Abstract
Tenosynovial giant cell tumor (TGCT) is a rare neoplasm closely associated with dysregulation of the colony-stimulating factor 1(CSF1)/CSF1R signaling pathway, faces high recurrence rates despite surgical intervention, prompting exploration of CSF1R inhibitors like pexidartinib. This retrospective pharmacovigilance study analyzed pexidartinib-associated adverse events (AEs) from FDA Adverse Event Reporting System (FAERS) data (Q4 2019–Q3 2024), employing disproportionality analyses (ROR, PRR, BCPNN, EBGM) and sensitivity assessments to evaluate 844 reports. Hepatic events (46.7% occurring within 30 days) and systemic reactions (fatigue, hair discoloration) dominated AE profiles, with median onset at 35 days (IQR 14–94). Sex-specific susceptibilities emerged, as females comprised 71.3% of cases and exhibited stronger signals for constipation and alopecia. Disproportionality analysis identified 84 significant Preferred Terms, while sensitivity analyses excluding confounders reinforced signal robustness. Despite therapeutic efficacy, hepatotoxicity and delayed-onset AEs (18.2% occurring after 6 months) necessitate rigorous adherence to risk mitigation protocols and long-term monitoring. These real-world data underscore sex-dimorphic AE patterns and validate FAERS as a critical tool for post-marketing surveillance, informing risk–benefit optimization in TGCT management.
Similar content being viewed by others
Introduction
Tenosynovial giant cell tumor (TGCT), formerly termed pigmented villonodular synovitis (PVNS), is a rare non-malignant neoplasm characterized by hyperplastic proliferative lesions arising from the synovium of joints, tendon sheaths, and bursae, predominantly affecting young adults with a female predilection1,2. Although histologically classified as benign, its locally aggressive behavior and propensity for recurrence may lead to substantial clinical morbidity. TGCT manifests in two distinct subtypes: Localized (L-TGCT) and diffuse (D-TGCT), which can occur either intra-articularly or extra-articularly. Typical clinical presentations include joint pain, swelling with hemorrhagic effusion, or palpable soft tissue masses, collectively imposing significant functional impairment and quality-of-life deterioration in affected individuals3,4.
The pathogenesis of TGCT remains incompletely elucidated, though it is believed to be associated with dysregulation of the colony-stimulating factor 1 (CSF1) signaling pathway, which drives the proliferation of tumor-associated macrophages and multinucleated giant cells within the lesion5. Surgical resection has historically served as the cornerstone therapeutic intervention for TGCT, aiming to achieve complete tumor excision and restore joint function6. Nevertheless, surgical feasibility is often compromised in cases of diffuse tumor growth or anatomically complex locations. The substantial recurrence rates (10% for localized TGCT and up to 40% for D-TGCT and frequent requirement for repeated surgical interventions underscore the urgent need for novel systemic or intra-articular treatment modalities7.
In recent years, pharmacological agents targeting the CSF1R pathway have emerged as promising therapeutic alternatives8,9. Tyrosine kinase inhibitors (TKIs) such as imatinib and nilotinib have demonstrated variable clinical efficacy. A retrospective analysis of imatinib therapy in advanced TGCT patients (n = 29) revealed an objective response rate (ORR) of 19% (5/27 evaluable cases). Conversely, nilotinib monotherapy exhibited a 5-year progression-free survival (PFS) rate of 53% in a single-arm study (n = 56), though 33% of patients (15/46) experienced clinical deterioration after a median follow-up of 11 months10,11. Notably, safety concerns persist as evidenced by a phase 2 trial where 11% of nilotinib-treated patients (6/56) developed grade 3 treatment-related adverse events, highlighting the necessity for improved therapeutic agents12. Pexidartinib, a novel oral small-molecule TKI, demonstrates enhanced selectivity for CSF1R with potent inhibitory activity. Its distinct pharmacological profile positions it as a promising candidate for addressing both efficacy limitations and safety challenges observed with earlier-generation TKIs13. Emerging clinical data suggest superior target engagement and reduced off-target effects compared to predecessors like imatinib and nilotinib.
Pexidartinib has demonstrated clinically meaningful efficacy in TGCT treatment across clinical trials. In a pivotal phase III trial involving 120 symptomatic TGCT patients, the pexidartinib arm achieved an overall response rate (ORR) of 39% (24/61) by RECIST criteria, with no responders observed in the placebo group (0%, 0/59)14. These findings underscore its potential as a disease-modifying therapy for patients with inoperable or recurrent TGCT. However, safety concerns remain a critical consideration. With extended follow-up (median 53 months), the cumulative ORR increased to 22%, suggesting sustained therapeutic effects. Notably, grade 3/4 adverse events (AEs) occurred in 44% (27/61) of pexidartinib-treated patients versus 12% (7/59) in the placebo cohort, with hepatic toxicity emerging as a dose-limiting concern (elevated ALT/AST in 29% of cases)14,15. Despite these risks, real-world evidence on long-term safety remains limited due to insufficient post-marketing surveillance data, particularly regarding rare events like irreversible cholestatic hepatitis.
