Abstract
A novel Corrax (CX) stainless steel was fabricated using the Selective Laser Melting (SLM) process. This study examined the influence of heat treatment processes on the microstructure and mechanical properties of CX stainless steel. Results indicate that SLM-fabricated CX samples mainly consist of martensite and residual austenite, with tensile strength and hardness of 1124 MPa and 337.4 HV, respectively. After solution treatment at 850 °C for 0.5 h (ST), the sample exhibited the highest martensite content, with nearly all residual austenite eliminated. Nickel and aluminum are fully dissolved within the matrix, leading to a supersaturated solid solution. This martensitic structure with a high dislocation density lays an important foundation for the subsequent aging treatment. Subsequent aging treatments at various temperatures demonstrate that when CX samples are subjected to solution aging at 850 °C for 0.5 h followed by aging at 525 °C for 4 h (ST + AT), all residual austenite is fully converted to martensite. The elements dissolved during the solution treatment precipitate, forming NiAl intermetallic compounds. This process leads to a substantial increase in tensile strength and surface hardness, achieving values of 1743 MPa and 526.8 HV, respectively. These results indicate that the solution and aging heat treatment significantly enhances the overall performance of CX samples.
Similar content being viewed by others
Introduction
Traditional manufacturing methods are often characterized by complex processes, significant material waste, and costly molds, which can hinder production efficiency and flexibility, particularly for intricate structures1. In contrast, 3D printing enables the construction of complex geometries layer by layer without the need for molds, substantially reducing material waste2. As a result of these advantages, additive manufacturing technologies have become increasingly prevalent across critical industries, including mold making, aerospace, marine engineering, and automotive manufacturing3. Among these, Selective Laser Melting (SLM) has garnered significant attention for its capacity to produce complex metal components while shortening production cycles, thereby proving highly valuable for industrial applications4.
Corrax (CX) stainless steel is a new type of martensitic precipitation-hardened stainless steel with extremely low carbon content and more than 11% chromium5. Due to its excellent corrosion resistance, dimensional stability, weldability, and superior mechanical properties, CX has become a preferred material for use in harsh environments6. Although SLM technology has successfully produced high-density metal components, the microstructure and mechanical properties of these parts still lag behind those produced by conventional methods. For instance, SLM-fabricated components may exhibit inferior surface quality and contain small pores and microcracks—metallurgical defects that can significantly diminish the lifespan of the components7. These issues can be effectively addressed through the application of appropriate heat treatment protocols.
In recent years, researchers have proposed that a reasonable heat treatment system can effectively improve the microstructure and mechanical properties of SLM formed deposition hardening stainless steel. Zhao et al.8 found that porous 17-4PH stainless steel prepared by SLM contained a large amount of martensite and a small amount of austenite. After solution aging treatment, the martensite transformed into a fine recrystallized structure, refining the martensite phase and significantly enhancing the material’s strength. Mutua et al.9 reported that the maximum tensile strength of SLM-formed martensitic stainless steel could reach approximately 2033 MPa following solution aging, representing a 1.2-fold increase compared to components manufactured using traditional methods. Sarkar et al. 10 investigated the microstructure and mechanical properties of 15–5 PH stainless steel components fabricated via SLM. After heat treatment of aging at 900 °C, the hardness of the SLM-processed 15–5 PH stainless steel was found to reach 43 HRC. Dong et al.11 suggested that dislocation strengthening is the primary hardening mechanism in SLM-processed CX stainless steel. This mechanism effectively hinders dislocation slip, significantly increasing the yield strength and tensile strength of the material. Yan et al.12 investigated CX stainless steel fabricated through SLM, highlighting its remarkable mechanical properties, with a tensile strength of 1043 MPa and a hardness of 350 HV. Jia et al.13 prepared CX stainless steel samples using SLM, observing a matrix dominated by martensite with minimal austenite. The fine-grained structure and precipitated phases provided good mechanical properties, although the high dislocation density adversely affected corrosion resistance. Zhao et al.14 investigated the heterogeneous microstructure of high strength, aging martensitic stainless steel formed by SLM, observing the presence of NiAl precipitates and alumina inclusions. The sample exhibited an ultimate tensile strength of 1647 MPa and a microhardness of 520 HV.
Research indicates that CX stainless steel fabricated through SLM exhibits extremely low porosity, along with favorable mechanical properties and hardness values. However, there has been limited systematic investigation into the microstructural characteristics and heat treatment processes of SLM-fabricated CX stainless steel. To address this gap, the present study examines the effects of three distinct heat treatment protocols —solution treatment (ST), aging treatment (AT), and solution aging treatment (ST + AT)—applied at varying temperatures on the microstructure and mechanical properties of SLM CX samples. Based on the experimental results, the study establishes the optimal heat treatment process, thereby providing a theoretical foundation for the practical application of CX stainless steel.
