Abstract
New studies have revealed an association between chronic heart failure, the severity of septic shock, and the blood urea nitrogen to albumin ratio (BAR). Nevertheless, its role in congestive heart failure patients admitted to the intensive care unit remains unclear. This study aimed to investigate the association between BAR and mortality among these patients. The present study analyzed data from the MIMIC-IV (version 2.2) database, targeting patients with congestive heart failure. The study outcome was all-cause mortality within the first year after discharge. Patients were categorized into three groups—T1, T2, and T3—based on tertiles of BAR levels. To explore the relationship between BAR and mortality, Kaplan-Meier survival curves and multivariate Cox proportional hazards models, adjusted for potential confounders, were employed. Additionally, a dose-response relationship between BAR and mortality risk was evaluated using a restricted cubic spline model and threshold effect analysis. Subgroup analyses were conducted across diverse populations to assess the prognostic value of BAR. Furthermore, the predictive capabilities of blood urea nitrogen, albumin, blood urea nitrogen combined with albumin, and BAR were assessed through receiver operating characteristic analysis. The cohort comprised 4506 patients diagnosed with congestive heart failure. Kaplan-Meier curves revealed that all-cause mortality was significantly elevated in patients within the higher BAR tertiles (p < 0.001). Multivariate Cox regression analysis indicated that patients in the T2 (hazard ratio (HR): 1.20, 95% confidence interval (CI): 1.06 ~ 1.36) and T3 groups (HR: 1.37, 95% CI: 1.18 ~ 1.57) had a significant increase in mortality risk relative to the T1 group (p for linear trend < 0.001). Most subgroups showed this association, with the exception of variations of levels in creatinine, blood urea nitrogen, alkaline phosphatase, and alanine aminotransferase. Notably, the BAR demonstrated superior predictive accuracy for mortality compared to blood urea nitrogen or serum albumin alone, while exhibiting comparable performance to their combined measure. Among ICU patients with congestive heart failure, an elevated BAR was associated with an increased risk of all-cause 1-year mortality, particularly in those with less impaired liver and kidney function. Therefore, BAR may be measured to comprehensively evaluate the patients’ prognosis.
Similar content being viewed by others
Introduction
Heart failure, as a terminal stage of heart disease, is marked by dysregulated neuroendocrine system activation and is frequently associated with multi-organ damage, including hepatic and renal impairment1. Despite significant advancements in pharmacological and device-based therapies, the comprehensive management of heart failure patients remains challenging, with a 5-year survival rate of approximately 50%2. Patients experiencing severe decompensated heart failure often necessitate admission to the intensive care unit (ICU), showing a 1-year all-cause mortality rate of about 20–30%3. To facilitate clinical treatment decisions, patients may be stratified by risk, which aims to improve their prognosis4. Despite the introduction of numerous risk prediction tools in recent years, their clinical application is often hindered by their suboptimal predictive performance and complexity5. Consequently, identifying additional non-invasive risk factors holds significance. Moreover, considering the regional variations in medical settings, the identification of convenient and easily accessible risk markers is essential.
Blood urea nitrogen (BUN) and albumin (Alb) are biochemical markers that can be rapidly detected and extensively utilized in clinical practice. BUN and Alb serve as direct and indirect measures of endogenous protein synthesis and catabolism. Notably, BUN may exhibit a lower sensitivity compared to serum creatinine, but elevations in BUN can be associated with the hemodynamic alterations resulting from decreased renal perfusion and subsequent neurohormonal activation6. Furthermore, Alb not only reflects the body’s nutritional status but also the regulation of osmotic pressure, vascular permeability, acid-base balance, and inflammation processes7. High levels of Alb may indicate myocardial fibrosis and a poor prognosis in chronic heart failure patients8. Consequently, researchers have proposed a novel prognostic marker, blood urea nitrogen to albumin ratio (BAR). BAR has demonstrated significance in evaluating the risk of ICU admission and mortality in patients with chronic heart failure9, gastrointestinal bleeding in the elderly10, and septic shock patients11. Furthermore, studies have indicated that BAR possesses a higher predictive value for the prognosis of community-acquired pneumonia compared to BUN or Alb alone12. Nonetheless, whether BAR can comprehensively integrate the prognostic information provided by both BUN and Alb remains undetermined. Meanwhile, as a relatively novel biomarker, BAR’s overall prognostic value in ICU patients with congestive heart failure remains uncertain.