The FDA Adverse Event Reporting System (FAERS) is a post-marketing surveillance database that collects and monitors adverse events associated with approved drugs16,17. FAERS provides valuable insights into the safety profile of medications in real-world settings, which can complement data from clinical trials. Given the limited real-world data on the safety of pexidartinib, leveraging FAERS can help identify potential safety signals and inform clinical decision-making. Our study aims to utilize FAERS data to evaluate the safety of pexidartinib in a larger patient population, thereby contributing to a more comprehensive understanding of its risk–benefit profile in the treatment of TGCT.
Methods and materials
Data source
This research adheres to the READUS-PV guidelines to ensure transparent and comprehensive reporting of disproportionality analyses, as detailed in Supplementary Table 1. FAERS is a publicly accessible database that aggregates AE reports submitted by healthcare professionals, patients, and pharmaceutical companies worldwide to support post-marketing drug safety surveillance. For this study, AE reports from the fourth quarter of 2019 to the third quarter of 2024 were extracted from FAERS. AE-related details are categorized into seven datasets within FAERS: DEMO, DRUG, REAC, INDICATION, OUTC, THER, and RSPR. All data utilized in this study are publicly accessible as ASCII data packages from the FAERS website (https://fis.fda.gov/extensions/FPD-QDE-FAERS/FPD-QDE-FAERS.html). Descriptions of these datasets in the FAERS database are detailed in Supplementary Table 2. Subsequent data processing and in-depth analysis were performed using RStudio (version 4.3.2).
Data processing
Data cleaning adhered to FDA recommendations: Duplicate reports were removed based on PRIMARYID and CASEID in the DEMO file. Specifically, reports with identical CASEID were retained if they had the most recent FDA_DT; when CASEID and FDA_DT coincided, the report with the higher PRIMARYID was selected. To enhance accuracy and validity, only AE reports where pexidartinib, including ingredients/trade name, was classified as the primary suspect (PS) in the DRUG file were included. Adverse events in FAERS are encoded using Preferred Terms (PTs) and categorized into System Organ Classes (SOCs) per the Medical Dictionary for Regulatory Activities (MedDRA).
For sensitivity analysis, reports containing other suspected drugs were excluded to retain cases where Pexidartinib was the sole suspected agent. Additionally, reports submitted by healthcare professionals (e.g., physicians, pharmacists) were prioritized based on information in the RSPR file.
Statistical analysis
Proportional disproportionality analyses were conducted using four metrics: Reporting Odds Ratio (ROR), Proportional Reporting Ratio (PRR), Bayesian Confidence Propagation Neural Network (BCPNN), and Empirical Bayesian Geometric Mean (EBGM) (see Supplementary Table 3 for formulas). This multi-method strategy aimed to mitigate limitations inherent to single-algorithm approaches, thereby improving reliability and precision in data mining. ROR: A signal was considered positive if ROR > 1 with a 95% confidence interval (lower limit) > 1 and ≥ 3 cases reported. PRR: A statistically significant association between the drug and target AE was defined as PRR ≥ 2, χ2 ≥ 4, and ≥ 3 reports. BCPNN: Significance threshold was set at IC025 > 0. EBGM: A signal was flagged if EBGM05 > 2.
To assess sex-based differences in AE occurrence post-Pexidartinib administration, we employed the refined ROR method following literature guidelines17. Details are shown in Supplementary Table 4.
Result
Descriptive analysis
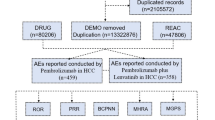
As illustrated in Fig. 1, this study extracted 844 Pexidartinib-related reports from 8,747,186 entries in the FAERS database spanning the fourth quarter of 2019 to the third quarter of 2024. The clinical characteristics of these reports are summarized in Table 1. Females and males accounted for 54.60% and 33.50% of cases, respectively. Pexidartinib-associated AEs predominantly occurred in patients aged 18 years or older. Half of the reports (50.20%) were submitted by consumers, with the overwhelming majority originating from the United States (99.90%). Non-serious outcomes comprised 78.80% of cases, while among serious outcomes, ‘‘Other serious’’ events were most frequently documented (11.70%), followed by ‘‘Hospitalization—initial or prolonged’’ (7.30%). Supplementary Fig. 1 highlights a marked increase in reporting frequency during 2024, with the number of reports from the first three quarters of 2024 already surpassing annual totals from previous years.
The process of searching Pexidartinib-associated adverse events from the FAERS.
