Abstract
In the Brazilian Amazon, cases of acute Chagas disease (ACD) are mostly associated with the consumption of açaí juice contaminated with Trypanosoma cruzi have become frequent. A large proportion of diagnosed patients and exposed individuals after outbreaks are not followed-up clinically since they reside in the remote interior of the state of Amazonas with limited access to healthcare services. This study aims to conduct serological surveillance of populations exposed to T. cruzi in orally transmitted ACD. We evaluated 66 individuals with a history of diagnosis and treatment (G1), and 49 individuals without proven infection after exposure to outbreaks (G2). Serological surveillance was performed using 3 commercially available tests and 2 genotype-specific tests using antigens from T. cruzi strains TcI and TcIV. Among the G1, 34.8% presented serological cure whereas 6.1% in G2 presented seroconversion for IgG anti-T. cruzi while 22.5% (G2) had indeterminate results due to discordance between IIF and ELISA tests, which use different principles. Genotype-specific ELISA (TcI-TcIV) tests showed detection rates between 30.3 and 31.8% in G1, and 14.3–22.4% in G2. These results highlight the importance of follow-up serological surveillance among Amazonian populations exposed to ACD outbreaks. It also underscores the need for more sensitive serological tests with greater agreement for to ensure accurate diagnosis and monitoring of populations with a history of exposure.
Similar content being viewed by others
Introduction
Chagas’ disease (CD) is a parasitic disease predominant in Latin America, but also found in other continents due to human migration1. The disease is caused by Trypanosoma cruzi, a flagellated protozoan with a complex biological cycle involving a variety of mammalian species2 and triatomines (Hemiptera) insects, popularly known as kissing bugs3,4. The genetic diversity results from the historic genetic exchange between ancestral strains5,6,7. Currently, seven T. cruzi genotypes, known as discrete typing units (DTU), have been identified: TcI to TcVI, and TcBat8,9.
In humans, infection with T. cruzi can occur through various routes1,10 with the incubation period varying depending on the transmission route. Exposed individuals may develop clinical manifestations in the acute phase of CD, which can be diagnosed through thick blood smear microscopy, as high parasitemia is typically evident. The chronic phase of CD, approximately 8 weeks post-infection, is diagnosed by detecting anti-T. cruzi IgG antibodies, through serological tests1,10. Confirmation requires reactivity in at least two tests based on different principles10,11. Enzyme-linked immunosorbent assay (ELISA), indirect immunofluorescence (IIF) and indirect hemagglutination (IHA) are the most commonly used serological test methods. With good quality kits, these tests can accurately diagnose approximately 98% of the samples. Positive results indicate either a past exposure or an active infection, while negative results suggest the absence of T. cruzi infection or may indicate a cured infection following treatment12,13.
Currently, the Brazilian Amazon has the highest number of acute Chagas disease (ACD) cases linked to outbreaks, primarily due to oral transmission. The main source of infection is the consumption of contaminated açaí juice14,15,16. Since 2004, outbreaks in Amazonas state linked to açaí consumption have been recorded and often affected family groups or close-knit communities16. The initial diagnosis is made early through thick smear microscopic examination during malaria surveillance17. However, most patients receive no clinical follow-up after ACD treatment, because they live in remote areas without access to healthcare service. Additionally, the region has no established programs for long-term medical monitoring of CD infected and exposed individuals16.
Amazonas state has 2 predominant T. cruzi genotypes in circulation. There’s the T. cruzi TcI genotype, reported in isolated ACD cases (vectors transmission presumed), chronic CD cases, as a well in vectors and in wild reservoirs18,19. There’s also the T. cruzi TcIV detected in acute cases associated with oral transmission from outbreaks16,20 and in domestic dogs21.
Inconsistencies in the available commercial serological tests have also been observed, further making confirming a chronic CD diagnosis difficult. Commercial diagnostic kits used in Amazonas often show high discordance, resulting in accuracy levels well below 98%22,23. These discrepancies can be caused by the use of T. cruzi antigens from regions other than those circulating in localities of the suspected cases being tested24,25. Low accuracy tests raise the risk of incorrect results thus undermining serological data essential for medical decision-making and patient follow-up.
This study aims to conduct serological surveillance of populations exposed to T. cruzi in orally transmitted ACD. Given the limitations of commercially available serological tests in the Amazon region, it also seeks to evaluate the performance of in-house serological test prototypes developed using antigens from T. cruzi TcI and TcIV, the strains circulating in the outbreak areas.
Results
Study participants
A total of 115 participants with a history of T. cruzi infection or exposure through oral transmission were identified during the active search. These individuals were native to the five municipalities affected by the outbreaks. In all outbreaks, T. cruzi was successfully isolated, and genotyped. Genotyping analysis confirmed the TcIV genotype in each case. Of the total participants, 66/115 (57.4%) were assigned to Group 1 (G1), consisting of individuals with a history of ACD and submitted to specific treatment. The remaining 49/115 (42.6%) were assigned Group 2 (G2), comprising individuals with a history of exposure to contaminated food but with no confirmed infection or clinical signs and symptoms at the time of the outbreak (Table 1).
