Abstract
This study investigates the potential of valorized wood bark waste as an efficient, low-cost adsorbent for removing Pb(II) ions from aqueous solutions. Valorization via pyrolysis significantly enhanced the surface area (from 105.3 to 344.7 m²/g) and pore volume (from 0.168 to 0.421 cm³/g), promoting higher adsorption capacity. Characterization using FT-IR, SEM-EDS, BET, and TGA confirmed structural and chemical modifications that improved Pb(II) uptake. Batch experiments optimized parameters such as pH (optimal at 5), contact time (100 min), temperature (55 °C), and adsorbent dosage (0.5 g). The valorized adsorbent (VWBWA) demonstrated superior performance, achieving a maximum adsorption capacity of 114.03 mg/g, nearly twice that of the raw form (59.20 mg/g). Adsorption followed both Langmuir and Freundlich isotherms, and kinetic data fit pseudo-second-order and intraparticle diffusion models, indicating chemisorption and pore diffusion mechanisms. Thermodynamic analysis revealed the process was spontaneous and endothermic, with higher enthalpy (39.54 kJ/mol) and entropy (13.44 J/mol·K) changes for VWBWA. Desorption studies confirmed reusability, with acetic acid showing optimal elution. These findings establish valorized wood bark waste as a promising and sustainable biosorbent for lead remediation in industrial wastewater treatment, aligning with circular economy and waste valorization principles.
Similar content being viewed by others
Introduction
The environment surrounding all living beings is crucial for survival and a clean and unpolluted ecosystem is essential for a healthy life1. An environment that is contaminated poses significant health risks to man and animals due to exposure to harmful chemicals. Pollution has worsened due to human and industrial activities, with substantial pollutant discharges into marine ecosystems, thus threatening coastal environments worldwide2,3. Mining of metals and other valuable minerals, and other industrial activities are significant contributors to environmental pollution and pollutants released from these operations negatively impact both the environment and living beings, especially with long-term exposure3,4. Among these metals that is often released to the environment is lead (Pb) which is the second most toxic metal, making up just 0.002% of Earth’s crust2. While naturally present in small amounts, its production is largely driven by human activities, including industrial processes, and automobiles2. Exposure primarily occurs through Pb salts and oxides found in dust, paints, battery emissions, industrial wastes, lead-based paint, and contaminated food5,6. The acceptable lead concentration in the environment ranges from 0.10 to 0.30 µg/m³2. As a result, both humans and the environment suffer from lead pollution and due to excessive pollution, while people living close to such environs often suffer from long-term respiratory and other diseases caused by the constant inhalation of dust particles7,8.
Lead is classified as carcinogen for humans and approximately 30–60% of ingested Pb is absorbed by the body, posing serious health risks2. Lead exposure can have harmful effects on nearly every organ and system in the human body, with children under six being particularly vulnerable7. In pregnant women for instance, lead stored in their bones can be released and cross the placental barrier, exposing the fetus to poisoning, which may result in reduced fetal growth, premature birth, and severe health complications for both the mother and baby7,8,9,10. Lead in children’s blood can lead to hearing and learning difficulties, anemia, behavioral issues, stunted growth, lower intelligence quotient, and hyperactivity9,10. In adults, prolonged lead exposure can lead to serious health issues, high blood pressure, including cardiovascular problems, malfunction of kidney and could contribute to reproductive issues7,8,9,10,11. The widespread effects of lead poisoning highlight the importance of reducing exposure, particularly in vulnerable populations such as children and pregnant women, to prevent long-term health consequences.
Various methods are used to remove lead ions from water and wastewater, and they include precipitation, coagulation-flocculation, ion exchange, adsorption, advanced oxidation, and membrane separation. However, precipitation and coagulation-flocculation generate large amounts of sludge and may not effectively reduce lead levels to safe limits. Ion exchange, while effective, is costly and requires pretreatment, as organic matter in wastewater can clog the exchanger matrices7,12,13. Adsorption is widely recognized as the most commonly used method for removing lead and other heavy metal ions from water and wastewater due to its simplicity, eco-friendly and do not generate secondary by-product14. Due to the intrinsic physicochemical properties and rich contents of lignocellulosic, the residues of agro-waste are increasingly being recognized as valuable resources for water treatment which aligns with growing governmental support for zero-waste initiatives and circular economy strategies15. One promising techniques for converting such agro-waste into useful products is carbonization, which offers an efficient and sustainable valorization pathway15. The role of adsorption method in the treatment of wastewater is notable for its simplicity, cost-effectiveness, and efficiency through the use of cheap materials, including raw or treated biomass, and activated biomass and their potential for reuse in treatment processes16. The development of cost-effective and cheap adsorbents from agricultural by-product for removing lead ions from water and wastewater is crucial as recent studies have reported commercial activated carbon (AC) to be expensive. Some of the adsorbents that have been successfully used for lead uptake from contaminated sites are acid activated bentonite clay17, modified coal fly ash18, sphagnum peat moss19, carbonized sawdust20, wheat straw21, agave bagasse22, and magnetic graphene oxide23. These adsorbents showed promise for being both affordable and efficient in lead removal. While various biomass materials have been explored as adsorbents, the use of valorised wood bark have not been comprehensively studied. Thus, turning wood bark waste into a value added product like adsorbent promotes circular economy principles.
This study introduces a novel, low-cost, and eco-friendly approach for the removal of toxic Pb²⁺ ions from aqueous systems using pyrolyzed wood bark waste which is a readily available and largely underutilized byproduct of the timber industry. Unlike conventional methods that require chemical activation or expensive commercial adsorbents, the valorization process involved the used thermal treatment alone, avoiding hazardous reagents while enhancing the adsorbent’s surface area and porosity. This chemical-free pyrolysis not only ensures environmental safety during preparation but also aligns with the principles of green chemistry and circular economy, offering a sustainable solution for waste-to-resource conversion. Given the widespread industrial use and environmental release of Pb²⁺, the development of efficient, accessible, and regenerable adsorbents is a critical need. Our valorized wood bark adsorbent serves as simple material for Pb²⁺ removal. as a promising, sustainable, and scalable alternative in real-world wastewater treatment applications. This research thus aimed at the use of valorised wood bark as an adsorbent for the removal of lead ions from aqueous solution via batch process and also optimize adsorption parameters (like pH, contact time, temperature, adsorbent dosage) for maximum Pb(II) removal efficiency. The physical properties of the wood bark waste adsorbent was examined using different spectroscopic and microscopic techniques like FT-IR, SEM-EDS, and TGA. Kinetics, thermodynamic and isotherms were evaluated to determine the nature of the adsorption process.
Methods
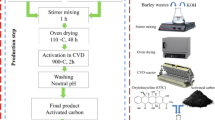
Pre-treatment of wood bark waste
The wood bark waste was obtained from a wood local factory at Ifo, Ogun State of Nigeria, washed with distilled water, dried to remove moisture and ground into a fine powder. Pyrolysis of the dried sample (100 g) was done at a temperatures of 600 °C in an inert atmosphere of furnace. Thereafter, the sample was removed from the furnace and allowed to cool down. The product formed was labeled as valorised wood bark waste adsorbent (VWBWA), while the portion left uncalcined was referred to as wood bark waste adsorbent (WBWA).
Characterization of adsorbent
Fourier transform infrared spectroscopy (FT-IR) (Agilent Cary 630 FTIR spectrometer, USA) was used to identified the functional groups, while Scanning electron microscopy (SEM, Phenom ProX by PhenomWorld Eindhoven, Netherlands) was used to examined the surface morphology and porosity. Brunauer-Emmett-Teller (BET) analysis was used to measured surface area and pore volume using Quantachrome corporation, (NOVA 4000e 1, Florida, USA). The thermogravimetric analysis (TGA) and differential thermal analysis (DTA) were measured using TGA instrument (TGA-4000, Perkins Elmer, Netherlands).
Preparation of Pb(II) stock solution
All the chemical used were purchased from Sigma-Aldrich, India and used without any forms of purification. 1000 mg/L Pb(II) stock solution was prepare using lead nitrate [Pb(NO₃)₂] dissolved in deionized water. Using this stock solution, different Pb(II) working solutions (50, 100, 150, 200, 250, and 300 mg/L) were prepared by appropriate dilution with deionized water. Adjustment of the pH of the solutions was done using 0.1 M HCl or 0.1 M NaOH, as required. The Point of zero charge (pHPZC) was done in which 25 mL solution of 0.01 M KNO₃ was transferred into conical flasks, and its initial pH was adjusted to a range of 2 to 8 using 0.1 M HCl or NaOH solutions. Subsequently, 0.5 g of the adsorbent was added to each flask, stirred, and left for 24 h, after which the final pH was measured. The pHPZC value was determined from the point where the plot of ΔpH against pH₀ intersects the pH line.