Therefore, a comprehensive analysis was conducted utilizing the Medical Information Mart in Intensive Care-IV (MIMIC-IV) database, investigating the potential association between BAR and 1-year all-cause mortality risk in ICU patients with congestive heart failure. Moreover, subgroup analyses were conducted to identify populations with enhanced prognostic efficacy.
Methods
Sample size calculations
The power for the endpoint all-cause mortality was calculated based on a two-sided t-test with a significance level of 5%. With a sample size of 896 or 670 subjects, respectively, the trial would achieve more than 90% or 80% power to detect a difference between low-level and high-level BAR9. In addition, 11 subgroup analyses were planned, with an adjusted significance level of 5‰. Sample sizes of 1475 or 1174 participants were required to exceed 90% or 80% power to detect the differences at this significance level, respectively.
Study population
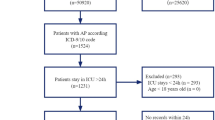
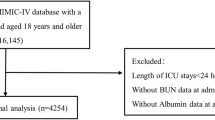
This observational study followed the STROBE guidelines. Study data were collected from MIMIC-IV v2.2, a single-center database comprising over 190,000 ICU admissions from 2008 to 2019. The diagnostic codes used in this study were from the 9th and 10th revisions of the International Classification of Diseases (Supplementary Table 1). The exclusion criteria were set: (a) age less than 18 years; (b) non-first ICU admission; (c) ICU stay less than 24 h; and (d) missing data of BUN or Alb. Based on the criteria, 4506 patients were included in the final analysis (Fig. 1).
Flow diagram of the number of study participants.
Considering that this study utilized anonymized patient data from publicly available datasets solely for secondary analysis, and that these data did not directly or indirectly identify individuals, the requirement to obtain informed consent from subjects was waived. No additional ethical review was required as the data used were derived from publicly accessible datasets.
Data acquisition and definition
Clinical data were gathered during the first 24 h after ICU admission. The variables with over 20% of missing values were discarded. Factors were collected, including age, gender, and comorbid conditions, such as chronic obstructive pulmonary disease (COPD), hypertension, and diabetes. Vital signs were recorded, including respiratory rate, heart rate, blood pressure, and saturation of peripheral oxygen (SpO2). The laboratory tests included white blood cell (WBC), neutrophil, hemoglobin, platelet, Alb, alanine aminotransferases (ALT), aspartate aminotransferases (AST), total bilirubin, alkaline phosphatase (ALP), creatinine, and BUN. In addition, the administration of inotropes or vasopressors (milrinone, phenylephrine, norepinephrine, epinephrine, vasopressin, dobutamine, dopamine), beta blockers, angiotensin-converting enzyme inhibitors/angiotensin Ⅱ receptor blockers (ACEI/ARB), furosemide, mechanical ventilation, and continuous renal replacement therapy (CRRT) was recorded. Moreover, the left ventricular ejection fraction (LVEF) was recorded due to its special role as a cardiac indicator, although data were missing in 24% of patients. Despite its essential role, B-type natriuretic peptide was excluded as data were missing in nearly 80% of patients.
The exposure was BAR, which was calculated using the following formula: BAR (mg/g) = BUN (mg/dL) / Alb (g/dL). The study outcome was all-cause mortality within one year.
Statistical analysis
Continuous variables conforming to a normal distribution were presented as mean and standard deviation, whereas the others were reported as median and interquartile range. Categorical variables were expressed as percentages. All analyses were run in R Studio (version 4.4.1), with a significance threshold at p < 0.005 in the Bonferroni-adjusted subgroup analysis, while a significance threshold of p < 0.05 was used for the rest of the analysis.
First, BAR levels were analyzed across the low, intermediate, and high-risk groups, which were categorized according to the tertiles of Acute Physiology Score Ⅲ (APS Ⅲ) and Sequential Organ Failure Assessment (SOFA).
