Abstract
To meet the growing need for sustainable energy solutions, improving the efficiency of thermal energy storage (TES) systems is important. In this study, the melting behavior of RT42 paraffin wax inside a hexagonal multi-cell was analyzed using numerical simulation based on the enthalpy-porosity method with ANSYS/FLUENT 16 software. The study focused on evaluating the effect of changing the thickness of the central air layer on the melting efficiency of the phase change material (PCM). Four cases were tested: one without an air layer and three others with thicknesses of 2 mm, 4 mm, and 6 mm. The results showed a clear quantitative relationship between the thickness of the air layer and the increase in melting time. The total time increased from 660 min (without air) to 780 min with a 2 mm thick air layer, an increase of 18%. The time reached 900 min at a thickness of 4 mm (37%) and 960 min at 6 mm (50%). These results show how important internal air layers are in reducing heat transfer efficiency, and how important it is to take them into account when designing phase change material-based thermal energy storage systems to get better performance and sustainability.
Similar content being viewed by others
Introduction
Energy is a key foundation for economic growth, technological advancement, and, ultimately, the general welfare of present-day society. It has played and continues to play a significant and transformative role in almost all infrastructure, industrial processes, and daily functions. However, some sources of energy are recognized to have severe environmental consequences, while others are limited resources that will eventually give out1,2. Recent years have seen a broad interest in phase change materials (PCMs) in passive thermal management due to their high thermal inertia. PCMs offer several advantages, including excellent temperature uniformity, low operating costs, high reliability, minimal aging effects, and widespread availability. They function by storing thermal energy in the form of latent heat, which can be released in response to temperature fluctuations. This characteristic makes PCMs a vital link between energy demand and supply over extended periods3,4,5. As a result, PCMs are employed in numerous applications, both domestically and industrially, such as in battery thermal management, energy storage systems, textile manufacturing, solar energy applications, heat exchangers, electronic cooling, air conditioning and refrigeration, building materials, and aerospace technologies. Recently, PCMs have also shown promise in biomedical applications6,7,8,9. In this context, Babaharra et al.10 investigated the integration of PCMs into hollow bricks for building applications under different climate conditions. Using the enthalpy-porosity method, they demonstrated that embedding PCMs within construction materials can significantly regulate internal temperatures and enhance thermal stability across variable climates.
The effect of phase change materials’ (PCMs) thermophysical properties on the efficiency of thermal energy storage systems has been a keen interest in various investigations. Tao and Carey11 experimentally investigated a tube-and-shell latent heat storage system to examine the effects of PCM properties on thermal storage performance. The study involved the flow of a heat transfer fluid in a cylindrical pipe, with an accent on the necessity of choosing phase change materials with specific characteristics to optimise storage efficiency. The study reached the conclusion that PCMs with high specific heat, melting enthalpy, density, as well as high thermal conductivity and a low melting temperature, significantly improve the velocity and efficiency of thermal storage systems.
Hasan et al.12 considered the contribution of phase change materials (PCMs) on thermal performance and indoor comfort levels. In the study, a 44 °C melting point phase change material was applied as an insulating material on the exterior walls constructed from 12 cm thick bricks, together with 1 cm thick adhesive mortar. The incorporation of a 10 mm thickness of phase change material (PCM) resulted in a significant decrease in indoor temperature and cooling load, with a 20.9% reduction in space cooling energy consumption. This finding indicates the potential for energy efficiency of PCMs when used in building applications by minimising the electrical consumption associated with cooling activities. A recent comprehensive review by Bouhezza et al.13 outlined a wide range of performance enhancement techniques for PCM applications, including the use of extended surfaces, nano-enhancements, porous matrix integration, and active/passive hybrid systems. Their findings reinforce the growing emphasis on geometry, material configuration, and internal thermal interface optimization as key drivers for thermal performance improvement. Ebrahimi et al.14 investigated the application of heat pipes in latent heat storage systems for their improvement. The research explored the use of U-shaped tubes to increase heat transfer efficiency, focusing on the belongings of impacts numbers of tubes and the orientations of the HTF plates. The researchers modelled at least thirteen different models with an enthalpy-porosity technique to examine the dynamics of phase change. The findings indicated that heat pipe usage surrounding the storage system would decrease melting time by as much as 91%. It was also found that raising the HTF plate angle would enhance the melting process, indicating that the system’s design plays a crucial role in optimising thermal efficiency. Jasim et al.15 Finding the most effective rod material copper, iron, aluminum, or silver heat transfer to phase-change materials inside a rectangular cell is the goal of this computer study. The study uses the ANSYS/FLUENT 16 software and the enthalpy-porosity model to investigate how various fin materials affect the dynamics of heat transmission in paraffin wax-based phase-change materials (RT58). The results show that the rod material affects heat transfer efficiency, with aluminum fins lowering the process of phase change by 6% and iron rods by 11% when compared to copper and silver fins. With potential uses in enhancing thermal management for contemporary electronic devices, these results emphasize the significance of selecting the right materials to improve heat transmission in systems of phase change. The work performed by Hammoodi et al.16,17 used CFD simulations to analyse the effects of air layers on melting processes for a phase change material (PCM) in spherical and semi-cylindrical containers. The authors aimed to examine the impacts of air voids on the thermal performance and phase change behavior of latent heat storage systems. The study shows that air layers improve thermal insulation and thus delay melting, with the delay time more prominent at lower density values. Cooling was beyond the measured reference due to the interference of air gaps within the heat transfer mechanism from the phase change material to the surrounding environment, thereby creating cooling spatial differences. The study showed how geometric configurations influence the air layers during the melting process. Spherical vessels exhibited a continuous thermal resistance effect due to the air layer, while semi-cylindrical vessels displayed isolated thermal resistance regions, resulting in prolonged melting time. The findings indicate that designs of PCM-based thermal storage systems ought to incorporate an air layer to enhance heat transmission. Hammoodi et al.17 extended this investigation by numerically analyzing the effect of circular air layers (1 mm and 2 mm) surrounding RT35 paraffin wax in cylindrical enclosures using the enthalpy-porosity method in ANSYS/FLUENT. Their results demonstrated a significant increase in melting time by 125% and 225%, respectively due to the suppression of natural convection and the insulating nature of the air layer.
