Abstract
This study aims to compare retinal vascular characteristics between ApoE ε4 carriers and non-carriers in healthy individuals. A total of 84 cognitively healthy subjects—64 non-carriers and 20 carriers of the ApoE ε4 allele—were analyzed using swept-source optical coherence tomography angiography (SS-OCTA). In the retinal vascular layer of OCTA images, parameters such as foveal avascular zone (FAZ) area, vessel perimeter, acircularity index (AI), and vascular density (FD) were quantified. Additionally, blood flow areas in the outer retina and choroidal capillaries, as well as the area without perfusion in the superficial vascular complex (SVC), were measured. Compared to non-carriers, carriers of ApoE ε4 exhibited significant reductions in the FAZ area, as well as increased foveal vessel density in both the deep and superficial vascular complexes. No significant differences were observed in AI, FD, blood perfusion area, or perifoveal blood vessel density between the groups. These findings suggest that reduced FAZ area and increased foveal blood flow density in ApoE ε4 carriers may indicate a compensatory mechanism in retinal blood flow, potentially serving as early indicators of Alzheimer’s disease (AD).
Similar content being viewed by others
Introduction
Alzheimer’s disease (AD) is a prevalent neurodegenerative disorder and ranks as a leading cause of dementia among the elderly population1,2. Driven by global population aging, AD prevalence and incidence are rising sharply, with worldwide cases projected to escalate from 46.8 million in 2015 to over 1.315 billion by 20502,3,4. Unfortunately, effective treatments for AD remain elusive; thus, prevention and early intervention have emerged as the most viable strategies to attenuate disease progression. Traditional diagnostic methods, including positron emission tomography (PET) and cerebrospinal fluid (CSF) analysis via lumbar puncture, are the mainstays for diagnosing AD5. However, patients often view these techniques unfavorably due to their high cost and invasive nature. Consequently, there is an urgent need for the development of non-invasive and cost-effective biomarkers that facilitate timely management and intervention, aiming to delay or prevent cognitive decline and the eventual onset of dementia.
The brain and eyes are closely related, with the retina being an extension of the optic vesicle derived from the neuroectoderm6. Additionally, there are intrinsic anatomical and physiological connections between the brain and eyes, making the eye a true window into brain health7,8. Recent studies have shown that pathological changes in the eye are associated with AD, primarily due to the transport of Aβ from the brain to the eye9. Retinal alterations related to AD, including microvascular abnormalities and retinal layer thinning, may serve as predictive biomarkers for Alzheimer’s disease10,11,12. Therefore, there is an urgent need to use the eye as a breakthrough point to observe earlier ocular indicators of brain disease in patients.
The ɛ4 allele of the apolipoprotein E (ApoE) gene is the most prominent genetic risk factor for Alzheimer’s disease, second only to age13. Emerging research has shown that the ApoE ɛ4 allele is also associated with cerebral hypoperfusion14,15, and a decline in cerebral blood flow (CBF) may precede the onset of clinical symptoms of Alzheimer’s disease16,17. However, changes in retinal blood flow among genetically susceptible individuals with Alzheimer’s disease remain unclear. Therefore, there is an urgent need for systematic ocular clinical observation in cognitively healthy ApoE ɛ4 carriers.
Based on this, we systematically analyzed the differences between ApoE ɛ4 allele carriers and non-carriers by comparing perfusion metrics obtained through swept-source optical coherence tomography angiography (SS-OCTA).
Methods
The study protocol is designed as a prospective observational investigation and has received approval from the Ethics Committee of the First Affiliated Hospital with Nanjing Medical University (Approval No: 2022-SR-023). Written informed consent was obtained from all participants. This study adheres to the ethical principles for human experimentation as outlined in the Declaration of Helsinki.
Participants
Participants aged between 50 and 70 years who exhibited no cognitive impairment and demonstrated normal findings on magnetic resonance imaging (MRI) were considered eligible for the study. Cognitive impairment was defined as a score of 25 or lower on the Montreal Cognitive Assessment (MoCA).
Exclusion criteria encompassed a range of medical conditions, including hypertension, diabetes, cancer, and other neurodegenerative disorders such as Parkinson’s disease and multiple sclerosis. Additionally, ocular exclusions included glaucoma, retinal ischemic conditions, epiretinal membranes (ERM), age-related macular degeneration, macular holes, previous retinal interventions (e.g., pars plana vitrectomy, laser photocoagulation), best corrected visual acuity of less than 20/40 (approximately Log MAR = 0.30), and significant refractive errors exceeding ± 3.00 DS (spherical equivalent). Furthermore, images of inadequate quality characterized by a signal strength of less than 6, extensive shadowing, decentration, motion artifacts, and extraneous noise that could hinder quantitative analysis were also excluded.
Neuropsychological assessment
Cognitive ability was assessed using the MoCA, a comprehensive and sensitive screening tool that evaluates various cognitive domains, including attention, executive functions, memory, language, visuospatial skills, orientation, calculations, and conceptual reasoning. The maximum score on the MoCA is 30 points, with a score of 26 or higher indicative of normal cognitive function.