Moreover, Kaplan-Meier survival curves stratified by BAR tertiles were employed to evaluate the risk of all-cause mortality.
To minimize confounding bias, BAR tertiles were treated as a categorical characteristic (T1 as the reference), a rank variable (one-tertile increment), and a continuous characteristic (per unit increase) in Cox proportional hazards models. These models, adjusted for various confounders, were used to calculate the hazard ratio (HR) for all-cause mortality, and a linear trend test was applied. Confounders were considered based on previous research and clinical experience, including demographics, markers of inflammation and anemia, liver and kidney function, therapy-related factors (drugs and devices), and disease severity (as indicated by SOFA and APS Ⅲ scores). In Model 0, only BAR was included. In Model 1, age, gender, neutrophil, hemoglobin, total bilirubin, and creatinine levels were adjusted. Model 2 was further adjusted for inotropes or vasopressors, ventilation, and CRRT. In Model 3, the SOFA and APS Ⅲ scores were additionally included. Additionally, these analyses were reproduced in patients with LVEF data. Specifically, LVEF was included in the analysis as a possible confounding factor.
Furthermore, restricted cubic spline (RCS) analysis was employed to investigate the dose-response effect of BAR on mortality, with threshold effect analysis performed as necessary across all the patients. Similarly, this analysis was replicated in patients with LVEF data.
Subgroup analysis and interaction testing were conducted using Model 3 to assess the prognostic value of BAR across different patient populations. These analyses considered factors such as age (≤ 65 vs. >65 years), gender, presence of hypertension or diabetes, use of inotropes or vasopressors, ventilation, and CRRT. Interaction terms, including serum creatinine × BAR, BUN × BAR, ALT × BAR, and ALP × BAR, were added to the models to evaluate the continuous interaction between these variables and BAR, with the results visualized using RCS.
In addition, differences in predictive power were compared between BAR, BUN, and albumin, using receiver operating characteristic (ROC) curves and the DeLong test.
Finally, to verify the robustness of the findings, relative risks (RRs) for BAR tertiles across both 6-month and 1-year periods were calculated using modified Poisson regression.
Results
Baseline characteristics
The study included a total of 4506 patients, providing a sufficient sample size for robust statistical inference. Table 1 presents the baseline information of all participants, stratified by BAR tertiles. Patients in the high BAR tertile exhibited elevated levels of neutrophils, RDW, hepatic impairment, renal impairment, SOFA scores, and APS Ⅲ, as well as a higher proportion of CRRT, and lower levels of hemoglobin and Alb. In addition, patients in the high BAR tertile exhibited higher rates of dopamine, norepinephrine, dobutamine, or vasopressin, and lower rates of beta blockers and ACEI/ARB usage (Supplementary Table 2). The overall incidence of endpoint events was 43.1%. Notably, patients with high BAR demonstrated increased all-cause mortality.
Relationship between BAR and markers of disease severity
As depicted in the accompanying bar graph, patients were classified into low-, intermediate-, and high-risk groups based on their SOFA and APS Ⅲ scores (Supplementary Fig. 1). Higher BAR values were observed with higher scores.
Prognostic value of BAR
Kaplan-Meier survival curves demonstrated a markedly higher 1-year all-cause mortality rate among patients with elevated BAR levels (Log-Rank test, p < 0.001, Fig. 2A). Further prognostic assessment using Cox models demonstrated a fully adjusted HR of 1.20 (95% CI: 1.06 ~ 1.36) for T2 and 1.37 (95% CI: 1.18 ~ 1.57) for T3, with a significant linear trend (p for trend < 0.001) (Table 2). When BAR was analyzed as a ranked variable, each increase in tertile corresponded to a 17% higher risk of mortality (HR: 1.17, 95% CI: 1.09 ~ 1.25). Alternatively, when considered a continuous variable, each unit increase in BAR was associated with a 1% rise in mortality risk (HR: 1.01, 95% CI: 1.00 ~ 1.02). Consistent findings were observed across Models 0, 1, and 2.
