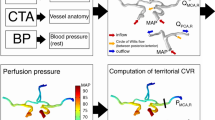
Abstract
The role of intravascular pressure ratio across carotid artery stenosis in assessing cerebral hemodynamics remains unclear. This study evaluated the utility of pressure-wire-based carotid pressure ratio (CPR) measurement during carotid artery stenting (CAS) in patients with internal carotid artery (ICA) stenosis. We prospectively enrolled 25 patients undergoing CAS for unilateral ICA stenosis. CPR was defined as the ratio of mean arterial pressure distal to proximal to the stenosis, measured before and after CAS using a pressure wire. We analyzed the correlation of CPR with cerebral vascular reserve (CVR), cerebral blood flow asymmetry index (CBF-AI), and stenosis-related parameters. Receiver operating characteristic (ROC) analysis identified optimal CPR thresholds. CPR significantly correlated with CVR (R2 = 0.38), CBF-AI, and stenosis severity. A CPR ≤ 0.80 accurately predicted impaired CVR (< 10%) with 100% sensitivity and 89% specificity. The CPR reached 1.0 when the minimal luminal diameter was ≥ 1.96 mm, suggesting resolution of the pressure gradient. No complications were observed with intraoperative CPR measurement. CPR measurement using a pressure wire is safe, minimally invasive, and reflects cerebral hemodynamic status. The CPR may complement imaging-based evaluation and support intra- and post-operative management in ICA stenosis. Further validation in larger multicenter cohorts is warranted.
Similar content being viewed by others
Introduction
Evaluation of cerebral blood flow (CBF) and cerebrovascular reserve (CVR) after the acetazolamide challenge using single-photon emission computed tomography (SPECT) is crucial in determining the indication for revascularization procedures such as carotid endarterectomy (CEA) or carotid artery stenting (CAS) for internal carotid artery stenosis1,2. A significant reduction in both rest CBF and CVR indicates a state close to misery perfusion, making revascularization an appropriate treatment option3. Furthermore, a decrease in CVR is a risk factor for cerebral hyperperfusion syndrome, making preoperative assessment particularly important4,5. However, acetazolamide challenge, which is used to measure CVR, has associated risks such as allergic reactions and the potential to induce cerebral infarction due to intracerebral steal phenomenon6. In recent years, the fractional flow reserve (FFR) has been used to evaluate lesions in coronary artery stenosis7. Coronary FFR, defined as the ratio of the blood pressure before stenosis to the blood pressure beyond the stenosis when a vasodilator is administered and the coronary artery is maximally dilated, well reflects the degree of myocardial ischemia and contributes to improving treatment prognosis8,9. However, few studies have investigated the ratio of intravascular pressure proximal and distal stenosis sites and cerebral hemodynamic impairment in carotid artery stenosis lesions; thus, the usefulness of this ratio remains unclear. Therefore, in this study, we measured the ratio of intravascular pressure at proximal and distal stenosis sites before and after carotid artery stenting (CAS) procedure and investigated its perioperative usefulness in patients with ICA stenosis.
Materials and methods
Patients
The study protocol was governed by the guidelines of the national government based on the tenets of the Declaration of Helsinki revised in 1983 and was approved by the Research Ethics Committee of University of Fukui. Informed consent was obtained from all subjects or their legal guardians. This prospective study included 25 cases with unilateral ICA stenosis (2 women; mean age 67 ± 10 years, range 46–76 years) from among a total of 27 cases in which carotid artery stenting was performed at our hospital between September 2022 and July 2024, after excluding 2 patients with contralateral or tandem lesions, and those undergoing retreatment.
Symptomatic lesions were defined as those accompanied by a history of stroke, amaurosis fugax, or transient ischemic attacks involving the ipsilateral carotid territory occurring within 180 days of the initial assessment10. Patients without severe stenosis (< 50%) and those with bilateral lesions, tandem lesions, or moyamoya disease were excluded from the study. The stenosis rate was measured using digital subtraction angiography using North American Symptomatic Carotid Endarterectomy Trial criteria11. The vascular diameter at the most stenotic site was measured using digital subtraction angiography (DSA) before and after the procedure in all patients. All DSA procedures were performed using biplanar digital subtraction angiography complemented by 3D-rotation angiography to allow accurate assessment of vessel diameter and lesion morphology.
