Abstract
Papillary thyroid ultra-microcarcinoma (PTUMC), defined as a tumor ≤ 0.5 cm in size, can be considered for active surveillance management. However, lateral neck node metastases also occur in patients with PTUMCs. This study evaluated the clinicopathological features of PTUMC and compare the clinicopathological characteristics of patients with PTUMC with and without lateral neck lymph node metastasis. The study included 3,004 patients with PTUMC treated between January 2009 and June 2013; of these, 89 (3.0%) had lateral neck node metastasis. Clinicopathological characteristics including sex, age, size, operation type, tumor location, multiplicity, thyroiditis, microscopic extrathyroidal extension (ETE), and nodal status were compared between the two groups. Patients with PTUMC presented with significant male sex (p = 0.014), microscopic ETE (p < 0.001), multiplicity (p < 0.001), upper pole lesions (p < 0.001), psammomatous calcification, and central node metastasis (p < 0.001). Multivariate analysis revealed microscopic ETE (p = 0.003), upper pole lesions (p < 0.001), psammomatous calcification (p = 0.002), central neck node metastases (p < 0.001) and aggressive subtype(p < 0.001) are independent risk factors for lateral neck metastasis in PTUMC, warranting careful consideration when deciding between active surveillance and surgical intervention in this patient population.
Similar content being viewed by others
Introduction
Papillary thyroid carcinoma (PTC) is a slowly progressing cancer1,2. With the technological advancement of diagnostic modalities including ultrasonography, papillary thyroid microcarcinoma has emerged as a predominant component of thyroid cancer diagnoses, accounting for 30–50% or more of cases internationally3,4,5.
Papillary thyroid microcarcinoma (PTMC) is characterized by a tumor size of less than 1 cm, and active surveillance has recently emerged as a notable management strategy for these lesions6,7,8. According to published studies, the long-term outcomes of patients with PTMC managed with active surveillance demonstrate that, over 10 to 20 years of follow-up, the incidence of disease progression remains low, ranging from approximately 0.8–3.8%9,10,11,12.
Although the incidence of lymph node metastasis during active surveillance is very low, it is important to recognize that, in clinical practice, lateral neck lymph node involvement may be present at the time of PTMC diagnosis or may be detected during the course of active surveillance13,14.
In the present study, the authors defined papillary thyroid ultra-microcarcinoma (PTUMC) as a markedly small malignancy, measuring less than 0.5 cm in greatest dimension on imaging studies. According to the American Thyroid Association (ATA)15 and European Thyroid Association (ETA)16 guidelines, fine-needle aspiration (FNA) biopsy is generally not recommended for nodules smaller than 0.5 cm.
Given the characteristics of PTUMC, even when malignancy is suspected, diagnostic procedures such as fine-needle aspiration (FNA) may be deferred. Furthermore, if PTUMC is diagnosed, active surveillance is often pursued; however, there remains a possibility that lateral neck metastasis may develop during follow-up.
This study aims to identify risk factors for lateral neck LN metastasis in patients with PTUMC and to clarify which factors should be carefully evaluated before initiating active surveillance.
Results
Clinicopathological results
Table 1 summarizes the demographics and clinical characteristics of patients with or without lateral neck LN metastasis.
A total of 3,004 patients with PTUMC were included in this study. Majority of patients were female (81.8%, n = 2,458), with a mean age of 45.8 ± 10.5 years, ranging from 13 to 81 years. The mean tumor size was 0.40 (range: 0.05–0.5) cm.
Total thyroidectomy was performed in 1,493 patients (49.7%), and the remaining 1,511 (50.3%) patients underwent less-than-total thyroidectomy. The distribution of primary tumor sites showed a relatively balanced occurrence between the right and left lobes of the thyroid gland. In total, 1,598 (50.3%) tumors were located in the right lobe, whereas 1,333 (44.4%) were located in the left lobe. A smaller proportion of tumors (73 cases; 2.4%) were identified in the isthmus. Analysis of the vertical distribution of tumors within the thyroid lobes revealed that 807 (26.8%), 1,553 (51.0%), and 664 (22.1%) cases were located in the upper, middle and lower poles, respectively. This distribution indicates a slight predominance of tumors in the middle third of the thyroid lobe.