Onset time of events
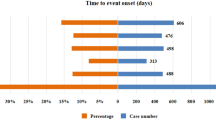
A total of 107 cases of Pexidartinib-associated AEs with available time-to-onset data were analyzed. The median time to AE onset was 35 days (interquartile range [IQR] 14–94 days). As illustrated in Fig. 2a, nearly half of the AE cases (n = 50, 46.73%) occurred within the first month following Pexidartinib initiation. Notably, a subset of AEs (n = 13, 12.15%) emerged beyond 360 days of continuous therapy. The cumulative incidence curve is presented in Fig. 2b. Additionally, the Weibull distribution analysis revealed a pattern of early failures, with the specific parameters detailed in Table 2.
Analysis of onset time. (a) Bar chart of time of onset; (b) Cumulative curve of time to onset.
Signal detection
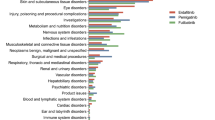
At the SOC level, Fig. 3 demonstrates that Pexidartinib-associated adverse events span 26 organ systems. These include General Disorders and Administration Site Conditions (cases: 1,230), Investigations (cases: 985), Skin and Subcutaneous Tissue Disorders (cases: 950), Injury, Poisoning, and Procedural Complications (cases: 813), and Gastrointestinal Disorders (cases: 782). Although multiple SOCs were implicated, most exhibited negative results in disproportionality analyses. Detailed outcomes of the PRR, EBGM, and BCPNN analyses are provided in Supplementary Table 5.
Signal strength of AEs of Pexidartinib at the SOC level in FAERS database.
In Supplemental Fig. 2, the Venn diagram reveals 84 PTs (22.8%) that displayed positive signals across all four analytical methodologies. Table 3 enumerates the top 10 SOCs by reporting frequency, with the five most frequently reported PTs listed under each SOC. Figure 4 illustrates the 50 most common AEs, where the top five PTs by incidence were Fatigue (cases: 372), Product Dose Omission Issue (cases: 341), Hair Colour Changes (cases: 300), Nausea (cases: 203), and Pruritus (cases: 165). Corresponding PRR, EBGM, and BCPNN analytical results are detailed in Supplementary Table 6. Additionally, all PTs were ranked by ROR values, with the top 50 PTs visualized in Supplemental Fig. 3.
Top 50 PTs with reported frequency of Pexidartinib-related AEs.
Subgroup analysis
As illustrated in Fig. 5, we enumerated the 20 most frequently reported adverse events among male and female patients. Corresponding PRR, EBGM, and BCPNN analytical results are detailed in Supplementary Table 7. Common reactions included fatigue, hair colour changes, pruritus, nausea, and rash. Given the observed disparities in incidence rates between genders, pharmacological responses to medications may exhibit sex-specific variations. Utilizing a refined ROR method for analysis, we identified constipation, alopecia, taste disorder, and nausea as adverse events demonstrating statistically significant positive signals (Fig. 6). Furthermore, the top 20 adverse events stratified by reporting frequency across different age cohorts are detailed in Supplementary Fig. 4.
Top 20 PTs with reported frequency of Pexidartinib-related AEs by gender.
Analysis of gender-differentiated risk signals in Pexidartinib.
Sensitivity analysis
In clinical practice, Pexidartinib is occasionally combined with other medications such as amlodipine, ibuprofen, and oxycodone/acetaminophen. To evaluate the independent AE profile of Pexidartinib, this study excluded reports where concomitant medications were suspected contributors to AEs. Specifically, we retained only those reports where Pexidartinib was the sole suspected agent. The results of four disproportionality analyses for each PT are detailed in Supplementary Table 8. As the FAERS is a spontaneous reporting database containing heterogeneous data quality—particularly for consumer-submitted reports—we further refined the analysis by exclusively including reports submitted by healthcare professionals (e.g., physicians, pharmacists). Outcomes of this subgroup analysis are presented in Supplementary Table 9.
To visualize the sensitivity analysis, we enumerated 84 signals consistently identified as positive across all four analytical methods in Supplemental Fig. 2. A heatmap was generated to illustrate the positivity status of these signals under two conditions: (1) reports with Pexidartinib as the sole suspected agent and (2) reports submitted by healthcare professionals. This comparative visualization is shown in Fig. 7.
Heat map for sensitivity analysis.
Discussion
As the only FDA-approved therapeutic agent for TGCT, pexidartinib warrants comprehensive post-marketing surveillance of its safety profile. This study provides a comprehensive pharmacovigilance analysis of pexidartinib through the FAERS database, addressing critical gaps in real-world safety evidence for this CSF1R inhibitor. Unlike prior clinical studies, our research utilized five-year real-world data. It described the demographic information of pexidartinib—related adverse events and listed and analyzed these events across subgroups based on gender and age differences. We also carried out a more thorough analysis of both common and uncommon adverse events, including common systemic reactions like fatigue and some rare hepatic injury responses. It was noted that neurological damage, rarely mentioned in previous studies, deserves attention, particularly the association between pexidartinib and memory impairment. In total, these findings provide viable data for the clinical use of pexidartinib and risk management strategies for TGCT drug therapy.