Single-photon emission computed tomography (SPECT) measurements
Preoperative clinical SPECT was performed following a dual-table autoradiographic protocol with dual administration of iodoamphetamine12. Briefly, two dynamic scans were acquired in quick succession with a 2-min interval between them. The first scan covered the initial 0–28-min period, while the second scan was acquired at 30–58 min. At 4 min/frame, each of the two dynamic scan periods produced seven frames. One-minute infusions of 123I-iodoamphetamine were administered into the antecubital vein at 0 and 30 min. Acetazolamide (17 mg/kg, 1000 mg maximum) was administered intravenously 20 min after the first injection and 10 min before the second iodoamphetamine injection. Projection data for the acquisition durations of the first and second scans were summed and reconstructed. SPECT data provide quantitative information on cerebral blood flow (CBF) at rest and after acetazolamide challenge, thereby providing information about the vascular reserve and the severity of hemodynamic brain ischemia. Regional cerebral vascular reserve (CVR) was defined as the ratio of the difference between acetazolamide-activated regional CBF and resting CBF to resting CBF: CVR = ([acetazolamide-activated CBF / resting CBF—1] × 100[%]). The asymmetry index (CBF-AI ) was defined as the ratio of CBF between the ipsilateral and contralateral sides: CBF-AI = (CBF on the ipsilateral side/CBF on the contralateral side). 123I-iodoamphetamine autoradiography was performed postoperatively in all patients. This method uses a single iodoamphetamine administration to assess CBF at rest13. The image reconstruction process was identical to that used in the dual-table autoradiographic protocol. Preoperative CBF and CVR were measured in all patients. A significant decrease was defined as CBF-AI < 0.8 and CVR < 10% based on previous research on the increased risk of stroke recurrence in medically treated patients1,4,5,8.
Carotid pressure ratio (CPR) measurements
CPR was defined as the ratio of mean blood pressure distal to proximal to the stenosis at rest and was measured using a pressure wire (0.014 inches, Omniwire®, Koninklijke Philips, Amsterdam, Netherlands). We used mean arterial pressure (MAP) because it reflects the steady-state perfusion pressure more accurately than systolic or diastolic values, which can be affected by transient hemodynamic fluctuations. In addition, the use of MAP is consistent with the methodology employed in FFR measurements in coronary physiology. This ratio-based approach is analogous to the concept of FFR in cardiology. Using a ratio allows for normalization across varying systemic blood pressures and provides a standardized, intuitive measure of hemodynamic significance, facilitating intraoperative decision-making. The pressure wire was first advanced through a guiding catheter to the common carotid artery, where the mean arterial pressure was measured in Pa. The pressure wire then traversed the stenosis and the mean blood pressure was measured, and the CPR was calculated. After CAS, the mean blood pressure was measured at the same location as before the procedure, and the CPR was recalculated to investigate the effects of endovascular therapy.
CAS procedure
All patients were administered 100 mg aspirin and 75 mg clopidogrel or 3.75 mg prasugrel daily as dual antiplatelet therapy for at least one week before the procedure to prevent thrombosis. Intravenous heparin was administered during the procedure. All procedures were performed by a neurosurgeon. In this study, 15 and 10 patients were treated under general and local anesthesia, respectively. A distal filter protection device alone was used in 10 patients, while 15 received a proximal common carotid artery balloon, an external carotid artery balloon, and a distal internal carotid artery filter with continuous flow reversal to the femoral vein. In cases treated with filter protection only, the pressure wire was advanced across the lesion prior to filter deployment, and CPR was measured. After the initial measurement, a distal protection device (Spider FX®, Medtronic, Minneapolis, MN, USA) was deployed in the distal ICA over the wire, followed by standard CAS procedures. In flow reversal cases, a guiding catheter was placed in the common carotid artery, and the pressure wire was first navigated across the stenosis to perform CPR measurement. After this, the pressure wire was removed, and both the external and common carotid arteries were occluded to initiate flow reversal. Post-stenting CPR measurements were performed after releasing all occlusions to restore antegrade flow.
