Abstract
An important limitation of the pivotal randomized controlled trials that compared transcatheter aortic valve replacement (TAVR) to surgical aortic valve replacement (SAVR) is that the SAVR arm scarcely included sutureless bioprosthetic valves. We identified 13 retrospective propensity-matched studies in low (n = 2), intermediate (n = 8), and high-risk (n = 3) patients, using EuroSCORE and STS score to assess perioperative risk. One large registry drove the outcome in low-risk patients, showing better early survival with TAVR, lower rates of stroke and acute kidney injury. Intermediate-risk patients showed improved early and medium-term survival with SU-AVR, whilst in high-risk patients, no significant differences were seen between treatment options. Overall, across all risk categories, the rates of moderate and severe aortic regurgitation and permanent pacemaker implantation were significantly lower with SU-AVR, while transprosthetic gradients and duration of hospital stay were higher compared to TAVR. The differences in survival in the intermediate risk group are not in line with conclusions of pivotal randomized trials comparing TAVR with SAVR. Specific features of SU-AVR may account for these survival differences, positioning SU-AVR as a valid and safe alternative for patients at intermediate risk. Awaiting confirmation in randomized trials, careful patient selection and consideration of either of the AVR options in the heart team remain crucial.
Similar content being viewed by others
Introduction
Degenerative aortic valve stenosis (AS) is the most prevalent heart valve disease in the western world, affecting 3% of the population above 75 years, and is one of the most frequently observed valve pathologies in daily clinical cardiology and cardiac surgery practice1. Surgical aortic valve replacement (SAVR) is the gold standard treatment for patients with symptomatic severe AS at low surgical risk below the age of 65 (American guidelines) or 75 (European guidelines)2,3. In contrast, patients > 80y (American guidelines) or ≥ 75y (European guidelines) should rather be considered for transcatheter aortic valve replacement (TAVR) when technically feasible. These recommendations leave room for an intermediate risk group (around 65-80y) in which both options are valuable alternatives. Final treatment choice is established through a process of shared decision making within the Heart Team, taking into account individual patient comorbidities, technical considerations, and importantly, patients’ preference. The guidelines do not specifically mention the use of sutureless or rapid deployment valves (RDV) for SAVR due to lack of randomized controlled trials (RCT) between sutureless aortic valve replacement (SU-AVR) and TAVR. In the search for innovative and less invasive alternatives to standard SAVR in a rapidly growing elderly population, another option has emerged besides TAVR (Central figure). This alternative avoids placement and tying of multiple sutures, and is referred to as sutureless aortic valve replacement (SU-AVR). The concept has existed for more than 50 years but the idea and the valves have been re-modernized4. Currently, two valve types are available: a sutureless valve called Perceval, and another valve that needs only 3 sutures, called Intuity (also referred to as RDV) The rapid-deployment Intuity Elite (Edwards Life Sciences, USA) works by a stainless steel cloth-covered frame that is delivered through balloon catheter expansion in the correct annular position5. The Perceval S or more recently the Perceval PLUS (Corcym, Italy, previously manufactured by Sorin group and LivaNova) utilizes a self-expanding nitinol frame that has a memory and deploys and positions the valve without the need for additional sutures6. Both approaches require a surgical incision, cardiopulmonary bypass (CPB), aortic cross clamping (AXC) and excision of the diseased calcified native aortic valve and debridement of the annulus. In comparison to the traditional stented aortic surgical prostheses, they do not need extensive suturing7. The main hypothesis is that faster implantation times may lead to shorter overall duration of the operation, with shorter CPB and AXC times, which is ideal in combination with minimally invasive access.
Because the sutureless valves, in their current form, are relatively new, there are few studies directly comparing TAVR with SU-AVR. The present systematic review therefore summarizes the available evidence and explores the potential benefit of SU-AVR over TAVR in patients at low, intermediate or high risk for SAVR.
Methods
Data sources and searches
We performed a systematic review of studies that compare TAVR and SU-AVR for the treatment of symptomatic severe AS. The electronic databases PUBMED, google scholar, LIMO and the Cochrane library were searched in October 2023. The MESH terms and appropriate free text was used: “sutureless aortic valve replacement”, “rapid deployment aortic valve replacement”, “surgical aortic valve replacement”, “transcatheter aortic valve implantation”, “transcatheter aortic valve replacement”, “minimally invasive aortic valve replacement”, “aortic stenosis”, “TAVI”, “TAVR”, “SAVR”, “AVR”, “AS”. The reference lists of articles found were also screened for relevance.