Abbas et al.18 conducted a numerical study on the effect of inserting an air layer around RT42 paraffin wax in spherical enclosures. Using the enthalpy-porosity method with ANSYS/FLUENT, their results showed that increasing the air gap thickness significantly delayed the melting process with a reported rise in melting time up to 125% for a 5 cm layer. This highlights the strong insulating effect introduced by air layers in PCM-based thermal storage systems.
In their study, Kadhim et al.19 look into how to improve the charging capacity of RT42 paraffin wax in a square enclosure (50 × 50 mm) that has copper fins of different lengths and orientations. Each cell utilized four fins, each with a uniform thickness of 1 mm. The fin lengths of each cell were 10, 20, and 30 mm, which accounted for the discrepancy. A heat flux-maintained steadiness was maintained on the wall containing fins, while other walls received insulation treatment. Among the tested cells, the longest 30 mm fins achieved the highest charging rate because they yielded a performance improvement of 55.56% compared to unfurnished cells. Placing the fins inside one of the insulated walls caused a reduction in charging speed since the fins should be positioned directly toward the thermal charging source.
Recent research highlights the growing interest in nano-enhanced phase change materials (NEPCMs) as a strategy for improving thermal energy storage and management. Gür et al. (2024)20 designed a solar-assisted radiant heating system incorporating B4C-based NEPCM for nearly zero-energy buildings. The results showed improved thermal regulation with minimal energy input during non-solar periods. Numerical analysis confirmed a temperature gain of 2.82 K compared to non-PCM systems, and a slight advantage of 0.62 K for NEPCM over conventional PCM. Öztop et al. (2024)21 extended this work by developing B4C-enhanced RT35HC nanocomposite PCMs. With increasing B4C content up to 2 wt%, thermal conductivity improved by 67.5% in solid and 15.3% in liquid states. Cp also increased, while latent heat slightly declined. The nanocomposite maintained stability over repeated use, indicating potential for long-term thermal storage applications. In another study by Kurt et al. (2024)22, a ternary fatty acid blend (LA–SA–UA) was implemented in a prototype data center. The PCM blend demonstrated promising thermal properties with conductivities reaching 0.245 W/m·K and high Cp values. Timed trials at various airflow speeds confirmed the PCM’s role in extending operational thermal buffering during peak loads. Finally, Gurgenc et al. (2024)23 analyzed how positioning a semi-circular adiabatic partition affects the melting and thermal performance of B4C/RT44HC nanocomposite PCMs. A thermal conductivity increase of 69.65% and energy storage enhancement of 19.68% were recorded, confirming the effectiveness of nanoparticle loading and spatial configuration.
This research makes an original contribution by analyzing the melting behavior of RT42 paraffin wax inside a hexagonal multicell container. Air layers of different thicknesses (2 mm, 4 mm, and 6 mm) were included in the center of the cell, in addition to a reference case without any air layer. Unlike previous studies that focused on spherical or cylindrical containers, this study is distinguished by the use of a more complex geometric shape that reflects the real-world challenges in the design of compact thermal storage systems. The originality of this work lies in evaluating the effect of internal air layers within this hexagonal configuration on phase change dynamics, natural convection behavior, and the total time required to complete melting. The study also included detailed quantitative comparisons to identify thermal delay mechanisms and highlighted the critical role of geometric constraints that have not been sufficiently addressed in previous literature.