Experimental materials and methods
Materials and sample preparation
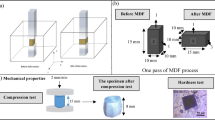
The SLM experiments were performed using an HBD-150 system (Guangdong Hanbang 3D Tech Co.). The working principle is illustrated in Fig. 1a and b. Key process parameters included a laser power of 200 W, scan speed of 900 mm/s, layer thickness of 30 μm, and hatch spacing of 90 μm. Commercial gas-atomized CX stainless steel powder served as the raw material. As shown in Fig. 1c, the powder consisted primarily of spherical particles with smooth surfaces and minor satellite particles, indicating favorable flow characteristics. Particle size distribution followed a normal pattern with a mean diameter of 40.1 μm , as shown in Fig. 1d. The chemical composition of the powder is provided in Table 1.
(a) Schematic diagram of SLM (b) Schematic diagram of cubic and tensile samples manufactured using SLM. (c) SEM image of CX stainless steel powder morphology (d) particle size distribution.
Before printing commenced, a 316L stainless steel substrate with thermophysical and chemical properties matching the powder was selected to ensure stable support, consistent print quality, and process reliability. To enhance powder flowability, the material was dried in a vacuum oven at 120 °C for 2 h prior to experimentation. High-purity nitrogen served as the protective atmosphere within the sealed build chamber during SLM processing. Oxygen levels were continuously monitored and maintained below 500 ppm to ensure smooth printing operations.
Heat treatment scheme
To establish the optimal heat treatment process, samples were aged at different temperatures and prepared under ideal parameters. First, the sample undergoes solution treatment. Subsequently, it is removed from the furnace and placed on a thermally stable surface to cool to room temperature. Aging treatment is then performed to achieve a homogeneous microstructure. All experiments were performed in an SX2-1013 box furnace with a heating rate of 10 ºC/min. The specific heat treatment procedures are displayed in Table 2.
Characterization methods
The microstructure of SLM CX samples before and after heat treatment was characterized as a plane parallel to the build direction. The printed specimens were mounted, then ground on an automated polishing machine using silicon carbide abrasive papers ranging from #80 to #2000 grit. Grinding was performed at a rotational speed of 400 r/min with a constant force of 15 N per specimen until a scratch-free surface was achieved. Subsequently, the samples were subjected to etching for approximately 30 s in Kalling’s solution (100 ml ethanol, 100 ml hydrochloric acid, and 5 g CuCl2). An optical microscope (OM, OPTIKA-CP20) and a scanning electron microscope (SEM) were utilized to examine the porosity and microstructure. Phase analysis of the samples before and after heat treatment was conducted using an X-ray diffractometer (XRD, D8 Advance) with a scanning range of 20°–100° and a scanning speed of 5°/min. Electron backscatter diffraction (EBSD) was employed to analyze microstructural information. For EBSD analysis, samples were first polished mechanically with SiC sandpaper, followed by electrolytic polishing in a solution of 10 vol% perchloric acid and 90 vol% acetic acid to relieve residual surface stress. The EBSD tests were performed at an accelerating voltage of 15 kV with a step size of 0.2 μm. Microstructural characteristics, including grain structure, phase distribution, grain size distribution, and geometrically necessary dislocation density (GNDs), were analyzed using AztecCrystal software.
The microhardness of the sample cross-sections was measured using a YZHV-1000C Vickers hardness tester. Before testing, samples were polished, with a load of 10 kgf applied for 10 s. Ten points were randomly selected on each sample’s cross-section for hardness testing, and their average was taken. Tensile tests at room temperature were conducted using a Roell Z50 universal testing machine with a stretching rate of 0.5 mm/min. Before the test, the samples were ground sequentially with sandpaper of 80, 240, and 400 grit along the loading direction until the surface showed no visible scratches, followed by a tensile test. Each tensile test was repeated at least three times, with the average result recorded as the final outcome.
Results and discussion
Microstructure and mechanical properties of the as-deposited SLMed CX
Figure 2 presents typical optical micrographs and SEM images at various magnifications of the S0 sample cross-section. The sample prepared with a power of 200W, and a scanning speed of 900 mm/s exhibited excellent cross-sectional morphology. Under these conditions, the sample’s surface exhibits a smooth morphology with only a few tiny spherical pores, and the interlayer bonding is strong. Figure 2c and d illustrate that, after etching, the sample’s surface microstructure reveals semicircular fish-scale melt pools that partially overlap15. Figure 2e reveals a significant presence of martensite. This structure forms because the high cooling rate within the fusion zone causes the austenite (γ-phase) to transform into martensite16. Near the fusion boundary, martensite undergoes tempering while reverted austenite decomposes. These processes promote dislocation rearrangement and grain refinement, resulting in fine grains (0.1–0.5 μm) and a refined substructure (Fig. 3f)17.
Cross sectional images of the as-built sample: (a) (b) OM images; (c) (d) OM images after corrosion; (e) (f) SEM images.
(a) XRD pattern of the as-built sample (b) Phase distribution polt of as-built sample.