Eligibility criteria and study selection
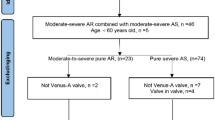
Studies that were included in this review directly compared TAVR with SU-AVR, using a propensity score matched analysis. Abstracts, expert opinions and editorials, case reports and case series with less than ten patients were excluded. If more studies were published from the same author, center or database, only the one with the most complete dataset was included. There were no restrictions on publication language, but all studies used in the analysis were in English. An overview of the studies is presented in (Fig. 1). Three studies were manually excluded: one very small non-propensity matched study8; another study by Kamperidis et al. because of the use of the 3f Enable sutureless valve, which was taken off the market in 20159. Lastly, a low-risk study by Muneretto et al. was excluded for risk of overlap with another study from the same author in patients at intermediate risk, the latter study being included10. The quality of studies was assessed by determining the level of evidence according to the criteria provided by the Oxford Centre for Evidence-based Medicine. All thirteen studies were retrospective matched cohort studies giving them a level 3b evidence. Every study has limitations besides the low number of patients, the absence of prospective randomization and propensity matching, not fully accounting for selection bias. Risk of bias was investigated using the ROBINS-I-tool and is shown in (Fig. 2).
PRISMA flowchart diaphragm from literature search to final analysis.
ROBINS-I-tool risk of bias summary with (A) traffic light plot and (B) summary plot. ROBINS-I: Risk of bias in non-randomized studies of interventions.
Study outcomes
The main study outcomes are in-hospital, 30-day, and 1 and 2-year survival. Other frequently reported outcomes are postoperative complications including stroke, acute kidney injury (AKI) and permanent pacemaker implantation (PPI). With regards to hemodynamics, most studies present postoperative echocardiographic parameters such as more than mild (para)valvular leakage (PVL) and the mean and peak transprosthetic gradients (TPG). Some studies also include these parameters in follow-up evaluations. Interestingly, some studies also report length of stay in the intensive care unit (ICU) or hospital, sometimes including a cost analysis of different techniques.
Statistical analysis
Meta-analysis was performed using SAS software (version 9.4) on the log Odds Ratios (OR) of binary outcomes (in-hospital mortality, 30-day mortality, stroke, PPI, AKI, PVL ≥ grade 2), on the log Hazard Ratio (HR) of the all-cause mortality survival outcome and on continuous outcomes (peak and mean postoperative TPG). For the continuous outcomes, medians and quartiles were transformed into means and standard deviations following Luo et al. (2017), and Wan et al. (2014)11,12. A sensitivity analysis was performed that excludes this transformed information to examine the robustness of the results.
The random-effects approach of DerSimonian and Laird13 was used to obtain a combined estimate. Between-study heterogeneity was evaluated using Chi² and I² statistics: I² = 100%*(Chi² - df)/df, where df is the degrees of freedom corresponding to the Chi² statistic14. I2 is the percentage of total variation in study estimates that is due to heterogeneity. Prediction intervals were reported alongside the effect estimates to show the extent of between-study variation. These analyses were performed overall and per surgical-risk subgroup. The results of the meta-analysis are presented using forest plots displaying the effect estimates and confidence intervals for the individual studies, the risk subgroups and the pooled study population. Funnel plots (Supplemental) were used to assess the publication bias of the included studies, with the addition of Egger’s regression tests. For none of the funnel plots was the p-value of the Egger’s regression test smaller than 0.05, indicating that the studies were symmetrically distributed. However, caution is warranted as the Egger’s regression test may be underpowered when the number of studies is lower than 10, which is the case for acute kidney injury, peak and mean postoperative gradient and all-cause mortality.
To perform a meta-analysis on the survival data and estimate the underlying Individual Person Data (IDP) from the Kaplan-Meier (KM) curves, the package IPDfromKM was used in Rstudio (version 2023.9.1.494)15. Then two statistical strategies were utilized. Firstly, we gathered the hazard ratios that were available in some individual studies and calculated their standard errors based on the confidence intervals. We combined them with hazards ratios and standard errors obtained from Cox regressions on the IPD data for which only Kaplan-Meier curves were available. Then we performed a random-effects meta-analysis on the log(HR). Secondly, we combined the IPD data from the individual studies into two Kaplan-Meier curves, one for SU-AVR and one for TAVR, and the 1-year and 2-year survival probabilities were estimated with 95% confidence intervals (CI). The two resulting Kaplan-Meier curves were compared with the log-rank test.
Weighted averages for each risk group were calculated as the sum of (number/percentage reported by a study multiplied by the number of patients in the study) for each study and then divided by the total patient number for each risk group.
Results
Patient characteristics
Baseline patient characteristics were similar between the two treatment groups due to propensity score matching across all studies (Table 1). Mean age ranged from 75.0 to 82.6 years. Surgical risk scores were reported using the logistic EuroSCORE, EuroSCORE II or STS score. Studies were stratified according to surgical risk, with a low (< 10%; < 1.5%; <4%), intermediate (10–20%; 1.6-5.0%; 4–8%) and high-risk group (> 20%; > 5%; > 8%), respectively16,17,18. The risk categorization of the study from Chung et al. 2021 was assumed to be intermediate based on the reported number of patients with a EuroSCORE II of ≥ 4%.