Numerical procedure
Mathematical models
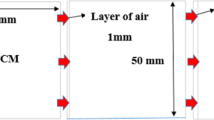
The multi-hexagonal cell analysed in this study has an outer-side length of 70 mm and an inner-side length of 20 mm. In order to analyse the effect of the air layer on melting, different air layer thicknesses of 2 mm, 4 mm, and 6 mm were considered. The introduction of an air layer prevents some heat transfer and produces additional thermal resistance that eventually changes the heat transfer and phase change behaviours. By considering these variations, it is expected to acquire complete knowledge about the influence of the presence/absence and thickness of an air layer on the melting rate, thermal distribution, and efficiency of the overall phase change process. Figure 1 shows a schematic representation of the hexagonal cell and the configuration of the air layer around it.
Configuration of physical model.
Governing equations
Simulating a hexagonal cell geometry using numerical methods allows for accurate predictions on melting in two dimensions. The modelling is two-dimensional, describing the laminar, unstable, and incompressible nature of the study. In so doing, only the most essential details consequential to this system are captured. The very nature of the system under study necessitates numerical analysis for effective and accurate solutions as opposed to complex three-dimensional simulations. The article also deals comprehensively with phase transition characteristics. In the numerical simulation of phase transformation, enthalpy-porosity, originally formulated in24,25, was applied. The method assumes that the liquid and solid phases of CPMS (phase change material) gain thermal equilibrium at their interface, and with such a fundamental assumption, it further simplifies the simulation characteristics in executing accurate phase transition simulations. It is believed that the system was well insulated, which led to ignoring the effects of viscous dissipation and thermal energy lost to the environment26,27. The assumptions also led to conservation equations for continuity, momentum, and energy28,29. During melting, it monitors temperature variation, thus accurately recording thermal response and latent heat effects of phase change processes in PCM by the enthalpy method. So, it can accurately simulate the thermal response and energy distribution of the system. Melting process modelling presents major difficulties due to the nonlinear, transient, and ever-changing solid-liquid interface. The careful control of various numerical constraints, which complicate the problem, is necessary for stable and accurate simulations. Such complex processes, in general, result from the motion of phase boundaries, rate of melting, and associated heat transfer. Complex numerical approaches are required to achieve satisfactory outcomes. PCMs are described by a fundamental set of differential equations that maintain the constant values of mass, momentum, and energy as a result of their capacity to undergo phase transitions in response to temperature. The thermal performance for a system is primarily estimated using Eqs. (1), (2), and (3), which illustrate the heat transfer and fluid flow within the system. The solutions to the governance equations enable experts to evaluate the distribution of temperatures, the transformation of phases, and the movement of heat through PCM storage systems to optimise their functionality. The system’s physical and thermal behaviour is governed by the following equation28:
The addition of the damping term (\(\:\overrightarrow{\text{S}})\) in the momentum equation is attributed to the influence of the phase transition event. This phrase aligns with the description of the damping term in the law of Darcy30, :
It has been found from the literature that the fixed Am for the mushy region is 105, as given in ref31,32,33. An energy scheme includes a source term to resolve latent heat impact and phase processes. The fluid fraction of the phase change material (PCM), represented as λ, is shown in34.
Where,
The meaning of the latent heat ability vary between zero (for a solid) and one (for a liquid) and the liquid fraction (β) can be written as:
The source term \(\:{\text{S}}_{\text{L}}\) in the energy equation is derived as follows:
Specified boundary conditions
The hexagonal cell analysed has an effect of the heat source from all sides on the phase change materials, and the temperature is (77 °C). Through the wall, Paraffin wax (RT42) is employed as a phase change material, and the thermal properties of paraffin are presented in Table 1.
Assumptions
In preparing the mathematical model to simulate the melting process inside the hexagonal cell, a set of assumptions were adopted to ensure physical realism and numerical efficiency. The flow was considered to be internally laminar, incompressible, and temporally unstable. The Boussinesq approximation was used to represent the buoyancy effects resulting from temperature differences within the PCM region. Viscosity dissipation effects were neglected due to their insignificance in normal low-speed loading conditions. It was assumed that phase change occurs without any volumetric expansion or contraction, i.e., using the constant volume assumption. The thermal boundaries of the system were also considered to be completely isolated, so that no heat exchange with the surrounding environment occurs. The thermal properties of the PCM were assumed to be constant in both the solid and liquid phases, while the air layer was considered to be a stationary and isolated region, with its resistance and thermal thickness neglected for simplicity Fig 2. All these assumptions contribute to simplifying the numerical model while preserving the essence of the physical phenomena of phase change.
Configuration of the mesh model.
Grid independence and code validation
A wide range of mesh independence studies was conducted to study the effect of mesh density on phase change behaviour in a particular geometric configuration. This step is necessary in the field of computational fluid dynamics (CFD) research to prevent the numerical outcomes from being affected by a modification of the mesh resolution. This analysis is intended to keep an accurate track of the progression of the phase change process. Four different mesh densities were chosen, representing groups with 32,456, 34,567, 36,789, and 38,790 (see Table 2) elements to cover a wide range of computational cases. An extensive assessment of the whole mesh ensemble was conducted, showing that the phase change progression was consistent across all element numbers. The uniformity observed implies that the numerical results were independent of mesh density, meaning refinement would not alter the results much. For obtaining a balance between computational efficiency and accuracy, the minimum element count of 32,456 was used for further investigations. Figure 3 illustrates the decision to lower the computational cost while maintaining the reliability of the results.