Adsorption experiments
Batch adsorption studies was utilized to analyzing the Pb(II) uptake under different conditions such as pH, adsorbent dose, pollutant concentration, temperature, and contact time. The experiments were perform in 250 mL Erlenmeyer flasks containing 50–300 mL of Pb(II) solution at a fixed volume and varying conditions. 0.5 g amount of the adsorbent was added to 50 mL in several 250 mL Erlenmeyer flasks and the pH adjusted. The content was stirred at 150 rpm at room temperature (25 °C). At a predetermined time, the content was separated, and the remaining Pb(II) concentration was measured using Atomic Absorption Spectroscopy (AAS) (iCE™ 3500, Thermo Fisher Scientific, Waltham, MA, USA). The parameters varied are: contact time (10 to 240 min), adsorbent doses (0.1 to 1.0 g), and temperature (25, 35, 45, 55, 65, and 75 °C).
The amount of lead ions sorbed and the percentage removal were computed from Eqs. 1 and 2 respectively:
With Qe (mg/g) denoting lead ions amount sorbed, V represent lead ions volume used in L, Co and Ce stand for initial and final concentrations of lead ions before and after adsorption in mg/L respectively. The experiments were performed in triplicate and the average value was used.
Desorption studies
To evaluate the feasibility of the biosorption process, sorption-desorption studies were performed on the used adsorbent. After equilibrium was attained, the exhausted adsorbent were carefully separated, and desorption was conducted using acitic acid as desorbing agent. After each process, the concentration of the lead ions washed was measured using a Atomic Absorption Spectroscopy (AAS) (iCE™ 3500, Thermo Fisher Scientific, Waltham, MA, USA). 0.1 M acetic acid was contacted with the adsorbent and equilibrated for 60 min at 150 rpm. The mixture was then filtered, and the filtrate was analyzed using Atomic Absorption Spectroscopy (AAS) (iCE™ 3500, Thermo Fisher Scientific, Waltham, MA, USA). This procedure was repeated for five cycles, following the same experimental protocol, and the percentage desorption (PD) was calculated using Eq. 3 below:
Where AD denote the amount (mg) of lead ions desorbed, and AA represent lead ions amount (mg) adsorbed.
Results and discussions
Thermogravimetric analysis (TGA) and differential thermal analysis (DTA)
The information on Thermogravimetric Analysis (TGA) and Differential Thermal Analysis (DTA) of wood bark adsorbent is shown in Fig. 1. The TGA curve indicates mass loss as shown in the sample with initial mass loss in the range of 100 to 200 °C which is likely due to moisture evaporation and release of volatile compounds24,25. The major decomposition occurred between 200 and 500 °C showing weight loss, indicating thermal degradation of organic components such as hemicellulose, cellulose, and lignin. Hemicellulose decomposes between 200 and 300 °C, cellulose decomposes around 300–400 °C, and lignin decomposes over a broader range (250–600 °C). The decomposition pattern aligns with typical lignocellulosic biomass pyrolysis, where hemicellulose, cellulose, and lignin break down in distinct stages24,25. The DTA shows endothermic and exothermic reactions during heating. Peak in the range of 350 to 450 °C corresponds to the maximum decomposition rate of cellulose and lignin. The TGA curve indicates that beyond 500 °C, most of the volatile matter has been lost, leaving behind carbonaceous char. Chen et al.24. in their study on the evaluation of agricultural residues pyrolysis noted three major decomposition stages which include initial weight loss due to moisture evaporation, next by the degradation of hemicellulose which took place at around 200–300 °C, the second stage cellulose, between 300 and 400 °C, and finally lignin, which decomposed between 550 and 600 °C. Also, ElSayed et al.25. reported similar trends, with hemicellulose decomposition occurring between 135 and 275 °C, cellulose in the region of 275–358 °C. These temperature ranges closely associated with the TGA curve of the wood bark adsorbent shown in Fig. 1 of the present study, which indicates an initial mass loss between 100 and 200 °C due to moisture and volatile release, followed by major decomposition between 200 and 500 °C corresponding to thermal breakdown of hemicellulose, cellulose, and lignin.
Thermogravimetric analysis (TGA) and differential thermal analysis (DTA) of wood bark adsorbent.
Scanning electron microscopy (SEM)
The SEM provide insight into the morphological changes in wood bark waste adsorbent at different stages of valorization and adsorption which is shown in Fig. 2. SEM image (a) denotes the wood bark waste adsorbent before valorization and the surface appears relatively compact, with a less porous and rough texture26. There are fewer visible cavities or cracks, suggesting limited surface area available for adsorption. The structure looks relatively dense, indicating that it has not undergone any modification to enhance porosity. Image (b) shows the adsorbent after valorization before adsorption. The surface now appears more porous, with larger cavities and cracks and this increase in porosity suggests that the valorization process has modified the structure, potentially enhancing its adsorption capacity26,27. The rougher texture and increased void spaces indicate improved accessibility for adsorbates. Image (c) shows the valorised adsorbent after adsorption, and it was observed that some of the visible pores and cavities seem to be partially filled, indicating the adsorption of the contaminants. The structure appears more compact compared to (b), suggesting that adsorption has led to material deposition within the porous network and this confirms that the valorized wood bark waste has effectively captured and retained the adsorbates. ElRayyes et al.26 noted that the surface of untreated wood biochar with initial dense, and compact structure on post-valorization, reveals a rough and highly porous surface riddled with cavities. After the uptake of the contaminant, pores blocking, smoother and denser surface were observed, confirming effective uptake. Ighalo and Adeniyi27 observed that the roughness and porosity were enhanced after thermal treatments, and that the SEM images captured after adsorption displayed structural smoothing and pore filling.Another study on activated carbons (KOH-activated) as reported by Kang et al.28 pointed out that after adsorption, SEM images show significantly fewer pores supporting the phenomenon of pore blockage in this current study.
SEM results for wood bark waste before calcination (a), after calcination (b), and calcined product after lead ions adsorption (c).
FT-IR results
The FT-IR analysis which provides information about the functional groups present in the wood material and how they change after calcination and Pb²⁺ adsorption is presented in Fig. 3. Before calcination (Fig. 3a), broad O–H stretching peak around 3310–3550 cm⁻¹ was seen, indicating the presence of hydroxyl (–OH) groups from lignin, cellulose, and hemicellulose29. The peak attributed to C–H stretching was observed at 2975 cm⁻¹corresponding to aliphatic compounds in organic biomass, while C = O stretching was found at 1780 cm⁻¹which could be from carboxyl and carbonyl functional groups. Peaks at 1675 cm⁻¹ correspond to C = C stretching, characteristic of lignocellulosic components29. Multiple peaks corresponding to C–O and aromatic C = C stretching were found at around 1200–1600 cm⁻¹ which is due to lignocellulosic components. Peaks related to C–O, and Si–O groups were found around 1050–1125 cm⁻¹29. After calcination (Fig. 3b), the broad O–H peaks initially seen at around 3310–3550 cm⁻¹ is reduced drastically, indicating dehydration and decomposition of hydroxyl-containing compounds30. The disappearance or weakening of C-H stretching (2975 cm⁻¹) confirms the breakdown of organic matter, while the formation of stronger C = O at around 1780 cm⁻¹ suggests the formation of carbonyl-rich surface30. The decrease in the intensity of the peaks corresponding to Si-O and C-O at around 1050–1125 cm⁻¹, indicate dehydration and decomposition of hydroxyl-containing compounds30.
After Pb²⁺ adsorption (Fig. 3c), shift and broadening of O-H stretching was seen with new peaks at 3450 to 3560 cm⁻¹ suggests Pb²⁺ binding with hydroxyl groups via hydrogen bonding or electrostatic interactions31. The broadening of the peak may be as a result of soaking the calcined adsorbent in aqueous medium of Pb2+ salts during the adsorption process. The reduction in C = O peak intensity at 1745 cm⁻¹ indicates the likely involvement of carboxyl groups in Pb²⁺ adsorption via ion-exchange or coordination31. The peaks around 500–700 cm⁻¹ corresponding to metal-O stretching vibrations, confirming successful Pb²⁺ adsorption. The Pb²⁺ ions interact mainly with oxygen-containing functional groups (O-H, C = O, and Si-O), and as the surface becomes saturated with Pb²⁺, it alters the peak intensities and positions. This analysis confirms that calcined wood bark waste is an effective adsorbent for Pb²⁺ removal, and the functional groups play a crucial role in the adsorption mechanism.