Overall
When considering the entire patient population there was no difference in early mortality (combining 30 days and in-hospital mortality) between SU-AVR and TAVR (OR 0.719 (95% CI: 0.370–1.399) (Fig. 3). No difference in stroke (OR 0.999 (95% CI: 0.593–1.681)) or AKI (OR 1.247 (95% CI: 0.451–3.448) was seen (Fig. 4A-B). SU-AVR was associated with less permanent pacemaker implantation (OR 0.659), less PVL (graded at least moderate) (OR 0.199), but slightly higher post-operative gradients (peak gradient: 3.108 mmHg (CI: 0.136–6.079)) and higher mean gradients: 2.866 mmHg (CI: 1.292–4.439))(Fig. 4C-F). Overall, in these observational studies, SU-AVR seems to be related with a lower mortality rate (HR: 0.665 CI: 0.488, 0.906)(Fig. 5) and the Kaplan-Meier curves are significantly different, favouring SU-AVR (1-year mortality: SU-AVR: 6.23% (CI: 4.9–7.9%) vs. TAVR: 9.15 (CI: 7.5–11.1%), 2-year mortality: SU-AVR: 9.90% (CI 8.1–12.1%) vs. TAVR: 13.87% (CI: 11.7–16.4%), Log-rank p = .0017)(Fig. 6A).
Forest-plot of early mortality showing Odds ratio and 95% confidence interval. SU-AVR: sutureless aortic valve replacement, TAVR: transcatheter aortic valve replacement.
Forest-plots of postoperative complication rates and hemodynamic performance showing Odds ratio or difference of the mean and 95% confidence interval. (A) Stroke; (B) AKI: acute kidney injury; (C) Permanent pacemaker implantation; (D) PVL: paravalvular leakage; (E) Peak postoperative gradient, (F) Mean postoperative gradient. SU-AVR: sutureless aortic valve replacement, TAVR: transcatheter aortic valve replacement.
Forest plot showing Hazard Ratios and 95% confidence intervals of all-cause mortality. SU-AVR: sutureless aortic valve replacement, TAVR: transcatheter aortic valve replacement.
Kaplan-Meier plots showing pooled overall survival. (A) Overall; (B) Low-risk patients; (C) Intermediate-risk patients; (D) High risk patients. SU-AVR: sutureless aortic valve replacement, TAVR: transcatheter aortic valve replacement.
Summary of upsides and downsides of SU-AVR and TAVR. AVR: aortic valve replacement, CPB(T): cardio-pulmonary bypass (time), GA: general anesthesia, PPI: permanent pacemaker implantation, PVL: paravalvular leakage, SU-AVR: sutureless aortic valve replacement, TAVR: transcatheter aortic valve replacement, XCT: aortic cross-clamp time.
Low-risk patients
Primary endpoint
Only two studies included low risk patients, both reporting early mortality (OR: 2.869 with 95% CI 1.479–5.567) (Fig. 3), which was significantly higher for SU-AVR in the study of Abdel-Wahad et al. (Table 2)19,20. This report comes from the German aortic valve registry (GARY) and includes two types of surgical valves (the sutureless Perceval and the rapid-deployment Intuity) (Fig. 7). Vilalta et al. found similar mortality and stroke rates, but with an increased risk of heart failure hospitalization for SU-SAVR patients at 2-year follow-up (Table 2). No significant difference for all-cause mortality was observed between both procedures (1-year mortality: SU-AVR: 8.19% vs. TAVR: 8.13%, 2-year mortality: SU-AVR: 13.02% vs. TAVR: 14.28%, HR: 0.97 (CI: 0.52, 1.82), p = .936) (Figs. 5 and 6B).
Secondary endpoints
Postoperative PPI was significantly lower in SU-AVR for both studies (OR: 0.597, CI 0.457–0.780), which was confirmed in our meta-analysis (Fig. 4C). Stroke rate was found to be significantly lower for TAVR by Abdel-Wahab et al. (OR: 2.442, CI: 1.242–4.804);19,20. Nevertheless, our meta-analysis could not show a significant difference between the two procedures regarding stroke in low-risk patients (Fig. 4A). The GARY registry also reported AKI (OR: 5.076 with CI: 2.107–12.229) (Fig. 4B), ICU stay (sutureless/RDV: 2 days vs. TAVR: 1 day, p < .001) and hospital length of stay (sutureless/RDV: 10 days vs. TAVR: 7 days, p < .001)19. The results of these outcomes are presented in Fig. 4; Table 2.
Hemodynamic performance at discharge
Hemodynamic performance was reported in both studies, with significantly lower mean TPG for TAVR. Meta-analysis: Difference of the means (DoM): 3.321 with 95% CI (-0.647–7.288). More than mild PVL (OR: 0.496 with CI (0.239–1.030) showed a non-significant trend towards less PVL in the low-risk SU-AVR patients. (Fig. 4D)19,20.