In pharmacovigilance studies utilizing the FAERS database, a combination of four disproportionality analyses (ROR, PRR, EBGM, and BCPNN) are commonly applied18. The ROR leverages an intuitive odds ratio framework for rapid signal screening, yet its neglect of prior distributions increases susceptibility to false positives for rare events with low reporting frequencies. The PRR, while offering enhanced stability in cross-drug comparisons through proportional rate analysis, risks overlooking weakly associated signals due to empirical threshold constraints (e.g., PRR ≥ 2)19. The EBGM employs a gamma-Poisson shrinkage model to mitigate random variability in sparse data, though its reliance on large-sample prior parameter estimation limits sensitivity for newly approved medications or rare AEs. The BCPNN quantifies signal uncertainty via information component (IC) metrics, demonstrating utility in multifactorial analyses, yet its probabilistic outputs require advanced statistical interpretation, and computational complexity hinders real-time clinical implementation20. Notably, the methodological limitations are exacerbated by the spontaneous reporting nature of FAERS, with consumer-submitted reports accounting for 50.2% of cases and a significant geographic bias in reports related to pexidartinib. In addition, ROR/PRR are vulnerable to selective reporting biases, while EBGM/BCPNN performance is highly dependent on data completeness. Consequently, the convergence of multiple algorithms (e.g., 84 PTs showing positive signals across all methods) combined with sensitivity analyses (e.g., exclusion of concomitant medication confounders) enhances signal reliability.
Within FAERS data, female patients accounted for a significantly higher proportion of pexidartinib-related reports compared to males. Among age-documented cases, the 18–64.9 age cohort predominated (84.6%), potentially reflecting TGCT’s epidemiological predilection for young adults and female populations2. Gender-stratified disproportionality analysis revealed positive signals for constipation, nausea, dysgeusia, and alopecia, suggesting potential sex-dependent variations in AE susceptibility. Notably, while only 844 pexidartinib-related reports were documented in FAERS, they encompassed 7311 PTs, indicating a high incidence of multi-AE co-reporting per patient (mean 8.66 PTs/report)-a phenomenon potentially attributable to pexidartinib’s AE-prone pharmacological profile21. Outcome analysis demonstrated that 78.80% of reports were classified as non-serious, with most AEs being manageable, possibly reflecting effective risk mitigation through the mandated Risk Evaluation and Mitigation Strategy (REMS) program22,23.
Our investigation identified multiple hepatic injury-related positive signals under the Investigations SOC, including Aspartate Aminotransferase Increased (ROR 28.34, 95% CI 24.76–32.43), Alanine Aminotransferase Increased (ROR 24.17, 95% CI 20.89–27.97), Gamma-Glutamyltransferase Increased (ROR 18.92, 95% CI 15.23–23.49), and Hepatic Enzyme Increased (ROR 15.64, 95% CI 12.89–18.97). Clinical trial data corroborate these findings, with 95% (133/140) of pexidartinib-treated patients experiencing hepatic AEs, predominantly reversible, low-grade dose-dependent transaminase elevations (91% of cases). However, 4% (5 cases) developed severe mixed/cholestatic liver injury requiring treatment discontinuation21,24. Critical cases necessitating liver transplantation have been documented25, prompting FDA-mandated REMS implementation to ensure appropriate risk management26. The mechanisms underlying pexidartinib-associated hepatotoxicity are not fully elucidated but likely involve metabolic and immune-mediated pathways. Pexidartinib is primarily metabolized by cytochrome P450 (CYP) 3A4 and uridine glucuronosyltransferase (UGT) 1A4, generating reactive intermediates that may directly damage hepatocytes or trigger immune responses27. Competitive inhibition of these enzymes by concomitant medications (e.g., CYP3A4 inducers or inhibitors) may alter pexidartinib’s pharmacokinetics and exacerbate hepatotoxicity28. Preclinical and clinical data suggest that CSF-1R inhibition itself may disrupt hepatic macrophage homeostasis, impairing Kupffer cell function and promoting cholestasis or inflammation29,30. Despite significant hepatotoxicity risks, pexidartinib remains the sole pharmacotherapeutic option for inoperable or recurrent TGCT patients31. Strict adherence to REMS protocols, including baseline/frequent hepatic monitoring and provider/patient education, enables effective risk–benefit balance in clinical practice23.