Data analysis
The correlations between the diameter at most stenosis site (DMS), stenosis rate (North American Symptomatic Carotid Endarterectomy Trial criteria), CBF, CVR, and CPR were analyzed using Spearman’s correlation test for rank-based association, and simple linear regression analysis was additionally performed to assess linear relationships and calculate R2 values for illustrative purposes. The optimal cut-off value was determined using receiver operating characteristic curve analysis. Statistical significance was set at P < 0.05, and JMP version 15 (SAS Institute Inc., Cary, NC, SAS Institute Inc.) was used to perform the statistical analyses. This study also examined the incidence of symptomatic cerebral infarction thought to be caused by CPR measurement techniques and hyperperfusion syndrome, defined as a CBF-AI of ≥ 1.2 or a quantitative CBF value of 2 × the preoperative level.
Results
In this study, nine patients who presented with minor strokes had symptomatic stenosis, with an average stenosis rate of 72%. The average resting CBF (29.7 ± 6.1 vs. 30.7 ± 6.6, P < 0.05) and CVR (47.4 ± 28.4 vs. 51.1 ± 23.0, P < 0.05) on the ipsilateral side measured by SPECT were significantly reduced compared with the contralateral side. Three cases showed significantly decreased CBF on the ipsilateral side, as measured by SPECT. Three cases showed a significantly decreased CVR (Fig. 1). The CAS procedure was successful in all patients, and on average, all parameters improved postoperatively (Table 1). However, these improvements reflect mean group values and were not necessarily observed uniformly in every individual case. Significant correlations were observed between CPR and DMS (y = 3.10x – 0.31, R2 = 0.27, P < 0.01), CPR and CVR (y = 72.6x – 12.6, R2 = 0.38, P < 0.01), CRP and CBF-AI (y = 0.18x + 0.83, R2 = 0.23, P < 0.01), and CPR and stenosis rate (y = -53.9x + 105, R2 = 0.23, P < 0.01) but not between CPR and CBF (Fig. 2). In the two cases in which CBF decreased but CVR remained normal, CPR did not decrease. Receiver operating characteristic analyses revealed a CPR cutoff of ≤ 0.80 to achieve a CVR of < 10% (100% sensitivity, 89% specificity) and a cutoff of 0.74 to achieve a CBF-AI of < 0.8 (75% sensitivity and 89% specificity). The cutoff value for the diameter at the most severe stenosis to achieve a CPR of 1.00 was 1.96 mm (75% sensitivity and 89% specificity) (Fig. 3, 4). Regardless of the CAS procedure, CPR measurements using pressure wires were possible in all cases, and no symptomatic complications were observed.
Scatterplot showing the relationship between cerebral blood flow asymmetry index (CBF-AI) and cerebral vascular reserve (CVR). Vertical dashed line: CBF-AI threshold (< 0.8); horizontal dashed line: CVR < 10%. These threshold values were adopted based on previously published studies1,4,5,8 indicating their association with cerebral hemodynamic impairment and hyperperfusion risk, and were not newly defined in this study.
Scatterplots illustrating the correlation between carotid pressure ratio (CPR) and key hemodynamic parameters: (A) cerebral blood flow asymmetry index (CBF-AI) (y = 0.18x + 0.83, R2 = 0.23, P < 0.01); (B) diameter of the most severe stenosis (DMS) (mm) (y = 3.10x – 0.31, R2 = 0.27, P < 0.01). (C) the degree of stenosis based on the NASCET method (%) (y = -53.9x + 105, R2 = 0.23, P < 0.01). (D) cerebral vascular reserve (CVR) (%) (y = 72.6x – 12.6, R2 = 0.38, P < 0.01). Each linear regression line represents the trend of the relationship. A statistically significant correlation was observed between CPR and all parameters. R2 values were calculated using simple linear regression analysis to illustrate the strength of linear trends in these relationships. Note: Scatter plots include both pre- and post-stenting values of CPR, DMS, and NASCET stenosis rate to visualize the full spectrum of hemodynamic changes.