In G1, the majority of participants were male (34/66; 51.5%), while in G2, the majority were female (32/49; 65.3%). Most individuals in both groups were adults aged 20–59 years at the time of the active search (65.1% in G1; 71.4% in G2). Among the G1 individuals, 58/66 (87.9%) were diagnosed with T. cruzi infection through a positive thick smear examination, while the remaining 8/66 (12.1%) were diagnosed based on positive IgM serology for T. cruzi antigens or IgG seroconversion, along with characteristic signs and symptoms of ACD. Risk behaviors associated with T. cruzi exposure, such as entering forested areas, consuming palm fruit, and eating bushmeat, were reported by over 60% of participants from either group. In contrast, less than 25% of participants in either group reported seeing triatomines in their homes or having a history of blood transfusion. The prevalence of previous leishmaniasis infection was below 5% in both groups (Table 1). A smaller proportion of G2 were involved in outbreaks that occurred between 6 and 10 years ago (12.2%), or over 10 years ago (0.0%), compared to those in more recent outbreaks (Table 1).
Serological results
The criteria for considering serological cure in G1 and seroconversion in G2 was seroreactivity in two tests with different principles (IFF + any other ELISA), following the Brazilian consensus on Chagas disease. Among the G1 individuals, 34.8% achieved serological cure following ACD treatment, while 65.2% remained IgG anti-T. cruzi positive over time, even after treatment following the outbreaks. In G2, 6.1% of individuals exhibited seroconversion for anti-T. cruzi antibodies, suggesting presumed CCD. Additionally, 71.4% were classified as seronegative (non-carriers of IgG anti-T. cruzi) indicating no prior exposure to T. cruzi antigens. The remaining 22.5% had indeterminate results due to discordance between serological tests with different working principles (IIF + ELISA) (Fig. 1A). Notably, only 5/115 (4.3%) were under medical supervision for CD.
Anti-T. cruzi IgG antibodies were detected in 3/66 (4.5%) of G1 participants, and in 9/49 (18.4%) of G2 exhibited exclusively through genotype-specific assays (ELISA-TcI and ELISA-TcIV). Notably, despite confirmed infection with T. cruzi TcIV in G1, there was no higher reactivity for ELISA-TcIV compared to other tests. Contrastingly, in the G2 group, 5/49 (10.2%) participants were identified as seropositive exclusively through ELISA-TcIV (Fig. 1B).
Among the commercial tests, IIF demonstrated the highest detection rate in G1 (50%), followed by ELISA-B (39.4%) and ELISA-W (30.3%). In G2, the detection rates were significantly lower: IIF (8.1%), ELISA-B (2.0%), and ELISA-W (4.1%).
To assess the effectiveness of serological assays using T. cruzi antigens derived from parasite genotypes circulating in the outbreak regions, the performance of standardized genotype-specific tests (ELISA-TcI and ELISA-TcIV) was also evaluated. Among G1, the detection rates for ELISA-TcI (31.8%) and ELISA-TcIV (30.3%) were comparable to those observed in commercial ELISA tests. However, among the G2, genotype-specific tests demonstrated a higher frequency of reactive results (ELISA-TcI: 14.3%, ELISA-TcIV: 22.4%) compared to commercial ELISA assays (Supplementary Table 1). When considering the commercial assays, the overall reactivity rate in G1 for any of the 3 tests was significantly higher than that of the 2 genotype-specific assays (60.6% vs. 34.8%, p < 0.05). Conversely, in G2, the commercial assays exhibited a lower detection rate (10.2%) compared to genotype-specific tests (24.5%) (Fig. 1B) (Supplementary Table 1).
Serology results for Chagas disease. A) Frequency of G1 participants with serological cure, or participants with anti-T.cruzi antibodies. Frequency of G2 participants; those without anti-T.cruzi antibodies, those who presented seroconversion with samples collected in the active search (presumed CCD), and those with indeterminate results due to divergence between the tests. B) Distribution of reactive test results in at least one serological test. The comparison of serological tests’ results (ELISA-B, IIF, ELISA W, ELISA-TcI, and ELISA-TcV) with samples from group 1 and group 2. Each column represents 1 participant.
A higher agreement was observed between between the ELISA-TcI and ELISA-TcIV tests (Kappa index [KI]: 0.749), followed by the ELISA-B and IIF tests (KI: 0.700), compared to other test pairings. Most test comparisons fell within the fair agreement category (KI range: 0.292–0.403), while a few demonstrated moderate agreement. The lowest agreement was noted between IIF and ELISA-TcIV (KI: 0.292) (Table 2).