Kaplan-Meier survival curve and subgroup analysis. Cumulative incidence of all-cause mortality according to BAR quartiles (A), and Hazard Ratios and error bars delineating 95% confidence intervals from Model 3 by subgroups (B). Drugs, inotropes or vasopressors.
In accordance with the above results, Cox analysis of the patients with LVEF data revealed an HR of 1.26 (95% CI: 1.09 ~ 1.47) for T2 and 1.42 (95% CI: 1.20 ~ 1.69) for T3, showing a significant linear trend (p for trend < 0.001) (Table 2). The analysis of Models 0, 1, and 2 presented consistent trends.
Similarly, the modified Poisson regression model 3 indicated that the 1-year RR of all-cause mortality, using T1 as a reference, was 1.18 (95% CI: 1.07 ~ 1.29) for T2 and 1.28 (95% CI: 1.15 ~ 1.42) for T3, displaying a significant linear trend (p for trend < 0.001) (Supplementary Table 3). When BAR was treated as a ranked variable, each tertile increase was associated with a 6% rise in mortality risk (RR: 1.06, 95% CI: 1.03 ~ 1.10). Likewise, 6-month RR of all-cause mortality, using T1 as a reference, was 1.22 (95% CI: 1.10 ~ 1.37) for T2 and 1.38 (95% CI: 1.22 ~ 1.55) for T3 (p for trend < 0.001). Each tertile increase was associated with a 17% rise in mortality risk (RR: 1.17, 95% CI: 1.10 ~ 1.24). Similar results were observed across Models 0, 1, and 2.
In the RCS model with full covariate adjustment, BAR demonstrated a positive but nonlinear relationship with all-cause mortality risk (p for nonlinearity = 0.002) (Fig. 3A). A two-segment Cox regression model, using a recursive algorithm, pinpointed the inflection point in the BAR-mortality relationship at 12.69 mg/g (Supplementary Table 4). Below this threshold, each unit increase in BAR was associated with a 4% rise in mortality risk (HR: 1.04, 95% CI: 1.02 ~ 1.06). In contrast, above this threshold, the mortality risk remained relatively stable (HR: 1.01, 95% CI: 1.00 ~ 1.01). The likelihood ratio test confirmed that the two-segment Cox model better described the BAR-mortality relationship (likelihood ratio test, p < 0.001).
Restricted cubic spline curves. The cubic spline model of the association between BAR and risk of all-cause mortality in the all patients (A), and in patients with data of left ventricular ejection fraction (B).
In addition, in the RCS model for the patients with data of LVEF, the risk of all-cause mortality rose accordingly and nonlinearly, emphasizing the robustness of the analysis (Fig. 3B).
Prognostic effects of BAR in subgroups
Subgroup analyses and interaction tests were conducted across various groups, including variables such as gender, age, hypertension, diabetes, inotropes or vasopressors, CRRT, and mechanical ventilation. The results showed a consistent association between BAR and all-cause mortality (p for interaction > 0.005 for all) (Fig. 2B). Furthermore, interactive RCS models were employed to examine potential interactions between BAR and biomarkers reflective of hepatic and renal function, like serum creatinine (p = 0.072, relatively close to 0.005), BUN (p < 0.001), ALT (p = 0.026, relatively close to 0.005), and ALP (p < 0.001), indicating that BAR exerted a stronger prognostic impact on mortality in patients with lower biomarker levels (Fig. 4).
Interactive restricted cubic spline curves. The interactive RCS models investigated potential interactions between BAR and creatinine (A), blood urea nitrogen (B), alanine aminotransferase (C) and alkaline phosphatase (D) which are indicative of hepatobiliary and renal damage.
Comparison of predictive values
To evaluate the predictive value of Alb, BUN, their combination, and BAR for all-cause mortality, the dataset was randomly split into a training set and a test set at a 7:3 ratio. These variables were employed as predictors in the training set to develop models, and their predictive performance was compared by analyzing the areas under the receiver operating characteristic curves (AUC) in the test set (Supplementary Fig. 2). At the 180-day mark, BAR exhibited a significantly higher AUC compared to those of BUN (Δ AUC: 0.028, p < 0.001) and Alb (Δ AUC: 0.027, p < 0.001). Similarly, at the 360-day time point, the BAR demonstrated the highest AUC of 0.642 (95% CI: 0.626 ~ 0.658), which was also significantly superior to BUN and Alb. Notably, the predictive performance of BAR was comparable to that of the combination of BUN and Alb at both time points (Δ AUC: 0.003, p = 0.221 at day 180; Δ AUC: 0.002, p = 0.764 at day 360).