Intermediate-risk patients
Primary endpoint
For early mortality, all eight studies except Gerfer et al. showed results favouring SU-AVR (OR: 0.425, CI (0.264–0.683)21,22,23,24,25,26,27,28. Figure 3). Most studies reported 1-year mortality (Sutureless/RDV: 1.7–8.4% vs. TAVR: 7-21.4%)23,24,25,26 and/or 2-year mortality (Sutureless/RDV: 4.9–8.4% vs. TAVR: 10.9%-33.8%)22,24. When all the intermediate risk studies were combined, 1-/2-year mortality was lower in the SU-AVR group (1-year mortality: SU-AVR: 7.39% vs. TAVR: 11.97%, 2-year mortality: SU-AVR: 9.68% vs. TAVR: 15.01%, HR: 0.579 (CI: 0.363–0.925), Log-rank p = .0014)(Figs. 5 and 6C).
Secondary endpoints
All studies reported PPI rates (OR: 0.906, CI (0.495–1.659), . 4C)21,22,23,24,25,26,27,28and meta-analysis confirms the wide spread of reported results, with an I² = 66.68 (p = .0038), showing substantial heterogeneity. Other reported postoperative complications included stroke (OR: 0.691 with CI (0.394–1.212)21,22,23,24,25,26,27,28 and AKI (OR: 0.824 with CI (0.313–2.170)21,22,23,24,26,28. Hospital length of stay (sutureless/RDV: 8.1–13 days vs. TAVR: 4.5–9.5 days) and ICU length of stay (sutureless/RDV: 1-5.9 days vs. TAVR 1-3.2 days) were longer in the SU-AVR group across all studies22,24,25,26,27,28.
Hemodynamic performance at discharge
Hemodynamic valve performance in terms of peak (DoM: 3.025 with 95% CI (-0.315–6.364) and mean TPG (DoM: 3.321 with 95% CI (-0.647–7.288) favored TAVR, although SU-AVR showed less PVL (OR: 0.125 with CI (0.064–0.247)21,22,23,24,25,26,27,28.
High-risk patients
Primary endpoint
Of the three high surgical risk studies, early mortality (OR: 1.195 with CI (0.398–3.585) did not differ between the two procedures (. 3))29,30. SU-AVR group seems to be associated with a lower mortality (1-year mortality: SU-AVR: 4.2% vs. TAVR: 8.3%, 2-year mortality: SU-AVR: 8.4% vs. TAVR: 20.8%, HR: 0.400 (CI:0.130 − 0.1.233), Log-rank p = .0793)(Figs. 5 and 6D).
Secondary endpoints
Two out of three studies reported PPI rates (OR: 0.260, CI (0.051– 1.328), Fig. 4C)29,30,31. Other postoperative complications were only described by Bruno et al., including stroke (OR: 1.000, CI (0.060– 16.763), Fig. 4A), AKI (OR: 3.101, CI (0121– 79.205), Fig. 4B), ICU length of stay (sutureless/RDV: 3.1 days vs. TAVR: 2.1 days)29 and hospital length of stay (sutureless/RDV: 7.7–12 days vs. TAVR 8-9.4 days)29,30.
Hemodynamic performance at discharge
Postoperative hemodynamic performance was only reported by Bruno et al. with peak TPG (DoM: 3.6 with CI (0.184–7.016)) and mean TPG (DoM: 2.5 with CI (0.396–4.604)) and PVL (OR: 0.187 with CI (0.009–4.062), (Fig. 4D-F)29. No significant differences between PVL rates were observed, possibly due to a limited power in the high-risk group, while post-operative TPG were significantly lower for the patients that underwent TAVR.
Discussion
Sutureless and rapid deployment valves have been compared to TAVR, though never in a RCT, as all RCTs involved mainly standard sutured valves in the surgical arm32,33,34,35,36,37. In absence of such RCTs, the information that can be extracted from propensity score matched comparative studies is still valuable, as they show results in a real world population.
The sutureless or rapid deployment valves used in the studies included in this analysis were the Perceval S valve and the Intuity valve. Although both valves often show very similar outcomes in terms of mortality and morbidity, the reduction of procedure times is larger for the Perceval valve whereas the Intuity shows better hemodynamic performance38,39,40,41. Likewise, there are differences in the valves that were used in the TAVR groups. The two main valve groups investigated were the balloon-expandable (BE) Sapien and self-expandable (SE) CoreValve/Evolut series. The Sapien series consist of the Sapien, Sapien XT and the Sapien 3 while Corevalve evolved to Evolut R and Evolut Pro, both exhibiting improved outcomes with every new generation42. Overall, the SE CoreValve series have shown higher rates of PPI which can be explained by the intrinsic radial forces required for self-expanding valve fixation42,43,44.
Most of the low-risk patients in our meta-analysis come from one registry: the GARY registry from Germany. Pooled analysis in this risk group confirms that TAVR outperforms SU-AVR in terms of early mortality, AKI, TPG, and shorter length of stay at the cost of more PPI and PVL. These favourable outcomes for TAVR can possibly be explained by the exclusively transfemoral approach in the GARY experience. Generally, the transfemoral approach has become the preferred access route as it has shown better immediate outcomes45,46. Of note, Vilalta et al. reported a surprisingly high early mortality (4.1%) in the sutureless group, which is higher than in most studies in the intermediate/high-risk groups. Long-term outcome data are scarce in this low-risk population. Vilalta et al., reported hazard ratio of 0.970 (CI 0.518–1.815), showing no difference between the two treatments.