Genotyping
Oropharyngeal swab samples were collected for the detection of ApoE genotypes. The genotyping was performed by analyzing two specific single nucleotide polymorphisms (SNPs), rs429358 and rs7412, utilizing the improved multiple ligase detection reaction (imLDR) technique. Participants who possessed one or two ApoE ε4 alleles were categorized as ApoE ε4 carriers (ε4/ε4 and ε4/ε3), while those lacking these alleles were classified as ApoE ε4 non-carriers (ε2/ε2, ε2/ε3, and ε3/ε3).
Optical coherence tomography angiography
Optical coherence tomography angiography (OCTA) images with a 3 × 3 mm configuration, centered on the fovea, were acquired by two skilled ophthalmic technicians using the AngioVue instrument (Optovue RTVue XR Avanti; Optovue, Inc., Fremont, CA, USA). Vascular segmentation was performed automatically through the AngioVue module. A quantitative assessment of the foveal avascular zone (FAZ) index and retinal blood flow area was executed using this advanced instrument.
The FAZ index encompasses several parameters: the FAZ area, the perimeter of the avascular region around the fovea (PERIM), the acircularity index (AI)-defined as the ratio of the measured circumference to the standard circumference of an equal area-and the foveal avascular zone blood density (FD), which refers to the blood flow density within a 300-micron-wide double ring surrounding the FAZ.
The retinal blood flow area was delineated into two regions: the outer retina, spanning from 10 µm below the outer plexiform layer (OPL) to 10 µm above the Bruch’s membrane (BRM), and the choriocapillaris, extending from 10 µm above the BRM to 30 µm below it. Additionally, the measurement of the Now-Flow area of blood was based on the superficial vascular complex (SVC), extending from the inner limiting membrane (ILM) to 10 µm above the inner plexiform layer (IPL).
Blood perfusion density, which refers to the proportion of the area occupied by blood flow structures, was measured at three different levels: the SVC, deep vascular complex (DVC), and radial peripapillary capillaries (RPC). The macular area was analyzed according to the standard Early Treatment Diabetic Retinopathy Study (ETDRS) macular grid (a foveal area of 1 mm in diameter, 1–3 mm around the fovea in the inner ring, and 3–6 mm for the outer ring)18.
Statistical analyses
All data analyses were performed using IBM SPSS Statistics for Windows (version 27; IBM Corp) and R software (version 4.3.2). To appropriately address the hierarchical structure of the data-accounting for repeated ocular measurements and inter-individual variability-Generalized Linear Mixed Models (GLMMs) were employed. Model selection involved choosing suitable distribution families and link functions according to the nature of the variables: Linear Mixed Models (LMMs) were applied for continuous variables with approximately normal distributions, while Gamma distributions with log-link functions were used for continuous variables exhibiting non-normal distributions. For multiple group comparisons, estimated marginal means (EMMs) were computed using the ‘emmeans’ package in R, with p-values adjusted for multiple testing using the Benjamini–Hochberg procedure to control the false discovery rate (FDR). Partial correlations, controlling for random effects, were calculated with the ‘ppcor’ package to assess inter-variable associations. Linear Mixed Models with random intercepts were further employed to estimate Pearson correlation coefficients for continuous variable relationships. Diagnostic performance of classification models was evaluated via receiver operating characteristic (ROC) curves constructed from predicted probabilities generated by binomial GLMMs with logit link functions using the ‘pROC’ package. Optimal cut-off points were determined by maximizing Youden’s index. The following AUC classification was used in our study: 0.5–0.6 = unsatisfactory, 0.6–0.7 = satisfactory, 0.7–0.8 = good, 0.8–0.9 = very good, and 0.9–1 = excellent. A threshold of p < 0.05 was established to determine statistical significance for all tests conducted.
Results
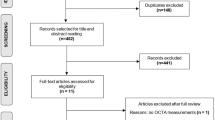
A total of 85 eyes from 64 non-carriers of ApoE ε4 and 29 eyes from 20 ApoE ε4 carriers were included in the study, with the specific selection process illustrated in Fig. 1. Females represented a higher proportion of participants, but no significant difference was observed between the groups (64.1% in non-carriers vs. 65.0% in carriers). Similarly, there were no significant differences in age or educational level between the two groups. Demographic data for the subjects are summarized in Table 1. (Fig. 2).
Flow diagram illustrating the selection of subjects and eyes. ApoE ε4-, noncarriers of ε4 allele; ApoE ε4 + , carriers of at least one e4 allele. OCTA, optical coherence tomography angiography.
30 × 30° OCTA images of the central perfusion area at three different levels from a 58-year-old ApoE ε4 carrier. (A) Foveal blood flow area based on choroidal capillaries, the small red circle is the fovea, and the yellow part indicates the area of blood perfusion; (B) Foveal blood flow area based on the outer retina, the small red circle is the fovea, and the yellow part indicates the area of blood perfusion; (C) No central perfusion area based on superficial vascular complex, the yellow part shows the area without blood perfusion.