Receiver operating characteristic (ROC) curve analysis for the diagnostic performance of CPR and the diameter at most stenosis site (DMS) in predicting impaired cerebral hemodynamics. (A) CPR cutoff of ≤ 0.80 predicted a CVR of less than 10% with an area under the curve (AUC) of 0.91. (B) CPR cutoff of ≤ 0.74 predicted a CBF asymmetry index (CBF-AI) of less than 0.8 with an AUC of 0.77. (C) DMS cutoff of ≥ 1.96 mm predicted a CPR of 1.0 with an AUC of 0.77.
Summary chart displaying the thresholds obtained by receiver operating characteristic analysis for CPR and DMS in predicting hemodynamic impairment. The ranges of CPR and DMS corresponding to specific CVR (< 10%) and CBF-AI (< 0.8) values are highlighted to show progression towards severe hemodynamic impairment. All thresholds were derived using ROC curve analysis, and the optimal cut-off values were identified based on the Youden index to maximize sensitivity and specificity. CPR, carotid pressure ratio; DMS, diameter of the most severe stenosis; CVR, cerebral vascular reserve; CBF-AI, cerebral blood flow asymmetry index.
Illustrative case
A 76-year-old man presented with a symptomatic right ICA stenosis due to a minor stroke. Preoperative CBF-AI was 0.79, and CVR was 3.0%. The DMS was 1.41 mm, and the degree of stenosis based on the NASCET method was 78%. The preoperative CPR in this patient was reduced to 0.73, but it improved to 1.02 after CAS, indicating the resolution of the pressure gradient (Fig. 5).
A 76-year-old man with symptomatic internal carotid artery stenosis. Diffusion-weighted imaging (A) and FLAIR (B) showed findings consistent with a minor stroke. Preoperative SPECT revealed decreased cerebral blood flow (CBF) on the affected side (C: 41.6 mL/100 g/min), with a CBF asymmetry index (CBF-AI) of 0.79 and a cerebrovascular reserve (CVR) of 3% (D). Digital subtraction angiography (DSA) demonstrated a minimum luminal diameter of 1.41 mm preoperatively (E). Pressure recordings showed a clear pressure gradient between the proximal (red arrow) and distal (blue arrow) segments of the stenosis, with a carotid pressure ratio (CPR) of 0.73. After carotid artery stenting, DSA (F) showed improvement of the stenosis to 3.6 mm, and the pressure gradient disappeared, with the CPR improving to 1.02.
Discussion
Several randomized clinical trials and non-randomized clinical trials have demonstrated the effectiveness of FFR-guided coronary revascularization for FFR values < 0.809,14,15. The CPR threshold for a CVR < 10% calculated in this study was 0.80, nearly identical to the FFR threshold for coronary revascularization. This may be because, despite the different pathologies of coronary and carotid artery diseases, in both diseases, disease onset is caused by blood flow impairment due to vascular stenosis. The high correlation between CPR and CVR (R2 = 0.38) suggests that blood flow restriction in ICA stenosis may directly affect cerebral blood flow regulation. Although from a hydrodynamic standpoint, pressure difference is more directly associated with flow resistance—as described by the Hagen–Poiseuille equation and Reynolds number—we used the pressure ratio (mean distal/proximal pressure) to normalize inter-individual variability in baseline blood pressure and better represent relative perfusion gradients. This approach also aligns with the concept of FFR, which is widely applied in coronary artery disease to assess physiological lesion severity. While absolute pressure values and pressure gradients are physiologically important—particularly in relation to thresholds for cerebral autoregulation—we chose the ratio-based approach for its practical clinical applicability and ability to standardize across patients. However, we acknowledge that absolute distal pressures may provide additional insight, especially in understanding autoregulatory failure in patients with critical ICA stenosis. Future studies should further investigate the relationship between absolute pressure metrics and cerebral hemodynamics. This finding is consistent with those reported by Hosoda et al., who identified cerebrovascular reactivity as a risk factor for post-CEA hyperperfusion syndrome4, and by Kuroda et al., who observed the effect of hemodynamic disturbances on cerebral blood flow regulation16.