Multifocal tumors were identified in 316 (10.5%) patients with unilateral lesions, 306 (10.2%) patients with bilateral lesions, and 2,382 (79.3%) patients with solitary lesions. The majority of the tumor types in our study were classified as infiltrative, accounting for 2,345 (78.1%) patients. A total of 658 (21.9%) patients presented with the expansion-type disease. Microscopic capsular invasion was observed in 986 (32.8%) patients while 2,018 (67.2%) showed no evidence of it. Psammomatous calcification was observed in 717 (23.9%) patients. In our study, underlying thyroiditis was histologically confirmed in 1,024 (34.0%) patients, and the remaining 1,980 (65.9%) had no evidence of thyroiditis.
Overall, 702 patients revealed significant LN involvement. Central LNs metastasis was detected in 702 (23.4%) patients, whereas 89 (3.0%) had lateral LNs metastasis with or without central neck node metastasis. Aggressive subtypes such as diffuse sclerosing, tall cell, and solid variants were identified in the final pathology of 25 patients (0.8%).
Our study compared the clinicopathological variables between the lateral neck node negative and positive groups in patients with PTUMC. The results are presented in Table 2.
The lateral neck node-negative group comprised 2,915 (97.0%) patients, while the lateral neck node-positive group included 89 patients (3.0%). The mean age was lower in the lateral neck node positive group (44.7 ± 11.5 years) compared to the negative group (45.8 ± 10.5 years) (p = 0.318). The male sex was more prevalent in the positive group (28.1% vs. 17.9%, p = 0.014).
The mean tumor size was significantly larger in the lateral neck node positive group (0.44 ± 0.08 cm) than in the negative group (0.40 ± 0.10 cm) (p < 0.001). Microscopic extrathyroidal extension (ETE) was significantly more frequent (50.8% vs. 33.5%, p < 0.001) and multiplicity was more common (33.7% vs. 20.3%, p < 0.001) in the positive than in the negative group. The tumor location was significantly more common in the upper pole in the lateral neck node positive group than in the negative group (47.2% vs. 26.2%, p < 0.001). The histopathological features of psammomatous calcification were significantly more common in the lateral neck node-positive group (48.3% vs. 23.1%, p < 0.001). The central LNs metastasis was significantly more prevalent in the lateral neck node-positive group (64.0% vs. 22.1%, p < 0.001). Furthermore, Aggressive subtypes were observed at a significantly higher rate in patients with lateral neck node positivity compared to those without (9.0% vs. 0.6%, p < 0.001).
Predictors of lateral neck LN metastasis in patients with PTUMC
Univariate and multivariate logistic regression analyses were conducted to identify risk factors for lateral neck node metastasis in patients with PTUMC, and the results is shown in Table 3.
Univariate analysis revealed several clinicopathological factors that were significantly associated with lateral neck node metastasis in PTUMC. Male sex (odds ratio [OR], 1.795; 95% confidence interval [CI], 1.120–2.877; p = 0.015), tumor multiplicity (OR: 1.995, 95% CI: 1.274–3.125, p = 0.03), and bilaterality (OR: 2.664, 95% CI: 1.596–4.448, p < 0.001) increased the risk of lateral neck node involvement. Microscopic ETE was associated with a higher likelihood of lateral neck node metastases (OR, 2.586; 95% CI, 1.691–3.955; p < 0.001). Tumors located in the upper pole of the thyroid gland were associated with an increased risk (OR, 2.511; 95% CI, 1.643–3.839; p < 0.001). Moreover, the presence of psammomatous calcification was a significant predictor (OR, 3.108; 95% CI, 2.033–4.752; p < 0.001). The presence of central lymph node metastasis emerged as significant predictor of lateral neck node involvement. (OR, 6.269; 95% CI, 4.031–9.750; p < 0.001). Notably, Aggressive subtypes were the strongest predictor of lateral neck node involvement (OR, 16.837; 95% CI, 7.062–40.141; p < 0.001).