Grid independency without fins.
The numerical simulation framework was verified and validated by a thorough validation method to guarantee the correctness and reliability of the numerical solution. This approach was supported by the addition of new functions into the simulation algorithm to enhance its ability to simulate the liquid fraction and properly model the geometric quantity of the phase transition process. The modifications were done to be similar to the computational structure of a fundamental research by Begum et al.36, which gives a detailed analysis of the effect on heat transfer and fluid flow behaviour in a multi-hexagonal cell phase change of the material phase transition (PCM). The adequacy of the augmented simulation SMPS framework was established on the grounds of a detailed comparison study. The numerical outcomes acquired from the enha nced model were meticulously compared with the experimental results given by Begum, L. This comparison asserted the credibility of the enhanced numerical method by finding an excellent convergence of the two data sets. Small variations in the computational approach and the configuration of the model produced few differences, thus validating that the generated simulation framework is correct and robust. As demonstrated by the obtained results, the effectiveness of the proposed model in replicating the key aspects of phase transition dynamics is proven. The results in the outcomes of this validation procedure indicate that on hexagonal geometries, the enhanced simulation framework can genuinely predict the thermal behaviour and phase transition characteristics of phase change materials. This verification is needed for establishing that the numerical method is a reliable tool for additional studies on heat-transport phenomena. Figure 4 presents the validation results as well as near agreement between the reference study and computational results. We find that the deviation between the two studies is 9%.
Distinction of the melting fraction versus operating time for this study against the research36.
Results and discussion
This research investigated four different cases of a multi-hexagonal cell incorporating phase change material (PCM) to analyse the influence of an air layer on the melting process. Case one serves as a comparative foundation by analysing the cell without the presence of an air layer. Subsequently, cases two, three, and four examine the effects of air layer thicknesses of 2 mm, 4 mm, and 6 mm, respectively, on heat transfer processes. This study examines the impact of air layer thickness on the rates of phase change and dissolution, thereby influencing melting efficiency and duration. Comparative analysis of these cases provides insights into the influence of air layers on heat transport dynamics and phase transition behaviours in multi-hexagonal cells containing phase change materials.
Case one (non-air layer)
In the absence of any air gap, the cell is under test. It is clear from Fig. 5 that the melting process begins with the conduction load, which primarily governs the area close to the wall. First phase is governed by heat transfer conduction, which causes melting on the phase-change material at the wall, and moving away from the wall, melting becomes increasingly governed by natural convection. Given that heat transfer depends on natural convection, it is evident that the melting process decelerates as the distance from the wall rises. Heat transmission to the phase-change materials at higher distances from the wall is slower, principally affected by natural convection rather than the more effective conduction. This process is clearly illustrated in Fig. 6, where the heat transfer dynamics are demonstrated to decelerate as the distance from the wall increases. Additional evidence is depicted in Fig. 7, which illustrates the progression of the melting front, originating near the wall and proceeding toward the phase-change materials. The shift from conduction-dominated heat transmission adjacent to the wall to convection-dominated heat transfer at a distance is a pivotal element of the melting process, underscoring the intricate interaction among various heat transfer processes.
Predicted evolution of the melting process without layer of air.
Temperature distributions with without layer of air.
Velocity distributions without a layer of air.
Case two (2 mm air layer)
A 2 mm layer of air is present during the investigation of the cell. Figure 8 illustrates that the air layer substantially impacts the conduction load, subsequently affecting the melting process. Heat is first transferred from the wall to the air layer, subsequently continuing to the phase-change materials along the layer. The melting process at this stage is predominantly governed by natural convection, observable as the phase-change material migrates away from the wall. The melting process decelerates with increasing distance from the wall, attributed to the reduced effectiveness of heat transfer via natural convection relative to conduction. Figure 9 illustrates the process of heat transfer to the phase-change materials, commencing with conduction from the wall, progressing through the air layer, and subsequently entering the phase-change materials. As the process advances, the impact of natural convection increasingly prevails, resulting in a reduced rate of heat transfer with increasing distance from the wall. Figure 10 illustrates the progression of the melting front, originating near the wall within the air layer and progressively advancing into the phase-change materials. This progression illustrates the influence of conduction and convection on the melting process, indicating that the melting rate decreases with increasing distance from the wall, while the dependence on natural convection intensifies.
Predicted evolution of the melting process with layer of air (2 mm).
Temperature distributions with layer of air (2 mm).
Velocity distributions with a layer of air (2 mm).