Receiver operating characteristic curve analysis in the present study showed that a CPR cut-off for CVR < 10% had high sensitivity (100%) and specificity (89%). The threshold values presented in this study—for example, CPR ≤ 0.80 to predict CVR < 10%, CPR ≤ 0.74 to predict CBF-AI < 0.8, and DMS ≥ 1.96 mm to predict CPR ≈1.0—were determined using receiver operating characteristic (ROC) curve analysis. The optimal cut-off points were calculated based on the Youden index, which identifies the value that maximizes the sum of sensitivity and specificity. This retrospective analysis was performed to assess whether intraoperative CPR could serve as a surrogate marker for preoperative cerebral hemodynamic impairment. Thus, this index may be useful for improving preoperative risk assessment and postoperative treatment planning. Although risk assessment ideally begins at the initial clinical consultation, the measurement of CPR is a novel technique in the context of carotid artery stenosis, and its applicability for preoperative risk stratification had not been previously established. In this study, considering ethical standards in clinical research and the lack of prior validation, we chose to perform CPR measurements intraoperatively during carotid artery stenting. This approach allowed us to safely evaluate the hemodynamic relevance of CPR without exposing patients to unnecessary preprocedural risks. Our findings suggest that intraoperative CPR can provide valuable information that may aid both intra- and post-operative management, including the prediction of cerebral hyperperfusion and the selection of stent strategy. Although CPR was measured intraoperatively, the ROC curve analysis in this study was conducted retrospectively by comparing CPR values with preoperative SPECT-based indicators such as CVR and CBF-AI. The aim was not to guide intraoperative decisions in real time, but rather to evaluate whether CPR could serve as a surrogate index for preoperative hemodynamic impairment in future practice. This retrospective correlation allows for objective threshold determination while avoiding the ethical and logistical challenges of prospective SPECT-based testing. The significant correlation between CPR and CBF-AI may reflect stenosis blood flow distribution and severity. This finding is consistent with those reported by Ohta et al., in which hyperperfusion was predicted by measuring cerebral blood flow using SPECT17. In addition, the cutoff value of CPR (< 0.74) at which CBF-AI is < 0.8 can be used to assess the risk of postoperative hyperperfusion. However, the lack of a significant correlation with CBF suggests that CBF may be influenced by other factors. CPR did not decrease in cases with decreased CBF; however, CVR was preserved in this study. Kuroda et al. reported that a reduced CBF and a normal CVR following acetazolamide administration characterize oxygen hypometabolism due to ischemia-related selective neuronal damage and a low recurrence rate of cerebral infarction with medical treatment16. CPR may be associated with functional severity and the incidence of cerebral infarction in patients with ICA disease. In addition, the cutoff value of diameter at the most severe stenosis at which CPR was 1.0 in the present study was 1.96 mm. This finding suggests that once the DMS reaches approximately 1.96 mm, the transluminal pressure gradient resolves (CPR = 1.0), and further dilation may not yield additional hemodynamic improvement. Thus, beyond this threshold, the minimum vascular diameter may no longer be a limiting factor for cerebral perfusion. These results may be useful in assessing the effectiveness of staged angioplasty18. Intravascular pressure can be easily measured using a pressure wire with a pressure sensor attached to the micro-guidewire used in CAS; thus, it may be used as an alternative to SPECT examination to reduce patient burden and medical costs. Compared to the Gee OPG method, which requires separate measurement of ophthalmic artery pressure, the pressure-wire technique is more practical because it is seamlessly integrated into the CAS procedure and does not impose additional burden on the patient19. Although breath-holding tests can be used as a simpler alternative to assess cerebrovascular reactivity, they are generally less reproducible and lack quantitative accuracy compared to acetazolamide challenge, which remains the gold standard for CVR evaluation20. In this study, CPR was safely and successfully measured using a pressure wire, with no additional intraoperative complications, thus demonstrating that CPR is a minimally invasive and useful diagnostic tool. A previous study also reported that hemodynamic assessment of intracranial large- vessel stenosis using the pressure wire technique is a safe, feasible, and useful method for assessing the functional severity of stenosis21. While the initial indication for CAS is typically determined preoperatively, intraoperative CPR measurement may still provide valuable information for refining treatment strategies. For example, CPR values may inform the decision to perform staged angioplasty, guide blood pressure management to mitigate hyperperfusion risk, or influence the selection and deployment of stents. Given the successful use of FFR measurements in coronary artery disease7,9,14, CPR may also play an important role in determining the treatment options for CAS (Supplementary information).