Discussion
This study undertook a comprehensive investigation into the independent association between the biomarker BAR and the risk of 1-year all-cause mortality in ICU patients diagnosed with congestive heart failure. This positive association was more pronounced in patients exhibiting less severe hepatobiliary and renal impairment.
The prognostic significance of BAR in cardiovascular diseases is of considerable interest. A recent study reported that elevated BAR levels are correlated with poorer functional status and more severe hemodynamics in patients with idiopathic pulmonary hypertension13. Studies have also demonstrated that elevated levels of BAR are independent risk factors associated with increased mortality in patients with heart failure14,15. Conversely, our study specifically targeted critically ill ICU patients with congestive heart failure, emphasizing the long-term risk of all-cause mortality. This focus potentially enhances the practical significance of our findings. Through rigorous analysis employing multiple statistical methods, a positive association was found between BAR and all-cause mortality risk. Notably, this association remained independent of SOFA and APS Ⅲ scores but reached a plateau after the threshold of 12.69 mg/g. Specifically, BAR levels below the threshold of 12.69 mg/g were more strongly correlated with outcomes, while changes in BAR above the threshold did not substantially affect prognosis. These results offer a more refined understanding of the association, potentially aiding clinicians in risk stratification. Furthermore, ROC curve analysis confirmed that BAR provided a significantly more accurate prediction of all-cause mortality risk compared to BUN or Alb alone. Notably, BAR was found to be equivalent to the combined predictive capacity of BUN and Alb in forecasting outcome events. This indicates that BAR not only surpasses the predictive value of BUN and Alb as individual indicators but also combines their prognostic information. Hence, BAR offers a practical assessment method for clinicians. These findings highlight the practical potential of BAR in clinical settings.
This study specifically examined the prognostic effects of BAR across various subgroups. In the ICU, congestive heart failure patients with hepatobiliary and renal impairment experience poorer outcomes. This consensus underscores the necessity for heightened clinical vigilance for these patients. Importantly, our subgroup analysis revealed that BAR demonstrated a higher prognostic stratification value in patients with lower serum creatinine, BUN, ALT, and ALP levels. This observation may be attributed to the relatively limited incremental predictive value of BAR in patients with severe hepatobiliary and renal impairment, who are already at a heightened risk of mortality. These findings indicate that BAR can effectively identify residual risk within the current evaluation framework. Consequently, BAR may attract increased attention from clinicians for high-risk patients within this cohort, potentially emerging as an integral component of the prognostic assessment system.
BAR integrates both BUN and Alb levels. Heart failure patients frequently present with concomitant kidney damage, contributing to a bidirectional relationship that exacerbates both conditions. Previous studies have demonstrated that persistently elevated BUN levels are linked to an increased risk of cardiovascular mortality and heart failure readmission, whereas normalization of BUN levels before discharge may correlate with improved clinical outcomes16. These results align with recent evidence suggesting that elevated BUN levels may indicate renal hypoperfusion rather than intrinsic renal insufficiency. This renal hemodynamic condition may be attributed to reduced cardiac output secondary to heart failure or congestion of the systemic venous system, including the renal veins9. BUN levels are hypothesized to arise from various factors, including hemodynamic alterations that lead to renal hypoperfusion and the subsequent activation of the renal sympathetic nervous system and the renin-angiotensin-aldosterone system. BUN levels are easily obtainable and serve as an indicator of this neurohormonal mechanism16. In addition, patients with heart failure frequently exhibit hypoalbuminemia, which is strongly correlated with adverse outcomes across various cardiovascular diseases17. Although heart failure itself may not directly induce hypoalbuminemia, it is commonly associated with concurrent conditions such as infection, chronic inflammation, malnutrition, liver impairment, and kidney disease, which may cause hypoalbuminemia. This condition, in turn, exacerbates pulmonary congestion and accelerates the progression of heart failure9. Considering its ability to integrate BUN and Alb, BAR emerges as a cost-effective, patient-independent tool that could play an essential role in evaluating disease severity and clinical outcomes in ICU patients with congestive heart failure. Further research is warranted to investigate its utility in assessing therapeutic effectiveness and its potential as a therapeutic target.