Multivariate logistic regression analysis identified four independent risk factors that were significantly associated with lateral neck LN metastasis in patients with PTUMC. Microscopic ETE was associated with a high risk of lateral neck LN metastasis (OR, 1.982; 95% CI, 1.269–3.094; p = 0.003). Tumors located in the upper pole of the thyroid gland were also associated with an increased likelihood of lateral neck node involvement (OR, 2.553; 95% CI, 1.634–3.989; p < 0.001). In addition, central LN metastasis was significantly correlated with a higher risk of lateral neck node involvement (OR, 4.516; 95% CI, 2.822–7.229; p < 0.001). The presence of aggressive subtypes was the strongest predictor, with an OR of 9.018 (95% CI, 3.384–24.032; p < 0.001). Additionally, pasmmomatous calcification (OR, 1.806; 95% CI, 1.134–2.897; p = 0.013). and bilaterality (OR, 2.387; 95% CI, 1.052–5.414; p = 0.037) were identified as an independent risk factor. These findings have important implications for risk stratification and management decisions in patients with PTUMC.
Discussion
Multivariate analysis revealed that microscopic ETE, upper pole location, psammomatous calcification, and central neck node metastasis were significant risk factors of lateral neck metastasis in PTUMC. These findings are similar to those reported in previously published studies on lateral neck LN metastasis in PTMC)13,14,17. In this study, we aimed to address whether PTUMCs are sufficiently indolent to warrant active surveillance or whether they are overlooked during diagnosis. Notably, approximately 3% of patients with PTUMC presented with lateral neck LN metastasis, suggesting that a subset of PTUMC cases may represent more aggressive thyroid cancers and should not be disregarded.
Previous studies reported similar risk factors for lateral neck metastases in patients with PTMC13,14,17,18. Our findings indicate that even small PTUMCs with these risk factors can metastasize to the lateral neck, highlighting the limitations of size alone as a criterion for determining the safety of active surveillance.
Our analysis demonstrated that the presence of aggressive subtypes of PTC serves as a significant risk factor for lateral neck metastasis in patients with PTUMC. The World Health Organization (WHO) classification of thyroid tumors recognizes variants such as tall cell, columnar cell, solid/trabecular, hobnail, and diffuse sclerosing types as exhibiting more aggressive clinical behavior19,20,21. However, accurately identifying these aggressive subtypes preoperatively through fine-needle aspiration biopsy (FNAB) remains challenging, thus limiting their utility in guiding clinical management prior to surgery22,23.
While the ATA guidelines suggest active surveillance as an option for PTC owing to its generally indolent nature, and many institutions have adopted this approach for PTMC8,15,16, a growing trend exists to extend this management strategy to PTUMC. This shift may lead to less aggressive FNA, and potentially result in the loss of regular follow-ups in some cases. Although only 3% of the patients with PTUMC in our study had lateral neck metastasis requiring surgery, a risk exists that some patients with progressive thyroid cancer may be misclassified as having localized disease, potentially delaying appropriate treatment.
Our findings suggest that for PTUMCs located in the upper pole or near the thyroid capsule (indicating microscopic ETE), a thorough preoperative ultrasound examination of the lateral neck LN is warranted. If LN enlargement or suspicious nodes are detected, additional imaging, such as neck CT scans should be considered for a more comprehensive analysis of lateral neck LN metastasis24,25.
While active surveillance is increasingly accepted as a safe management option for early stage thyroid cancer, applying the risk factors identified in this study for patients with PTUMC with lateral neck metastasis could help in more accurately selecting suitable candidates for this approach. Furthermore, patients with risk factors identified in univariate analysis, such as male sex, multiplicity, or bilaterality, should be thoroughly evaluated and counseled before opting for active surveillance.
Understanding these risk factors for lateral neck metastasis in patients with PTUMC can not only aid in selecting appropriate candidates for active surveillance, but also in determining the optimal surgical extent for small thyroid cancers.
Although not analyzed in the current study, emerging molecular tests are increasingly being utilized to predict the prognosis of thyroid nodules22,26,27. For patients with small tumors at risk of lateral neck LN metastasis, molecular profiling of high-risk genetic alterations, such as BRAF V600E, TERT promoter mutations, and RET/PTC rearrangements may aid in risk stratification28,29. While cost-effectiveness remains debated, recently commercialized multi-gene panels, such as ThyroSeq v3 and Afirma Gene Sequencing Classifier enable prognostic assessment using FNA samples30,31. Although not universally applicable, these tests may serve as viable adjuncts in selecting patients with PTUMC suitable for active surveillance, potentially reducing unnecessary interventions.