These results are in line with the findings in the large RCTs comparing TAVR with SAVR in low-risk patients, in which TAVR was found to be non-inferior or even superior to SAVR in terms of the primary outcome. Despite the all-comer nature in the registries included in our meta-analysis, patient risk profile and outcomes were comparable to those in the RCTs, with STS scores around 2% and similar stroke and early mortality rates34,37. In this respect, the use of sutureless or RDV does not seem to present a particular advantage to conventional surgical valves.
In contrast with low-risk patients, intermediate risk patients seem to fare exceptionally well with SU-AVR, with a significantly lower early mortality rate as compared with TAVR in this meta-analysis. This benefit was confirmed in the studies reporting longer term follow-up. While the difference in hemodynamic performance of the implanted valves was similar to low-risk patients (higher TPG but less PVL for sutureless or RDV), there are conflicting results regarding stroke, AKI and PPI (some studies show less, equal or more stroke, AKI or PPI after TAVR). Chung et al. reported a remarkably high incidence of AKI (21%) after SU-AVR, however, this could potentially be due to the differences in definitions used by the different studies. Regarding the need for PPI, the higher incidence of PPI with TAVR was mainly driven by those studies using a larger proportion of SE valves42.
The lower early and medium-term mortality with SU-AVR in intermediate risk patients is in contrast with the findings in the large RCTs comparing TAVR with SAVR in this risk category. However, the weighted average STS risk score in our meta-analysis were at least as high (5.7%) as compared with the RCTs with BE (5.8%) and SE (4.4%) TAVR valves. Interestingly, early mortality rates were higher for TAVR in the meta-analysis as compared to the TAVR arms of the RCTs (weighted average: 4.9% vs. BE: 3% and SE: 0.5%, respectively), while early mortality was lower in the surgical arms of the SU-AVR meta-analysis as compared to the RCTs (weighted average: 2% vs. BE: 4.1% and SE: 1.3%, respectively)33,36. These differences suggest improved performance of SU-AVR of sutured valves in this patient population, as well as a poorer TAVR outcomes in daily practice as compared to the RCTs. Another contributing factor may be some degree of bias being introduced in the treatment selection in the registry studies, despite the propensity matching that has been applied. Indeed, it is conceivable that patient characteristics not included in the propensity matching may have driven the choice for SAVR rather than TAVR, or vice versa, in this patient population.
According to treatment guidelines, high-risk patients with severe AS should preferably be treated with transfemoral TAVR, when technically feasible. In our meta-analysis, only limited data is available regarding the comparison between SU-AVR and TAVR in this group. The studies from Bruno and Santarpino et al. are very similar, both using the SE Corevalve with 100% transfemoral approach in TAVR patients. Within the few outcomes reported by both studies, there are noticeable differences in the in-hospital mortality and hospital LOS. Another important difference is in the need for PPI in the TAVR groups, which is almost three times as high for Bruno et al. compared to Santarpino et al. A possible explanation might be that the latter has a larger patient population, with a longer inclusion time, resulting in a better learning curve to optimize outcome47. While the presented data hardly permit one to make solid conclusions regarding the role of SU-AVR versus TAVR in this high-risk population, these studies confirm that SU-AVR may be a good alternative to conventional SAVR for these patients, especially when TAVR is contraindicated. Shorter XCT and procedure times are of particular benefit in an often frail and aged population with multiple comorbidities.
Limitations
The present systematic review and meta-analysis has several limitations that are inherent to the nature of the studies included. Reconstructed IPD rely on the level of information given, e.g., number at risk and the quality of the graphics. When lines are tangled, it can be difficult to separate out the KM-curves for digitizing. Moreover, the underlying algorithm presumes uniform censoring, an assumption that may not hold in all studies. The original authors have also indicated the need for subsequent research to investigate the variability of test statistics (even though their limited investigations showed promising results). In general, propensity score matching across studies tries to match patient characteristics. However, even when applying a correct statistical methodology, propensity matching cannot fully correct for the heart team decision. Not all discussed variables, such as patient frailty, mobility, mental health etc., are systematically reported and hence not included in the model. Overall, even after matching, the TAVR population was a slightly older and of higher risk than the SU-AVR group. This difference is not negligeable and could have an influence on the outcomes presented in this meta-analysis. Truly balanced (randomized) analyses should include patients who fully qualify for both treatment options before treatment selection. Moreover, older studies included differences in learning curve, mainly for TAVR, but also for SU-AVR. Another aspect that increases the difficulty to interpret the results is the many different valve types used in the included studies. Furthermore, valve designs are continuously changing (mainly of the TAVR valves) and sometimes implantation techniques were adapted (location of suture of the Perceval valve to avoid PPI and a change in sizing strategy to prevent oversizing). Moreover, the included studies contain many rather small sample sizes. Specifically in the low- and high-risk groups, the number of included studies is limited and have a noticeable disbalance in sample size within each group. Additionally, for the low- and high-risk groups, the length of follow-up is limited compared to the intermediate risk group. All are factors that warrant attention to the interpretation of results and comparison with the intermediate risk group. Ideally a large, randomized trial comparing SU-AVR with TAVR (ideally in low to intermediate surgical risk patients) should be performed in the future.