Comparison of FAZ index between ApoE ε4 carriers and controls
Figure 2 illustrates eye blood perfusion in a 58-year-old woman. In the FAZ index, the FAZ area (mm2) was significantly lower in ApoE ε4 carriers compared to controls; a trend toward group differences was observed in PERIM (mm) (p = 0.05). No statistically significant differences were found between the groups in AI and FD (%) (Fig. 3).
FAZ index between ApoE ε4 carriers and controls. (A) The area of the fovea without blood vessels; (B) The perimeter of the avascular region around the fovea; (C) Acircularity index; (D) The blood flow density within a 300-micron-wide double ring surrounding the FAZ.
Comparison of retinal blood flow area between ApoE ε4 carriers and controls
Within the retinal blood flow area, no differences were observed in Now-Flow area based on SVC, the blood flow area of the outer retina and choriocapillaris between the groups (see Fig. 4).
Retinal blood flow area between ApoE ε4 carriers and controls. (A) Foveal blood flow area based on the outer retina; (B) Foveal blood flow area based on choroidal capillaries; (C) No central perfusion area based on superficial vascular complex.
Comparison of blood flow density between ApoE ε4 carriers and controls
The blood perfusion density at three different levels including SVC, DVC and RPC were compared (see Figs. 5, 6 and 7). The blood perfusion density in the fovea at the SVC and DVC levels was significantly higher in ApoE ε4 carriers compared to the control group. Conversely, the blood perfusion density in inside disc of RPC was significantly lower in ApoE ε4 carriers. No statistically significant differences in microvascular perfusion density were detected within the peripheral regions at three levels between the two groups (p > 0.05).
Blood Flow Density of the Superficial Vascular Complex Between ApoE ε4 Carriers and Controls. (A) Whole Image; (B) Superior-Hemi; (C) Inferior-Hemi; (D) Fovea; (E) ParaFovea; (F) Tempo-ParaFovea; (G) Superior-ParaFovea; (H) Nasal-ParaFovea; (I) Inferior-ParaFovea.
Blood Flow Density of Deep Vascular Complex Between ApoE ε4 Carriers and Controls. (A) Whole Image; (B) Superior-Hemi; (C) Inferior-Hemi; (D) Fovea; (E) ParaFovea; (F) Tempo-ParaFovea; (G) Superior-ParaFovea; (H) Nasal-ParaFovea; (I) Inferior-ParaFovea.
Blood flow density of RPC Between ApoE ε4 Carriers and Controls. (A) Whole Image; (B) Inside Disc; (C) Peripapillary; (D) Superior-Hemi-Peripapillary; (E) Inferior-Hemi-Peripapillary; (F) Tempo-Peripapillary; (G) Superior-Peripapillary; (H) Nasal-Peripapillary; I Inferior-Peripapillary.
Retinal parameters of ApoE ε4 carriers and control group were compared based on gender stratification
Notably, sex-stratified analyses demonstrated that FAZ area was the sole parameter exhibiting significant intergroup divergence among all hemodynamic metrics examined. Post hoc multiple comparisons revealed that both female (0.34 ± 0.11mm2) and male (0.37 ± 0.13mm2) ApoE ε4 non-carriers displayed significantly larger FAZ areas compared to male carriers (0.22 ± 0.10mm2) (see Table S2–S3).
Univariate ROC model and multimodal ROC model to distinguish between ApoE ε4 carriers and controls
Univariate receiver operating characteristic (ROC) analyses of retinal vascular parameters, including FAZ area, Deep_Fovea, Sup_Fovea, and RPC_In, demonstrated discriminative capacity for identifying ApoE ε4 carriers, with FAZ area emerging as the strongest classifier. The FAZ model achieved an area under the curve (AUC) of 0.70 (95% CI: 0.58–0.81), which remained stable at 0.70 (95% CI: 0.59–0.80) after model correction (see Fig. 8A–D). A Youden’s index of 0.38 was utilized to establish a cutoff value of ≤ 0.33 mm2 for identifying ApoE ε4 carriers, demonstrating a sensitivity of 79.0% and a specificity of 59.0% (see Table S1).
ROC Curves for Separation of ApoE ε4 Carriers and ApoE ε4 Noncarriers Based on Different Retinal Blood Flow Parameters. (A) ROC curve illustrating the model based on FAZ area. (B) ROC curve illustrating the model based on Deep_Fovea. (C) ROC curve illustrating the model based on Sup_Fovea. (D) ROC curve illustrating the model based on RPC_In. (E) ROC curve illustrating a model incorporating FAZ area, Deep_Fovea, and Sup_Fovea. (F) ROC curve illustrating a model incorporating FAZ area, Deep_Fovea, Sup_Fovea, and RPC_In. Deep_Fovea, foveal density of deep vascular complex (DVC); Sup_Fovea, foveal density of superficial vascular complex (SVC).