In this study, the most frequently reported SOC was General Disorders and Administration Site Conditions, with the predominant PTs being Fatigue, Pain, and Feeling Abnormal. Notably, Fatigue emerged as the most commonly reported adverse event across all PTs and demonstrated a positive signal in disproportionality analysis (ROR: 4.1 with 95% CI 3.7–4.56). This finding aligns with observations from other CSF-1R antagonists, where fatigue is frequently documented32,33,34.
Eye Disorders also exhibited a positive ROR signal. Signal analysis revealed that pexidartinib induced multiple ocular adverse events, including periorbital swelling, with high reporting frequency and signal strength. Consistent with our results, these ocular adverse reactions were previously identified in clinical trials35,36. As a CSF-1R inhibitor, pexidartinib reduces macrophage recruitment and activity in peripheral tissues29,31. Given that CSF-1R is also expressed in ocular tissues, such as retinal microglia and choroidal macrophages37, which are crucial for maintaining ocular immune privilege and tissue homeostasis, disrupting CSF-1R signaling may impair these functions. Considering macrophages are implicated in lymphatic drainage and interstitial fluid homeostasis, inhibition of CSF-1R may disrupt these processes, leading to localized fluid retention and edema. A clinical study involving 41 subjects treated with the CSF-1R inhibitor PLX3397 reported periorbital edema, fatigue, hair color changes, nausea, and dysgeusia as common adverse events38. These findings were corroborated by both FAERS database reports and positive disproportionality signals, likely attributable to CSF-1R inhibition.
Nervous System Disorders also warrant attention. Beyond commonly reported and generally tolerable events such as headache, dizziness, and somnolence, pexidartinib was associated with taste disorder and memory impairment, both demonstrating positive disproportionality signals. Pexidartinib selectively inhibits CSF-1R, which predominantly targets microglia. Prolonged treatment results in the elimination of ~ 99% of brain-wide microglia, a phenomenon further validated in vitro39. Preclinical models suggest that CSF1R inhibition may alter microglial dynamics, potentially leading to neuroinflammation or synaptic remodeling40. While clinical trials in TGCT patients have not reported direct neurotoxicity, the drug’s ability to cross the blood–brain barrier warrants caution41. Intriguingly, studies have exploited pexidartinib’s microglial-depleting effects to restore hippocampal synaptic plasticity and ameliorate social isolation-induced emotional deficits42. However, microglia are critical for normal brain function, and their dysfunction is implicated in numerous neurological disorders43,44. Specifically, microglial synaptic pruning is essential for synaptic plasticity and memory consolidation45,46,47. Although microglial populations gradually recover after pexidartinib discontinuation, the long-term neurological implications—particularly with chronic use—remain to be fully elucidated.
This study has several inherent limitations due to the nature of the FAERS database and the methodologies employed. First, as a spontaneous reporting system, FAERS data suffer from underreporting, selective reporting, and incomplete information (e.g., missing demographic details such as age and weight). Second, disproportionality analyses (e.g., ROR, PRR, BCPNN) may generate false-positive signals due to confounding factors like concomitant medications or underlying diseases, which retrospective analyses cannot fully adjust for. Third, FAERS data lack clinical context such as medication dosage, treatment duration, and patient comorbidities, making causal inference challenging. Fourth, there exists geographical and ethnic bias, with pexidartinib adverse event reports predominantly originating from the United States, potentially misrepresenting AE profiles in Asian or other populations. Finally, it should be noted that while the FAERS database offers substantial data volume, it cannot establish causality between drugs and adverse events. Future studies should incorporate prospective designs or real-world data to validate these findings and address these limitations.
Conclusion
This pharmacovigilance study provides a comprehensive evaluation of pexidartinib, the first FDA-approved systemic therapy for TGCT, through an analysis of FAERS data spanning five years. The most frequently reported AEs include hepatic injury, fatigue, and dermatological reactions, with significant signals identified in hepatobiliary, neurological, and ocular disorders. These findings highlight the necessity for vigilant monitoring, particularly during initial treatment phases and in patients requiring prolonged therapy. A critical observation is the sex-based disparity in AE susceptibility, with females disproportionately affected and exhibiting distinct signals for gastrointestinal and dermatological reactions. This aligns with preclinical evidence suggesting hormonal modulation of CSF1R pathway activity, underscoring the potential for sex-specific risk stratification in clinical management. Furthermore, the identification of delayed-onset AEs beyond 360 days emphasizes the importance of long-term surveillance, even in patients with initially favorable tolerability. The robustness of these findings is reinforced by multimethod disproportionality analysis, which prioritized high-confidence signals such as hepatic enzyme abnormalities and periorbital edema. Sensitivity analyses excluding concomitant medications and non-professional reports further validated these associations, mitigating confounding from polypharmacy and reporting biases. Clinically, strict adherence to REMS protocols—including baseline hepatic function assessments and patient education—remains paramount to optimize therapeutic outcomes.