In the study conducted on beech wood subjected to heat treatment by Timar et al.29, exhibited significant changes in the FTIR spectra with decrease in the intensity of the C = O stretching vibration at 1643 cm⁻¹, indicative of degradation of conjugated carbonyl groups. Simultaneously, lignin-associated peaks at 1506 and 1595 cm⁻¹ increased, indicating lignin condensation or structural rearrangement which is similar to what is observed after calcination in this present study. In a study conducted by Will30, it was observed that the presence of typical functional groups like broad O–H stretches (~ 3300–3550 cm⁻¹), aliphatic C–H (~ 2900 cm⁻¹), and C = O stretches (~ 1700–1780 cm⁻¹) can be seen in different wood species which are similar to what was observed in raw wood bark prior to calcination in this study as depicted in Fig. 3a. Further, El Hajam et al.31 studied chemically activated sawdust and found that after acid-base treatment, O–H stretching bands (around 3390 cm⁻¹) weakened, while lignin and C–O–C peaks at 1500, 1381, and 1163 cm⁻¹ diminished significantly, confirming decomposition of oxygenated groups and enhancement of cellulose content which is closely related to our observations on post-calcination (Fig. 3b). After metal adsorption, El Hajam et al.31. noted shifts in O–H peaks and the appearance of metal–O vibrations between 500 and 700 cm⁻¹, confirming the binding of Pb²⁺ to oxygen-containing functional groups and this directly aligns with our post-adsorption results (Fig. 3c), where the broadening of the O–H band and emergence of metal–O signals confirms the interaction of Pb²⁺ ions with hydroxyl, carbonyl, and silanol groups.
FT-IR of wood bark waste before calcination (a), after calcination (b), and adsorbed lead ions wood bark waste (c).
The BET (Brunauer-Emmett-Teller) and BJH (Barrett-Joyner-Halenda) analysis, which provides the specific surface area, pore size and pore volume of the wood bark waste adsorbent is shown in Fig. 4. The BET shows surface area of 105.313 m²/g and 344.703 m²/g for raw and heat treated samples respectively, as higher BET surface area generally improves the adsorption capacity of materials since it provides more available surface sites for metal ion interactions. The value of 344.703 m²/g for the heat treated sample suggests that the wood bark waste is highly porous and can effectively adsorb Pb²⁺ ions. The pore volume for the raw and calcined samples are 0.168 cm3/g and 0.421 cm3/g respectively. The pore volume represents the total space available for Pb²⁺ ion accommodation within the adsorbent, as higher pore volume means more Pb²⁺ ions can diffuse into the adsorbent, leading to increased adsorption efficiency. The pore size obtained are 2.062 nm and 5.108 nm for the raw and calcined samples respectively. These pore sizes fall within the mesoporous range (2–50 nm), meaning the material is a mesoporous adsorbent. Mesopores materials are ideal for heavy metal adsorption, as they allow ions to diffuse efficiently into the material without blocking. Pb²⁺ ions (hydrated radius ~ 0.24 nm) can easily enter 2.062–5.108 nm pores, allowing for deep penetration and strong adsorption.
Mabaso et al.32. examined bamboo-chopstick biochar and reported BET surface areas ranging from 165 to 414 m²/g and pore sizes between 3.7 and 10.6 nm, confirming mesoporous structure and linking enhanced surface area and porosity to improved adsorption of heavy metals including Pb²⁺ which is consistence with our findings. In a similar vain, Rahimi et al.33 characterized biomass-derived carbons from almond shell and olive stone treated at 600 °C emphasized that higher BET surface area and pore volume enhanced Pb²⁺ and Cu²⁺ adsorption performance, inferring that greater surface area improves the diffusion channels significantly boost adsorption efficiency. Roy et al.34 reported that pyrolyzed mesoporous activated carbon from biomass waste, resulted in large surface area and increased pore volume which are standard criteria for efficient adsorbents in the sorption of adsorbate which is similar to the obtained results in this current study.
BET (Brunauer-Emmett-Teller) and BJH (Barrett-Joyner-Halenda) analysis.
Adsorption trend over time and initial Pb2+ concentration
The results on the adsorption of lead ions by wood bark waste adsorbent at different initial lead ion concentrations (50 mg/L to 300 mg/L) over time (0 to 180 min) is shown in Figs. 5 and 6 respectively. At all initial lead ion concentrations, adsorption increases rapidly in the first 30 min due to available vacant sites on the adsorbent surface. After 120 and 100 min for WBWA and VWBWA, adsorption reaches equilibrium, as indicated by the constant values in subsequent time intervals (120–180 min), and this suggests that the adsorption sites on the wood bark waste become saturated over time. Such behavior aligns with findings from studies on the adsorption of heavy metals using biosorbents derived from organic waste by Tang35. The adsorption process is greater and faster in the valorized wood bark waste and by 100 min, the valorized adsorbent achieves approximately 109.3 mg/g (300 mg/L), whereas the non-valorized one only reaches 50.8 mg/g at 120 min. The enhancement in adsorption capacity and rate observed in valorized wood bark waste adsorbents can be attributed to the increased accessibility and availability of binding sites resulting from the valorization process. This improvement facilitates more efficient capture of lead ions, as evidenced by studies demonstrating that modifications of biosorbents can significantly enhance their heavy metal uptake capabilities20,35.
For the effect of initial concentration, higher initial lead ion concentrations resulted in greater adsorption capacity. For example, at equilibrium (100 and 120 min onward), adsorption reaches approximately 16.1 mg/g and 21.8 mg/g for WBWA and VWBWA at 50 mg/L but 109.3 mg/g (valorized) compared to 50.5 mg/g (non-valorized) at a higher lead concentration of 300 mg/L, more than double the adsorption capacity. This indicates that wood bark waste has a higher adsorption capacity when lead ion concentration increases, likely due to a greater concentration gradient driving the mass transfer36. For instance, studies have demonstrated that increasing the initial metal ion concentration resulted in higher adsorption capacities13,36. At all lead ion concentrations and contact time tested, the valorized wood bark waste adsorbent demonstrates a higher adsorption capacity compared to the non-valorized version. Valorization processes, enhance the adsorbent’s ability to capture lead ions by increasing surface area and porosity. These treatment remove components like hemicellulose and lignin, resulting in a more porous structure and enlarged specific surface area, which facilitate greater interaction between heavy metal ions and the adsorbent36,37.
Mabaso et al.32 observed that the sorption of Pb(II) by biochar adsorbents obtained at a temperature of 450 °C W and 700 °C occurred in two stages with a very fast step at the commencement (from 0 to 1 h) and a second phase of a slow step (from 1 to 24 h) attaining maximum efficiencies of 99.9% and 99.8% for adsorbent obtained at 450 and 700 °C at 24 h respectively. This was alluded to the large surface areas observed for the biochars, but the removal of Pb(II) using biochar obtained at 200 and 500 °C revealed lower removal efficiencies under the same experimental condition, resulting in lower removal efficiency when compared with biochar at 450 and 700 °C which was attributed to the lower surface area, pore volume, and accessibility of available adsorption sites32. Natrayan et al.38. observed the influence of contact time on the sorption of Pb2+ and noted that Pb ion concentration rose with the increase in the equilibrium duration until saturation was attained and this phenomenon was said to be due to the presence of greater active zones for adsorptions. A similar observation was reported by Chen et al.39.
Adsorption of lead ions by raw wood bark waste adsorbent at different initial lead ion concentrations (50 mg/L to 300 mg/L) over time (0 to 180 min).
Adsorption of lead ions by valorised wood bark waste adsorbent at different initial lead ion concentrations (50 mg/L to 300 mg/L) over time (0 to 180 min).
Effect of adsorbent dosage
Results presented in Fig. 7 shows the effect of adsorbent dosage (g) on the adsorption of lead ions using wood bark waste adsorbent (WBWA) and valorized wood bark waste adsorbent (VWBWA). It was observed that as the adsorbent dosage increases from 0.1 g to 0.5 g, the adsorption capacity increases for both WBWA and VWBWA. The valorized wood bark waste adsorbent consistently shows higher adsorption capacity compared to WBWA, confirming the improved efficiency of the valorized material. Increasing adsorbent dosage increases adsorption because more surface area and active binding sites are available for lead ion removal40. However, after 0.5 g, the adsorption capacity levels off and even slightly decreases (72.2 to 69.3% for WBWA and 88.4 to 85.2% for VWBWA). This decline at higher dosages suggests that adsorbent saturation occurs, meaning additional adsorbent does not improve lead removal. This could be due to particle aggregation, reducing the effective surface area. Also, saturation of available binding sites, where excess adsorbent does not contribute to further adsorption could also caused this decline41.