Limitations
This single-institution study included only 25 cases. Moreover, the study population was biased towards cases with mild cerebral hemodynamic impairment, in which CBF and CVR were maintained within the normal ranges. These limitations affect the generalizability of the results. Therefore, large-scale multicenter studies conducted at other institutions are required. Additionally, the study participants were limited to those with certain conditions (e.g., unilateral carotid artery stenosis and exclusion of retreatment cases), limiting the applicability of these findings to other cases (e.g., bilateral and tandem lesions). Although correlations among CPR, CVR, and other parameters have been demonstrated, their causal relationships have not been clarified. Further investigations into how CPR contributes to clinical outcomes are required. Although we intended to focus on patients with moderate to severe ICA stenosis, the scatter plot includes both pre- and post-procedural data to illustrate the range of CPR and stenosis changes. Limiting the analysis to only preoperative values would have reduced the number of analyzable data points, compromising statistical power. We acknowledge this as a limitation of the study design.
Conclusions
The results of this study demonstrated the safety and reliability of CPR measurement using a pressure wire in CAS. Moreover, these measurements are significantly associated with preoperative CVR and CBF-AI. CPR is a useful indicator for predicting a decline in CVR, with a cutoff value of ≤ 0.80 showing high sensitivity and specificity. Furthermore, CPR measurement using a pressure wire is a safe, physiologically informative, and comparatively minimally invasive technique for evaluating intraoperative and postoperative cerebral hemodynamics. Unlike conventional modalities such as SPECT or PET, this method avoids ionizing radiation to the brain and does not require pharmacologic vasodilators such as acetazolamide, which may carry risks of adverse effects. Therefore, pressure ratio measurement can offer a safer and more accessible alternative for selected patients, especially when conventional imaging is contraindicated or unavailable. Given the success of FFR-guided treatment for coronary artery disease, CPR measurements may also play an important role in decision-making for treatment planning for carotid artery disease.
Data availability
The datasets used during the current study available from the corresponding author on reasonable request. All data generated or analyzed during this study are included in this published article.
Abbreviations
- CBF:
-
Cerebral blood flow
- CBF-AI:
-
Asymmetry index for cerebral blood flow
- CAS:
-
Carotid artery stenting
- CEA:
-
Carotid endarterectomy
- CPR:
-
Carotid pressure ratio
- CVR:
-
Cerebral vascular reserve
- FFR:
-
Fractional flow reserve
- ICA:
-
Internal carotid artery
- SPECT:
-
Single-photon emission computed tomography.
- DMS:
-
The diameter at most stenosis site
References
Ogasawara, K. et al. Intracranial hemorrhage associated with cerebral hyperperfusion syndrome following carotid endarterectomy and carotid artery stenting: retrospective review of 4494 patients. J. Neurosurg. 107, 1130–1136. https://doi.org/10.3171/JNS-07/12/1130 (2007).
Isozaki, M. et al. Silent ischemic lesion laterality in asymptomatic internal carotid artery stenosis relates to reduced cerebral vasoreactivity. Surg. Neuro. Int. 8, 6. https://doi.org/10.4103/2152-7806.198733 (2017).
Okazawa, H. et al. Can the detection of misery perfusion in chronic cerebrovascular disease be based on reductions in baseline CBF and vasoreactivity?. Eur. J. Med. Mol. Imaging 34, 121–129 (2007).
Hosoda, K. et al. Cerebral vasoreactivity and internal carotid artery flow help to identify patients at risk for hyperperfusion after carotid endarterectomy. Stroke 32, 1567–1573. https://doi.org/10.1161/01.str.32.7.1567 (2001).
Imaizumi, M. et al. Detection of misery perfusion with split-dose 123I-iodoamphetamine single-photon emission computed tomography in patients with carotid occlusive diseases. Stroke 33, 2217–2223. https://doi.org/10.1161/01.str.0000027638.19392.7e (2002).
Hartkamp, N. S. et al. Intracerebral steal phenomenon in symptomatic carotid artery disease. J. Neuroradiol. 46, 173–178 (2019).
Paolucci, L. et al. Fractional flow reserve-guided coronary revascularization: evidence from randomized and non-randomized studies. Diagnostics 12, 2659. https://doi.org/10.3390/diagnostics12112659 (2022).