Case three (4 mm air layer)
During the investigation of the cell, a 4-mm layer of air existed above it. As shown in Fig. 11, the air layer plays a greater role in the melting process than the 2-mm layer, the reason being predominantly the conduction load that initially affects the air layer. After that, heat transfer proceeds from the air layer to the phase-change materials according to the advancement of the layer. The melting of the phase-change materials under heating is mainly affected by natural convection as the material moves away from the wall. Thereafter, a sharp drop in melting rate is seen away from the wall since heat transfer to phase-change materials starts depending on natural convection but not on conduction. Heat transfer to the phase-change material starts predominantly in the air layer, which explains the rapid widening of this layer. Heat subsequently transfers into the phase-change materials; however, as the distance from the wall increases, the rate of heat transfer diminishes due to the effects of natural convection. Figure 12 illustrates temperature distributions in this case, while Fig. 13 illustrates the melting process, showing it near the wall in the air and gradually moving into the phase-change materials. This reflects a balance between conduction and natural convection, with the latter taking greater importance further away from the wall, thus suppressing the melting rates.
Predicted evolution of the melting process with layer of air (4 mm).
Temperature distributions with layer of air (4 mm).
Velocity distributions with a layer of air (4 mm).
Case four (6 mm air layer)
An investigation of the cell reveals a 6 mm air layer. The melting process is clearly shown in Fig. 14 to be markedly impacted by the air layer. Because heat moves from the wall to the air, first, the conduction load affects the air layer. Following the same path around the layer, heat moves from the air layer to the phase change substances. The process of melting shifts from conduction to mostly depending on spontaneous convection as heat is added to the phase change materials. As the phase change material starts to separate from the wall, one can see the change in action. It becomes clear as one gets further from the wall that the melting process slows down, causing dependency on natural convection. In contrast to conduction, natural convection is a slower process, therefore changing the mechanism of heat transfer and hence delaying the melting of the material.
Figure 15 illustrates the mechanism of heat transfer to the phase-change materials. Initially, heat is transferred to the air layer, influencing its thickness or width. Heat transfers through the air layer and subsequently enters the phase-change materials. The initial transfer from the air to the phase-change material is contingent upon the conduction load. As the distance from the wall increases, the process slows due to heat transfer becoming more reliant on natural convection, which is less efficient than conduction. The gradual slowdown results from heat transfer depending on the slower, less efficient convection mechanism, as opposed to the more rapid conduction mechanism found nearer to the wall. However, Fig. 16 illustrates the melting process as it initiates near the wall within the air layer and subsequently advances into the phase-change materials. This progression demonstrates the integrated impacts of conduction and natural convection. Initially, heat from the wall is transferred through the air layer by conduction; however, as the phase-change material moves away from the wall, natural convection becomes the primary mode of heat transfer. This transition results in a significant reduction in the melting rate as one moves further into the phase-change material. The transition from conduction to natural convection is a demand for understanding the interaction of these mechanisms in the design of systems employing phase-change materials, particularly in cases with air layers.
Predicted evolution of the melting process with layer of air (6 mm).
Temperature distributions with layer of air (6 mm).
Velocity distributions with a layer of air (6 mm).
Results comparison
Studies on the various cases demonstrate the substantial impact of the air layer’s melting. The air layer is a significant component in the heat transfer procedure, which affects the period that is required for the process of melting to be completed. The duration of the melting process without an air gap to be accomplished by 660 min. Adding a 2 mm air layer extends the melting time so that completion is reached at 780 min. The melting process is more prolonged in the presence of a 4 mm air layer, finishing at the 900th minute. Finally, the melting process concludes at the 960th minute with a 6 mm air layer. The observations indicate that the air layer significantly influences the dissolution process, with an increase in the air layer thickness correlating to a longer duration of the process. Figure 17 compares the different cases examined and demonstrates the impact of the air layer on the heat transfer process within the cell. The increase in air layer thickness significantly impedes the heat transfer process, resulting in a prolonged melting period. This effect is significant, as it illustrates how the air layer hinders efficient heat transfer, thereby delaying the overall phase change. The movement of the phase-change material is significantly influenced by the presence of the air layer. The melting process decelerates as the material distances itself from the wall and the air layer thickness increases, indicating the effects of diminished heat transfer and natural convection. Figure 18 illustrates the influence of the air layer on the total duration necessary for the melting process. The presence of a 2 mm-thick air layer leads to an 18% increase in the time required for the melting process. A 4 mm air layer results in a 37% increase, whereas a 6 mm air layer contributes to a 50% increase in melting time. The findings demonstrate the significant role of the air layer in impeding the melting process, confirming that a thicker air layer prolongs the time required for the phase change to occur.
Comparison of the melting process between the all cases.
Variation of melt fraction.