BAR represents a multifaceted biomarker that can optimize heart failure management by integrating assessments of renal function, systemic inflammation, microcirculatory dysfunction, and nutritional status. Elevated blood urea nitrogen levels are strongly associated with decreased renal perfusion, protein catabolism, and increased neurohormone activation18. Meanwhile, a decrease in serum albumin indicates malnutrition, hepatic dysfunction, and increased vascular permeability—all markers of systemic inflammation and endothelial dysfunction19. This dual mechanism highlights the value of BAR in simultaneously reflecting the severity and inflammatory burden of cardiorenal syndrome. In our cohort, elevated BAR levels demonstrated strong associations with adverse outcomes, which are consistent with previous studies linking BAR to increased risk of inferior vena cava congestion and long-term mortality20. The nonlinear correlations suggest that BAR’s prognostic utility may vary across different severity spectra, potentially reflecting distinct pathophysiological states ranging from compensated cardiorenal dysfunction to decompensated multi-organ failure. Critically, the overlap between heart failure and sepsis represents a particularly challenging clinical scenario in which BAR’s integrative nature offers prognostic insight. Sepsis-induced cardiomyopathy affects up to 40% of septic patients and significantly worsens prognosis when complicated by pre-existing heart failure21. These concurrent conditions can exacerbate myocardial dysfunction and hemodynamic instability22. In these patients, BAR may serve as a composite marker reflecting: (1) the severity of sepsis-induced myocardial dysfunction through elevated catabolism and reduced hepatic albumin synthesis; (2) microcirculatory dysfunction manifested by impaired tissue perfusion and increased capillary leak; and (3) the inflammatory cascade activation that bridges sepsis and heart failure pathophysiology23. Recent evidence supports BAR’s role as an inflammatory and microcirculatory marker. Elevated BAR correlates with increased inflammatory cytokines (IL-6 and TNF-α) and endothelial dysfunction markers in heart failure patients24. Furthermore, a previous study reported that elevated BAR levels parallel microcirculatory alterations assessed by sublingual microvascular flow index and perfused vessel density, suggesting its utility in monitoring tissue-level perfusion deficits25. This microcirculatory perspective is relevant given that heart failure and sepsis share common pathways of endothelial dysfunction, glycocalyx degradation, and capillary leak syndrome. Moreover, BAR’s diagnostic utility extends beyond prognostic evaluation. As demonstrated by Zhang et al., elevated BAR levels can be used to effectively distinguish heart failure from other conditions with similar presentations, including asthma and COPD26. Recent studies have also validated BAR’s predictive value in diverse clinical contexts, including febrile urinary tract infections in infants, suggesting broad applicability across different inflammatory conditions27. In conclusion, BAR emerges as a valuable integrative biomarker in critically ill heart failure patients, particularly those with concurrent sepsis or systemic inflammation. Its ability to reflect cardiorenal interactions, inflammatory burden, and microcirculatory dysfunction positions it as a practical tool for risk stratification and therapeutic guidance. Future research should focus on validating treatment algorithms based on BAR and exploring its utility in monitoring treatment response in this complex patient population.
Nevertheless, the limitations of the present study should be acknowledged. Firstly, this investigation is a post-hoc analysis, and certain confounding factors may not have been accounted for. Specifically, B-type natriuretic peptide, as a key covariate, was excluded from this analysis due to a high rate of omission in the available data. Secondly, determining whether heart failure was the primary cause of ICU admission was a challenge due to database limitations. However, congestive heart failure often presents with other critical illnesses, resulting in a complex condition, warranting ICU admission. Database limitations also prevented the analysis of other outcomes of interest, such as favorable outcomes or subsequent hospitalizations related to heart failure. Future prospective studies should address these issues. Thirdly, biomarkers collected within the first 24 h of ICU admission may only reflect the pre-treatment status of patients; thus, caution is warranted when interpreting biomarker data obtained after treatment initiation. Moreover, although subgroup analyses and multiple comparisons of Bonferroni’s correction were performed to assess the prognostic consistency of BAR in various clinical situations, these analyses remain exploratory and retrospective in nature. The results are intended to develop hypotheses for future studies. In the future, prospective studies should be conducted to validate these exploratory results.