Ezeonuegbu et al.42. observed that the osorption efficiency of sugarcane bagasse rose with increase in adsorbent dosage from 0.3 to 0.5 g and this was attributed to an increase in the number of active sites available for the pollutant adsorption. They reported further that when the adsorbent dosage was increased beyond 0.5 g, no significant increase in pollutant removal efficiency was seen and this was ascribed to overlapping of adsorption sites as a result of overloading of the adsorption site42. The trend observed from this study is consistent with recent studies where increasing the adsorbent dosage provides more available binding sites, improving metal uptake; and beyond this, slight decline in adsorption capacity were common which was attributed to particle aggregation, that reduces effective surface area per unit mass, and binding site saturation, as as such, additional adsorbent no longer contributes to adsorption38,43,44.
Adsorption of lead ions by raw and valorised wood bark waste at different adsorbent dosage.
Trend of pH influence
The data presented in Fig. 8 illustrates the influence of pH (conducted in the pH range of 2 to 8) on the sorption of Pb2+ using wood bark waste adsorbents (WBWA) and valorized wood bark waste adsorbents (VWBWA). A crucial factor governing this characteristics properties of adsorbent is the point of zero charge (pHpzc) of the adsorbents. For WBWA and VWBWA, the pHpzc values were determined to be 4.13 and 4.25, respectively and at solution pH values above these points, the surface of the adsorbents acquires a net negative charge, enhancing the electrostatic attraction between the adsorbent and positively charged Pb²⁺45.
The sorption capacity of both WBWA and VWBWA rose steadily as the pH increase from 2 to 5, reaching a maximum at pH 5 (71.6% for WBWA and 86.4% for VWBWA). This trend is consistent with the progressive reduction in surface protonation and the enhanced availability of active binding sites. At low pH (2–3), adsorption was considerably lower due to the high concentration of H⁺, which compete with Pb²⁺ for the same binding sites. As the pH approaches and exceeds the pHₚzc, this competition diminishes, and the surface becomes more negatively charged, allowing greater Pb²⁺ uptake via electrostatic attraction with negatively charged functional groups like hydroxyl and carboxyl moieties, as also confirmed by FT-IR analysis45,46,47. The role of metal speciation is also vital in interpreting this property. Between pH 4 and 6.5, Pb²⁺ exists predominantly as free divalent ions (Pb²⁺), which is the most adsorbable form due to its strong electrostatic interaction with negatively charged surfaces. However, above pH 6, a decline in adsorption is observed which is most notably at pH 8 which can be attributed to the precipitation of Pb²⁺ as Pb(OH)₂, effectively reducing the concentration of free Pb²⁺ available for adsorption47. This phenomenon aligns with earlier reports where the optimal pH for Pb²⁺ adsorption was around 3.0–5.0, with decline performance at higher pH values due to metal hydroxide precipitation7,46,47,48.
Adsorption of lead ions by raw and valorised wood bark waste adsorbent at different pH.
Kinetics of lead ion adsorption by WBWA and VWBWA
Pseudo-first-order kinetics
The pseudo-first-order model assumes that adsorption occurs primarily through physisorption (physical attraction) and its linear form is given as13,49,50:
Where qt and qe are the adsorption capacities at time t and equilibrium, respectively (mg/g), and k1 is the rate constant (min− 1). By plotting log(qe−qt)against t, a straight line with slope − k1/2.303 and intercept logqe can be obtained as shown in Fig. 9a with constant values listed in Table 1. The experimental adsorption capacity (Qe(exp)) values are slightly different from the calculated values (Qe(cal)), while the rate constant (k1) for VWBWA is higher than WBWA across all concentrations, indicating faster adsorption kinetics in VWBWA due to enhanced surface properties. The correlation coefficient (R²) values are high (> 0.99 for most cases), meaning the first-order model fits the data well, especially for lower concentrations (50–200 mg/L). Sum of squared errors (SSE) values are low, further confirming a good fit for this model. The PFO model predicts the physical adsorption of Pb2 onto the adsorbent, where the adsorption rate is directly related to the active adsorption sites availability on the adsorbent (Haider et al. 2025). The results from the study carried out by Haider et al.51 indicated that the kinetic models analysis confirmed that the PFO gave the best fit for Pb adsorption which is consistent with our results, as identified the PFO model to be suitable for the adsorption kinetics of Pb2+. Similar report was observed by Ngueagni et al.43 where the uptake of Pb2+ from aqueous solutions achieved via calcined cow dung adhered strong with the PFO model which also corroborate our findings in this present study.
Pseudo-second-order kinetics
The pseudo-second-order model assumes that the adsorption process is chemically controlled (chemisorption) and involves valency forces or electron sharing between Pb²⁺ ions and the adsorbent surface and the linear form can be expressed as13,49,50:
Where k2 denotes the rate constant (g/mg·min). By plotting t/qt against t as shown in the plots in Fig. 9b can be obtained and parameters listed in Table 1. The Qe(cal) values from the second-order model are closer to Qe(exp) than in the first-order model, indicating a better fit and the rate constant (k2) for VWBWA is higher than WBWA across all concentrations, meaning VWBWA reaches equilibrium faster due to improved surface area and functional groups from valorization. The correlation coefficient (R²) values are higher (> 0.99 for VWBWA, and > 0.89 for WBWA), confirming that the second-order model also fit the kinetics data. SSE values are lower in the second-order model compared to the first-order model, reinforcing the suitability of this model as well. The sorption of Pb²⁺ onto the calcined wood bark was best described by the pseudosecondorder (PSO) model, with superior R² values and close agreement between modeled and experimental qe values. This is an indication that the rate-limiting step involves chemisorption, possibly via electron exchange or sharing between the surface functional groups like hydroxyl and carbonyl groups and the Pb²52. Similar reports have been documented in other studies where lignocellulosic materials, like modified sawdust31 and acid-treated banana peel biochar41 where pseudo-second-order kinetics were attributed to specific interactions between oxygen-containing groups and metal ions. Additionally, while the PSO model often correlates with chemisorption, recent analyses by Hubbe et al.53 propose that PSO fitting can also reflect diffusion-limited processes, especially in porous, heterogeneous materials. This is in agreement with our findings from intra-particle diffusion modeling, which showed a multi-step adsorption involving both pore diffusion and boundary-layer control.
Kinetics plots of lead ion adsorption by WBWA and VWBWA from (a) pseudo-first-order, (b) pseudo-second-order, and (c) intraparticle diffusion models.
Intraparticle diffusion model
The intraparticle diffusion model is used to determine if diffusion through pores controls adsorption. The linear form can be expressed as13,54:
Where ki is the intraparticle diffusion rate constant (mg/g·min1/2) and Ci represents the boundary layer effect. The plot of qt against t1/2 is shown in Fig. 9c, while their constants are listed in Table 1. The diffusion rate constant (Kp) is higher for VWBWA than WBWA, indicating faster internal diffusion, likely due to the improved pore structure in VWBWA. The R² values are high (~ 0.99), indicating that intraparticle diffusion plays a role but may not be the only controlling mechanism. The transport of molecules within particles generally occurs in three key stages: (1) external mass transfer also known as film diffusion, is where molecules travel from the bulk solution to the outer surface of the solid particle; (2) intraparticle diffusion which is also called pore diffusion, is where molecules penetrate the internal pores of the solid, often serving as a rate-limiting step, particularly in microporous materials; and (3) interaction with binding sites on the solid surface55,56. While intraparticle diffusion plays a role in adsorption, the fact that the plots do not pass through the origin suggests that additional mechanisms, such as surface adsorption and chemical interactions, also contribute to the process.
Adsorption isotherms for Pb(II) uptake by WBWA and VWBWA
Langmuir isotherm model
The Langmuir model assumes that adsorption occurs at specific homogeneous sites on the adsorbent surface and that once a site is occupied, no further adsorption can occur at that location (monolayer coverage) and its linear form expressed as13,57:
Where Qm is the maximum adsorption capacity (mg/g), and and b indicates the Langmuir constant in L/mg. The constants were obtained from Fig. 10a, while their values are provided in Table 2.
Separation factor (RL)
This is the separation factor also know as the dimensionless constant from the Langmiur isotherm which determines the favorability of adsorption and can be represented as4,13:
If 0 < RL<1, the adsorption is favorable, if RL>1, it’s unfavorable adsorption, when RL=1, it’s a linear adsorption, and when RL= 0, it’s said to be irreversible adsorption4,13. It was observed that the maximum adsorption capacity for VWBWA is 114.032 mg/g which is greater than that of WBWA (59.196 mg/g) and this means that VWBWA can adsorb almost twice the amount of Pb²⁺ ions compared to WBWA, indicating that valorization enhances adsorption capacity. The RL values for WBWA is 0.423 indicating moderate adsorption favorability compared to VWBWA with value of 0.026 indicating highly favorable adsorption. Since 0 < RL < 1, the adsorption is favorable for both adsorbents, but VWBWA exhibits much stronger Pb²⁺ uptake due to lower RL values. From the values of R² obtained, (WBWA: 0.977 and VWBWA: 0.989), the model fit the adsorption data of the two adsorbents, though with better fir for VWBWA.