This study had some limitations. Despite analyzing a substantial dataset, the study was retrospective in nature and compared a small number of patients with lateral neck LN metastasis to a larger cohort without metastasis. Additionally, as lateral neck LN dissection was not performed in all patients, there is a potential for error in assessing the LN metastasis status. Future studies employing more accurate methods to evaluate lateral neck LN metastasis or prospective designs incorporating lateral neck LN dissection could help overcome these limitations.
In conclusion, microscopic ETE, upper pole location, psammomatous calcification, and central neck node metastasis are significant independent risk factors for lateral neck metastasis in patients with PTUMC. The presence of these risk factors in patients with PTUMC warrants careful consideration when deciding between active surveillance and surgical intervention. Active surveillance protocols for PTUMC should be applied judiciously, considering the identified risk factors to avoid undertreatment of potentially aggressive diseases. The risk stratification model developed in this study may aid in selecting appropriate candidates for active surveillance and in optimizing the extent of surgery in patients with PTUMC.
Materials and methods
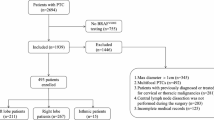
The study included patients treated at the Department of Surgery of Gangnam Severance Hospital between January 2009 and June 2013. Overall, 3,004 patients with PTUMC were analyzed.
Inclusion and exclusion criteria
Patients with a tumor size < 0.5 cm based on final pathological results, complete medical records, diagnosis of PTC confirmed by final pathological examination, history of thyroidectomy and LNs dissection (including the central and lateral neck), and no evidence of distant metastasis, were included in this study. Patients with PTC with a tumor size exceeding 0.5 cm based on the final histopathological examination results, those with missing medical record, those with pathological findings indicating malignancies other than PTC, and those who did not undergo LNs dissection during thyroid surgery, were excluded.
Preoperative evaluation
All patients were diagnosed with PTC, classified as Bethesda category ≥ 5 through preoperative neck ultrasonography and FNA biopsy. Additionally, neck CT scan was performed as part of the preoperative evaluation.
Surgical procedure
Five endocrine surgeons performed all the surgeries, including thyroidectomy and central neck dissection, using standardized techniques. Prophylactic central neck dissection was performed on the ipsilateral side of the tumor in cases in which imaging studies showed no evidence of central LN metastasis. Lateral neck LNs dissection was performed when lateral LN metastases were diagnosed based on imaging studies and biopsy results, with the extent of dissection covering levels II–Vb.
Data collection
Patient demographic information, such as sex and age, was anonymized by assigning encrypted individual identifiers. Data from preoperative imaging studies, surgical records, and final pathological reports were collected to summarize variables, including operation type, tumor size, tumor location, multiplicity, thyroiditis, microscopic ETE, and nodal status.
Statistical analysis
Data were organized using Microsoft Excel, and statistical analyses were conducted using SPSS (version 21.0; IBM Corp., Armonk, NY, USA). Univariate analyses were performed using χ² tests, t-tests, and Fisher’s exact tests. Multivariate analysis of factors associated with lateral neck LN metastasis in patients with PTC was conducted using unconditional logistic regression for variables with a p-value < 0.05.
Data availability
All data generated or analysed during this study are included in this published article [supplementary information files].
References
Schmidbauer, B., Menhart, K., Hellwig, D. & Grosse, J. Differentiated thyroid cancer—treatment: state of the Art. Int. J. Mol. Sci. 18, 1292. https://doi.org/10.3390/ijms18061292 (2017).
Zhu, H. N. et al. Progress in long non-coding RNAs as prognostic factors of papillary thyroid carcinoma. Pathol. Res. Pract. 256, 155230. https://doi.org/10.1016/j.prp.2024.155230 (2024).
Chen, K. et al. Bibliometric insights in advances of papillary thyroid microcarcinoma: research situation, hot points, and global trends. Front. Endocrinol. 13, 949993. https://doi.org/10.3389/fendo.2022.949993 (2022).
Lin, J. D. Increased incidence of papillary thyroid microcarcinoma with decreased tumor size of thyroid cancer. Med. Oncol. 27, 510–518. https://doi.org/10.1007/s12032-009-9242-8 (2010).