Furthermore, the multimodal ROC model based on FAZ area, Deep_Fovea, Sup_Fovea, exhibited better classification performance than the univariate ROC analysis (see Fig. 8E–F). Notably, the multimodal ROC model combining FAZ area, Deep_Fovea, Sup_Fovea outperformed a model that included all four retinal parameters (FAZ area, Deep_Fovea, Sup_Fovea, and RPC_In) with an AUC of 0.72 (95% CI = 0.61–0.82). After model correction, it maintained an AUC of 0.72 (95% CI 0.59–0.80), achieving a sensitivity of 69.0% and a specificity of 76.0%.
Correlation analysis of blood perfusion parameters and blood density parameters in ApoE ε4 carriers
Correlation analysis revealed that the FAZ area (p < 0.001) and PERIM (p = 0.0012) were negatively correlated with the central blood flow density of the SVC in ApoE ε4 carriers. However, RPC_In showed no significant correlation with the central blood flow density of the SVC) and DVC (Fig. 9).
The blood perfusion parameters and blood density parameters in ApoE ε4 Carriers. (A–C) Linear correlation between FAZ area, PERIM, RPC_In and foveal density of SVC. (D–F) Linear correlation between FAZ area, PERIM, RPC_In and foveal density of DVC. SVC, superficial vascular complex; DVC, deep vascular complex.
Discussion
In this study, we identified a significant reduction in the FAZ area and the central blood flow density of RPC among ApoE ε4 carriers compared to non-carriers, while blood flow density in the fovea was increased in both the SVC and DVC. Notably, parameters such as the AI, FD, and retinal blood flow in both the outer retina and choriocapillaris exhibited no significant differences between the two groups. Previous studies have shown that female carriers of ApoE ε4 experience earlier cognitive decline compared to their male counterparts19. Considering these gender differences, we conducted a further stratified analysis. Sex-stratified analysis demonstrated larger FAZ areas in male and female ApoE ε4 non-carriers compared to male carriers.
Additionally, we developed a statistically robust ROC model capable of effectively differentiating ApoE ε4 carriers from controls, based on individual analyses of FAZ area, Sup_Fovea, Deep_Fovea, and RPC_In, with the FAZ parameter achieving the highest AUC value. Furthermore, we conducted ROC curve analysis across multiple modalities and found that the multimodal ROC models for the FAZ area, Sup_Fovea, and Deep_Fovea have strong AUC for distinguishing between groups.
Our study found that retinal blood flow significantly increased in ApoE ε4 carriers, which contradicts the conclusions of previous research indicating that retinal blood flow perfusion decreases in patients with AD20,21,22. This discrepancy may stem from variations in study design, population characteristics, or stages of the disease. For example, all participants in our study were healthy individuals without significant vascular risk factors, while prior studies often focused on populations with cognitive impairment or those with existing vascular lesions. Additionally, differences in imaging techniques and data analysis methods may also account for the divergent results.
ApoE ε4 is one of the primary genetic risk factors for AD, and its influence on blood flow is complex and multifaceted, reflecting a dynamic and adaptive process. In cognitively normal ApoE ε4 carriers, the allele may induce an increase in retinal blood flow through various compensatory mechanisms. First, reduced perfusion can trigger a retinal autoregulatory response, leading to macular edema and neovascularization23,24. Vascular endothelial growth factor (VEGF), which is upregulated under hypoxic conditions, plays a pivotal role in this process23,25. VEGF promotes increased blood flow by enhancing endothelial cell survival and regulating vascular permeability26. Notably, studies have demonstrated that VEGF treatment can mitigate cognitive deficits and improve cerebral ischemic function in ApoE ε4 female mice, both in the early and late stages of AD pathology27,28. Additionally, the metabolic demands of brain and retinal tissues in ApoE ε4 carriers may be elevated29, prompting local blood vessels to dynamically adapt to these demands. This adaptation is mediated by mechanisms such as the short-term activation of endothelial nitric oxide synthase (eNOS), which enhances blood supply30,31. Furthermore, neuronal metabolic activity can influence blood flow regulation. Interactions between neurons and blood vessels may induce vascular dilation through neurogenic signaling, further augmenting local blood flow31,32,33.
In the late stages of AD, ApoE ε4 engages in a mutually reinforcing vicious cycle with cerebrovascular dysfunction through multidimensional pathological mechanisms, which can be summarized as three interconnected pathological axes. First, ApoE ε4 disrupts vascular homeostasis via dual pathways: On one hand, it activates the NF-κB pathway to persistently induce the secretion of pro-inflammatory factors such as TNF-α and IL-634,35, leading to endothelial dysfunction characterized by reduced vasodilation capacity and abnormally increased vascular permeability36. On the other hand, it suppresses eNOS activity, significantly reducing the synthesis of nitric oxide (NO), a critical mediator of vasodilation30,37. This dual insult synergistically induces microvascular pathology and cerebral hypoperfusion, laying the pathological foundation for ischemic injury. Second, at the blood–brain barrier (BBB)-lipid metabolism axis, ApoE ε4 activates cyclophilin A in pericytes, triggering the NF-κB/MMP-9 cascade38, thereby compromising BBB structural integrity. Concurrently, this genotype drives lipid metabolism dysregulation, promoting the abnormal accumulation of atherogenic lipoproteins39,40. Such structural-metabolic dual damage markedly exacerbates cerebrovascular degeneration. Notably, ApoE ε4 carriers with AD often exhibit reduced cardiac output41, suggesting that systemic hemodynamic alterations may accelerate cerebrovascular injury through brain-systemic circulation coupling mechanisms. Third, in the Aβ-vascular interaction axis, ApoE ε4 significantly impairs β-amyloid (Aβ) clearance efficiency via dysregulated low-density lipoprotein receptor-related protein 1 (LRP1)42,43,44,45, leading to perivascular Aβ deposition. These amyloid plaques synergistically amplify vascular pathology by activating oxidative stress pathways and upregulating endothelin-1 expression46,47,48, ultimately causing decompensation of cerebrovascular autoregulation49. This multi-target cascade mechanism reveals that ApoE ε4 not only directly accelerates neurodegeneration in late-stage AD but also drives disease progression by constructing a vascular-metabolic-inflammatory network. The complexity of its pathological mechanisms suggests that combined interventions targeting vascular protection, inflammatory modulation, and lipid metabolism balance may offer new directions to overcome current therapeutic limitations.