Due to discrepancies between the ELISA-TcI and ELISA-TcIV tests and the commercial assay kits, Western blot (WB-TcI and WB-TcIV) was used to determine the specific antigenic fractions recognized by antibodies present long after the ingestion of T.cruzi-contaminated food. This analysis included both individuals with a history of ACD (G1) and those without the diagnosis (G2). A representative sampling of sera from G1 (n = 22) and G2 (n = 07) revealed that the primary antigens recognized were located in the 70k −100 kDa range for both soluble TcI and TcIV antigens (Fig. 2).
Western blot representative of the immunodominant fractions present in the soluble antigens TcI or TcIV. Uncropped Blots image at Supplementary Fig. 2.
To evaluate whether the reactivity indices of the quantitative ELISAs were associated with the time of outbreak occurrence, a correlation analysis was performed between these two variables. Although statistically significant, the observed positive correlations between time and RI were weak (r < 0.5; Supplementary Fig. 1). Additionally, when comparing the ELISA reactivity indices across participant groups categorized by outbreak location and date, the Coari-2007 group exhibited significantly higher reactivity than the Labrea-2018 group across quantitative ELISAs (p < 0.05). Significant differences were observed between Coari-2007 and Labrea-2022 for the ELISA-W and ELISA-TcI tests (p < 0.05), and between Coari-2007 and Carauari-2015 (p < 0.05) (Fig. 3).
Reactivity index (RI) of antibodies specific to T. cruzi antigens in different serological tests A) ELISA-W; B) ELISA-TcI; C) ELISA-TcIV). Serum samples were separated by the location and time of the outbreak occurrence. Each point represents one participant. For each group, the central horizontal line represents the median RI, and the lower and upper lines represent the interquartile ranges (25% and 75%, respectively). * p < 0.05; ** p < 0.01.
Discussion
Chagas disease represents a major health problem in Latin America, as it significantly affects vulnerable populations living in rural areas, exposed to the T. cruzi cycle, and with limited access to health care. For a long time, CD was not given priority it deserved in public health programs despite its prevalence and associated morbidity. Initially, difficulties in the diagnosis often undermined its existence and visibility, due to the fact that it generally affects the most vulnerable segment of the population, whose needs are not understood or heard by policy makers1,26.
For decades, there has been emphasis on the need for active surveillance of undiagnosed CD cases. Experts have also highlighted that CD transmission in the Amazon has unique peculiarities, that require the adoption of a surveillance model different from that used in the country’s traditionally at-risk areas27. Currently, this region reports the highest number of acute cases, particularly from oral transmission14 with the state of Amazonas ranking third in number of reported cases.
In this context, the main challenge was monitoring patients after diagnosis and treatment, considering that majority of the affected population predominantly live in remote, hard to access rural areas, with limited access to healthcare services. Although they were diagnosed and treated early, most did not undergo clinical follow-up compared to the 7% who were followed up after treatment. As a result, the evolution of most of these individuals remains unknown16,28,29,30,31. Given the lack of surveillance to ACD-related outbreaks, our data demonstrated that prolonged monitoring of exposed individuals is necessary, not only for individuals diagnosed with ACD, but also for those exposed.
In this study, serology data indicated a serological cure in 34.8% of the patients treated, and 71.4% of those exposed to contaminated açaí had negative results in all tests. According to the Ministry of Health10 serological negativity in two tests employing different principles is the criterion used to evaluate the cure of ACD in patients. However, the lack of a gold standard method makes the interpretation of the evaluation difficult10,12,32,33. The constant divergence in sensitivity and specificity of the commercially available serological kits has been a recurring problem, hindering accurate and timely diagnosis. Although easily available, these kits are not produced using the T. cruzi antigens circulating in the region.
The serological reactivity observed in both the treated and exposed individuals demonstrates the importance of serological surveys to understand the progression of acute CD to the chronic phase, or even to detect CD in those who evolved to chronic forms without having manifested an acute condition, according to the criteria established for diagnosing the disease1,10.
Anti-T. cruzi antibody levels may gradually decline within four years after ACD treatment, eventually falling below the cutoff value13. However, studies by Pinto et al. (2013) and Diaz-Bello et al. (2021) demonstrated that 73–86% of evaluated cases exhibited persistent serological reactivity12,32. In this study, 65.2% of the patients remained seropositive, and of these, including 41% who had been diagnosed with ACD more than 10 years ago. This indicated no clear no inverse relationship between the time since the outbreaks and a decrease in serological reactivity index for CD.