Conclusion
The BAR is independently associated with the risk of 1-year all-cause mortality in ICU patients with congestive heart failure, exhibiting a pronounced effect in those with relatively preserved hepatobiliary and renal function. BAR may be applied as a valuable metric for a comprehensive assessment of patient prognosis and disease severity.
Data availability
The data that support the findings of this study are available from MIMIC-IV repository but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the corresponding authors upon reasonable request and with permission of MIMIC-IV repository.
References
Becher, P. M., Lund, L. H., Coats, A. J. S. & Savarese, G. An update on global epidemiology in heart failure. Eur. Heart J. 43 (32), 3005–3007. https://doi.org/10.1093/eurheartj/ehac248 (2022).
Savarese, G. et al. Global Burden of Heart Failure: A Comprehensive and Updated Review of Epidemiology. Cardiovascular Research 118(17):3272–3287. (2022). https://doi.org/10.1093/cvr/cvac013%J. Cardiovascular Research.
Foroutan, F. et al. Global comparison of readmission rates for patients with heart failure. J. Am. Coll. Cardiol. 82 (5), 430–444. https://doi.org/10.1016/j.jacc.2023.05.040 (2023).
Lee, D. S. et al. Trial of an intervention to improve acute heart failure outcomes. N Engl. J. Med. 388 (1), 22–32. https://doi.org/10.1056/NEJMoa2211680 (2023).
Chen, Z., Li, T., Guo, S., Zeng, D. & Wang, K. Machine Learning-Based in-Hospital mortality risk prediction tool for intensive care unit patients with heart failure. Front. Cardiovasc. Med. 10, 1119699. https://doi.org/10.3389/fcvm.2023.1119699 (2023).
Khoury, J. et al. Blood Urea nitrogen variation upon admission and at discharge in patients with heart failure. ESC Heart Fail. 6 (4), 809–816. https://doi.org/10.1002/ehf2.12471 (2019).
Wiedermann, C. J. Hypoalbuminemia as surrogate and culprit of infections. Int. J. Mol. Sci. 22 (9). https://doi.org/10.3390/ijms22094496 (2021).
Prenner, S. B. et al. Serum albumin is a marker of myocardial fibrosis, adverse pulsatile aortic hemodynamics, and prognosis in heart failure with preserved ejection fraction. J. Am. Heart Assoc. 9 (3), e014716. https://doi.org/10.1161/jaha.119.014716 (2020).
Lin, Z. et al. Blood Urea nitrogen to serum albumin ratio as a new prognostic indicator in critical patients with chronic heart failure. ESC Heart Fail. 9 (2), 1360–1369. https://doi.org/10.1002/ehf2.13825 (2022).
Bae, S. J., Kim, K., Yun, S. J. & Lee, S. H. Predictive performance of blood Urea nitrogen to serum albumin ratio in elderly patients with Gastrointestinal bleeding. Am. J. Emerg. Med. 41, 152–157. https://doi.org/10.1016/j.ajem.2020.12.022 (2021).
Pereira, A. G. et al. Urea to albumin ratio is a predictor of mortality in patients with septic shock. Clin. Nutr. ESPEN. 42, 361–365. https://doi.org/10.1016/j.clnesp.2021.01.007 (2021).
Smith, D. K., Kuckel, D. P. & Recidoro, A. M. Community-Acquired pneumonia in children: rapid evidence review. Am. Fam Physician. 104 (6), 618–625 (2021).
Zhang, S. et al. Blood Urea nitrogen to serum albumin ratio as a new indicator of disease severity and prognosis in idiopathic pulmonary artery hypertension. Respir Med. 227, 107643. https://doi.org/10.1016/j.rmed.2024.107643 (2024).