Lin, Y. & Wu, Y. Trends in incidence and overdiagnosis of thyroid cancer in china, japan, and South Korea. Cancer Sci. 114, 4052–4062. https://doi.org/10.1111/cas.15909 (2023).
Jeon, M. J., Kim, W. G., Kim, T. Y., Shong, Y. K. & Kim, W. B. Active surveillance as an effective management option for low-risk papillary thyroid microcarcinoma. Endocrinol. Metab. 36, 717–724. https://doi.org/10.3803/EnM.2021.1042 (2021).
Yoon, J. H. et al. Active surveillance for low-risk papillary thyroid carcinoma as an acceptable management option with additional benefits: a comprehensive systematic review. Endocrinol. Metab. 39, 152–163. https://doi.org/10.3803/EnM.2023.1794 (2024).
Sugitani, I. et al. Indications and strategy for active surveillance of adult low-risk papillary thyroid microcarcinoma: consensus statements from the Japan association of endocrine surgery task force on management for papillary thyroid microcarcinoma. Thyroid 31, 183–192. https://doi.org/10.1089/thy.2020.0330 (2021).
Ito, Y. et al. Patient age is significantly related to the progression of papillary microcarcinoma of the thyroid under observation. Thyroid 24, 27–34. https://doi.org/10.1089/thy.2013.0367 (2014).
Lee, E. K. et al. Progression of low-risk papillary thyroid microcarcinoma during active surveillance: interim analysis of a multicenter prospective cohort study of active surveillance on papillary thyroid microcarcinoma in Korea. Thyroid 32, 1328–1336. https://doi.org/10.1089/thy.2021.0614 (2022).
Liao, L. J., Ono, Y., Hung, S. F., Chen, Y. C. & Hsu, W. L. Active surveillance in early thyroid cancer: a meta-analysis. Diagnostics 14, 2628. https://doi.org/10.3390/diagnostics14232628 (2024).
Miyauchi, A. et al. Long-term outcomes of active surveillance and immediate surgery for adult patients with low-risk papillary thyroid microcarcinoma: 30-year experience. Thyroid 33, 817–825. https://doi.org/10.1089/thy.2023.0076 (2023).
Lim, J., Lee, H. S., Heo, J. H. & Song, Y. S. Clinicopathological features and molecular signatures of lateral neck lymph node metastasis in papillary thyroid microcarcinoma. Endocrinol. Metab. 39, 324–333. https://doi.org/10.3803/EnM.2023.1885 (2024).
Kim, K. et al. The contributing factors for lateral neck lymph node metastasis in papillary thyroid microcarcinoma (PTMC). Endocrine 69, 149–156. https://doi.org/10.1007/s12020-020-02251-2 (2020).
Haugen, B. R. et al. 2015 American thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American thyroid association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid 26, 1–133. https://doi.org/10.1089/thy.2015.0020 (2016).
Durante, C. et al. 2023 European thyroid association clinical practice guidelines for thyroid nodule management. Eur. Thyroid J. 12, e230067. https://doi.org/10.1530/etj-23-0067 (2023).
Back, K., Kim, J. S., Kim, J. H. & Choe, J. H. Superior located papillary thyroid microcarcinoma is a risk factor for lateral lymph node metastasis. Ann. Surg. Oncol. 26, 3992–4001. https://doi.org/10.1245/s10434-019-07587-2 (2019).
Zhan, S., Luo, D., Ge, W., Zhang, B. & Wang, T. Clinicopathological predictors of occult lateral neck lymph node metastasis in papillary thyroid cancer: a meta-analysis. Head Neck. 41, 2441–2449. https://doi.org/10.1002/hed.25762 (2019).
Chiba, T. Molecular pathology of thyroid tumors: essential points to comprehend regarding the latest WHO classification. Biomedicines 12, 712. https://doi.org/10.3390/biomedicines12040712 (2024).
Jung, C. K., Bychkov, A. & Kakudo, K. Update from the 2022 world health organization classification of thyroid tumors: a standardized diagnostic approach. Endocrinol. Metab. 37, 703–718. https://doi.org/10.3803/EnM.2022.1553 (2022).