Conclusions
This study confirms the influential role of internal air layers on the melting behavior of phase change materials (PCMs) inside hexagonal cells. A numerical simulation was performed using the enthalpy-porosity model in ANSYS/FLUENT 16 to study the effect of different air layer thicknesses on the thermal response of RT42 paraffin wax. The main results are as follows:
Melting time prolongation:
-
• Without an air layer, the total melting time was 660 min.
-
• With a 2 mm thick air layer, the time increased to 780 min (+ 18%).
-
• At 4 mm, the time reached 900 min (+ 37%).
-
• With 6 mm, it extended to 960 min (+ 50%).
Physical explanation:
The presence of air creates thermal resistance and impedes heat transfer, both by conduction and natural convection, slowing down the melting process and delaying the advance of the phase front.
Implications for design:
These results highlight the importance of considering the thickness of the air layer when designing PCM-based thermal energy storage systems. Ignoring this factor can significantly reduce the thermal response and overall performance of the system.
Challenges
Several difficulties were encountered in the study of the effect of air layers on the melting process of the phase change materials (PCMs) in a multi-hexagonal cell:
-
The modelling of the interaction between the phases presents in the melting process, which involves solid, liquid, and air phases, means facing huge difficulties due to the complex interactions between these phases. The changes between these phases, especially with changing air layers, made the simulations complicated.
-
Mesh Dependency: Achieving mesh independence proved to be a difficult task, as the results’ accuracy was heavily dependent on the appropriate mesh density to be used. One has to compromise between computational speed and accuracy, since meshes are much finer when accuracy is high.
-
Appropriate representation of boundary conditions between the air layer and phase change material (PCM – especially in phase transitions) was one of the major problems. It was crucial to examine the temperature gradients at the boundaries very carefully to get an accurate simulation result.
-
Reaching numerical stability during the phase-change process, especially with the enthalpy-porosity method, was difficult due to the transient nature of the process and large changes in the material properties that occur during the phase change.
Calibration and validation of the simulations required correlation with data from earlier experiments. This process entailed fine-tuning of the material properties, heat transmission coefficients, and wall condition to match with the experimental records.
Future work
Based on the evidence generated through this study, several opportunities for further inquiry and advancement can be explored:
-
Validation of experiments is necessary; while numerical simulations provide valuable information, performing experiments on the melting process for various air layer thicknesses would provide more credible data. Future work needs to include designing experiments for measuring temperature distributions, phase change processes, and overall melting times for comparison with simulation results.
-
There is a need for further research to establish the ideal thickness of the air layer that optimizes the performance of the melting process. This could involve studying the effects of air layers combined with different cell geometries or phase change materials (PCMs).
-
Advanced numerical models that include variables such as turbulence and non-Newtonian fluid dynamics can be used to improve the predictive accuracy of the melting process in real-world applications.
-
It is essential to investigate the long-term thermal performance and stability of multi-hexagonal cells with phase change materials (PCMs) at different temperatures and environmental conditions to increase the lifespan and efficiency of energy storage systems.
-
Future research may aim to integrate optimized multi-hexagonal PCM cells with renewable energy systems such as solar energy to evaluate their potential for large-scale energy storage applications. This may contribute to enhanced sustainability and efficiency of thermal energy management systems.
Data availability
The data that support the findings of this study are available on request from the corresponding author.
References
Bull, S. R. Renewable energy today and tomorrow, Proceedings of the IEEE, vol. 89, no. 8, pp. 1216–1226, (2001). https://doi.org/10.1109/5.940290
Ibrahim, O. A. A. M., Kadhim, S. A., Hammoodi, K. A., Rashid, F. L. & Askar, A. H. Review of hydrocarbon refrigerants as drop-in alternatives to high-GWP refrigerants in VCR systems: the case of R290. Clean. Eng. Technol. 23, 100825. https://doi.org/10.1016/j.clet.2024.100825 (Dec. 2024).
Jouhara, H., Żabnieńska-Góra, A., Khordehgah, N., Ahmad, D. & Lipinski, T. Latent thermal energy storage technologies and applications: A review, International Journal of Thermofluids, vol. 5–6, p. 100039, Aug. (2020). https://doi.org/10.1016/j.ijft.2020.100039
Barrak, E. S., Hussain, H. M. & Habeeb, L. J. Experimental study for controlling airborne contaminant exposure in Iraqi negative pressure isolation rooms. Int. J. Heat Technol. 42 (3), 777–785. https://doi.org/10.18280/ijht.420307 (2024).
Al-Tajer, A. M. et al. A Numerical Simulation to Select the Optimal Thermal Agents for Building Parts, Mathematical Modelling of Engineering Problems, vol. 9, no. 5, pp. 1393–1398, Dec. (2022). https://doi.org/10.18280/mmep.090530
Hussein, H. Q., Khalaf, A. F., Jasim, A. K. & Rashid, F. L. Experimental investigation for the influence of a basement inside collector on solar chimney effectiveness. J. Mech. Eng. Res. Developments. 44 (4), 346–354 (2021).