Zhao, L. et al. The prognostic value of blood Urea nitrogen to albumin ratio on patients with heart failure. Int. Heart J. 64 (6), 1010–1017. https://doi.org/10.1536/ihj.23-383 (2023).
Zhang, Y. Y., Xia, G., Yu, D., Tu, F. & Liu, J. The association of blood Urea nitrogen to serum albumin ratio with Short-Term outcomes in Chinese patients with congestive heart failure: A retrospective cohort study. Nutr. Metab. Cardiovasc. Dis. 34 (1), 55–63. https://doi.org/10.1016/j.numecd.2023.10.011 (2024).
Jujo, K. et al. Persistent high blood Urea nitrogen level is associated with increased risk of cardiovascular events in patients with acute heart failure. ESC Heart Fail. 4 (4), 545–553. https://doi.org/10.1002/ehf2.12188 (2017).
Mene-Afejuku, T. O. et al. The relevance of serum albumin among elderly patients with acute decompensated heart failure. J. Geriatr. Cardiol. 16 (7), 522–528. https://doi.org/10.11909/j.issn.1671-5411.2019.07.005 (2019).
Núñez, J., Miñana, G., Santas, E. & Bertomeu-González, V. Cardiorenal syndrome in acute heart failure: revisiting paradigms. Rev. Esp. Cardiol. (Engl Ed). 68 (5), 426–435. https://doi.org/10.1016/j.rec.2014.10.016 (2015).
Arques, S. Human serum albumin in cardiovascular diseases. Eur. J. Intern. Med. 52, 8–12. https://doi.org/10.1016/j.ejim.2018.04.014 (2018).
Parrinello, G. et al. Blood Urea nitrogen to creatinine ratio is associated with congestion and mortality in heart failure patients with renal dysfunction. Intern. Emerg. Med. 10 (8), 965–972. https://doi.org/10.1007/s11739-015-1261-1 (2015).
Hollenberg, S. M. & Singer, M. Pathophysiology of Sepsis-Induced cardiomyopathy. Nat. Rev. Cardiol. 18 (6), 424–434. https://doi.org/10.1038/s41569-020-00492-2 (2021).
Hiraiwa, H. et al. Clinical impact of visually assessed right ventricular dysfunction in patients with septic shock. Sci. Rep. 11 (1), 18823. https://doi.org/10.1038/s41598-021-98397-8 (2021).
Joffre, J., Hellman, J., Ince, C. & Ait-Oufella, H. Endothelial responses in sepsis. Am. J. Respir Crit. Care Med. 202 (3), 361–370. https://doi.org/10.1164/rccm.201910-1911TR (2020).
Dick, S. A. & Epelman, S. Chronic heart failure and inflammation: what do we really know?? Circ. Res. 119 (1), 159–176. https://doi.org/10.1161/circresaha.116.308030 (2016).
Jung, C., Lauten, A. & Ferrari, M. Microcirculation in cardiogenic shock: from scientific bystander to therapy target. Crit. Care. 14 (5), 193. https://doi.org/10.1186/cc9244 (2010).
Zhang, J., Zhou, L. & Zhang, Y. Diagnostic values of blood Urea nitrogen (Bun), creatinine (Cr), and the ratio of bun to cr for distinguishing heart failure from asthma and chronic obstructive pulmonary disease. Comput. Math. Methods Med. 2022 (4586458). https://doi.org/10.1155/2022/4586458 (2022).
Hyun, H., Lee, Y. H., Kang, N. Y. & Suh, J. S. Clinical relevance of blood Urea nitrogen to serum albumin ratio for predicting bacteremia in very young children with febrile urinary tract infection. Kidney Res. Clin. Pract. 43 (3), 348–357. https://doi.org/10.23876/j.krcp.22.289 (2024).
Funding
None.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by J.Y., J.W. and K.W. The first draft of the manuscript was written by J.Y. and K.W., and all authors reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Yang, J., Wang, J. & Wang, K. Independent prognostic importance of blood urea nitrogen to albumin ratio in critically ill patients with congestive heart failure. Sci Rep 15, 29971 (2025). https://doi.org/10.1038/s41598-025-16383-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-16383-w