Further, a lack of clinical follow-up of individuals exposed to the ACD outbreaks raises important questions and hypothesis. The most pertinent is the possibility of therapeutic failure, particularly among those who tested positive for anti-T. cruzi IgG despite them reporting completion of treatment. Interestingly, there were exposed participants who presented anti-T. cruzi IgG, without demonstrating symptoms of acute disease. It is important to highlight that these individuals likely continued to be exposed to the disease transmission cycle, through work activities or eating habits, in their daily routines, implying the possibility of reinfection.
This study combined conventional and in-house methodologies to enhance sensitivity in anti-T. cruzi IgG detection. However, discrepancies in the serological test performances persisted. This underscores the ongoing challenge of selecting diagnostic tests with both high sensitivity and specificity for reliable CCD diagnosis within this region. Tests employing total antigens tend to be more sensitive than those based on recombinant antigens, however they also exhibit higher cross-reactivity25,34. In this context, preliminary results from Western blot analysis using TcI and TcIV total antigens may contribute to the future development of more specific serological assays tailored for CD diagnosis in the Amazon, where disease dynamics differ significantly from other regions of Brazil. Considering the risk of TcI and TcIV mixed infections35this in house test is versatile for both DTUs.
Study Limitations.
This study had a number of limitations. One limitation was the lack of data on clinical follow-up, as already described in the discussion. Another critical issue in the serological diagnosis of CCD is the potential for cross-reactivity with other parasitic infections, a well-documented challenge34,36,37. The antigenic similarity between T. cruzi and other trypanosomatids, such as Leishmania spp., both of which are endemic in the Amazon region38can lead to the production of antibodies that recognize antigens from both protozoa. Although only a small percentage of participants in our two groups reported a history of leishmaniasis, the possibility of asymptomatic Leishmania infection cannot be ruled out. Such infections could induce antibody production with cross-reactive potential, complicating the interpretation of serological tests for Chagas disease.
Final considerations
Chagas disease remains a significant public health concern in the Amazon region, yet many aspects of its epidemiology and transmission dynamics remain poorly understood and require further investigation. The states with the highest reported cases of oral transmission include Pará, Amapá, and Amazonas, with sporadic cases also recorded in other states39. These findings highlight the emergence of Chagas disease as a growing public health issue in the Brazilian Amazon region.
Our study findings underscore the urgent need for improved serological diagnostic tools and long-term clinical follow-up for CD affected individuals. Enhanced clinical and serological surveillance extend beyond individuals who developed acute disease encompassing also those exposed to transmission cycles during outbreaks but who remained asymptomatic. This approach would provide pivotal insights into the long-term health status of exposed individuals and help determine whether asymptomatic cases progress to chronic Chagas disease over time.
Methods
Study design, study population and definitions
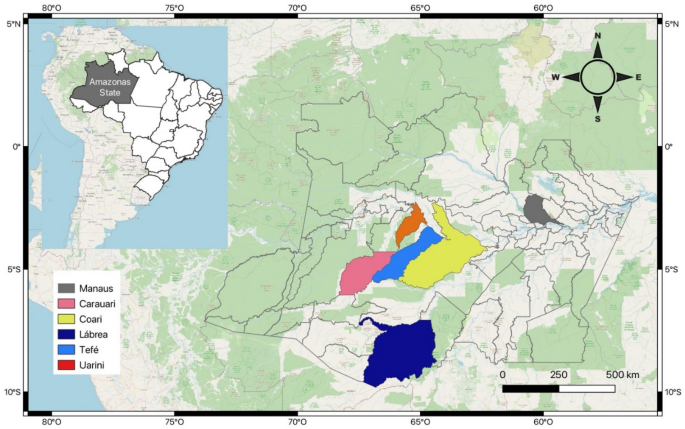
This serological survey was conducted between July 2022 and June 2023 to identify individuals affected by CD outbreaks linked to oral transmission through the consumption of açaí juice (Euterpe spp.). Active case finding was employed for comprehensive participant identification. These outbreaks occurred in five municipalities in the state of Amazonas (Carauari, Coari, Lábrea, Tefé, and Uarini) between 2004 and 2022 (Fig. 4). All individuals exposed/infected during these outbreaks received medical care from the CD service team at FMT-HVD, a national tertiary reference center for tropical and infectious diseases. During active case-finding visits, a structured questionnaire was administered to collect sociodemographic data (sex, age), clinical history (previous leishmaniasis diagnosis, history of blood transfusion), and epidemiological/behavioral information (forest exposure, ability to identify triatomines at home, consumption of palm fruits and bushmeat).
Map of the state of Amazonas highlighting the five municipalities where the study was conducted. The ACD outbreaks peer municipality resulting from açaí ingestion occured between 2004 and 2021: Tefé (outbreak in 2004), Coari (outbreak in 2007), Carauari (outbreaks in 2001 and 2015), Lábrea (outbreaks in 2018 and 2022), and Uarini (outbreaks in 2019 and 2021). The map was created using QGIS 3.32 software.