Lee, J. S. et al. Aggressive subtypes of papillary thyroid carcinoma smaller than 1 cm. J. Clin. Endocrinol. Metab. 108, 1370–1375. https://doi.org/10.1210/clinem/dgac739 (2023).
Lukyanov, S. A. et al. Prediction of the aggressive clinical course of papillary thyroid carcinoma based on fine needle aspiration biopsy molecular testing. Int. J. Mol. Sci. 25, 7090. https://doi.org/10.3390/ijms25137090 (2024).
Zhu, Y. et al. Cytomorphologic features as predictors of aggressiveness in patients with pT1 papillary thyroid carcinoma: a retrospective study of associations with clinicopathological parameters in 226 fine-needle aspirates. Gland Surg. 10, 319–327. https://doi.org/10.21037/gs-20-618 (2021).
Alabousi, M. et al. Diagnostic test accuracy of ultrasonography vs computed tomography for papillary thyroid cancer cervical lymph node metastasis: a systematic review and meta-analysis. JAMA Otolaryngol. Head Neck Surg. 148, 107–118. https://doi.org/10.1001/jamaoto.2021.3387 (2022).
Dong, L. et al. CT radiomics-based nomogram for predicting the lateral neck lymph node metastasis in papillary thyroid carcinoma: a prospective multicenter study. Acad. Radiol. 30, 3032–3046. https://doi.org/10.1016/j.acra.2023.03.039 (2023).
Nikiforov, Y. E. Role of molecular markers in thyroid nodule management: then and now. Endocr. Pract. 23, 979–988. https://doi.org/10.4158/ep171805.Ra (2017).
Nylén, C. et al. Molecular markers guiding thyroid cancer management. Cancers 12, 2164. https://doi.org/10.3390/cancers12082164 (2020).
Vuong, H. G., Altibi, A. M. A., Duong, U. N. P. & Hassell, L. Prognostic implication of BRAF and TERT promoter mutation combination in papillary thyroid carcinoma—a meta-analysis. Clin. Endocrinol. 87, 411–417. https://doi.org/10.1111/cen.13413 (2017).
Zhang, W., Lin, S., Wang, Z., Zhang, W. & Xing, M. Coexisting RET/PTC and TERT promoter mutation predict poor prognosis but effective RET and MEK targeting in thyroid cancer. J. Clin. Endocrinol. Metab. 109, 3166–3175. https://doi.org/10.1210/clinem/dgae327 (2024).
Chiosea, S. et al. Molecular profiling of 50 734 Bethesda III-VI thyroid nodules by ThyroSeq v3: implications for personalized management. J. Clin. Endocrinol. Metab. 108, 2999–3008. https://doi.org/10.1210/clinem/dgad220 (2023).
Alzahrani, A. S. Clinical use of molecular data in thyroid nodules and cancer. J. Clin. Endocrinol. Metab. 108, 2759–2771. https://doi.org/10.1210/clinem/dgad282 (2023).
Acknowledgements
I would like to express my deepest gratitude to my advisor, Professor Hang-Seok Chang, for his invaluable guidance and support throughout the study.
Funding
This research received no specific grants from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Conceptualization: Hyeung Kyoo KimMethodology: Hyeung Kyoo KimFormal Analysis: Hyeung Kyoo Kim, Ho Jung JeongInvestigation: Yong Sang Lee, Hang-Seok ChangResources: Jin Seok Lee, Soo Young KimData Curation: Yong Sang Lee, Hang-Seok ChangWriting – Original Draft: Hyeung Kyoo KimWriting – Review & Editing: Hang-Seok ChangVisualization: Hyeung Kyoo KimSupervision: Hang-Seok Chang.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This study adhered to the ethical principles outlined in the Declaration of Helsinki for medical research involving human participants. This study was approved by the Institutional Review Board of Yonsei University (approval number: 3-2025-0072). Informed consent was waived from IRB due to the retrospective design.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kim, H.K., Jeong, H.J., Lee, J.S. et al. Risk factors for lateral neck lymph node metastasis in papillary thyroid ultra micro carcinoma with implications for active surveillance. Sci Rep 15, 30640 (2025). https://doi.org/10.1038/s41598-025-16519-y
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-16519-y