When combining all risk groups, our meta-analysis reveals no difference in early mortality between SU-AVR and TAVR, however SU-AVR was associated with an increased survival at 1- and 2-years. Stroke and AKI rates were not different between the two procedures. SU-AVR resulted in lower rates of PPI and significant paravalvular leakage, while TAVR patients had significantly better peak and mean post-operative TPG and a shorter hospital stay. On the basis of this analysis, we can also conclude that both approaches are associated with a different pattern of complications, something that may guide the choice for the individual patient, confirming again the importance of the Heart Team and individual tailored treatment strategies.
Conclusion
Based on our meta-analysis, which is based on a limited number of non-randomized studies, SU-AVR is not inferior to and possible superior to TAVR for certain outcomes, especially in the intermediate risk patients. Until stronger evidence is available SU-AVR should be discussed as a potential valid treatment in patients with severe AS that are discussed for TAVR. Randomized clinical trials, ideally with long-term follow-up are essential to exactly determine the ideal patients for both procedures.
Data availability
All data generated or analyzed during this study are available from the corresponding authors on reasonable request.
Abbreviations
- AKI:
-
Acute kidney injury
- AS:
-
Aortic valve stenosis
- AXC:
-
Aortic cross clamp
- BE:
-
Balloon-expandable
- CBP:
-
Cardiopulmonary bypass
- CVVH:
-
Continuous venovenous hemofiltration
- DoM:
-
Difference of the means
- ECC:
-
Extra corporeal circulation
- HR:
-
Hazard ratio
- ICU:
-
Intensive care unit
- IPD:
-
Individual person data
- KM:
-
Kaplan-Meier
- Mi-AVR:
-
Minimally invasive aortic valve replacement
- OR:
-
Odds ratio
- PARTNER trial:
-
Placement of aortic transcatheter valves trial
- PCI:
-
Percutaneous coronary intervention
- PPI:
-
Permanent pacemaker implantation
- PVL:
-
Paravalvular leakage
- RCT:
-
Randomized controlled trial
- RDV:
-
Rapid deployment valve
- ROBINS-I:
-
Risk of bias in non-randomized studies of interventions
- SAVR:
-
Surgical aortic valve replacement
- SE:
-
Self-expandable
- STS:
-
Society of thoracic surgeons
- STS PROM:
-
Society of thoracic surgeons predicted risk of mortality
- SU-AVR:
-
Sutureless aortic valve replacement
- TAVR:
-
Transcatheter aortic valve replacement
- TPG:
-
Transprosthetic gradient
References
Nkomo, V. T. et al. Burden of valvular heart diseases: a population-based study. Lancet 368, 1005–1011. https://doi.org/10.1016/S0140-6736(06)69208-8 (2006).
Otto, C. M. et al. ACC/AHA guideline for the management of patients with valvular heart disease: A report of the American college of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 143, e72-e227 https://doi.org/10.1161/CIR.0000000000000923 (2021).
Vahanian, A. et al. 2021 ESC/EACTS guidelines for the management of valvular heart disease. Eur. J. Cardiothorac. Surg. 60, 727–800. https://doi.org/10.1093/ejcts/ezab389 (2021).
Chambers, J. B. Aortic stenosis. Eur. J. Echocardiogr. 10, i11–19. https://doi.org/10.1093/ejechocard/jen240 (2009).
Horstkotte, D. & Loogen, F. The natural history of aortic valve stenosis. Eur Heart J 9 Suppl. E 57–64. https://doi.org/10.1093/eurheartj/9.suppl_e.57 (1988).
Nishimura, R. A. et al. AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 63, e57-185 https://doi.org/10.1016/j.jacc.2014.02.536 (2014).
Perceval S Sutureless Valve for Aortic Valve Replacement: A Review of the Clinical Effectiveness, Safety, and Cost-Effectiveness (Canadian Agency for Drugs and Technologies in Health Copyright © 2015 Canadian Agency for Drugs and Technologies in Health., (2015).
Doss, M., Buhr, E., Moritz, A. & Martens, S. Sutureless aortic valve replacement: catheter-based transapical versus direct transaortic implantation. J. Heart Valve Dis. 21, 758–763 (2012).
Kamperidis, V. et al. Surgical sutureless and transcatheter aortic valves: hemodynamic performance and clinical outcomes in propensity score-matched high-risk populations with severe aortic stenosis. JACC Cardiovasc. Interv. 8, 670–677. https://doi.org/10.1016/j.jcin.2014.10.029 (2015).
Muneretto, C. et al. Sutureless and rapid deployment vs. Transcatheter valves for aortic stenosis in Low-Risk patients: Mid-Term results. J. Clin. Med. 12, 4045 (2023).