Although we found no significant sex-based differences in retinal blood flow among ApoE ε4 carriers, numerous previous studies have established that female ApoE ε4 carriers exhibit significant gender-specific differences in the progression of AD, with distinct patterns of cognitive decline and pathological mechanisms compared to males. Studies have shown that female carriers face a higher risk of transitioning from normal cognitive status to mild cognitive impairment (MCI) and AD50,51, which may be closely linked to hormonal fluctuations and metabolic compensation imbalances. During the perimenopausal to postmenopausal stages, declining estrogen levels accelerate amyloid deposition, reduce brain glucose metabolism, and decrease gray and white matter volumes52. Concurrently, compensatory hemodynamic and metabolic adjustments occur, such as increased cerebral blood flow and enhanced ketone metabolism53. Notably, the cerebrovascular system in female carriers demonstrates dynamic fragility54,55: hormonal fluctuations during perimenopause lead to transient reductions in blood flow56,57, while postmenopausal compensatory increases in cerebral blood flow may exacerbate metabolic dysregulation, potentially driving AD progression57.
In addition to hemodynamic changes, immune pathological mechanisms further underscore the unique vulnerability of female ApoE ε4 carriers. Their immune systems exhibit a paradoxical activation state, with significant elevations in pro-inflammatory factors (e.g., IL-17, IL-1) alongside upregulated immunosuppressive factors (e.g., IL-10, TGF-β)58. This suggests that compensatory immunosuppression is insufficient to counteract excessive neuroinflammation. Furthermore, abnormal expression of immune checkpoint molecules (e.g., LAG3, PD-1) reflects immune cell exhaustion, which may accelerate neuronal damage58. Critically, the interplay between hormones and immunity forms a key pathological loop: postmenopausal estrogen decline regulates microglial function and increases susceptibility to Aβ pathology51,59, creating a self-reinforcing cycle of “metabolic dysregulation-immune activation-neuroinflammation.” However, the temporal and spatial relationships between hemodynamic changes and immune responses, as well as the specific regulatory roles of ApoE ε4 in these processes, remain to be elucidated through integrated multi-omics studies.
Our study reveals significant alterations in retinal hemodynamics among ApoE ε4 carriers, offering new insights into the role of ApoE ε4 in the early pathology of AD. Notably, the changes in retinal blood flow dynamic parameters indicate their potential as novel biomarkers for the early diagnosis of AD. The ROC model developed in this study effectively distinguishes ApoE ε4 carriers from the control group. This model serves as an objective detection tool with significant clinical application value for early AD screening.
This study has several limitations: Firstly, its cross-sectional design makes it difficult to establish a causal temporal relationship between ApoE ε4-related hemodynamic changes and AD pathology, necessitating validation through longitudinal follow-up studies. Secondly, the exclusion of subjects with vascular risk factors may limit the applicability of the findings to high-risk AD populations. Thirdly, variations in retinal imaging equipment and differing analysis algorithms could affect the comparability of the results, underscoring the need for a standardized detection protocol. Finally, the sample size of the study was small, and there was no in-depth investigation into the specific mechanisms behind the differences in retinal vasculature between groups.
Conclusions
In this study, our findings indicate that changes in retinal blood flow could serve as promising early indicators of AD, particularly in individuals with a genetic predisposition. As we strive for non-invasive and effective diagnostic approaches, incorporating ocular assessments into routine clinical practice may significantly enhance our ability to identify those at risk for AD, ultimately supporting the development of timely interventions aimed at mitigating cognitive decline.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Change history
08 January 2026
The original online version of this Article was revised: The Funding information section was missing from this article and should have read 'This study was funded by the National Natural Science Foundation of China (Grant numbers 81772454 and 81971237), the Jiangsu Municipal Science and Technology Bureau (BE2017734) and the Key Project of Jiangsu Province's Key Research and Development Program (BE2023023-2).' The original article has been corrected.