The findings from this study is consistence with the reports by Haider et al.51. where it was observed that Langmuir model best fit Pb adsorption with maximum sorption capacity of 43.7 mg g−1 and value of R² of 0.97. The obtained is also similar to literature report by Elkhaleefa et al.44. as Langmuir mode was indicated as the best applicable mode for describing Pb2+ uptake. In a different study by Ngueagni et al.43, Pb2+ adsorption by cellulose-base adsorbent of waste animal origin showed strong adherence to the Langmuir model. This is an indication that the adsorption occurred on a homogeneous monolayer surface, where each site can only accommodates one adsorbate molecule, and where all sites possess equal energy with negligible adsorbed species interactions43.
Isotherms plots for the uptake of Pb(II) by WBWA and VWBWA for (a) Langmuir and (b) Freundlich models.
Freundlich isotherm model
The Freundlich model describes adsorption on a heterogeneous surface with multiple binding sites and allows for multilayer adsorption and its linear form given as48,58:
Where KF (L/g) and n stand for Freundlich constants, indicating adsorption capacity and intensity. If 1/n is between 0 and 1, the adsorption is favorable, if n = 1, the adsorption is linear, indicating identical adsorption energies across sites, and if 1/n > 1, adsorption is unfavorable58. The graphical representation of this model is shown in Fig. 10b, and the values obtained are listed in Table 2. The values of KF obtained (VWBWA: 68.381 mg/g) and WBWA: 39.765 mg/g) further demonstrated that VWBWA has a higher Pb²⁺ uptake capacity, which is consistent with the Langmuir model results. The values of 1/n shows that 0 < 1/n < 1 for both adsorbents, indicating favorable adsorption, but VWBWA has a stronger adsorption affinity (lower 1/n). The R² values (WBWA: 0.944 and VWBWA: 0.955), suggest that the Freundlich model fits the adsorption data for the two adsorbent but with better with VWBWA. Isotherm modeling revealed that the Langmuir model provided a better fit than the Freundlich model, indicating monolayer adsorption on a homogeneous surface. The maximum Langmuir adsorption capacity of the calcined wood bark was 114.032 mg/g, which is comparable to or higher than values reported for other low-cost adsorbents such as carbonized sawdust (87.7 mg/g)20 and cold KOH activated camphor leaves biochar (90.09 mg/g)59.
Comparison of adsorption capacities of WBWA and VWBWA with other adsorbents for lead (Pb²⁺) removal
Table 3 presents the adsorption capacities (Qmax, mg/g) of various adsorbents, comparing them with wood bark waste adsorbent and valorized wood bark waste adsorbent for lead (Pb²⁺) removal and the goal is to evaluate the effectiveness of WBWA and VWBWA relative to other materials commonly used in adsorption studies. The results show that the VWBWA with adsorption capacity of 114.032 mg/g gave superior adsorption capacity, outperforming most conventional adsorbents.
Temperature influence and thermodynamic study
Figure 11a presents data on the effect of temperature on the adsorption of lead ions using WBWA and VWBWA and the results shows that for WBWA, adsorption remains relatively stable between 67.3% (25 °C) and 74.1% (45 °C), peaking at 45 °C, then slightly decreasing. For VWBWA, adsorption continuously increases from 63.2% (25 °C) to 87.3% (55 °C) and then remains stable at higher temperatures (85.4% at 65 °C). At lower temperatures (25–35 °C), the adsorption is lower because molecular movement is slower, leading to less interaction between Pb²⁺ ions and the adsorbent surface. At moderate temperatures (45–55 °C), both adsorbents show their highest adsorption capacities in this range. However, at higher temperatures (65–75 °C), WBWA slightly declines (72.1% at 75 °C), indicating possible desorption due to weakening interactions between Pb²⁺ and the adsorbent, but VWBWA maintains high adsorption efficiency (85.2% at 75 °C), suggesting it is more thermally stable. These observations align with findings from other studies, which indicate that temperature influences the adsorption capacity of biosorbents60. For example, a review on the removal of lead ions using various adsorbents noted that adsorption capacity can vary with temperature, depending on the nature of the adsorbent and the specific conditions employed7. Additionally, research on eucalyptus wood charcoal as a biosorbent for lead(II) ions highlighted the importance of considering temperature effects during adsorption studies37.
Haider et al.51. reported that the sorption of Pb increases with temperature up to an optimum around 37 °C, and thereafter declines beyond that temperature as a result of weakening interactions between Pb²⁺ and binding site which is consistent with our observation of a peak followed by a slight drop for WBWA. Also, Mohammed et al.64. in their study on the use of agricultural-waste-based biosorbents found optimal Pb removal efficiency to be around 99% in the temperature range of 30–50 °C, with little decline above.
Temperature effect (a) and thermodynamic analysis (b) of lead (Pb²⁺) adsorption by WBWA and VWBWA.
Gibbs free energy (ΔG), enthalpy (ΔH) and entropy (ΔS) changes were determined to further provide explanations into the adsorption process using the expressions listed in Eq. 9 to 1149,65:
Where KD denotes the equilibrium constant, R stands for the gas constant (8.314 J/mol·K), and T represent temperature (K). These parameters were obtained using Fig. 11b, and their values provided in Table 4. The ΔG values are negative at all temperatures, indicating that the adsorption of Pb²⁺ onto both WBWA and VWBWA is spontaneous and feasible60. As the temperature increases (from 298 K to 348 K), ΔG values become more negative, suggesting that adsorption becomes more favorable at higher temperatures. For WBWA, ΔG decreases from − 0.22 kJ/mol (at 298 K) to − 2.15 kJ/mol (at 348 K), and for VWBWA, ΔG decreases from − 0.51 kJ/mol (at 298 K) to − 3.5 kJ/mol (at 348 K). The higher magnitude of ΔG for VWBWA compared to WBWA suggests that VWBWA has a stronger driving force for Pb²⁺ adsorption. For the enthalpy change, WBWA had a value of 15.82 kJ/mol, while VWBWA had a value of 39.54 kJ/mol. Since ΔH is positive for both adsorbents, the adsorption process is endothermic, meaning that Pb²⁺ adsorption is more favorable at higher temperatures14. Since VWBWA has a higher ΔH value (39.54 kJ/mol) compared to WBWA (15.82 kJ/mol), it suggests that VWBWA undergoes a stronger heat-driven adsorption process. Thermodynamic analysis further showed that the positive ΔH° (in the range of 15–40 kJ/mol) suggests that the process is primarily driven by physisorption with possible contributions from chemisorption56,57. These trends are consistent with previous reports on metal adsorption by thermally treated agro-waste materials, where ΔH° values between 40 and 80 kJ/mol were linked to ion exchange and chemisorption mechanisms, while those less than 20 kJ/mol associated with physisorption mechanism66,67. Comparable findings have been documented for lead removal using biochar adsorbents, where ΔH° values between 20 and 80 kJ/mol were ascribed to coordination with surface functional groups68. In the case of the entropy change, positive ΔS values indicate an increase in randomness at the solid-liquid interface, meaning that Pb²⁺ ions become more disordered as they are adsorbed onto WBWA and VWBWA surfaces14,65. The higher ΔS value for VWBWA (13.44 J/mol·K) compared to WBWA (4.56 J/mol·K) suggests that the adsorption process for VWBWA leads to a more increase in randomness, possibly due to greater surface heterogeneity or a larger number of active sites. This aligns with results from biochar-based adsorbents as reported by Mahmood et al.69. where the uptake of Pb²⁺ was driven by increased randomness at the interface (positive ΔS°).
Desorption study
The desorption efficiency of various agents for recovering Pb(II) ions from raw wood bark waste adsorbent (WBWA) and valorized wood bark waste adsorbent (VWBWA) provides insight into both the strength of metal-adsorbent interactions and the reusability of the adsorbents as shown in Fig. 12. Acetic acid emerged as the most effective desorbing agent for both WBWA and VWBWA with desorption efficiencies of 76.3% and 85.37% respectively. Its ability to desorb Pb(II) efficiently is likely due to its mild acidity, which can protonate surface functional groups, weakening the interaction between Pb²⁺ and the adsorbent. The high performance, especially with VWBWA, suggests that acetic acid can disrupt metal-ligand coordination effectively without damaging the adsorbent. Hydrochloric acid also shows high desorption efficiency (WBWA: 72.2%, VWBWA: 75.6%). Its strong acidic nature enhances proton exchange, replacing Pb(II) ions bound to negatively charged sites but due to its aggressive acid conditions, which could alter or degrade surface functional groups over repeated cycles, acetic was selected over it. Water has the lowest desorption efficiencies (WBWA: 59.3%, VWBWA: 64.9%), as expected, due to the lack of strong competing ions or chelating ability. The results indicate that acetic acid is the most effective and possibly most sustainable desorbing agent and was therefore selected for susequent studies.