Kataoka, H. et al. Results of prospective cohort study on symptomatic cerebrovascular occlusive disease showing mild hemodynamic compromise [Japanese extracranial-intracranial bypass trial (JET)-2 study]. Neurol. Med. Chir. (Tokyo) 55, 460–468. https://doi.org/10.2176/nmc.oa.2014-0424 (2015).
De Bruyne, B. et al. Fractional flow reserve-guided PCI versus medical therapy in stable coronary disease. N. Engl. J. Med. 367, 991–1001. https://doi.org/10.1056/NEJMoa1205361 (2012).
Timaran, C. H. et al. Reporting standards for carotid interventions from the Society for Vascular Surgery. J. Vasc. Surg. 53, 1679–1695. https://doi.org/10.1016/j.jvs.2010.11.122 (2011).
North American Symptomatic Carotid Endarterectomy Trial Collaborators, Barnett HJM, Taylor DW, et al. Beneficial effect of carotid endarterectomy in symptomatic patients with high-grade carotid stenosis. N. Engl. J. Med., https://doi.org/10.1056/NEJM199108153250701. (1991).
Kim, K. M. et al. Quantitative mapping of basal and vasareactive cerebral blood flow using, split-dose 123I-iodoamphetamine and single photon emission computed tomography. Neuroimage 33, 1126–1135. https://doi.org/10.1016/j.neuroimage.2006.06.064 (2006).
Iida, H. et al. Quantitative mapping of regional cerebral blood flow using iodine-123-IMP and SPECT. J. Nucl. Med. 35, 2019–2030 (1994).
Xaplanteris, P. et al. Five-year outcomes with PCI guided by fractional flow reserve. N. Engl. J. Med. 379, 250–259. https://doi.org/10.1056/NEJMoa1803538 (2018).
van Nunen, L. X. et al. Fractional flow reserve versus angiography for guidance of PCI in patients with multivessel coronary artery disease (FAME): 5-year follow-up of a randomized controlled trial. Lancet 386, 1853–1860. https://doi.org/10.1016/S0140-6736(15)00057-4 (2015).
Kuroda, S. et al. Reduced blood flow and preserved vasoreactivity characterize oxygen hypometabolism due to incomplete infarction in occlusive carotid artery disease. J. Nucl. Med. 45, 943–949 (2004).
Ohta, T. et al. Prediction of cerebral hyperperfusion after carotid artery stenting by cerebral angiography and single-photon emission computed tomography without acetazolamide challenge. Neurosurgery 81, 512–519. https://doi.org/10.1093/neuros/nyx041 (2017).
Uchida, K. et al. Experience of staged angioplasty to avoid hyperperfusion syndrome for carotid artery stenosis. Neurol. Med. Chir. (Tokyo) 55, 824–829. https://doi.org/10.2176/nmc.oa.2015-0102 (2015).
Ricotta, J. J. et al. Use of oculopneumoplethysmography (OPG-Gee) following carotid endarterectomy. J. Vasc. Surg. 2, 415–418 (1985).
Zhao, M. Y. et al. Reproducibility of cerebrovascular reactivity measurements: A systematic review of neuroimaging techniques. J. Cereb. Blood. Flow. Metab. 42, 700–717 (2022).
Han, Y. F. et al. Severity assessment of intracranial large artery stenosis by pressure gradient measurements: A feasibility study. Catheter. Cardiovasc. Interv. 88, 255–261. https://doi.org/10.1002/ccd.26414 (2016).
Acknowledgements
This study was supported by JSPS KAKENHI (grant number: 24K10907).
Author information
Authors and Affiliations
Contributions
M.I., H.A., and K.K. performed the statistical analysis and wrote the main manuscript text. K.M., A.A., and M.K. contributed to data acquisition related to the intervention procedures. H.O. supervised the statistical analysis and data collection regarding the SPECT imaging. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Isozaki, M., Matsuda, K., Akazawa, A. et al. Usefulness of intravascular pressure ratio measurement for cerebral hemodynamics in carotid artery stenting patients. Sci Rep 15, 29874 (2025). https://doi.org/10.1038/s41598-025-15939-0
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-15939-0