Figure 3 shows the XRD pattern of the S0 sample, where α-phase (BCC, martensite) coexists with a small amount of γ-phase (FCC, austenite). Additionally, anisotropy in the phase components was detected in the sample. This is primarily due to the selective laser melting (SLM) process, where regions that cool rapidly produce more martensite, while slower-cooling areas retain more residual austenite or form precipitates of different phases18. This uneven phase transformation results in varying proportions of phase components in different directions 19.
EBSD was employed to further investigate the phase distribution in the deposited sample, as shown in Fig. 3b. The EBSD analysis revealed that the austenite content in the S0 sample does not exceed 0.5%, while the average grain size (dv) was about 1.43 μm. This is primarily due to the low carbon and nickel content, where low nickel levels favor the stability of martensite. Additionally, in-situ heat treatment occurring during the SLM process promotes the transformation of austenite to martensite20. Moreover, traces of residual austenite were observed in the microstructure of the S0 sample, particularly at the boundaries of the martensitic laths21. The residual austenite did not fully transform into martensite, which is mainly attributed to the segregation and diffusion of nickel (an austenite-stabilizing element) along the boundaries of the martensitic laths22.
The mechanical properties of the S0 sample are summarized in Table 3. The SLM-prepared CX specimens exhibit a tensile strength of 1124 ± 8 MPa, yield strength of 885 ± 10 MPa, elongation of 9.6 ± 0.8%, and hardness of 337.4 ± 8 HV. During the printing process, the rapid cooling characteristic refines the grains, and the high density of martensite contributes to the material’s elevated tensile strength and hardness. However, residual stresses generated during cooling, particularly tensile components, induce localized stress concentrations within the specimen, promoting premature initiation and propagation of micro-cracks, thereby resulting in diminished elongation23. The strengthening mechanisms of CX stainless steel include precipitation strengthening, grain refinement strengthening, and second phase strengthening, with dislocation strengthening being the primary mechanism24.
Effects of solution treatment and aging treatment on microstructure and mechanical properties of SLMed CX stainless steel
Microstructure evolution after solution or aging treatment
The XRD pattern of the S1 sample is shown in Fig. 4, where only the peaks corresponding to the α-Fe phase are clearly observed, indicating the detection of martensite exclusively. In contrast, Fig. 3 shows that the XRD pattern of S0 without ST also display peaks from the residual γ-Fe phase alongside the α-Fe phase. This indicates that ST promotes the transformation of residual γ-Fe phase into α-Fe phase, resulting in a reduction in austenite content. Additionally, the α-Fe phase exhibits a preferential growth trend on the (110) crystallographic plane.
XRD pattern of solution-treated sample.
EBSD analysis was conducted to further examine the microstructure of the S1 sample. As shown in Fig. 5, the morphology of martensite changed after solution treatment compared to the S0 sample. Recrystallization led to the formation of a more uniform lath martensite structure, with a preferential orientation along the (111) crystallographic plane. This low-carbon lath martensite maintained a balance of strength and toughness, achieved through moderate refinement and a more uniform distribution. Additionally, the orientation of martensite grains became more uniform, with an increase in the proportion of randomly oriented grains25. This indicates that the material achieved effective microstructural control at high temperatures, significantly enhancing structural stability.
Microstructure of solution treatment sample: (a) IPF map; (b) Phase distribution; (c) KAM map; (d) Particle size distribution.
As shown in Fig. 5b, the solution treatment resulted in more complete martensite formation, with the retained austenite content reduced to less than 0.1%, yielding a martensite phase content of 99.9% in the S1 sample. The increase of martensite is attributed to the complete dissolution of alloying elements in the austenite phase, which facilitated the transformation of austenite into martensite during cooling. In contrast, the as-deposited sample exhibited incomplete transformation due to rapid cooling and local compositional inhomogeneity, leading to a higher retained austenite content 26. Figure 5c shows a more uniform distribution of misorientation angles in the KAM map, with calculated GNDs of approximately 6.77 × 1014 m-2 using AZtecCrystal software. This indicates a reduction in high angle misorientation regions, lower dislocation density, and the relaxation of internal residual stress. As depicted in Fig. 5d, solution treatment also promoted rapid grain growth, with the maximum lath martensite length reaching 19.3 μm and an average grain size (dv) of approximately 3.69 μm.
Figure 6 presents the XRD patterns of samples S2, S3, and S4. The (110) diffraction peak of the α-Fe phase is the most prominent in all aged samples, with the diffraction peak corresponding to the γ-Fe phase also present. As the aging temperature increases, the (111) diffraction peak intensity of the γ-Fe phase significantly increases, indicating that elevated temperatures promote reverted austenite formation. Concurrently, the enhanced intensity of the α-Fe (110) peak primarily results from the decomposition of the martensitic matrix. Notably, at higher aging temperatures, slight shifts in the diffraction peak position of the α-Fe phase may occur, likely due to changes in lattice parameters or microstress variations during heat treatment27.