References
Lane, C. A., Hardy, J. & Schott, J. M. Alzheimer’s disease. Eur. J. Neurol. 25(1), 59–70. https://doi.org/10.1111/ene.13439 (2018).
Alzheimer’s disease facts and figures. Alzheimers Dement. 19(4), 1598–1695. https://doi.org/10.1002/alz.13016 (2023).
Yu, N. et al. Ganoderma lucidum triterpenoids (GLTs) reduce neuronal apoptosis via inhibition of ROCK signal pathway in APP/PS1 transgenic Alzheimer’s disease mice. Oxid Med. Cell Longev. 2020, 9894037. https://doi.org/10.1155/2020/9894037 (2020).
Chu, J.-C. et al. Synthesis and biological evaluation of C-4 substituted phenoxazine-bearing hydroxamic acids with potent class II histone deacetylase inhibitory activities. J. Enzyme Inhib. Med. Chem. 38(1), 2212326. https://doi.org/10.1080/14756366.2023.2212326 (2023).
Khan, S., Barve, K. H. & Kumar, M. S. Recent advancements in pathogenesis, diagnostics and treatment of Alzheimer’s disease. Curr. Neuropharmacol. 18(11), 1106–1125. https://doi.org/10.2174/1570159X18666200528142429 (2020).
Coulombre, A. J. Experimental embryology of the vertebrate eye. Invest. Ophthalmol. 4, 411–419 (1965).
Garcia-Martin, E. et al. Retina measurements for diagnosis of Parkinson disease. Retina 34(5), 971–980. https://doi.org/10.1097/IAE.0000000000000028 (2014).
Archibald, N. K., Clarke, M. P., Mosimann, U. P. & Burn, D. J. The retina in Parkinson’s disease. Brain 132(Pt 5), 1128–1145. https://doi.org/10.1093/brain/awp068 (2009).
Cao, Q. et al. Transport of β-amyloid from brain to eye causes retinal degeneration in Alzheimer’s disease. J. Exp. Med. https://doi.org/10.1084/jem.20240386 (2024).
Golzan, S. M. et al. Retinal vascular and structural changes are associated with amyloid burden in the elderly: ophthalmic biomarkers of preclinical Alzheimer’s disease. Alzheimers Res. Ther. 9(1), 13. https://doi.org/10.1186/s13195-017-0239-9 (2017).
Li, B. et al. Differential reductions in the capillary red-blood-cell flux between retina and brain under chronic global hypoperfusion. Neurophotonics. 10(3), 035001. https://doi.org/10.1117/1.NPh.10.3.035001 (2023).
Ko, F. et al. Association of retinal nerve fiber layer thinning with current and future cognitive decline: A study using optical coherence tomography. JAMA Neurol. 75(10), 1198–1205. https://doi.org/10.1001/jamaneurol.2018.1578 (2018).
López-Cuenca, I. et al. Early visual alterations in individuals at-risk of Alzheimer’s disease: A multidisciplinary approach. Alzheimers Res. Ther. 15(1), 19. https://doi.org/10.1186/s13195-023-01166-0 (2023).
Rawle, M. J. et al. Apolipoprotein-E (Apoe) ε4 and cognitive decline over the adult life course. Transl. Psychiatry. 8(1), 18. https://doi.org/10.1038/s41398-017-0064-8 (2018).
Thambisetty, M., Beason-Held, L., An, Y., Kraut, M. A. & Resnick, S. M. APOE epsilon4 genotype and longitudinal changes in cerebral blood flow in normal aging. Arch. Neurol. 67(1), 93–98. https://doi.org/10.1001/archneurol.2009.913 (2010).
Wolters, F. J. et al. Cerebral perfusion and the risk of dementia: A population-based study. Circulation 136(8), 719–728. https://doi.org/10.1161/CIRCULATIONAHA.117.027448 (2017).
Bracko, O., Cruz Hernández, J. C., Park, L., Nishimura, N. & Schaffer, C. B. Causes and consequences of baseline cerebral blood flow reductions in Alzheimer’s disease. J. Cereb. Blood Flow Metab. 41(7), 1501–1516. https://doi.org/10.1177/0271678X20982383 (2021).
Grading diabetic retinopathy from stereoscopic color fundus photographs—An extension of the modified Airlie House classification. ETDRS report number 10. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology. 1991;98(5 Suppl):786–806.
Cho, S. H. et al. Disease progression modelling from preclinical Alzheimer’s disease (AD) to AD dementia. Sci. Rep. 11(1), 4168. https://doi.org/10.1038/s41598-021-83585-3 (2021).
Arthur, E. et al. Retinal mid-peripheral capillary free zones are enlarged in cognitively unimpaired older adults at high risk for Alzheimer’s disease. Alzheimers Res. Ther. 15(1), 172. https://doi.org/10.1186/s13195-023-01312-8 (2023).
Corradetti, G. et al. Choriocapillaris and retinal vascular alterations in presymptomatic Alzheimer’s disease. Invest Ophthalmol. Vis. Sci. 65(1), 47. https://doi.org/10.1167/iovs.65.1.47 (2024).