While FAERS data offer invaluable real-world insights, limitations such as geographic reporting skew and incomplete clinical context necessitate cautious interpretation. Future studies should integrate pharmacokinetic modeling and biomarker exploration to elucidate dose-toxicity relationships and refine risk prediction. In conclusion, pexidartinib’s therapeutic value in TGCT must be balanced against its safety challenges, with personalized monitoring strategies and multidisciplinary management essential to maximize clinical benefit.
Data availability
The original contributions presented in the study are included in the article/Supplementary Material. Other requests about the data in the manuscript can be submitted in writing to the corresponding author.
References
Ottaviani, S., Ayral, X., Dougados, M. & Gossec, L. Pigmented villonodular synovitis: A retrospective single-center study of 122 cases and review of the literature. Semin. Arthritis Rheum. 40, 539–546. https://doi.org/10.1016/j.semarthrit.2010.07.005 (2011).
Monestime, S. & Lazaridis, D. Pexidartinib (TURALIO): The first FDA-indicated systemic treatment for tenosynovial giant cell tumor. Drugs R D 20, 189–195. https://doi.org/10.1007/s40268-020-00314-3 (2020).
Ofluoglu, O. Pigmented villonodular synovitis. Orthop. Clin. N. Am. 37, 23–33. https://doi.org/10.1016/j.ocl.2005.08.002 (2006).
Dania, V. et al. Treatment modalities for refractory-recurrent tenosynovial giant cell tumor (TGCT): An update. Medicina https://doi.org/10.3390/medicina60101675 (2024).
Gounder, M. M., Thomas, D. M. & Tap, W. D. Locally aggressive connective tissue tumors. J. Clin. Oncol. 36, 202–209. https://doi.org/10.1200/JCO.2017.75.8482 (2018).
Staals, E. L., Ferrari, S., Donati, D. M. & Palmerini, E. Diffuse-type tenosynovial giant cell tumour: Current treatment concepts and future perspectives. Eur. J. Cancer 63, 34–40. https://doi.org/10.1016/j.ejca.2016.04.022 (2016).
Mastboom, M. J. L. et al. Surgical outcomes of patients with diffuse-type tenosynovial giant-cell tumours: An international, retrospective, cohort study. Lancet Oncol. 20, 877–886. https://doi.org/10.1016/S1470-2045(19)30100-7 (2019).
Blay, J. Y., El Sayadi, H., Thiesse, P., Garret, J. & Ray-Coquard, I. Complete response to imatinib in relapsing pigmented villonodular synovitis/tenosynovial giant cell tumor (PVNS/TGCT). Ann. Oncol. 19, 821–822. https://doi.org/10.1093/annonc/mdn033 (2008).
Stacchiotti, S. et al. Response to imatinib in villonodular pigmented synovitis (PVNS) resistant to nilotinib. Clin. Sarcoma Res. 3, 8. https://doi.org/10.1186/2045-3329-3-8 (2013).
Cassier, P. A. et al. Efficacy of imatinib mesylate for the treatment of locally advanced and/or metastatic tenosynovial giant cell tumor/pigmented villonodular synovitis. Cancer 118, 1649–1655. https://doi.org/10.1002/cncr.26409 (2012).
Spierenburg, G. et al. Long-term follow-up of nilotinib in patients with advanced tenosynovial giant cell tumours: Long-term follow-up of nilotinib in TGCT. Eur. J. Cancer 173, 219–228. https://doi.org/10.1016/j.ejca.2022.06.028 (2022).
Gelderblom, H. et al. Nilotinib in locally advanced pigmented villonodular synovitis: A multicentre, open-label, single-arm, phase 2 trial. Lancet Oncol. 19, 639–648. https://doi.org/10.1016/S1470-2045(18)30143-8 (2018).
Lamb, Y. N. PExidartinib: First approval. Drugs 79, 1805–1812. https://doi.org/10.1007/s40265-019-01210-0 (2019).
Tap, W. D. et al. Pexidartinib versus placebo for advanced tenosynovial giant cell tumour (ENLIVEN): A randomised phase 3 trial. Lancet 394, 478–487. https://doi.org/10.1016/S0140-6736(19)30764-0 (2019).
Lamb, Y. N. Correction to: Pexidartinib: First approval. Drugs 80, 447. https://doi.org/10.1007/s40265-020-01280-5 (2020).
Sakaeda, T., Tamon, A., Kadoyama, K. & Okuno, Y. Data mining of the public version of the FDA adverse event reporting system. Int. J. Med. Sci. 10, 796–803. https://doi.org/10.7150/ijms.6048 (2013).