Percentage desorption of Pb²⁺ by WBWA and VWBWA at different desorbing agents.
Figure 13 illustrates the desorption efficiency of lead (Pb²⁺) using two types of adsorbents: untreated wood bark waste adsorbent (WBWA) and valorized wood bark waste adsorbent (VWBWA), across six desorption cycles with acetic acid as the desorbing agent. In the first cycle, WBWA shows a desorption efficiency of 74.2%, while VWBWA achieves 83.8%. This trend continues across all subsequent cycles, with both adsorbents showing a gradual decline in PD% over time. By the sixth cycle, WBWA drops to around 65.4%, whereas VWBWA maintains a relatively higher PD% of 70.3%. This decreasing trend is common in reuse studies and may be attributed to structural degradation of the adsorbent, pore blockage, or irreversible binding of metal ions to the adsorbent surface. It is evident from the chart that VWBWA consistently outperforms WBWA in terms of percentage desorption (PD%) throughout all six cycles. This suggests that the valorization process significantly enhances the material’s desorption capabilities, likely by improving surface area, porosity, or introducing additional functional groups that facilitate lead ion release. The desorption data supports the use of wood bark waste adsorbent as effective and reusable adsorbent for lead removal in aqueous environments.
Percentage desorption of Pb²⁺ by WBWA and VWBWA.
Proposed adsorption mechanism of pb²⁺ on calcined wood bark
The uptake of Pb²⁺ ions by calcined wood bark may proceeds through a combination of electrostatic attraction, surface complexation, ion exchange, and metal–oxygen coordination mechanisms based on the information obtained already from FT-IR, isotherms, kinetics and thermodynamics investigations. First, information from FT-IR evidence reveals the functional groups involvement as the spectra provided critical insights into those groups present on the wood bark surface before and after adsorption. The broad O–H stretching bands (3310–3550 cm⁻¹) initially seen in the raw sample significantly reduced on heat treatment, suggesting thermal decomposition or reduction of hydroxyl-bearing compounds (e.g., cellulose, hemicellulose, and lignin). However, after the sorption of Pb², the broadening and the reappearance of O–H bands (3450–3560 cm⁻¹) indicate hydrogen bonding and possible electrostatic interactions between negatively charged surface hydroxyl groups and the positively charged Pb²⁺. a similar situation was noted in the intensity of the C = O peak seen at 1745–1780 cm⁻¹ before adsorption which decrease after sorption confirming the involvement of carboxyl groups in metal binding, likely through complexation or coordination bonding. Furthermore, the appearance of new bands seen in the range of 500–700 cm⁻¹ region, which could be attributed to metal–oxygen (Pb–O) vibrations, further corroborate the formation of Pb²⁺–ligand complexes on the wood bark surface.
Information from adsorption isotherms shows that the Langmuir isotherm model gave a better fit than Freundlich, based on higher R² values, indicating that Pb²⁺ adsorption took place via a homogeneous surface via monolayer coverage. This indicates that interactions like surface complexation or ion exchange at defined active sites, rather than multilayer physical adsorption. Kinetic analysis revealed that the sorption by the biochar followed the pseudo-second-order model, inferring chemical sorption (chemisorption) adsorption mechanism which is in line with the formation of chemical bonds between Pb²⁺ ions and electron-donating surface groups, such as O–H, C = O, and Si–O as initially suggested from FT-IR analysis. The sorption of the raw biomass which fit with the pseudo-first-order model suggests that physical adsorption (e.g., van der Waals forces) plays significant role. Additionally, thermodynamic parameter of ΔH⁰ obtained between 15 and 30 kJ/mol confirm that the process is most likely governed by physisorption with some chemisorption contributions66,67. Figure 14 illustrates the schematic representation of the adsorption mechamism between Pb2+ and the surface of the wood bark waste.
Schematic representation of the adsorption mechanism.
Economic feasibility of the adsorbent
The use of calcined wood bark as an adsorbent showcase a cost-effective and sustainable solution for heavy metal elimination from wastewater. Wood bark is an abundant agro-forestry byproduct that is often discarded or underutilized as waste, making it readily available at little to no cost. In this study, the heat treatment process involves only thermal treatment without the need for hazardous or expensive chemical reagents, thereby minimizing environmental impact and the costs of production. On like conventional commercial adsorbents, like activated carbon, the preparation of calcined wood bark is less energy-intensive and simpler. In addition, the calcined wood bark demonstrated promising adsorption capacity that can be regenerated and reused, which further enhanced its economic viability for large-scale water treatment applications.
Because the preparation method is thermal-only (pyrolysis at 600 °C) with no chemical activation, the main costs are:
-
Raw material cost: $0.00 (because wood bark waste, it is considered a free agro-forestry byproduct in most cases).
-
Drying and Grinding Cost: Energy cost of drying and size reduction is estimated at ~$0.05/kg.
-
Pyrolysis Energy Cost: This is the major component which can be estimated using the following method.
Cost of pyrolyze 1 kg of dry wood bark can be estimated as follows:
-
Heating rate: up to 600 °C is assumed to energy of 1.5–3.0 MJ/kg and electricity price at $0.10 per kWh (standard industrial rate in Nigeria). If 1 MJ = 0.2778 kWh = 0.2778 kWh, and for 1.5–3.0 MJ, we have 0.4167 kWh to 0.8334 kWh.
-
Energy Cost (USD): Cost = Energy (kWh) × Electricity rate\text.
-
Cost = 0.4167 × 0.10 = $0.042 to 0.8334 × 0.10 = $0.083.
-
So pyrolysis energy cost ≈ $0.04–$0.08 per kg.
Table 5 presents the total estimated cost for producing 1 kg of the wood bark-based adsorbent which ranges from $0.16 to $0.28. The raw material cost is estimated at $0.00, as the wood bark waste is typically sourced at no charge from sawmills or wood-processing industries. Being industrial or an agricultural by-product, it is often disposed of as waste, making it an economically and environmentally sustainable choice for the production of adsorbent. The drying and grinding stage incurs a cost between $0.02 and $0.05 per kilogram which includes manual efforts for drying the material (which may be sun-dried or oven-dried), and grinding or milling to reduce the particle size. Pyrolysis, which involves thermally treating the biomass to convert it into a biochar adsorbent, is estimated to cost between $0.04 and $0.08 per kilogram, based on energy consumption alone. Labor and miscellaneous expenses, which include wages for personnel, water usage, safety gear, equipment maintenance, and other indirect costs such as packaging, range from $0.10 to $0.15 per kilogram. Overall, the cost of $0.16 to $0.28 per kilogram highlights the economic viability of the wood bark-based adsorbent, especially when compared to commercial activated carbon, which can cost between $1.00 and $3.00 per kilogram. The low production cost, combined with effective adsorption capabilities, positions this material as a promising, scalable, and cost-effective solution for wastewater treatment, particularly in resource-limited or developing regions13.
Conclusion
The adsorption of lead ions using raw and valorised wood bark waste adsorbents was investigated. The adsorption of Pb²⁺ onto wood bark waste adsorbents is highly pH-dependent, with maximum adsorption at pH 5. Valorized wood bark waste (VWBWA) demonstrates superior adsorption compared to non-valorized wood bark waste (WBWA), with higher capacity, faster kinetics, and improved equilibrium uptake. The adsorption mechanism follows both the Langmuir and Freundlich models, indicating a multiple mechanisms, making the process effective across different conditions. Its structural modifications, as confirmed by SEM analysis, reveal that calcination increases porosity, and surface area, significantly enhancing Pb²⁺ adsorption. FT-IR analysis shows that valorization improves oxygen containing functional groups which enhances the surface characteristics and improves the active sites for Pb²⁺ binding. In conclusion, WBWA is a highly efficient, thermally stable, and environmentally friendly adsorbent for Pb²⁺ removal, making it a promising material for industrial wastewater treatment.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Ghosh, S., Mohanty, S., Akcil, A., Sukla, L. B. & Das, A. P. A greener approach for resource recycling: manganese bioleaching. Chemosphere 154, 628–639. https://doi.org/10.1016/J.CHEMOSPHERE.2016.04.028 (2016).
Kshyanaprava, R. & Alok, P. D. Lead pollution: impact on environment and human health and approach for a sustainable solution. Environ. Chem. Ecotoxicol. 1, 79–85. https://doi.org/10.1016/j.enceco.2023.02.001 (2023).
Ghosh, S., Gandhi, M., Van-Hullebusch, E. D. & Das, A. P. Proteomic insights into Lysinibacillus sp.-mediated biosolubilization of manganese Environ. Sci. Pollut. Res. 28, 40249–40263 (2021).