XRD pattern of aging treatment samples at 475, 525, and 575℃
As shown in Fig. 7a, the microstructure of the matrix after aging treatment primarily consists of fine blocky martensite, with a preferred orientation along the (111) crystallographic plane. The arrangement of crystals is relatively uniform, and the aging temperature has a minimal impact on the morphology and size of the martensite bundles. In contrast to the lamellar martensite formed in the S1 sample, simple aging treatment leads to the refinement of the martensite grains, resulting in a more uniform and stable blocky structure. Figure 7b shows a phase distribution dominated by the α-Fe phase with a smaller amount of γ-Fe phase. The γ-Fe phase content reaches 1.1%, indicating that direct aging treatment promotes the transformation from martensite to austenite. Figure 7c presents the KAM map for the S3 sample, with the GNDs calculated by AZtecCrystal software to be approximately 7.05 × 1014 m⁻2. The dislocation density is higher compared to that of the S1 sample, primarily due to the formation of precipitates during aging, which obstructs dislocation motion and promote dislocation accumulation, thereby increasing the dislocation density28. As shown in Fig. 7d, the average grain size (dv) is about 1.79 μm, indicating that aging treatment contributed to some grain growth; however, the relatively low temperature limited the grain growth process.
Microstructure of S3 sample: (a) IPF map; (b) phase distribution; (c) KAM map; (d) particle size distribution.
Changes in mechanical properties after solution or aging treatment
To investigate the mechanical properties of samples after solution and aging treatments, hardness and tensile tests were conducted. Table 4 presents statistical data on mechanical properties. Following solution treatment, the tensile strength and Vickers hardness of S1 decreased to 954 MPa and 314.9 HV, respectively, representing reductions of approximately 15% and 7% compared to the as-built sample. During the solution phase, alloying elements like Ni and Al dissolve into the martensitic matrix, and the disappearance of cell structure along with reduced residual stress lowers dislocation movement resistance, resulting in reduced tensile strength and hardness29. At an aging temperature of 475 °C, the limited precipitation of secondary phase particles weakens the strengthening effect. As the aging temperature increases, the formation of a greater number of precipitates within the martensitic matrix progressively enhances the strengthening effect, reaching a peak hardness of 498.4 HV at 525 °C, which represents a 47% increase relative to the as-built sample. However, upon reaching 575 °C, over-aging occurs, leading to a rapid reduction in hardness.
Figure 8 presents the microhardness and stress–strain curves of the CX samples after heat treatment at various aging temperatures. As shown in Fig. 8a, the hardness of the samples initially increases and then decreases with rising aging temperatures, remaining within the range of 350 to 500 HV. Figure 8b shows the stress–strain curves of the aged samples. The tensile strength aligns with the hardness, initially increasing and then decreasing. During the early aging stages, the solubility of alloying elements in the matrix decreases, causing supersaturated solute atoms to diffuse and form nucleation sites at dislocations or grain boundaries, leading to nucleation. As the aging temperature rises, elements precipitate in the martensitic matrix, forming fine NiAl hard secondary-phase particles, which generate a strong aging strengthening effect that rapidly increases the sample’s strength. At 525 °C, the precipitate particles reach a critical size, maximizing the secondary phase strengthening effect and the material’s strength. A peak tensile strength of 1678 MPa was attained under this processing condition. With further increases in aging temperature, the Orowan mechanism becomes dominant30, leading to over-aging as precipitate size and particle spacing increase, reducing the dispersion and uniformity of the secondary phase and thereby lowering the sample’s strength.
Mechanical properties of SLMed samples at different aging temperatures: (a) Vickers hardness; (b) Stress–strain curve.
The elongation of the CX sample exhibits an opposite trend, decreasing initially and then increasing. After aging at 475℃, the grain structure is relatively uniform, with minimal formation of hard precipitates, which helps to maintain good elongation. After aging at 525℃, the elongation is lowest due to the precipitation of a significant amount of hard phases, which increases the brittleness of the sample and further reduces its elongation. As shown in Fig. 6, the diffraction peak corresponding to the γ-Fe phase in sample S3 exhibits the highest peak intensity relative to other samples, which is attributed to the extensive formation of reverse austenite. Considering that austenite typically exhibits higher ductility than martensite, the observed trend in γ-Fe peak prominence aligns with the mechanical behavior. After aging at 575℃, the increase in austenite phase compensates for the negative impact of grain coarsening on ductility, resulting in improved elongation of the sample.
The effects of solution aging heat treatment on the microstructure and mechanical properties of SLMed CX stainless steel.