Luo, D., Wan, X., Liu, J. & Tong, T. Optimally estimating the sample mean from the sample size, median, mid-range, and/or mid-quartile range. Stat. Methods Med. Res. 27, 1785–1805. https://doi.org/10.1177/0962280216669183 (2018).
Wan, X., Wang, W., Liu, J. & Tong, T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 14, 135. https://doi.org/10.1186/1471-2288-14-135 (2014).
DerSimonian, R. & Laird, N. Meta-analysis in clinical trials. Control Clin. Trials. 7, 177–188. https://doi.org/10.1016/0197-2456(86)90046-2 (1986).
Higgins, J. P. & Thompson, S. G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 21, 1539–1558. https://doi.org/10.1002/sim.1186 (2002).
Liu, N., Zhou, Y. & Lee, J. J. IPDfromKM: reconstruct individual patient data from published Kaplan-Meier survival curves. BMC Med. Res. Methodol. 21, 111. https://doi.org/10.1186/s12874-021-01308-8 (2021).
Leontyev, S. et al. Aortic valve replacement in octogenarians: utility of risk stratification with EuroSCORE. Ann. Thorac. Surg. 87, 1440–1445. https://doi.org/10.1016/j.athoracsur.2009.01.057 (2009). https://doi.org/https://doi.org/
Paparella, D. et al. Risk stratification for in-hospital mortality after cardiac surgery: external validation of EuroSCORE II in a prospective regional registry. Eur. J. Cardiothorac. Surg. 46, 840–848. https://doi.org/10.1093/ejcts/ezt657 (2014).
Balan, P. et al. The society of thoracic surgery risk score as a predictor of 30-Day mortality in transcatheter vs surgical aortic valve replacement: A Single-Center experience and its implications for the development of a TAVR risk-Prediction model. J. Invasive Cardiol. 29, 109–114 (2017).
Abdel-Wahab, M. et al. Transcatheter versus Rapid-Deployment aortic valve replacement: A Propensity-Matched analysis from the German aortic valve registry. JACC Cardiovasc. Interv. 13, 2642–2654. https://doi.org/10.1016/j.jcin.2020.09.018 (2020).
Vilalta, V. et al. Midterm outcomes following sutureless and transcatheter aortic valve replacement in Low-Risk patients with aortic stenosis. Circ. Cardiovasc. Interv. 14, e011120. https://doi.org/10.1161/circinterventions.121.011120 (2021).
Biancari, F. et al. Immediate outcome after sutureless versus transcatheter aortic valve replacement. Heart Vessels. 31, 427–433. https://doi.org/10.1007/s00380-014-0623-3 (2016).
Repossini, A. et al. Stentless sutureless and transcatheter valves: a comparison of the hemodynamic performance of different prostheses concept. Minerva Cardioangiol. 66, 180–190. https://doi.org/10.23736/s0026-4725.17.04564-9 (2018).
Al-Maisary, S. et al. Are sutureless and Rapid-Deployment aortic valves a serious alternative to TA-TAVI? A Matched-Pairs analysis. J. Clin. Med. 10 https://doi.org/10.3390/jcm10143072 (2021).
Miceli, A. et al. Minimally invasive aortic valve replacement with a sutureless valve through a right anterior mini-thoracotomy versus transcatheter aortic valve implantation in high-risk patients. Eur. J. Cardiothorac. Surg. 49, 960–965. https://doi.org/10.1093/ejcts/ezv210 (2016).
D’Onofrio, A. et al. The rise of new technologies for aortic valve stenosis: A comparison of sutureless and transcatheter aortic valve implantation. J. Thorac. Cardiovasc. Surg. 152, 99–109. https://doi.org/10.1016/j.jtcvs.2015.11.041 (2016). e102.
Chung, Y. H. et al. Transcatheter aortic valve replacement versus sutureless aortic valve replacement: A single center retrospective cohort study. Yonsei Med. J. 62, 885–894. https://doi.org/10.3349/ymj.2021.62.10.885 (2021).
Gerfer, S. et al. Comparison of Self-Expanding RDV perceval S versus TAVI ACURATE neo/tf. Thorac. Cardiovasc. Surg. 69, 420–427. https://doi.org/10.1055/s-0040-1722692 (2021).
Muneretto, C. et al. Sutureless versus transcatheter valves in patients with aortic stenosis at intermediate risk: A multi-institutional European study. Surgery 174, 1153–1160. https://doi.org/10.1016/j.surg.2023.08.001 (2023). https://doi.org/https://doi.
Bruno, P. et al. Rapid-deployment or transcatheter aortic valves in intermediate-risk patients? Asian Cardiovasc. Thorac. Ann. 25, 264–270. https://doi.org/10.1177/0218492317704773 (2017).
Santarpino, G. et al. Sutureless versus transcatheter aortic valve replacement: A multicenter analysis of real-world data. J. Cardiol. 79, 121–126. https://doi.org/10.1016/j.jjcc.2021.08.022 (2022). https://doi.org/https://doi.
Ferrara, J. et al. Rapid deployment versus trans-catheter aortic valve replacement in intermediate-risk patients: A propensity score analysis. J. Card Surg. 36, 2004–2012. https://doi.org/10.1111/jocs.15483 (2021).