Based on retrospective medical record analysis, participants were categorized into two groups according to the inclusion criteria:
Group 1 (G1): Individuals who, during the outbreak of oral transmission, were diagnosed with T. cruzi infection. Diagnosis was confirmed through thick smear examination, positive IgM serology (or IgG seroconversion), along with symptoms consistent with ACD. All participants in this group were treated with benznidazole, experiencing clinical improvement post-treatment.
Group 2 (G2): Individuals who were exposed, took the same contaminated food responsible for the outbreak cases, but did not present any clinical or laboratory indications of a T. cruzi infection.
The non-inclusion criteria for the study included: individuals diagnosed and treated during outbreak events, and unwilling participate in the serological survey.
Participants in G1 were assessed for serological cure following treatment. Serological cure was defined as having non-reactive results in all conventional serological tests (IIF, ELISA-B and ELISA-W) and in the in-house species-specific tests (ELISA-TcI and ELISA-TcIV) using serum samples collected during the active search.
For G2, participants were classified based on seroconversion. Seroconversion was defined as reactive serological results in at least two tests with different principles (ELISA + IIF), using serum samples from the active search, indicating presumed CCD. All participants were also asked whether they were receiving medical follow-up for CD.
Sample collection
During the active search, 10 mL blood were collected from each participant into vacutainer tubes without anticoagulant. The samples were then transported to the Nelson Fé Entomology Unit at Fundação de Medicina Tropical Doutor Heitor Vieira Dourado (FMT-HVD) in Manaus, AM. Here, the coagulated blood was centrifuged at 1200 g for 10 min at room temperature. The resulting serum sample was separated, aliquoted, and stored at −20 °C until when needed.
Commercial serological tests
All commercial tests were performed according to the manufacturers’ recommendations. The serological procedures for screening the samples were initially performed at the Central Laboratory of Amazonas (LACEN), where serum samples were tested using two commercial kits: the ELISA Kit Chagas Bioclin (ELISA-B), that use recombinant antigens, with a sensitivity of 99.9% and specificity of 99.3%; and the Indirect Immunofluorescence (IIF) CHAGAS kit – Biomanguinhos, the manufacturer did not provide the specific DTU used in this test, with sensitivity and specificity of ≥ 90%. Both assays provided only qualitative results (reactive or non-reactive). In parallel, at FMT-HVD, the same samples were tested using an additional commercial assay ELISA kit Chagatest v.4.0, Wiener lab (ELISA-W), that use recombinant antigens (SAPA, 1, 2, 13, 30 and 36), with sensitivity range of 99.1–99.9% and specificity of 98.3–99.7%. This test provided quantitative results (reactivity index - RI).
Antigen preparation for in-house assays
The T. cruzi TcI or TcIV antigens used here were produced as described by Cervantes-Landín et al. (2014)40 with some modifications. The T. cruzi TcI or TcIV strains isolated from blood samples of patients diagnosed with acute Chagas disease (ACD) native to the Amazon were used to prepare the parasite mass.
Briefly, cryopreserved T. cruzi isolates were activated in Novy-MacNeal-Nicolle (NNN) medium thereafter cultured in Schneider medium supplemented with 20% fetal bovine serum (FBS) for the multiplication of epimastigote forms for 3 days (TcI) or 2 days (TcIV), at 28 °C. After growth of the isolates, the parasite suspension was centrifuged at 3,500 g for 20 min, discarding the supernatant. The precipitate containing the parasite biomass was washed thrice with Phosphate Buffer Solution (PBS) under the same centrifugation conditions. Approximately 6 × 105 parasitic forms were suspended in 500 µl of 10 mM TRIS-HCl buffer, pH 8.2 with protease inhibitor (12 mM EDTA, 0.1 mM leupeptin) and sonicated for 30 min (UltraCleaner 750 A). The resulting lysate was centrifuged at 12,000 g for 20 min, at 4 °C, and then the supernatant, containing soluble antigens, aliquoted and stored at −20 °C. Protein concentration was determined by the Pierce BCA Protein Assay Kit (Thermo Scientific) following the manufacturer’s instructions. The generated parasite antigens were subsequently used in ELISA-TcI, ELISA-TcIV, WB-TcI, and WB-TcIV assays.