Yeh, T.-C., Kuo, C.-T. & Chou, Y.-B. Retinal microvascular changes in mild cognitive impairment and Alzheimer’s disease: A systematic review, meta-analysis, and meta-regression. Front. Aging Neurosci. 14, 860759. https://doi.org/10.3389/fnagi.2022.860759 (2022).
Pournaras, C. J., Rungger-Brändle, E., Riva, C. E., Hardarson, S. H. & Stefansson, E. Regulation of retinal blood flow in health and disease. Prog. Retin Eye Res. 27(3), 284–330. https://doi.org/10.1016/j.preteyeres.2008.02.002 (2008).
Harris, A., Ciulla, T. A., Chung, H. S. & Martin, B. Regulation of retinal and optic nerve blood flow. Arch. Ophthalmol. 116(11), 1491–1495 (1998).
Antes, R. et al. VEGF mediates ApoE4-Induced neovascularization and synaptic pathology in the choroid and retina. Curr. Alzheimer Res. 12(4), 323–334 (2015).
Melincovici, C. S. et al. Vascular endothelial growth factor (VEGF)—key factor in normal and pathological angiogenesis. Rom. J. Morphol. Embryol. 59(2), 455–467 (2018).
Zaldua, S. et al. Epidermal growth factor treatment of female mice that express APOE4 at an age of advanced pathology mitigates behavioral and cerebrovascular dysfunction. Heliyon. 6(5), e03919. https://doi.org/10.1016/j.heliyon.2020.e03919 (2020).
Thomas, R., Morris, A. W. J. & Tai, L. M. Epidermal growth factor prevents APOE4-induced cognitive and cerebrovascular deficits in female mice. Heliyon. 3(6), e00319. https://doi.org/10.1016/j.heliyon.2017.e00319 (2017).
Protas, H. D. et al. Posterior cingulate glucose metabolism, hippocampal glucose metabolism, and hippocampal volume in cognitively normal, late-middle-aged persons at 3 levels of genetic risk for Alzheimer disease. JAMA Neurol. 70(3), 320–325 (2013).
Seddon, M. D., Chowienczyk, P. J., Brett, S. E., Casadei, B. & Shah, A. M. Neuronal nitric oxide synthase regulates basal microvascular tone in humans in vivo. Circulation 117(15), 1991–1996. https://doi.org/10.1161/CIRCULATIONAHA.107.744540 (2008).
O’Gallagher, K. et al. Neuronal nitric oxide synthase regulates regional brain perfusion in healthy humans. Cardiovasc. Res. 118(5), 1321–1329. https://doi.org/10.1093/cvr/cvab155 (2022).
Williams, H. C. et al. APOE alters glucose flux through central carbon pathways in astrocytes. Neurobiol. Dis. 136, 104742. https://doi.org/10.1016/j.nbd.2020.104742 (2020).
Østergaard, L. et al. The capillary dysfunction hypothesis of Alzheimer’s disease. Neurobiol. Aging. 34(4), 1018–1031. https://doi.org/10.1016/j.neurobiolaging.2012.09.011 (2013).
Ophir, G. et al. Apolipoprotein E4 enhances brain inflammation by modulation of the NF-kappaB signaling cascade. Neurobiol. Dis. 20(3), 709–718 (2005).
Fan, Y.-Y. et al. APOE ε4 allele elevates the expressions of inflammatory factors and promotes Alzheimer’s disease progression: A comparative study based on Han and She populations in the Wenzhou area. Brain Res. Bull. 132, 39–43. https://doi.org/10.1016/j.brainresbull.2017.04.017 (2017).
Almutary, A. G. et al. Inflammatory signaling pathways in Alzheimer’s disease: Mechanistic insights and possible therapeutic interventions. Ageing Res Rev. 104, 102548. https://doi.org/10.1016/j.arr.2024.102548 (2025).
Seddon, M. et al. Effects of neuronal nitric oxide synthase on human coronary artery diameter and blood flow in vivo. Circulation 119(20), 2656–2662. https://doi.org/10.1161/CIRCULATIONAHA.108.822205 (2009).
Bell, R. D. et al. Apolipoprotein E controls cerebrovascular integrity via cyclophilin A. Nature 485(7399), 512–516. https://doi.org/10.1038/nature11087 (2012).
Mahley, R. W. & Rall, S. C. Apolipoprotein E: Far more than a lipid transport protein. Annu. Rev. Genomics Hum. Genet. 1, 507–537 (2000).
Yang, L. G., March, Z. M., Stephenson, R. A. & Narayan, P. S. Apolipoprotein E in lipid metabolism and neurodegenerative disease. Trends Endocrinol Metab. 34(8), 430–445. https://doi.org/10.1016/j.tem.2023.05.002 (2023).
Bown, C. W. et al. Apolipoprotein E genotype modifies the association between cardiac output and cognition in older adults. J Am. Heart Assoc. 8(15), e011146. https://doi.org/10.1161/JAHA.118.011146 (2019).