Younsi, Z., Joulin, A., Zalewski, L., Lassue, S. & Rousse, D. R. Phase Change Materials: A Numerical Method for the Behaviour Predictions, Proceedings of the Fourth International Conference on Thermal Engineering: Theory and Applications, vol. 33, no. 0, pp. 1–7, (2009).
Shokouhmand, H. & Kamkari, B. Experimental investigation on melting heat transfer characteristics of lauric acid in a rectangular thermal storage unit. Exp. Therm. Fluid Sci. 50, 201–212. https://doi.org/10.1016/j.expthermflusci.2013.06.010 (2013).
Sun, X., Chu, Y., Mo, Y., Fan, S. & Liao, S. Experimental investigations on the heat transfer of melting phase change material (PCM). Energy Procedia. 152, 186–191. https://doi.org/10.1016/j.egypro.2018.09.079 (2018).
Babaharra, O. et al. Thermal performance analysis of hollow bricks integrated phase change materials for various climate zones, Heat Transfer, vol. 53, no. 4, pp. 2148–2172, Jun. (2024). https://doi.org/10.1002/htj.23031
Tao, Y. B. & Carey, V. P. Effects of PCM thermophysical properties on thermal storage performance of a shell-and-tube latent heat storage unit. Appl. Energy. 179, 203–210. https://doi.org/10.1016/j.apenergy.2016.06.140 (2016).
Hasan, M. I., Basher, H. O. & Shdhan, A. O. Experimental investigation of phase change materials for insulation of residential buildings, Sustain Cities Soc, vol. 36, no. October pp. 42–58, 2018, (2017). https://doi.org/10.1016/j.scs.2017.10.009
Bouhezza, A. et al. Effective techniques for performance improvement of phase change material applications: A review. J. Energy Storage. 105, 114671. https://doi.org/10.1016/J.EST.2024.114671 (Jan. 2025).
Ebrahimi, A., Hosseini, M. J., Ranjbar, A. A., Rahimi, M. & Bahrampoury, R. Melting process investigation of phase change materials in a shell and tube heat exchanger enhanced with heat pipe. Renew. Energy. 138, 378–394. https://doi.org/10.1016/j.renene.2019.01.110 (2019).
Jasim, A. K., Alwan, S. H., Nemah, A. K., Hussein, H. Q. & Hammoodi, K. A. A numerical study to improve heat transfer in a rectangular cell filled with phase change materials using several types of rods. Int. J. Heat Technol. 42 (6), 2108–2114. https://doi.org/10.18280/ijht.420629 (2024).
Hammoodi, K. A. et al. Effect of air layer on PCMs melting process inside a spherical container: A numerical investigation. Results Eng. 24, 103088. https://doi.org/10.1016/j.rineng.2024.103088 (Dec. 2024).
Hammoodi, K. A. et al. Investigation of the influence of the air layer on the phase change material melting process inside a hemicylindrical enclosure: A numerical approach. Results Eng. 24, 103337. https://doi.org/10.1016/j.rineng.2024.103337 (Dec. 2024).
Khalaf, A., Rashid, F., AL-OBAIDI, M., Ameen, A. & Mohammed, H. Numerical investigation of the effect of an air layer on the melting process of phase change materials. Mater. Renew. Sustain. Energy May. https://doi.org/10.1007/s40243-024-00261-y (2024).
Kadhim, S. A. et al. Enhancing the melting rate of RT42 paraffin wax in a square cell with varied copper fin lengths and orientations: A numerical simulation. Int. J. Thermofluids. 24, 100877. https://doi.org/10.1016/J.IJFT.2024.100877 (Nov. 2024).
Gür, M., Gürgenç, E., Coşanay, H. & Öztop, H. F. Solar-assisted radiant heating system with nano-B4C enhanced PCM for nearly zero energy buildings. Case Stud. Therm. Eng. 65, 105544. https://doi.org/10.1016/j.csite.2024.105544 (Jan. 2025).
Öztop, H. F., Gürgenç, E. & Gür, M. Thermophysical properties and enhancement behavior of novel B4C-nanoadditive RT35HC nanocomposite phase change materials: Structural, morphological, thermal energy storage and thermal stability, Sol. Energy Mater. Sol. Cells, vol. 272, p. 112909, Aug. (2024). https://doi.org/10.1016/j.solmat.2024.112909
Kurt, K., Öztop, H. F., Abu-Hamdeh, N. & Gür, M. An experimental study on cooling of model data center building by using eutectic phase change material in their walls, J. Therm. Anal. Calorim., vol. 148, no. 14, pp. 7233–7258, Jul. (2023). https://doi.org/10.1007/s10973-023-12203-3
Gurgenc, E., Gur, M., Cosanay, H., Gurgenc, T. & Oztop, H. F. Effects of position of semi-circular body on melting of a novel B4C/RT44HC PCM nanocomposite in a closed space. Case Stud. Therm. Eng. 65, 105628. https://doi.org/10.1016/j.csite.2024.105628 (Jan. 2025).