In-house ELISA
Two non-commercial ELISA, developed and standardized at FMT-HVD, (ELISA-TcI and ELISA-TcIV) were used to evaluate the capacity to detect species/lineage-specific antibodies from the serological surveillance. For this purpose, 96-well polystyrene plates (Thermo Fisher Scientific) were sensitized with 10 µg/ml of antigen (TcI or TcIV), incubated overnight at 4 °C followed by washing with PBS containing 0.05% Tween 20 (PBS-T). Nonspecific sites were then blocked with PBS containing 10% FBS with a 2 h incubation step. After washing the wells again, 50 µl/well of serum samples diluted at 1:100 was added and incubated overnight at 4 °C. After six washing cycles, the plates were incubated for 1 h with the anti-human IgG conjugate (INVITROGEN) at a dilution of 1:500. After another wash step, the OPD (O-Phenylenediamine) substrate was added before incubating in the dark for 10 min. The reactions were stopped using 2 N HCl and absorbance readings obtained using a microplate reader (ThermoPlate) at a wavelength of 492 nm. The results were expressed as a reactivity index. Receiver Operating Characteristic (ROC) curve generated using known negative and positive control samples was used to establish cut-off for distinguishing between reactive and non-reactive field samples. Sensitivities of 92% and specificities of 91.6% were observed in both assays.
In-house Western blot
To identify the subfractions of soluble antigens recognized by ELISA-TcI and ELISA-TcIV, a representative sample from G1 (n = 22) and G2 (n = 7) was evaluated using customized Western Blot tests (WB-TcI and WB-TcIV). Here, soluble antigens were separated using 8% SDS-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to PVDF membrane in a semi-dry Power Blotter System. The membrane was then cut into strips (approximately 20 µg of antigen/strip) which were subsequently placed into individual channels for non-specific binding site blocking. The blocking was done incubating in PBS-Tween 0.05% (PBS-T) containing 5% skimmed milk for 2 h under rocking motion. The strips were the washed with PBS-T solution, before adding diluted serum samples (1:100) and incubated with agitation overnight at 4 °C. Individual strips were six times using PBS-T solution, before adding anti-human IgG conjugate (INVITROGEN) diluted in 1:500. After a 2-hour incubation under agitation, the strips underwent another wash cycle before adding the DAB substrate (Pierce™) until the bands appeared. The reaction was stopped using distilled water. The WB reactivity results were presented based on the frequency of detected band profile among participants.
Statistical analysis
Sociodemographic, epidemiological and clinical variables, as well as qualitative results of serological tests, were assessed regarding the frequency of occurrence in each of the groups G1 and G2. The age variable was categorized into age groups. Contingency analyses (Fisher’s exact test or Chi-square test) were performed to identify differences in frequencies between the groups. For each group, numerical variables such as reactivity indexes (RI; defined by the sample absorbance value/test cut-off value) were represented by median values and interquartile ranges (25% and 75%). Statistical differences between the groups were assessed by analysis of variance (ANOVA). Spearman correlations were performed between the time of outbreak occurrence and the IR for each ELISA test. A 95% confidence interval (p < 0.05) was considered for all analyses. The analysis of agreement between the tests was performed using the KAPPA index (between 0.21 and 0.4 – reasonable agreement; 0.41 and 0.6 – moderate agreement; 0.61 and 0.8 – substantial agreement; 0.81 and 1.0 – almost perfect agreement). All analyses were performed using the Graphpad PRISM program version 10.4.1.
Ethical aspects
The study was submitted to and approved by the Human Research Ethics Committee - CEP of the Fundação de Medicina Tropical Doutor Heitor Vieira Dourado (FMT-HVD), under certification CAAE: 97505318.7.0000.0005. All participants willingly provided informed and signed consent before participating in study-related procedures after receiving a detailed explanation of the study protocol. All methods were performed in accordance with the relevant guidelines and regulations, including the Helsinki Declaration.
Data availability
The data that support the findings of this study are available on request from the corresponding author MGVBG. The data are not publicly available due to their containing information could compromise the privacy of research participants.
References
Pérez-Molina, J. A. & Molina, I. Chagas disease. Lancet 391, 82–94 (2018).
Jansen, A. M., Xavier, S. C. D. C. & Roque, A. L. R. Trypanosoma cruzi transmission in the wild and its most important reservoir hosts in Brazil. Parasites Vectors. 11, 1–25 (2018).
Galvão, C. Taxonomy Triatominae - The biology of Chagas disease vectors. in Triatominae - Biology Chagas Disease Vectors 5, 15–38 (Springer, 2021).
Carcavallo, I., Jurberg, J. & Lent, H. Atlas dos vetores da doença de Chagas nas americas. (1998).
Murta, S. M. F. & Romanha, A. J. Characterization of Trypanosoma cruzi. Mem. Inst. Oswaldo Cruz. 94, 177–180 (1999).
Miles, M. et al. Do radically dissimilar Trypanosoma cruzi strains (zymodemes) cause Venezuelan and Brazilian forms of chagas’ disease? Lancet 317, 1338–1340 (1981).
Brisse, S., Barnabé, C. & Tibayrenc, M. Identification of six Trypanosoma cruzi phylogenetic lineages by random amplified polymorphic DNA and multilocus enzyme electrophoresis. Int. J. Parasitol. 30, 35–44 (2000).