Microstructure evolution after solution aging treament
After solution treatment, the microstructure primarily consists of large blocky martensite. Due to the high solution treatment temperature and short holding time, residual austenite rapidly transforms into large plate-like martensite, with lengths reaching up to 15 μm (seen in Fig. 9d). Following the aging treatment, the microstructure exhibits a uniform, fine, and well-dispersed structure. This is attributed to the low aging temperature and prolonged holding time, which accelerate martensitic transformation and increase austenite content. After solution aging, the microstructure contains a large number of fine plate-like martensite, with the original austenite grain boundaries clearly visible. Optical microscopy observations in Fig. 9a, b, and c reveal that the S6 sample displays greater uniformity, with smaller interparticle distances compared to the other samples. Additionally, white particles were observed near the grain boundaries of S3 and S6 samples (seen in Fig. 9e and f). As reported by Zhang et al.31 the CX alloy is strengthened by the formation of NiAl phases. Therefore, in order to further analyze the component of the particles, EDS analysis was carried out at the grain boundaries (M1 and M2 in Fig. 9e). The EDS results, presented in Fig. 10, show that the primary elements present in the particles are Fe, Cr, Ni, and Al. Therefore, based on the chemical composition of the CX samples, it can be inferred that the white particles are primarily NiAl intermetallic compounds32.
OM and SEM images of CX after corrosion: (a), (d) S1; (b), (e) S3; (c), (f) S6.
Results of the EDS analysis (mass. %) of the asterisk positions shown in Fig. 9(e).
Figure 11 presents the XRD patterns for the S5, S6, and S7 samples, which exhibit a distinct α-Fe phase following solution aging at varying temperatures. The intensity of the α-Fe diffraction peaks increases with higher aging temperatures. The precipitation of NiAl phases reduces aluminum solubility in the α-Fe matrix and relieves lattice distortion, thereby enhancing the structural regularity of the α-Fe phase and consequently intensifying its diffraction peak in XRD patterns. Previous studies have shown that, during the initial stages of aging, segregation of Ni and Al atoms leads to the formation of solute clusters, known as GP zones or transitional phases33. In this metastable state, NiAl clusters are prone to transforming into the NiAl precipitate phase34. Additionally, a weak diffraction peak of the γ-Fe phase is observed on the (200) plane, while the (111) peak of γ-Fe is more pronounced than the (200) peak, suggesting the onset of martensitic transformation to austenite. The diffraction peaks for the S6 and S7 samples remain largely unchanged in position and width, indicating that the α-Fe phase undergoes minimal variation within this temperature range, retaining the grain size established during solution treatment. However, the intensity of the (111) and (220) diffraction peaks of the γ-Fe phase in sample S7 is greater than that in samples S6 and S5, indicating that the relatively high aging temperature has provided more sufficient conditions for promoting the formation of reversed austenite.
XRD patterns of solution-treated samples after aging treatment at 475, 525, and 575 °C.
Figure 12 presents the microstructure of S6 sample detected by EBSD technology. As shown in Fig. 12a, the sample retains a typical lath martensite structure after aging treatment, with grain orientation preferentially aligned towards the (111) crystal plane and stable grain alignment. In Fig. 12b, the phase distribution remains predominantly α-Fe, with a γ-Fe phase content of 0.2%, slightly higher than that observed in solution-treated samples, indicating the initiation of reverse austenite formation. Figure 12c presents the KAM map for the S6 sample, showing GNDs calculated at approximately 6.91 × 1014 m-2 using AZtecCrystal software, which is lower than that of the S1 sample. This reduction is attributed to the enhanced diffusion of alloy elements in the matrix during aging treatment, facilitating active dislocation movement, rearrangement, or mutual annihilation, thereby reducing dislocation density. As depicted in Fig. 12d, the average grain size (dv) is around 3.48 μm, smaller than that of the S1 sample. This reduction is a result of the formation of white precipitate particles during aging, which restricts further grain growth and stabilizes grain size. These observations confirm that precipitate strengthening in the S6 sample effectively hinders dislocation motion, significantly enhancing the sample’s strength and hardness while reducing residual strain and internal stress35.
Microstructure of S6 sample: (a) IPF map; (b) phase distribution; (c) KAM map; (d) particle size distribution.
Mechanical properties after solution aging treament
An analysis of the mechanical properties of CX stainless steel samples after solution aging treatment is summarized in Table 5 and Fig. 13. It can be seen that the hardness values of the samples initially increase and then decrease as the post-solution aging temperature rises. Due to the slow formation rate of the precipitated phase, the hardness of the sample increases slowly during the low-temperature aging stage. During the medium-temperature aging stage, the hardness is significantly enhanced, mainly because of the precipitation strengthening effect of the NiAl precipitated phase36. The S6 sample remains the highest microhardness of 526.8 HV, which is 56.1% higher than that of the S0 sample. However, the microhardness of the S7 sample decreases compared to that of S5 and S6. This is mainly because of that over aging phenomenon occurs in S7 sample due to the extremely high post-solution aging temperature37. At this stage, some of the martensite transforms into tempered martensite or residual austenite. The combined effects of precipitate coarsening and a small amount of phase transformation contribute to the reduction in hardness.
Mechanical properties of SLMed samples after solution aging treatments: (a) Vickers Hardness; (b) Stress–Strain Curves.