Mahdi, J. M. & Nsofor, E. C. Melting enhancement in triplex-tube latent heat energy storage system using nanoparticles-metal foam combination. Appl. Energy. 191, 22–34. https://doi.org/10.1016/j.apenergy.2016.11.036 (2017).
Talebizadeh Sardari, P., Walker, G. S., Gillott, M., Grant, D. & Giddings, D. Numerical modelling of phase change material melting process embedded in porous media: Effect of heat storage size, Proceedings of the Institution of Mechanical Engineers, Part A: Journal of Power and Energy, vol. 234, no. 3, pp. 365–383, (2020). https://doi.org/10.1177/0957650919862974
Shahsavar, A., Goodarzi, A., Mohammed, H. I., Shirneshan, A. & Talebizadehsardari, P. Thermal performance evaluation of non-uniform fin array in a finned double-pipe latent heat storage system. Energy 193, 116800. https://doi.org/10.1016/j.energy.2019.116800 (2020).
Shahsavar, A., Shaham, A. & Talebizadehsardari, P. Wavy channels triple-tube LHS unit with sinusoidal variable wavelength in charging/discharging mechanism, International Communications in Heat and Mass Transfer, vol. 107, no. June, pp. 93–105, (2019). https://doi.org/10.1016/j.icheatmasstransfer.2019.05.012
Wang, P. et al. Thermal energy charging behaviour of a heat exchange device with a zigzag plate configuration containing multi-phase-change-materials (m-PCMs). Appl. Energy. 142, 328–336. https://doi.org/10.1016/j.apenergy.2014.12.050 (2015).
Abdul-Ghafoor, Q. J., Abed, S. H., Kadhim, S. A. & Al-Maliki, M. A. Experimental and numerical study of a linear Fresnel solar collector attached with dual axis tracking system, Results in Engineering, vol. 23, p. 102543, Sep. (2024). https://doi.org/10.1016/j.rineng.2024.102543
Esapour, M., Hosseini, M. J., Ranjbar, A. A., Pahamli, Y. & Bahrampoury, R. Phase change in multi-tube heat exchangers. Renew. Energy. 85, 1017–1025. https://doi.org/10.1016/j.renene.2015.07.063 (2016).
Mat, S., Al-Abidi, A. A., Sopian, K., Sulaiman, M. Y. & Mohammad, A. T. Enhance heat transfer for PCM melting in triplex tube with internal-external fins. Energy Convers. Manag. 74, 223–236. https://doi.org/10.1016/j.enconman.2013.05.003 (2013).
Ye, W. B., Zhu, D. S. & Wang, N. Numerical simulation on phase-change thermal storage/release in a plate-fin unit. Appl. Therm. Eng. 31, 17–18. https://doi.org/10.1016/j.applthermaleng.2011.07.035 (2011).
Assis, E., Katsman, L., Ziskind, G. & Letan, R. Numerical and experimental study of melting in a spherical shell. Int. J. Heat. Mass. Transf. 50, 9–10. https://doi.org/10.1016/j.ijheatmasstransfer.2006.10.007 (2007).
Al-Abidi, A. A., Mat, S., Sopian, K., Sulaiman, M. Y. & Mohammad, A. T. Internal and external fin heat transfer enhancement technique for latent heat thermal energy storage in triplex tube heat exchangers. Appl. Therm. Eng. 53 (1), 147–156. https://doi.org/10.1016/j.applthermaleng.2013.01.011 (2013).
Hammoodi, K. A. et al. Influence of air velocity on enhancing heat transfer rate in phase change materials for efficient device temperature control. Heliyon 11 (6), e43003. https://doi.org/10.1016/j.heliyon.2025.e43003 (Mar. 2025).
Begum, L., Hasan, M. & Vatistas, G. H. Energy storage by melting commercial change phase materials in hexagonal-shaped heat exchangers. J. Thermophys. Heat. Trans. 32 (4), 1013–1030. https://doi.org/10.2514/1.T5341 (2018).
Author information
Authors and Affiliations
Contributions
Karrar A. Hammoodi: Writing – review & editing, Writing – original draft, Methodology, Investigation.Walaa Nasser Abbas: Writing – original draft, Conceptualization.Elaf S. Barrak: Writing – original draft.Ravishankar Sathyamurthy: InvestigationAbdallah Bouabidi: Project administration.Issa Omle: Writing – review & editing, software.Mujtaba A. Flayyih: Investigation.Hasan Qahtan Hussein: validation.Saif Ali Kadhim: Writing – review & editing, Validation, Supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Hammoodi, K.A., Abbas, W.N., Barrak, E.S. et al. Numerical investigation of the influence of air layer on the melting behavior of RT42 PCM in a multi-hexagonal cell. Sci Rep 15, 34368 (2025). https://doi.org/10.1038/s41598-025-17026-w
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-17026-w