Zingales, B. et al. A new consensus for Trypanosoma cruzi intraspecific nomenclature: second revision meeting recommends TcI to TcVI. Mem. Inst. Oswaldo Cruz. 104, 1051–1054 (2009).
Ramirez, J. et al. First report of human Trypanosoma cruzi infection attributed to TcBat genotype. Zoonoses Public. Health. 61, 477–479 (2014).
Pinto Dias, J. C. et al. II Consenso Brasileiro em Doença de Chagas, Epidemiol. e Serviços Saúde 25, 1–10 (2015).
World Health Organization & Panamerican Health Organization. Guidelines for Diagnosis and Treatment of Chagas Disease. (2019).
Pinto, A. Y. et al. Clinical Follow-Up of responses to treatment with benznidazol in amazon: A cohort study of acute Chagas disease. PLoS One. 8, 1–9 (2013).
Pinto, A., Ferreira, A., Valente, V., Harada, G. & Valente, S. Urban outbreak of acute Chagas disease in Amazon region of brazil: four-year follow-up after treatment with benznidazole. Rev. Panam. Salud Publica. 25, 77–83 (2009).
Secretaria de Vigilância. Em Saúde-Ministério Da Saúde-Brasil. Bol. Epidemiológico 46, 1–9 (2015).
Pacheco, L. V., Santana, L. S., Barreto, B. C., de Santos, E. & Meira, C. S. S. Transmissão oral Da doença de Chagas: Uma Revisão de literatura. Res. Soc. Dev. 10, e31910212636 (2021).
Sousa, D. et al. Acute Chagas disease associated with ingestion of contaminated food in Brazilian Western Amazon. Trop. Med. Int. Heal. 1–10. https://doi.org/10.1111/tmi.13899 (2023).
Monteiro, W. M. et al. Driving forces for strengthening the surveillance of Chagas disease in the Brazilian Amazon by training the eyes of malaria microscopists. Rev. Soc. Bras. Med. Trop. 53, 10–12 (2020).
Santana, R. A. G. et al. Trypanosoma cruzi strain TcI is associated with chronic Chagas disease in the Brazilian Amazon. Parasite 7, 1–7 (2014).
Magalhães, L. et al. Bioecological aspects of triatomines and marsupials as wild Trypanosoma cruzi reservoirs in urban, peri-urban and rural areas in the Western Brazilian Amazon. Med. Vet. Entomol. 35, 389–399 (2021).
Santana, R. A. G. et al. Oral transmission of Trypanosoma cruzi. Brazilian Amazon. 25, 25–28 (2019).
Vasconcelos, L. A. S. et al. Trypanosoma cruzi discrete typing unit TcIV implicated in a case of acute Chagas disease in a domiciliated dog in the Western Amazon. Rev. Soc. Bras. Med. Trop. 54, e0873 (2021).
da Silveira, J. F., Umezawa, E. S. & Luquetti, A. O. Chagas disease: Recombinant Trypanosoma cruzi antigens for serological diagnosis. Trends Parasitol. 17, 286–291 (2001).
Sáez-alquezar, A. et al. A Origem geográfica de Pessoas com a Doença de Chagas crônica no Brasil impacta o desempenho de testes comerciais Para IgG anti-T cruzi. Mem Inst. Oswaldo Cruz 116, 37–47 (2021).
Sánchez, B., Monteón, V., Reyes, A. & Espinoza, B. Standardization of Micro-Enzyme-Linked immunosorbent assay (ELISA) and Western blot for detection of Trypanosoma cruzi antibodies using extracts from Mexican strains as antigens. Arch. Med. Res. 32, 382–388 (2001).
Guzmán-Gómez, D. et al. Highly discordant serology against Trypanosoma cruzi in central veracruz, Mexico : role of the antigen used for diagnostic. Parasit. Vectors. 8, 1–8 (2015).
Coura, J. R., Viñas, P. A., & Junqueira, A. C. V. Ecoepidemiology Short history and control of Chagas disease in the endemic countries and the new challenge for non-endemic countries. Mem. Inst. Oswaldo Cruz. 109, 856–862 (2014).
Coura, J. R. & Junqueira, A. C. V. Surveillance, health promotion and control of Chagas disease in the Amazon Region - Medical attention in the Brazilian Amazon region: A proposal. Mem. Inst. Oswaldo Cruz. 110, 825–830 (2015).
Barbosa, M. G. V. et al. Chagas disease in the state of amazonas: history, epidemiological evolution, risks of endemicity and future perspectives. Rev. Soc. Bras. Med. Trop. 48, 27–33 (2015).
de Souza-Lima, R. Outbreak of acute Chagas disease associated with oral transmission in the Rio Negro region, Brazilian Amazon. Rev. Soc. Bras. Med. Trop. 46, 510–514 (2013).