Jackson, R. J. et al. APOE2 gene therapy reduces amyloid deposition and improves markers of neuroinflammation and neurodegeneration in a mouse model of Alzheimer disease. Mol. Ther. 32(5), 1373–1386. https://doi.org/10.1016/j.ymthe.2024.03.024 (2024).
Fongang, B. et al. A genome-wide association meta-analysis of all-cause and vascular dementia. Alzheimer’s Dementia. 20(9), 5973–5995. https://doi.org/10.1002/alz.14115 (2024).
Ossenkoppele, R. et al. Prevalence of amyloid PET positivity in dementia syndromes: A meta-analysis. JAMA 313(19), 1939–1949. https://doi.org/10.1001/jama.2015.4669 (2015).
Liu, C.-C. et al. ApoE4 accelerates early seeding of amyloid pathology. Neuron https://doi.org/10.1016/j.neuron.2017.11.013 (2017).
Paris, D. et al. Vasoactive effects of A beta in isolated human cerebrovessels and in a transgenic mouse model of Alzheimer’s disease: role of inflammation. Neurol Res. 25(6), 642–651 (2003).
Thomas, T., Thomas, G., McLendon, C., Sutton, T. & Mullan, M. beta-Amyloid-mediated vasoactivity and vascular endothelial damage. Nature 380(6570), 168–171 (1996).
Palmer, J. C., Barker, R., Kehoe, P. G. & Love, S. Endothelin-1 is elevated in Alzheimer’s disease and upregulated by amyloid-β. J Alzheimers Dis. 29(4), 853–861. https://doi.org/10.3233/JAD-2012-111760 (2012).
Chen, F. et al. The vascular contribution of apolipoprotein E to Alzheimer’s disease. Brain 147(9), 2946–2965. https://doi.org/10.1093/brain/awae156 (2024).
Altmann, A., Tian, L., Henderson, V. W. & Greicius, M. D. Sex modifies the APOE-related risk of developing Alzheimer disease. Ann. Neurol. 75(4), 563–573. https://doi.org/10.1002/ana.24135 (2014).
Neu, S. C. et al. Apolipoprotein E genotype and sex risk factors for Alzheimer disease: A meta-analysis. JAMA Neurol. 74(10), 1178–1189. https://doi.org/10.1001/jamaneurol.2017.2188 (2017).
Mosconi, L. et al. Sex differences in Alzheimer risk: Brain imaging of endocrine vs chronologic aging. Neurology 89(13), 1382–1390. https://doi.org/10.1212/WNL.0000000000004425 (2017).
Mosconi, L. et al. Menopause impacts human brain structure, connectivity, energy metabolism, and amyloid-beta deposition. Sci Rep. 11(1), 10867. https://doi.org/10.1038/s41598-021-90084-y (2021).
Viticchi, G. et al. Framingham risk score and the risk of progression from mild cognitive impairment to dementia. J. Alzheimers Dis. 59(1), 67–75. https://doi.org/10.3233/JAD-170160 (2017).
Suri, S. et al. Reduced cerebrovascular reactivity in young adults carrying the APOE ε4 allele. Alzheimers Dement. https://doi.org/10.1016/j.jalz.2014.05.1755 (2015).
Moreau, K. L. & Hildreth, K. L. Vascular aging across the menopause transition in healthy women. Adv. Vasc. Med. 2014(1), 204390 (2014).
Guo, W. et al. Effect of menopause status on brain perfusion hemodynamics. Stroke 55(2), 260–268. https://doi.org/10.1161/STROKEAHA.123.044841 (2024).
Rosenzweig, N. et al. Sex-dependent APOE4 neutrophil-microglia interactions drive cognitive impairment in Alzheimer’s disease. Nat. Med. 30(10), 2990–3003. https://doi.org/10.1038/s41591-024-03122-3 (2024).
Nemes, S. et al. Sex and APOE ε4 carrier effects on atrophy, amyloid PET, and tau PET burden in early-onset Alzheimer’s disease. Alzheimers Dement. 19(Suppl 9), S49–S63. https://doi.org/10.1002/alz.13403 (2023).
Funding
This study was funded by the National Natural Science Foundation of China (Grant numbers 81772454 and 81971237), the Jiangsu Municipal Science and Technology Bureau (BE2017734) and the Key Project of Jiangsu Province's Key Research and Development Program (BE2023023-2).
Author information
Authors and Affiliations
Contributions
TW and QHL conceived and proposed the initial hypotheses for the study. QCC, JZ, TW, and QHL designed the study’s framework. JZ, SIY, and XYD were responsible for recruiting and screening subjects. QHL provided the imaging equipment. QCC, JZ, WJC, SGY and YCW conducted image acquisition and organized the data. JZ, WJC and SGY performed the statistical analysis and interpreted the results. QCC and JZ drafted the initial manuscript, while WYW, WTG, JZ, WRX, PY, TW, and QHL reviewed the manuscript and offered detailed feedback. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, J., Cao, Q., Chen, W. et al. Compensatory retinal blood flow enhancement in cognitively normal ApoE ε4 carriers. Sci Rep 15, 32349 (2025). https://doi.org/10.1038/s41598-025-16770-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-16770-3