The stress–strain curves of the solution-aged samples are presented in Fig. 13b. After solution aging treatment, the tensile strength of the S5, S6, and S7 samples showed significant improvement. During the post-solution aging process, the nucleation rate of precipitate phases (such as intermetallic compounds like NiAl) is moderate, with small and uniformly distributed sizes. These fine and evenly distributed precipitates effectively hinder dislocation movement, increase the stress required for dislocation slip, and thus enhance both yield strength and tensile strength38. Aging treatment samples at 525 °C after Solution-treated, the density and distribution of the precipitates reach an optimal state, resulting in the most significant strengthening effect on the material. The tensile strength reaches a maximum of 1743 MPa, which is an increase of 51.8% compared to the printed state. As the aging temperature increases to 575℃ after Solution-treated, the precipitates at the grain boundaries coarsen and cluster, which reduces the effectiveness of precipitation strengthening due to over-aging phenomena39. Additionally, solution treatment eliminates the high-density dislocation networks and fine substructures formed during SLM. Although subsequent aging increases strength, it sacrifices ductility.In contrast, direct aging leverages this "metastable, high-defect" initial microstructure to promote uniform, fine precipitate strengthening. This approach enhances strength while retaining greater ductility.Therefore, directly aged samples exhibit higher elongation than solution aging treatment samples40. Overall, solution aging treatment SLMed CX stainless steel exhibits superior mechanical properties.
Conclusions
A novel high-performance CX stainless steel (PHSS) was fabricated using SLM technology. The effects of three different heat treatment regimens on the microstructural evolution, precipitation strengthening behavior, and mechanical properties of the SLMed CX stainless steel were systematically investigated. The main conclusions can be generalized as follows:
-
(1)
SLM can effectively produce CX stainless steel with minimal defects and a fine martensitic microstructure. The as-deposited specimens exhibit a matrix composed predominantly of fine martensite, with small amounts of reverted austenite. Solution treatment at 850 °C for 0.5 h leads to the transformation of residual austenite into martensite. The tensile strength and hardness of the solution-treated specimens are 954 ± 10 MPa and 314.9 ± 6.5 HV, respectively, representing approximately 84% and 93% of those of the as-deposited samples.
-
(2)
Following aging treatment, the matrix consists primarily of fine lath martensite. The aging temperature has minimal impact on the morphology and size of martensitic packets. However, increasing the aging temperature promotes the reversion of martensite to austenite. Maximum hardness (498.8 ± 10.2 HV) and tensile strength (1678 ± 15 MPa) are achieved at an aging temperature of 525 °C.
-
(3)
After solution and aging treatments, a significant number of massive martensite structures are observed in the specimens. At lower aging temperatures (475℃), although no austenite peak is detected, the original austenite grain boundaries are preserved. Additionally, in the S3 and S6 samples, fine and dispersed NiAl precipitates are observed on the martensite matrix. The SLMed specimens treated with a solution treatment at 850 °C for 0.5 h followed by aging at 525 °C for 4 h exhibit the highest hardness (526.8 ± 13.1 HV) and ultimate tensile strength (1743 ± 19 MPa), which represent an increase of approximately 52% compared to the as-deposited samples.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Zhao, L. et al. Microstructure and mechanical properties of 316L stainless steel manufactured by multi-laser selective laser melting (SLM). Mater. Sci. Eng.: A 913, 147053. https://doi.org/10.1016/j.msea.2024.147053 (2024).
Ma, Y., Gao, Y., Zhang, H., Men, Z. & Yang, L. A comprehensive study on the microstructure evolution and mechanical property characterization of selective laser melted 18Ni300 stainless steel during heat treatment processes. J. Market. Res. 33, 51–60. https://doi.org/10.1016/j.jmrt.2024.09.054 (2024).
Asapu, S., Gupta, A. & Jha, S. K. Effect of laser scan strategy and heat treatment microstructural and mechanical characterization of selective laser melted 17–4 PH stainless steel. J. Mater. Eng. Perform. 33, 202–213. https://doi.org/10.1007/s11665-024-09470-y (2024).
Jiang, C.-M., Ho, J.-R., Tung, P.-C. & Lin, C.-K. Fatigue crack growth behavior of selective laser melted martensitic stainless steel. Int. J. Fatigue 179, 108060. https://doi.org/10.1016/j.ijfatigue.2023.108060 (2024).
Shahriari, A. et al. Microstructure and corrosion behavior of a novel additively manufactured maraging stainless steel. Electrochi. Acta 339, 135925. https://doi.org/10.1016/j.electacta.2020.135925 (2020).
Hooper, P. A. Melt pool temperature and cooling rates in laser powder bed fusion. Addit. Manuf. 22, 548–559. https://doi.org/10.1016/j.addma.2018.05.032 (2018).
Karlsson, D. et al. Elemental segregation in an AlCoCrFeNi high-entropy alloy—A comparison between selective laser melting and induction melting. J. Alloy. Compd. 784, 195–203. https://doi.org/10.1016/j.jallcom.2018.12.267 (2019).