Ofudje, E. A. et al. Mechanism of Cu2+ and reactive yellow 145 dye adsorption onto eggshell waste as low-cost adsorbent. Chem. Ecol. 37, 268–289 (2021).
Tripathy, B., Dash, A. & Das, A. P. Detection of environmental microfiber pollutants through vibrational spectroscopic techniques: recent advances of environmental monitoring and future prospects. Crit. Rev. Anal. Chem. 1, 1–11 (2022).
Mishra, S., Dash, D., Al-Tawaha, A. R. M. S. & Das, A. P. A review on heavy metal ion adsorption on synthetic microfiber surface in aquatic environments. Appl. Biochem. Biotechnol. 194, 4639–4654. https://doi.org/10.1007/S12010-022-04029-W/METRICS (2022).
Chowdhury, I. R., Chowdhury, S., Mazumder, M. A. J. & AlAhmed. A. Removal of lead ions (Pb2+) from water and wastewater: A review on the low-cost adsorbents. Appl. Water Sci. 12, 185. https://doi.org/10.1007/s13201-022-01703-6 (2022).
Dey, S., Tripathy, B., Kumar, M. S. & Das, A. P. Ecotoxicological consequences of manganese mining pollutants and their biological remediation. Environ. Chem. Ecotoxicol. https://doi.org/10.1016/J.ENCECO.2023.01.001 (2023).
Wani, A. L., Ara, A. & Usmani, J. A. Lead toxicity: A review. Interdiscip Toxicol. 8 (2), 55–64 (2015).
Charkiewicz, A. E. & Backstrand, J. R. Lead toxicity and pollution in Poland. Int. J. Environ. Res. Public. Health. 17 (12), 4385 (2020).
Neskoromnaya, E. A. et al. Adsorption of lead ions (Pb2+) from wastewater using effective nanocomposite go/cmc/fenps: kinetic, isotherm, and desorption studies. Colloids Surf., A. 655, 130224. https://doi.org/10.1016/j.colsurfa.2022.130224 (2022).
Crini, G. & Lichtfouse, E. Advantages and disadvantages of techniques used for wastewater treatment. Environ. Chem. Lett. 17, 145–155 (2019).
Ofudje, E. A., Al-Ahmary, K. M. & Alzahrani, E. A. Sugarcane Peel Ash as a sorbent for methylene blue. BioResources 19 (4), 9191–9219. https://doi.org/10.15376/biores.19.4.9191-9219 (2024).
Ogundiran, A. A., Ololade, O. B., Ogundiran, O. O. & Ofudje, E. A. Utilization of unmodified and acid modified snail shells as low-cost adsorbents for the removal of congo red. MUJAST 2 (1), 27–38 (2022).
Akkari, I., Kaci, M. M. & Pazos, M. Revolutionizing waste: Harnessing agro-food hydrochar for potent adsorption of organic and inorganic contaminants in water. Environ. Monit. Assess. 196, 1035. https://doi.org/10.1007/s10661-024-13171-3 (2024).
Kaci, M. M. et al. Recent trends in remediating basic red 46 dye as a persistent pollutant from water bodies using promising adsorbents: a review. Clean. Technol. Environ. Policy. 27, 773–788. https://doi.org/10.1007/s10098-024-03026-3 (2025).
Budsaereechai, S., Kamwialisak, K. & Ngernyen, Y. Adsorption of lead, cadmium and copper on natural and acid activated bentonite clay. KKU Res J. 17 (5), 800–810 (2012).
Astuti, W., Chafdz, A., Al-Fatesh, A. S. & Fakeeha, A. H. Removal of lead (Pb(II)) and zinc (Zn(II)) from aqueous solution using coal Fy Ash (CFA) as a dual-sites adsorbent. Chin. J. Chem. Eng. 34, 289–298 (2020).
Lubbad, S. H. & Al-Batta, S. N. Ultrafast remediation of lead-contaminated water applying sphagnum peat moss by dispersive solidphase extraction. Int. J. Environ. Stud. 77, 382–397 (2020).
Aigbe, R. & Kavaz, D. Unravel the potential of zinc oxide nanoparticle-carbonized sawdust matrix for removal of lead(II) ions from aqueous solution. Chin. J. Chem. Eng. 29, 92–102 (2021).
Cao, Y. et al. Carbonization and ball milling on the enhancement of Pb(II) adsorption by wheat straw: competitive effects of ion exchange and precipitation. Bioresour Technol. 273, 70–76. https://doi.org/10.1016/j.biortech.2018.10.065 (2019).
Cholico-Gonzalez, D., Ortiz Lara, N., Fernandez Macedo, A. M. & Salas, J. C. Adsorption behavior of Pb(II), Cd(II), and Zn(II) onto Agave bagasse, characterization, and mechanism. ACS Omega. 5, 3302–3314. https://doi.org/10.1021/acsomega.9b03385 (2020).
Zhang, J. et al. Simultaneous removal of humic acid/fulvic acid and lead from landfill leachate using magnetic graphene oxide. Appl. Surf. Sci. 370, 335–350 (2016).
Chen, J., Wang, Y., Lang, X., Ren, X. & Fan, S. Evaluation of agricultural residues pyrolysis under non-isothermal conditions: thermal behaviors, kinetics, and thermodynamics. Bioresour. Technol. 241, 340–348. https://doi.org/10.1016/j.biortech.2017.05.0 (2017).
El-Sayed, S. A., Khass, T. M. & Mostafa, M. E. Thermal degradation behaviour and chemical kinetic characteristics of biomass pyrolysis using TG/DTG/DTA techniques. Biomass Convers. Bioref. 14, 17779–17803. https://doi.org/10.1007/s13399-023-03926-2 (2024).
El-Rayyes, A. et al. Sawmill residue valorization as adsorbent for Cd 2+ from aqueous solution. BioResources 20 (3), 7048–7074 (2025).
Ighalo, J. O. & Adeniyi, A. G. A mini-review of the morphological properties of biosorbents derived from plant leaves. SN Appl. Sci. 2, 509. https://doi.org/10.1007/s42452-020-2335-x (2020).
Kang, S. et al. Valorization of biomass hydrolysis waste: Activated carbon from humins as exceptional sorbent for wastewater treatment. Sustainability 10 (6), 1795. https://doi.org/10.3390/su10061795 (2018).
Timar, M. C., Varodi, A., Hacibektasoglu, M. & Campean, M. Color and FTIR analysis of chemical changes in Beech wood (Fagus sylvatica L.) after light streaming and heat treatment in two different environments. BioRes 11 (4), 8325–8343 (2016).
Will, W. FT-IR Spectroscopy to Accurately Identify Native Commercial Wood Species. https://www.spectroscopyonline.com/view/ft-ir-spectroscopy-to-accurately-identify-native-commercial-wood-species?utm_source=chatgpt.com (2024).
El Hajam, M. et al. Pb2+ ions adsorption onto raw and chemically activated Dibetou sawdust: application of experimental designs. J. King Saud Univ. Sci. https://doi.org/10.1016/j.jksus.2020.02.027 (2020).
Mabaso, T., Lo, S. L. & Chiueh, P. T. Effect of pyrolytic temperature on the adsorption of Pb(II) from synthetic wastewater onto bamboo chopstick biochar: A conventional vs. microwave-assisted pyrolysis approach. Sustain. Environ. Res. 34, 31. https://doi.org/10.1186/s42834-024-00238-6 (2024).
Rahimi, V. et al. Development and characterization of biomass-derived carbons for the removal of Cu2+ and Pb2+ from aqueous solutions. C2025 11 (1), 2. https://doi.org/10.3390/c11010002 (2024).
Roy, D., Roy, B. & Manna, A. K. Pyrolyzed mesoporous activated carbon preparation from natural rubber common effluent biosludge: characterization, isotherms, kinetics, thermodynamics, and ANN modeling during phenol adsorption. Groundw. Sustain. Dev. 23, 101020. https://doi.org/10.1016/j.gsd.2023.101020 (2023).
Tang, K. H. D. Valorization of organic waste as biosorbents for wastewater treatment. Water Emerg. Contam. Nanoplastics. 3, 25. https://doi.org/10.20517/wecn.2024.53 (2024).
Sen, T. K. Agricultural solid wastes based adsorbent materials in the remediation of heavy metal ions from water and wastewater by adsorption: A review. Molecules 28, 5575. https://doi.org/10.3390/molecules28145575 (2023).
Meez, E., Rahdar, A. & Kyzas, G. Z. Sawdust for the removal of heavy metals from water: A review. Molecules 26, 4318. https://doi.org/10.3390/molecules26144318 (2021).
Natrayan, L. et al. Development and characterization of carbon-based adsorbents derived from agricultural wastes and their effectiveness in adsorption of heavy metals in waste water. Bioinorg. Chem. Appl. 23, 1659855. https://doi.org/10.1155/2022/1659855 (2022).