Xie, W. L., Li, H. H., Li, D., Li, J. Y. & Deng, A. P. A real-world pharmacovigilance analysis for agalsidase beta: Findings from the FDA adverse event reporting database. Expert Opin. Drug Saf. https://doi.org/10.1080/14740338.2024.2446429 (2024).
Chen, K., Huang, H., Chen, Y. & He, W. Association between atorvastatin and erectile dysfunction: A comprehensive analysis incorporating real-world pharmacovigilance and Mendelian randomization. Front. Pharmacol. 15, 1382924. https://doi.org/10.3389/fphar.2024.1382924 (2024).
van Manen, R. P., Fram, D. & DuMouchel, W. Signal detection methodologies to support effective safety management. Expert Opin. Drug Saf. 6, 451–464. https://doi.org/10.1517/14740338.6.4.451 (2007).
Huang, L., Guo, T., Zalkikar, J. N. & Tiwari, R. C. A review of statistical methods for safety surveillance. Ther. Innov. Regul. Sci. 48, 98–108. https://doi.org/10.1177/2168479013514236 (2014).
Lewis, J. H. et al. Pexidartinib long-term hepatic safety profile in patients with tenosynovial giant cell tumors. Oncologist 26, e863–e873. https://doi.org/10.1002/onco.13629 (2021).
Salas, M. et al. Evaluation of patient/caregiver and healthcare provider knowledge, attitudes and behavior for safety and use of pexidartinib. J. Comp. Eff. Res. 10, 953–967. https://doi.org/10.2217/cer-2020-0253 (2021).
Dharmani, C. et al. Turalio risk evaluation and mitigation strategy for treatment of tenosynovial giant cell tumor: Framework and experience. Future Oncol. 18, 1595–1607. https://doi.org/10.2217/fon-2021-1475 (2022).
Palmerini, E., Longhi, A., Donati, D. M. & Staals, E. L. Pexidartinib for the treatment of adult patients with symptomatic tenosynovial giant cell tumor: Safety and efficacy. Expert Rev. Anticancer Ther. 20, 441–445. https://doi.org/10.1080/14737140.2020.1757441 (2020).
Piawah, S. et al. A case report of vanishing bile duct syndrome after exposure to pexidartinib (PLX3397) and paclitaxel. NPJ Breast Cancer 5, 17. https://doi.org/10.1038/s41523-019-0112-z (2019).
Toyserkani, G. A., Lee, J. H. & Zhou, E. H. The risk evaluation and mitigation strategy (REMS) public dashboard: Improving transparency of regulatory activities. Pharmaceut. Med. 37, 349–353. https://doi.org/10.1007/s40290-023-00489-5 (2023).
Su, Y. et al. Exploring the effect of compound glycyrrhizin and silybinin on the metabolism of pexidartinib in rats based on cyp3a4 and cyp2c9. Adv. Pharmacol. Pharm. Sci. 2023, 6737062. https://doi.org/10.1155/2023/6737062 (2023).
Zahir, H. et al. Evaluation of absorption and metabolism-based DDI potential of pexidartinib in healthy subjects. Clin. Pharmacokinet. 61, 1623–1639. https://doi.org/10.1007/s40262-022-01172-9 (2022).
Qin, X. et al. Identifying the reactive metabolites of tyrosine kinase inhibitor pexidartinib in vitro using LC-MS-based metabolomic approaches. Chem. Res. Toxicol. 36, 1427–1438. https://doi.org/10.1021/acs.chemrestox.3c00164 (2023).
Wen, J., Wang, S., Guo, R. & Liu, D. CSF1R inhibitors are emerging immunotherapeutic drugs for cancer treatment. Eur. J. Med. Chem. 245, 114884. https://doi.org/10.1016/j.ejmech.2022.114884 (2023).
McCabe, C., Wright, H., Polson, K. & Wagner, A. J. Supporting patients in the transition to the revised pexidartinib dosing regimen: Perspectives from the multidisciplinary clinical and allied health professional team. Orphanet. J. Rare Dis. 18, 313. https://doi.org/10.1186/s13023-023-02926-9 (2023).
Machiels, J. P. et al. Phase Ib study of anti-CSF-1R antibody emactuzumab in combination with CD40 agonist selicrelumab in advanced solid tumor patients. J. Immunother Cancer https://doi.org/10.1136/jitc-2020-001153 (2020).
Gomez-Roca, C. et al. Anti-CSF-1R emactuzumab in combination with anti-PD-L1 atezolizumab in advanced solid tumor patients naive or experienced for immune checkpoint blockade. J. Immunother. Cancer https://doi.org/10.1136/jitc-2021-004076 (2022).
Autio, K. A. et al. Immunomodulatory activity of a colony-stimulating factor-1 receptor inhibitor in patients with advanced refractory breast or prostate cancer: A Phase I study. Clin. Cancer Res. 26, 5609–5620. https://doi.org/10.1158/1078-0432.CCR-20-0855 (2020).