Zhao, Z. et al. Microstructure and properties of porous 17–4PH stainless steel prepared by selective laser melting. Trans. Indian Inst. Met. 75, 1641–1648. https://doi.org/10.1007/s12666-021-02388-2 (2022).
Mutua, J., Nakata, S., Onda, T. & Chen, Z.-C. Optimization of selective laser melting parameters and influence of post heat treatment on microstructure and mechanical properties of maraging steel. Mater. Des. 139, 486–497. https://doi.org/10.1016/j.matdes.2017.11.042 (2018).
Sarkar, S., Mukherjee, S., Kumar, C. S. & Kumar Nath, A. Effects of heat treatment on microstructure, mechanical and corrosion properties of 15–5 PH stainless steel parts built by selective laser melting process. J. Manuf. Process. 50, 279–294. https://doi.org/10.1016/j.jmapro.2019.12.048 (2020).
Dong, D. et al. Selective laser melting (SLM) of CX stainless steel: Theoretical calculation, process optimization and strengthening mechanism. J. Mater. Sci. Technol. 73, 151–164. https://doi.org/10.1016/j.jmst.2020.09.031 (2021).
Yan, X. et al. Study of the microstructure and mechanical performance of C-X stainless steel processed by selective laser melting (SLM). Mater. Sci. Eng.: A 781, 139227. https://doi.org/10.1016/j.msea.2020.139227 (2020).
Zhao, X., Jia, D., Wei, S., Gao, Y. & Liu, H. Investigation of tribo-corrosion behaviors of SLM-printed CX stainless steel under different loads. J. Market. Res. 30, 5266–5277. https://doi.org/10.1016/j.jmrt.2024.04.244 (2024).
Zhao, X., Zhao, K., Gao, Y. & Wang, D. Influence of Microdefect on Mechanical Behaviors of CX Stainless Steel Produced by Selective Laser Melting. Met. Mater. Int. https://doi.org/10.1007/s12540-024-01740-7 (2024).
Pirgazi, H. et al. Texture evolution in selective laser melted maraging stainless steel CX with martensitic transformation. J. Mater. Sci. 56, 844–853. https://doi.org/10.1007/s10853-020-05290-2 (2020).
Li, Y. & Gu, D. Thermal behavior during selective laser melting of commercially pure titanium powder: Numerical simulation and experimental study. Addit. Manuf. 1–4, 99–109. https://doi.org/10.1016/j.addma.2014.09.001 (2014).
Prakash, P., Midawi, A., Muhammad, W., Wells, M. & Hadadzadeh, A. Role of heat treatment conditions in the high-temperature deformation behavior of laser-powder bed fused Fe–Cr–Ni–Al maraging stainless steel. Mater. Sci. Eng.: A 909, 146837. https://doi.org/10.1016/j.msea.2024.146837 (2024).
Huang, C. et al. Ductilization of selective laser melted Ti6Al4V alloy by friction stir processing. Mater. Sci. Eng., A 755, 85–96. https://doi.org/10.1016/j.msea.2019.03.133 (2019).
Krakhmalev, P., Yadroitsava, I., Fredriksson, G. & Yadroitsev, I. In situ heat treatment in selective laser melted martensitic AISI 420 stainless steels. Mater. Des. 87, 380–385. https://doi.org/10.1016/j.matdes.2015.08.045 (2015).
Fang, R. et al. Effect of selective laser melting process parameters on the microstructure and properties of a precipitation hardening stainless steel. Mater. Des. 212, 110265. https://doi.org/10.1016/j.matdes.2021.110265 (2021).
Ateba Betanda, Y. et al. Measurement of stored energy in Fe–48%Ni alloys strongly cold-rolled using three approaches: Neutron diffraction, dillamore and KAM approaches. Mater. Sci. Eng.: A 614, 193–198. https://doi.org/10.1016/j.msea.2014.07.037 (2014).
Moussa, C., Marc Bernacki, R. & Besnard, N. B. About quantitative EBSD analysis of deformation and recovery substructures in pure tantalum.. IOP Conf. Ser.: Mater Sci. Eng. 89, 1–8 (2015).
Li, C. et al. Effect of heat treatment on microstructure and mechanical properties of 17–4PH stainless steel manufactured by laser-powder bed fusion. J. Market. Res. 26, 5707–5715. https://doi.org/10.1016/j.jmrt.2023.08.283 (2023).
Sun, L. et al. A novel ultra-high strength maraging steel with balanced ductility and creep resistance achieved by nanoscale β-NiAl and Laves phase precipitates. Acta Mater. 149, 285–301. https://doi.org/10.1016/j.actamat.2018.02.044 (2018).
Bösing, I. et al. Influence of heat treatment on the microstructure and corrosion resistance of martensitic stainless steel. AIP Adv 9, 065317. https://doi.org/10.1063/1.5094615 (2019).
