation of data: TDD, MS, BAS, PS, RB, NK, DP, MB. Drafting of the manuscript: MB, TDD. Supervision: MB. Reviewed the paper: all authors. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
All authors are employees of OncoStem Diagnostics Pvt Ltd, Bangalore, India.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Durgekar, T.D., Sunder, M., Savitha, B.A. et al. Real-world data of CanAssist Breast- first immunohistochemistry and AI-based prognostic test. Sci Rep 15, 30430 (2025). https://doi.org/10.1038/s41598-025-15736-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-15736-9