Smith, C. R. et al. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl. J. Med. 364, 2187–2198. https://doi.org/10.1056/NEJMoa1103510 (2011).
Leon, M. B. et al. Transcatheter or surgical Aortic-Valve replacement in Intermediate-Risk patients. N Engl. J. Med. 374, 1609–1620. https://doi.org/10.1056/NEJMoa1514616 (2016).
Mack, M. J. et al. Transcatheter Aortic-Valve replacement with a Balloon-Expandable valve in Low-Risk patients. N Engl. J. Med. 380, 1695–1705. https://doi.org/10.1056/NEJMoa1814052 (2019).
Barker, C. M. & Reardon, M. J. The corevalve US pivotal trial. Semin Thorac. Cardiovasc. Surg. 26, 179–186. https://doi.org/10.1053/j.semtcvs.2014.10.001 (2014).
Reardon, M. J. et al. Surgical or transcatheter Aortic-Valve replacement in Intermediate-Risk patients. N Engl. J. Med. 376, 1321–1331. https://doi.org/10.1056/NEJMoa1700456 (2017).
Popma, J. J. et al. Transcatheter Aortic-Valve replacement with a Self-Expanding valve in Low-Risk patients. N Engl. J. Med. 380, 1706–1715. https://doi.org/10.1056/NEJMoa1816885 (2019).
Berretta, P. et al. Sutureless versus rapid deployment aortic valve replacement: results from a multicenter registry. Ann. Thorac. Surg. 114, 758–765. https://doi.org/10.1016/j.athoracsur.2021.08.037 (2022).
D’Onofrio, A. et al. Surgical aortic valve replacement with new-generation bioprostheses: Sutureless versus rapid-deployment. J. Thorac. Cardiovasc. Surg. 159, 432–442.e431 https://doi.org/10.1016/j.jtcvs.2019.02.135 (2020).
Flynn, C. D. et al. Sutureless valve and rapid deployment valves: a systematic review and meta-analysis of comparative studies. Ann. Cardiothorac. Surg. 9, 364–374. https://doi.org/10.21037/acs-2020-surd-27 (2020).
Wang, C. et al. Sutureless vs. rapid-deployment valve: a systemic review and meta-analysis for a direct comparison of intraoperative performance and clinical outcomes. Front. Cardiovasc. Med. 10, 1123487. https://doi.org/10.3389/fcvm.2023.1123487 (2023).
Eikelboom, R. et al. Current and future transcatheter aortic valve replacement valves. Curr. Opin. Cardiol. 37, 173–179. https://doi.org/10.1097/hco.0000000000000935 (2022).
van Nieuwkerk, A. C. et al. Balloon-Expandable versus Self-Expandable valves in transcatheter aortic valve implantation: complications and outcomes from a large international patient cohort. J. Clin. Med. 10 https://doi.org/10.3390/jcm10174005 (2021).
Deharo, P. et al. Impact of Sapien 3 Balloon-Expandable versus evolut R Self-Expandable transcatheter aortic valve implantation in patients with aortic stenosis. Circulation 141, 260–268. https://doi.org/10.1161/CIRCULATIONAHA.119.043971 (2020).
Kumar, N., Khera, R., Fonarow, G. C. & Bhatt, D. L. Comparison of outcomes of transfemoral versus transapical approach for transcatheter aortic valve implantation. Am. J. Cardiol. 122, 1520–1526. https://doi.org/10.1016/j.amjcard.2018.07.025 (2018).
Nicolas, J. & Mehran, R. Access routes in transcatheter aortic valve replacement: all roads lead to Rome but only one is paved. Catheter Cardiovasc. Interv. 97, 1470–1471. https://doi.org/10.1002/ccd.29776 (2021).
Wassef, A. W. A. et al. The learning curve and annual procedure volume standards for optimum outcomes of transcatheter aortic valve replacement: findings from an international registry. JACC Cardiovasc. Interv. 11, 1669–1679. https://doi.org/10.1016/j.jcin.2018.06.044 (2018).
Author information
Authors and Affiliations
Contributions
LM, PV, CD and BM conceived the experiments. LM and ML conducted the systematic review. LM, ML and LVL analyzed the results and wrote the main manuscript text. LVL conducted the meta-analysis. LM, ML, LVL, PV, JB, CD and BM reviewed and contributed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
BM & PV are consultants to Corcym srl. CD receives speaker fees from Corcym srl. CD is transcatheter heart valve (THV) proctor for Edwards Lifesciences. The Research Foundation Flanders (FWO) supported L. Minten with a personal research grant (1194521-23N). Corcym supported M. Lamberigts by a research grant.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Minten, L., Lamberigts, M., Van Langenhoven, L. et al. Sutureless versus transcatheter valves for the treatment of aortic valve stenosis: a systematic review and meta-analysis. Sci Rep 15, 34540 (2025). https://doi.org/10.1038/s41598-025-17857-7
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-17857-7