Mao, C., Liu, C., Yu, L., Li, H. & Liu, Y. Discontinuous lath martensite transformation and its relationship with annealing twin of parent austenite and cooling rate in low carbon RAFM steel. Mater Des 197, 109252. https://doi.org/10.1016/j.matdes.2020.109252 (2021).
Nong, X. D. et al. Selective laser melting and heat treatment of precipitation hardening stainless steel with a refined microstructure and excellent mechanical properties. Scripta Mater. 178, 7–12. https://doi.org/10.1016/j.scriptamat.2019.10.040 (2020).
Asgari, H. & Mohammadi, M. Microstructure and mechanical properties of stainless steel CX manufactured by direct metal laser sintering. Mater. Sci. Eng., A 709, 82–89. https://doi.org/10.1016/j.msea.2017.10.045 (2018).
Li, S. et al. Effects of heat treatment influencing factors on microstructure and mechanical properties of a low-carbon martensitic stainless bearing steel. Mater. Sci. Eng., A 605, 229–235. https://doi.org/10.1016/j.msea.2014.03.061 (2014).
Casati, R., Lemke, J., Tuissi, A. & Vedani, M. Aging behaviour and mechanical performance of 18-Ni 300 steel processed by selective laser melting. Metals 6(9), 218. https://doi.org/10.3390/met6090218 (2016).
Zhang, J. et al. Effect of process parameters and heat treatment on the properties of stainless steel CX fabricated by selective laser melting. J. Alloys Compd 877, 160062. https://doi.org/10.1016/j.jallcom.2021.160062 (2021).
Tseng, C.-Y. et al. Effect of repeated-tempering induced martensite on the microstructural evolution in 17–7 PH stainless steel. Mater. Charact. 214, 114091. https://doi.org/10.1016/j.matchar.2024.114091 (2024).
Huang, J. & Zhang, D. Effect of heat treatment on precipitation behavior of second phase and property evolution of martensitic stainless steel. Mater. Today Commun. 37, 107267. https://doi.org/10.1016/j.mtcomm.2023.107267 (2023).
Wu, L. et al. Unveiling the cellular microstructure–property relations in martensitic stainless steel via laser powder bed fusion. Int. J. Miner. Metall. Mater. 31, 2476–2487. https://doi.org/10.1007/s12613-024-2947-z (2024).
Shahriari, A. et al. Quasi in-situ study of microstructure in a laser powder bed fusion martensitic stainless steel. Metall. and Mater. Trans. A. 55, 1302–1310. https://doi.org/10.1007/s11661-024-07353-4 (2024).
Hadadzadeh, A., Shahriari, A., Amirkhiz, B. S., Li, J. & Mohammadi, M. Additive manufacturing of an Fe–Cr–Ni–Al maraging stainless steel: Microstructure evolution, heat treatment, and strengthening mechanisms. Mater. Sci. Eng.: A 787, 139470. https://doi.org/10.1016/j.msea.2020.139470 (2020).
Chiu, P.-H., Chen, Z.-W., Chen, C.-Y. & Yang, J.-R. Effect of sub-zero treatment on the microstructural characteristics and mechanical properties of maraging stainless steel. J. Mater. Eng. Perform. 33, 4815–4830. https://doi.org/10.1007/s11665-023-08294-6 (2023).
Jiao, Z. B., Luan, J. H., Miller, M. K. & Liu, C. T. Precipitation mechanism and mechanical properties of an ultra-high strength steel hardened by nanoscale NiAl and Cu particles. Acta Mater. 97, 58–67. https://doi.org/10.1016/j.actamat.2015.06.063 (2015).
Shi, Y. et al. Mechanical property evaluation of a SLMed martensitic stainless steel. Acta Metall Sin (English Letters) 33, 1466–1476. https://doi.org/10.1007/s40195-020-01128-7 (2020).
He, C. et al. The new design to improve the stability of retained austenite and mechanical properties in super martensitic stainless steel. Mater. Charact. 217, 114342. https://doi.org/10.1016/j.matchar.2024.114342 (2024).
Acknowledgements
This research was funded by the Shandong Province Key Research and Development Program—Research and industrialization of high-precision intelligent mine crusher, grant No. 2024TSGC0902. This research was also funded by the Shandong Jiaotong University discipline backbone research start-up fund. This research was also supported by the technology development project: Lightweight Intelligent Fast Boat Technology (2024370105122331).
Author information
Authors and Affiliations
Contributions
Shaoqian Wu: Formal Analysis, Writing—original draft, Visualization. Tianshu Wang: Data curation, Supervision. Shuo Wu: Resources, Funding acquisition, Writing—Review & Editing. Shilong Xing: Conceptualization, Writing—Review & Editing. Jiabin Hou: Supervision. Yuantao Zhao: Investigation. Zongan Li: Visualization. Yanbo Liu: Project Administration.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Wu, S., Wang, T., Wu, S. et al. Effect of heat treatment on the microstructure and mechanical properties of CX stainless steel fabricated by selective laser melting. Sci Rep 15, 32257 (2025). https://doi.org/10.1038/s41598-025-17582-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-17582-1