Desai, J. et al. A phase 4, multicenter, global clinical study to evaluate discontinuation and rechallenge of pexidartinib in patients with tenosynovial giant cell tumor previously treated with pexidartinib. Cancer 131, 35634. https://doi.org/10.1002/cncr.35634 (2025).
Vaynrub, A., Healey, J. H., Tap, W. & Vaynrub, M. Pexidartinib in the management of advanced tenosynovial giant cell tumor: Focus on patient selection and special considerations. Onco. Targets Ther. 15, 53–66. https://doi.org/10.2147/OTT.S345878 (2022).
Oskam, J. A. & Danesh-Meyer, H. V. Neuro-ophthalmic complications of modern anti-cancer drugs. Graefes Arch. Clin. Exp. Ophthalmol. 262, 2269–2281. https://doi.org/10.1007/s00417-023-06350-4 (2024).
Tap, W. D. et al. Structure-guided blockade of csf1r kinase in tenosynovial giant-cell tumor. N Engl. J. Med. 373, 428–437. https://doi.org/10.1056/NEJMoa1411366 (2015).
Elmore, M. R. et al. Colony-stimulating factor 1 receptor signaling is necessary for microglia viability, unmasking a microglia progenitor cell in the adult brain. Neuron 82, 380–397. https://doi.org/10.1016/j.neuron.2014.02.040 (2014).
Weyer, M. P., Strehle, J., Schafer, M. K. E. & Tegeder, I. Repurposing of pexidartinib for microglia depletion and renewal. Pharmacol. Ther. 253, 108565. https://doi.org/10.1016/j.pharmthera.2023.108565 (2024).
Shaikh, S. N. et al. CSF-1R inhibitor PLX3397 attenuates peripheral and brain chronic GVHD and improves functional outcomes in mice. J. Neuroinflamm. 20, 300. https://doi.org/10.1186/s12974-023-02984-7 (2023).
Wang, L. et al. Pexidartinib (PLX3397) through restoring hippocampal synaptic plasticity ameliorates social isolation-induced mood disorders. Int. Immunopharmacol. 113, 109436. https://doi.org/10.1016/j.intimp.2022.109436 (2022).
Streit, W. J., Xue, Q. S., Tischer, J. & Bechmann, I. Microglial pathology. Acta Neuropathol. Commun. 2, 142. https://doi.org/10.1186/s40478-014-0142-6 (2014).
Pallares-Moratalla, C. & Bergers, G. The ins and outs of microglial cells in brain health and disease. Front. Immunol. 15, 1305087. https://doi.org/10.3389/fimmu.2024.1305087 (2024).
Tan, S. et al. CD33/TREM2 signaling mediates sleep deprivation-induced memory impairment by regulating microglial phagocytosis. Neuromolecular. Med. 25, 255–271. https://doi.org/10.1007/s12017-023-08733-6 (2023).
Puigdellivol, M. et al. The microglial P2Y(6) receptor mediates neuronal loss and memory deficits in neurodegeneration. Cell Rep. 37, 110148. https://doi.org/10.1016/j.celrep.2021.110148 (2021).
Hasegawa, Y. et al. Microglial cannabinoid receptor type 1 mediates social memory deficits in mice produced by adolescent THC exposure and 16p11.2 duplication. Nat. Commun. 14, 6559. https://doi.org/10.1038/s41467-023-42276-5 (2023).
Funding
This study was funded by the Wuhan Science and Technology Bureau (2023020201020542).
Author information
Authors and Affiliations
Contributions
Methodology, Wen-long Xie and Dan Chen. Software, Dan Chen, Jing Fu and Deng-zheng Zhang. Data curation, Dan Chen, Xia-wen Yang, and Jing Fu. Formal analysis, Dan Chen, Xia-wen Yang, Guo-qing Chen and Wei Cai. Supervision, Wen-long Xie and Wei Cai. Writing original draft, Dan Chen, Wei Cai and Wen-long Xie. All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics statement
Ethical approval was not required for the study involving humans in accordance with the local legislation and institutional requirements. Written informed consent to participate in this study was not required from the participants or the participants’ legal guardians/next of kin in accordance with the national legislation and the institutional requirements.
Generative AI statement
The authors utilized DeepSeek (https://www.deepseek.com) for grammatical improvements in this manuscript. The authors take full responsibility for the content of the publication.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, D., Yang, Xw., Fu, J. et al. A disproportionality analysis of adverse events caused by pexidartinib from the FDA adverse event reporting system. Sci Rep 15, 31689 (2025). https://doi.org/10.1038/s41598-025-17232-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-17232-6
Keywords
This article is cited by
-
Pexidartinib-related AEs reported to FAERS
Reactions Weekly (2025)