Brito, A. K. S. B. et al. Acute micro-outbreak of Chagas disease in the southeastern amazon: a report of five cases. Rev. Soc. Bras. Med. Trop. 55, e0687 (2022).
Borborema, M. et al. Doença de Chagas urbana Em Tefé-AM: Relato de nove Casos com suspeita de Transmissão oral. Revista Da Sociedade Brasileira De Med. Tropical 38, (2005).
Díaz-Bello, Z. et al. Ten-year follow-up of the largest oral Chagas disease outbreak. Laboratory biomarkers of infection as indicators of therapeutic failure. Acta Trop 222, 1-9 (2021).
Pinto, A. Y. D. N., Valente, V. C., Valente, S. A. S., Motta, T. A. R. & Ventura, A. M. R. S. Clinical, cardiological and serologic Follow-Up of Chagas disease in children and adolescents from the Amazon region, brazil: longitudinal study. Trop. Med. Infect. Dis. 5, 1–12 (2020).
Flores-Chávez, M. et al. Comparación de técnicas convencionales y no convencionales Para El Diagnóstico de La Enfermedad de Chagas importada En España. Enferm Infecc Microbiol. Clin. 28, 284–293 (2010).
Cruz-Saavedra, L. et al. Exploring Trypanosoma cruzi transmission dynamics in an acute Chagas disease outbreak using next-generation sequencing. Parasites Vectors. 17, 1–12 (2024).
Abras, A. et al. Serological diagnosis of chronic Chagas disease: is it time for a. J. Clin. Microbiol. 54, 1566–1572 (2016).
Santos, S. et al. In-house ELISA method to analyze anti- Trypanosoma cruzi IgG reactivity for differential diagnosis and evaluation of Chagas disease morbidity. Rev. Soc. Bras. Med. Trop. 45, 35–44 (2012).
Guerra, J.A.O., Ribeiro, J. A. S., Coelho, L. I., Barbosa, M. G. V. & Paes, M. G. Epidemiologia da Leishmaniose tegumentar Na Comunidade São joão, Manaus, Amazonas, Brasil epidemiology of tegumentary leishmaniasis. Cad Saúde Pública. 22, 2319–2327 (2006).
Sistema de Informação de Agravos de Notificação- SINAN. Doença de Chagas Agudo. (2023). http://portalsinan.saude.gov.br/doenca-de-chagas-aguda
Cervantes-Landín, A. Y., Martínez, I., Schabib, M. & Espinoza, B. High molecular weight proteins of Trypanosoma cruzi reduce Cross-Reaction with leishmania spp. In serological diagnosis tests. Biomed. Res. Int. https://doi.org/10.1155/2014/365403 (2014).
Acknowledgements
The authors thank all the medical and support staff of the Fundação de Medicina Tropical Doutor Heitor Vieira Dourado (FMT-HVD), Francisca Mendes Hospital, Foundation Dr. Rosimary Costa Pinto (FVS-RCP/AM) and the Centro de Arritmias Cardíacas de Manaus. The Municipal Health Departments of the affected by the outbreaks and Fundação de Amparo à Pesquisa do Estado do Amazonas for their financial support in acquiring materials. We also recognize the immense support from the Entomology Department staff, particularly Mr. Nelson Fe (in memoriam) and Yolanda Noguth. We would also like to thank the patients who agreed to participate in this study.
Funding
This work was funded by the Amazonas Research Foundation (Fundação de Amparo a Pesquisa do Estado do Amazonas-FAPEAM) EDITAL N. 002/2018 - UNIVERSAL AMAZONAS and EDITAL N. 010/2021- CT&I ÁREAS PRIORITÁRIAS. Amazonas State Research Support Foundation – FAPEAM - NOTICE No. 038/2022 - PDPG/CAPES/FAPEAM - COORDINATOR/FINANCIAL AID.
Author information
Authors and Affiliations
Contributions
SSD, MGVBG and JAOG conceived the idea for the project. SSD, AGS and ESM designed and analyze the assays. SSD, ESM and MMM performed the assays. EIGM, DRTS, GMA and JVO collected the samples and epidemiological data. SSD and AGS analyzed the data. SCBJ, MGVBG, JAOG and KNC were involved in funding acquisition. JMBF, ARJB, AGS, MGVBG and JAOG supervised the studies. SSD, MGVBG, AGS and JAOG wrote the original draft. SCBJ, ARJB, KNC, JMBBF, YPA and LPRS edited the original draft. All authors authorized the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential competing interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Smith-Doria, S., Guerra, J.A.d., Guevara-Moctezuma, E.I. et al. Serological surveillance of orally acquired acute Chagas disease in the Brazilian Amazon using conventional and in house assays. Sci Rep 15, 35792 (2025). https://doi.org/10.1038/s41598-025-15973-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-15973-y