Chen, L. et al. Predicting Cd(II) adsorption capacity of Biochar materials using typical machine learning models for effective remediation of aquatic environments. Sci. Total Environ. 944, 173955 (2024).
Singh, B., Walia, B. S. & Arora, R. Eucalyptus wood charcoal as biosorbent for removal of lead(II) ions from aqueous solution. Int. J. Res. Anal. Reviews. 5 (4), 993–998 (2018).
Raji, Z., Karim, A., Karam, A. & Khalloufi, S. Adsorption of heavy metals: mechanisms, kinetics, and applications of various adsorbents in wastewater remediation—A review. Waste 1, 775–805. https://doi.org/10.3390/waste1030046 (2023).
Ezeonuegbu, B. A. et al. Agricultural waste of sugarcane Bagasse as efficient adsorbent for lead and nickel removal from untreated wastewater: biosorption, equilibrium isotherms, kinetics and desorption studies. Biotechnol. Rep., 30, e00614. https://doi.org/10.1016/j.btre.2021.e00614
Ngueagni, P. T. et al. Cellulose-based adsorbent of animal waste for the adsorption of lead and phenol. BioResources 20 (2), 3923–3952 (2025).
Elkhaleefa, A. et al. Evaluation of the adsorption efficiency on the removal of Lead(II) ions from aqueous solutions using Azadirachta indica leaves as an adsorbent. Processes 9 (3), 559. https://doi.org/10.3390/pr9030559 (2021).
Bakatula, E. N., Richard, D., Neculita, C. M. & Zagury, G. J. Determination of point of zero charge of natural organic materials. Environ. Sci. Pollut. Res. 25 (8), 7823–7833 (2018).
Kaushal, A. & Singh, S. K. Adsorption phenomenon and its application in removal of lead from waste water: a review. Int. J. Hydrol. 1 (2), 38–47. https://doi.org/10.15406/ijh.2017.01.00008 (2017).
Mamatha, M., Aravinda, H. B., Manjappa, S. & Puttaiah, E. T. Kinetics and mechanism for adsorption of lead in aqueous and industrial effluent from Pongamia pinnata tree bark. J. Environ. Sci. Toxicol. Food Technol. 2 (3), 1–9 (2012).
Blessing, A. E. et al. Agricultural waste of sugarcane bagasse as efficient adsorbent for lead and nickel removal from untreated wastewater: Biosorption, equilibrium isotherms, kinetics and desorption studies. Biotechnol. Rep. 30, e00614 (2021).
Shagun, K., Piyush, S. & Pandey, O. P. Green sorbents from agricultural wastes: A review of sustainable adsorption materials. Appl. Sur Sci. Adv. 19, 100562. https://doi.org/10.1016/j.apsadv.2023.100562 (2024).
Adeogun, A. I., Ofudje, E. A., Idowu, M. A. & Ahmed, S. A. Biosorption of Cr(VI) ion from aqueous solution by maize husk: isothermal, kinetic and thermodynamic study. J. Chem. Soc. Pak. 34, 1388–1396 (2012).
Haider, B. et al. Adsorptive removal of lead from wastewater using Pressmud with evaluation of kinetics and adsorption isotherms. Sci. Rep. 15, 22823. https://doi.org/10.1038/s41598-025-05169-9 (2025).
Yang, W. et al. Removal of Pb(II) from aqueous solution and adsorption kinetics of corn stalk Biochar. Separations 10, 438. https://doi.org/10.3390/separations10080438 (2023).
Hubbe, M. A., Azizian, S. & Douven, S. Implications of apparent pseudo-second-order adsorption kinetics onto cellulosic materials: A review. BioRes 14 (3), 7582–7626 (2019).
Duran, C., Ozdes, D., Gundogdu, A. & Senturk, H. B. Kinetics and isotherm analysis of basic dyes adsorption onto almond shell (Prunus dulcis) as a low cost adsorbent. J. Chem. Eng. Data. 56 (5), 2136–2147. https://doi.org/10.1021/je101204j (2011).
Díaz, E., García, L. & Ordóñez, S. Adsorption of stable and labile emerging pollutants on activated carbon: degradation and mass transfer kinetic study. Appl. Water Sci. 14, 79. https://doi.org/10.1007/s13201-023-02087-x (2024).
Sahmoune, M. N., Moussa, A. & Mohamed, T. Understanding the rate-limiting step adsorption kinetics onto biomaterials for mechanism adsorption control. Prog. React. Kinet. Mech. 49, 1–26. https://doi.org/10.1177/14686783241226 (2024).
Ademoyegun, A. J., Babarinde, N. A. A. & Ofudje, E. A. Sorption of Pb(II), Cd(II), and Zn(II) ions from aqueous solution using Thaumatococcus Danielli leaves: kinetic, isotherm, and thermodynamic studies. Desalin. Water Treat. 273, 162–171. https://doi.org/10.5004/dwt.2022.28877 (2022).
Ibrahim, M. N. M. et al. A novel agricultural waste adsorbent for the removal of lead (II) ions from aqueous solutions. J. Hazard. Mat. 182, 377–385. https://doi.org/10.1016/j.jhazmat.2010.06.044 (2010).
Li, C., Wang, H. & Zhang, Y. Pb(II) removal from aqueous solution by cold KOH activated Biochar of Camphor leaves: isotherms, kinetics and thermodynamics. Desalination Water Treat. 161, 327–336. https://doi.org/10.5004/dwt.2019.24312 (2019).
Ghahremani, A., Manteghian, M. & Kazemzadeh, H. Removing lead from aqueous solution by activated carbon nanoparticle impregnated on lightweight expanded clay aggregate. J. Environ. Chem. Eng. 9, 104478 (2021).
Wang, Z., Xu, J., Yellezuome, D. & Liu, R. Effects of cotton straw-derived Biochar under different pyrolysis conditions on Pb (II) adsorption properties in aqueous solutions. J. Anal. Appl. Pyrol. 157, 105214 (2021).
Tan, Y. et al. Novel Zn-Fe engineered Kiwi branch Biochar for the removal of Pb(II) from aqueous solution. J. Hazard. Mater. 424, 127349 (2022).
Lian, W. et al. Utilization of Biochar produced from invasive plant species to efficiently adsorb cd (II) and Pb (II). Bioresour Technol. 317, 124011 (2020).
Mohammed, A. H., Shartooh, S. M. & Trigui, M. Biosorption and isotherm modeling of heavy metals using Phragmites australis. Sustainability 17 (12), 5366. https://doi.org/10.3390/su17125366 (2025).
El-Rayyes, A. et al. Hot water-treated cow waste use as an efficient adsorbent for Cresol red dye and chromium VI removal from aqueous solutions. BioResources 20 (2), 3252–3285 (2025).
Li, N. et al. Magnetic graphene oxide functionalized by Poly dimethyl Diallyl ammonium chloride for rfficient removal of Cr(VI). J. Taiwan. Inst. Chem. Eng. 91, 499–506 (2018).
Naat, J. N. et al. Histamine adsorption from aqueous solution using silica-based SiO2@BSA adsorbent from natural sand: adsorption parameters, kinetic models, isotherms, and thermodynamics. Iran J. Chem Chem Eng. 43 (2), 635–651 (2024).
Sahmoune, M. N. Evaluation of thermodynamic parameters for adsorption of heavy metals by green adsorbents. Environ. Chem. Lett. 17, 697–704. https://doi.org/10.1007/s10311-018-00819-z (2019).
Mahmood, U., Abid, S., Qadir, B., Nazir, A. & Hussain, T. Thermodynamic and kinetic study of adsorptive removal of lead by the nanocomposite loaded nanofibers. Turk. J. Chem. 46 (2), 342–355. https://doi.org/10.3906/kim-2107-67 (2021).
Acknowledgements
The authors express their gratitude to Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2025R581), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Funding
This work was funded by Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2025R581), Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Salah Ud Din carried out the statistical analysis, and characterizations, Arogundade Ibrahim conceived the work, and performed the experiments, Khairia Mohammed Al-Ahmary performed statistical analysis and read the first draft, Saedah R. Al-Mhyawi performed experiments, and performed data analysis, Hamad AlMohamadi contributed resources and performed statistical analysis, Nuha Y. Elamin and Ibtehaj F. Alshdoukhi contributed resources, performed data analysis and contributed in characterization, Jawaher Saud Alrashood provided financial assistance and contributed resources.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Din, S.U., Arogundade, I., Al-Ahmary, K.M. et al. Utilization of valorised wood bark waste as an adsorbent for the uptake of Pb(II) ions. Sci Rep 15, 31099 (2025). https://doi.org/10.1038/s41598-025-17021-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-17021-1